Abstract
Background
Bacterial sexually transmitted infections (STIs) continue to increase in the United States. Despite evidence of the effectiveness of doxycycline post-exposure prophylaxis (Doxy-PEP) to prevent STIs, little is known about providers’ attitudes and willingness to implement Doxy-PEP.
Methods
An online questionnaire was sent to 575 clinical providers in New York State in September 2022.
Results
Ninety-one eligible individuals responded. Most providers served men who have sex with men (MSM) (84%); reported willingness to recommend Doxy-PEP (98%, 77%, and 67% for chlamydia, syphilis, and gonorrhea, respectively); preferred Doxy-PEP administration for MSM and transgender populations; believed recurring bacterial STIs (88%) and reported condomless sex (85%) were the most important characteristics to consider for recommending Doxy-PEP; and were concerned about antibiotic resistance (90%) (primarily for
Conclusions
Study results suggest high willingness and support for implementing Doxy-PEP. As this survey was administered prior to national guidelines on the use of Doxy-PEP, these data can be used as a baseline to compare to studies conducted after their release to formulate appropriate messages for providers to improve implementation.
Introduction
Despite increasing rates of bacterial sexually transmitted infections (STIs), evidence-based, effective biomedical preventions have been limited to the latex condom, until recent promising data on doxycycline post-exposure prophylaxis (Doxy-PEP). 1 Doxycycline is a tetracycline with high efficacy and tolerability and is widely employed to prevent and treat various bacterial infections. 2 While commonly used for STI treatment,3,4 using doxycycline to prevent bacterial STIs is a relatively new approach involving chemoprophylaxis before or after potential exposure(s) to an STI, as a pre-or post-exposure prophylaxis (Doxy-PrEP / Doxy-PEP, respectively).
In clinical trials, with high tolerance and minimal side effects, Doxy-PEP reduces the number of new cases of chlamydia, syphilis, and gonorrhea, though the effect on reducing gonorrhea is less pronounced, presumably due to the emergence of drug-resistant strains.5–8 These trials predominantly involve cohorts of men who have sex with men (MSM) and transgender women: living with HIV or on HIV-PrEP and with a previous history of bacterial STIs. A positive community-level impact has also been demonstrated in modeling studies when doxycycline is used among MSM with a higher likelihood of contracting STIs.9,10 A decrease in the monthly number of male rectal chlamydia, male rectal gonorrhea, and adult male syphilis cases was demonstrated in San Francisco following the release of a clinical guideline on Doxy-PEP use. 11
In addition to the effectiveness and safety, the literature consistently shows high acceptance and willingness of priority populations (predominantly men who have sex with men) to use doxycycline for the prevention of bacterial STIs.10,12–14 Some healthcare authorities also recommend using Doxy-PEP in specific circumstances.15,16 Successful implementation of Doxy-PEP relies on healthcare providers’ willingness to prescribe doxycycline prophylaxis. However, existing literature is scarce and show inconsistent data on clinical providers’ attitudes and beliefs. The lack of clarity often hampers the planning of the effective educational interventions for providers. With intention to address this gap, we conducted a baseline study to assess the willingness of clinical providers to prescribe doxycycline prophylaxis in New York State before state and federal guidelines were released.
Materials and methods
Provider questionnaire, study population, and recruitment
We developed an online questionnaire for clinical providers to assess attitudes, beliefs, and willingness to provide Doxy-Pre- Exposure Prophylaxis (Doxy-PrEP) and/or PEP for three bacterial STIs: chlamydia, gonorrhea, and syphilis. Initially, the questionnaire was pilot tested among 12 clinical providers funded by the New York State Department of Health AIDS Institute and minor adjustments were made for clarity following pilot feedback. Pilot responses were not included in the final analyses.
