Abstract
Background
People living with HIV (PLWH) starting or switching to an integrase strand transfer inhibitor-based regimen are more likely to experience weight gain than other classes of antiretroviral regimens. The aim of this study was to evaluate the weight gain and metabolic disturbances in PLWH who start antiretroviral therapy (ART) with bictegravir/emtricitabine/tenofovir alafenamide and in individuals who switch from another ART to BIC/FTC/TAF after 48 weeks.
Methods
A prospective longitudinal study was conducted in an HIV clinic in Mexico. Weight and metabolic parameters were measured at baseline, 24 and 48 weeks. A paired t test and Wilcoxon signed-rank test were applied to evaluate weight and metabolic changes.
Results
160 participants completed measurements, median age was 29 (IQR 26-32) and 30 (IQR 27–34) years old for the treatment-naïve and switch group respectively. In the treatment-naïve group, mean weight change was 3.8 kg (±5.8) (p < .001) and BMI increased 1.3 kg/m2 (±2) (p < .001) at 48 weeks. Incidence of BMI >25 kg/m2 was 28% (95%CI; 18%–40%) and BMI >30 kg/m2 was 7% (95%CI; 2%–16%) at 48 weeks in treatment-naïve individuals. In the switch group, mean weight gain and BMI change at 48 weeks was 2.8 kg (±5.9) and 0.9 kg/m2 (±2.0) respectively (p < .001). Incidence of BMI >25 kg/m2 was 17% (95%CI; 8%–32%) and BMI >30 kg/m2 12.8% (95%CI; 5%–26%) at 48 weeks respectively.
Conclusions
Weight gain should be considered when men PLWH are treated with BIC/FTC/TAF regimen. They should be informed about this possible adverse event and strategies of intervention.
Background
The integrase strand transfer inhibitor (INSTI) class has demonstrated a good tolerability profile. However, recent evidence suggests that people living with HIV (PLWH) starting on or switching to an INSTI based regimen are more likely to experience weight gain than those on antiretroviral regimen based on other classes.1,2
There have also been reports of increased weight gain with Tenofovir Alafenamide (TAF) use in antiretroviral therapy (ART)-naïve individuals in the ADVANCE trial. 3 In addition, in treatment-naïve PLWH, a return to health effect in part characterized by a restoration of immune function and weight gain, has been postulated. 4 However, these possible causes for weight gain have to be balanced against an increase in overweight/obesity due to diet and sedentary lifestyle in the general population also affecting PLWH. 5 Weight gain may lead to visceral fat accumulation, dyslipidemia, insulin resistance, and arterial hypertension, increasing the risk of type-2 diabetes and cardiovascular disease (CVD).6,7,8
The aim of this study was to evaluate weight gain and metabolic changes in PLWH who start ART with bictegravir/emtricitabine/tenofovir alafenamide (BIC/FTC/TAF) and in individuals who switch from another ART regimen to BIC/FTC/TAF.
Methods
Design
A prospective longitudinal study was conducted from September 2020 to November 2021, in men PLWH treated at the Hospital de Infectología “La Raza'” National Medical Center, in Mexico City. The HIV clinic is a tertiary level reference center for people with social security coverage.
Study population
Study subjects were invited to participate if they were men (male sex assigned at birth) who have sex with men (MSM) living with HIV and ≥18 years old. Two cohorts were included, ART naïve PLWH and those on ART virologically suppressed for more than 3 months, defined as HIV-1 RNA viral load <40 copies without previous virological failure, who were switched to BIC/FTC/TAF. Study subjects were excluded if they did not complete the measurements until 48 weeks of treatment for any cause (change of HIV clinic, loss of social security, informed consent withdrawal or switch to another regimen for adverse events).
Measurements
Weight and height were measured during routine clinical care, and recorded in electronic health records. Repeated measures of weight in kilograms (kg) were done at baseline, 24 weeks and 48 weeks on BIC/FTC/TAF.
