Abstract
Background
For women living with HIV (WLHIV), co-infection with herpes simplex virus type 2 (HSV-2) causes severe genital ulcers and presents additional challenges for their HIV care. To inform preventive strategies, we aimed to determine the incidence and risk factors of HSV-2 positivity in a prospective cohort of South African women.
Methods
The CAPRISA 002 study enrolled women at acute HIV infection between 2004 and 2020. HSV-2 testing was conducted by multiplex polymerase chain reaction (PCR) assay on collected vaginal swabs up to twice annually during follow-up. We calculated incidence as the number of new cases per 100 person-years (PYs) and used Cox-proportional-hazard regression to identify factors associated with time-to-HSV-2 PCR positivity.
Results
At enrolment, the median age of 171 women was 24 years, interquartile range (IQR 21–28), and the estimated median days since HIV infection was 42 (IQR 22–65). Of participants tested at enrolment, HSV-2 antibody prevalence was 81.4% (105/129), and 10.6% (12/113) were positive by PCR. Among 147 women with a prior negative HSV-2 PCR diagnosis, we observed 47 new HSV-2 PCR positive cases over 424.4 PYs of follow-up, yielding an incidence rate of 11.1 cases per-100-PYs. HSV-2 PCR positivity incidence was higher among younger women (<25 years: adjusted Hazard Ratio [aHR] = 5.91, 95%CI 3.02–11.6), those with bacterial vaginosis (BV) (Nugent score 7–10: aHR = 2.17, 95%CI 1.15–4.10) and lower CD4 counts (<500 cells/μl: aHR = 2.04, 95%CI 1.08–3.87).
Conclusion
After acute HIV infection in women, the incidence of HSV-2 PCR positivity was associated with younger age, BV diagnosis and lower CD4 count.
Keywords
Introduction
Sexually transmitted infections (STIs) facilitate Human Immunodeficiency Virus (HIV) infection and transmission. 1 Herpes simplex virus (HSV) type 2 is among the leading risk factors for HIV infection mediated through genital ulcers or herpes disease. 2 HSV type 1 also causes genital herpes, but recurrent disease and subclinical viral shedding are more frequent with HSV-2, which affects more women than men.3,4 Comparing global estimates, the highest contribution of HSV-2 infection to population-level HIV incidence comes from the World Health Organisation (WHO) Africa Region, with an estimated population attributable fraction of 37.1% compared to 11.6 to 21.3% from the rest of the world. 5
Among women living with HIV (WLHIV), the risk of HSV-2 infection5,6 and HSV-2 shedding at the vaginal mucosa 7 are higher than in HIV-negative women. In South Africa, a global hotspot of the HIV epidemic, 8 two studies have estimated 13.7% prevalence of HSV-2 in men and women living with HIV 9 and 10.8% prevalence in WLHIV. 10 In WLHIV, HSV-2 coinfection with HIV causes increased morbidity through prolonged genital herpes recurrences, especially among patients with lower immune status. 3 This makes their HIV care more burdensome due to more clinic visits and treatments, often leading to poorer clinical outcomes. 11
Considering the chronicity of HSV-2 infection and the current non-availability of an effective cure, prevention is preferable, especially among WLHIV to reduce their healthcare burden and risk of HIV transmission, but no effective vaccine is currently available. 3 To strengthen the evidence on HSV-2 occurrence and inform public health prevention efforts, this study aimed to determine HSV-2 positivity incidence and the associated demographic, sexual-behavioural and clinical risk factors among women enrolled at acute HIV infection and followed up before, after, and during antiretroviral treatment (ART) in South Africa.
Methods
Study design, population, and setting
This retrospective cohort study used data from the Centre for the AIDS Programme of Research in South Africa (CAPRISA) acute HIV infection study (CAPRISA 002) conducted from 2004 to 2020 to investigate the disease progression of HIV-1 subtype C infection among South African adult women. 12 Participants were enrolled from other CAPRISA HIV prevention studies upon HIV seroconversion. 12 The eligibility criteria for the study included being 16 years or older, having a haemoglobin >9.0 g/dl and a negative HIV antibody test result within 5 months of HIV infection. The study confirmed HIV infection at enrolment with Enzyme Immunoassay (EIA) antibody testing, and HIV reverse transcription polymerase chain reaction (RT-PCR) testing was conducted when the antibody result was negative. The time of HIV infection was determined as the midpoint date between the last documented negative and the first positive HIV antibody test. For participants with a negative antibody test at enrolment, the estimated HIV infection date was 14 days before the positive RT-PCR test. Testing and management of HSV-2 infection and other STIs were conducted bi-annually before and annually for up to 5 years after ART initiation, with an additional 15 years of optional follow-up offered thereafter. Participants started ART based on the applicable South African guidelines at the time. HIV pre-and post-test counselling, risk reduction counselling, male and female condoms, access to clinical care, and treatment for STIs were provided to all participants throughout the study. The Biomedical Research Ethics Committee (BREC) of the University of KwaZulu-Natal approved the CAPRISA 002 study (E013/04) and this analysis (BREC/00003808/2022).
