Abstract
Humans are exposed to environmental and economic threats that can profoundly affect individual survival and group functioning. Although anecdotal evidence suggests that threat exposure can increase collective action, the effects of threat on decision-making have been mainly investigated at the individual level. Here we examine how threat exposure and concomitant physiological responses modulate cooperation in small groups. Individuals (N = 105, ages 18–34 years) in groups of three were exposed to threat of electric shocks while deciding how much to contribute to a public good. Threat of shock induced a state of physiological freezing and, compared with no-threat conditions, reduced free riding and enabled groups to maintain higher cooperation over time. Exploratory analyses revealed that more cooperative responses under threat were driven by stronger baseline prosociality, suggesting that habitual prosociality is reinforced under threat. The current results support the view that human groups respond to outside threat with increased cooperation.
Climate change, contagious diseases, and terrorist attacks pose threats that can profoundly affect individual well-being and social welfare. Humans, similar to other animals, react to threatening situations with neurophysiological changes that influence decision-making (Fanselow & Lester, 1988; Roelofs, 2017). Whereas existing work on threat responding has mainly considered individual decision-making (Blanchard et al., 2011; Bradley et al., 2001; Fanselow & Lester, 1988; Roelofs, 2017), threats arising from climate change, contagious diseases, and terrorist attacks typically affect and are experienced by groups of individuals whose behavioral responses influence not only their own but also each other’s welfare (Gross et al., 2020; Van Lange & Rand, 2022). Unfortunately, however, we poorly understand how threat exposure and concomitant neurophysiological responses modulate group functioning. Indeed, although there is reason to assume that threat exposure can lead individuals to prioritize personal interests and self-preservation, leading to more selfishness (Engelmann et al., 2019), there is also evidence that threat exposure can increase group cohesion and facilitate cooperation (Bauer et al., 2016; Calo-Blanco et al., 2017; De Dreu et al., 2022).
To better understand whether and how anticipatory threat and concomitant neurophysiological responses modulate group cooperation, we experimentally examined public-good provision in which cooperation increases the welfare of all group members at a personal cost and not cooperating increases personal outcomes at a cost to the group. Individuals were under a threat of electric shocks and repeatedly made decisions, allowing us to investigate how threat influences cooperation across time. If threat undermines cooperation, we should see more free riding and smaller contributions to the public good under threat, compared with control conditions without threat of electric shocks. If, alternatively, threat promotes cooperation, we should observe larger contributions to the public good and fewer free-riding decisions under threat.
In conditions of anticipatory threat, humans and animals display a freezing response (Fanselow & Lester, 1988; Kozlowska et al., 2015; Roelofs, 2017). At the physiological level, freezing is associated with the coactivation of the parasympathetic and sympathetic nervous systems, with the dominant parasympathetic activation reflected in a transient heart rate deceleration and sympathetically controlled increases in skin conductance (Kozlowska et al., 2015; Roelofs, 2017). Freezing puts the individual in a state of hypervigilance and enhanced sensory processing. This enables the detection and processing of threat-relevant cues and other information relevant to optimal threat responding (Blanchard et al., 2011; Lang et al., 1997; Lojowska et al., 2018). Freezing in response to threat exposure enables individuals to increase their chances of survival. Such enhanced focus on self-preservation may lead the individual to prioritize personal interests over those of others, including the interests of the overarching group (Engelmann et al., 2019; FeldmanHall et al., 2015). If this is true, threat exposure should reduce the willingness to benefit others at a cost to oneself—threat exposure should reduce cooperation and increase attempts to free-ride on the cooperative efforts of other group members. On the other hand, individual survival is often tied to and promoted by group affiliation (Ioannou, 2017; Lehtonen & Jaatinen, 2016), and collective action is a common behavioral adaptation to threat in a range of animals, including fish (Hoare et al., 2004), birds (Beauchamp, 2004), and humans (Bauer et al., 2016; Tedeschi et al., 2021). Threat may increase a common-fate experience, leading to more bonding among the group members and increased within-group cooperation (Calo-Blanco et al., 2017; De Dreu et al., 2022; Insko et al., 2013). Indeed, by sticking together under threat and coordinating collective action, individuals can benefit from a safety-in-numbers principle (Ioannou, 2017; Lehtonen & Jaatinen, 2016). From this perspective, threat exposure should increase the individual’s concern for other group members’ well-being, and therefore, cooperative provision of public goods may increase and maintain. If true, this would point to a physiological mechanism that can explain why environmental stress sometimes enhances cooperation in real-life social dilemmas (Rabinovich et al., 2020; Wit & Wilke, 1998).
Method
Participants
To determine sample size for our study, we used the simr package in R (Green & MacLeod, 2016), which is designed to calculate power for linear mixed models, and pilot data on three groups. This revealed that a sample size of 30 three-person groups was needed to detect an effect size (b) of 0.99 (threat − safe) at 80% power. We therefore tested 35 groups in total (N = 105 individuals; 64% female; average age = 23.2 years, SD = 3.7). All groups were mixed gender.
Statement of Relevance
Nowadays, humans are exposed to threats from climate change or terrorist attacks. Many of these threats are experienced collectively, and individuals are often faced with a choice of whether to respond in a way that benefits everyone in the group or only themselves. The current study shows that individuals exposed to external threat (i.e., electric shocks) cooperate with others more compared with individuals in nonthreatening conditions. Such cooperative behavior under threat is observed particularly among more prosocial individuals and is associated with a specific autonomic physiological response pattern (i.e., reduced heart rate and increased skin conductance). These findings show that autonomic responses under threat, which are thought to promote individual survival, may do so through increased cooperation with others in the presence of collective threats, in line with the safety-in-numbers principle.