The laboratory test performed were glucose (mg/dL), triglycerides (TG) (mg/dL), total cholesterol (TC) (mg/dL), HDL-cholesterol (HDL-c) (mg/dL), LDL-cholesterol (LDL-c) (mg/dL), CD4+ cells count (cells/mm3) and HIV-1 RNA (Log10). Metabolic disturbances were defined as impaired fasting glucose (≥100 mg/dL), hypertriglyceridemia (≥150 mg/dL), low HDL-cholesterol (≤40 mg/dL) according to the definition of metabolic syndrome from the National Cholesterol Education Program (NCEP) Adult Treatment Panel III (ATP III). 6 We calculated BMI, and it was classified as <18.5 kg/m2 underweight, 18.5 to <25 kg/m2 healthy weight, 25.0 to <30 kg/m2 overweight and 30.0 kg/m2 or higher obesity according to the World Health Organization (WHO).
Sample size
The sample size was calculated with a 95% confidence level and an 80% of power, using a formula for a single cohort study with a sample size of 74 participants in each group for a total of 148 subjects.
Statistical analysis
The qualitative variables were analyzed through frequency and percentage tables. Quantitative variables, according to their non-normal distribution (Kolmogorov-Smirnov test), were summarized using median and interquartile ranges (IQR). Percentage was obtained in order to evaluate the weight gain classification. We assessed weight change and BMI at 48 weeks with a paired t test and metabolic disturbances with Wilcoxon signed-rank test. All analyses were conducted using SPSS software (version 22; SPSS IBM Corp., Armonk, NJ, USA). Figures were created with GraphPad Prism v.9.0.1.
Ethical consideration
The study was approved by the local committee for health research 3502 at the Hospital de Infectología “La Raza” National Medical Center, with registration number R-2021-3502-084. Written informed consent was obtained from all participants.
Results
Baseline characteristics of the study population.
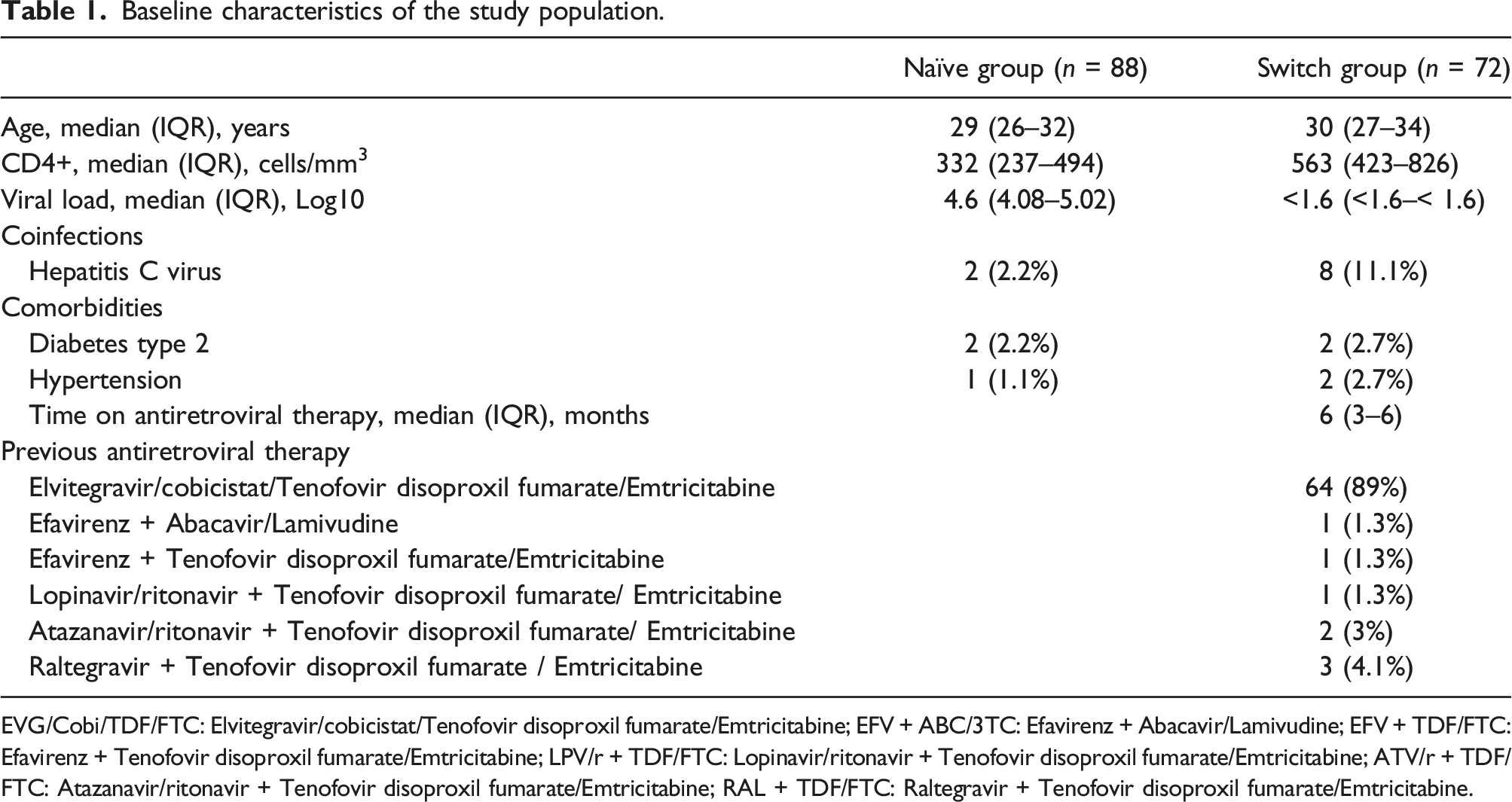
EVG/Cobi/TDF/FTC: Elvitegravir/cobicistat/Tenofovir disoproxil fumarate/Emtricitabine; EFV + ABC/3TC: Efavirenz + Abacavir/Lamivudine; EFV + TDF/FTC: Efavirenz + Tenofovir disoproxil fumarate/Emtricitabine; LPV/r + TDF/FTC: Lopinavir/ritonavir + Tenofovir disoproxil fumarate/Emtricitabine; ATV/r + TDF/FTC: Atazanavir/ritonavir + Tenofovir disoproxil fumarate/Emtricitabine; RAL + TDF/FTC: Raltegravir + Tenofovir disoproxil fumarate/Emtricitabine.
