Abstract
Introduction
Globally, high-risk human papillomavirus (hrHPV) causes 90%–100% of anal cancers. 1 Anal cancer incidence data have been collected primarily in high-resource countries and incidence data from sub-Saharan Africa (SSA) are limited, 2 although anal cancer may be the most common HPV-associated cancer among men in some parts of SSA. 3
HIV-positive persons and some HIV-negative persons, for example men who have sex with men, are at substantially increased risk for anal cancer. 4 Given that HIV prevalence in SSA is the highest in the world and that increased availability of HIV antiretroviral therapy supports increased lifespans among persons with HIV, 5 increased anal cancer incidence in SSA seems probable among both HIV-positive and HIV-negative MSM.
HPV vaccination is highly effective at preventing infection with multiple hrHPV types, including HPV-16 which is responsible for the large majority of anal cancers, especially in HIV-negative persons. 1 Vaccination programs are increasing in SSA; 6 however, developing efficient screening programs that target persons at highest risk for anal cancer is needed since the full benefit of vaccination will not be realized for decades.
HIV infection, coupled with hrHPV, is highly associated with anal precancers 7 which has resulted in recommendations in high-resource countries to screen HIV-positive persons, especially MSM, using anal Pap cytology and Digital Anal Rectal Examinations (DARE). 8 The benefit of anal cancer screening among HIV-negative MSM is less clear although the Centers for Disease Control and Prevention now encourages screening using DARE among HIV-negative MSM with a history of receptive anal sex. 9
Anal hrHPV prevalence among MSM differs by country and size of city in Africa10,11 with the prevalence of HPV-16 ranging from 0% in the Central African Republic 12 to 23.1% in a large city in central Nigeria 13 in studies using PCR-based assays. To our knowledge there have been no reports of anal hrHPV prevalence among Tanzanian MSM. Nor are there reports from African countries of factors associated with anal hrHPV among HIV-negative MSM. Knowledge of these factors may support development of anal cancer screening programs in these settings.
Our objective was to assess hrHPV prevalence among both HIV-positive and HIV-negative MSM in Dar es Salaam, Tanzania to inform the development of anal cancer screening standards in SSA.
Methods
Study design and participant recruitment
Data and specimens were collected in 2011–2012 in Dar es Salaam (metropolitan area population >4 million), Tanzania for a study of sexual behaviour, HIV, and other sexually transmitted infections in Tanzanian MSM. Details of study activities have been previously described. 14 Briefly, given the highly stigmatized and criminalized nature of homosexuality in Tanzania at the time of the study and to the present, 15 MSM were recruited using respondent-driven sampling (RDS) which provided access to MSM utilizing established networks of friends and acquaintances. Five initial respondents were contacted in Dar es Salaam and these persons referred up to 3 persons each using study coupons. These men in turn recruited additional waves of MSM in a similar, iterative fashion until 200 men were enrolled. Oral informed consent was collected from each participant.
Questionnaire data
All participants completed a self-administered interview on a laptop in Swahili or English. Additional open-ended questions were completed with the assistance of study staff. Participants self-reported their sexual identity as either “gay/homosexual,” “straight/heterosexual,” “bisexual,” “undecided,” or “other.” All men identified as gay or bisexual except for four persons who identified as “undecided.” These were merged with persons identifying as “bisexual.”
Specimen collection and assays
Participants were asked to undergo blood-based testing for HIV-1 antibodies, syphilis antibodies, hepatitis B surface antigen, and urine-based testing for Neisseria gonorrhoea and Chlamydia trachomatis. In addition, an anal swab was used to assess rectal N. gonorrhoea and C. trachomatis. Finally, a second dry Dacron swab was used to collect exfoliated cells from the anal canal. This swab was immersed in specimen transport media (Hologic, San Diego, California, USA) and immediately placed into −20°C storage. A total of 172 persons agreed to sampling with the second swab. Persons declining the swab were more likely to have an unknown HIV status (p < 0.001) and were more likely to be married, separated, divorced, widowed, or living with a partner (p = 0.01). The specimens remained frozen until 2019 when they were sent to a lab in Kenya (Pathologists Lancet Kenya, Lancet Laboratories Tanzania, Uganda & Rwanda) for HPV testing.
