Abstract
Background
Routine opt-out HIV testing in healthcare settings is often not implemented to its fullest extent. We assessed factors contributing to missed HIV testing opportunities at an academic medical center in Chicago, Illinois, with a routine HIV screening program.
Methods
Retrospective analysis of HIV testing in clinical encounters was performed using multivariate regession models. Missed opportunities were defined as 1) an encounter during which an HIV test was not conducted on a patient later diagnosed with HIV, or 2) an encounter in which a bacterial STI test was performed without HIV testing.
Results
Of 122 people newly diagnosed with HIV from 2011-2018, 98 patients had 1215 prior encounters, of which 82.8% were missed opportunities. Female gender, persons not known to be men who have sex with men, and encounter location other than inpatient had higher odds of a missed opportunity. Nearly half (48.4%) of 104,678 bacterial STI testing encounters were missed opportunities. Female gender, older age, lack of syphilis testing, and location outside the emergency department had higher odds of a missed opportunity.
Conclusions
We found a high number of missed HIV testing opportunities, which could be reduced by strengthening routine screening and increasing targeted testing concurrent with STI screening.
Keywords
Introduction
Routine screening for human immunodeficiency virus (HIV) is recommended for all individuals aged 13–64 years once in a lifetime and at least annually for persons at risk for infection per the United States Centers for Disease Control and Prevention (CDC).1,2 In addition, all persons presenting for sexually transmitted infection (STI) evaluation are recommended to undergo HIV screening regardless of other risk factors and past HIV testing. 3 Despite this policy, an estimated 13% of people living with HIV (PLWH) remain unaware of their diagnosis in the United States (US), and late diagnosis of HIV remains too common.4–7 This delay to diagnosis and care has detrimental consequences to the well-being of individual patients and public health; early diagnosis, linkage to care, and initation of antiretroviral therapy result in improved morbidity and mortality for PLWH as well as decreased community viral load and less transmission.8–10
Several studies have identified socioeconomic, age-related, and comorbidity-related factors that are associated with missed opportunities for diagnosis of HIV in resource-rich settings in North America.5,6,11–15 One factor that has been shown to contribute to missed opportunities is the lack of concurrent HIV testing in individuals being tested for other sexually transmitted infections (STIs). 16 In one study of patients presenting to an academic emergency department (ED), less than 4% of individuals tested for chlamydia or gonorrhea were concurrently tested for HIV. 17 Routine screening helps improve rates of HIV testing, but may not be fully implemented in healthcare settings or may not capture those who may need repeat testing over time due to changing or ongoing potential exposure.5,11,13,15 Routine HIV screening was initiated at University of Chicago Medicine (UCM) in 2011 as part of the public health-funded Expanded HIV Testing and Linkage to Care (xTLC) program, which has been described previously.18–20 The xTLC program supports routine healthcare-based HIV testing and timely linkage to care. While the program initially used an opt-in model focused on annual screening in the ED, after legislative changes in 2016 it changed to an opt-out model and expanded to include once in a lifetime HIV screening in outpatient primary care clinics and inpatient settings.
UCM’s catchment area is at the intersection of three neighborhoods in Chicago that consistently rank among the community areas with highest HIV incidence rates in the city. 21 While HIV incidence in Chicago has decreased, HIV disproportionately affects certain populations, including men who have sex with men (MSM) and Black communities. 21 Late diagnoses account for 18.8% of new HIV diagnoses between 2014 and 2019 in Chicago.21,22 Understanding patterns of HIV testing in healthcare settings, and missed opportunities for testing, is important as we work to improve early diagnosis in PLWH. In this study we assess sociodemographic and clinical factors associated with missed opportunities for HIV testing at an academic medical center with a routine HIV testing program. In order to best understand opportunites to improve timely HIV diagnosis within our medical center, we assess missed opportunities in two cohorts: persons newly diagnosed with HIV who had a prior encounter in our healthcare system, and persons tested for bacterial STIs.
