Abstract
Introduction
Acute HIV infection (AHI) diagnosis is challenging due to complex diagnostics and low prevalence. We tested different targeting approaches for AHI testing using data from a cross-sectional study of STI burden in Eswatini.
Methods
From June 2022 to April 2023, adults underwent routine HIV and viral load (VL) testing (Xpert) and completed a questionnaire on socio-demographics, behavioral characteristics and current symptoms. AHI was defined as negative/discordant serial HIV rapid diagnostic test and VL ≥10,000 copies/mL. We used lasso regression to determine risk factors for AHI and build study-specific predictor risk score (PRS). We evaluated the ability of the PRS and other targeting approaches to predict AHI.
Results
Of 1064 participants, ten (0.9%) had AHI. The 10-parameter PRS at cut-off ≥10.8 had the area under the curve (AUC) 0.87 (0.86–0.89), sensitivity 100% (69.2–100), and proportion needed to test (PNT) 26.1%. At the cut-off ≥14.1 the AUC was 0.85 (0.71–0.98), sensitivity 80% (44.4–97.5) and PNT 11.5%. A previously developed PRS and targeting young women reporting AHI symptoms also performed well, but not the WHO-recommended clinical screening criteria for AHI (sensitivity 40%).
Conclusions
Targeted approaches that combine AHI symptoms and locally relevant characteristics may be an efficient way to support the scale-up of AHI testing.
Keywords
Introduction
Acute HIV infection (AHI) is a period of a few weeks between viral acquisition and the appearance of HIV antibodies, 1 associated with high viral load (VL), seeding of viral reservoirs and a disproportionally high likelihood of onward transmission.1–3 Undiagnosed and untreated acute and early HIV infection may be interfering with benefits of HIV control strategies, as despite impressive progress in HIV control, the HIV incidence remains high, with 1.3 million new infections in 2023, more than three-times the UNAIDS 2025 target. 4
The relative role and contribution of AHI in onward transmission is likely to become increasingly important as the proportion of the population who know their HIV status, are on antiretroviral therapy (ART) and have an undetectable VL increases. 4 Missed AHI may allow inappropriate initiation of pre-exposure prophylaxis (PrEP), potentially leading to HIV resistance, especially with long-acting injectable PrEP.5,6
Diagnosis of AHI is challenging because it cannot be detected by routinely used rapid diagnostic tests (RDTs), duration is short and overall prevalence is low. In high-income countries AHI is routinely diagnosed with sensitive fourth-generation antibody/antigen immunoassays, but these are not available in resource-limited settings. Molecular testing became more widely available through point-of-care platforms (i.e. Xpert), but it remains expensive. AHI is rarely diagnosed in sub-Saharan Africa and there are currently no World Health Organization (WHO) or other guidance to support AHI testing. 7
Different targeting strategies for AHI testing have been proposed to reduce the proportion needed to test (PNT) and increase the AHI yield, for example targeting testing of patients seeking urgent care for malaria,8,9 or attending sexually transmitted infection (STI) clinics.10,11 Several predictor risk scores (PRS) algorithms to identify individuals at high risk of AHI who should be prioritized for the molecular testing have been developed in sub-Saharan Africa.12–19 Despite the promising performance of many PRSs, the uptake in routine practice has been limited, possibly because the PRSs are often context-specific16,17 or implementation in routine settings is challenging.
Eswatini is a country with high HIV prevalence in the general population (24.8% among adults in 2021) and excellent HIV control, achieving the 95–95–95 targets (93.7–97.3–96.2), but although decreasing, HIV incidence remains high (0.6% overall in 2021). 20 Novel and innovative approaches are needed to address the persistently high HIV incidence, including identifying HIV infection during the AHI period. We used data from a cross-sectional study of STI burden in rural Eswatini to develop and test different targeting approaches for AHI testing.
Methods
This was a cross-sectional study, nested within a larger study evaluating burden of STIs, among individuals presenting to health facilities and HIV care sites in Shiselweni region in Eswatini between July 2022 and March 2023.21–23 The analysis in this paper includes participants who underwent routine HIV testing and had a negative or discordant HIV RDT result.
Study setting
Shiselweni is a predominantly rural region in southern Eswatini, with a population of just over 200,000 people and a generalized HIV epidemic, with HIV prevalence of 26.5% among adults. 20 The study was conducted at six sites: Nhlangano second-level health center, two rural primary-level health clinics, one factory clinic and two stand-alone MSF-supported community sites. All sites offer HIV testing services (HTS) and HIV prevention and care services, reproductive health care and management of STIs. All care is provided free of charge.