Participant-inclusion criteria were age between 18 and 35 years old; good understanding of written English; no history of cardiovascular, psychiatric, or neurological conditions; no pregnancy (or doubt about being pregnant); and no use of psychotropic drugs within the past 2 weeks. The study was approved by the ethics committee of the Faculty of Social Science of Leiden University (No. 2020-0-07-M.Lojowska-V3-1999). All participants provided written informed consent and received financial compensation or course credits for their participation (i.e., €3.50 or two course credits). In addition, participants earned extra money with the decisions they made during the experiment (range: €4.82–€19.89). To preserve anonymity, we computed payments individually and in private, and we paid participants immediately after the end of the task by mobile bank transfer.
Threat-induction and physiological measures
To investigate how threat influences group cooperation, we induced anticipatory threat states through a chance of receiving unpleasant but not painful electric shocks during our group-cooperation task (see below for task details). Electric shocks were delivered transcutaneously through the participant’s fourth and fifth distal phalanges of the dominant hand using Digitimer Constant Current Stimulator DS5 (www.digitimer.com) and standard Ag/AgCl electrodes. The duration of the electric stimulation was 200 ms, with a 50-Hz repetition of 250-µS pulses. The intensity of electric shocks varied between 1.2 to 10 mA. Shock intensity was adjusted at the individual level to ensure that the shocks were unpleasant but not painful. Shock calibration was performed using a standard shock-calibration procedure comprising five shock presentations, after which shock intensity was adjusted according to the participants’ verbal reports of its unpleasantness on a scale from 1 (not unpleasant at all) to 5 (very unpleasant). The final shock intensity obtained with this method was used in the group-cooperation task.
We introduced two control conditions to isolate the effect of threat on cooperation. In the safe condition, participants did not experience any shocks. In the mild-stimulation condition, we replaced electric shocks with mild sensory stimulations that were set to an intensity of 0.6 mA for all participants. We used this intensity as a control stimulation on the basis of a pilot study in which the majority of the participants rated it as a 1 on a scale from 1 (not unpleasant at all) to 5 (very unpleasant). In the actual study, the intensity of the mild stimulation was rated on average as 1.5 by the participants, suggesting that it was experienced as not unpleasant. The mild-stimulation condition allowed us to check whether the behavioral effects under threat are due to the actual threat manipulation or rather due to sensory stimulation. Because mild stimulation was rated as not unpleasant and was physiologically comparable with our safe condition (see the Results section), we expected no changes in behavior across the safe and mild-stimulation conditions.
To assess whether our threat manipulation was successful, we recorded heart rate and skin-conductance levels (SCLs). Heart rate was used for off-line assessment of the parasympathetically controlled heart rate deceleration (i.e., physiological index of freezing), whereas SCL was used as an index of sympathetic activity. Heart rate and SCL were acquired throughout the task using a BIOPAC MP 150 system (Biopac Systems, Goleta, CA). The sample rate was set to 1000 Hz. Heart rate was measured using disposable electrocardiogram electrodes attached to the participant’s chest. Skin-conductance data were collected with two standard Ag/AgCl electrodes attached to the second and third distal phalanges of the participant’s nondominant hand.
Experimental procedure
All participants received the information brochure with the study description and exclusion criteria at least 24 hr before the experiment. No deception was used in the study. Participants were informed that they would perform the experiment with other volunteers present in the lab and that their decisions had real monetary consequences for themselves and for the other participants. Participation was anonymous: Although participants were in the same room, they were seated in separate cubicles, were not able to see each other before or during the experiment, and were informed that their identity would not be revealed to other participants (to avoid reputation concerns). Furthermore, during the experiment, participants were asked to wear ear mufflers so that they were unable to hear each other (e.g., key presses, reactions to shock administration). To assure that participants believed that the other participants were actually present and subjected to the experimental treatments, we conducted the study in a laboratory designated for interactive, nondeception studies only, and the participants were clearly informed that no deception would be used. Only 7.6% of participants (8 out of 105) expressed their doubts in an exit questionnaire about whether the other participants in the study were real.
Heart rate, skin-conductance, and shock electrodes were subsequently attached. Next, participants were asked to complete the social-value-orientation (SVO) task (Murphy et al., 2011) to measure baseline prosociality, as well as questionnaires measuring risk preferences (i.e., staircase risk-elicitation task) and state and trait anxiety (for references, see the Supplemental Material available online). The outcome of these assessments was not analyzed in the current study (Fig. 1a).
