Abstract
People living with HIV (PLWH) are at higher risk of reactivation of Chagas disease, a neglected tropical disease, caused by Trypanosoma cruzi. There are no data from UK HIV clinics on the prevalence of T. cruzi. We implemented T. cruzi screening at our clinic as part of routine care for PLWH with epidemiological risk factors. Among 86 patients screened, none had positive serology: one seropositive patient was identified due to increased clinician awareness. Implementing T. cruzi screening as part of routine clinical care was feasible, though labour intensive and identified at-risk individuals.
Keywords
Introduction
Chagas disease is caused by Trypanosoma cruzi, a parasite spread by triatomine bugs. It is endemic in South, Central and parts of North America. The World Health Organisation has classified Chagas disease as a neglected tropical disease, that is, a condition mainly prevalent in tropical areas, affecting people living in impoverished communities and causing devastating health, social and economic consequences to more than one billion. 1 In HIV-negative patients, Chagas disease mainly causes a chronic disease leading to cardiomyopathy or to a megacolon/oesophagus. People living with HIV (PLWH) are at higher risk of reactivation of infection which tends to present with acute disease, mainly neurological, but occasionally affecting the heart. 2 HIV clinics in Spain have found T. cruzi seroprevalence of up to 10% among PLWH originating from Latin America,3,4 but there are no equivalent published data from UK HIV clinics. The British HIV Association (BHIVA) guidelines 2 recommend all PLWH with epidemiological risk factors or with a history of blood transfusions or intravenous drug use with contacts from these areas should be screened for T. cruzi. We implemented T. cruzi screening as part of routine care for PLWH born in Mexico, Central and South America attending a large central London HIV clinic. Our main aim was to determine the yield of screening for co-infection in our cohort.
Methods
People living with HIV born in Central and South America categorized as “currently in care” (attendance within the previous 12 months) at the start of the project, were identified from our clinic database and flagged for screening in our electronic patient record (EPR). The electronic patient record of patients currently in care was updated to include to T. cruzi antibody screening at their next attendance/phlebotomy. T.cruzi antibodies were tested using an IgG ELISA. Screened patients were asked to complete a questionnaire including questions about geographical history in Central and South America (where they had lived and for how long), family history of Chagas disease, previous symptoms (gastrointestinal, cardiac and neurological) and prior knowledge of Chagas disease and vector. Patients were asked to either self-complete the questionnaire at the time of blood sampling, by the clinician seeing the patient or by a member of the project team via telephone call or email, where permission was granted on the EPR. Data were collated on an Excel spreadsheet. A patient information leaflet was offered to patients, available in both English and Spanish, and designed to be shared with their families. Patients with positive results were referred to the Hospital for Tropical Diseases for further assessment and management. All staff were trained on Chagas disease and T. cruzi screening. This was a quality improvement project and ethical approval was therefore not sought.
Results
Country of origin of PLWH screened for Chagas disease.
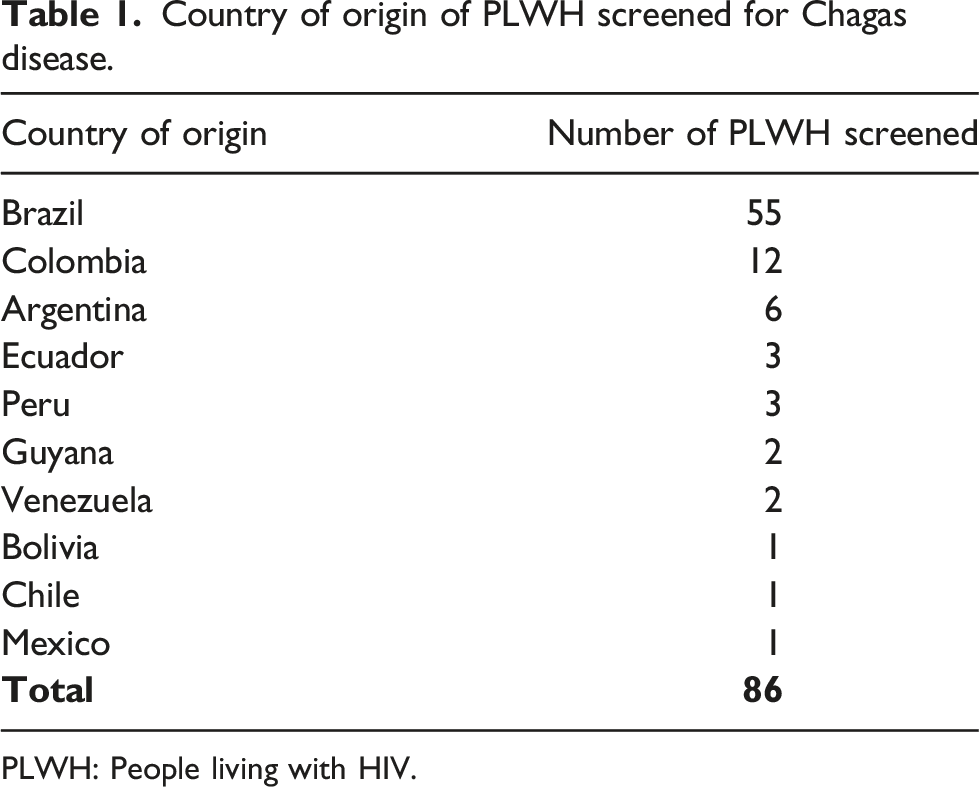
PLWH: People living with HIV.
Discussion
To our knowledge this is the first quality improvement project in a UK HIV clinic to systematically screen at-risk patients for T.cruzi. While there was no positive serology for T. cruzi among 86 patients screened, one person with T. cruzi infection was tested by his clinician based on better awareness due to the project rather than by the project screening itself. The lack of positive cases is due, at least in part, to the small number tested overall and in particularly the low number of patients from Bolivia. Of 60 patients at the Hospital for Tropical Diseases with serologically proven Chagas disease between 1995 and 2018, three quarters were originally from Bolivia. 5 In addition, with an expected prevalence around 2% for the region as a whole, chance may have contributed to there being no positives out of the 86 patients screened. Data from the USA, Spain and endemic countries (Argentina, Brazil, Bolivia), showed a prevalence of 0%, up to 10.5%3–5 and 32%,5,6 respectively, which may be higher due to different migration patterns.
Our electronic patient records made it easy to identify at-risk individuals. Our co-location with a specialist centre for tropical diseases facilitated referral and management. Screening provides an opportunity to disseminate information to at-risk individuals, their wider communities, and raise awareness of the condition among clinicians. HIV centres serving patient populations from endemic areas should consider routine screening at enrolment into care, particularly among individuals with advanced HIV disease in whom reactivation of Chagas disease is more likely. Earlier initiation of ARVs should also reduce the incidence of reactivation disease. Ultimately given the impact and consequences of Chagas as a neglected tropical disease, coupled with constant changing migration patterns due to natural, man-made and humanitarian crises, it is important for clinicians to be aware of, and screen for, conditions that could be prevented and managed, reducing the morbid and mortality as well as the impact on the healthcare.
Footnotes
Authors contributions
AG, SH, AAP, PB, SE conceptualised and designed the project; NA, HR and PC additionally supported implementation, data collection and analysis; all authors contributed to the writing and reviewing of the publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Patients who underwent screening had informed consent taking by staff doing the screening.
