Abstract
Background
The increasing prevalence of drug-resistant Neisseria gonorrhoeae (NG) infections has caused great concern. Ciprofloxacin remains the empiric antimicrobial recommended to treat NG infections in Peru disregarding the susceptibility profile of circulating NG strains. We report the prevalence of individuals infected with NG strains presenting mutations in the gyrA gene that confers ciprofloxacin resistance.
Methods
We conducted a descriptive study assessing extragenital swab samples collected from a cohort of men who have sex with men and transgender women in Lima, Peru. Anal and pharyngeal NG positive swabs for Aptima Combo 2 assay (Hologic Inc., USA) were used for DNA extraction. We performed TaqMan real time PCR assays to detect a point mutation at codon Ser91 of the gyrase A (gyrA) gene.
Results
From 156 individuals who had at least one positive sample for NG reported by the Aptima assay, 80 individuals had at least one amplified DNA for the gyrA gene. We found that 67 of them (84.0%) were infected with a gyrA-mutated NG strain at the Ser91 codon.
Conclusions
We report a high prevalence of gyrA mutation conferring ciprofloxacin resistance among individuals with extragenital NG infection. Empirical treatment of NG needs to be urgently updated in Peru in concordance with international guidelines.
Introduction
Neisseria gonorrhoeae (NG), etiological agent of gonorrhea, is the second most common bacterial sexually transmitted infection (STI), with an estimated incidence of 82.4 million new cases among people aged 15–49 years worldwide in 2020. 1 Key populations such as men who have sex with other men (MSM) and transgender women (TW) have a disproportionate burden of gonorrhea globally. 2 In Peru, the prevalence of gonorrhea in general population was around 0.1%; 3 while, prevalence of extragenital NG infections among MSM and TW is estimated at up to 9.6%. 4
NG has developed resistance to most antimicrobials that have been used to treat it over the last 80 years. 5 Because of the sustained increase of quinolone resistance, the CDC stopped recommending ciprofloxacin as the empiric treatment for NG infections more than 10 years ago. 6 However, the use of ciprofloxacin is useful if the sensitivity of NG to this antibiotic is known through antimicrobial susceptibility testing. 7 In Peru, ciprofloxacin remains the recommended treatment for NG infections, regardless of antibiogram test results. 8 There have been few studies reporting the presence of NG ciprofloxacin resistance in Latin American countries, 9 especially in Peru,10–13 where recently a study reported that 66 (94.3%) of 70 culture isolate of NG infections from MSM were ciprofloxacin resistant. 13 In addition, a systematic review confirmed that detection of the Ser91 codon mutation in the gyrase A (gyrA) gene, by molecular techniques like quantitative real-time polymerase chain reactions (qPCR), predicts NG susceptibility to ciprofloxacin (sensitivity and specificity >98%). 14
Because NG infections among MSM and TW have frequently occurred in anal and pharyngeal locations, and most of these cases are asymptomatic, undiagnosed, and untreated, the high prevalence has continued among them. 2 Moreover, NG strains circulating in these populations were described as having increased antimicrobial resistance and rapid spread. 15
In this study, we aimed to determinate the gyrA genotype in stored NG swabs samples to better document the prevalence of NG mutations conferring ciprofloxacin resistance among a cohort of MSM and TW in Lima, Peru.
