Abstract
Background
Understanding the cost-effectiveness of the HPV vaccine from a global perspective is important to assess from a policy perspective and to support current and future HPV vaccination programs.
Objectives
The aim of this analysis was to conduct a targeted literature review of published pharmacoeconomic literature on the cost-effectiveness of the HPV vaccine to treat patients in various countries, with a focus on cost-savings and their impact on vaccine recommendations.
Methods
We searched cost-effectiveness studies in HPV published in peer-reviewed literature from 2012 to 2020 using MEDLINE via the PubMed database and Google Scholar.
Results
HPV vaccine cost-effectiveness was found to be greatest in low-income countries where screen programs were not yet in place, additionally, in adolescent males and females. The majority of the economic evaluations viewed the implementation of the HPV vaccine as cost-effective and recommended national HPV vaccination.
Conclusion
The majority of economic studies favored national HPV vaccination for adolescent males and females in various countries. Feasibility of this strategy and implementation remains an open question, in addition to screening coverage rate in countries with no vaccine programs or countries yet to introduce national HPV vaccination.
Introduction
Human papillomavirus (HPV) is a viral infection that is primarily transmitted through sexual contact.1,2 There are over 200 distinct types of HPV, with some subtypes associated with the development of different types of cancer, including cervical, anal, oropharyngeal, vaginal, vulvar, and penile cancer. In addition, HPV can also cause benign conditions such as warts on the skin and genitals. HPV can infect basal cells of the vulva, vagina, anus, cervix, neocervix, oropharynx, intestinal vaginoplasty, penis, and perianal region. The virus is known to replicate slowly once its viral DNA has integrated into the host genome; it maintains a low copy number allowing it to evade humoral immune detection. 3 Individuals with multiple sexual partners and persistent HPV infection are at higher risk of acquiring more HPV subtypes. 4 HPV transmission can occur during vaginal or anal intercourse, and via oral-genital or genital-genital contact due to mucosal trauma. Hence, no differences have been seen in the HPV transmission rates reported between heterosexual women and women who have only had sex with women. 3
In 2022, approximately 79 million people in the United States were infected with HPV. With 14 million new infections occurring annually in sexually active people, the HPV infection rate remains high across all ages in the U.S. 3 While there is currently no cure for HPV, there are vaccines available that can prevent acquisition of many HPV strains. Though there are treatments for some of the symptoms that HPV causes, there are no treatments for the virus itself. Today, there are primary (vaccination) and secondary prevention programs (cervical screening) that can show benefit and impact cancer prevention. Despite these approaches, many are not available for those at greater risk. 5
Currently, there are more than 200 HPV serotypes. 40 of these serotypes are transmitted through direct sexual contact and can be categorized into low-risk and high-risk serotypes (based on risk of cancer development following infection). Low-risk HPV serotypes (such as 6 and 11) can cause anogenital warts and respiratory papillomatosis, and affect the cutaneous squamous epithelium of the skin. High-risk HPV serotypes (such as 16, 18, 31, 33, 45, 52, and 58) are commonly associated with cervical, vulvar, vaginal, anal, oropharyngeal, and penile cancers. 3 Cervical cancer is known to be primarily caused by HPV. Over 300,000 women died from cervical cancer in 2018, 90% of these preventable deaths were from low-income and middle-income countries (LMICs). While the pap smear has been used as a preventative measure for cervical cancer since the 1950s, the development of primary prevention approaches has become a beneficial innovation. 6 Since the first licensed HPV vaccine in 2006, it has shown great promise in effectively reducing incidence rates of cervical cancer. 7 Early development of the HPV vaccine has demonstrated to target strains that are 70% responsible for cervical cancer (HPV-16 and HPV-18), which have shown high efficacy against infection in adolescent girls. Additional strains (HPV-11 and HPV-6) are now included in the quadrivalent vaccine which offers prevention of precursors of certain cancers and of precancerous cervical lesions. 6 The HPV vaccine Gardasil-9 offers protection against nine HPV types including 6, 11, 16, 18, 31, 33, 45, 52, and 58 which are known to cause the majority of cervical, vulvar, vaginal, and anal cancers, as well as genital warts. These HPV types are responsible for approximately 90% of cervical cancer cases and a significant proportion of other HPV-related cancers. 8
Since the global call to action against HPV was established from the World Health Organization (WHO) in 2018, the need to achieve high levels of vaccine coverage illustrates the importance of the cost-effectiveness of the vaccine. 7 With this interest in mind, we conducted a targeted literature review evaluating the various cost-effective analysis (CEA) of HPV vaccination to analyze intervention methods, the effect of factors on the methods, and interpret country-level recommendations of vaccination programs.