In the treatment-naïve group (n = 88) the median age was 29 years (IQR: 26–32), median CD4+ cell count 332 cells/mm3 (IQR: 237–494) and median HIV RNA 4.6 log (IQR: 4.08–5.02). For the switch group (n = 72) the median age was 30 years (IQR: 27–34.7) with a median CD4+ cell count of 563 cells/mm3 (IQR: 423–826), all individuals had HIV RNA <40 copies/ml at baseline.
The mean initial weight in the treatment-naïve group was 71 kg (±13) and mean BMI was 24.2 kg/m2 (±4.2); for the switch group, the mean weight at baseline was 72.5 kg (±12.7) and mean BMI was 24.6 kg/m2 (±3.2). In the treatment-naïve group, 27% (95%CI; 19%–37%) individuals had a BMI >25 kg/m2 before starting ART, and 7.9% (95%CI; 4%–15%) participants had a BMI >30 kg/m2.
In the treatment-naïve group, the mean weight change was 3.8 kg (±5.8) at 48 weeks (p<.001); mean BMI increase was 1.3 kg/m2 (±2) at 48 weeks (p <.001) (Figure 1). Incidence of BMI >25 kg/m2 was 28% (95%CI; 18%–40%) and BMI >30 kg/m2 was 7% (95%CI; 2%–16%) at 48 weeks in treatment-naïve individuals. (Figure 2). A total of 23/88 individuals (26%; 95%IC 18%–36%) had 5%–10% weight