STI burden study procedures
Details are described elsewhere. 21 Briefly, adults (18 years and older) were invited to participate in the study, if they attended HTS, received ART refills or received syndromic diagnosis of STI at one of the study sites between July 2022 and March 2023. Those who provided informed consent were invited to complete a detailed questionnaire about socio-demographic and behavioral characteristics, and current and past (2 weeks) symptoms; either self-administered electronic or paper questionnaire or they could be assisted by study staff. Participants provided venous blood for HIV VL and further STI diagnostics, and urine sample for pregnancy test, urine leucocyte esterase and molecular STI diagnosis. A nurse conducted clinical examination.
HIV testing and linkage to care
Participants with negative or unknown HIV status were eligible for routine HIV testing, performed by HTS counselors on finger-prick blood using the standard serial RDT algorithm used in Eswatini (Determine™ as the first-line test, followed by Uni-Gold™ if positive). HIV VL was measured in the study laboratory in Nhlangano health center on Cepheid’s GeneXpert® platform using plasma.
Established HIV infection was defined according to the national serial HIV testing algorithm, based on detectable HIV antibodies on Determine™ and Uni-Gold™ HIV RDT and detectable HIV VL. AHI was defined as a negative or discordant (Determine™ positive and Uni-Gold™ negative) HIV test result on the serial RDT algorithm, combined with either (1) a VL >10,000 copies/mL, or (2) an initially detectable VL <10,000 copies/mL confirmed by a second detectable VL on a plasma sample collected prior to ART initiation.24,25 Serial HIV RDT algorithm negative or discordant samples with undetectable HIV VL were considered negative.
Participants who tested positive for HIV (established HIV or AHI) were linked to care. Participants testing negative for HIV were offered HIV prevention: PrEP (oral PrEP or vaginal ring), condoms and/or voluntary medical male circumcision. Participants diagnosed with syndromic STI were treated as per national algorithm, and their treatment was adapted based on the laboratory results.
Eligibility criteria for AHI analysis
Participants who underwent routine HTS and had an HIV negative or discordant result based on routine HTS algorithm were included in the analysis. Exclusion criteria included missing VL result and missing complete risk factor questionnaires.
Data management
We coded all binary variables as “1” for positive response and “0” for all other answers (negative, missing, or not knowing the answer), assuming those without characteristics and symptoms would be more likely to omit reporting. We used one-hot encoding for categorical variables with more than two categories, thus representing categorical variables as group of binary variables with each variable representing one category. This allowed the lasso regression model to handle each category separately and benefit from L1 regularization. 26
Statistical analysis
Statistical analyses were performed using Stata Statistical Software: Release 18 (College Station, TX: StataCorp LLC). We described baseline characteristics using counts and proportions for categorical variables and medians with interquartile range (IQR) for continuous variables. Categorical variables were compared using Fisher exact test.
