Abstract
Background
About 1.2 million people aged 13 years and older are living with HIV in the United States;1,2 among the newly diagnosed, approximately one in four are adolescents and emerging adults (youth) aged 13 to 24.3,4 Youth with HIV (YWH) are less likely than their older peers to successfully navigate the HIV care continuum.5,6 Half of YWH are unaware of their HIV status; 7 among those who know about their HIV diagnosis, half lack consistent access to treatment and antiretroviral therapy (ART), a precursor to achieving and maintaining viral suppression.7–9 YWH face numerous barriers to obtaining viral suppression; 55% of YWH are not retained in care, and 22% of those prescribed ART do not achieve viral suppression.10,11 Among people with HIV (PWH), Black populations and youth who identify as sexual or gender minorities have lower rates of care engagement, ART adherence, and viral suppression.9,12,13 Suboptimal rates of viral suppression have been attributed to ART non-adherence related to insufficient or inconsistent access to HIV care, inability to follow daily pill regimens, and competing health demands related to co-morbid health conditions such as mental illness and sexually transmitted infections (STI).14–16 Thus, engaging YWH, particularly minoritized YWH, in supportive care is a high priority in ending the epidemic.
In the United States, barriers to health and well-being are exacerbated among those who identify both as racial and sexual and gender minorities.17,18 Black and sexual and gender minority youth (e.g., men who have sex with men or MSM, transgender women, and other sexual minorities) are more likely to experience structural racism and discrimination, live in unsafe neighborhoods, receive lower quality of care, and internalize intersectional stigma related to their race, gender, orientation, and HIV status. 19 All of the aforementioned have been associated with worse health outcomes across the HIV continuum of care and affect other aspects of health and wellbeing.9,15,20–22
Further, YWH in the southern United States are located in a region with the highest rates of STIs, including chlamydia and gonorrhea. 23 Youth aged 15–24 account for over half of new STI diagnoses in the United States.16,24,25 One study found that 67% of YWH had at least one STI during a three-year study period, with 14% of YWH having two or more STIs. 16 Co-occurring STIs are associated with elevated risk of HIV transmission; specifically, having an STI can increase viral shedding and higher concentration of HIV in the genital tract resulting in higher likelihood of transmitting HIV to sexual partners. 26 Southern YWH often struggle with obtaining and maintaining viral suppression due to personal and structural barriers to care, while also being highly susceptible to STI acquisition.10,11,16,27
Considering the urgency of addressing the epidemic among YWH, especially YWH in the southern United States where health disparities persist in HIV, STI, and related outcomes, 28 the objective of this exploratory study is two-fold: (1) Examine rates of viral suppression among YWH receiving care from a clinic in the southern United States, and (2) Examine the relationship between STIs (testing and positive diagnosis) and viral suppression among a sample of YWH. This study is novel in its inclusion of YWH ages 10 to 24 years, given that YWH have unique care needs, may require changes in medications over time to stay suppressed, and are navigating their transition to adulthood while managing HIV as a chronic condition.28–30
Methods
Data and participants
Our sample includes YWH aged 10–24 years from a Health Resources and Services Administration (HRSA)-funded Ryan White Part B and D clinic providing services to women, infants, children, adolescents, and young adults with HIV in Alabama (N = 139). Data were extracted manually from multiple electronic health records systems. Patient profiles were aggregated from 933 clinic visits that occurred from 2017 to 2020, with the index visit being defined as the first clinical record within this time. The index visit may represent the point at which the youth was established as a patient or the first record of an established patient during our time period. Missing data were not extrapolated or imputed.