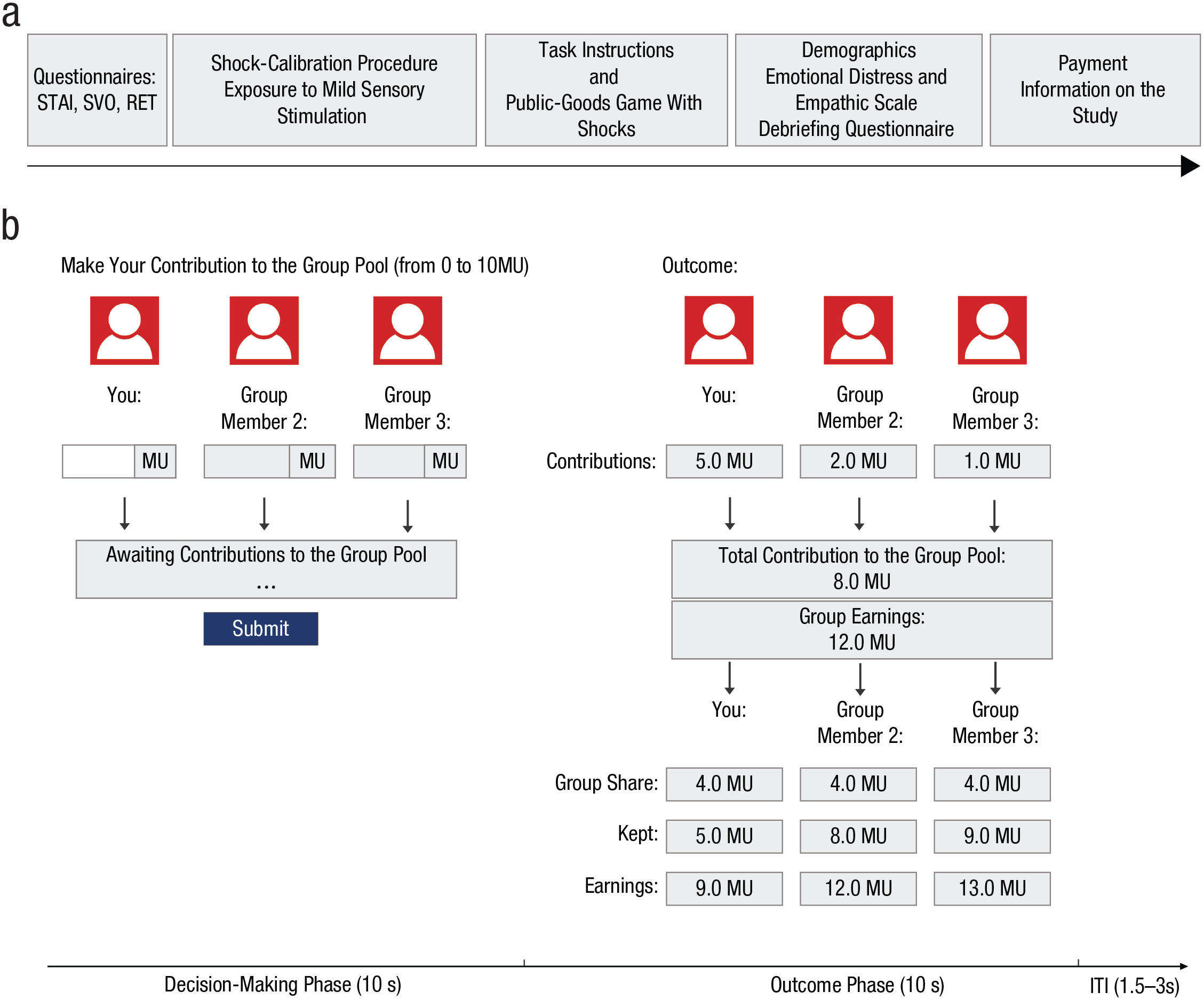
Experimental set-up. The experimental timeline and measurements are shown in (a), and the public-goods game as implemented in the threat condition (indicated to participants by the red icons) is shown in (b). The figure shows the timeline of one trial with exemplary decision input. Each trial consisted of a decision-making phase (10 s), outcome phase (10 s), and a variable intertrial interval (ITI; 1.5–3 s). In threat trials, participants could receive electric shocks at any time during the decision-making and outcome phases. STAI = State and Trait Inventory; SVO = social-value orientation; RET = risk-elicitation task; MU = monetary unit.
The SVO task confronts participants with six decisions on how to allocate money between themselves and another (unknown) person. For each decision, money can be allocated self-servingly or prosocially (sacrificing points to benefit the other person). For example, in one decision problem, the participant has to choose one out of nine possible allocations ranging from allocating 100 points to oneself and 50 points to the other person (maximal proself option) to allocating 50 points to oneself and 100 points to the other person (maximal prosocial option). On the basis of the decision pattern, the task allows to calculate an SVO angle, with larger values indicating stronger baseline prosociality (Murphy et al., 2011).
After completing these measurements, each participant underwent the individual shock-calibration procedure (as outlined above) and was exposed to two mild sensory stimulations. Participants then read instructions for our group-cooperation task (the public-goods game) and completed comprehension questions. When participants had answered all the comprehension questions correctly, the public-goods game under threat began (see below).
At the end of the experiment, participants answered demographic questions about their age, gender, and field of study, as well as a set of questions assessing their reputation concerns, beliefs in the study, and empathy (for reference, see the Supplemental Material). These questionnaires were not analyzed in the current study. Participants were then debriefed about the purpose of the experiment and received information about their earnings from the experiment. Total earnings included the participant’s fee plus earnings from the SVO task, the staircase risk-elicitation task, and one randomly chosen block from the public-goods game.
Cooperation under threat
To test our hypotheses about the effects of threat on cooperation, we used a linear public-goods game that groups performed under the threat condition and the two control conditions (safe and mild stimulation).
The rules and structure of the public-goods game were the same across all conditions (Fig. 1b). At the start of each trial, each participant received an endowment of 10 monetary units. One monetary unit was equivalent to €0.80. Each trial started with a 10-s decision phase during which participants had to decide how much of this endowment they would like to contribute to the group pool (i.e., the public good). Participants were informed that every monetary unit contributed to the group pool would be multiplied by a factor of 1.5 and divided equally among all three group members. The public-goods game confronts groups with a social dilemma. If individuals fully cooperate, by investing all of their resources to the public good, they create a total benefit of 15 monetary units for each group member. However, if only one group member cooperates fully, they receive a return of 5 monetary units (10 × 1.5/3) from their contribution, which is lower than keeping their monetary units for themselves. Although full contribution of all group members to the group pool (i.e., perfect and reciprocal cooperation) maximizes group earnings, individual group members earn even more by withholding their own monetary unit and benefiting from the contributions of others (free riding). Therefore, the rational-selfish choice in this game is to not contribute anything to the group pool, regardless of what others do.
In each trial, after participants simultaneously decided how many of their monetary units to contribute to the group pool, they were presented with a 10-s feedback screen showing the following information: each participant’s contribution to the group pool, each participant’s remaining endowment, the sum of contributions to the group pool, the sum of contributions to the group pool multiplied by 1.5, and individual earnings from the group pool. If participants did not make a decision within the decision window, their contribution was chosen randomly by the computer, but the participants themselves earned nothing in that specific trial. This was done to encourage participants to make active decisions on each trial. This procedure was known to participants beforehand and happened in only 0.77% of all trials, which were removed from the analyses. Feedback was followed by a 2- to 4-s intertrial interval.