Primary outcome
The primary outcome was HSV-2 positivity incidence based on PCR testing. HSV-2 positivity incidence was defined as any new diagnosis after an initial negative test at enrolment.
Exposure variables
Exposure variables included demographic characteristics namely age and educational status, sexual behavioural characteristics namely age at first sexual intercourse, number of sexual partners and condom use and clinical characteristics including viral load, CD4 count, BV and STIs.
Laboratory measures
For HSV-2 PCR testing on vaginal swabs we used an in-house multiplex PCR assay at the National Institute for Communicable Diseases (NICD) laboratories in Johannesburg, South Africa. Blood specimens were collected and sent to Global laboratories in Durban for HSV-2 antibody testing based on HSV-2 glycoproteins G-2 with the HerpeSelect-2 EIA (Focus Technologies, Cypress, California, USA). The FACSCalibur flow cytometer (BD Biosciences, San Jose, California) was used to measure CD4+ T cell count. ROCHE COBAS Ampliprep-COBAS TaqMan version 2.0 and COBAS Amplicor version 1.5 RT-PCR and assays (Roche Diagnostics, Rotkreuz, Switzerland) were used to measure HIV viral loads. Urine specimens and vaginal swabs were collected and tested for Neisseria gonorrhoeae (NG), Chlamydia trachomatis (CT), Trichomonas vaginalis (TV) and Mycoplasma genitalium (MG), with either laboratory-based (in-house multiplex assay) or point-of-care (GeneXpert) assays. Gram-stained vaginal swabs were assessed with a light microscope by a certified laboratory technician using Nugent’s criteria for diagnosing BV (scores 7–10).
Data collection
Data capture for demographic information, physical examination, sexual behavioural risk assessments and clinical measures were conducted using structured case report forms (CRFs). Trained research assistants completed the CRFs at the study clinics and faxed them to the CAPRISA data management system using the iDataFax system (DF/Net Research, Seattle, USA). All data entry activities followed the CAPRISA data management Standard Operating Procedures for quality control.
Statistical analyses
We performed all statistical analyses and plotted graphs with R 4.2.1 (R Foundation for Statistical Computing, Vienna, Austria). 13 We summarized participant characteristics at baseline with proportions and medians stratified by HSV-2 PCR testing status (‘Negative’, ‘Positive’, ‘Not tested’) since not all participants were tested at enrolment. We compared the baseline differences in proportions and medians with the Chi-squared and Kruskal-Wallis tests.
All subjects who were HSV-2 PCR negative at their first time of testing, either at enrolment or subsequently for those not tested at enrolment, were included in the incidence analyses. We conducted a single-failure survival analysis to determine the incidence of HSV-2 PCR positivity measured as the number of new cases per 100 person-years (PYs) follow-up. The time at risk started from the first HSV-2 PCR negative to the first positive date or censoring. HSV-2 PCR was assumed to occur on the testing date. We then fitted Cox-proportional-hazard regression models using Breslow’s method for tied survival times to determine the risk factors associated with the time to HSV-2 PCR positivity adjusted for age, number of lifetime casual sex partners, condom use for contraception, co-occurrence of BV, CD4 count and HIV viral load status. We evaluated the multivariable Cox regression model for proportionality of hazard rates over time with the Schoenfeld's residuals test.
To provide a graphical representation of the probability of HSV-2 PCR positivity over time, based on the multivariable Cox model, we fitted adjusted Cox failure curves for each statistically significant variable. These were done by plotting the HSV-2 PCR failure probabilities and observation time obtained from separate fitted Cox models where each statistically significant variable was specified as the strata and adjusted for the other covariates. On the Y axis of the Cox failure curves, we plotted the adjusted probability of HSV-2 PCR positivity and plotted the person-time or years of follow-up after acute HIV infection on the X axis. We used the log-rank test to determine the equality of failure curves over time and a 5% significance level for all hypothesis tests.