Methods
Search strategy
The studies in this review were based on a targeted literature search conducted through PubMed and Google Scholar on economic studies analyzing the cost-effectiveness of the HPV vaccine for cervical cancer, and any other HPV-related diseases for both males and females. Our analysis focused on the cost-effectiveness of the HPV vaccine on a global scale. We excluded articles that did not clearly state their recommendation for the HPV vaccine, the cost-effectiveness of the HPV vaccine, or the efficacy of the HPV vaccine. Systematic studies were chosen to incorporate a wide range of countries for analysis. To include all relevant studies, the search was conducted on published literature between 2012 and 2020. Bibliographic references from each study were reviewed for additional reports of similar analyses (Figure 1). Flow diagram of the included studies by search strategy.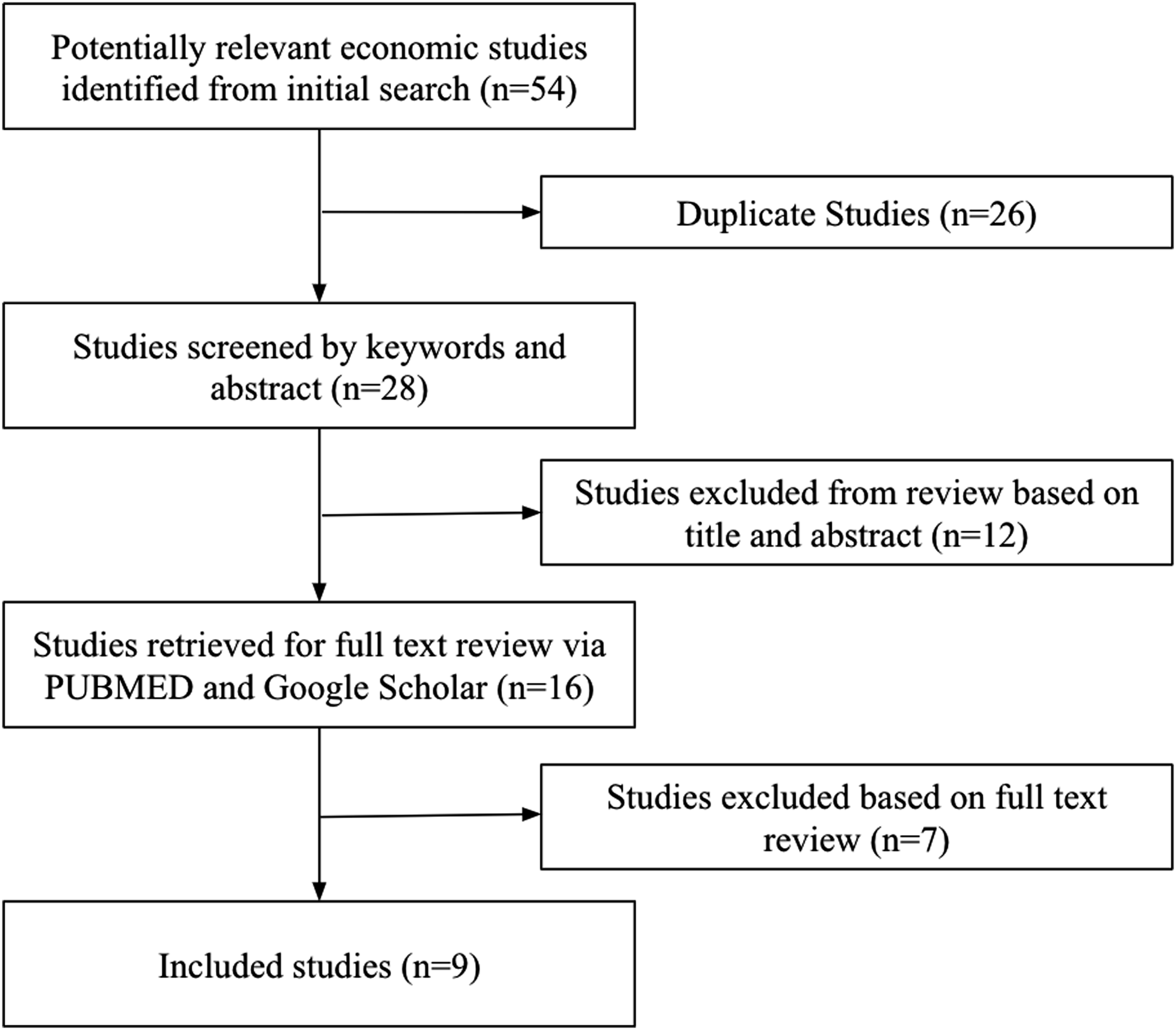
Keywords
Cost-effectiveness, economic analyses, human papillomavirus, vaccination, immunization, vaccine, HPV vaccination programs, sexually transmitted infection, cervical cancer, HPV prevention, HPV infection, health economic, targeted systematic reviews, national immunization, cervical cancer screening, vaccine-preventable diseases.
