Abstract
Introduction
People with HIV (PLWH) have an increased risk myocardial infarction (MI), and evidence suggests that MI is under-diagnosed in Tanzania. However, little is known about barriers to MI care among PLWH in the region.
Methods
In this qualitative study grounded in phenomenology, semi-structured interviews were conducted in northern Tanzania. Purposive sampling was used to recruit a diverse group of providers who care for PLWH and patients with HIV and electrocardiographic evidence of prior MI. Emergent themes were identified via inductive thematic analysis.
Results
24 physician and patient participants were interviewed. Most participants explained MI as caused by emotional shock and were unaware of the association between HIV and increased MI risk. Providers described poor provider training regarding MI, high out-of-pocket costs, and lack of diagnostic equipment and medications. Patients reported little engagement with and limited knowledge of cardiovascular care, despite high engagement with HIV care. Most provider and patient participants indicated that they would prefer to integrate cardiovascular care with routine HIV care.
Conclusions
PLWH face many barriers to MI care in Tanzania. There is a need for multifaceted interventions to educate providers and patients, improve access to MI diagnosis, and increase engagement with cardiovascular care among this population.
Introduction
In people living with HIV (PLWH), cardiovascular diseases (CVD) such as myocardial infarction (MI) are a leading cause of mortality.1,2 PLWH are at increased risk for CVD for a variety of reasons, including chronic inflammation from HIV infection and antiretroviral therapy exposure.2–4 Moreover, PLWH are living longer due to advances in HIV care in sub-Saharan Africa (SSA), 5 and are facing increased age-related cardiovascular complications. 6 Simultaneously, the prevalence of cardiovascular risk factors like hypertension and obesity is rapidly growing among this population.7–9 Given these trends, MI poses a serious threat in PLWH in SSA.
MI care in some parts of SSA is complicated by resource limitations. 10 Recent studies among the general population from Tanzania, Kenya, and South Africa have demonstrated that MI is frequently misdiagnosed and not treated according to evidence-based guidelines due to inadequate provider training, lack of awareness among patients, and a lack of diagnostic equipment.11–14 Despite an elevated risk of MI in PLWH, few studies probe specific barriers to MI care among this population in SSA.
There is a pressing need to understand barriers to MI care among PLWH in SSA for several reasons. First, PLWH likely face a unique set of barriers, such as stigma and lack of education about the link between HIV and CVD.13,15–17 Second, growing evidence suggests that the burden of MI among PLWH in the region is substantially greater than among the general population: a recent study in Tanzania found that ECG findings suggestive of prior MI were significantly more common among PLWH than Tanzanians without HIV. 18 Furthermore, since PLWH in the region have access to free HIV clinics and are familiar with chronic disease management and daily HIV medication adherence, improvements to chronic CVD could be built on existing HIV care pathways.
Further understanding of barriers and facilitators to MI care is needed to inform the development of interventions to improve cardiovascular care in this high-risk patient population, particularly as policy-makers increasingly advocate for integration of HIV and cardiovascular care in the region. 19 Thus, the aim of this qualitative study is to examine provider and patient perspectives on barriers to MI care among PLWH in Tanzania.
Methods
Setting
This study was conducted in northern Tanzania from March to September 2021. We recruited providers caring for MI in PLWH from healthcare facilities where patients with HIV receive care. Participants with HIV were recruited from those seeking routine outpatient HIV care at Majengo Care and Treatment Center (MCTC). MCTC is a government-funded clinic that provides routine care for about 1200 PLWH in Moshi, Tanzania. MCTC provides free HIV care including provision of anti-retroviral medications, blood testing for viral load and CD4 count, and HIV-related counseling, but MCTC does not provide cardiovascular care such as treatment of hypertension or electrocardiography.
Sampling
Purposive sampling was used to recruit various types of providers (physicians, clinical officers, nurses, and social workers) of different ages, genders, and educational backgrounds from various healthcare facilities in northern Tanzania that provide care to PLWH, including HIV clinics and general hospitals. Patients were enrolled from an ongoing prospective observational study at MCTC in which participants underwent electrocardiogram (ECG) screening. Patients were eligible for in-depth interviews if they self-reported a history of MI or if they had an ECG demonstrating evidence of prior MI, as per Fourth Universal definition of MI criteria. 20 Purposive sampling was also used to recruit patients with a diverse range of ages, sexes, and education levels. Interim analyses were conducted concurrently with data collection; interviews were conducted until thematic saturation was reached.