Methods
We performed a retrospective analysis using data extracted from UCM electronic medical records (EMR) for persons 18 years or older in two cohorts: 1) those newly diagnosed with HIV; and 2) those who underwent STI testing for chlamydia and gonorrhea (CT/GC), and/or syphilis. Data were provided by the Center for Research Informatics (CRI) at UCM. This study was approved by the Institutional Review Board of the Biological Sciences Division at the University of Chicago.
Cohort 1: missed opportunities for HIV testing among people newly diagnosed with HIV
We identified persons with a new diagnosis of HIV at UCM from 2011 to 2018 and examined all healthcare encounters from these patients, if any, at UCM in the five year period prior to their diagnosis. This resulted in the inclusion of encounters between 2006 and 2018 that were within the 5 years preceding each patient’s HIV diagnosis. This five year review period for encounters was chosen to be consistent with a recent study that used a similar approach. 5 Diagnosis of HIV was based on the CDC testing algorithm, 23 defined as a PLWH without a previously documented diagnosis of HIV or prior positive HIV test results. In addition, all new diagnoses were confirmed with the Chicago Department of Public Health by the xTLC linkage to care coordinator at the time of test result. All encounters following a confirmed HIV positive test were excluded, as were encounters from outpatient locations where diagnostic or screening tests are not typically ordered, such as physical therapy, occupational therapy, diagnostic radiology, and interventional procedures. An encounter during which an HIV test was not conducted was considered a missed opportunity. Encounters within six months of a prior HIV test, regardless of whether an HIV test was performed, were not deemed missed opportunities.
Sociodemographic variables included age, race, ethnicity, chart-identified gender, and zip code. Behavioral variables included alcohol use, illicit drug use, tobacco use, sexual activity, and sexual behavior. Encounter-level variables included location (ED, inpatient, or outpatient) and encounter year, dichotomized as 2006-2010 vs. 2011-2018. We chose 2011 as the cutoff for the dichotomization as this coincided with the implementation of UCM’s xTLC routine screening program. As encounter-level insurance data were unavailable, zip code was used to identify patients residing in high economic hardship community areas according to the 2010-2014 Chicago Community Area Economic Hardship Index. 24 Zip codes representing communities with a hardship index value of 45.1 or greater were considered to be high hardship community areas. 24 Alcohol use, illicit drug use, tobacco use and sexual activity were drawn from structured fields in the ‘social history’ section of the EMR. Patient sexual behavior (e.g. men who have sex with men (MSM)) was confirmed on chart review for all patients diagnosed with HIV. Patients were classified as not known to be MSM (Not MSM), known to be MSM at time of the encounter (Known MSM), or identified as MSM after HIV diagnosis (MSM Later Known). Chart review was performed to determine HIV pre-exposure prophylaxis (PrEP) use prior to HIV diagnosis for those diagnosed in 2012 or later. Finally, we defined late HIV diagnosis as an initial CD4 count <200 cells/mm3. 25
Cohort 2: missed opportunities for HIV testing in encounters with bacterial STI evaluation
Concurrent STI and HIV tests were examined for all STI testing encounters from 2011 to 2018. Encounters where bacterial STI testing was performed in the absence of a concurrent HIV test were considered missed opportunities, excluding encounters within 30 days of a previous HIV test. All encounters following a confirmed HIV positive test were excluded. Patient-level variables included age, chart-identified gender, race, and ethnicity, while encounter-level variables included year (examined as discrete 2-year intervals), location, STI test(s) performed (chlamydia & gonorrhea (CT/GC), syphilis, or all), and previous positive STI test results. We performed chi-square analysis of the proportion of missed testing opportunities in two-year intervals (2011-2012, 2013-2014, 2015-2016, and 2017–2018) to assess trends in missed testing opportunities over time in Cohort 2.