The screening of the targeted literature was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement.
9
Information related to the health economic analysis was extracted according to the Consolidated Health Economics Evaluation Reporting Standards (CHEERS) statement.
10
The information extracted from each study were first authors’ name and year of publication, number of countries represented, study population, currency and base, unit of cost effectiveness, and conclusion. Different strategies for the HPV-vaccine (cervical cancer screening and the nine-valent or sixteen/18 vaccine) were categorized and compared to four alternative strategies: ➢ Cervical cancer screening and the HPV vaccine for girls only; ➢ Cervical cancer screening and the HPV vaccine for boys only; ➢ Cervical cancer screening and the HPV vaccine for both girls and boys only; ➢ Screening strategy only
Health economic parameters
To simplify the study comparison, only cost-effective analyses of systematic reviews were included in the review (with the exception of the PRIME modeling study).9,10 Two types of representations for the incremental cost-effectiveness ratio (ICER) for the health outcome measured were described: Life-years (LYs) gained or lost, and the health-related quality of life measured by quality-adjusted-life-years (QALYs). Figure 1— reports the ICERs associated with female or male vaccination depending on vaccination coverage, available screening programs, and the total vaccination cost.
Health outcomes
Summary of results of included studies.
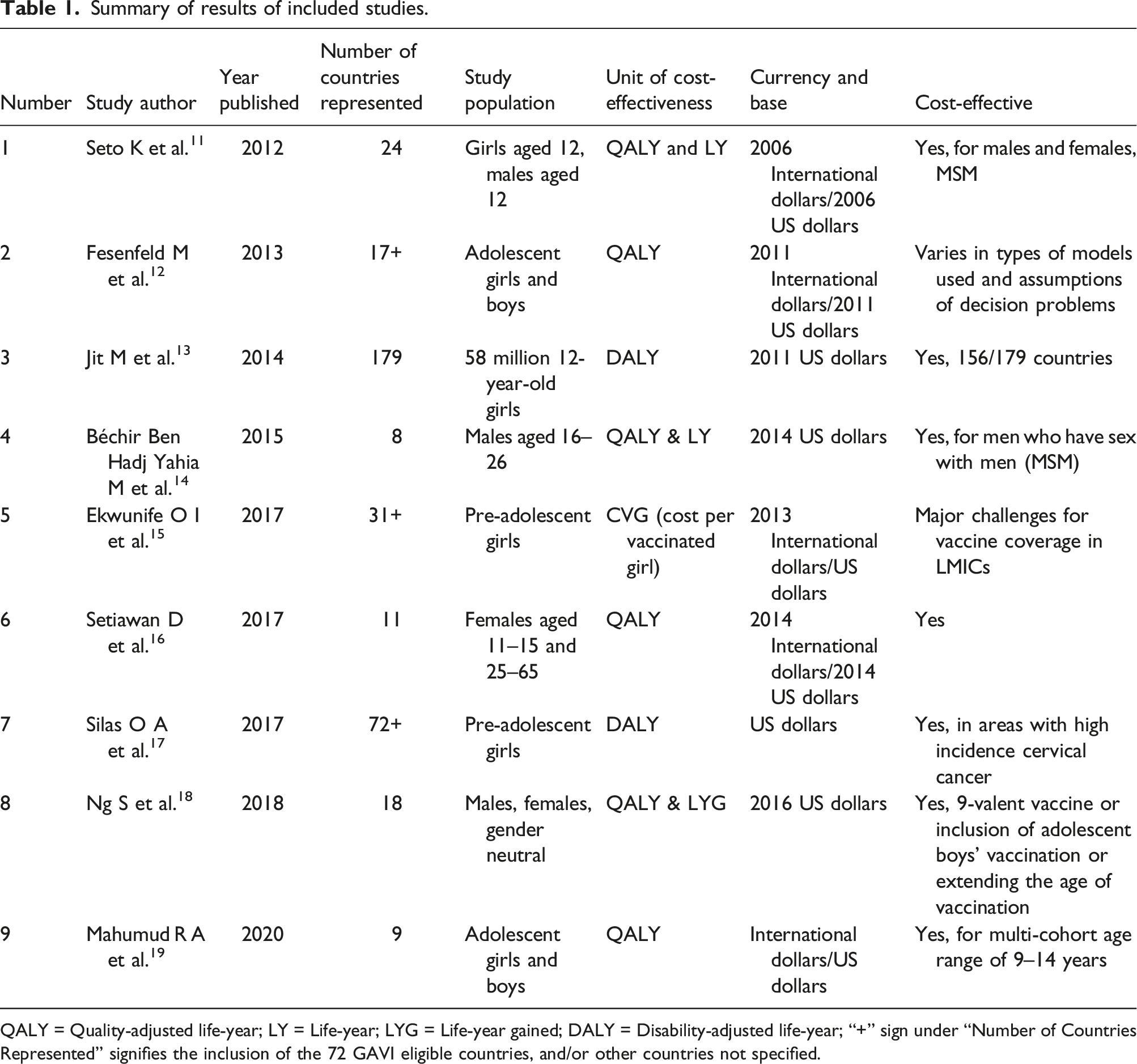
QALY = Quality-adjusted life-year; LY = Life-year; LYG = Life-year gained; DALY = Disability-adjusted life-year; “+” sign under “Number of Countries Represented” signifies the inclusion of the 72 GAVI eligible countries, and/or other countries not specified.
Results
The targeted literature identified a total of 9 CEA and systematic reviews representing 179 countries which were included. Upon review of these various economic analyses, we learned that there were differences in the models (static, dynamic, or hybrid) used, varying vaccination strategies (vaccination alone or vaccination plus screening), varying health outcomes (cost per QALY, LYG, LY, DALY, or cost per vaccinated girl) and differences in