After the pilot, a total of 575 potential providers were identified through the New York State Department of Health AIDS Institute Voluntary HIV PrEP/PEP Provider Directory and clinical advisory groups. Everyone from the list received three recruitment emails, including an initial invitation and two reminders between August 30 and September 26, 2022. Email invitations included an opportunity for potential participants to share the questionnaire link with other eligible providers in their agency. Participants were eligible if they were authorized to prescribe medication in New York State.
The questionnaire included questions on (1) provider occupational characteristics; (2) attitudes and (3) beliefs toward doxycycline pre- and/or post-exposure prophylaxis. Attitude-related questions included the perceived need and appropriateness of Doxy-PrEP/PEP based on risk behavior and patient population. In this section, we assessed providers’ opinions on the potential consequences of Doxy-PrEP/PEP, including drug resistance and side effects. Belief-related questions included the efficacy of doxycycline prophylaxis for preventing specific STIs and facilitators and barriers to the provision of Doxy-PrEP/PEP. Attitudes and beliefs were primarily assessed using 5-item Likert scales to determine agreement with questions of interest; the degree of preference for using doxycycline prophylaxis and concern about potential drug resistance of
Statistical analyses
Descriptive statistics were performed to assess demographic and occupational characteristics of survey respondents. Some responses were grouped due to small sample sizes. For the number of years of experience as a clinician/medical provider, less than 5 years and 5–10 years were categorized as “Less than 10 years.” Similarly, region within New York State was categorized as New York City (the Bronx, Brooklyn, Manhattan, Queens, and Staten Island) and rest of state (the remaining counties in New York State). Additionally, since the majority of respondents were medical doctors, those who had current roles as nurse practitioners, physician assistants, and HIV medical directors were categorized as “Other” for bivariate analyses.