Results
Of the women enrolled at acute HIV infection in the CAPRISA 002 study, 171 had at least one HSV-2 PCR test performed during follow-up between October 2004 and November 2020 and were included in this study. Among the 171 women, the median follow-up time was 9.2 years (interquartile range [IQR] 6.2–11.5) with a median of 6 (IQR 4–8) visits, where HSV-2 PCR testing was conducted. During follow-up, 136/171 women (79.5%, 95% Confidence Interval [CI] 72.7–85.3) started ART at a median of 4.8 (IQR 2.6–6.5) years after enrolment.
Characteristics of participants at enrolment
Characteristics of participants at enrolment stratified by HSV-2 PCR testing status.
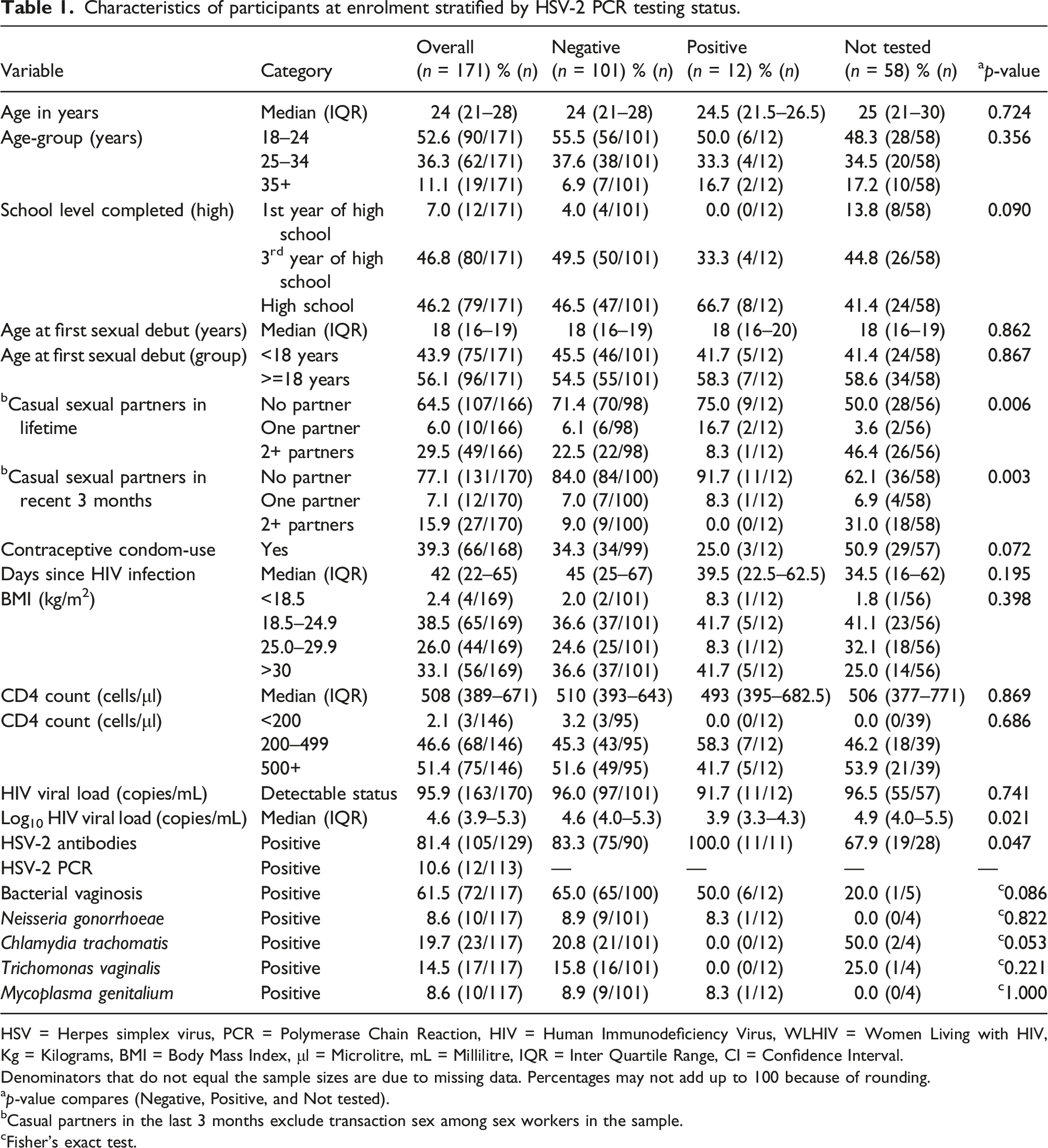
HSV = Herpes simplex virus, PCR = Polymerase Chain Reaction, HIV = Human Immunodeficiency Virus, WLHIV = Women Living with HIV, Kg = Kilograms, BMI = Body Mass Index, μl = Microlitre, mL = Millilitre, IQR = Inter Quartile Range, CI = Confidence Interval.