We established a study specific PRS for identification of AHI, using lasso regression for prediction with 7-fold cross-validation. 26 Lasso allows for use of large number of predictors; it performs automatic feature selection by shrinking coefficients towards zero, eliminating irrelevant ones and thus performing variable selection,26,27 and it can select models with a limited number of coefficients.
Characteristics of patients included in the study, by AHI status.
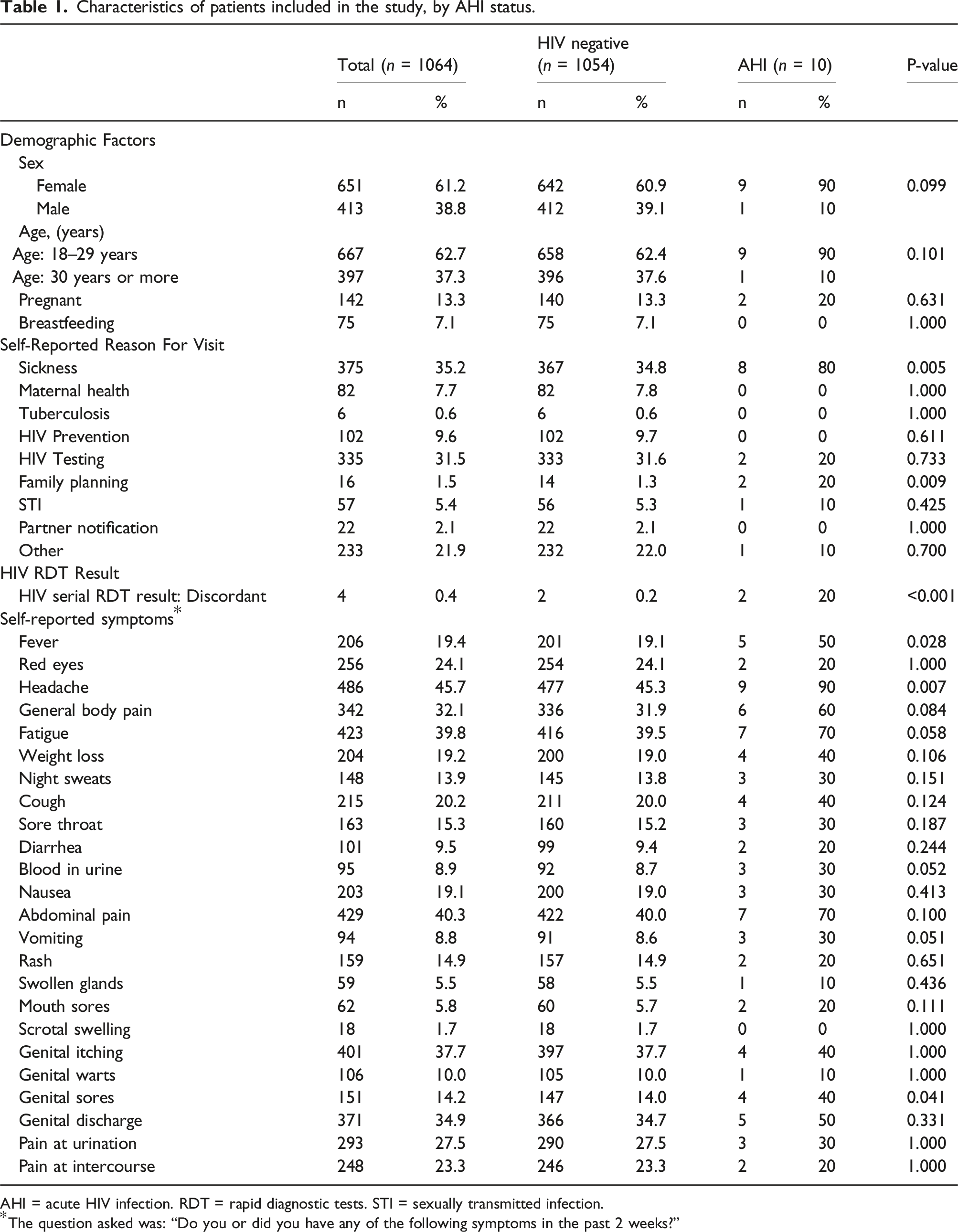
AHI = acute HIV infection. RDT = rapid diagnostic tests. STI = sexually transmitted infection.
*The question asked was: “Do you or did you have any of the following symptoms in the past 2 weeks?”
To obtain participant’s individual risk of AHI we summed up the different penalized standardized beta coefficients for each predicting factor and multiplied it by 10 (to enhance interpretability and facilitate score calculation). We plotted and cross-tabulated the sensitivity, specificity and PNT estimates at each cut-off level of the PRS, chose the optimal cut-off point that would give best sensitivity with lowest PNT and evaluated performance of PRS at each selected cut-off point. PNT was defined as the proportion of participants who scored at, or above the score threshold or fulfilled targeted criteria and would qualify for HIV VL testing and the “number needed to test” (NNT) as the number of participants who would have to be tested to identify one AHI.
Other targeting strategies
We evaluated performance of two previously established PRSs: “Nhlangano 2019 PRS” established in the same setting 17 and “consensus PRS” developed from three cross-sectional and one prospective study in sub-Saharan Africa. 16 Details of the previously established PRSs at their best performing cut-off points are shown in Tables S1 and S2
We also evaluated performance of different simple screening approaches that would not require calculating PRS and could be more easily implemented in routine settings. We tested performance of “symptoms suggestive of AHI” (sore throat, fever (reported or measured as >37.5°C at time of clinical consultation) or any reported STI symptom) as used to select participants in Nhlangano 2019 study, 17 “WHO AHI symptoms” (fever, rash, sore throat, aches and pains, lymphadenopathy, mouth sores and/or headache) (6) , and combination of “WHO AHI symptoms” with recent (last 14 days) sexual intercourse, as proxy for recent exposure, as recommended screening criteria for delaying PrEP initiation by WHO. 6 In addition, as young women are carrying most of the burden of HIV incidence in Eswatini, 20 we also evaluated performance of simple criteria “young woman” and a combination of being a “young woman” with “WHO AHI symptoms”.
Ethics
All participants provided written informed consent. The study was approved by the Eswatini Health and Human Research Review Board (EHHRRB096/2021) and the MSF Ethics Review Board (ID:2154).
Results
Of 1195 participants who underwent HIV testing, 39 (3.3%) were newly diagnosed with established HIV infection, 22 (2.9%) were missing information on final HIV status and 70 (5.9%) were missing questionnaire data and were excluded from further analysis (Figure S1). Finally, data of 1064 participants were included in the analysis, their median age was 27 years (IQR 22–33) and 651 (61.2%) were female. Ten (0.9%) participants were diagnosed with AHI. Participants who visited study sites because of illness or family planning, reported to have had fever, headache or genital sores and those with discordant serial HIV RDT result were more likely to be diagnosed with AHI (Table 1). All risk factors measured are described in Tables 1 and S3.