HIV-1 positivity was confirmed by a positive result on two rapid tests: Determine (Abbott Laboratories, Chicago, Illinois, USA) which was used as a screening test and Unigold (Trinity Biotech, Bray, Ireland) which was used as a confirmatory test. Pre-test and post-test HIV counselling were provided according to Tanzanian national guidelines and all HIV-seropositive men were referred on to the Muhimbili Hospital HIV Centre in Dar es Salaam. N. gonorrhoea and C. trachomatis were assessed using the APTIMA Combo2 Hologic Gen-Probe (San Diego, California, USA). Syndromic sexually transmitted infection (STI) examination and treatment was provided at the time of specimen collection.
HPV genotypes were determined by COBAS 4800 (Roche Molecular Systems, Inc, Branchburg, NJ) which uses PCR amplification for type-specific detection of HPV-16 and HPV-18, in addition to a group of 12 high-risk HPV types (HPV-31, -33, -35, -39, -45, -51, -52, -56, -58, -59, -66, and -68). β-globin was detected to assess specimen adequacy. A positive and negative control from the kit was included on each run. Of 172 persons providing a swab in Dar es Salaam, 17 swabs were lost in transit to Nairobi, Kenya leaving 155 swabs for HPV genotyping. Of these, 25 specimens were inadequate due to lack of β-globin. Inadequacy was associated with older age at first sex (ptrend = 0.046), having fewer lifetime male partners (ptrend < 0.001), having fewer partners in the past month where the participant is the receptive partner in anal sex (ptrend < 0.001), a bisexual identity (Pearson χ2 p = 0.007), and HIV-negative status (Pearson χ2 p = 0.045). One additional swab had a missing HPV result and 13 individuals had unknown HIV status leaving 116 observations available for analyses.
Statistical and epidemiological analysis
Prevalence for the outcomes of any hrHPV (including HPV-16, -18, and the group of 12 hrHPV types) and for HPV-16 alone was estimated. Prevalence was not adjusted using RDS weights. Exposures chosen for assessment were based on prior HPV literature including age, sexual identity, age at first sex, number of sex partners (male or female) during the lifetime, in the last 1 month and the last 6 months, marital status, employment, engaging in sex for pay or purchasing sex, HIV status, and presence of Treponema pallidum, N. gonorrhoea, and C. trachomatis. In addition, the exposures of number of close gay friends and use of lubrication for anal sex were assessed. Pearson χ2, the Cochran-Armitage test for trend, and Fisher’s Exact tests were used to assess for the association between hrHPV and each categorical exposure.
Since hrHPV was almost ubiquitous among HIV-positive MSM in these data, we assessed factors associated with hrHPV among HIV-negative MSM. Point estimates were derived using Poisson regression with a robust variance estimator 16 and without using RDS weights. 17 Variables with a p-value < 0.20 in bivariate regression analysis were included in multivariable modelling. While age was identified as a potential confounder using Directed Acyclic Graphs, 18 it was not associated with the outcome of anal HPV infection and thus not included in multivariable modelling. Independent risk factors for the outcome were identified using a manual backward-elimination method. Variables with a p-value ≥ 0.05 on a score test were removed manually until a final set of factors remained. Since sexual identity (gay or bisexual) was highly correlated with the number of non-paying partners with whom the participant engaged in receptive anal sex (rspearman = −0.58) and insertive anal sex (rspearman = 0.42), only the behavioural variables were used in multivariable analysis. Syphilis was not included in multivariable regression due to sparse data. Regression analysis was conducted with SAS 9.4 (SAS Institute, Cary, North Carolina, USA).
Patient and public involvement
A non-governmental organization in Dar es Salaam (which cannot be identified for safety reasons) was involved in the RDS selection of “seed” participants and questionnaire piloting and modification. Participants then recruited their friends and acquaintances.
Results
Characteristics of participants, anal high-risk human papillomavirus (HPV) prevalence, and HPV-16 prevalence among men who have sex with men, 2011-2012, Dar es Salaam, Tanzania.