Denominators that do not equal the sample sizes are due to missing data. Percentages may not add up to 100 because of rounding.
ap-value compares (Negative, Positive, and Not tested).
bCasual partners in the last 3 months exclude transaction sex among sex workers in the sample.
cFisher’s exact test.
Incidence of HSV-2 PCR positivity after acute HIV infection among women
Of the 171 women tested during follow-up in the CAPRISA 002 study, 155 (90.6%) were HSV-2 PCR negative at their first testing episode. Of these 155 women, 8 were lost to follow-up, and the remaining 147 were included in the incidence analysis. Over a total of 424.4 PYs, 47 new HSV-2 PCR positive cases were observed, at a cumulative incidence of 32.0% (95% CI 24.5–40.2) and a crude incidence of 11.1 cases per 100 PYs (95% CI 8.3–14.6).
Factors associated with time to HSV-2 PCR positivity after acute HIV infection among women
Cox regression for factors associated with time to HSV-2 PCR positivity among WLHIV.
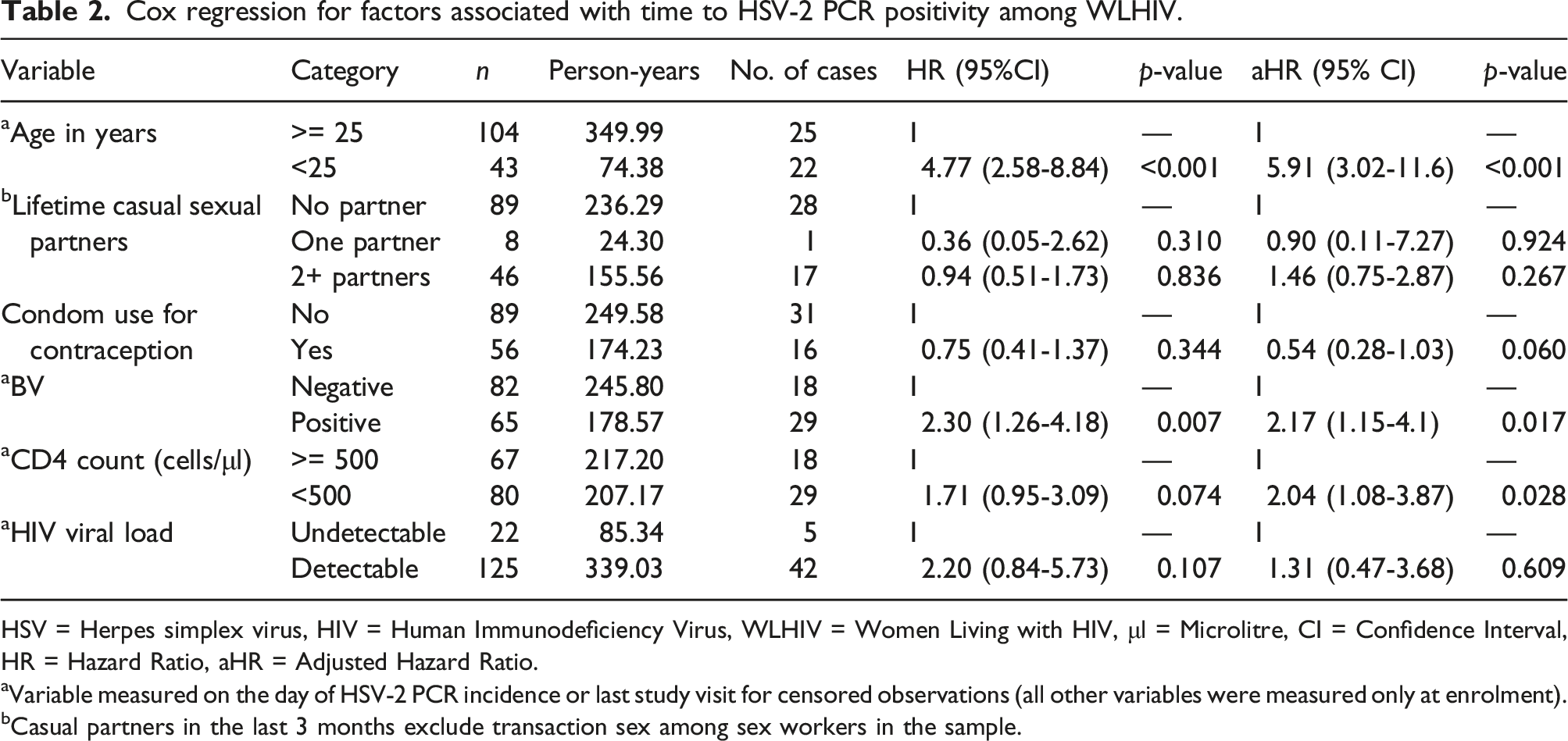
HSV = Herpes simplex virus, HIV = Human Immunodeficiency Virus, WLHIV = Women Living with HIV, μl = Microlitre, CI = Confidence Interval, HR = Hazard Ratio, aHR = Adjusted Hazard Ratio.