The McNemar-Bowker test of symmetry was used to assess differences in providers’ willingness and preferences, as well as concerns and perceived barriers to prescribing Doxy-PEP. With this test, often used with pre/post-tests, we were able to determine if responses between two questions were symmetrical (e.g., when asking about chlamydia vs gonorrhea vs syphilis, or Doxy-PEP vs Doxy-PrEP). Due to smaller sample sizes, the exact test was used.
17
To assess the differences in attitudes and beliefs towards using Doxy-PrEP/PEP by provider characteristics, Rao-Scott chi-square tests were used. Fisher’s exact test was performed where expected cell counts were less than five. All analyses were conducted in SAS 9.4. Tests with a
Results
Though the survey included questions on Doxy-PrEP, the results presented have been restricted to Doxy-PEP, as the analysis did not show significant differences between the survey results for Doxy-PrEP versus Doxy-PEP, except that more providers are willing to prescribe Doxy-PEP regardless of the Centers for Disease Control and Prevention (CDC) approval (
Demographic characteristics
A total of 99 respondents accessed the survey. Eight participants were excluded from the final sample size as they were not authorized to prescribe medicine (
Characteristics of respondents (
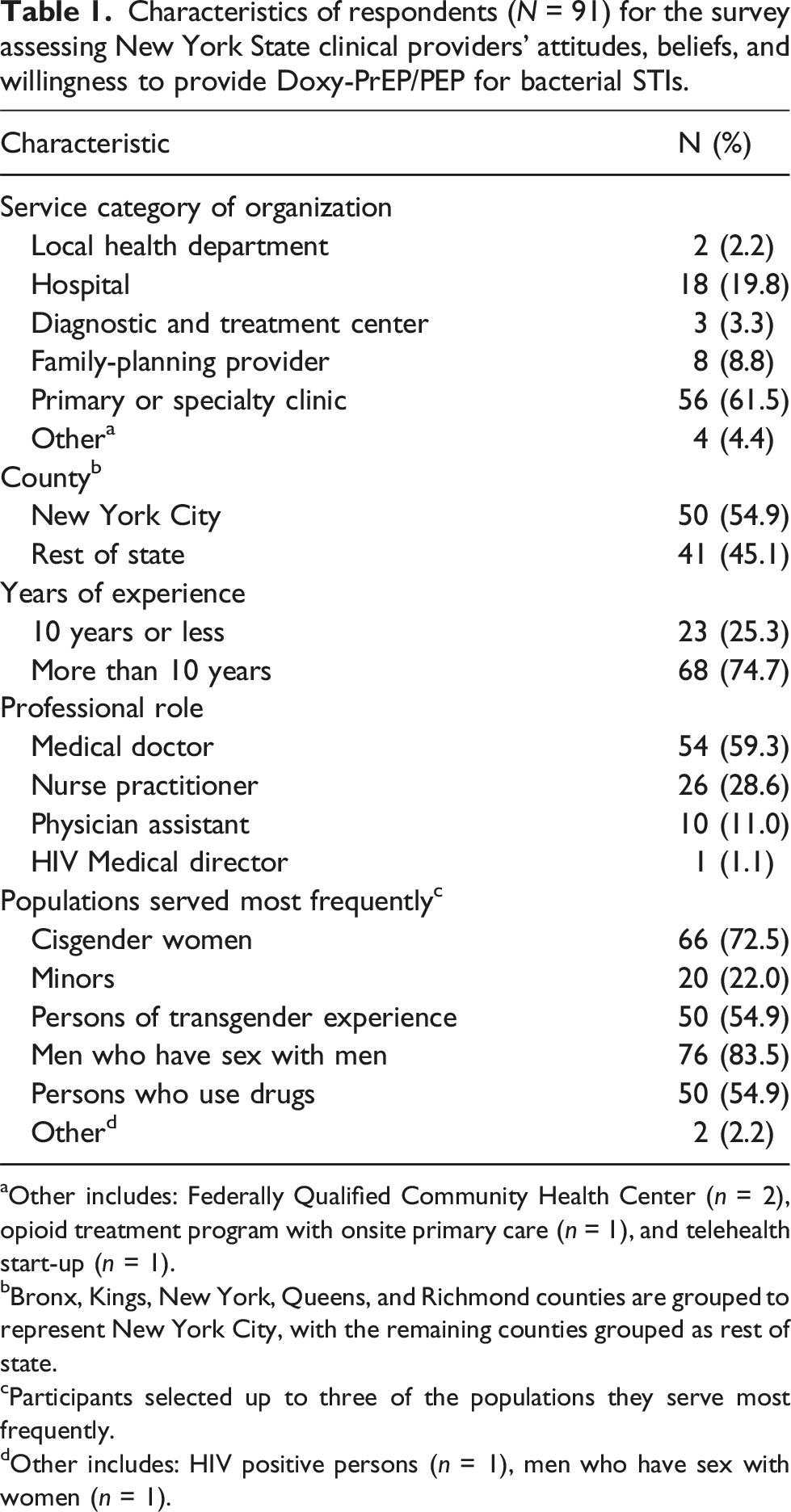
aOther includes: Federally Qualified Community Health Center (
bBronx, Kings, New York, Queens, and Richmond counties are grouped to represent New York City, with the remaining counties grouped as rest of state.
cParticipants selected up to three of the populations they serve most frequently.