vaccine type. The countries that were more frequently represented in these analyses were Canada, Mexico, the United States, Australia, Italy, and the United Kingdom. The general consensus was that the HPV vaccine was found to be a cost-effective method, thus recommended. However, there were some exceptions to this. It was found that only three out of the 11 represented South Asian countries in the Setiawan et al. 16 study have actually implemented a comprehensive prevention for cervical cancer. However, Iran was demonstrating a non-cost-effective result to introduce HPV vaccination due to high HPV vaccine prices, as reported by Giraldi et al. 17
Out of the 33 studies from Ekwunife et al., 15 26 acknowledged the cost-per-vaccinated-girl and vaccine coverage rate (21 studies) as particular challenges for LMICs, while only nine studies identify screening coverage rate as a challenge. 10 out of the 12 studies from Mahumad et al. concluded that 9-valent vaccination was cost-effective. When adolescent boys were included in immunization programs, vaccination was cost-effective when vaccine price and coverage were low. They also found that when vaccination coverage for females was more than 75%, gender-neutral HPV vaccination was less cost-effective than vaccination only targeting girls aged 9–18 years. Multi-cohort immunization was found to be cost-effective in the age range of 9–14 years; the upper-age limit found the vaccine to not be cost-effective. 19
Similarly, as reported by Ng et al., it was found that vaccination programs for adolescent boys were cost-effective if the vaccine price and coverage were low; when female coverage was over 75%, they found that gender-neutral vaccination was less cost-effective than when targeting only girls aged from 9-18 years. Note that their study did not provide conclusive evidence of greater cost-effectiveness of the 9-valent vaccine in comparison to older HPV vaccines since the price for the 9-valent vaccine was still uncertain.18,20 It can be concluded that apparent country disparities exist for HPV vaccination; countries with most to gain with the introduction of national HPV vaccination have yet to implement this program. Vaccination is most cost-effective in settings where vaccination screening programs are not yet in place.
