Abstract
Background
Several co-factors for HPV oncogenesis have been proposed, including co-infection with HSV-2. We assessed the relationship between HSV-2 infection and HPV-related outcomes in quadrivalent HPV-vaccinated (qHPV) women living with HIV (WLWH).
Methods
In this multi-site study of immunogenicity and efficacy of the qHPV vaccine in WLWH, visits took place at months −3, 0, 2, 6, 12, 18, 24, and annually thereafter. Participants provided clinical data and cervico-vaginal swabs for HPV DNA detection; baseline serum was tested for HSV-2 type-specific antibodies. We used non-parametric statistics to compare HPV-related outcomes by HSV-2 serostatus and use of anti-HSV medication.
Results
151 baseline serum samples underwent HSV-2 testing. At baseline, median age was 39 years, median CD4 count was 500 cells/mm3, and 70% had an HIV viral load of <50 copies/mL. Baseline HSV-2 seroprevalence was 76.2%. HSV-2 seropositivity was associated with increased age (p = 0.006). Controlling for age and median CD4 count, HSV-2 seropositivity was not associated with HPV incidence, persistence, and precancerous lesions. The use of anti-HSV medications was associated with higher odds of HSIL cytology (OR = 3.35, 95% CI = 1.03,11.26) and a greater number of HPV types detected (OR = 1.18, 95% CI = 1.00,1.39). Results were similar in sensitivity analyses using an index value of 3.5. The presence of HSV lesions during the study was not associated with HPV outcomes.
Conclusions
HSV-2 seropositivity was common in this cohort of WLWH in Canada but was not associated with multiple measures of HPV incidence, persistence, and precancerous lesions. However, the use of anti-HSV medications was associated with HSIL cytology and number of HPV types detected.
Keywords
Introduction
Human papillomavirus (HPV) disproportionately affects women living with human immunodeficiency virus (WLWH) compared to the general population. Prevalence of HPV is doubled in WLWH, with approximately 50% of WLWH experiencing HPV infection compared to approximately 25% in women without HIV. 1 As a result, WLWH consistently face adverse HPV-related outcomes, including higher rates of persistent HPV infection (24 vs 4%) and increased incidence of invasive cervical cancer (16 per 100 000 person-years vs 5 per 100 000 person-years).2–4 A strong association between HIV and HPV is present throughout the literature, with studies showing that advanced HIV may exacerbate the effects of HPV and lead to worse histological and cytological outcomes by suppressing the host immune response.5,6
HIV is also highly associated with herpes simplex virus 2 (HSV-2) infection. More than half of people living with HIV experience co-infection with HSV-2 and risk of HIV acquisition is nearly tripled among those who are HSV-2 positive.7,8 HIV and HSV-2 infections consistently co-occur and are thought to exacerbate one another.7,8 Mechanistically, HSV-2 increases the presence of activated CD4+ T cells and induces damage in the genital mucosa, thereby facilitating HIV entry and establishment of infection. 8 HIV infection contributes to worse HSV-2 associated outcomes through reduced immune functionality. This is evidenced by higher rates of subclinical HSV-2 shedding, as well as more frequent and severe genital lesions found in people living with HIV. 9 Co-infection with HIV also enables further HSV-2 transmission by enhancing genital shedding. 8
While the relationships between HPV and HIV, as well as HSV-2 and HIV, are well documented, the association between HPV and HSV-2 in the context of WLWH has been difficult to characterize. Some evidence indicates that HSV-2 co-infection is important for HPV oncogenesis.9–11 These studies postulate that HSV-2 may interfere with the host immune response and induce tumorigenesis in HPV-infected cells by amplifying the viral and oncogenic activity of HPV.9,10 However, findings in the literature have been conflicting, with some research showing little to no correlation between HSV-2 and cervical oncogenesis.12-14
In the present study, we sought to better understand the role of HSV-2 in HPV-related outcomes within a cohort of quadrivalent HPV-vaccinated WLWH. Both serostatus and use of anti-HSV medication were used to explore the impacts of HSV-2 infection, as treatment with suppressive HSV therapy may suggest more active disease. Co-infection with HSV-1 was not investigated in this study. Though HSV-1 is an important cause of genital herpes, there has been minimal evidence to suggest that it plays a role in cervical oncogenesis. 8