Newly established PRS
Factors identified as predictors of AHI in the long and simplified lasso model with their non-zero penalized beta coefficients (multiplied by 10 as used in the score).

Negative coefficients indicate protective factors. AHI = acute HIV infection.
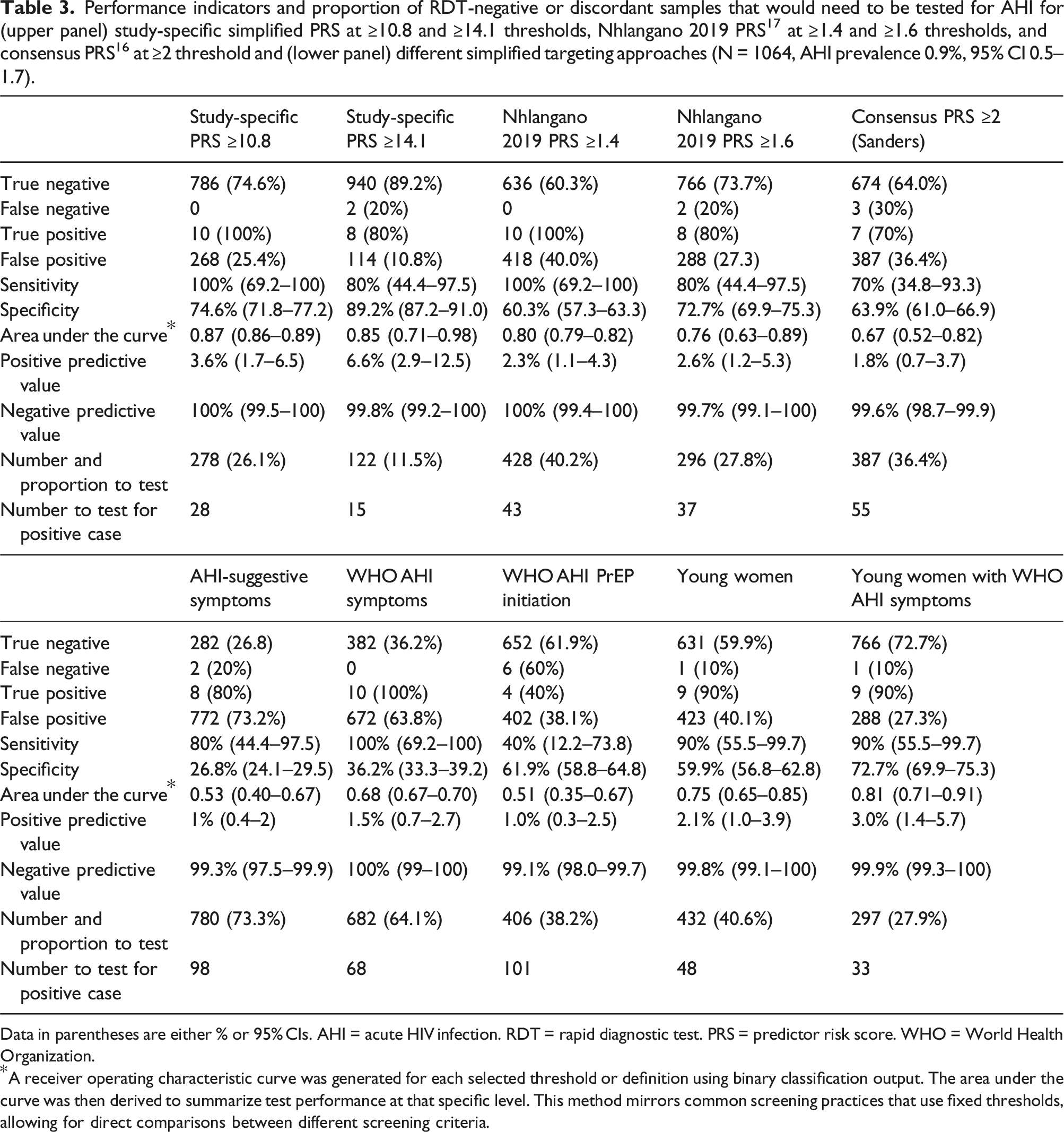
We identified two well performing cut-off thresholds for the simplified PRS (Figure 1 and Table 3). The cut-off threshold at ≥10.8 points had AUC 0.87 (95% CI 0.86–0.89), sensitivity 100% (95% CI 69.2–100) and specificity 74.6% (95% CI 71.8–77.2). The PNT was 26.1% and NNT 28 (Table 3). PRS at the cut-off threshold ≥14.1 points had similar overall performance with AUC 0.85 (0.71–0.98), but lower sensitivity (80%, 44.4–97.5), higher specificity (89.2%, 87.2–91.0) and a lower PNT (11.5%) and NNT (15), and would miss two (20%) AHI. Estimates of sensitivity, specificity and proportion needed to test at different PRS cut-off thresholds. The dashed vertical line indicates the cut-off thresholds at ≥10.8 points (sensitivity 100%, specificity 74.6% and proportion needed to test with molecular test 26.1% of the sample) and ≥14.1 points (sensitivity 80%, specificity 89.2% and proportion needed to be tested of 11%). *X-axis values represent cut-off thresholds of predicted scores, displayed in increasing order. The values are not