gain and 21/88 participants (23%; 95%IC 16%–33%) gain >10% (Figure 3). Weight gain from baseline to 48 weeks in treatment-naïve and switch groups. Distribution of participants by percentage change in BMI from baseline to 48 weeks in treatment-naïve and switch groups (<25, 25–30, >30 kg/m2). Distribution of participants by percentage change in weight from baseline to 48 weeks in treatment-naïve and switch groups (<5, 5–10, >10%).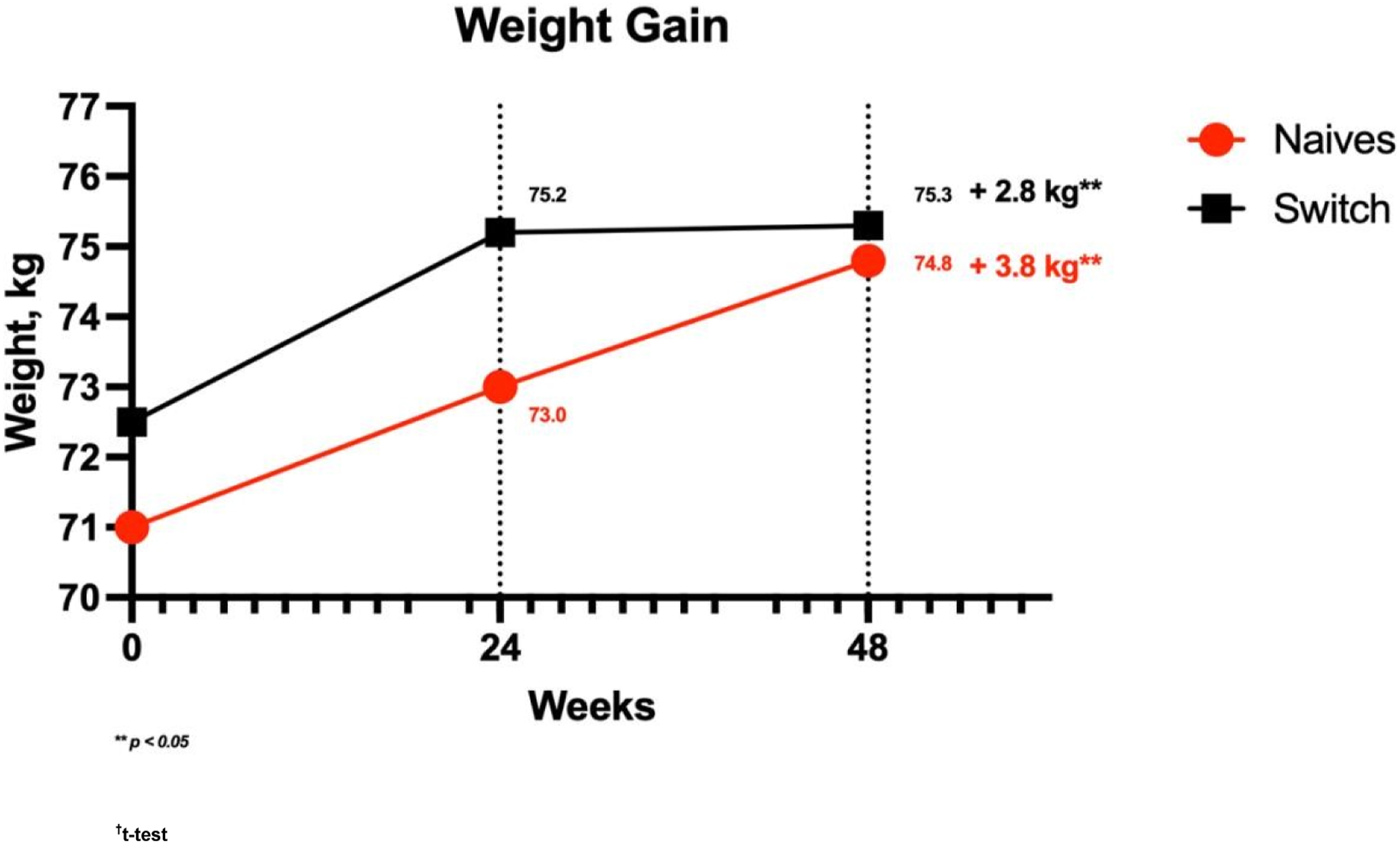
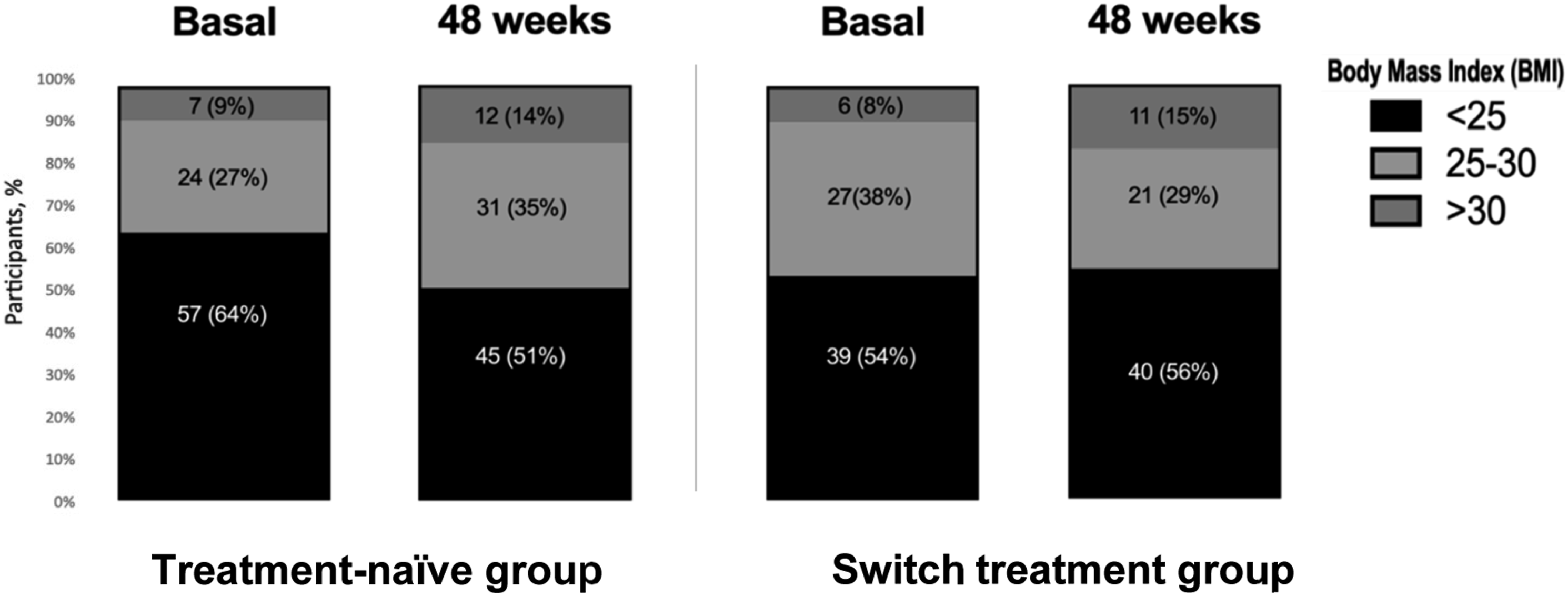

In the switch group, mean weight gain and BMI change at 48 weeks was 2.8 kg (±5.9) and 0.9 kg/m2 (±2.0) respectively (p <.001) (Figure 1). Incidence of BMI >25 kg/m2 was 17% (95%CI; 8%–32%) and BMI >30 kg/m2 12.8% (95%CI; 5%–26%) at 48 weeks (Figure 2). A total of 22/72 individuals (30.5%; 95%CI 21%–41%) had 5%–10% weight gain and 14/72 participants (19.5%; 95%CI 12%–30%) gain >10% (Figure 2). Two patients started atorvastatin during the period of the study, both of them were in the switch group.
Measurements in treatment-naïve individuals from baseline to 48 weeks (n = 88).
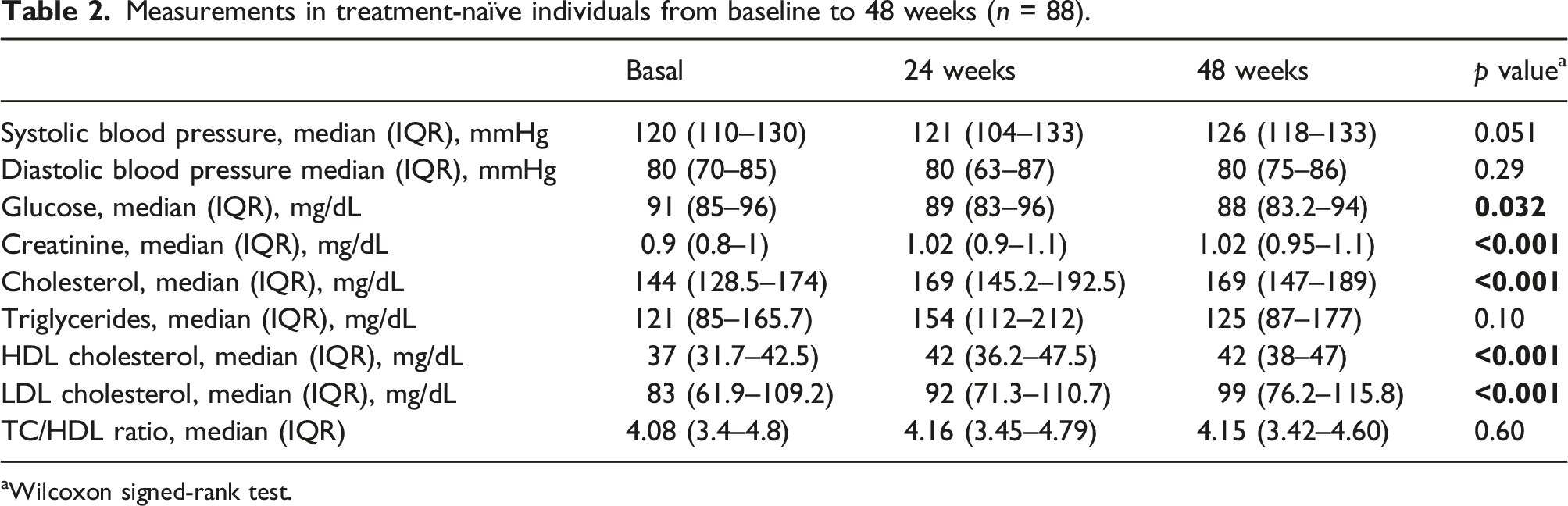
aWilcoxon signed-rank test.
