Abstract
Background
To assess whether human papillomavirus (HPV) vaccination is associated with herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) infection among young women in the United States.
Methods
We analyzed data from the 2011–2016 National Health and Nutrition Examination Survey, including sexually active women aged 18–35 years with HSV-1 and HSV-2 serology and self-reported HPV vaccination status. Weighted prevalence estimates and logistic regression models were used to evaluate associations, adjusting for demographic and socioeconomic covariates. Propensity score–weighted analyses were conducted as sensitivity checks.
Results
Among 1643 women, 30.1% reported HPV vaccination. Vaccinated women had lower unadjusted HSV-1 (41.9% vs 47.8%, p = 0.034) and HSV-2 prevalence (9.9% vs 15.2%, p = 0.007). In adjusted models, HPV vaccination was not significantly associated with HSV-1 (aOR = 1.26, 95% CI: 0.94–1.70) or HSV-2 infection (aOR = 0.86, 95% CI: 0.56–1.31). Sensitivity analyses confirmed these findings.
Conclusions
HPV vaccination was not linked to increased risk of HSV-1 or HSV-2 infection, providing HSV-specific evidence against sexual risk compensation and supporting public health messaging promoting HPV immunization.
Introduction
Human papillomavirus (HPV) is the most prevalent sexually transmitted infection (STI) worldwide and a major cause of cervical and other anogenital cancers. HPV vaccination has proven highly effective in preventing infection with oncogenic types and has become a cornerstone of STI prevention strategies.1,2
Despite these well-documented benefits, concerns about “risk compensation”—the belief that vaccination may encourage earlier or riskier sexual activity—remain a persistent barrier to broader coverage. Previous studies, however, have consistently found no evidence that HPV vaccination increases sexual activity or the incidence of other STIs such as Chlamydia trachomatis or Neisseria gonorrhoeae.3–7
Herpes simplex virus types 1 and 2 (HSV-1 and HSV-2) remain highly prevalent in the United States and are closely linked to sexual behavior, especially HSV-2.4,8 Although the behavioral implications of HPV vaccination have been widely examined,9–11 few studies have directly explored its relationship with HSV infection. Understanding whether HPV vaccination is associated with HSV-1/2 provides a disease-specific assessment of the sexual risk compensation hypothesis.
Therefore, using nationally representative data from the National Health and Nutrition Examination Survey (NHANES) 2011–2016, we evaluated the association between HPV vaccination and HSV-1/2 seropositivity among sexually active U.S. women. These findings may help clarify behavioral and epidemiologic patterns related to HPV immunization and guide public health communication.
Methods
Study design and population
We used data from three cycles of the U.S. National Health and Nutrition Examination Survey (NHANES 2011–2012, 2013–2014, and 2015–2016), a nationally representative cross-sectional survey conducted by the National Center for Health Statistics (NCHS), Centers for Disease Control and Prevention (CDC). NHANES employs a complex, multistage probability design to assess the health of the non-institutionalized civilian U.S. population. Detailed survey procedures, ethics approvals, and informed consent processes are described in NCHS documentation. 12
Our analysis was restricted to sexually active women aged 18–35 years, defined as reporting any lifetime vaginal, anal, or oral sex. 13 Across the three cycles, 29,902 individuals were surveyed, of whom 2845 were women aged 18–35. After applying exclusions for sexual inactivity (n = 692), missing HPV vaccination data (n = 120), missing HSV serology (n = 127), and incomplete covariates (n = 263), the final analytic sample included 1643 participants.
Exposure: HPV vaccination
The primary exposure was HPV vaccination status, defined as receipt of ≥1 dose of HPV vaccine. This was based on self-report to the question: “Have you ever received one or more doses of the HPV vaccine (such as Cervarix, Gardasil, or Gardasil 9)?” (Yes/No). Although participants reported number of doses and age at first dose, we analyzed vaccination as a binary measure to maximize sample size. Self-reported HPV vaccination has demonstrated high validity compared with medical records (≈90% sensitivity, 80% specificity). 14 Provider-verified vaccination dates were unavailable; thus, temporal ordering of vaccination and HSV infection could not be confirmed.