Methods
Clinical swab specimens from pharyngeal and anal anatomic locations were collected quarterly during 24 months between June 2013 and July 2016, as part of the PICASSO cohort, a study that aimed to estimate the incidence and characterize the syphilis infection under social, diagnostic, immunological and molecular determinants, among MSM and TW in Lima, Peru. More details of the cohort methods were described previously. 16 In summary, participants were recruited from two STI clinics and were included if they were ≥18 years old and had at least three of the following high-risk criteria for acquiring syphilis: (1) positive rapid test of syphilis in the last 2 years, (2) being HIV seropositive, (3) having a genital ulcer, (4) having any STI diagnosis in the last six months, (5) 5 years of sexual activity at minimum, (6) five or more sexual partners in the last 3 months, (7) five or more episodes of condomless anal intercourse in the last 6 months. 17
Participants completed a computer-based survey assisted by a trained interviewer at baseline and at each follow-up visit. They reported socio-demographic characteristics, sexual behavior, history of previous and current STIs, and alcohol and substance use. After receiving HIV/STI pre-test counseling, they were referred to the laboratory to provide venous blood samples for HIV and syphilis testing, a pharyngeal swab specimen collected by the staff, and to receive instructions for self-collection of an anal swab specimen for Chlamydia trachomatis (CT) and NG testing. Screening for HIV and syphilis was performed at the clinic sites, using a third-generation rapid test (Alere Determine™ HIV 1/2, Alere Inc., USA), and a nontreponemal Plasma Reagin Rapid (RPR) test (BD Macro-Vue™ RPR Card Test Kit, Becton Dickinson, USA), respectively.
Aliquots of serum and swab specimens were transported maintaining cold chain to the Laboratory of Sexual Health at Universidad Peruana Cayetano Heredia in Lima and stored at −20°C until processing. Confirmatory HIV testing was performed using the EIA fourth generation test (Genscreen™ ULTRA HIV Ag-Ab, Bio-Rad, USA) and Western Blot test (NEW LAV BLOT I, Bio-Rad, USA). For syphilis confirmation, the Treponema pallidum particle agglutination (TPPA) test (Serodia, Fujirebio Diagnostics Inc., Japan) was used. A recent syphilis infection was considered if the RPR title was ≥1:16 and the TPPA test was positive. Finally, NG and CT detection was done using a transcription mediated amplification (TMA) assay (Aptima Combo 2 CT/NG, Hologic Inc., USA). All participants received the corresponding treatment if they tested positive for any STI, and were referred to the national HIV treatment program in case of confirmed HIV new infection.