To model associations with missed opportunities for HIV testing we created bivariate and multivariate regression models using encounter-level data, which allowed for all encounters from a single patient to be included. In order to control for repeat encounters amongst individuals, Generalized Estimating Equation (GEE) models were used with an exchangeable correlation structure. All p-values are two-sided, and the level of significance was set at 5%. Associations were measured via odds ratios (OR) or adjusted odds ratios (aOR) and 95% confidence intervals (95% CI). Adjusted models apriori contained demographics (race, chart-identified gender, and ethnicity) as well as all factors identified as significant in bivariate analyses. All analysis was performed in SAS Version 9.4 (SAS Institute, Cary, NC).
Results
Cohort 1: missed opportunities for HIV testing in people newly diagnosed with HIV
Demographic information for patients newly diagnosed with HIV from 2011-2018.

Abbreviations: IQR, interquartile range; SD, standard deviation; MSM, men who have sex with men.
Gender is a binary male/female categorization drawn from the electronic medical record and represents the patient’s legal gender.
**Economic hardship was determined based on zip code of the patient, using the Chicago Community Area Economic Hardship Index 24 .
***Includes heterosexual men and all women.
Encounters of patients newly diagnosed with HIV from 2011-2018.
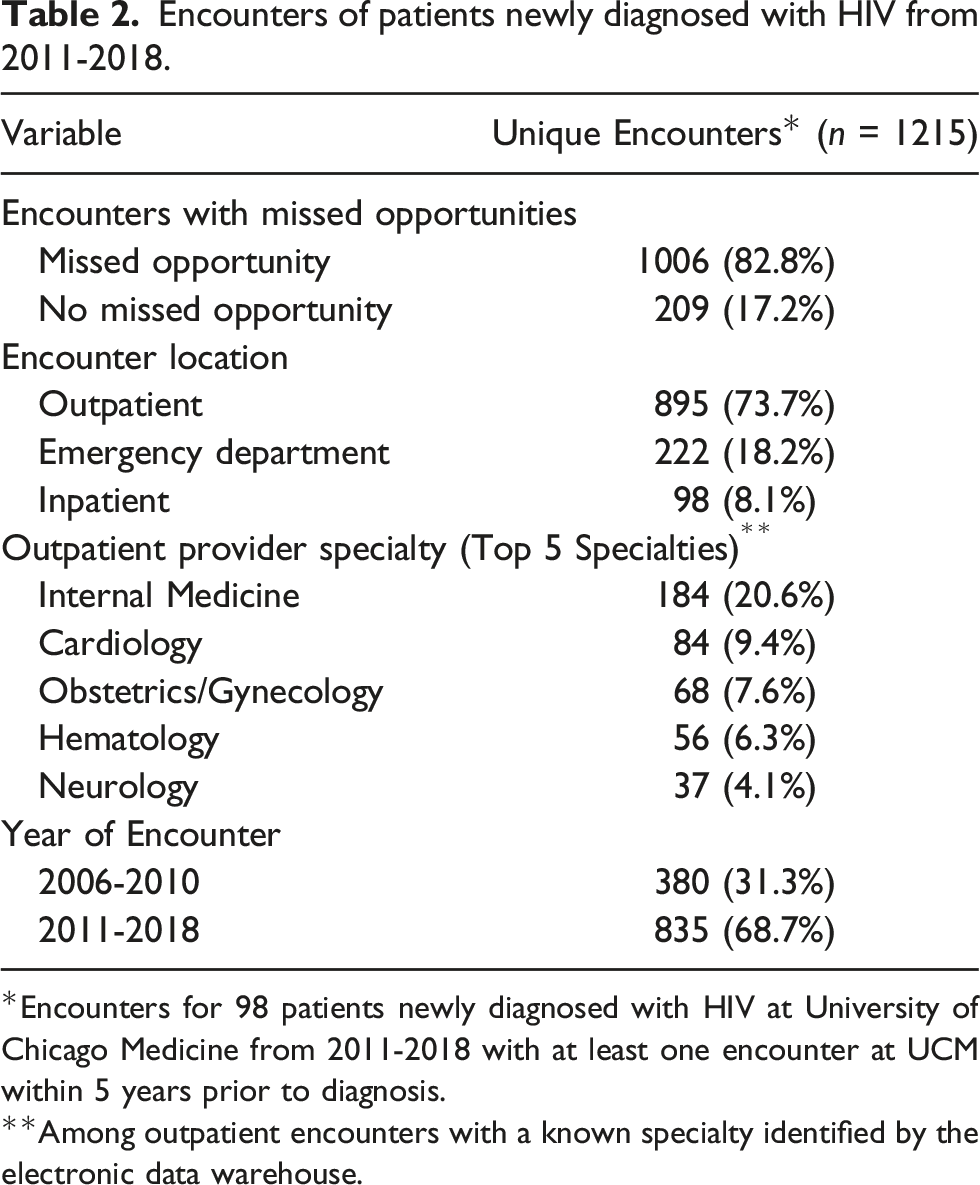
*Encounters for 98 patients newly diagnosed with HIV at University of Chicago Medicine from 2011-2018 with at least one encounter at UCM within 5 years prior to diagnosis.