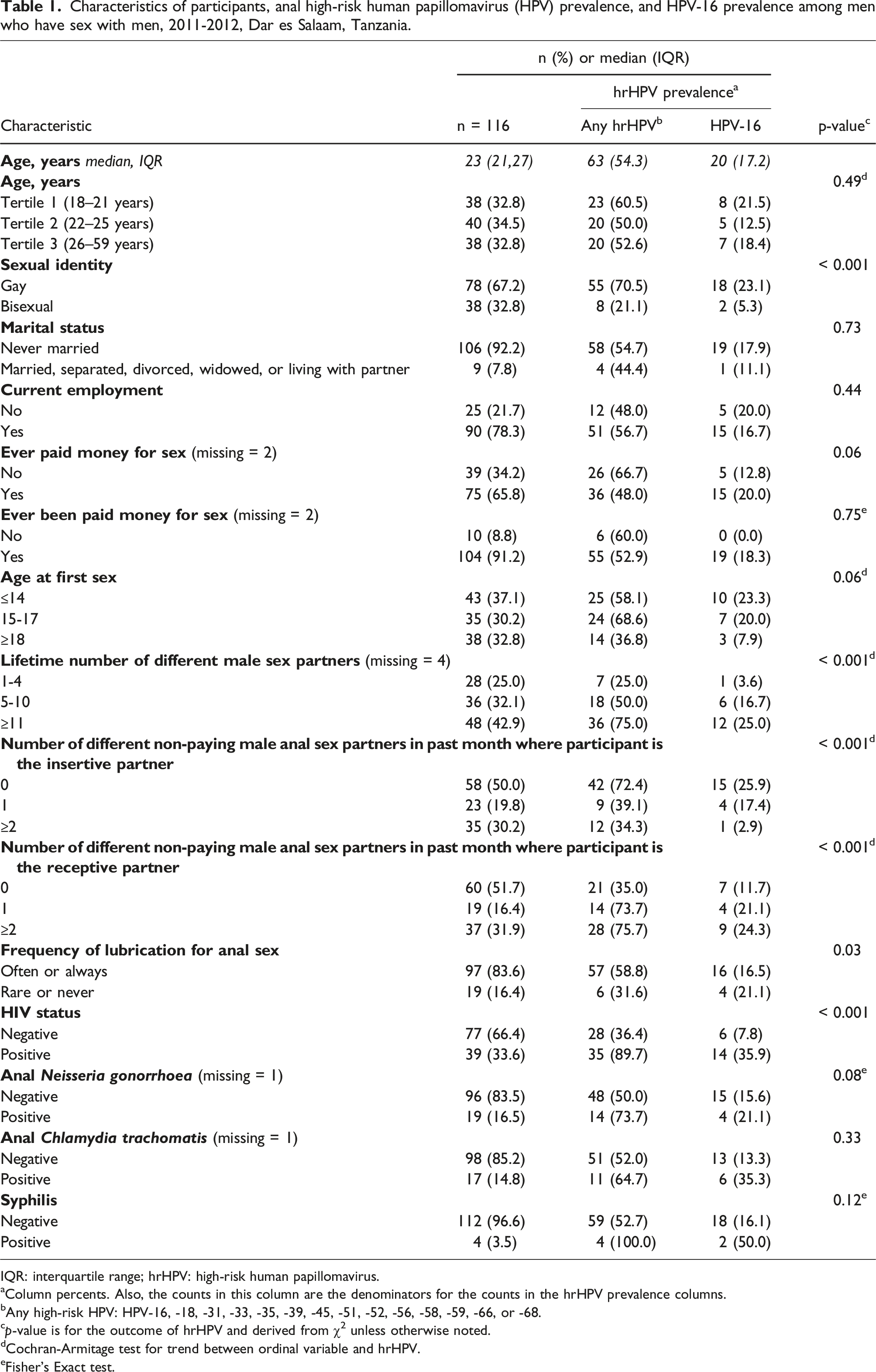
IQR: interquartile range; hrHPV: high-risk human papillomavirus.
aColumn percents. Also, the counts in this column are the denominators for the counts in the hrHPV prevalence columns.
bAny high-risk HPV: HPV-16, -18, -31, -33, -35, -39, -45, -51, -52, -56, -58, -59, -66, or -68.
cp-value is for the outcome of hrHPV and derived from χ2 unless otherwise noted.
dCochran-Armitage test for trend between ordinal variable and hrHPV.
eFisher’s Exact test.