The PICASSO study enrolled 401 participants. 17 The prevalence of anal and pharyngeal NG infection at baseline was 9.0% and 6.2%, respectively, while the incidence rate of anal and pharyngeal was 28.1 and 21.3 cases per 100 person-years, respectively. For this estimate, incident cases were defined as a newly positive NG Aptima test during follow-up with no previous infection or documented treatment of a previous infection. 18
Primers and probes.
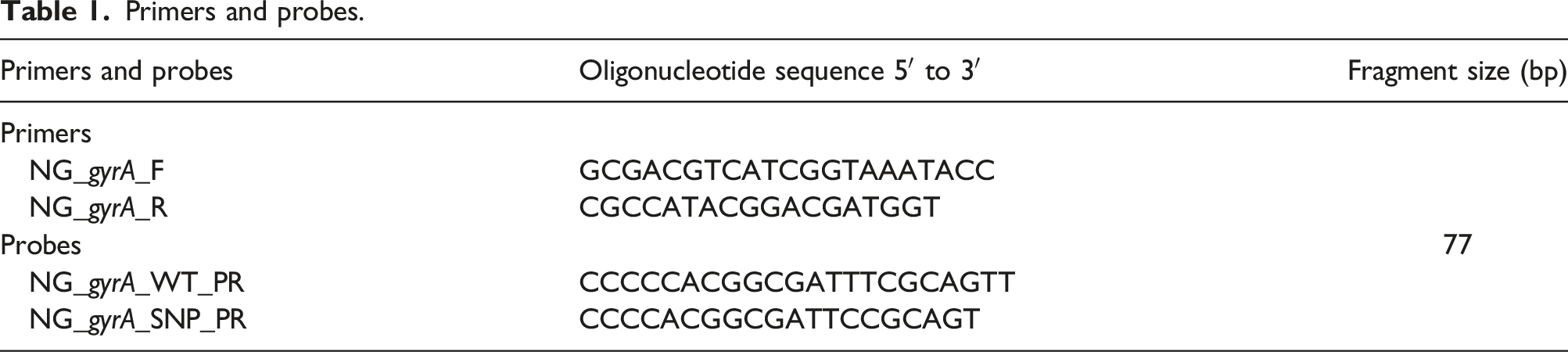
Master mix was prepared using iTaq™ universal probes supermix (Bio-Rad Laboratories, Inc., USA), with a concentration of 0.12 μmol for each primers, 0.15 μmol of WT probe, 0.10 μmol of SNP probe and 5 μL of DNA template for a final volume of 20 μL. Thermal cycler CFX96 (Bio-Rad Laboratories, Inc., USA) was used for amplification with the following conditions: 95°C for 5 min, 45 cycles of 95°C for 30 s and 60°C for 60 s; also, we used the fluorescent channel: FAM (510–530 nm) for WT probe and VIC (560–580 nm) for SNP probe. After amplification, probe curves were analyzed using Bio-Rad CFX Manager software version 2.0. Samples were considered to have the gyrA mutation if their cycle of quantification (Cq) were below 40 cycles and their value of relative fluorescence units (RFU) were higher than 480 and 380, which were the background fluorescence for both FAM and for VIC probe, respectively. For the analysis, an individual was considered NG–infected when there was at least one Aptima-positive sample (either anal or pharyngeal) in any follow-up visit, and was included for gyrA mutation assessment if follows the Cq and RFUs already describe criteria.
Bacterial load was estimated for each amplified anal sample by real time qPCR, according to the standard curves. These were estimated using five serial dilutions from 105 to 101 genome copies/μL of WT and mutant strains by calculating the mass of the NG genome using: m = (n) (1.096 × 10−21 g/bp), according to Applied Biosystems protocol. 20 The genome size (n) of NG considered was 2′153,922 bp (NC_002946.2, NCBI). Standards were run in each qPCR assay (in triplicate) only with WT or mutant samples (in duplicate) using the corresponding probe and a correlation coefficient (R2) greater than 0.89.
For statistical analysis, we described the medians and proportions of socio-demographic characteristics, previous and current STIs diagnosed, antibiotic use prior to diagnosis (at baseline), and bacterial load of anal samples from individuals with an available PCR result for gyrA gene. We also compared these variables according to gyrA mutation pattern using Fisher exact test for categorical variables and Mann–Whitney U test for continuous variables. A p-value of less than 0.05 was considered statistically significant. All data analysis was conducted using the Stata software (version 15.0, Stata Corporation Inc., USA).
Results
Characteristics, gyrA mutation pattern, and bacterial load of MSM and TW infected with a gyrA wild type or Ser91codon mutant strain of Neisseria gonorrhoeae.