**Among outpatient encounters with a known specialty identified by the electronic data warehouse.
Unadjusted & adjusted odds ratios for missed opportunities in persons newly diagnosed with HIV.
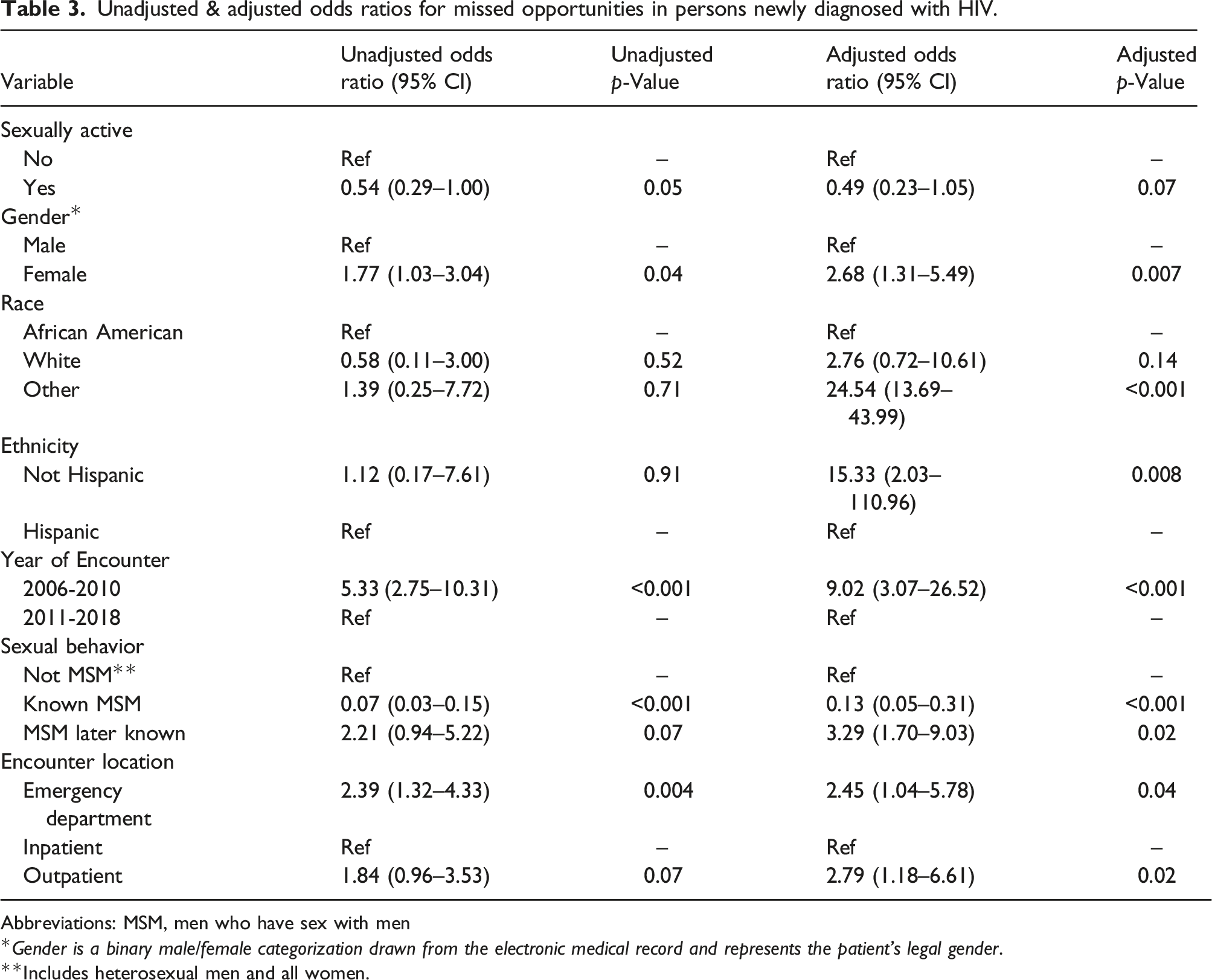
Abbreviations: MSM, men who have sex with men
Gender is a binary male/female categorization drawn from the electronic medical record and represents the patient’s legal gender.
Includes heterosexual men and all women.
Cohort 2: missed opportunities for HIV testing in encounters with bacterial STI evaluation
Patient demographics and STI testing encounter information, 2011-2018.
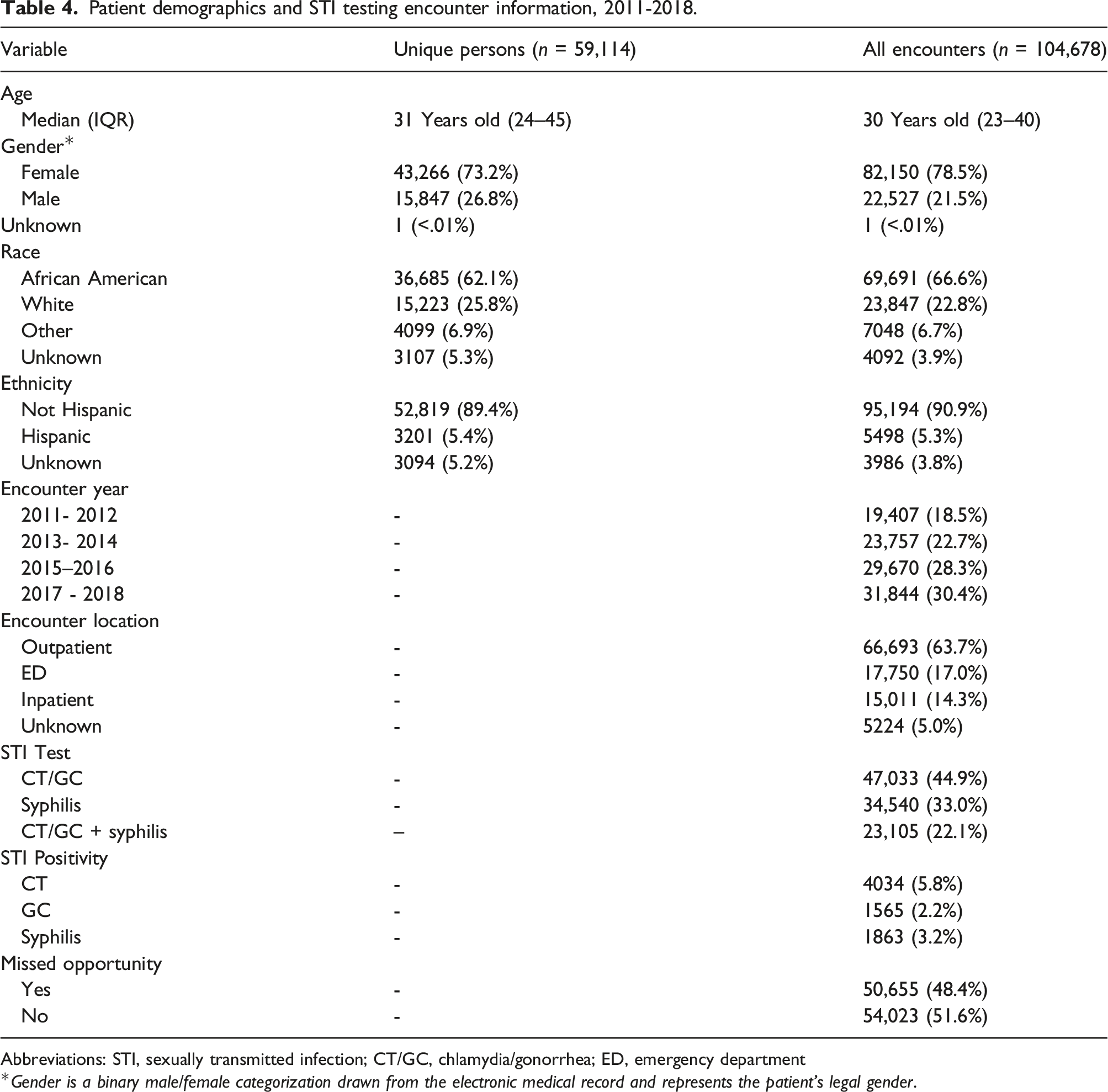
Abbreviations: STI, sexually transmitted infection; CT/GC, chlamydia/gonorrhea; ED, emergency department
Gender is a binary male/female categorization drawn from the electronic medical record and represents the patient’s legal gender.
Unadjusted & adjusted odds ratios for missed HIV testing opportunities in STI testing encounters.
