Abstract
Tuberculosis (TB) remains a leading cause of morbidity and mortality among people living with HIV. HIV-associated TB disproportionally affects African countries, particularly vulnerable groups at risk for both TB and HIV. Currently available TB diagnostics perform poorly in people living with HIV; however, new diagnostics such as Xpert Ultra and lateral flow urine lipoarabinomannan assays can greatly facilitate diagnosis of TB in people living with HIV. TB preventive treatment has been underutilized despite its proven benefits independent of antiretroviral therapy (ART). Shorter regimens using rifapentine can support increased availability and scale-up. Mortality is high in people with HIV-associated TB, and timely initiation of ART is critical. Programs should provide decentralized and integrated TB and HIV care in settings with high burden of both diseases to improve access to services that diagnose TB and HIV as early as possible. The new prevention and diagnosis tools recently recommended by WHO offer an immense opportunity to advance our fight against HIV-associated TB. They should be made widely available and scaled up rapidly supported by adequate funding with robust monitoring of the uptake to advance global TB elimination.
Introduction
With the rapid upsurge of the global HIV epidemic in the late 1980s, TB incidence rose in HIV-prevalent countries despite successful implementation of the Directly Observed Treatment Short-course (DOTS). This eventually led to criticism about the failure of the DOTS strategy in addressing HIV-associated TB. 1 Since then, collaborative TB and HIV activities have been established and scaled up. 2 Due to its implementation, deaths from HIV-associated TB declined globally by 60% between 2000 and 2018. 3 Nonetheless, TB remains the leading cause of deaths in people living with HIV. In this review, we summarize HIV-associated TB epidemiology, pathogenesis, clinical presentation and care, the current global programmatic response, and key challenges.
Epidemiology of HIV-associated TB
TB is the leading cause of HIV-associated hospitalization and death among adults and children living with HIV worldwide, 4 and accounted for a third (208,000) of the estimated 690,000 HIV-related deaths globally in 2019, 81% of these deaths occurring in the African region. 3 A systematic review of postmortem studies from resource-limited settings found that almost half (46%) of TB deaths were undiagnosed at the time of death. 5 While over half (62%) of new TB cases occur in the Southeast Asia and Western Pacific regions, almost three-quarters (595,000) of all new HIV-associated TB cases occur in the African region; a quarter (24%) of TB cases in the African region are estimated to be among people living with HIV, rising to around 60% in Lesotho, Eswatini, and South Africa. In 2019, only 56% of the estimated 815 000 incident HIV-positive TB cases were notified globally. 3
TB and HIV intersect where overcrowding and impoverishment meet, particularly affecting populations living in informal settlements, prisoners, people who inject drugs, migrants, and mine workers. Environmental factors such as poor living conditions characterized by overcrowding and poor ventilation and high prevalence of individual risk factors (e.g., smoking, undernutrition, and alcohol use) among these populations create the perfect environments for TB transmission. Poor access to health care exacerbates the epidemic further. For example, TB notification rates in South Africa’s townships are among the highest in the world, exceeding 1000 per 100,000 persons. 6 The risk of TB disease in prisons is around 23 times higher than the general population. 7 TB prevalence among HIV-positive inmates is as high as 30%, while HIV prevalence among inmates with active TB can reach 40%–70%. 8 These groups face additional challenges, making TB control even more difficult. Migrants and refugee populations experience language barriers, discrimination, unstable legal status and consequent fear of deportation, and mobility. 9 Many mine workers face occupational risk factors, including poorly ventilated working environments and silica dust exposure. 10 Given the variety of challenges they face, the need for a multisectoral response beyond health programs cannot be overemphasized.