As described above, groups were confronted with the public-goods game across our three different conditions: threat, mild stimulation, and safe. Each of these conditions were indicated by a different color: Trials in the threat condition were indicated by red icons, trials in the mild-stimulation condition by yellow icons, and trials in the safe condition by green icons (Fig. 1b). In the threat and mild-stimulation trials, participants could receive an electric shock or a mild sensory stimulation, respectively, at any point during the decision-making and feedback parts of the trial. Shock and mild sensory stimulation were both incidental (i.e., independent of behavioral responses), and all participants received stimulations simultaneously. During the safe condition, no sensory stimulation was administered. In the task instructions, participants were informed that the chance of receiving the shocks was the same for all participants in the group and that the shocks would be administered at the same time to everyone. Individuals within groups should thus not expect within-group differences in threat exposure.
We used a within-subjects design with each threat condition organized in blocks of 10 consecutive trials, repeated two times. These resulted in a total number of 6 blocks (with the order randomized across groups). Blocks were separated by breaks of 10 s and were randomized between participants. Ten percent of all trials were with shocks and 10% with mild stimulation, resulting in a total of six shocks and six mild stimulations administered during the task. Shocks and mild stimulations were administered randomly, and in a single trial participants could receive one to three shocks or mild stimulations.
Physiological and behavioral measures and analyses
The analysis of physiological data (heart rate and SCL) was performed off-line using in-house software implementation in MATLAB (The MathWorks, Natick, MA), allowing for visual assessment and removal of signal artifacts. Because delivery of the shocks could interfere with the process of decision-making, and we were primarily interested in anticipatory threat states rather than threat exposure itself, trials with shocks were excluded from the analysis (Table S8 in the Supplemental Material gives results including these trials). Heart rate data for seven participants (from seven different groups) were discarded because of extensive noise and artifacts (e.g., poor peak detection). To assess parasympathetic and sympathetic activity during the cooperation task, we calculated baseline-corrected heart rate responses and SCL for each trial. Heart rate responses were quantified by calculating the mean heart rate between 3 and 10 s following trial onsets corrected for the baseline represented by the mean of the heart rate in 10-s breaks preceding a given block of trials. Changes in SCL were quantified using the same time window. Behavioral data—that is, individual contributions (range 0–10)—were used as an index of cooperation. We also calculated an index of free riding, defined as the number of trials on which the individual contributed nothing. The analysis of free-riding decisions is complementary to the pattern of contributions by focusing specifically on maximally selfish decisions (i.e., keeping all resources for oneself).
Statistical analyses were carried out in R (Version 3.5.1; R Core Team, 2018). The analyses were performed using a linear mixed-effects model as implemented by the lmer function (lmer4 package, Version 3.3.1; Bates et al., 2015). Mixed models were used to account for repeated measures across individuals nested in groups. Each model included a group-level fixed effect coding our main predictor—threat condition (threat, mild stimulation, safe). To test for the presence of habituation effects in heart rates and SCLs over time, we also included fixed effects for the block number (1 or 2) and trial number (1–10). Fixed effects for the interactions between threat condition, block number, and trial number were also included. Within-subjects repeated measures were modeled by including a per-participant random adjustment to the fixed intercept (random intercept) as well as random adjustments to the slopes of predictors whenever no convergence errors were observed. All correlations among random effects were also included in the models. Continuous predictors were centered, and all categorical predictors were coded using treatment coding. Free-riding decisions were coded as a binary variable for which a logistic mixed-effects model, as implemented in the glmer function (lmer package, Version 3.3.1; Bates et al., 2015), was fitted. The models were fitted maximally with respect to the random effects to avoid inflated Type I errors (Barr et al., 2013). As a general strategy, we first used an omnibus model investigating the main effects of threat and its interactions with block and trial. To this end, we determined p values using Type 3 likelihood-ratio tests implemented in the mixed function of the package afex (Singmann et al., 2018). In linear mixed-effects models, point estimates (b) were used as a measure of the magnitude of the effects, and their corresponding p values were obtained using the lmerTest package (Kuznetsova et al., 2017). In logistic mixed-effects models, odds ratios and their 95% confidence intervals (CIs) were calculated as a measure of significance of the effects.
Results
Physiological responses
To investigate the effectiveness of our threat-manipulation procedure, we first examined physiological responses at the group level. This was done specifically to check whether a pattern of reduced heart rate and increased SCL responses previously observed for anticipatory threat states was also present in our data and hence would provide evidence that (a) our threat manipulation indeed led to anticipatory threat states and (b) that this was only the case when electric shocks were administered (i.e., in the threat condition only). We first tested the effect of our threat manipulation on parasympathetic activation as reflected in heart rate changes. Threat of shock resulted in a time-dependent reduction of heart rate in the first, but not the second, block of decision-making. Specifically, there was a significant interaction between threat condition, block, and trial, χ2(2, N = 98) = 4.94, p = .084, with a significant interaction between threat condition and trial in Block 1, χ2(2, N = 98) = 13.57, p = .001 (Fig. 2a), but not in Block 2, χ2(2, N = 98) = 0.84, p = .66. A planned linear-contrast analysis revealed that heart rate responses in Block 1 decreased differently for the conditions over trials, χ2(2, N = 98) = 13.80, p = .001. Specifically, stronger reduction in heart rate over trials was observed for threat compared with safe trials, β = −5.42, SE = 1.65, t(2640) = −3.26, p = .001, and compared with mild-stimulation trials, β = −5.26, SE = 1.66, t(2640) = −3.17, p = .0015. Heart rate responses in the mild and safe conditions did not differ over time, β = −0.16, SE = 1.66, t(2640) = −0.10, p = .920 (for full model results, see Table S1 in the Supplemental Material).