evenly spaced numerically, they are plotted at uniform visual intervals for interpretability. Performance indicators and proportion of RDT-negative or discordant samples that would need to be tested for AHI for (upper panel) study-specific simplified PRS at ≥10.8 and ≥14.1 thresholds, Nhlangano 2019 PRS
17
at ≥1.4 and ≥1.6 thresholds, and consensus PRS
16
at ≥2 threshold and (lower panel) different simplified targeting approaches (N = 1064, AHI prevalence 0.9%, 95% CI 0.5–1.7). Data in parentheses are either % or 95% CIs. AHI = acute HIV infection. RDT = rapid diagnostic test. PRS = predictor risk score. WHO = World Health Organization. *A receiver operating characteristic curve was generated for each selected threshold or definition using binary classification output. The area under the curve was then derived to summarize test performance at that specific level. This method mirrors common screening practices that use fixed thresholds, allowing for direct comparisons between different screening criteria.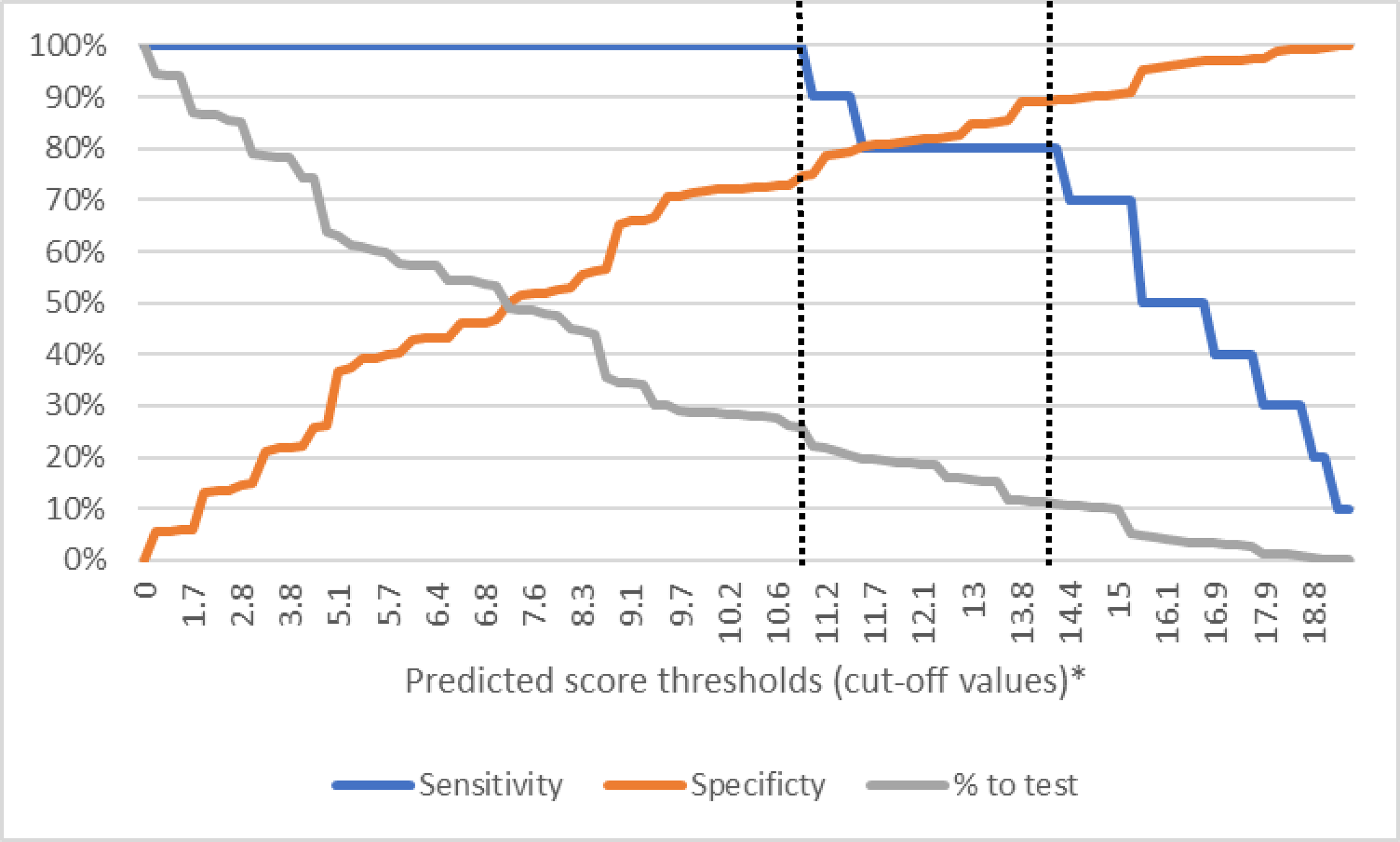
Validation of other PRSs
The “Nhlangano 2019” score performed well at the cut-off ≥1.4: AUC 0.80 (0.79–0.82), sensitivity 100% (69.2–100), specificity 60.3% (57.3–63.3), and a PNT of 40.2%, and slightly less well at the cut-off of ≥1.6: AUC 0.76 (0.63–0.89), sensitivity 