Abstract
Biological therapies represent the gold-standard treatment of severe forms of plaque psoriasis. However, people living with HIV are often under-treated for psoriasis because very limited data are available on the use of biologics in this population. We report four cases of patients affected by HIV and moderate-to-severe plaque psoriasis, all treated with risankizumab, a monoclonal antibody that selectively targets interleukin-23. After 16 weeks, all patients experienced complete or almost complete skin clearance without any adverse events. Data on the effectiveness and safety of biological therapies in people living with HIV are limited to case reports or small case series, especially for the most recently approved inhibitors of interleukin-23. Our experienced, although limited, supports the role of risankizumab as a safe and effective therapy for psoriasis amongst patients living with HIV.
Introduction
Psoriasis is one of the most common inflammatory skin diseases affecting approximately up to 3% worldwide. 1 It has been reported that the psoriasis prevalence in people living with HIV (PLHIV) is comparable with the general population. 2 In PLHIV, psoriasis can present with several and severe manifestations. Moreover, the relationship between the severity of chronic HIV and psoriasis extension has been widely demonstrated. 2 The treatment of psoriasis in PLHIV is challenging because conventional systemic immune-suppressive drugs cannot be used, and the prescription of biological therapies should also be cautious. 3
Interleukin(IL)-23 inhibitors represent the most recently approved class of biologics for plaque psoriasis. 4 They showed excellent efficacy and safety profile in clinical trials; however, data on PLHIV are extremely limited. 5
We report four male patients with concomitant psoriasis and HIV infection who were successfully treated with risankizumab, an anti-IL-23 monoclonal antibody.
Cases presentation
The first patient received a diagnosis of HIV at the age of 45 years and started antiretroviral therapy (ART) with rilpivirine/tenofovir alafenamide/emtricitabine in 2011. He had suffered from plaque psoriasis since the age of 25 years. Given the severity of his psoriasis (with a Psoriasis Area and Severity Index (PASI) of 22) and the impact on his quality of life (with a Dermatology Life Quality Index (DLQI) of 17), and as successful viro-immunological control was maintained for years, we prescribed risankizumab 150 mg at weeks 0, 4 and then every 12 weeks. As a result, complete skin clearance was achieved after 16 weeks (Figure 1), and the patient is still on treatment without any adverse events (AEs). 