Outcomes: HSV-1 and HSV-2 Infection
Serological status for HSV-1 and HSV-2 was determined from blood samples collected at NHANES mobile examination centers and tested using a solid-phase enzymatic immunodot assay with virus type–specific glycoproteins. This in-house assay, developed and performed by Emory University for NHANES, has been reported to possess high sensitivity and specificity, although exact performance metrics are not provided in the NHANES documentation. Results were categorized as positive, negative, or indeterminate according to standardized NHANES laboratory criteria.
Covariates
Potential confounders were selected a priori based on the disjunctive cause criterion and a directed acyclic graph (DAG). 15 Candidate variables included age (continuous), race/ethnicity, education, poverty-income ratio (PIR, continuous), marital status, health insurance, smoking history, alcohol consumption, body mass index (BMI, continuous), and NHANES cycle. To further explore the role of sexual behavior factors, we included the mediating variables (age at first sexual debut, condom use, and number of partners) together with the candidate variables in the full model. Backward elimination guided by Akaike information criterion (AIC) was applied to derive the most parsimonious models. 16
Statistical analysis
Survey weights, strata, and primary sampling units were incorporated to account for the complex NHANES design. Group differences were assessed using survey-weighted χ2 tests for categorical variables and weighted linear regression for continuous variables. To evaluate potential selection bias due to missing data, we also compared sociodemographic characteristics between included participants and sexually active women excluded because of incomplete vaccination or HSV serology information. Standardized mean differences (SMDs) > 0.2 were considered meaningful. We estimated weighted prevalence of HSV-1 and HSV-2 by vaccination status and compared groups using Rao–Scott χ2 tests. Logistic regression was used to estimate odds ratios (ORs) for HSV infection. Three models were fitted:
Unadjusted (crude) ORs
Multivariable-adjusted ORs including covariates retained after backward elimination Propensity score–weighted ORs as a sensitivity analysis.
Propensity scores were estimated using gradient boosting machines based on all identified confounders, with stabilized inverse probability of treatment weights (IPTW) applied. Covariate balance after weighting was confirmed by SMD <0.2 across variables.
All analyses were performed using R version 4.4.2 (survey package). Statistical tests were two-sided, with p < 0.05 considered significant.
Ethics
NHANES protocols were approved by the NCHS Ethics Review Board, and all participants provided written informed consent.
Results