Materials and methods
As part of the CTN 236 HPV in HIV Study, 420 girls and WLWH aged nine and older were recruited from 14 clinics across Canada between 2008–2012 and were followed for up to eight years. All participants provided informed consent and ethics approval for national study coordination was received from the University of British Columbia Clinical Research Ethics Board (approval H14-02,364). Doses of quadrivalent HPV vaccine were administered at months 0, 2, and 6, and visits were scheduled for months −3, 0, 2, 6, 12, 18, 24, and annually thereafter. To determine cervical cytology, cervico-vaginal samples were collected at months 0, 6, 12, 18, 24, 36, 48, 60, 72, 84, and 96 and assessed using ThinPrep® liquid-based cytology processed at the British Columbia Cancer Agency Cervical Cancer Screening Laboratory. Findings were reported in accordance with the Bethesda criteria. Participants were referred for clinical colposcopy as per the clinical care recommendations in their region. Histology results were abstracted from pathology reports. Aliquots from cytology samples were collected in PreservCyt®, processed, and PCR tested as described previously for the detection of 36 HPV genotypes using the Linear array assay.15,16 The following HPV types were tested for using this method: 6, 11, 16, 18, 26, 31, 33, 34, 35, 39, 40, 42, 44, 45, 51, 52, 53, 54, 56, 58, 59, 61, 62, 66, 67, 68, 69, 70, 71, 72, 73, 81, 82, 83, 84, and CP6108. Pelvic examination was performed on participants who were post-menarchal and sexually active at months 0, 6, 12, 18, 24, 36, 48, 60, 72, 84, and 96. Clinical data, including presence of visible lesions clinically deemed to be caused by HSV-2, were collected on all participants. To assess the impact of HSV-2 serostatus on clinical and pathologic outcomes, all available baseline serum samples taken at month 0 were tested for HSV-2 serostatus using the Focus HerpeSelect-2 enzyme immunoassay. As this was an exploratory analysis, all available samples were used. Samples with an index value >1.1 were considered seropositive for HSV-2 as per the manufacturer’s recommendations.
Girls aged <16 were removed from analysis due to lack of pelvic examination in this group. Univariate analyses were conducted using Welch’s T-test, Fisher’s Exact test, and Wilcoxon Rank Sum test to determine associations between each marker of HSV-2 (baseline serostatus, lesion presence [yes/no during the study], and anti-HSV medication) and clinical variables, including age, region of origin, total lifetime sexual partners, baseline CD4+ T cell count, CD4+ T cell nadir, and HIV viral load suppression (<50 copies/mL). Use of anti-HSV medications included both chronic and episodic use of acyclovir, valacyclovir, or famciclovir. Controlling for age and median CD4 count, a separate logistic regression model was performed to compare each HPV-related outcome, including HPV persistence, clearance of incident HPV, number of HPV types detected during the study (across follow-up), presence of high-grade squamous intraepithelial lesion (HSIL) cytology at least once during the study, and cervical intraepithelial neoplasia of grade 2 and higher (CIN2+) at least once during the study, according to three measures of HSV infection or treatment: HSV-2 serostatus, HSV lesion presence, and use of anti-HSV medication. Median CD4 count was adjusted for to account for levels of immunosuppression, which could play a role in facilitating HPV outcomes. Persistent HPV infection was defined as the detection of the same incident HPV type in samples collected at two or more consecutive study visits (>6 months apart) or detection of an HPV type at the last available visit. 17 Clearance of incident HPV was defined as the absence of an HPV type that was incident in the previous visit ≥6 months prior. Subsequently, we conducted a sensitivity analysis to determine whether findings differed when the HSV-2 index value cut-off was increased to ≥3.5.
Results
Participant characteristics by HSV-2 status (n = 151), n (%) or median (IQR).
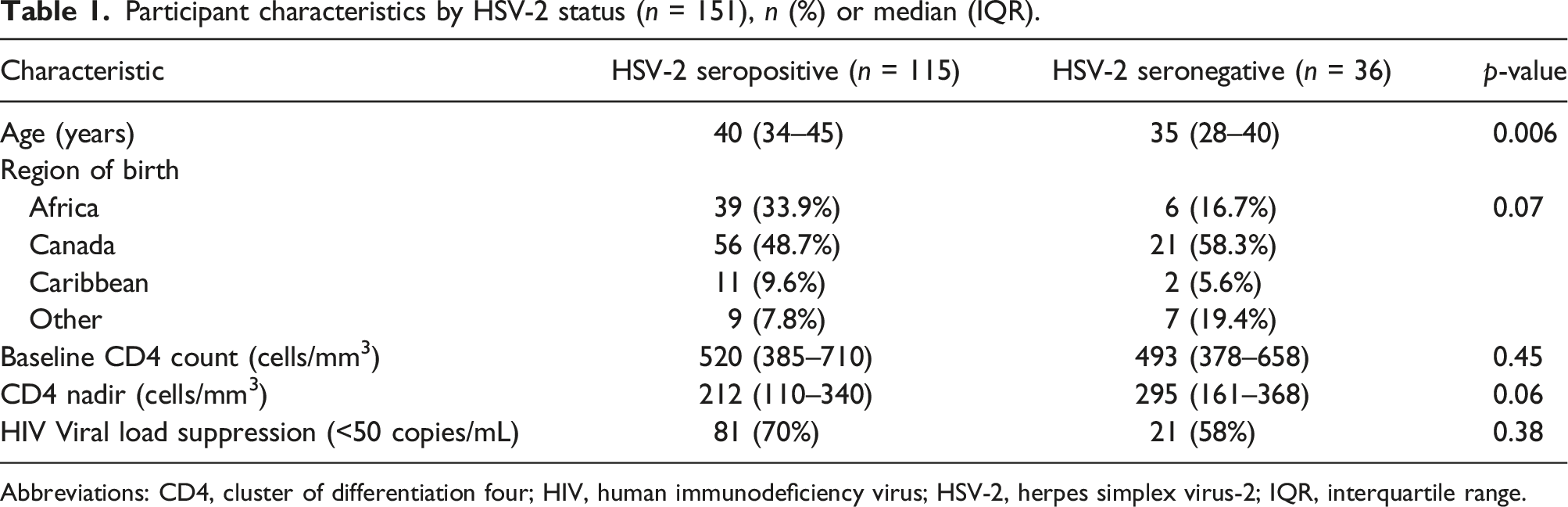
Abbreviations: CD4, cluster of differentiation four; HIV, human immunodeficiency virus; HSV-2, herpes simplex virus-2; IQR, interquartile range.