Sociodemographic measures
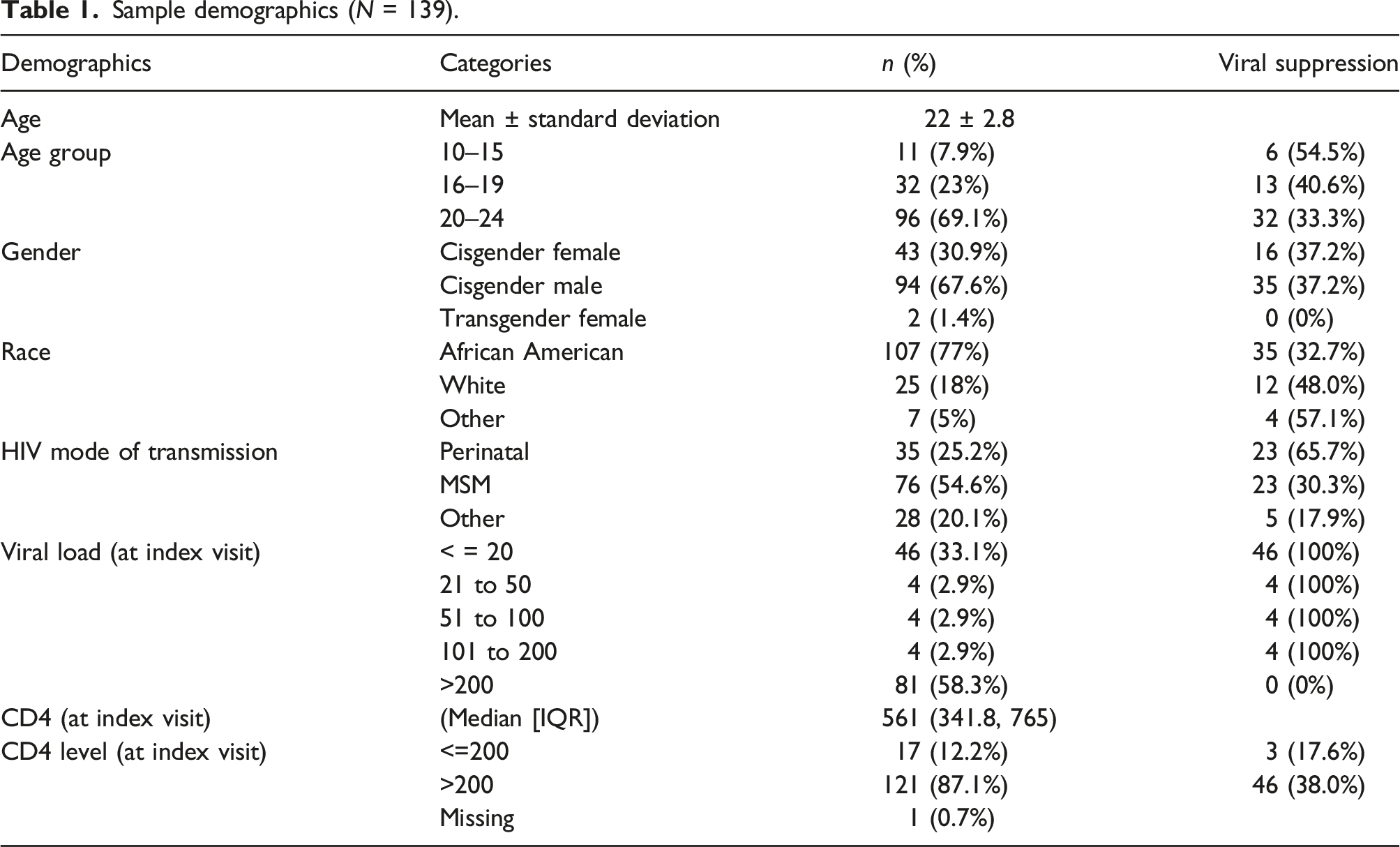
Age, gender, and race were identified from the index visit. Age was included in our model as a continuous variable ranging from 10 to 24 years. Gender, rather than sex assigned at birth, was categorized as male, female, and transgender female. No records indicated gender of transgender male. Race was coded as Black and Other. White and Other race (Asian) were combined as one group due to the small sample size. We did not include ethnicity in our analyses due to small sample size (Hispanic, n = 3).