aVariable measured on the day of HSV-2 PCR incidence or last study visit for censored observations (all other variables were measured only at enrolment).
bCasual partners in the last 3 months exclude transaction sex among sex workers in the sample.
Cox failure curves showing the adjusted probability of HSV-2 PCR positivity over time
Figure 1 displays trends of the adjusted probabilities of HSV-2 PCR positivity by age group, BV and CD4 count in panels A, B and C. The graphs represent estimates from the multivariable Cox regression analysis, as explained in the Methods section. The Y axis of the graph shows the adjusted probability of HSV-2 PCR positivity, and the X axis shows the years of follow-up after acute HV infection. The curves show a significantly faster trend in HSV-2 PCR positivity by (A) younger age of < 25 versus >= 25 years, (B) BV positive versus negative status and (C) lower CD4 count of < 500 versus >= 500 cells/μl. The log-rank test p-values for all failure curves were less than 0.05. Adjusted Cox failure curves showing trends of the probability of HSV-2 PCR positivity by (a) Age-group, (b) Bacterial vaginosis status and (c) CD4 count (cells/microliter). All curves were adjusted for other variables in the multivariable Cox regression: Age-group, Lifetime and casual sexual partners, BV status, condom use for contraception, CD4 count and HIV viral load.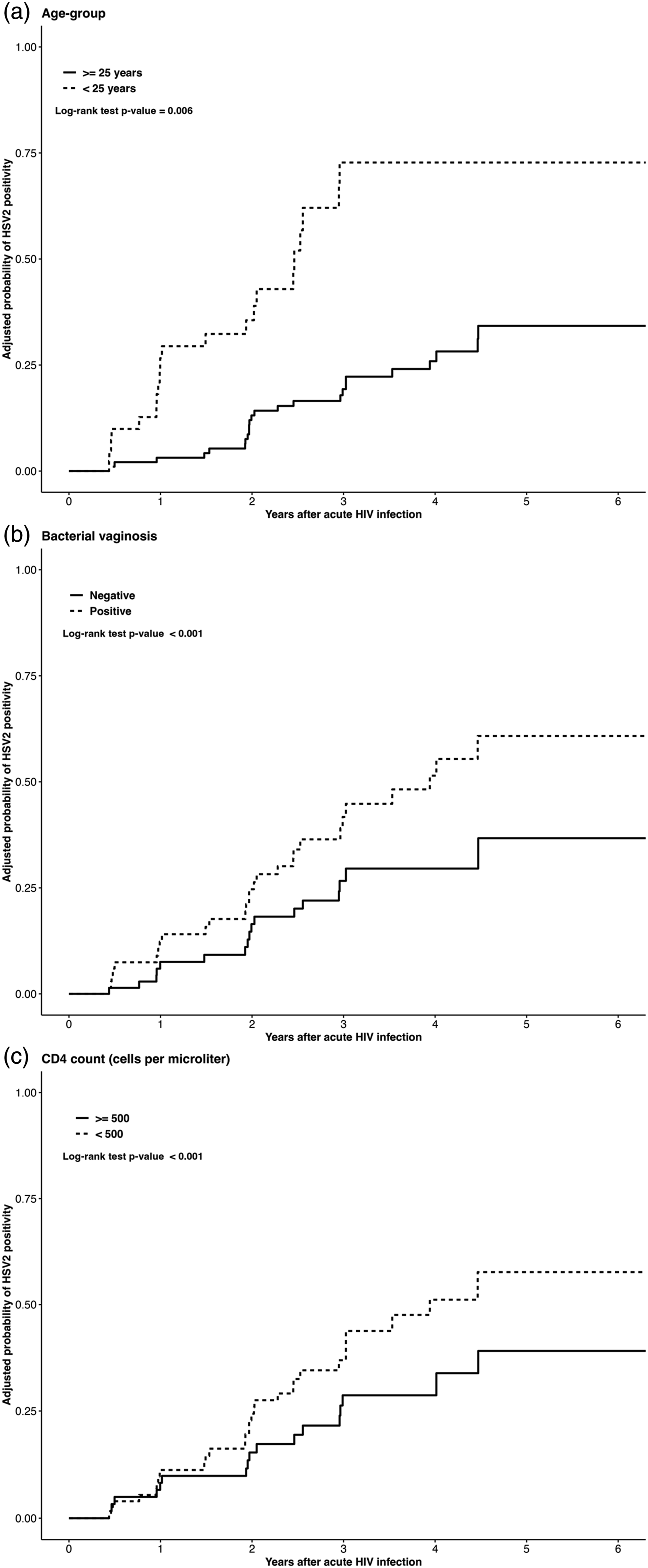
Multivariable Cox regression model goodness of fit: the proportional-hazards assumption
Figure 2 shows a graphical representation of results from the multivariable Cox regression model goodness of fit test. It displays plots of the scaled Schoenfeld’s residuals against observation time in years for each variable in the multivariable Cox regression. The figure also shows the p-values of the Schoenfeld test of the proportional hazard assumption for each covariate and the global p-value for the multivariable model. The Schoenfeld residual is the difference between each covariate’s observed and expected hazard ratios at the time of HSV-2 PCR occurrence. A stable trend of the residuals against time indicates the proportionality of the HSV-2 PCR hazard rates over time.
14