Participant characteristics
Survey-weighted bivariate comparisons among sexually active females aged 18–35 participating in NHANES 2011–2016, by HPV vaccination status (n = 1643).
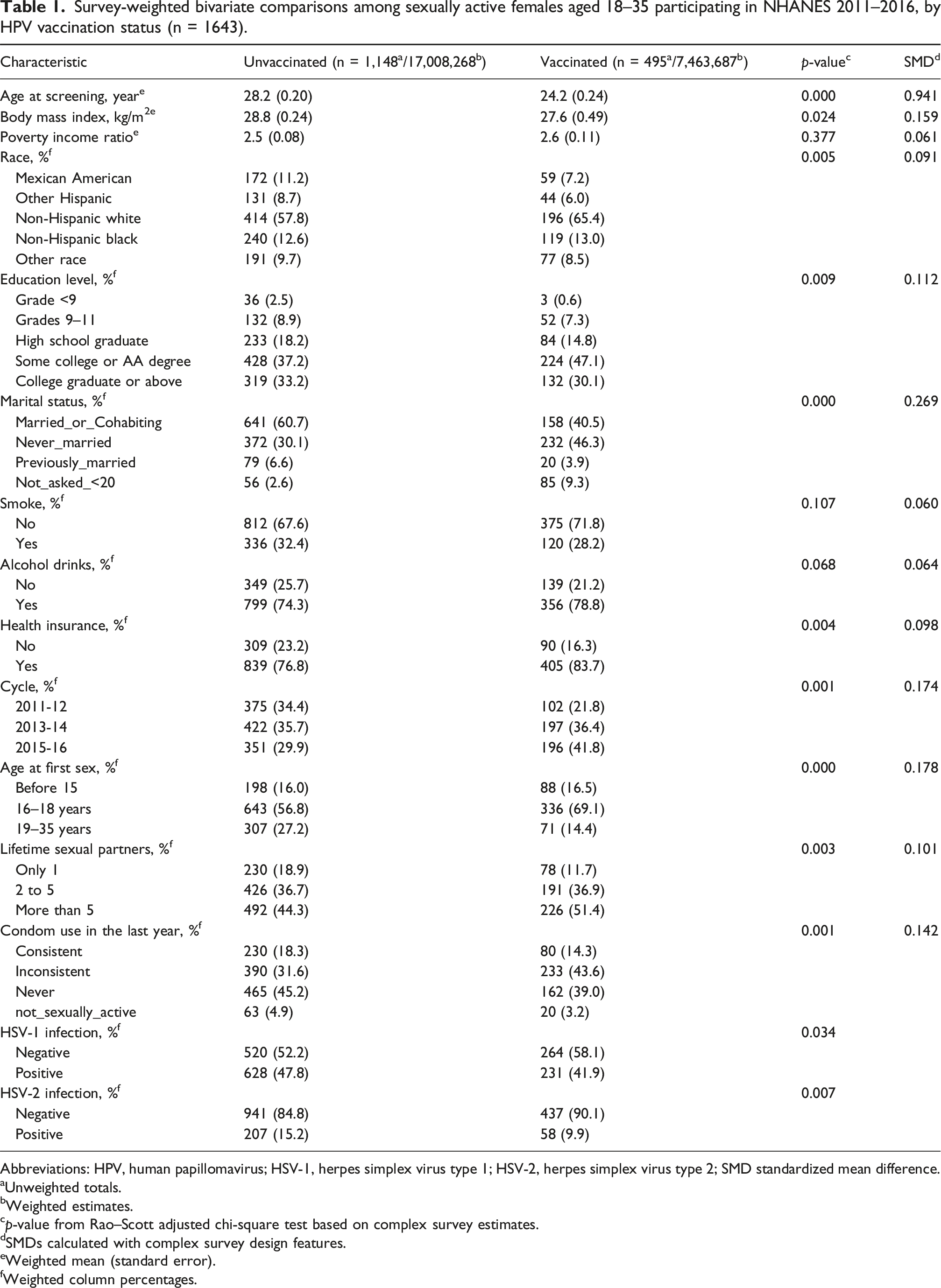
Abbreviations: HPV, human papillomavirus; HSV-1, herpes simplex virus type 1; HSV-2, herpes simplex virus type 2; SMD standardized mean difference.
aUnweighted totals.
bWeighted estimates.
cp-value from Rao–Scott adjusted chi-square test based on complex survey estimates.
dSMDs calculated with complex survey design features.
eWeighted mean (standard error).
fWeighted column percentages.
Association between HPV vaccination and HSV infection
Survey-weighted crude and adjusted associations of HPV vaccination with HSV-1 infection among females aged 18–35 with a history of sexual activity in NHANES 2011–2016 (n = 1643).
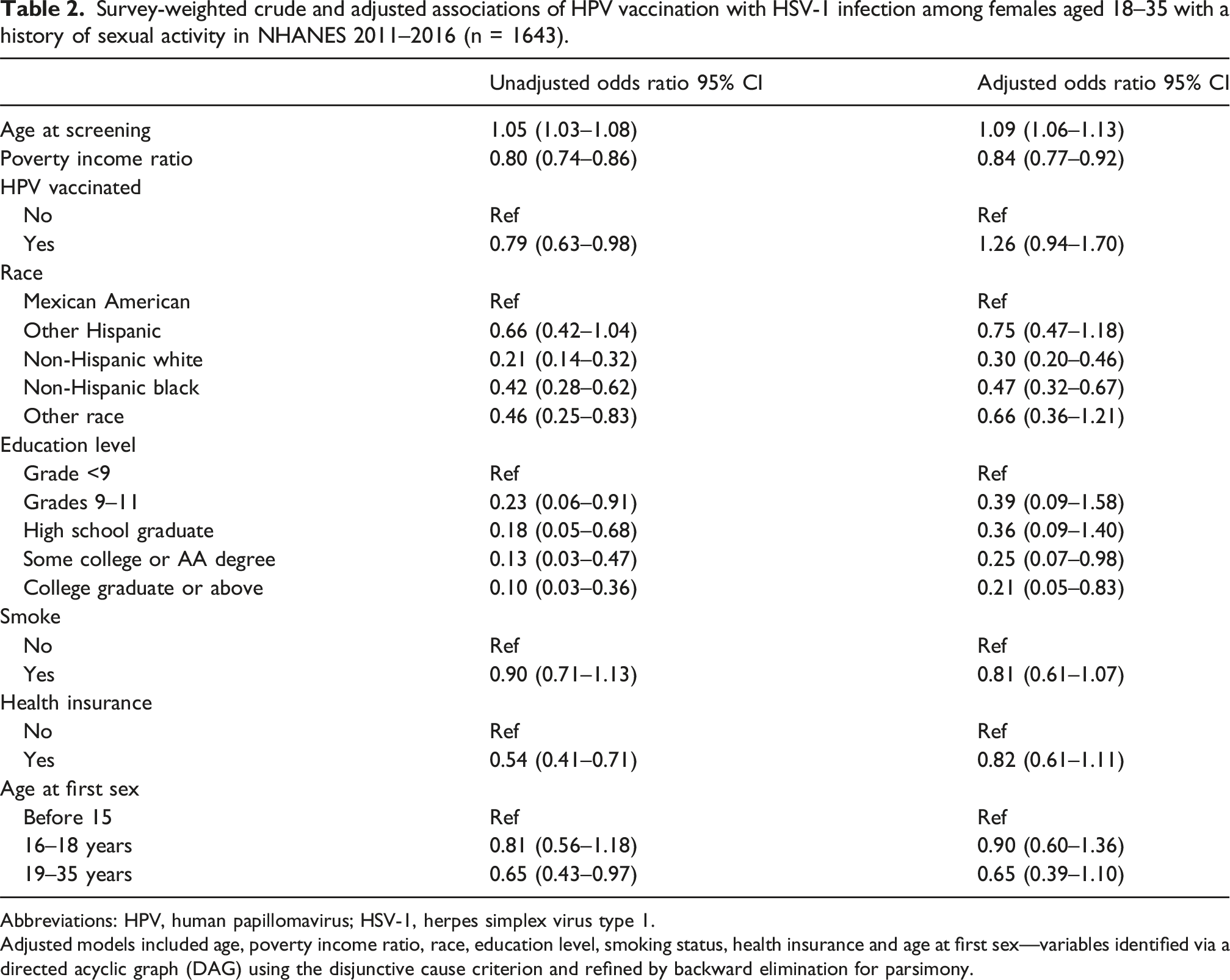
Abbreviations: HPV, human papillomavirus; HSV-1, herpes simplex virus type 1.
Adjusted models included age, poverty income ratio, race, education level, smoking status, health insurance and age at first sex—variables identified via a directed acyclic graph (DAG) using the disjunctive cause criterion and refined by backward elimination for parsimony.
Survey-weighted crude and adjusted associations of HPV vaccination with HSV-2 infection among females aged 18–35 with a history of sexual activity in NHANES 2011–2016 (n = 1643).