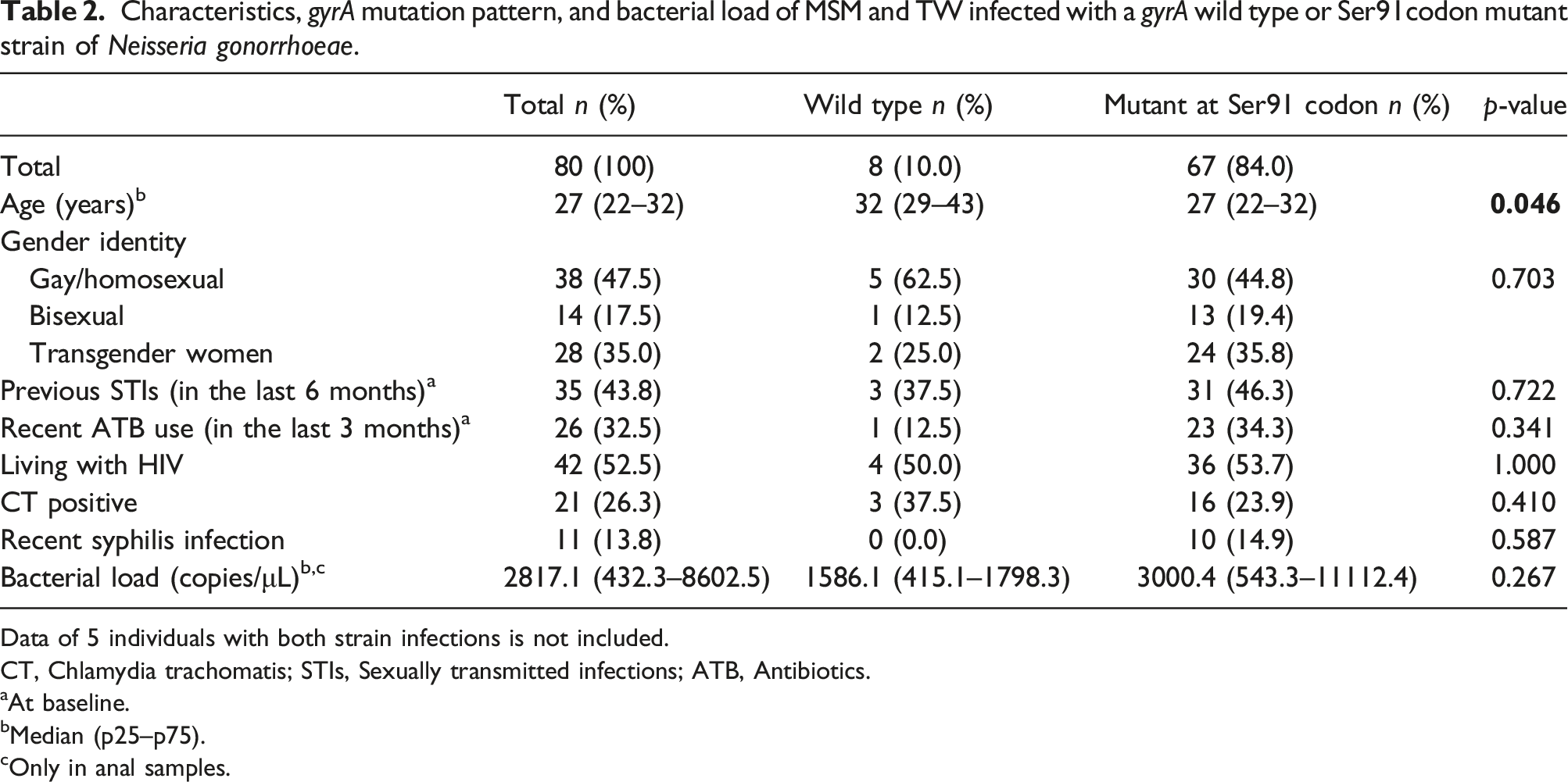
Data of 5 individuals with both strain infections is not included.
CT, Chlamydia trachomatis; STIs, Sexually transmitted infections; ATB, Antibiotics.
aAt baseline.
bMedian (p25–p75).
cOnly in anal samples.
Regarding gyrA gene NG pattern, we found that 67 individuals (84.0%) had at least one sample with a mutation at Ser91 codon of gyrA gene NG strain, 8 (10.0%) had a WT NG strain, and 5 (6.0%) alternated between WT and mutant NG infections during follow up in the same anatomical site (these infections were detected between 3 and 21 months apart). Among the 67 individuals infected only with mutant NG strains, 10 (14.9%) were diagnosed with a recent syphilis infection, 36 (53.7%) were people living with HIV, 31 (46.3%) reported previous STIs in the last 6 months, and 23 (34.3%) used antibiotics in the 3 months prior to their enrolment visit; however, there were no statistically significant differences compared to individuals with a WT NG strain. Additionally, the median bacterial load in subjects with mutant NG in any anal sample was higher than WT NG; however, it was not statistically significant (3000.4 copies/μL versus 1586.1 copies/μL, p = 0.267).