Procedures
Participants were approached by Tanzanian research assistants with prior experience conducting qualitative research. 12 After providing written informed consent, participants engaged in one-on-one interviews. The interviews followed semi-structured interview guides (Supplementary Material) developed by an interdisciplinary team of researchers and physicians from both the United States and Tanzania, informed by the Health Belief Model to provide a framework for the interactions between beliefs and health-related behaviors. 21 All interviews were conducted face-to-face in Swahili by the Tanzanian research assistants, who had been trained with the interview guide via didactics and piloting. The interviews were audiotaped, translated, and transcribed into English by a local translator with quality checks provided by an author (JTH) fluent in both languages.
Data analysis
Both the provider and patient interview guide elicited responses from participants regarding their perceptions and knowledge of MI, current patterns of MI diagnosis and treatment, as well as perspectives on barriers to care. Although the Health Belief Model was used to inform the development of the semi-structured interview guide, a purely inductive approach was used in thematic analysis. Inductive thematic analysis was conducted to identify emergent themes and a codebook was developed by an interdisciplinary team, including an American physician (JTH), two Tanzanian clinical officers (JJM and GLK), a Tanzanian social scientist (TGT), and a qualitative expert (SP). 22 All transcripts were independently double-coded in NVivo 12, and the codebook was further iteratively refined through the coding process through discussion. Discrepancies in coding were resolved through discussion and consensus-building among the coding team. In nearly all cases, unanimous consensus was reached; in rare cases where unanimous consensus was not achieved, a simple majority was used for final adjudication. The final codebook consisted of 67 overall codes (Supplementary Material) organized into eight themes across 3 overarching domains.
Ethics
This study was approved by institutional review boards at the Tanzania National Institute of Medical Research (NIMR/HQ/R.8a/Vol. IX/3437), Kilimanjaro Christian Medical Centre (Proposal 893), and Duke Health (Pro00090902).
Results
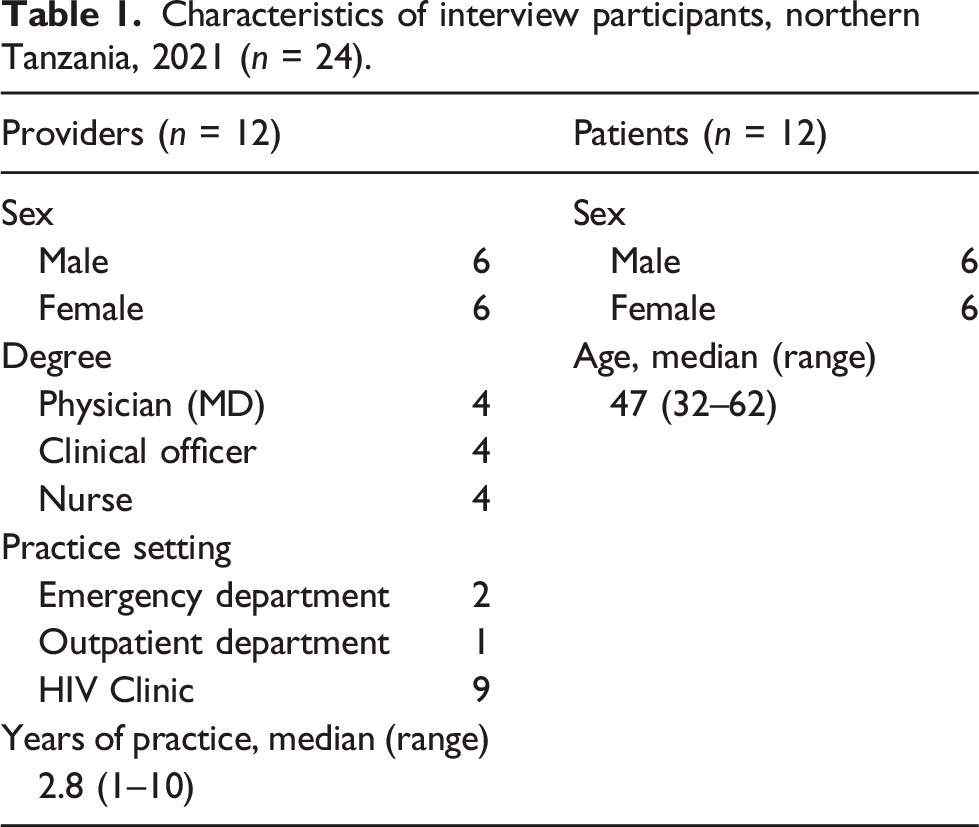
Characteristics of interview participants, northern Tanzania, 2021 (n = 24).