Overall prevalence of any high-risk HPV type was 54.3% while HPV-16 prevalence was 17.2%. Age was not associated with hrHPV prevalence (p = 0.49). HrHPV prevalence was substantially higher among MSM identifying as gay compared with bisexual persons (70.5% and 21.1%, respectively, p < 0.001), among HIV-positive than HIV-negative persons (89.7% and 36.4%, respectively, p < 0.001), and those reporting receptive anal sex with ≥2 partners or 1 partner as compared with 0 partners in the last month (75.7%, 73.7% and 35.0%, respectively, ptrend < 0.001) (Table 1). Prevalence of hrHPV and HPV-16 was 91.7% and 36.1%, respectively among gay persons with HIV. In contrast, prevalence was 17.1% and 2.9% for hrHPV and HPV-16 among HIV-negative bisexual persons (Figure 1). Human papillomavirus (HPV)-16 and high-risk HPV prevalence by HIV and sexual identity among men who have sex with men, 2011-2012, Dar es Salaam.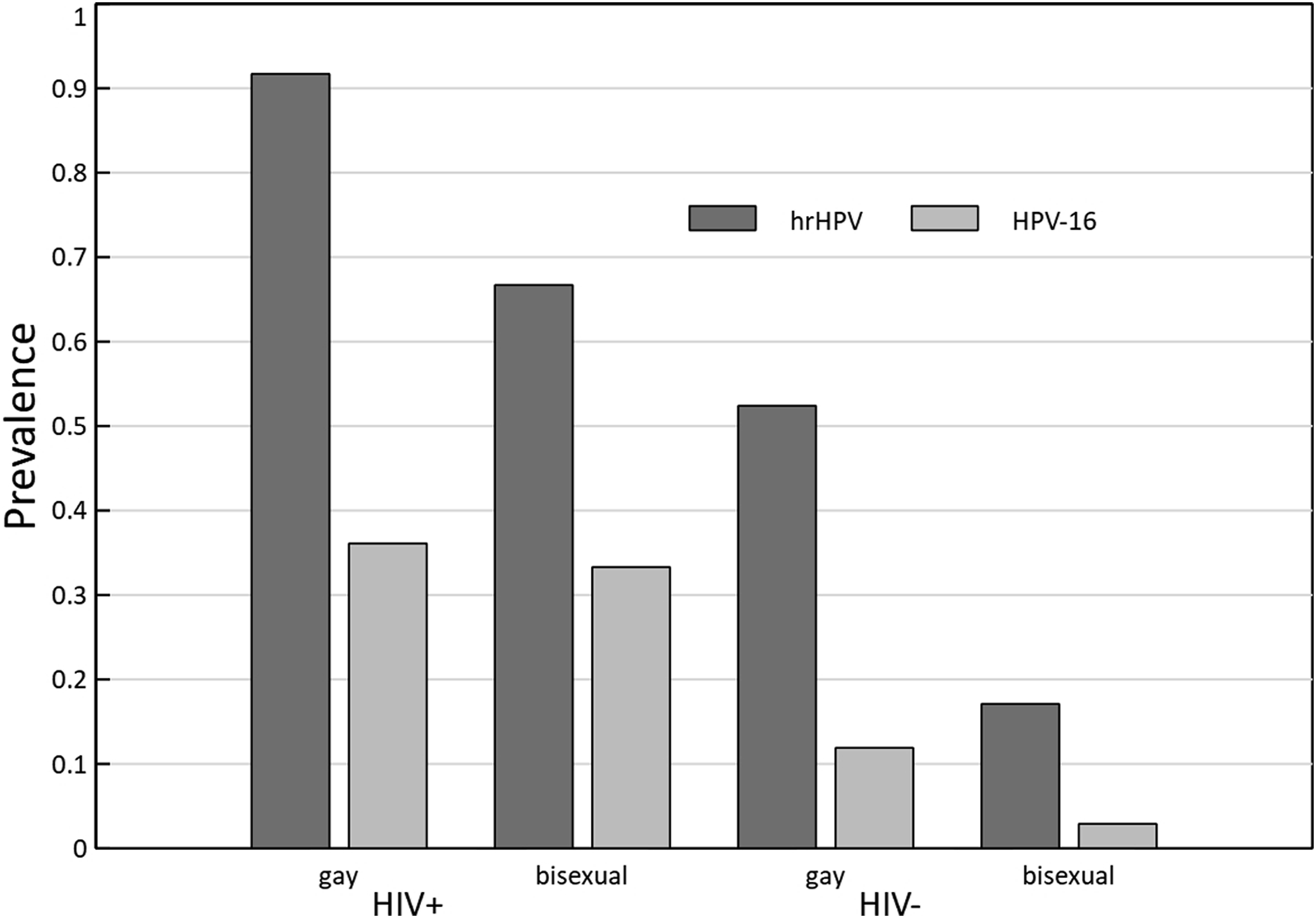
Bivariate analysis: Factors associated with anal high-risk human papillomavirus* infection among HIV-negative men who have sex with men, 2011-2012, Dar es Salaam, Tanzania.