53-year-old man with erythematous and scaly plaques involving the trunk and four limbs at baseline (a, c, e) and after 16 weeks of treatment (b, d, f).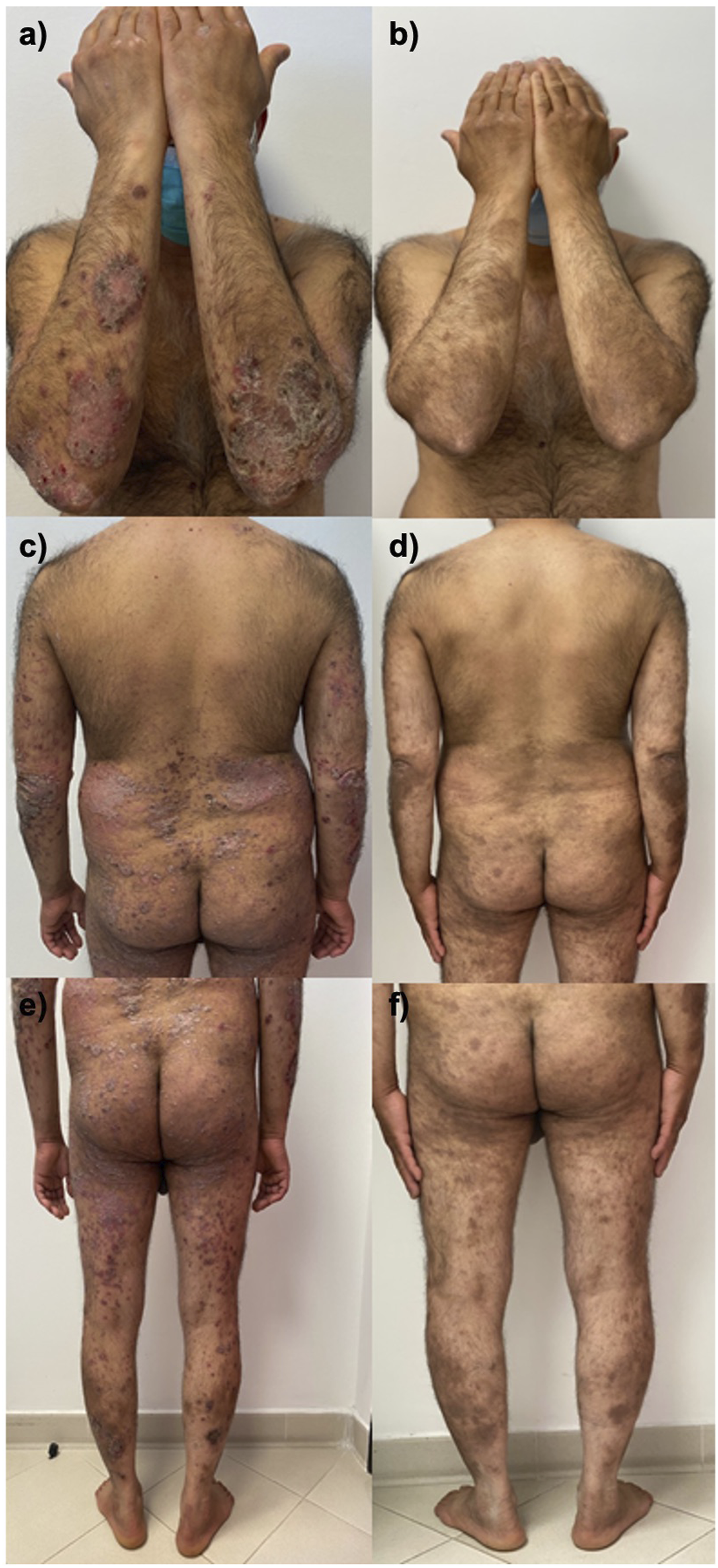
The second patient is a 32-year-old man who received both diagnoses of HIV and psoriasis in 2020. He was prescribed ART with doravirine/tenofovir disoproxil fumarate/emtricitabine soon after, gaining complete immunological control. However, a year later, he showed a worsening of his plaque psoriasis, which involved the trunk and four limbs (PASI 30). As a result, we decided to start treatment with risankizumab, which showed a successful response after 16 weeks (PASI 0).
The third patient is a 58-year-old man who presented to our dermatology unit with severe plaque psoriasis (PASI 20). He referred a history of AIDS with Pneumocystis jirovecii pneumonia in 1999 and Kaposi sarcoma in 2000. Ha was on treatment with rilpivirine/emtricitabine/tenofovir alafenamide with successful control of HIV (CD4+ lymphocytes 439 cells/mm3) and undetectable viral load. We prescribed risankizumab, achieving a PASI of 1 after 16 weeks.
The last patient is a 52-year-old man with a diagnosis of HIV in 1996, successfully controlled with lamivudine/dolutegravir. He referred the onset of psoriasis in 2018 after a surgical amputation of his left leg following bacterial osteomyelitis.
Due to the severity of his psoriasis (PASI 10) and the undetectable viral load, we decided to start risankizumab. After 16 weeks, the patient showed complete skin clearance.
In all cases, the treatment with risankizumab was well tolerated, and no AEs or viral reactivation were observed, with two patients completing already two years of follow-up.
Demographic characteristics and characteristics of HIV and psoriasis in all our four patients.
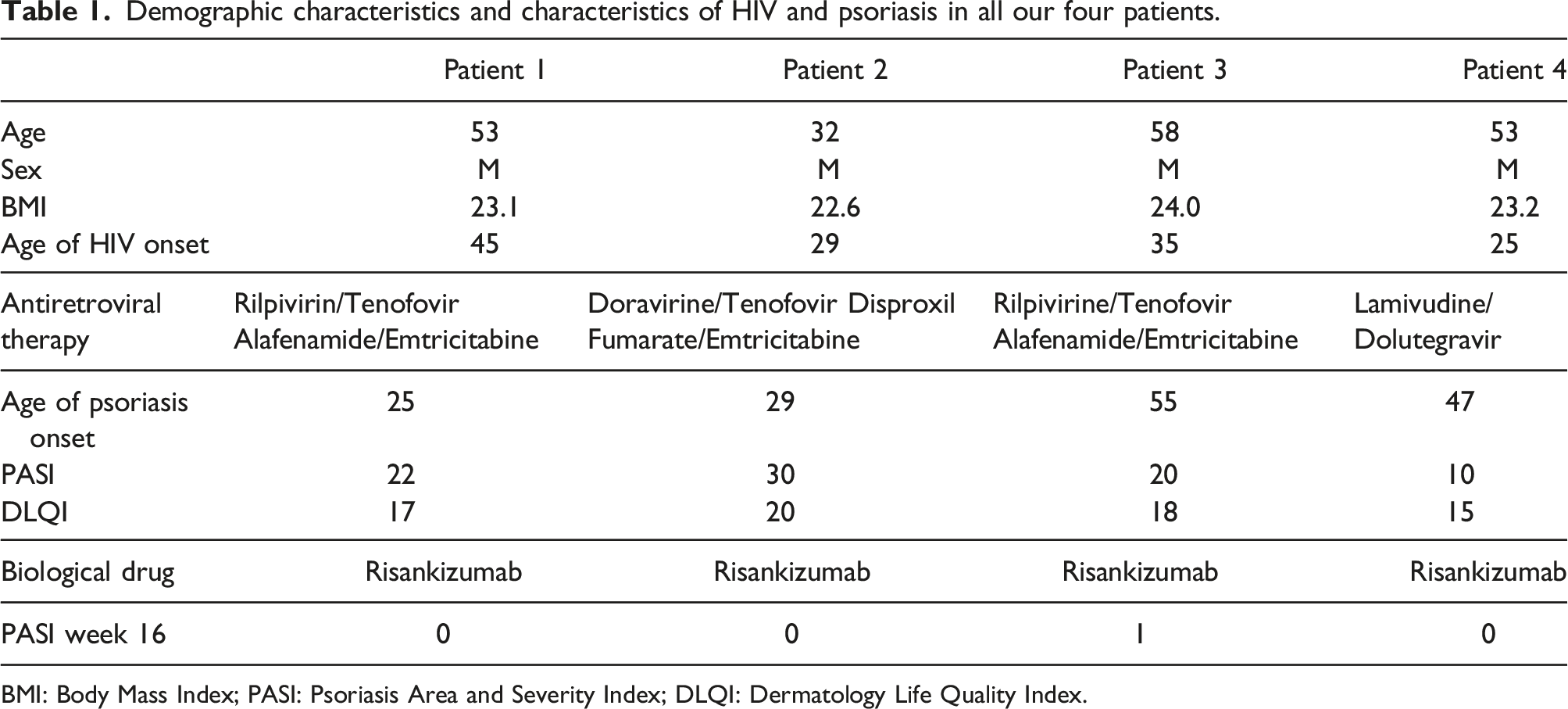
BMI: Body Mass Index; PASI: Psoriasis Area and Severity Index; DLQI: Dermatology Life Quality Index.
Discussion