Measurements in switch treatment individuals from baseline to 48 weeks (n = 72).
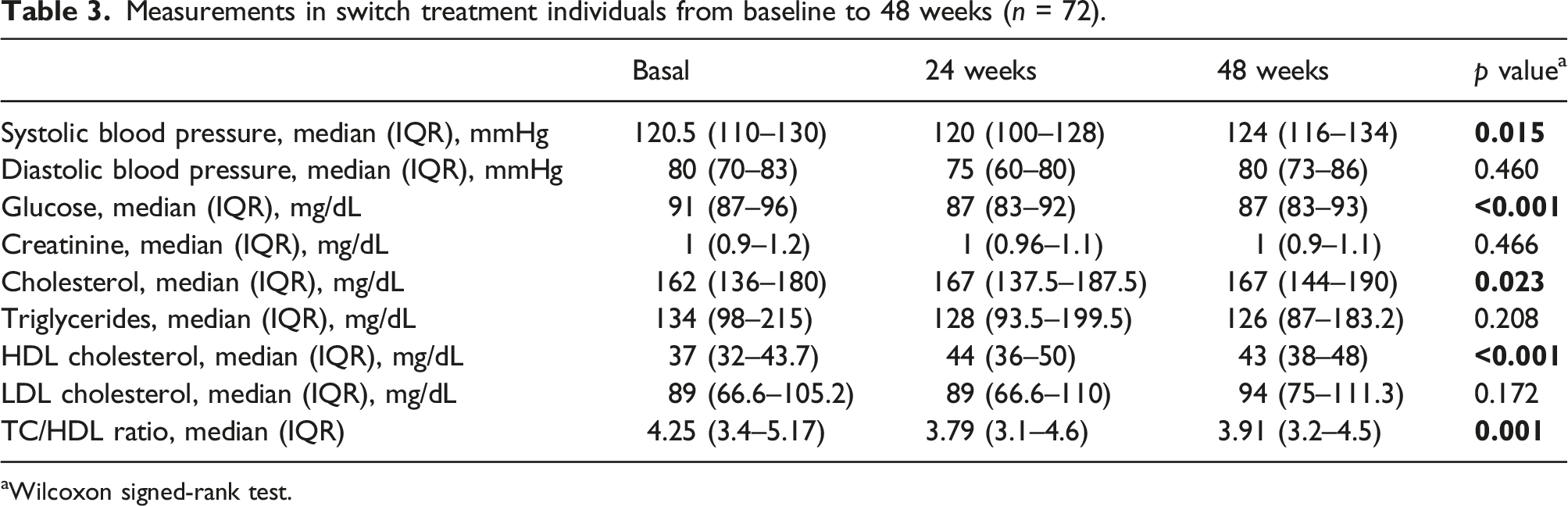
aWilcoxon signed-rank test.
Discussion
In this prospective cohort study in Mexican MSM living with HIV, weight gain was observed in PLWH initiating and switching from stable ART to BIC/FTC/TAF. In the treatment-naïve group weight gain may be considered at least partially to be due to a return to health. However, in PLWH already on stable ART this phenomenon should not be a major contributor to the weight gain observed. For this, it can be assumed that in this group the weight gain is not associated with the control of a consummating disease, but due to a direct effect of the ART. Interestingly, the general patterns of weight gain and metabolic changes in both groups were comparable.