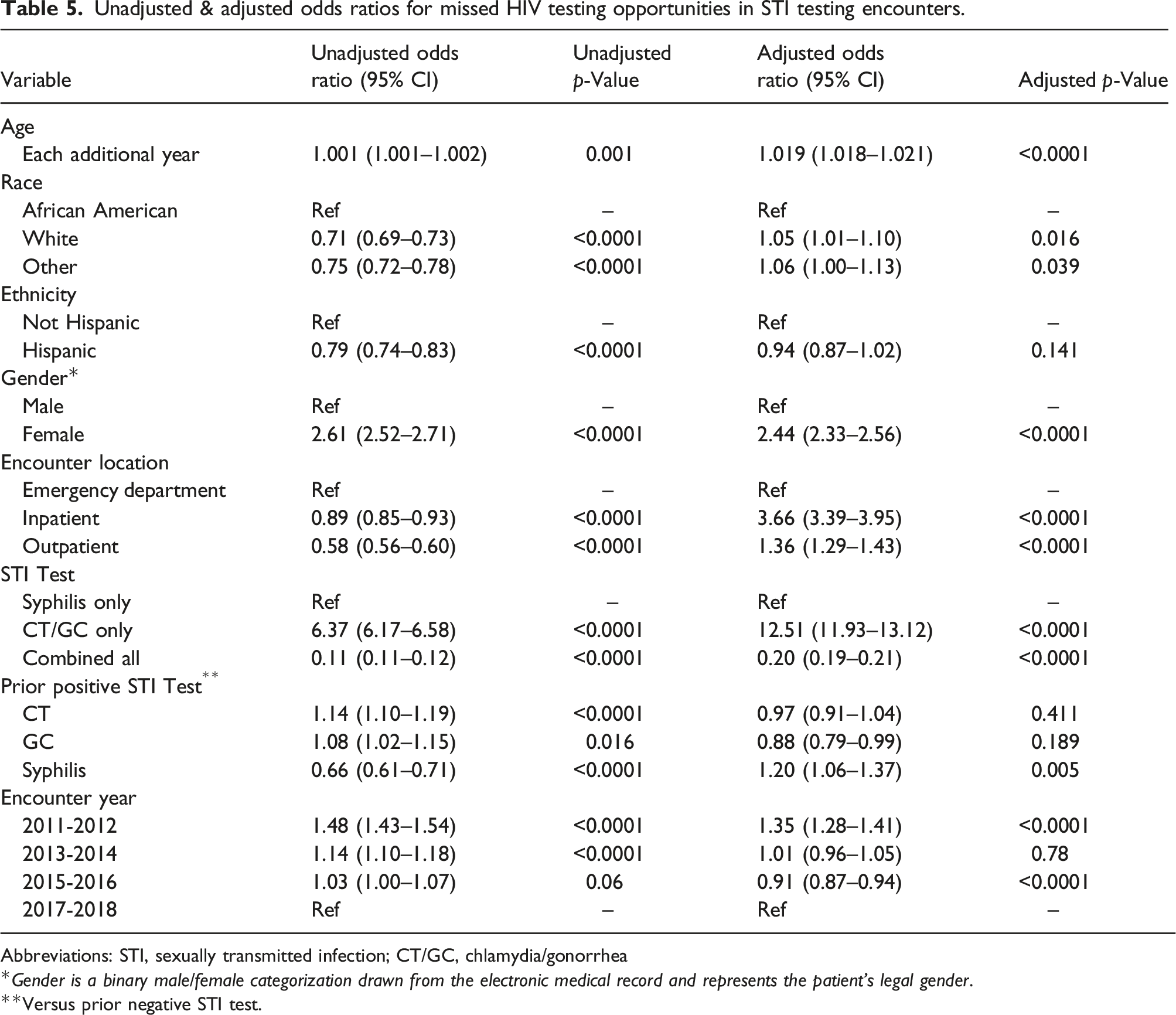
Abbreviations: STI, sexually transmitted infection; CT/GC, chlamydia/gonorrhea
Gender is a binary male/female categorization drawn from the electronic medical record and represents the patient’s legal gender.
Versus prior negative STI test.
Discussion
Our analysis includes two different methods to identify and characterize missed opportunities for HIV testing and earlier diagnosis. Overall, missed opportunities remained quite high during the time period assessed, and we found several associated factors. In newly diagnosed PLWH, 83% of prior encounters were missed opportunities, and late diagnosis of HIV was 28.7%, higher than the 23% reported in the US in 2014 and the 18.8% in the city of Chicago from 2014 to 2019.21,22,26 Targeting factors associated with missed opportunities in future health system initiatives may significantly improve early diagnosis and minimize late presentation of HIV, which is associated with significant morbidity and mortality. 27