dOther includes: HIV positive persons (
Providers’ willingness and preferences to prescribe Doxy-PEP
We evaluated providers’ willingness to prescribe Doxy-PEP across various scenarios. Of 89 respondents, 36 (40.5%) indicated that they would recommend Doxy-PEP to prevent bacterial STIs regardless of approval by the CDC, 48 (53.9%) would only recommend Doxy-PEP if it was approved by the CDC, and five (5.6%) would not recommend Doxy-PEP even if it was approved by the CDC (Figure 1). Most survey respondents would recommend Doxy-PEP to prevent bacterial STIs (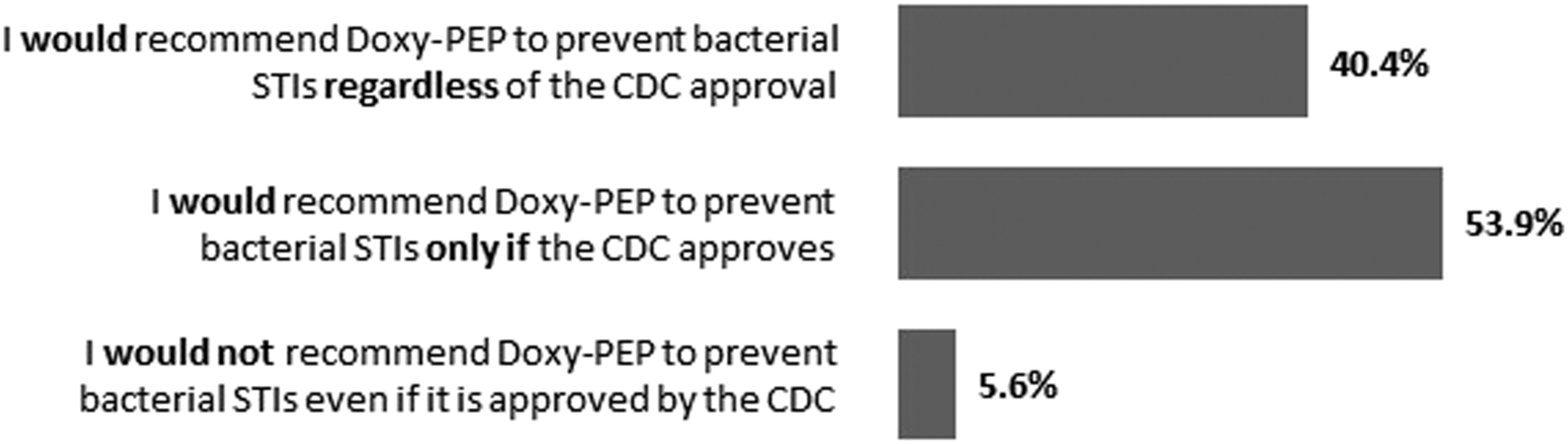
If administration of Doxy-PEP for the prevention of STIs was approved by the CDC, respondents were most likely to recommend Doxy-PEP for the prevention of chlamydia (Figure 2). Of 84 respondents, 82 (97.6%) would recommend Doxy-PEP for the prevention of chlamydia, versus 65 (77.4%) for syphilis, and 56 for gonorrhea (66.7%). The differences in the responses for the three STIs were found to be statistically significant using the McNemar-Bowker test of symmetry ( Percent of respondents