HPV-related outcomes by HSV-2 serostatus and use of anti-HSV medication (n = 151), n (%) or median (IQR).

Abbreviations: CI, confidence interval; CIN2+, cervical intraepithelial lesion of grade 2 or higher; HPV, human papilloma virus; HSIL, high-grade squamous intraepithelial lesion; HSV-2, herpes simplex virus-2; IQR, interquartile range.
aRate Ratio presented for number of HPV types.
bAdjusted for age and Median CD4 count.
*p-value < 0.10, **p-value < 0.05.
Discussion
The aim of this exploratory study was to assess the relationship between markers of HSV-2 infection and HPV-related outcomes, including HPV incidence and persistence among WLWH. Notable differences in HPV-related outcomes, when compared by HSV-2 serostatus, were not seen in this population of women. Conclusions were unchanged in sensitivity analyses in which we used a higher cutoff for defining HSV-2 seropositivity. Despite these findings, we did see significant differences in number of HPV types detected and prevalence of HSIL cytology when stratifying by use of anti-HSV medication.
While our cohort did not demonstrate notable differences in HPV-related outcomes when compared by HSV-2 serostatus, there are several possible explanations for these findings. First, HSV-2 seropositivity does not necessarily indicate active disease or viral activity. Twenty-eight percent of the study population had received either chronic or episodic suppressive HSV therapy and when compared against the rest of the cohort, odds of HSIL cytology and number of HPV types detected was significantly higher in this group. As such, we hypothesize that anti-HSV medication use may be a stronger indicator for active disease than serostatus, and therefore may have greater correlation with HPV-induced cervical oncogenesis. These findings should be interpreted with caution, as we had low numbers of HPV types and HSIL cytology detected during the study, as well as wide confidence intervals for these measures. It is also likely that some patients in this cohort were on suppressive HSV therapy to manage other HSV infections, so we cannot assume that use of medication was equivalent to HSV-2 reactivation. Furthermore, this analysis had a limited sample size and high proportion of seropositive individuals (only 36 participants were seronegative), which may have precluded our ability to detect significant differences by serostatus. To draw more definitive conclusions, larger sample sizes in both comparison groups would be required. Median age was also 5 years older in the HSV-2 seropositive group and HIV viral suppression rates were higher. Both of these variables are known to be important predictors of HPV-associated outcomes. Matching by age and HIV viral load suppression across groups would be ideal to account for confounders such as differences in disease course and management. Finally, all study participants received three doses of the quadrivalent HPV vaccine and therefore had low rates of HPV-related outcomes such as HPV incidence and persistence, HSIL cytology, and CIN2 + histology. This may have limited our ability to detect prognostic differences between the HSV-2 seropositive and seronegative group.
Of note, the HSV-2 seroprevalence in this cohort was high at 76.2%. This is consistent with other studies of HSV-2 seroprevalence among people living with HIV, suggesting that HSV-2 is very common in this population.18,19 Additionally, the literature supports that HSV-2 seropositivity is higher in women, and in sub-Saharan Africa where 30% of our participants had immigrated from.18–20 The correlation we documented between HSV-2 seropositivity and older age, as well as lower CD4 nadir, is also consistent with findings from prior studies.18,20
Our study emphasizes the significant burden of co-infections among WLWH in Canada. In a cohort of 151 WLWH, 74.8% had persistent HPV infections and 76.2% were seropositive for HSV-2. Moreover, 21% of HSV-2 seropositive participants had active symptomatic HSV-2 infections at least once over the course of the study. These high rates of stigmatizing and physically damaging viral infections highlight the need for increased screening, education, and care management within this population.