In the context of HIV infection, the onset or worsening of psoriasis could reflect the direct impact of viral infection on cytokine balance and the cell-mediated inflammatory processes associated with the immune regeneration following antiretroviral therapy. 6 A topical or UVB-based approach is usually not an adequate option to achieve remission in cases characterized by high severity. Therefore, systemic treatment becomes mandatory to restore proper quality of life and to avoid potential musculoskeletal and cardiovascular involvement.
Limited data are available in the literature on the treatment of psoriasis in PLHIV, mostly regarding inhibitors of TNF-alfa.7,8 The inhibitors of the IL-23/17 axis have shown promising results in patients with chronic viral infections. 9 A few case reports have demonstrated that anti-IL-17 are a safe option for treating psoriasis and psoriatic arthritis in PLHIV.10,11 Real-life studies on IL-23 inhibitors have also included patients with concomitant chronic infections, including HIV, showing no adverse events throughout the treatment period.12–14 Additionally, two case reports of patients treated with IL-23 inhibitors during chronic HIV infection have been reported in the literature.15,16 In a recent meta-analysis, risankizumab was found to have high efficacy and low risk compared with other biologics currently available for psoriasis management, even in a long-term evaluation. 17
Conclusion
There is an urgent need to find safe and effective treatments for PLHIV with moderate-to-severe plaque psoriasis. In our experience, we choose to prescribe risankizumab because of its safety profile, high effectiveness, and the availability of data on interleukin inhibitors in the management of patients with psoriasis and chronic infection. However, more extensive prospective studies are strongly needed to support our findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L. Gargiulo has been a consultant for Almirall. A. Costanzo has served as an advisory board member, consultant and has received fees and speaker’s honoraria or has participated in clinical trials for Abbvie, Almirall, Biogen, LEO Pharma, Lilly, Janssen, Novartis, Pfizer, Sanofi Genzyme, and UCB-Pharma. A. Narcisi has served on advisory boards, received honoraria for lectures and research grants from Almirall, Abbvie, Leo Pharma, Celgene, Eli Lilly, Janssen, Novartis, Sanofi‐Genzyme, Amgen and Boehringer Ingelheim. The other authors have nothing to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