In each graph, the black dotted points represent the residuals versus time plot, the solid black lines represent the smoothing spline fit to the plot, and the black dashed lines represent the 95% confidence interval bands. All plots display a stable trend of the residuals over time, indicating that the model meets the proportional hazard assumption (all covariates p > .05 and global p = .903). Plots of the scaled Schoenfeld’s residuals against observation time for all covariates in the multivariable Cox regression model. The black dotted points represent the residuals versus time plot. The solid black lines represent the smoothing spline fit to the plot. The black dashed lines represent the 95% confidence interval bands.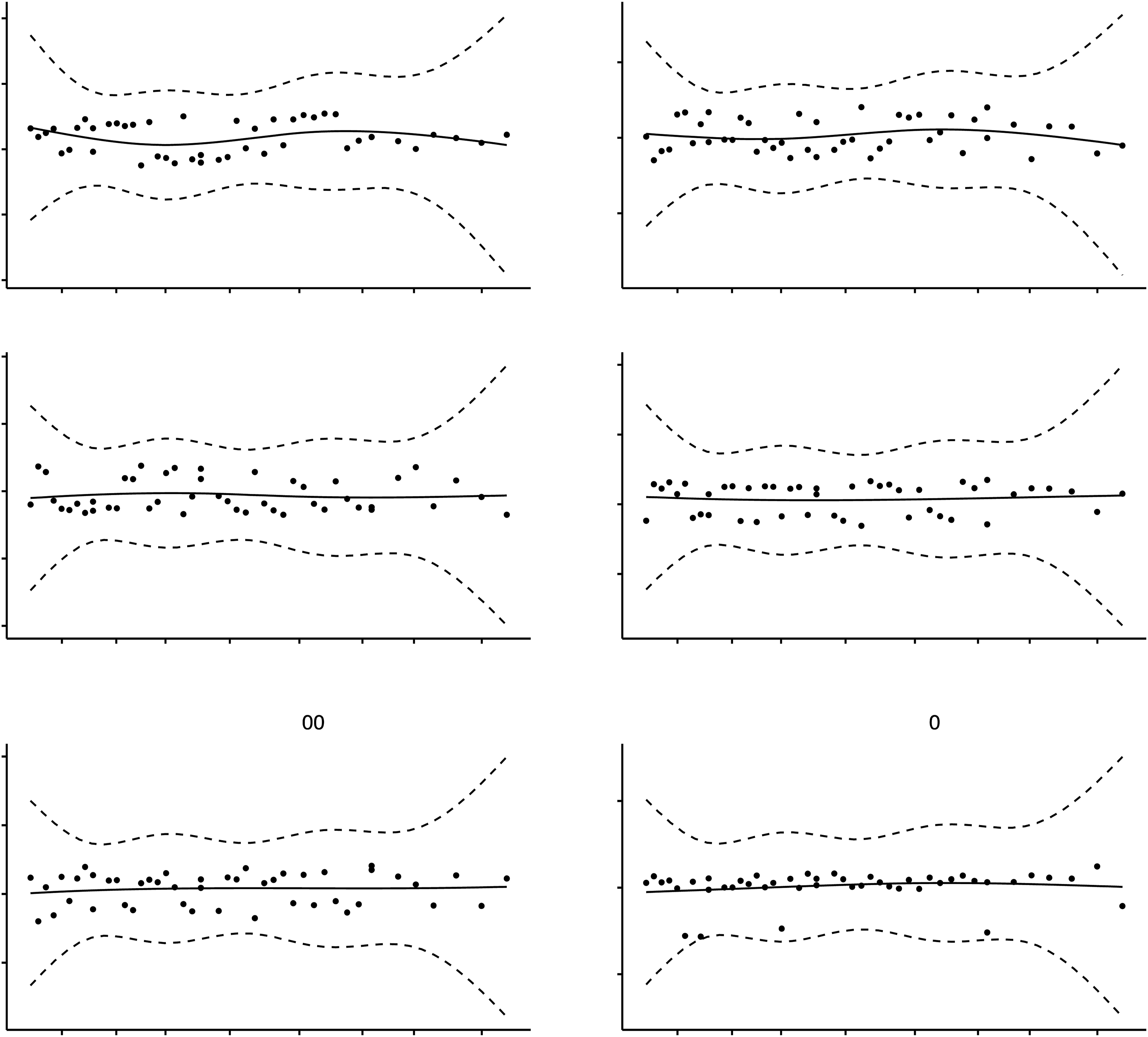
Discussion
This study investigated the incidence and risk factors associated with HSV-2 PCR positivity from acute to chronic HIV infection in a cohort of women from high-risk communities in South Africa. Although the crude incidence of HSV-2 PCR positivity was 11.1 cases per 100 PYs among women with an initial negative test, the incidence was higher among younger women, those with BV and lower CD4 counts after adjusting for other participant characteristics in the multivariable Cox regression analysis. Of note, the incidence analysis included only women with a prior negative HSV-2 PCR diagnosis, but of the 171 women included in this study, 81.4% (n = 105/129) of those tested at enrolment were HSV-2-antibody-positive indicating a prior HSV-2 infection. Thus, our HSV-2 PCR positivity outcome predominantly represents HSV-2 reactivation or viral shedding from existing disease with occasional instances of newly acquired infection. Studies have shown that HSV-2 viral shedding or re-activation in WLHIV is associated with increased HIV viral shedding15,16 which can further increase HIV infectiousness.2,5 Aligned with these studies, our findings emphasize the importance of reducing the risk of HSV-2 incidence, viral shedding, or re-activation in WLHIV to decrease population level HIV transmission.