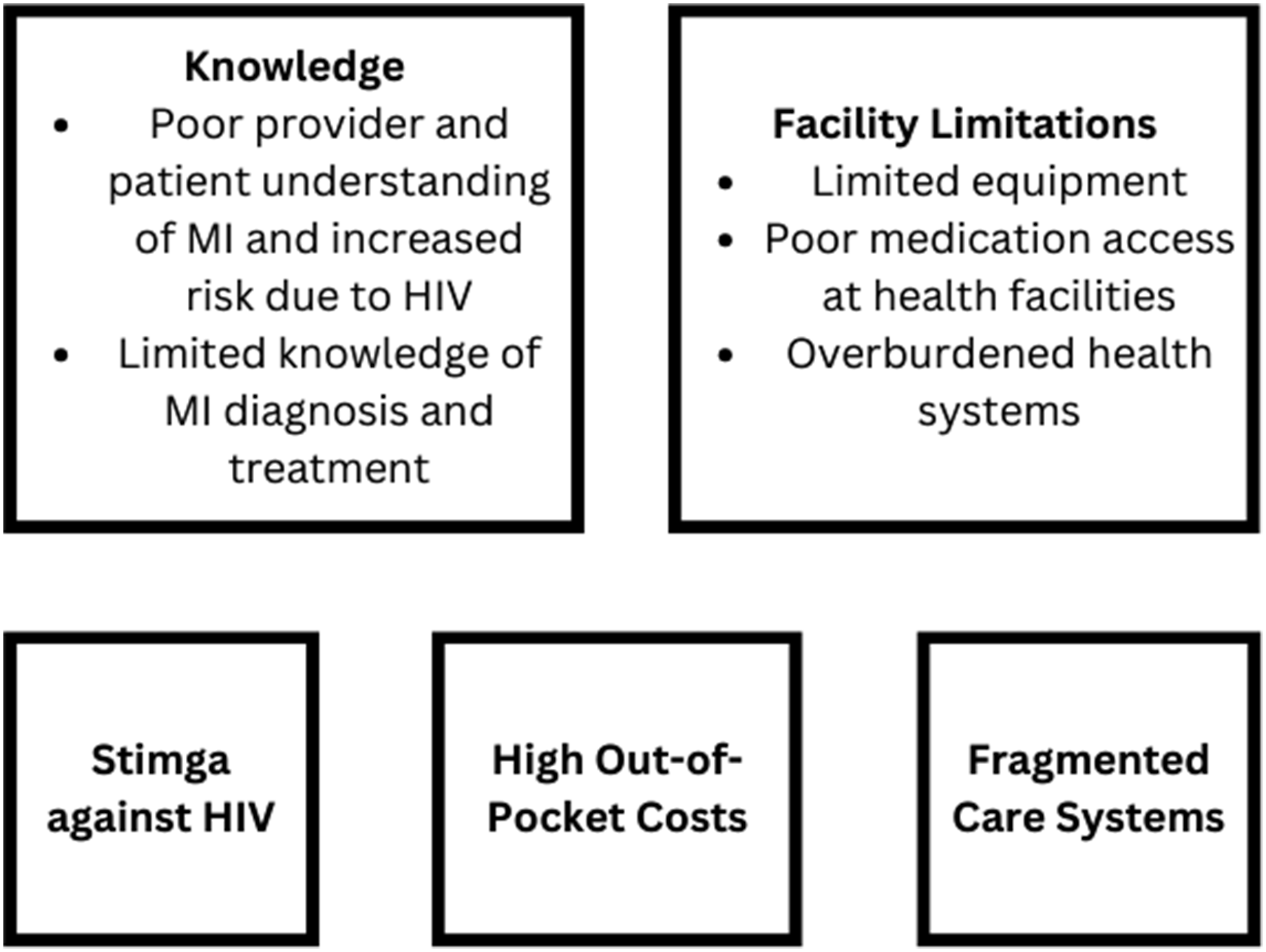
Overview of the barriers to myocardial infarction care among people living with HIV, Northern Tanzania, 2021.