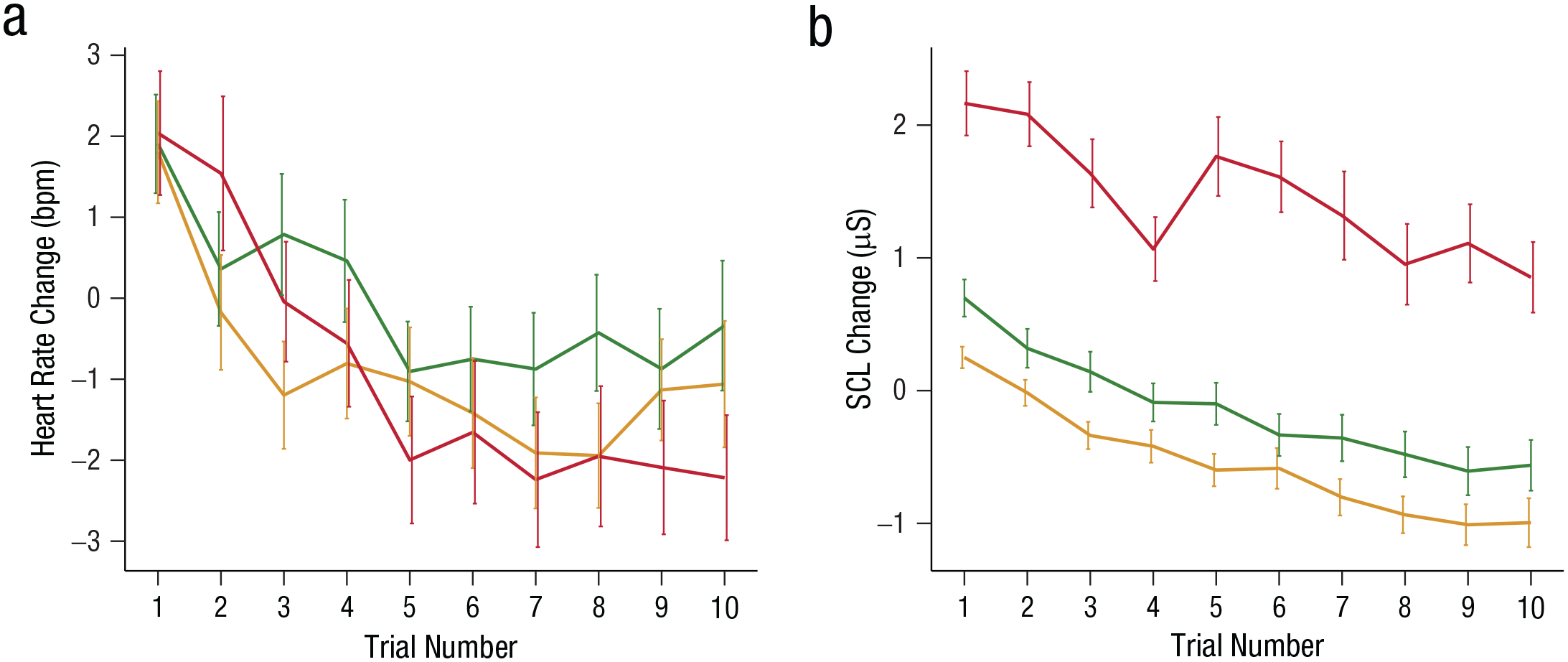
Physiological responses in the first decision-making block, represented by baseline-corrected responses in the threat condition (red), mild-stimulation condition (yellow), and safe condition (green). Threat-induced reduction in heart rate (a) reflects parasympathetic activation, and threat-induced increase in skin-conductance levels (SCLs; b) reflects sympathetic activation. Error bars represent standard errors of the mean. bpm = beats per minute.
Next, we examined sympathetically controlled changes in SCLs. We found that threat of shocks induced larger SCL responses compared with the mild-stimulation and safe conditions. Specifically, we found a main effect of threat condition on SCL responses, χ2(2, N = 105) = 64.27, p < .001; SCL responses were higher in the threat condition compared with the mild-stimulation condition, β = 1.59, SE = 0.24, t(104) = 6.71, p < .001, and the safe condition, β = 2.00, SE = 0.25, t(104) = 7.83, p < .001 (Fig. 2b). Larger SCL responses were also observed on mild-stimulation trials compared with safe trials, β = 0.41, SE = 0.19, t(104) = 2.13, p = .035. SCL responses decreased as a function of trial number, χ2(1, N = 105) = 391.09, p < .001, and this decrease did not differ between the threat, safe and mild stimulation conditions, as suggested by a nonsignificant interaction between threat condition and trial number, χ2(2, N = 105) = 3.72, p = .16. The remaining interactions were nonsignificant (p > .05; for full model results, see Table S1 in the Supplemental Material).
These results support the conclusion that threat of shock evoked both sympathetically and parasympathetically driven autonomic responses, with the latter observed primarily in the first decision-making block.