HPV: human papillomavirus; PR: prevalence ratio.
* High-risk HPV: HPV-16, -18, −31, −33, −35, −39, −45, −51, −52, −56, −58, −59, −66, or −68.
Discussion
For this non-clinic population of MSM, hrHPV was almost ubiquitous (89.7%) among HIV-positive MSM while HPV-16 was detected in 35.9% of HIV-positive men. The prevalence of hrHPV was also high (52.4%) among HIV-negative men identifying as gay; however, only 17.1% and 2.9% of HIV-negative men identifying as bisexual harboured hrHPV and HPV-16, respectively.
Two other studies in SSA using RDS recruitment found similar anal HPV prevalence as the current study. Studies of 342 MSM in Nigeria 13 and 207 MSM in Togo 19 found 85.8% and 85.2% of HIV-positive MSM having hrHPV, respectively, and 23.3% and 26.0% of HIV-positive MSM having HPV-16, respectively. Other studies from SSA which used clinic-based recruitment or convenience community samples reported prevalence for hrHPV among HIV-positive MSM ranging 62.0%–81.2%.10,12,20,21 One study from Rwanda, adjacent to northwest Tanzania, reported a prevalence of only 19.7% for hrHPV among HIV-positive MSM. 11
HPV-16 prevalence in this study was 17.2%. In comparison, a recent study of cervical HPV in Tanzania reported an HPV-16 prevalence of 3.5% at the cervix. HPV vaccination is highly effective at preventing HPV infection and associated disease, including cervical and anal precancers; 22 however, Tanzania only started vaccinating girls in 2018 which could not have affected our estimates of prevalence in 2011-2012. 23 Even if there were current gender-neutral HPV vaccination programs in Tanzania, most MSM would not receive its full disease preventive potential; thus, urban Tanzanian MSM will need anal cancer screening for years to come.