Domain 1: Understanding of MI in PLWH
Theme 1: Incomplete understanding of MI
About half the providers (n = 7) were able to correctly explain the pathophysiology of an MI as a lack of blood flow to the heart. Other providers (n = 7) and patients (n = 5) described an MI as something that “happens suddenly after receiving some shocking news” (Provider 2, MA), such as an HIV diagnosis. Some patients had never heard of the term MI or heart attack, although their ECGs displayed signs of prior MI. “Heart attack is what happens when you lost someone and your heartbeat starts to change. But later on, when you have accepted the news then you feel better and your heartbeat gets normal” (Patient 4).
Theme 2: Perceptions of MI risk in PLWH
Half of providers did not think MI was a common problem, and a few providers (n = 2) were unsure if MI is a common problem in PLWH because their facility does not have the capacity to diagnose MI. A handful of providers (n = 4) were aware of the increased risk of MI in PWH, and no patient knew that their HIV status increased their risk for CVD. “Being shocked about your [HIV] status and it may affect your heart. You know the heart responds to what your mind triggers…[but HIV and MI] are not related.” (Patient 12).
Domain 2: Barriers to diagnosis and treatment of MI
Theme 1: Inadequate provider training in MI diagnosis
More than half of the providers (n = 7) reported that they did not know any signs or symptoms of MI, while others incorrectly reported that MI typically presents with heart palpitations and that blood pressure measurement could be used to diagnose MI. When asked how their facility diagnosed MI, one provider responded that they “do tests like checking blood pressure, their blood glucose levels…” (Provider 4, RN).
Some providers (n = 4) reported that they would primarily suspect infectious diseases such as tuberculosis in a patient with HIV complaining of chest pain. Among physicians and clinical officers responsible for diagnosis (n = 8), only about half of indicated that they could interpret an ECG (n = 4), had ever heard of cardiac biomarker testing for MI diagnosis (n = 5), or were familiar with the Tanzanian national guidelines for MI care (n = 3).
Finally, providers reported a lack of adequate experience with MI. A few providers (n = 4), mostly those at HIV health centers, indicated that they had never cared for an MI patient. Most providers (n = 10) indicated that they only received “shallow trainings” on MI in school (Provider 2, RN); those with shorter professional training such as nurses indicated that more thorough training regarding CVD was only available for physicians. Patient participants had little knowledge of MI diagnosis and therefore did not report any barriers related to provider training in MI diagnosis.
Theme 2: Patient care-seeking behavior for MI symptoms
Most providers (n = 9) felt that limited patient awareness leads PLWH to not seek care for MI symptoms and neglect appropriate follow-up care after an MI event. Only a few patients (n = 3) indicated that they would seek medical care for symptoms such as acute chest pain and shortness of breath while others (n = 5) indicated that they only seek emergency care when they felt very ill. Most patients (n = 11) reported having attributed prior episodes of chest pain to infectious diseases such as the flu and not heart disease; one provider explained that “many [patients] think that when [they] have a chest pain it means you have pneumonia, so they buy antibiotics” (Provider 7, MD, Emergency Department).
Both providers (n = 6) and patients (n = 4) indicated that stigma limits PLWH from seeking appropriate care due to fear of disclosing their HIV status to additional providers. “When [PLWH] are sick they run to the HIV clinic because they know there is good confidentiality there. If they go to other healthcare facilities, they are afraid that their status will be disclosed to many people if their acute diagnosis is connected to HIV” (Provider 6, MD, Outpatient Department). “Most [PLWH] have disclosed to only one person… You find that they are looking for the appropriate time to come to the [HIV clinic] unnoticed” (Patient 1).