In sub-Saharan Africa, a moderate number of studies17–19 have reported a high HSV-2 seroprevalence among WLHIV, with an estimated 93.3% prevalence reported in KwaZulu-Natal, South Africa 19 where this study was conducted. We found two studies in South Africa that performed diagnostic testing to determine active HSV-2 infection prevalence of 10.8% in WLHIV 10 and 13.7% 9 in PLHIV. However, few studies have determined the incidence and predictors of HSV-2 PCR positivity among PLHIV in sub-Saharan Africa. One study in Uganda enrolled 924 participants, including 45 (4.9%) PLHIV, to determine the seroprevalence and incidence of genital ulcer infections, including HSV-2. 20 Of the 45 PLHIV who were initially HSV-2 seronegative, 5 seroconverted based on serological testing during 15.0 person-years of observation at an incidence rate of 333.3 cases per 1000 PYs. 20 Among 343 young PLHIV included in the Reaching for Excellence in Adolescent Care and Health (REACH) study in the United States, HSV-2 incidence was 8.5 cases per 100 person-years based on antibody testing in participants who were seronegative at baseline. 21
We found an association between younger age and the incidence of HSV-2 PCR positivity, an indication of higher risk of HSV-2 infection or reactivation3,22 among this age group. This is consistent with established evidence of higher STI infection risk in younger women 23 as previously demonstrated in this same cohort of WLHIV for other STIs, including NG, CT, TV or MG. 24 Several reasons have been reported for the high STI burden in younger women including riskier sexual practices like multiple partners and inconsistent condom use,1,25 often exacerbated by gender and age-related power imbalances in sexual relationships. 26
HSV-2 PCR positivity incidence was also associated with BV diagnosis in our study. BV is known to be related to genital inflammation through the production of inflammatory cytokines 27 in the vaginal mucosa, which further increases the risk of new HSV-2 infections and HSV-2 viral shedding 28 in women with pre-existing subclinical disease. Moreover, women with HSV-2 infection usually have a higher occurrence of BV,29,30 as HSV-2 also causes genital inflammation, generally resulting in an increased presence of BV-causing bacteria. 31 Findings from these studies and ours, thus, confirm a bidirectional association between HSV-2 infection and BV occurrence in women, 32 an interaction increasing the risk for opportunistic infections and worsening reproductive and sexual health in women.