HIV measures
Route of HIV transmission was divided into three unique groups: vertical transmission that occurred during pregnancy, delivery, or breastfeeding (coded = Perinatal), transmission through sexual intercourse between men who have sex with men (coded = MSM), and other types of transmission, including but not limited to intravenous drug use or heterosexual contact (coded = Other). HIV viral load is the quantity of virus in each volume of fluid or blood and indicates if a PWH is virally suppressed or unsuppressed; being suppressed indicates that the patient cannot transmit HIV. Viral load was classified into five categories: less than or equal to 20, 21–50, 51–100, 101–200, and 200+. 31 Viral suppression was defined as having less than 200 copies of HIV per milliliter (ml) of blood. 32 CD4 cell count is an indicator of immune function among PWH and is therefore also important to patient health. 33 CD4 count was categorized into two groups: < = 200 cells/mL or >200 cells/mL.
Measures of STI testing
Gonorrhea and chlamydia testing were co-located within the clinic; testing for syphilis required YWH to visit a separate laboratory within the same building as clinic. We included multiple STI testing measures as indicated by ICD-10 codes from clinical records. We created an indicator of STI testing using the patient’s health records at each visit. For records that included an STI test, we created a series of measures. Each YWH who had received STI testing was assigned separate indicators for chlamydia, gonorrhea, and syphilis; a reactive or positive test was coded as one under each of these measures. A non-reactive or negative test was coded as 0. Each STI test in the visit record was considered individually even if there were multiple tests administered for a given STI. We then created an aggregated STI measure for having received any STI testing and if any test result was reactive or positive. Herpes and HPV tests were excluded due to insufficient sample size. All visit records during the three-year period were used in the analysis, including the index visit.
Statistical analyses
Generalized linear mixed models with random intercepts to account for repeated measures within patients were employed to estimate unadjusted odds ratios (ORs), 95% confidence intervals (CIs) and p-values. We employed a stepwise method (backward) to generate the parsimonious model. We considered adjusted models containing demographic information, route of HIV transmission, and one of the following: STI results (positive test result, negative test result, and no test result for each of the three STIs), STI testing (tested or not tested for each STI), and any STI test status (tested for any STI or no test). We used backward selection to determine which variable remained in each