Theme 3: Resource-related barriers
Most providers (n = 11) and patients (n = 10) reported that high out-of-pocket costs are a major barrier to accessing diagnostic tests and MI care, and little can be done when a patient is unable to afford care other than encouraging them to ask family and friends for the funds. One provider explains that “there is a temporary exemption for in-patients to [provide medicines and perform diagnostic tests]… but upon discharge, they will be required to pay for all those costs” (Provider 10, MD).
Most providers (n = 11) also reported that limited equipment complicates MI diagnosis and treatment. Providers from HIV clinics indicated that they only have access to basic equipment like manual blood pressure cuffs and this equipment does not always function properly. Providers who worked at larger hospitals also reported equipment-related barriers to MI diagnosis. “you will find that the BP machines are faulty. When you want to check the blood pressure, you can’t at that particular time because the only BP machine has been taken somewhere else” (Provider 4, RN). “There used to be only 1 [ECG machine] for the entire hospital so you can see a hospital caring for about 1,000 patients has only 1 ECG machine” (Provider 10, MD).
Domain 3: Barriers Related to long-term MI Care
Theme 1: Missed opportunities for MI care along care pathways
Even though each patient’s ECG demonstrated evidence of prior MI, no patient had been formally diagnosed with MI. Half of patients (n = 6) reported that they had never been screened for cardiovascular risk factors, and all reported that such screening was not routine practice at their HIV clinic. Even among those with known cardiovascular comorbidities, patients reported little engagement with noncommunicable disease care: only one of four diagnosed patients was currently seeking treatment for their hypertension. One patient who had been diagnosed with hypertension explained that his physician had not prescribed him medication, but instead advised him “to drink a lot of glucose solution, avoid noisy areas and avoid quarrels” (Patient 6). This poor engagement in CVD care was in stark contrast to high levels of engagement with HIV-care: all patients stated that they routinely attended HIV clinic appointments and adhered to their antiretroviral medications.
Theme 2: Ill-equipped health facilities
Most providers (n = 11) reported facility-related barriers to long-term MI treatment, such as a lack of medications for cardiovascular conditions. For example, pharmacies at HIV clinics only stock HIV-related medications. Other identified barriers include overburdened health systems with long wait-times and a lack of specialists like cardiologists. These limitations, combined with the lack of diagnostic equipment and inadequate provider training, require many health centers to refer patients with CVD to specialized cardiac clinics. Most providers (n = 8) attested that the only long-term MI care they provide for patients is lifestyle counseling: “They can get counselling advice but on treatment we mostly link them to another facility so that they can get medication” (Provider 4, RN).
Theme 3: Integration of cardiovascular care with routine HIV-related care
Integrated cardiovascular and HIV care is currently not standard practice in Tanzania. Most providers (n = 10) supported integrating cardiac services into routine HIV care, preferring a “one-stop shop where [patients] get all their services [including HIV-specific care and heart care] in one place ” (Provider 12, CO). However, all providers expressed feasibility concerns: they were worried that the HIV clinics did not have capacity or expertise to provide integrated CVD care. Patients were also interested in receiving cardiovascular care at their HIV clinic; most (n = 8) stated that they typically seek information and care for noncommunicable conditions from the HIV clinic first and would prefer to receive care for their noncommunicable comorbidities at the HIV clinic.
Discussion
This study, among the first to examine barriers to MI care among PLWH in SSA, identified key gaps in MI care across the care continuum, including in provider and patient understanding, in timely diagnosis, and in the provision of comprehensive long-term care. These findings highlight the need for interventions to strengthen care systems for managing MI in PLWH.
We identified an incomplete biomedical understanding of MI and inaccurate risk perceptions of MI in PLWH among both patients and providers. This is consistent with prior work in SSA identifying gaps in provider training in CVD12,13,23 and poor understanding of MI symptoms among the general population and PLWH.15,24–27 Additional provider education has been shown to improve the quality of MI care elsewhere in SSA,28,29 and a similar approach should be considered among HIV providers. Moreover, patient-centered educational interventions have demonstrated efficacy in disease self-management in high-income settings,30–33 and may prove effective in Tanzania.