We found an association between lower CD4 count and the incidence of HSV-2 PCR positivity among WLHIV. Consistent with the relationship between immune status and other opportunistic infections, our finding emphasises the potential benefit of immune reconstitution through ART in reducing HSV-2 co-infection and related clinical consequences in WLHIV. Consistent with our finding, a cohort study conducted in Burkina Faso revealed that staying longer on ART was associated with reduced odds of HSV-2 viral shedding and genital ulcer disease, mainly in participants who achieved HIV viral suppression. 18 After introducing the universal ‘Test and Treat’ policy for HIV care in South Africa, a large-scale programmatic data analysis has shown better treatment outcomes with early ART initiation at CD4 count >500 cells/μ. 33 Our findings, thus, underscore the need to sustain such interventions with high treatment adherence for improved outcomes and prevention of opportunistic co-morbidities among PLHIV.
Our study had some limitations. Firstly, the sexual-behavioural characteristics, like condom use and number of sexual partners, were self-reported. Thus the related results could have been prone to social desirability 34 and recall biases. 35 Second, our study cohort included women recruited from high-risk communities for STI/HIV and may therefore not represent the general population of South Africa. For these reasons, we included variables on sexual partners in our multivariable regression models. Third, HSV-2 seroprevalence was high in this cohort at the time of enrolment. We were, therefore, unable to determine whether all new HSV-2 PCR positive cases were truly new infections or represented intermittent shedding of HSV-2.
In conclusion, our study has demonstrated a higher risk of HSV-2 infection or reactivation in younger WLHIV. It is important to promote safe sexual practices such as condom use through evidence-based behavioural interventions during chronic HIV infection with a special focus on younger women. The findings also support the importance of immune reconstitution with ART for improving not only HIV-treatment outcomes but also reducing HSV-2 co-infection or viral shedding and, as a result, reduce the risk of HIV transmission. Thus, it is necessary to improve HIV treatment continuation, retention-in-care and adherence through enhanced adherence counselling particularly in participants with poorer HIV treatment outcomes during ART. Our findings also demonstrate the role of genital inflammation through BV diagnosis for HSV-2 risk. This underscores the need for comprehensive STI/BV care, including HSV-2 testing and management as part of routine HIV care, to control STIs in general and reduce HSV-2 incidence and reactivation or shedding in WLHIV, specifically. Put together, adherence to these recommendations can improve the quality of life of WLHIV and reduce their HIV infectiousness which can result in lower population level HIV transmission.
Footnotes
Acknowledgements
We are grateful to all CAPRISA 002 study participants for participating in this research and the study team for conducting the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the CAPRISA 002 Acute HIV Infection Study was received from the Comprehensive International Program of Research on AIDS (CIPRA), funded by the National Institute of Allergy and Infectious Disease (NIAID), National Institutes of Health (NIH) and the US Department of Health and Human Services (DHHS) (Grant#1 U19 AI51794). KA was supported by HEARD scholarship with funding provided by Swedish International Development Cooperation (SIDA). The funders played no role in the study design, data collection and analysis, publication decision, or manuscript preparation.
Ethical statement
Contributors
KA, AT, and NG designed the study and are accountable for the overall content of the manuscript. KA conducted the statistical analysis and prepared the manuscript. All authors critically reviewed and approved the final submitted version of the manuscript.