80% (44.4–97.5), specificity 72.7% (69.9–75.3), and a PNT of 27.8% (Table 3). The “consensus score” with threshold of ≥2 points performed less well: AUC 0.67 (0.52–0.82), sensitivity 70% (34.8–93.3), specificity 63.9% (61.0–66.9), and a PNT of 36.4%. Figure 2 (upper panel) compares AUCs of different PRS tested. Comparison of area under the receiver operator characteristic curves (AUC)* for (upper) study-specific simplified PRS at ≥10.8 and ≥14.1 thresholds, Nhlangano 2019 PRS (17) at ≥1.4 and ≥1.6 thresholds, and consensus PRS (19) at ≥2 threshold and (below) different simplified targeting approaches. * A receiver operating characteristic curve was generated for each selected threshold or definition using binary classification output. The area under the curve was then derived to summarize test performance at that specific level. This method mirrors common screening practices that use fixed thresholds, allowing for direct comparisons between different screening criteria.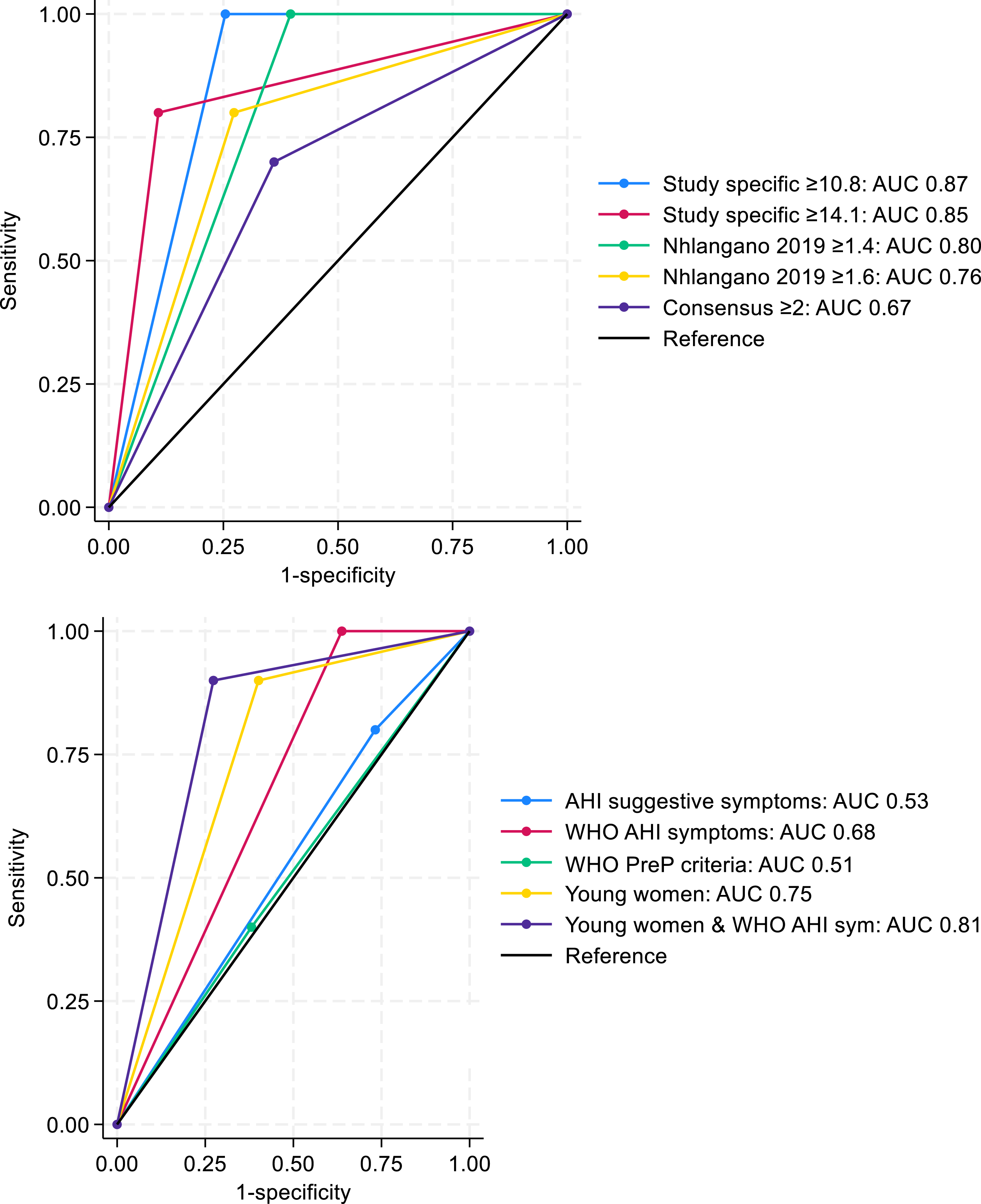
Simpler non-PRS targeted approaches
We explored several alternative targeting approaches that do not require additional screening to what should be done in routine care (Table 3). The “symptoms suggestive of AHI” had poor predictive ability (AUC 0.53, 0.40–0.67), with a large PNT (73.3%) and NNT (98), while missing two (20%) AHI.