Female gender was a strong risk factor for having a missed opportunity, consistent with other studies, 12 despite women comprising 30% of new HIV diagnoses at our health center and a quarter of new diagnoses in the US.7,20 Yet women remain an important population to engage in HIV testing and prevention efforts, particularly African American women who are disproportionately impacted by HIV. 28 Women may be at increased risk for a missed opportunity due to frequent asymptomatic CT/GC testing as part of routine gynecologic evaluations or due to clinician perception of lower risk. Further, we found that concurrent syphilis testing, which requires a blood draw, carried lower odds of a missed opportunity as compared to GC/CT testing alone; this may reflect a desire to avoid additional blood draws, which has been described as a barrier to HIV testing, particularly in younger persons. 29 Some routine HIV testing programs have linked the HIV screening to other orders requiring a blood draw, such as a complete blood count. 18 However, this limits the reach of routine testing and may be a particular limitation for patients presenting only for STI testing or routine gynecologic care.
In newly diagnosed PLWH, chart documentation of MSM sexual behavior was associated with lower odds of a missed opportunity. The majority of new HIV diagnoses in the US continue to be among gay and bisexual men, and physicians likely have awareness of this sexual behavior as a “can’t-miss” indication for HIV testing.14,15,17,30 However, failure to accurately obtain and/or document MSM sexual behavior in the EMR is a barrier to appropriate testing, as encounters in persons who were identified as MSM only after HIV diagnosis had higher odds of a missed opportunity. Sexual history taking remains a major weakness, 31 and some patients may also be reluctant to disclose sexual orientation, activity, and behaviors, 32 all of which may hinder understanding of current need for initial and repeat testing.