Discussion
We found an 84% prevalence of NG infections with mutations in the Ser91 codon of the gyrA gene of stored NG-positive specimens collected from extra-genital anatomic sites among MSM and TW in Lima, Peru. Sanchez et al. found that 24% of MSM diagnosed with NG between 2012 and 2013, were infected with an NG strain which had a mutation in the quinolone resistance-determining region of the gyrA gene. 10 Differences could be explained by the differences in study populations. Our study was performed among a population at high-risk for syphilis, which may reflect similar higher risk for antimicrobial-resistant NG and these could explain why the presence of gyA-mutant NG strains is higher in our study. Recently, Jorge-Berrocal et al. 13 reported a 95.2% prevalence of ciprofloxacin-resistant NG culture isolates as a part of a Peruvian NG sentinel surveillance system. Certainly, all of these estimates are sufficiently high to call into question the overall effectiveness of using ciprofloxacin as empiric therapy for management of NG infections in Peru.21,22
In our study, recent antibiotic use was reported by 34.3% of individuals with mutant NG infection, compared with 12.5% of individuals with WT NG. Studies have shown that self-prescribed antibiotics and recent antibiotic use are associated with quinolone-resistant NG strains;23,24 moreover, a high prevalence of self-prescribed antibiotics has been reported in our country. 25
In anal samples, we found that relative bacterial load in mutant NG infections was twice the value of WT NG, although it was not statistically significant. In vitro studies reported that bacterial fitness could be enhanced by point mutations related to quinolone resistance, increasing bacterial load; 26 which could facilitate the transmission of NG-resistant strains. 27
Our study reported no difference in the proportion of HIV-positive patients between gyrA profiles. Living with HIV has been associated with an NG resistance profile, although reports have been inconclusive. 28 However, screening for extragenital gonorrhea infections among high-risk groups should be implemented in the context of HIV transmission and acquisition.
The main limitation of this study was that almost half of the samples did not amplify by PCR assay, especially the samples from the pharyngeal location, since despite the high number of positive results by Aptima Combo 2 assay, we obtained few pharyngeal samples to assess for gyrA mutation analysis which may have affected our results, and may be caused by low NG bacterial load in this anatomical site, the buffer in which the sample was resuspended (might not be appropriate for the PCR assay) or other PCR inhibitors from commensal Neisseria species.29,30 Importantly, prior studies have documented a higher prevalence of antimicrobial resistance among pharyngeal reservoirs; 31 thus our findings may underestimate the prevalence of ciprofloxacin resistance.
In this study, we documented a high prevalence of gyrA mutations conferring ciprofloxacin resistance among extra-genital NG infections. This finding may warrant a change in the empirical treatment of NG infections. Recently, the Sentinel Surveillance Group of the Peruvian National Institute of Health (promoter of public health policies and norms) does not recommend the use of fluoroquinolones; 13 however, the National Guidelines for STIs Management have not yet been updated since 2009. In addition, in key populations, extragenital screening and the use of molecular genetic markers to identify antimicrobial susceptibility should be considered in order to detect asymptomatic NG infection early, provide the correct treatment, and interrupt the chain of transmission.
Footnotes
Author Contributions
All authors listed on this manuscript certify that they have participated sufficiently in the work to take public responsibility for the content, including participation in the concept, design, analysis, writing, or revision of the manuscript.
Declaration of conflicts of interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JDK reports consulting fees from Danaher, Cepheid, Talis Bio, Curative, Abbott, GSK, Roche, Chembio, and SpeeDx. KAK reports employment at both UPCH and UCLA. JQ, SKV, ME, FV, DD, LTAB, TJO, CFC.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Allergy and Infectious Diseases (NIAID; R01AI099727-01) and Team Klausner Saving Lives.