Strengths of this analysis include the longitudinal follow up of a national cohort of WLWH and use of standardized assessments for HPV outcomes. Our findings were reinforced by including a second analysis in which the cutoff for HSV-2 seropositivity was increased to an index value of ≥3.5, which has demonstrated improved sensitivity compared to the manufacturer cutoff of 1.1. 21
For decades, HSV-2 has been suggested as a possible cofactor of cervical oncogenesis, but this remains a subject of debate today. In this cohort of WLWH, HSV-2 seropositivity was common but not associated with multiple measures of HPV incidence, persistence, and precancerous lesions. However, chronic and episodic use of suppressive HSV treatment was correlated with detection of a greater number of HPV serotypes and increased HSIL cytology, suggesting that active disease rather than serostatus may be an important indicator for HPV viral activity and the development of precancerous lesions. Our findings add important data to the literature regarding HSV-2 infection in HPV-vaccinated WLWH. Further study of the role of active HSV-2 infection is required to determine if these results are representative of the larger population of HPV-vaccinated WLWH.
Footnotes
Acknowledgements
The authors would like to thank the participants without whom this research would not be possible. The authors would like to acknowledge the CTN 236 HPV in HIV Study Team, in alphabetical order: Ariane Alimenti, MD (University of British Columbia), Arezou Azampanah, MSc (Women’s Health Research Institute), Ari Bitnun, MD (University of Toronto), Sandra Blitz, MSc (University Health Network), Jason Brophy, MD (University of Ottawa), Jan Christilaw, MD (University of British Columbia), Andrew Coldman, PhD (British Columbia Cancer Agency), Simon Dobson, MD (Vaccine Evaluation Centre), Laurie Edmiston (Canadian AIDS Treatment Information Exchange), Catherine Hankins, MD, PhD (Amsterdam Institute for Global Health and Development), Christos Karatzios, MD (McGill University Health Centre), Mel Krajden, MD (British Columbia Centre for Disease Control), Normand Lapointe, MD (CHU Sainte Justine), Nancy Lipsky, BA (Women’s Health Research Institute), Jessica McAlpine, MD (University of British Columbia), Dianne Miller, MD (University of British Columbia), Dirk van Niekerk, MD (British Columbia Cancer Agency), Gina Ogilvie, MD, DrPH (University of British Columbia), Neora Pick, MD (University of British Columbia), Lindy Samson, MD (University of Ottawa), Julie van Schalkwyk, MD (University of British Columbia), David Scheifele, MD (Vaccine Evaluation Centre), Joel Singer, PhD (CIHR Clinical Trials Network), Sarah Stone, MD (British Columbia Centre for Excellence in HIV/AIDS), Gavin Stuart, MD (University of British Columbia), Marcie Summers (Positive Women’s Network), Laura Vicol, MN, NP (University of British Columbia), and Melissa Watt (Women’s Health Research Institute). The authors wish to thank all of the additional clinicians and research staff for their important contributions to participant enrollment and study visits.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EM has no conflicts to declare. DHST has received investigator-initiated research grants from Abbvie, Gilead Sciences and Viiv Healthcare, and he is a Site Principal Investigator for clinical trials sponsored by Glaxo Smith Kline. SB has no conflicts to declare. AA has no conflicts to declare. FC has received grants for research projects given to his institution from Roche Diagnostics, Becton Dickenson, and Merck, Sharp, and Dome, honoraria for presentations from Merck, Sharp, and Dome and Roche Diagnostics, and has participated in an expert group by Merck, Sharp, and Dome, outside the submitted work. ML has received honoraria from Merck Canada Inc. SW has received grants, personal fees and non-financial support from Merck Canada Inc., ViiV Healthcare, Gilead, AbbVie, Janssen and Bristol Meyers Squibb for participation on advisory boards, presentations, meetings, studies, workshops and symposia for each, outside the submitted work. GO is a co-investigator on an investigator-led trial funded by Hologic Inc and Roche. DM has received grants from GSK and Merck Canada Inc. for conducting sponsored vaccine trials. She also reports grants from Novartis and Sanofi for conducting sponsored vaccine trials in an unrelated area. She has received personal fees for symposium participation from Merck Canada Inc., outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Le Rseau sida et maladies infectieuses, Michael Smith Foundation for Health Research (Trainee Award), Canadian HIV Trials Network, Canadian Institutes of Health Research, Merck Canada, Canadian Foundation for AIDS Research, Canada Research Chairs, Ontario HIV Treatment Network, Canadian Institutes of Health Research (MOP 136784).