Using “WHO AHI symptoms” would identify all 10 AHI, with better performance (AUC 0.68, 0.67–0.70), but with large PNT (64.1%). Performance of WHO-recommended screening criteria for AHI prior PrEP initiation was poor (AUC 0.51, 0.35–0.67) with lower PNT (38.2%), and missing six (60%) AHI.
Targeting young women performed better, with better predictive ability (AUC 0.75, 0.65–0.85), similar PNT (40.6%), and missing one (10%) AHI. Targeting young women with “WHO AHI symptoms” had the best performance (AUC 0.81, 0.71–0.91) with lowest PNT (27.9%), while missing one (10%) AHI. Figure 2 (lower panel) compares AUCs of different simplified strategies.
Discussion
Our study explored various targeting strategies for AHI testing. Several approaches performed well, reducing the PNT from 100% to 10-40% while still detecting most infections. Simplified approaches combining AHI symptoms with locally relevant characteristics (young women) worked particularly well.
Our study-specific PRS included similar predictive factors to previously established PRSs (Table S2), i.e. general flu-like symptoms or STI-suggestive symptoms and signs. Younger age, being female, and discordant RDT results have all been previously included, but some studies only targeted younger individuals, men who have sex with men (MSM), and others excluded participants with discordant RDT results, thus making direct comparisons difficult. The two newly identified predictors (reason for clinic visit being family planning or feeling sick) are specific to the study design, recruiting participants from the general population attending health facilities. Feeling sick was the strongest predictor, and confirms findings from previous studies that patients with AHI often seek care and present at facilities, 28 which presents the opportunity for targeted AHI screening.
The performance of “Nhlangano 2019 PRS” 17 in our study data set was good, but the “consensus PRS” 16 performed less well. As documented previously,16–18 the PRSs commonly perform less well outside study settings (Table S1 compares performance of published PRS in original and other datasets), as performance depends on local HIV epidemiology, type of population included in the study, study setting and methodology, as well as how different variables were assessed and ascertained. This highlights the importance of local validation of even very well performing PRSs.
Overall, the good performance of the study-specific PRS in our study population compares to other PRS developed in sub-Saharan Africa in the past decades.12–19 But although many PRS performed well, there has been limited real-life uptake, with the exception of studies in coastal Kenya, where Sanders et al implemented the “consensus” score in primary clinics and explored different approaches to AHI testing.16,29 One reason for low uptake is the complexity of integrating PRS into routine care, with overburdened healthcare workers struggling with additional workload and already performing other screening strategies (e.g, tuberculosis). However, the digital era offers new opportunities, for example digital tools integrating several screening algorithms or offering self-administration, either at facility or at home, within HIV self-testing strategies. Further research is needed to test these possibilities.