that would recommend Doxy-PEP for the prevention of each STI, if approved by the CDC. Respondents would be most willing to recommend Doxy-PEP for the prevention of chlamydia, followed by syphilis and gonorrhea (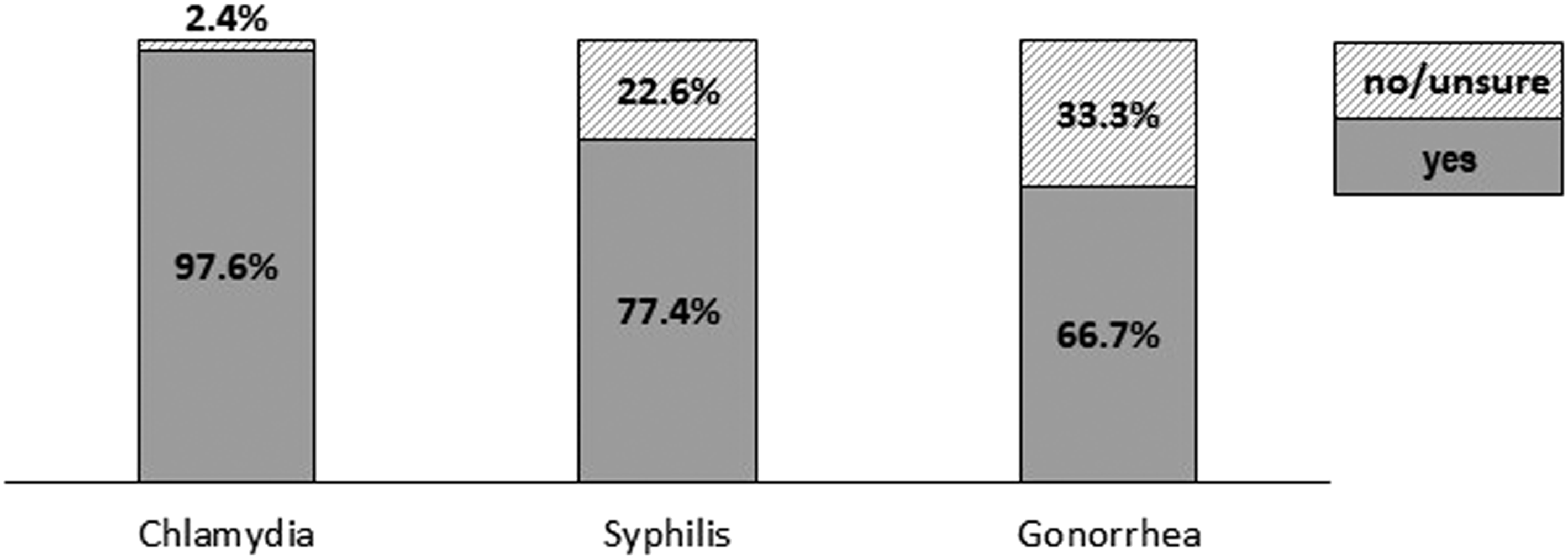
Respondents indicated the strongest preference to recommend Doxy-PEP for MSM (45.5%, 30/66), followed by persons of transgender experience (37.9%, 25/66). Respondents identified recurrence of bacterial STIs and practice of condomless sex as the most important characteristics to consider in identifying a person’s need or appropriateness for Doxy-PEP to prevent STIs (87.9%, 58/66 and 84.8%, 56/66, respectively), while HIV status and HIV PrEP status were stated as the least important (19.7%, 13/66 and 16.7%, 11/66, respectively).
Providers’ concerns and perceived barriers to prescribing Doxy-PEP
When asked about concerns about the potential consequences of Doxy-PEP use, 90.9% (60/66) of respondents were concerned about potential antibiotic resistance, with the greatest concern for antibiotic resistance in Concern of respondents towards the potential drug resistance of 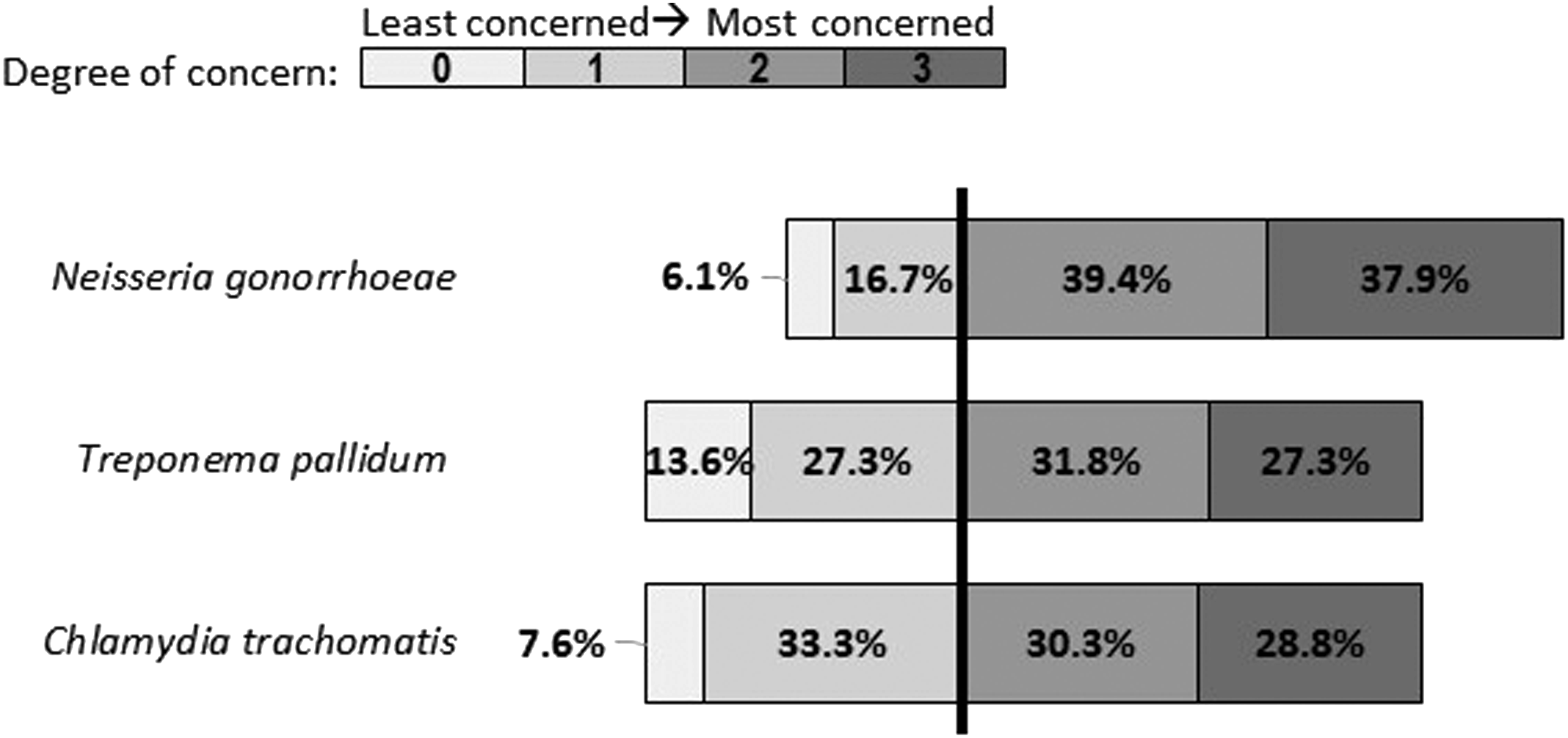
Providers’ attitudes and beliefs by provider characteristics
There was no significant relationship between the level of support for a recommendation for Doxy-PEP to prevent bacterial STIs and years of experience (
Discussion
This baseline study assessing New York State clinical providers’ attitudes and beliefs towards the use of Doxy-PEP to prevent syphilis, chlamydial and gonorrheal infections suggests that most providers accepted Doxy-PEP, particularly if recommended by the CDC. This finding is important as the high level of acceptance noted in this evaluation was in place prior to recommendations on Doxy-PEP put forth by the CDC in 2024. Most respondents in our survey would recommend Doxy-PEP for MSM and persons of transgender experience, as well as for persons engaging in condomless sexual activity and having a history of prior bacterial STIs. Antimicrobial resistance was the most cited concern, whereas insurance cost was perceived as the most difficult barrier to overcome in implementing Doxy-PEP.