Discussion
Vaccine preventable diseases are diseases that are mostly avoidable by immunization, such as HPV. According to the Association for Professionals in Infection Control and Epidemiology 21 (APIC), vaccination is a highly effective method to prevent certain infectious (communicable) diseases. Not only do vaccinations protect the recipient, they also prevent the disease and illness from spreading to others. According to the WHO fact sheet, 22 cervical cancer is the fourth most frequent cancer in women with an estimated 604,000 new cases in 2020. Of the estimated 342,000 deaths from cervical cancer in 2020, about 90% of these occur in low- and middle-income countries. Moreover, in all world regions the contribution of HIV to cervical cancer falls disproportionately on younger women. In the current targeted literature review study, we assessed the cost-effectiveness of the HPV vaccination across various countries. The majority of studies were in favor of HPV vaccination and supported HPV vaccination/immunization programs. However, some countries found vaccination programs to be non-effective. The country of Iran, from Silas et al., 17 found HPV vaccination to be non-cost effective due to the vaccine’s high price. Cost-effectiveness of HPV vaccination is sensitive to changes to the cost of vaccine supply, delivery, and implementation. A vaccine’s high price will raise the overall cost of vaccination programs, thus posing a barrier to the cost-effectiveness of HPV vaccination. 23 Similarly, Seto et al. 11 found the addition of boys to a vaccination program to exceed traditional cost-effective thresholds. Reasons for the HPV vaccine not being cost-effective in some countries could be that the cost (.i.e. QALY) outweighs the health benefit. For example, according to the WHO, the target age group for HPV vaccination is from 9 to 26 years. Some countries may have found vaccination programs outside these age ranges not to be cost-effective, thus it is important to vaccinate at the appropriate age and timing to prevent this disease. 24
Optimizing the number of doses administered for HPV vaccination can strengthen vaccination programs. A single-dose for HPV vaccination has been suggested to improve cost-effectiveness of vaccination programs. Optimizing the HPV dosing schedule is expected to improve access to the vaccine, reduce challenges with costly follow-ups, and allows reallocation of resources to other health priorities. Efficacy and durability of varying dose schedules for both males and females are still being researched. 25
Despite our review revealing that a majority of our studies were not from LMICs, more studies conducted in LMIC countries would grant specificity and warrant better support for local decision-making. These areas were found to have the most benefit of the vaccination program and were shown to be more cost-effective. In LMIC, there is limited access to these preventative measures and cervical cancer is often not identified until it has further advanced and symptoms develop. In addition, access to treatment of cancerous lesions (for example, cancer surgery, radiotherapy and chemotherapy) may be limited, resulting in a higher rate of death from cervical cancer in these countries. 22
Access to the HPV vaccination is essential not only within the United States, but from a global perspective as well. The World Health Assembly adopted the global strategy to accelerate the elimination of cervical cancer as a public health problem. The definition of elimination of cervical cancer has been set up as a country reaching the threshold of less than 4 cases of cervical cancer per 100 000 women per year. To reach this threshold by the end of the 21st century, WHO has set up targets to be reached by 2030 and to be maintained. 26 The targets are population that include: 90% of girls fully vaccinated with HPV vaccine by age 15; 70% of women are screened with a high-performance test by 35, and again by 45 years of age; and 90% of women identified with cervical disease receive treatment (90% of women with pre-cancer treated; 90% of women with invasive cancer managed). 26
Limitations
This was a retrospective review of targeted literature which is accompanied by some limitations. No evaluation of the effects of vaccination after any HPV-related treatment was made. A clear recommendation on how to implement HPV vaccination in a country should be required. It is important to note that the literature search was done using one database rather than multiple. This could have led to a greater number of included studies in addition to more significant economic analyses. Though more current studies were used, by eliminating any studies older than 2012 allowed analysis of more current data and of methods that have advanced since the introduction of the HPV vaccine in 2006. 7 Though, additional studies within the peer-literature review include studies prior to 2006. Additionally, the strain targeted from each HPV vaccine was not thoroughly discussed as the aim of this review was to analyze the overall effectiveness of the HPV vaccine. For example, some studies specified the vaccine they used was the nonovalent vaccine, whereas others did not specify the strain. Further research can be done to analyze the effectiveness of the HPV vaccine across different virus strains. Majority of reviews on HPV vaccine research conducted on LMICs are conducted from the developed countries hence bias should be considered. The inadequate vaccine coverage in LMICs is strong due to the lack or absence of health institutions to support such projects in these countries. Poverty, illiteracy, lack of stable political systems, taboos against vaccination, are all challenges against the success of HPV vaccine implementation in LMICs. A comprehensive health promotion is commonly required in areas with inadequate health services/access, rural areas, or populations with low awareness of health. Furthermore, only two databases (Pubmed and Google Scholar) were used to screen for studies. This may have limited information represented in this study.
Footnotes
Acknowledgements
The author would like to thank Dr. Nazia Rashid from Keck Graduate Institute for kindly providing guidance, additional information, and review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