Contributions and free riding
Having shown that our threat manipulation led to physiological changes associated with threat experience, we subsequently tested whether contributions to the public good (i.e., cooperation) changed as a function of threat condition, block, and trial. We found that the decline of cooperation over trials that is commonly observed in public-good-provision experiments was present in the mild-stimulation and safe conditions and that this decline was significantly reduced under threat of shocks (Fig. 3a). As with parasympathetic and sympathetic activations, these effects of threat on behavior were observed in the first decision-making block, but not the second decision-making block. Specifically, there was a significant interaction between threat condition, block, and trial, χ2(2, N = 105) = 15.77, p < .001. For contributions in the first block, we observed a main effect of threat condition, χ2(2, N = 105) = 14.16, p < .001, and a significant interaction between threat condition and trial, χ2(2, N = 105) = 9.39, p = .009. Contributions across time were higher in the threat condition compared with both the mild- stimulation condition and the safe condition, β = 0.17, SE = 0.08, t(2791.76) = 2.00, p = .046, and β = 0.26, SE = 0.08, t(2791.76) = 3.01, p = .003, respectively. The difference between the mild-stimulation and safe trials was nonsignificant, β = 0.09, SE = 0.08, t(2789.92) = 1.01, p = .310. Additionally, we checked whether contributions on trial N tracked the individual’s own contributions and the other group members’ contributions on the previous trial (N – 1) and whether such tracking was modulated by the threat condition. The analysis revealed that contributions in trial N – 1 positively predicted contributions on trial N (p < .001), but this effect did not differ between the threat conditions (p > .05; see Table S7 in the Supplemental Material). Finally, the effect of threat on contributions in Block 2 was also nonsignificant (p > .05; see Tables S2 and S3 in the Supplemental Material).
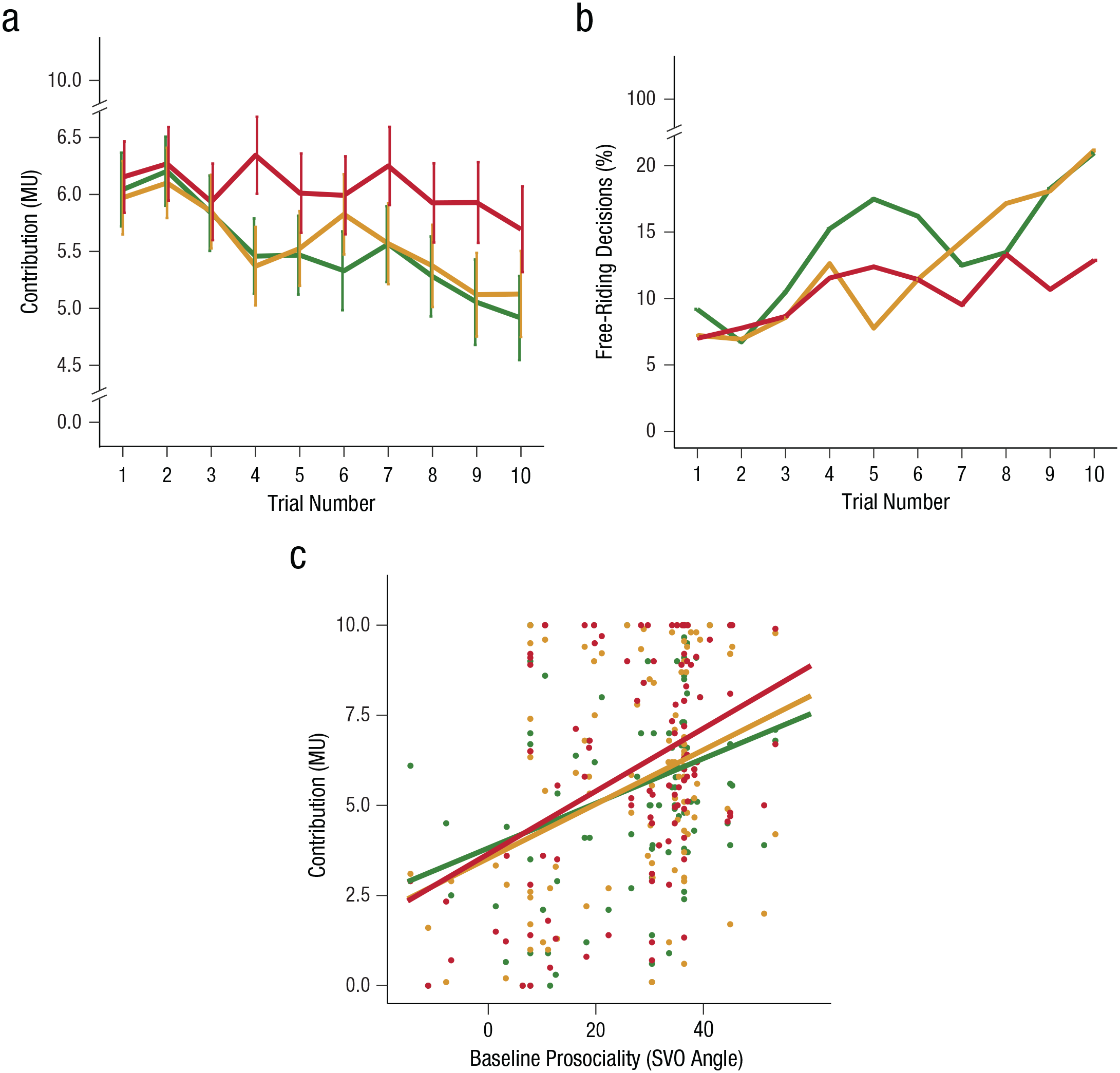
Individual contribution to the public good, with higher contributions indicating more cooperation. More cooperation (a) and fewer free-riding decisions (b) were observed when individuals anticipated unpleasant shocks (red) rather than mild stimulation (yellow) or no shocks (green) in the first decision-making block. Baseline prosociality (c) predicted cooperation, especially when group members anticipated painful rather than mild stimulation or no shocks. Error bars represent standard errors of the mean. MU = monetary unit; SVO = social-value orientation.