candidate model. We selected the final parsimonious model by comparing the model fit of each candidate model using Pseudo-Likelihood and selecting the best-fitted model. Adjusted ORs (AOR), 95% CIs and p-values are reported. All analyses were performed with SAS (Version 9.4, Cary, North Carolina, USA), with PROC GLIMMIX procedure approximating the marginal likelihood by using Laplace’s method. The empirical (“sandwich”) estimators were applied as the computational option to increase robustness.
Results
Sample characteristics
Sample demographics (N = 139).
Unadjusted correlates of viral suppression (viral load <200 copies/ml).
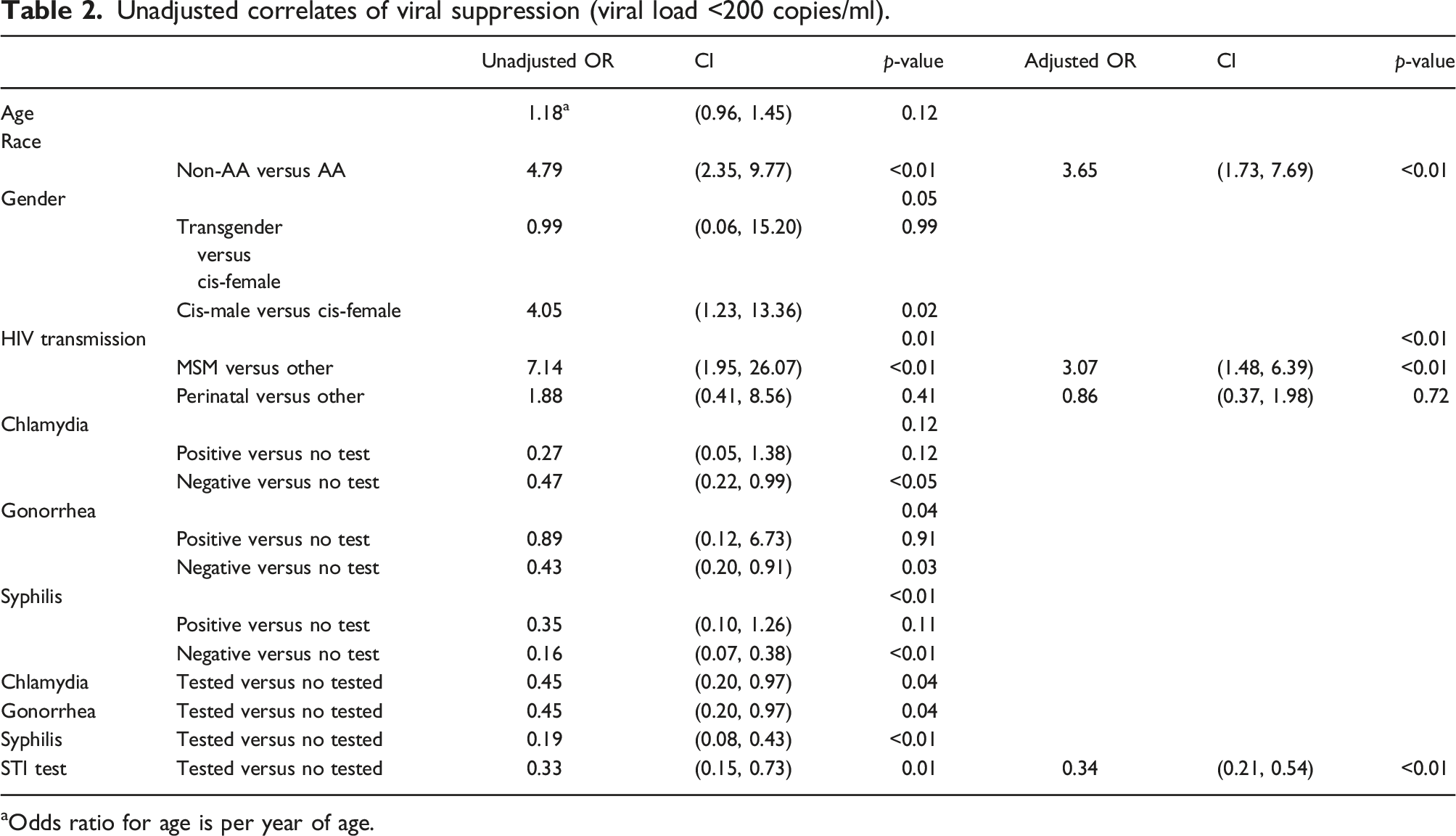
aOdds ratio for age is per year of age.
Associates of HIV viral suppression
YWH who identified as White or of other races had 4.79 higher odds of being viral suppressed as compared to Black YWH (p < .01, OR = 4.79, 95% CI: 2.35–9.77). MSM had 7.14 higher odds of being viral suppressed compared to their peers who acquired HIV via other modes of transmission (p < .01, OR = 7.14, 95% CI: 1.95–26.07).
HIV viral suppression and STI testing
When examining the effects of STI testing via a binary equation (test or no test), testing of all three STIs were associated with lack of viral suppression (p < .05 for chlamydia, p < .05 for gonorrhea, and <0.01 for syphilis). Specifically, YWH who received STI testing had lower odds of viral suppression compared to peers who did not require testing or were not offered testing for chlamydia (OR = 0.45, 95% CI: 0.20–0.97), gonorrhea (OR = 0.45, 95% CI: 0.20–0.97), and syphilis (OR = 0.19, 95% CI: 0.08–0.43). We also examined the effects of STI testing through a singular universal test or no test. The result of this method was identical to the second method. We found that YWH with any STI test in their medical records had 33% lower odds of being virally suppressed compared to YWH without record of receiving STI testing (p = .01, OR = 0.33, 95% CI: 0.15–0.73).
Adjusted model
Unadjusted models indicated that chlamydia, gonorrhea and syphilis testing were all associated with lack of HIV viral suppression, specifically those who received these STI tests had lower odds of being HIV virally suppressed. After performing candidate adjusted models, the model containing race, mode of HIV transmission, and indictor of any STI test was selected. In this parsimonious adjusted model, White and Other race YWH had significantly higher odds of viral suppression (AOR = 3.65, 95% CI = 1.73–7.69) compared to Black YWH. MSM YWH had 3.07 higher odds of being viral suppressed compared to their peers who acquired HIV via other modes of transmission. YWH who received STI testing had lower odds of viral suppression (AOR = 0.34, 95% CI = 0.21–0.54) compared to those who were not tested for STIs.