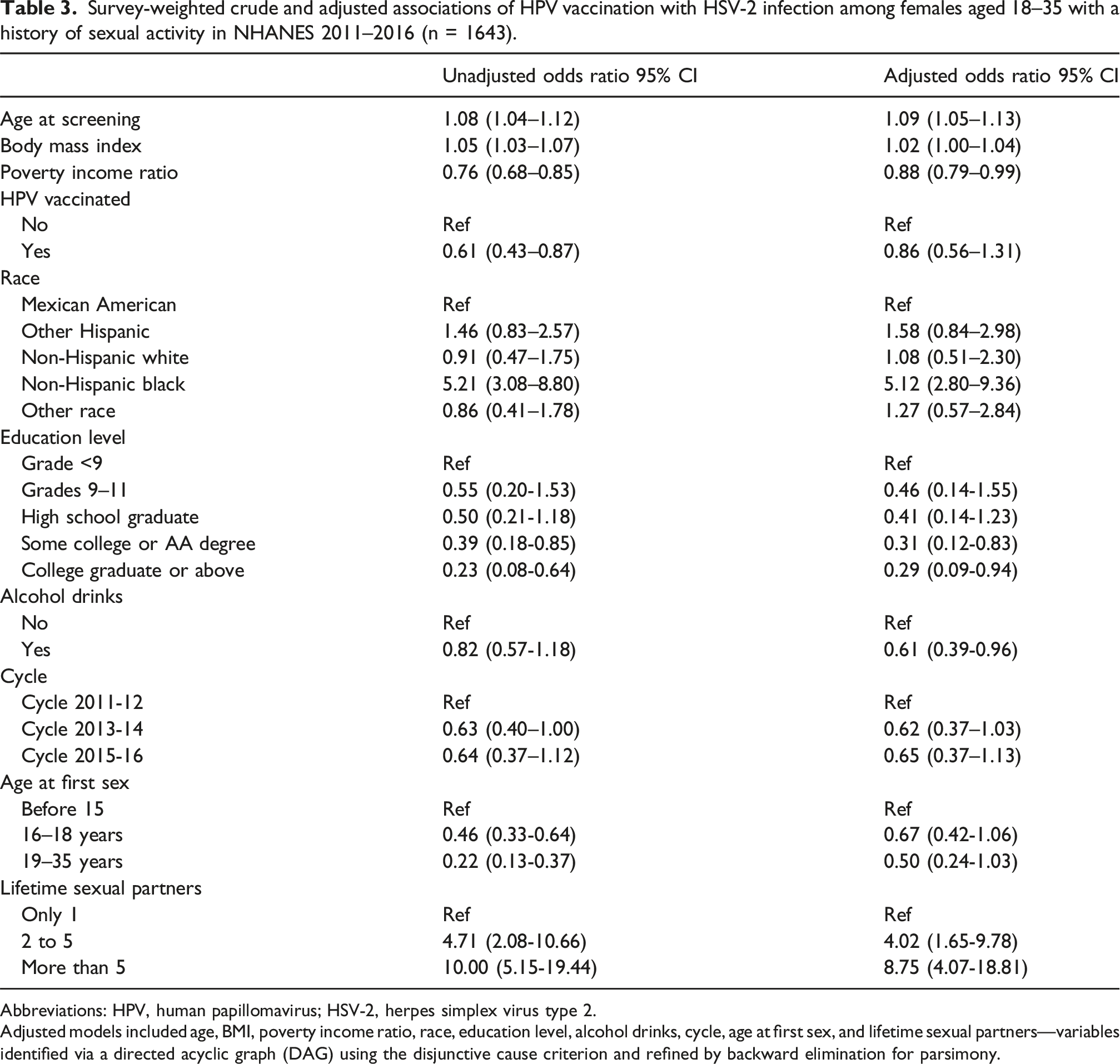
Abbreviations: HPV, human papillomavirus; HSV-2, herpes simplex virus type 2.
Adjusted models included age, BMI, poverty income ratio, race, education level, alcohol drinks, cycle, age at first sex, and lifetime sexual partners—variables identified via a directed acyclic graph (DAG) using the disjunctive cause criterion and refined by backward elimination for parsimony.
For HSV-1, independent predictors included older age (aOR 1.09, 95 %CI 1.06–1.13), lower poverty-income ratio (aOR 0.84, 95% CI 0.77–0.92), and Hispanic ethnicity compared with Mexican American reference groups.
For HSV-2, significant predictors included older age (aOR 1.09, 95% CI 1.05–1.13), higher BMI (aOR 1.02, 95% CI 1.00–1.04), non-Hispanic Black race/ethnicity (aOR 5.12, 95% CI 2.80–9.36), lower poverty-income ratio (aOR 0.88, 95% CI 0.79–0.99), and alcohol use (aOR 0.61, 95% CI 0.39–0.96). Later sexual initiation and fewer lifetime sexual partners were associated with reduced HSV-2 seropositivity.
Sensitivity analyses
Propensity score–weighted models achieved good covariate balance (all SMD <0.2). Effect estimates were consistent with the primary analyses, showing no significant association between HPV vaccination and HSV-1 or HSV-2 infection.