Our findings regarding healthcare providers’ willingness to prescribe Doxy-PEP are supported by a previous study restricted to looking at willingness to utilize doxycycline prophylaxis among MSM that showed 89.5% of clinical providers were willing to if it was recommended by the CDC. 12 Both our study and the supporting study were concluded before the CDC’s shared decision-making recommendations on Doxy-PEP released in 2024, so we can assume that more clinical providers would be willing to prescribe Doxy-PEP. Our findings on providers’ willingness to use Doxy-PEP based on patients' previous STI history and population characteristics also align with priority populations identified in most previous studies on doxycycline prophylaxis. Moreover, the New York State Department of Health AIDS Institute (NYSDOH AI) Clinical Guidelines on Doxycycline Post-exposure Prophylaxis to Prevent Bacterial Sexually Transmitted Infections, released after this survey, also suggest using Doxy-PEP for individuals with similar characteristics. Notably, Doxy-PEP is recommended for cisgender men and persons with transgender experiences who have condomless sex with partners assigned male sex at birth and who have a history of bacterial STIs in previous 12 months. Additionally, the guideline does not recommend prescribing Doxy-PEP for those with no or unknown history of STIs. 18
Our results suggest that providers are predominantly concerned about the potential development of antimicrobial resistance for bacterial STIs, which aligns with previous results stating that 80% of providers are worried about potential antimicrobial resistance. 12 However, existing literature does not provide sufficient evidence to determine the risks of antibiotic resistance among Doxy-PEP users. 19 Yet, as the practice of doxycycline self-administration among men who have sex with men increases, antibiotic stewardship has become a major focus.14,20 Insurance costs was identified by clinical providers as another major barrier to Doxy-PEP implementation despite doxycycline being one of the most affordable medications. Previous results indicated that community members also regarded cost as one of the most severe potential barriers. 12 It is surprising that concerns about cost arise, given that doxycycline is inexpensive and would not generally be subject to insurance or formulary restriction lists. Therefore, the implementation of Doxy-PEP should incorporate messaging to address these concerns around potential antibiotic resistance and cost.
There were limitations to our study. First, this study had a small sample size, which gradually decreased as the survey progressed. This was partially addressed analytically through the categorization of variables and the use of statistical methods appropriate for small cell counts. Second, respondents’ beliefs, attitudes, and willingness may differ by provider characteristics such as age, sex, and gender which are not included in the study. Finally, while this survey serves as an important baseline assessment of providers’ attitudes and beliefs towards Doxy-PEP, this survey was conducted before the release of many novel studies supporting the effectiveness of Doxy-PEP and the release of jurisdictional recommendations regarding Doxy-PEP use in specific circumstances.19,20 Thus, the percentage of providers willing to implement Doxy-PEP may be higher if the survey were conducted today. Future research should examine providers’ attitudes and beliefs since the release of Doxy-PEP guidelines.
To our knowledge, this study represents the first attempt to assess US healthcare providers’ attitudes toward using doxycycline for the prevention of each bacterial STI, individually. However, the implementation and success of doxycycline prophylactic programs rely on clinical providers’ use of this prevention strategy. Specifically, it is necessary to understand providers’ attitudes towards Doxy-PEP use across care scenarios and perceived barriers and concerns regarding implementation. These study results and future relevant research after the release of the guidelines regarding using Doxy-PEP will support a deeper understanding of strategies to promote and implement doxycycline prevention programs and address both community and provider needs and concerns.
Footnotes
Acknowledgements
The authors wish to acknowledge the contributions of Dr Cord Stone and colleagues from the AIDS Institute’s Division of HIV, STI, HCV Prevention; Division of HIV and Hepatitis Health Care; Office of Drug User Health; and Office of the Medical Director for their support of the survey development. The authors would also like to acknowledge the clinical providers who took the time to respond to this survey.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Esma Imerlishvili is supported by the Fogarty International Center and the National Institute of Alcohol Abuse and Alcoholism of the National Institutes of Health under Award Number D43 TW011532. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. This analysis was further made possible through the support of Centers for Disease Control and Prevention’s Strengthening Sexually Transmitted Disease Prevention and Control for Health Departments Grant (No.6 MH25PS005175-01) and the Grant or Cooperative Agreement Number 1NU62PS924546. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the Centers for Disease Control and Prevention or the Department of Health and Human Services.