Identified barriers to MI diagnosis include limited provider experience in managing MI, delayed patient care-seeking behaviors, high-out-of-pocket costs, and a lack of diagnostic equipment. Task-shifting has been shown to be feasible and effective for the management of noncommunicable diseases among the general population elsewhere in SSA,34–36 and similar strategies could be considered for PLWH in Tanzania. Both equipment- and cost-related barriers are frequently reported as barriers to CVD care in SSA in existing literature.12,13,23,37 Since MI is a potentially life-threatening emergency—survival is reduced by 20% in patients who are initially misdiagnosed38–40—these findings indicate the need for interventions to improve patient access to facilities that are equipped to diagnose MI or expansion of diagnostic capacity to a wider network of facilities, including HIV clinics.
We found little routine engagement with preventative cardiovascular care and poor continued engagement with cardiac care following a cardiovascular event. Most patients reported only accessing routine HIV care, and many indicated they had never been screened for CVD risk factors prior to our study. This poor engagement with cardiovascular screening and treatment has been observed in general populations elsewhere in SSA,5,16,41,42 but is particularly concerning among this high-risk population. Primary healthcare systems remain under-resourced in many parts of SSA, making the management of the growing burden of chronic noncommunicable disease difficult;43–45 however, PLWH in Tanzania and elsewhere in SSA have access to a separate primary healthcare system for their HIV, which presents unique opportunities for leveraging CVD care. 46 Nonetheless, our study found that most HIV clinics were not stocked with CVD medications, requiring the patient to go to two locations for medications to different conditions. Siloed systems limiting access to common CVD medications and complicating follow-up care were also identified in other studies in SSA.15,23,37 These findings highlight the need for interventions to improve screening for CVD and linkage with long-term care for PLWH. Prior studies from elsewhere in SSA have shown that integrating hypertension and diabetes care into existing HIV care systems is feasible and could improve care engagement.47–50 In our study, most participants preferred integrating cardiovascular services into HIV care to promote ease and accessibility of these services. Additional study is needed to determine whether integration of care in Tanzania would improve outcomes.
This study had several limitations. First, while we provided a safe, private space for participants to share their honest opinions, participants’ responses may have been influenced by social desirability bias. Second, this study recruited PLWH engaged with HIV care and does not capture the experiences of individuals with undiagnosed or untreated HIV. Finally, this study did not include policy-makers or healthcare administrators; the perspectives of these individuals would have provided additional important information about political and systems-related barriers to MI care among PLWH in Tanzania.
In conclusion, to address the barriers to MI care identified in this study, multifaceted interventions are needed to improve provider and patient knowledge of MI among PLWH and to improve quality of MI care for PLWH in Tanzania. Additional research is needed to design interventions to improve the quality of cardiovascular health screening and subsequent care in this population.
Supplemental Material
Supplemental Material - Patient and provider perspectives on barriers to myocardial infarction care among persons with human immunodeficiency virus in Tanzania: A qualitative study
Patient and provider perspectives on barriers to myocardial infarction care among persons with HIV in Tanzania: A qualitative study by Sainikitha Prattipati, Tumsifu G Tarimo, Godfrey L Kweka, Jerome J Mlangi, Dorothy Samuel, Francis M Sakita, Anna Tupetz, Janet P Bettger, Nathan M Thielman, Gloria Temu and Julian T Hertz in International Journal of STD & AIDS
Supplemental Material
Supplemental Material - Patient and provider perspectives on barriers to myocardial infarction care among persons with HIV in Tanzania: A qualitative study
Patient and provider perspectives on barriers to myocardial infarction care among persons with HIV in Tanzania: A qualitative study by Sainikitha Prattipati, Tumsifu G Tarimo, Godfrey L Kweka, Jerome J Mlangi, Dorothy Samuel, Francis M Sakita, Anna Tupetz, Janet P Bettger, Nathan M Thielman, Gloria Temu and Julian T Hertz in International Journal of STD & AIDS
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: National Heart, Lung, and Blood Institute (1K23-HL155500-01), Society for Academic Emergency Medicine, Center for AIDS Research, Duke University (P30 AI064518).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