Conclusions
The objectives of this exploratory study were to (1) Examine rates of viral suppression among YWH receiving care from a clinic in the southern United States, and (2) Explore the relationship between STI testing and viral suppression among our sample of YWH in the southern United States. We found multiple factors with significant associations with viral suppression. Race (p < .01) and mode of HIV transmission (p=.01) were both statistically significant in unadjusted models, with Black YWH experiencing notable inequities.8,15,34 Considering that our sample was nearly 80% Black and were between the ages of 20 to 24 years, future research using similar data could explore outcomes among YWH who identify as sexual and gender minorities, focusing on the role of intersectionality in these patients’ care experiences and outcomes given the unique context of the southern United States.
Irrespective of test result, we found that having received an STI test was associated with lower likelihood (odds) of viral suppression among clients. YWH receiving STI testing were less likely to be suppressed. Multiple explanations exist for this finding. In the case of non-testers, these youths' sexual behaviors may not warrant STI testing (e.g., non-sexually active). For those who received testing, it is possible that testing was recommended due to the presence of STI symptoms, a known determinant of HIV-related complications given that viral non-suppression rates were also high in this population.34–36 More HIV-discordant couples are having conversations about condom use and adherence to ARTs prior to engaging in sex, which may allow for more appropriate STI risk assessments and condom negotiation. 37 Therefore, it is possible that these results may reflect the evolution of sexual behavior among YWH who are virally suppressed and have applied their health knowledge to STI prevention.
We did not find evidence of age-related correlates of viral suppression in our sample. This may have been due to a smaller sample size. A recent study found that children who received intensive adherence counseling did not achieve increased viral suppression, although this is a World Health Organization recommended strategy for promoting viral suppression among perinatal cases. 38
Limitations
Viral suppression is not a direct measure of ART adherence. Analyses performed were cross-sectional, which prevents the ascertainment of causal direction of associations among the variables of interest, and potential temporal fluctuations. Our small sample size may have led to a loss of statistical power to detect significant findings and could have inflated CIs. We employed a generalized linear mixed model (GLMM), thereby ensuring every client visit would contribute to the model's fit. Although the 933 visit records bolsters our study’s power, the sample size of 139 clients is small and limits generalizability. In this study, the index visit does not necessarily correspond to the patient's initial visit (establishment in care date). Recognizing the varying visit patterns between new patients and those who have been in care for years, we conducted sub-group analyses for those who commenced care 6-months prior to the study period. Model fitting complications arose due to the reduced sample size (n = 73), leading to a loss of statistical power. Due to limitations of our electronic health systems, “male” and “men” (e.g. gender and sex) were considered equivalent. Body site of chlamydia and gonorrhea testing (pharyngeal, rectal, or genital) was not available in our data.
Conclusions
Despite limitations, results support a need for further exploration of social and structural factors affecting STI testing among YWH in the southern United States. Among young clients, attention should be given to the dynamics of their personal relationships and STI knowledge. Additionally, with high rates of STI positive test results (0.13 per person-year in this study), STI counseling in pediatric HIV care settings may be warranted. More research is needed to understand YWHs’ experiences with receipt of care and long-term determinants of viral suppression, such as considering STI co-morbidities. Moreover, our preliminary results support the need for multi-component interventions to concurrently improve viral suppression rates and STI testing in YWH, to promote a higher level of health and wellbeing.
Footnotes
Author contributions
HB is the lead author and conceptualized the study with the input of other authors. TS is the senior author. DML and JH conducted the statistical analyses. SH and ACMB contributed to the framing and study orientation. All authors contributed to the writing and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institute of Mental Health (NIMH) of the National Institutes of Health (NIH) under Award Number K01MH116737. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Data availability statement
All available materials are included in this manuscript.