To complement the analysis of cooperation rates, we analyzed free riding (defined as the frequency of zero contributions). Both measures are interrelated but are not exactly the same. Analyzing free-riding decisions allowed us to see whether threat also influences the choice to be maximally selfish (i.e., keeping all resources for oneself) and allowed us to cross-check results with a similar, but not perfectly correlated, alternative dependent variable. For free riding, a similar pattern emerged: The likelihood that individuals would free-ride during the threat condition was smaller than during the mild-stimulation and safe conditions. Specifically, there was a significant interaction between threat condition and block, χ2(2, N = 105) = 6.77, p = .03, with a significant main effect of threat in Block 1, χ2(2, N = 105) = 8.32, p = .02, but not in Block 2, χ2(2, N = 105) = 1.65, p = .44. In Block 1, the odds of deciding to free-ride were significantly larger in the safe condition, OR = 0.56, 95% CI = [0.36, 0.82], χ2(1, N = 105) = 9.43, p = .002, and the mild-stimulation condition, OR = 0.65, 95% CI = [0.42, 1.09], χ2(1) = 5.70, p = .02 (across time), compared with the threat condition (Fig. 3b). The difference between the mild-stimulation and safe conditions was nonsignificant, OR = 0.78, 95% CI = [0.55, 1.09], χ2(1, N = 105) = 2.23, p = .13. As with cooperation rates, the effects of threat on free-riding decisions in Block 2 was nonsignificant (p > .05; see Tables S2 and S3).
To further understand these general effects of threat on cooperation, we performed an exploratory analysis in which we examined how individual variation in baseline prosociality, proxied with the SVO angle, modulated the contributions in the threat conditions. Individuals with prosocial tendencies are commonly found to cooperate more than proself individuals (Van Lange, 1999), especially when the group is exposed to environmental stress or threat from enemy groups (Abbink et al., 2012; Campos-Mercade et al., 2021; De Dreu et al., 2021). The expression of more prosocial behavior, especially among highly prosocial individuals under threat, would also resonate with the observation that stress generally increases habitual responses (Hartogsveld et al., 2020; Schwabe & Wolf, 2009). In line with these findings, we found that stronger baseline prosociality was associated with higher contributions overall, β = 0.26, SE = 0.17, χ2(1, N = 105) = 14.65, p < .001 (Fig. 3c). More interestingly, we found that baseline prosociality marginally modulated the interaction between threat condition and contributions, χ2(2, N = 105) = 5.86, p = .053, with stronger prosociality predicting higher contributions in the threat condition compared with the safe condition, β = 0.36, SE = 0.15, t(103.20) = 2.37, p = .020. Contributions on the threat versus the mild-stimulation condition, β = 0.17, SE = 0.17, t(103.13) = 1.00, p = .32, and the mild-stimulation versus the safe condition, β = 0.18, SE = 0.21, t(103.06) = 0.88, p = .38, did not differ significantly as a function of baseline prosociality. A similar pattern emerged for free-riding decisions as a function of baseline prosociality (see Table S4 in the Supplemental Material).
Physiological threat responding and contributions
We concluded our exploratory analyses by examining whether and how threat-related changes in parasympathetic responding, reflected in heart rate and skin-conductance changes, predicted individual contributions in the public-goods game. We were specifically interested in whether a stronger reduction in heart rate—a parasympathetically controlled response specific for freezing—was linked to more contributions in the threat condition. Focusing on Block 1 decision-making, we found that heart rate and SCL responses did not predict contributions decisions (p > .05; see Table S5 in the Supplemental Material).
Given the above association between SVO and contributions, we subsequently explored whether the association between physiological responses and contributions was modulated by baseline levels of prosociality. Indeed, we found that threat-induced reduction in heart rate was marginally associated with smaller contributions, but only among less prosocial individuals, β = −0.21, SE = 0.10, χ2(1, N = 98) = 3.83, p = .050 (Fig. 4). This relationship remained after controlling for sympathetic activation (i.e., SCL), β = −0.21, SE = 0.10, χ2(1, N = 98) = 3.85, p = .0497. No significant relationship was observed between SCL responses and contributions, β = 0.06, SE = 0.12, χ2(2, N = 105) = 5.86, p = .053, suggesting that among less prosocial individuals, cooperation under threat is modulated specifically by parasympathetic responses to threat. No associations between free-riding decisions, physiological responses, and baseline prosociality (SVO) were found (p > .05; see Table S6 in the Supplemental Material). These results tentatively suggest that baseline prosocial tendencies modulate the extent to which the underlying and dominant parasympathetic responses, typical for the anticipatory state of threat, influence cooperative decisions. However, these findings are based on exploratory analyses for which some effects were only marginally significant and, hence, should be interpreted with caution.

Baseline prosociality modulated the relationship between threat-induced changes in heart rate and contributions. A stronger reduction in heart rate was associated with lowered contributions in the threat compared to the safe condition, especially among less prosocial individuals. SVO = social value orientation; MU = monetary unit.
Discussion
Individuals in our experiment were initially cooperative, yet over trials, the provision of the public good dropped substantially. This breakdown of cooperation has often been observed and explained in terms of participants’ fear that other group members may free-ride on their cooperation (see Fehr & Gachter, 2002; for a review, see van Dijk & De Dreu, 2021). Here we find that anticipatory threat of physically unpleasant electric shocks not only reduces free riding during public-good provision but also significantly slows down the decline in cooperation. Rather than producing a simple increase or decrease in cooperation overall, anticipatory threat helps participants maintain cooperation longer. Our exploratory analysis also showed that such positive effects of anticipatory threat may be particularly prominent among more prosocial individuals who care for the well-being of others and may be modulated by parasympathetic responses at the physiological level. Future studies are needed to provide further support for these exploratory findings. That being said, the observed increase in cooperation stability on the group level under threat aligns with findings of studies on nonhuman animals, in which the primary response to threat was found to be collective action (Ioannou, 2017). Further, the interaction with baseline prosociality levels resonates with the idea that habitual responses may become more prevalent in stressful situations (Hartogsveld et al., 2020; Schwabe & Wolf, 2009).