Discussion
In this nationally representative cross-sectional study of young U.S. women, we found no evidence that HPV vaccination was associated with HSV infection. After adjustment for demographic and socioeconomic, and behavioral factors, no independent relationship between HPV vaccination and HSV seropositivity was observed. These findings argue against the hypothesis that HPV vaccination promotes sexual risk compensation and subsequent STI acquisition.
Although approximately 24% of sexually active women were excluded due to incomplete data, their demographic differences were limited to age and socioeconomic factors that were adjusted for in all models, suggesting minimal selection bias.
For HSV-1, infection was primarily associated with age, socioeconomic status, race/ethnicity, and educational attainment. Older age, lower poverty-income ratio, Hispanic ethnicity, and lower educational attainment were strong predictors of HSV-1 seropositivity, consistent with the known cumulative exposure pattern and social gradient of HSV-1 infection in the United States and underscoring persistent disparities across sociodemographic groups. 17 In contrast, HSV-2 infection was strongly influenced by sexual-behavior variables. Earlier age at sexual debut, a greater number of lifetime sexual partners, and higher body mass index were independently associated with HSV-2 seropositivity, whereas HPV vaccination was not. These results emphasize that HSV-2 risk is shaped predominantly by behavioral and socioeconomic factors rather than HPV vaccination status. Interestingly, vaccinated women, who tended to report earlier sexual debut and more lifetime partners, did not exhibit higher HSV-2 prevalence after adjustment. This may reflect greater engagement with preventive health services and safer sexual practices—such as condom use and regular screening—among vaccine recipients.
Our results align with prior research showing no association between HPV vaccination and increased sexual risk-taking or STI incidence. For example, systematic reviews by Kasting et al. 4 and others 18 consistently report no behavioral risk compensation. Donken et al. 19 and Bednarczyk et al. 20 likewise observed no increases in pregnancy or non-HPV STIs after vaccination. Our study extends this evidence base specifically to HSV, which has rarely been examined separately.
Although our adjusted estimate for HSV-2 was slightly below 1 (aOR ≈0.9), this was not statistically significant and is unlikely to represent a biological effect. Instead, it may reflect health-seeking behaviors among vaccine recipients, including regular gynecological care and STI screening. 21 Large-scale surveillance from the United Kingdom similarly reported no post-vaccine increases in adolescent sexual activity. 5
Several factors likely explain the absence of increased HSV risk: Preventive behavior clustering – HPV-vaccinated women are more likely to participate in other preventive practices, including cervical cancer screening. 22 Provider recommendation and ongoing care – Vaccination typically occurs within ongoing healthcare, where counseling emphasizes condom use and STI screening.23–25 Psychosocial factors – Vaccination encounters may serve as opportunities for accurate sexual health education, mitigating misconceptions. 9
We used multivariable adjustment and IPTW to address confounding. While unmeasured factors (e.g., religiosity, parental influence) may remain, they would likely bias toward higher STI rates among vaccinated women—yet this pattern was not observed. The cross-sectional design limits causal inference, and some women may have been vaccinated after HSV acquisition. Nevertheless, if risk compensation were present, higher HSV prevalence would still be expected in vaccinated participants. Limitations also include reliance on self-reported vaccination, lack of temporal data on vaccination and sexual debut, potential residual confounding, and restriction to women, limiting generalizability.
Despite these limitations, the present results have important public health implications.
They support the conclusion that HPV vaccination does not increase HSV risk and should reassure providers, parents, and policymakers. HPV vaccination should continue to be promoted for its proven cancer-prevention benefits, alongside counseling that it does not protect against non-HPV STIs such as HSV, Chlamydia trachomatis, or HIV. Dispelling misconceptions about sexual disinhibition may strengthen public confidence and improve vaccine uptake, while supporting comprehensive sexual health strategies.
Supplemental Material
Supplemental Material - Human papillomavirus vaccination and herpes simplex virus infection: A cross-sectional analysis of young women in the United States
Supplemental Material for Human papillomavirus vaccination and herpes simplex virus infection: A cross-sectional analysis of young women in the United States by Jiachang Hu, Hongyu Han, Jianlong Zhu and Ying Shan in International Journal of STD & AIDS.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research was supported by the Specialty Feature Construction Project of Pudong Health and Family Planning Commission of Shanghai (Grant No. PWZzb2022-29).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