Based on the current analysis, HIV-positive and HIV-negative MSM may benefit from anal cancer screening. While there is limited infrastructure for high-resolution anoscopy even in high-resource settings, DARE may still be available, 24 although, the lack of availability of diagnostic and treatment resources for anal cancer may also limit the use of DARE. 25
The only independent risk factor associated with hrHPV among HIV-negative MSM was the number of different non-paying male anal sex partners in the past month where the participant was the receptive partner. Other African studies have not assessed factors associated with hrHPV among HIV-negative MSM. Our results are consistent with studies from high-resource settings. 26 MSM who reported paying for sex had 44% reduced prevalence of anal HPV although this characteristic was not retained in multivariable modelling after adjusting for confounders. A substantial proportion of participants reported both paying for and being paid for sex, an added complexity which we explored previously in this sample of MSM in Dar es Salaam. 27
In addition to number of anal sex partners, there were strong associations between hrHPV, HIV status and sexual identity in bivariate analysis. HIV-negative MSM identifying as bisexual had low HPV-16 prevalence (2.9%), the genotype responsible for about 90% of anal cancers in HIV-negative persons. 1 While societal constructions of minority sexual identities vary across cultures and change over time, 28 it is a repeated finding in SSA countries that either a gay sexual identity (vs bisexual) or engaging in receptive anal sex (vs insertive anal sex) is associated with increased anal hrHPV prevalence.10-12,21 While this suggests that behaviour and/or sexual identity might be utilized in risk-based anal cancer screening algorithms, disclosure of behaviour and sexual identity may not occur since both are highly stigmatized. This barrier not only has consequences for the epidemiologic construction of risk and the interpretation of patterns of vulnerability to STIs and disease outcomes like anal cancer, but it is also possible that persons who generally identify as gay among their social network may not identify as gay to a health care provider. It was common among MSM enrolled in the parent study to say that they would report only heterosexual contact to avoid stigma when going for sexually transmitted infection 29 Strategies to manage stigma may include self-sampling which has been found feasible/acceptable among Tanzanian women for cervical cancer screening. 30
Participants in Dar es Salaam who declined an anal swab were more likely to be married, separated, divorced, widowed, or living with a partner. Since persons in this group were somewhat less likely to have hrHPV in the final dataset, our prevalence estimates may have been somewhat lower if they had agreed to swabbing. Likewise, 25 specimens were β-globin negative which also may have somewhat inflated our prevalence estimates since inadequacy was associated with HIV-negative status, a bisexual identity, and other characteristics that had slightly lower hrHPV prevalence in the final dataset. We have previously observed higher specimen inadequacy among men who have sex with women compared to MSM. 31 It is possible that receptive anal sex might abrade the anal canal mucosa allowing more productive collection of exfoliated swabs, although using lubrication for sex was not associated with inadequacy in the current data (Fisher’s Exact p = 0.17). It is also possible that faecal matter might increase the risk for an inadequate specimen given the potential for bacterial growth that may interfere with PCR assays. 32 It is plausible that persons who have receptive anal sex less often may clean the anus less and therefore leave increased faecal material on a swab.
In this analysis, limited sample size likely prevented the detection of associations with other exposures in multivariable analysis. Justification of sample size was based on the primary parent study goal of assessing HIV prevalence. 33 Also, estimates of the prevalence of bisexual and gay men had not stabilized when recruitment ended in the parent study 34 which may overrepresent gay men and thus possibly inflate overall prevalence. Nevertheless, the prevalence we observed is consistent with anal HPV prevalence estimates of HIV-negative and HIV-positive MSM in other urban areas, worldwide. We did not use RDS weighting in the analysis which may overestimate or underestimate true prevalence; however, RDS recruitment may provide more generalizable results compared to clinic-based samples or community convenience samples. 35 The questionnaire did not ask for number of recent partners for transactional sex which may have prevented us from observing associations between transactional sex and hrHPV. Finally, these data collected in Dar es Salaam may limit generalization of study conclusions to other large urban areas.
The current study observed a high prevalence of anal hrHPV infection in young HIV-positive MSM and in HIV-negative MSM identifying as gay in Dar es Salaam in 2011–2012. These results and others from SSA may foreshadow high anal cancer incidence in the coming decades especially as HIV treatment extends the lives of persons with HIV. Risk-based anal cancer screening programs utilizing participant characteristics might support earlier detection of anal cancers and precancers.
Footnotes
Acknowledgements
The authors give special thanks to the persons volunteering for the study. Also, thanks to Maritza Pallo, Dr Rabia Mukadam, Kevin Brown, and Dr Jennifer S. Smith.
Author’s Contribution
AGN wrote the manuscript and analysed the data; MWR designed the study; KM and JN edited and reviewed the manuscript.
Data availability
Data are available on reasonable request. Data previously presented, in part, at the 33rd International Papillomavirus Conference (Virtual) July 20-24, 2020 and the 34th International Papillomavirus Conference (Virtual) November 13-15, 2021.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The protocol was approved by The Tanzanian National Institute for Medical Research (NIMR/HQ/R.8a/Vol. IX/1088) and The University of Texas Health Science Center’s Institutional Review Board (HSC-SPH-10-0033).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the United States’ National Institutes of Health, National Institute of Mental Health grant number 5R21MH090908 to MWR.