Unaddressed AHI is of particular concern in the context of PrEP, 5 especially with the introduction of injectable PrEP. More accessible laboratory testing would not only detect missed AHI, but also avoid missed opportunities for PrEP initiation, as current guidelines recommend delaying PrEP initiation for one month for those who screen positive for AHI.6,30 This AHI screening criteria however performed poorly in our study. While “AHI symptoms” alone identified all AHI, restricting to potential exposure in the past 14 days reduced the sensitivity to 40%. The sensitivity of “AHI symptoms” was better than expected; HIV subtype C, which is prevalent in Eswatini, 31 was previously described as less likely to present with AHI symptoms, 32 although more recent work has linked AHI symptoms to innate immunity rather than HIV subtype. 33 But the sensitivity of possible risk exposure was low; only four of 10 participants diagnosed with AHI reported their last sexual episode within the past 14 days (Table S3). Participants may not recall the exact date of their last sexual contact, or choose to omit reporting because of social desirability. 34 Alternatively, the 14-day period since high-risk exposure may be too short to capture the full window period35,36; for example Delaney et al. estimated the median window period for sensitive antibody RDT to be 31.1 days (95%CI 26.2, 37.0), more than twice longer. 37 The sensitivity of the currently recommended clinical screening criteria for AHI in the context of PrEP initiation should be verified in further studies, as this is currently the standard of care in most routine settings in sub-Saharan Africa.
Zambia, the first country in sub-Saharan Africa to introduce injectable PrEP in routine care recently implemented blanket molecular testing for AHI before injectable PrEP initiation. In the first 3 months they identified six AHI among 927 individuals tested (0.65%), highlighting the importance of AHI testing but also substantial amount of resources needed in the case of blanket screening (154 tests to detect one AHI). 38
Restricting targeting AHI testing to young women with AHI symptoms performed well (sensitivity 90%, AUC 0.81). This finding probably reflects the context of Eswatini, where HIV incidence is concentrated in young women, 20 but might be different elsewhere. Nevertheless, as we and others have demonstrated,16,17,19 targeted approaches combining AHI symptoms and locally relevant characteristics may be an efficient way forward.
Limitations and strengths
The main limitation of our study is the small number of AHI cases detected; we established the study-specific PRS based on only 10 AHI. We used lasso prediction model to handle a large number of predictors. However, on the individual level, a long questionnaire might have impacted the reliability of answers, and behavioral characteristics in addition are prone to social desirability bias. 39 The generalizability of our findings is unknown, as PRS and other targeted approaches are known to be context-specific.16,17,19 However, unlike most of the existing PRSs which were developed in specific high-risk populations, such as STI clinics,14,19 among MSM 15 or sex workers 13 or within prospective studies,12,13,15,18 our study included the general population attending HTS.
Conclusions
We identified several well-performing targeting approaches for AHI screening that reduced the PNT while still identifying most infections; in particular, the study-specific PRS and the simple approach of targeting young women with AHI symptoms performed well. We also show that current WHO-recommended clinical screening for AHI may miss some AHI. We reaffirm the importance of AHI symptoms in targeting AHI strategies, and the need to adapt and validate screening approaches to the local context. Simple and accessible strategies are needed to address AHI in high burden settings, and targeted approaches may be one way forward until there is universal access to accurate and affordable AHI testing.
Supplemental Material
Supplemental Material - Targeted approaches for acute HIV infection diagnosis in rural Eswatini
Supplemental material for Targeted approaches for acute HIV infection diagnosis in rural Eswatini by Iza Ciglenecki, Bernhard Kerschberger, Esther Mukooza, Skinner Lekelem, Nombuso Ntshalintshali, Mpumelelo Mavimbela, Sindisiwe Dlamini, Lenhle Dube, Nomvuyo Mabuza, Nelly Staderini, Melat Haile, Tom Ellman, Antonio Flores, Laurence Toutous Trellu, Olivia Keiser, Sindy Matse, Alexandra Calmy in International Journal of STD & AIDS
Footnotes
Acknowledgements
This study was funded by MSF, Operational center Geneva. We thank the study participants and MOH and MSF staff at participating sites who supported the study. We also thank MSF staff in Nhlangano, Mbabane and Geneva for supporting the study and Olaya Astudillo for support in the manuscript preparation.
Author contributions
IC, BK, and AC conceived and designed the analysis. BK, EM, SK and NN curated and verified the data. IC and BK performed the analysis. IC wrote the original draft. All authors contributed to the interpretation of the findings and reviewed the paper.
Declaration of conflicting interests
MSF provided support in the form of salaries for IC, BK, EM, SL, NB, NL, MH, ET and AF. AC declares unrestricted educational grants from Gilead Sciences, ViiV Healthcare and MSD, paid to the institution. All other authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Médecins Sans Frontières.
Data Accessibility Statement
The minimal data set underlying the findings of this paper are available on request, in accordance with the legal framework set forth by MSF data sharing policy. MSF is committed to sharing and disseminating health data from its programmes and research in an open, timely, and transparent manner to promote health for populations while respecting ethical and legal obligations towards patients, research participants, and their communities. The MSF data sharing policy ensures that data will be available upon request to interested researchers while addressing all security, legal, and ethical concerns. Readers can contact the MSF generic address
Ethics statement
All participants provided written informed consent. The study was approved by the Eswatini Health and Human Research Review Board (EHHRRB096/2021) and the MSF Ethics Review Board (ID:2154).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