Similar to our findings, prior studies have suggested that INSTI-based regimens were associated with weight gain. In this study the weight gain in treatment-naïve men individuals was 3.8 kg, almost identical to the NAMSAL trial with a weight gain of 4 kg in the men group receiving dolutegravir (DTG)-based regimen. Regarding weight percentage, we observed a weight gain >10% in 21 (24%) individuals, similar to the NAMSAL trial where they found weight gain >10% in 27 (26%) individuals. 9 In the ADVANCE trial, there was significantly more weight gain in DTG-containing regimens, especially in combination with TAF, than with Efavirenz (EFV) plus Tenofovir Disoproxil Fumarate/Emtricitabine (TDF/FTC) regimen. At week 48, absolute and the percentage of weight gain of PLWH in whom obesity emerged during treatment were highest in the TAF-based group (6 kg, 14% new obesity), but the values in the TDF-based group (3 kg, 7% new obesity) were also higher than those in the EFV-group (1 kg, 6% new obesity). Our findings on the incidence of obesity were similar with the results among men in the ADVANCE trial. 3
Weight gain has been reported with TAF use among virologically controlled ART-experienced PLWH who switched to this drug from any other regimen. In the OPERA cohort, the average weight gained was 1.5 kg after 12 months, lower than the results observed in this study of 2.8 kg. This difference could be associated with older age of individuals in OPERA and the decrease of exercise in individuals in this study caused by the lockdown. 10
Most of the individuals in the switch group in this study were on EVG/cobi/TDF/FTC and more than 99% of them were on TDF-based regimen and it means a higher opportunity to gain weight according to OPERA study; in addition, most of them changed from a first generation INSTI (EGV/c) to BIC and weight gain has also found for this switch; Tiraboschi J et al., found in patients switching from EVG/c/FTC/TAF to BIC/FTC/TAF after 6 months, that the median body weight change was statistically significant (0.6 kg, p = .003); this shows that not only TAF was associated to weight gain, second generation INSTI was associated too.10,11
According with Sax et al., normal baseline BMI was associated with ≥10% weight gain when compared to individuals with overweight or obese baseline BMI (normal vs overweight: OR, 1.54 [95% CI, 1.27–1.87]; p < 0.001 and normal vs obese: OR, 1.66 [95% CI, 1.29–2.15]); In this study, in the naive group, only 2 (2.7%) and 5 (5.6%) in the switch group were underweight; then, for the analysis we included with the normal weight category (BMI <25 kg/m2). 12
The use of antiretrovirals may be associated with metabolic complications including dyslipidemia and increased cardiovascular events. 13 Mallon et al., indicate that TAF is associated with greater increases in TC, LDL-c and TG compared with TDF based regimens. 14 Milinkovic et al described an increase of TC more than 30 mg/dL after switching from TDF to TAF. In our findings, the impact of TC and HDL-c was less than the Milinkovic study, but with no difference in TC/HDL ratio in both groups of individuals; 15 in addition, in the switch group two individuals were eligible for treatment with atorvastatin medication.
We have also several limitations in the current analysis. First of all, we do not have a control group for both cohorts and we were not able to adjust for weight gain over time due to lifestyle including data on the type of food being consumed by the participants or rather eating habits; secondly, just weight was measured as part of clinical assessment, rather than measurement of body composition than could give more data for this study in order to distinguish if the weight gain was fat or muscle or if the fat gain was in trunk, limbs or visceral areas; another limitation in this study was that all the participants were men and we were not able to control the impact of the COVID-19 lockdown in the decrease of exercise. In addition, we are unable to differentiate between the possible contributions of TAF or bictegravir because all individuals were treated with BIC/FTC/TAF. We found that in Latin American PLWH who start BIC/FTC/TAF regimen weight gain was common, in addition; we were able to determine ≥5% and ≥10% weight gain in individuals with similar observation time and demographic characteristics, >5% of weight gain in the first 6 months was associated with diabetes and >10% of weight gain is associated with increased cardiovascular risk.
Conclusions
Routine measurement and monitoring of BMI, waist, and body composition may provide important insights and identify opportunities for interventions in Men PLWH.
Weight gain should be taken into account, when treating PLWH with TAF and INSTI based regimens. PLWH should be informed about this possible adverse event and make strategies of interventions.
Footnotes
Acknowledgements
We would like to express our gratitude to the participants of this study
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