The finding that threat exposure increases cooperation is consistent with the results of studies showing that the threat of losing against a competing out-group can enhance within-group cooperation (e.g., Abbink et al., 2012; Bornstein & Ben-Yossef, 1994). Current findings also align with work showing that environmental and financial stress can lead groups to cooperate more (e.g., De Dreu et al., 2022; Milinski et al., 2008; Zhang, 2019). In both lines of work, cooperation was instrumental in mitigating or annihilating the threat to personal resources. In the present experiment, threat was independent of whether and how much group members cooperated. This provides for a clean and unconfounded assessment of how threat in itself induces behavioral and cognitive adaptations with the ultimate goal of assuring individual survival and self-preservation, which could also be reflected in prioritizing personal interests and less cooperation under threat.
Accordingly, our experiment contributes to these earlier lines of work in two ways. First, it provides evidence for collective action during a physiologically and functionally distinct state of anticipatory threat. Second, it shows that enhanced cooperation following threat emerges even when cooperation is not instrumental to threat avoidance. Put differently, incidental threats lead people to prioritize collective rather than selfish interests.
Our findings also contribute to the extant literature on the linkages between anticipatory threat, underlying physiological responding, and behavioral decisions (Bradley et al., 2001; Fanselow & Lester, 1988; Lang et al., 1997). This literature considers individuals operating in isolation, and to our knowledge, our experiment is the first to link threat to physiological responses and decision-making in individuals interacting in small groups. Anticipatory threat, and specifically reduction in heart rate—a parasympathetic index of freezing—has commonly been associated with sensory intake and enhanced processing of environmental cues, allowing for optimal threat avoidance and self-preservation (Blanchard et al., 2011; Fanselow & Lester, 1988; Lang et al., 1997; Roelofs, 2017). Here, too, however, we found that a stronger reduction in heart rate under threat predicted less cooperation among less prosocial individuals. The current result suggests that individual differences in baseline prosociality may play an important role in modulating (a) the interactions between the underlying affective (autonomic) processes and social decision-making and (b) the extent to which autonomic defensive responses are associated with more self-regarding versus other-regarding behavior. It should be noted, however, that the effects supporting these conclusions were small and not all of them were significant at conventional levels. Future studies are needed to confirm the robustness of these findings and to further understand the relationship between autonomic responses, individual personality traits, and cooperation under threat.
In contrast to previous studies in which decisions were made after the anticipatory threat periods, the anticipatory state in the current study was accompanied by an active process of decision-making (i.e., contributions to the public good). This process may have been also arousing, especially initially, as supported by the time-dependent reduction in skin conductance during decision-making in the safe condition. Perhaps such initially dominant arousing process of decision-making may explain why parasympathetically-controlled heart-rate responses were observed later and as a function of time in our task. In addition, threat-related increase in sympathetically controlled skin conductance was observed in both blocks, whereas threat-induced changes in heart rate were observed only in the first decision-making block. This may suggest that both sympathetic and parasympathetic responses to threat are required for groups to maintain the provision of public goods and resist the temptation to free-ride on others’ cooperation. An alternative explanation could be that responses in Block 2 reflected more complex interactions between what has been learned about the other group members in the previous blocks and threat responses. For example, nonsuccessful cooperation in the preceding block could override facilitating effects of threat in the subsequent block, which could possibly explain the observed lack of threat effects on cooperation in Block 2. We did not find significant effects of block order on cooperation. Yet we had only a limited number of observations per cell (order of presented blocks), and hence we do not have enough sensitivity to systematically investigate this possibility with our data. Future studies could investigate how previous experience within groups may modulate subsequent effects of threat on cooperation.
The current study shows that cooperation emerges in a group of individuals with equal chances of exposure to threat. Previous studies, however, have shown that inequality in financial resources leads to lower contributions to the public good, especially by the rich (Hargreaves Heap et al., 2016). Furthermore, when joint contributions are required to avoid a financial risk, inequality leads to more self-reliance among the richest, making the poorer more susceptible to losing their resources (Gross et al., 2020). On the other hand, unequal exposure to threat (of electric shocks) has been found to evoke autonomic defensive responses in individuals exposed to the harm to others and has predicted subsequent helping behavior (Hein et al., 2011). This suggests that inequality in exposure to threat may in fact foster cooperation. An interesting line for future research, therefore, would be to specifically investigate how cooperation emerges during unequal exposure to threat and the role of vicarious autonomic responses therein.
Humans are continuously exposed to a range of threats, from economic and climate shocks to the presence of predators and hostile conspecifics. Here, we identified how anticipatory threat can change, at both the physiological and behavioral level, the microfoundations of cooperation and collective action. Physiological freezing enables humans to produce both self- and other-regarding behavior. In particular, more prosocial individuals may be more likely to maintain cooperation under threat and, as a result, may enable groups to maintain public goods from which the group and its individual members benefit.
Supplemental Material
sj-docx-1-pss-10.1177_09567976221104037 – Supplemental material for Anticipatory Threat Mitigates the Breakdown of Group Cooperation
Supplemental material, sj-docx-1-pss-10.1177_09567976221104037 for Anticipatory Threat Mitigates the Breakdown of Group Cooperation by Maria Lojowska, Jörg Gross and Carsten K. W. De Dreu in Psychological Science
Footnotes
Acknowledgements
The authors would like to thank Luuk Snijder and Brandon Keytel for their help with data collection.
Transparency
Action Editor: Eddie Harmon-Jones
Editor: Patricia J. Bauer
Author Contributions
M. Lojowska, J. Gross, and C. K. W. De Dreu designed the experiment; M. Lojowska collected the data; and M. Lojowska, J. Gross, and C. K. W. De Dreu performed the analysis. All the authors wrote the manuscript and approved the final version for submission.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