Encounters in later years (after 2011 in newly diagnosed PLWH and after 2012 in those evaluated for bacterial STI) were less likely to have a missed opportunity. Of note, we also examined dichotomizing encounters around 2016 (data not shown) in newly diagnosed PLWH (2006-2015, 2016–2018) and similar results were seen with the later period of encounters having a reduced risk of missed opportunities (unadjusted OR 0.41; 95% CI [0.23–0.74]). This follows national trends in HIV testing and may also have resulted from the implementation of routine HIV screening.19,33 However, true opt-out testing could not be performed until 2016 when the Illinois AIDS Confidentiality Act was revised, allowing HIV testing to occur under the consent for general medical care rather than a separate consent process. 34 This legislation allowed for EMR-prompted ordering to be implemented in the ED if patients did not opt out of screening. Leveraging EMR tools such as best practice advisories and linked orders sets may also prove beneficial in the outpatient setting, where the majority of STI-related missed testing opportunities occurred.
There are several limitations worth noting in our study. First, our data did not allow us to determine if an HIV test was offered by a physician but declined by the patient. Additionally, there were limitations based on our EMR system, including that information on sexual behavior of patients is not always collected in structured fields that can be drawn from the electronic data warehouse. We were also unable to analyze the effects of birth sex versus gender, as our EMR system only collected information on legal sex at the time of this study. We did not have information on PrEP use in the STI testing cohort, and were unable to differentiate between symptomatic and asymptomatic STI testing. While a prior positive syphilis result was associated with higher odds of a missed opportunity in STI testing encounters, this is likely explained by the need for serial rapid plasma reagin (RPR) quantification in patients diagnosed and treated for syphilis in shorter intervals than HIV testing may be required. This is supported by the fact that this variable did not become significant until we included the 31–180 day range following a prior encounter to qualify as a missed opportunity—a period of time when the first post-treatment RPR would likely be assessed without an absolute need for repeat HIV testing. While inpatient encounters were associated with lower odds of a missed opportunity in Cohort 1 and higher odds of a missed opportunity in Cohort 2, we were unable to account for severity of illness at time of encounter, which may impact HIV testing. Finally, our data were drawn from a single, urban academic medical center in an area of high HIV incidence and prevalence, so our findings may not be generalizable to other healthcare settings or geographic areas.
In this study, missed opportunities for HIV testing remained common among both persons eventually diagnosed with HIV and those who received bacterial STI testing. Routine, opt-out HIV testing has reduced missed opportunities over time, but these could be reduced further by strengthening targeted and repeat testing. Employing complementary HIV screening approaches is likely to be most effective at identifying new diagnoses. 35 To supplement routine screening in a systemic way, we need better sexual history-taking and structured EMR documentation to identify those patients with reasons to test more frequently, as well as coupling HIV testing with other bacterial STI testing, all while leveraging automated processes within the EMR as much as possible.36,37 Expanding the reach of HIV testing can improve earlier diagnosis of HIV and increase the number of PLWH who are aware of their diagnosis. Improving HIV screening among persons being tested for STIs may also help identify persons who would benefit from HIV PrEP, as HIV testing serves as entry into care for prevention services, 38 a key component in ending the HIV epidemic. Further work is needed to understand and document barriers to testing for patients and providers as well as to incorporate PrEP linkage into HIV testing programs across inpatient, outpatient, and ED settings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Data for this study were provided by the Clinical Research Data Warehouse maintained by the Center for Research Informatics (CRI) at University of Chicago. CRI is funded by the Biological Sciences Division, the Institute for Translational Medicine/CTSA (NIH UL1 TR000430) at the University of Chicago. The Expanded Testing and Linkage to Care Program is funded by the Chicago Department of Public Health. Moira McNulty is supported by the National Institute of Mental Health (K23MH118969).