The relationship between HIV and drug-resistant TB (DR-TB) is inconsistent. Epidemic levels of DR-TB have been reported in South Africa, the country with the greatest number of people living with HIV. 11 Data from over 100,000 TB patients did not find a consistent association between multidrug-resistant TB (MDR-TB) and HIV, although poor data quality and limited coverage of HIV testing were limitations to this review 12 ; nevertheless, in 11 of the countries included in the review, HIV-positive TB patients had a significantly higher odd of MDR-TB than HIV-negative individuals. Primary resistance acquired through transmission is estimated to account for a majority of DR-TB. 13 However, the gap remains in the detection of DR-TB, with only 44% of the estimated incident rifampicin-resistance/MDR-TB cases notified globally. 3 The treatment success rate for MDR-TB remains suboptimal. In a large individual patient data meta-analysis, MDR-TB treatment success rate was 68% in HIV-negative patients, with a lower success rate of 55% in HIV-positive patients. 14
Pathogenesis and risk of TB in people living with HIV
TB is the most common opportunistic infection in people living with HIV who have increased susceptibility to TB infection and are at high risk for progression to TB disease. 15 In most healthy individuals, TB infection is carried as latent TB infection (LTBI). Untreated HIV infection results in a progressive reduction in CD4 count, which increases the risk for progression from TB infection to active disease. There is an increased risk of TB soon after HIV infection, even at high CD4 count 16 ; this suggests that the increased risk is not solely because of CD4 cell depletion but also possibly because of functional impairment of TB-specific T cells.16,17 It is also suggested that HIV induces innate immune defects. HIV infection appears to limit macrophage ability to restrict the growth of TB bacilli. 16 This underpins the importance of ART, regardless of CD4 count to prevent TB. 18
ART induces immune reconstitution, thereby reducing the risk of TB disease by two-thirds. 19 CD4 count recovery and viral suppression are associated with a reduction in TB incidence while HIV viral load alone is not predictive of TB risk. 20 The risk of TB in people living with HIV remains higher than HIV-negative individuals even after initiation of ART. 21 A rapid expansion of TB-specific CD4 cells induces an exaggerated innate immune response, and in the presence of a high TB antigen load ART may induce TB-immune reconstitution inflammatory syndrome (IRIS). 16
TB may in turn exacerbate HIV infection. Increased HIV viral load in lungs, blood, and cerebrospinal fluid is observed in people with TB. 16 This is likely due to increased viral replication at the sites of granulomatous inflammation with abundant activated T cells and upregulation of HIV transcription by pro-inflammatory cytokines produced through the host immune response against TB. 16 Epidemiological studies have shown an increased incidence of opportunistic infections in HIV-positive people with TB. 16
TB prevention
Trials have shown that immediate ART initiation even at a high level of CD4 count significantly reduces the risk of TB.22,23 Thus, achieving the 95–95–95 targets—95% of people living with HIV knowing their HIV status, 95% of those diagnosed receiving ART, and 95% of them with viral suppression—is critical for TB prevention. Since 2016, the WHO has recommended ART for all, regardless of CD4 count. As of July 2019, 96% of low- and middle-income countries (LMICs) have adopted the recommendation. 24 However, only 67% of people living with HIV were on ART at the end of 2019. 25 Moreover, ART alone is insufficient to prevent TB. 26 TB preventive treatment (TPT) provides additive protection even at higher CD4 counts and reduces mortality.22,27,28
The sensitivity of LTBI tests in people living with HIV is limited and even lower in those with lower CD4 count, whose TB risk is higher. 29 Trials in high TB burden countries have shown benefits of TPT regardless of LTBI test results.27,28,30Hence, LTBI testing is not required as recommended by WHO except in settings with low TB incidence. 31
Six months of daily isoniazid, known as isoniazid preventive therapy (IPT), has been the mainstay of treatment with protective efficacy ranging between 40 and 60% for people living with HIV.32,33 However, studies in high TB transmission settings showed waning protection.34,35 In studies conducted in South Africa and Botswana, IPT of 36 months or longer was associated with lower TB incidence than 6 months of IPT.36,37 Therefore, WHO conditionally recommends at least 36 months of IPT in people living with HIV in high-transmission settings who have an unknown or a positive-LTBI test; however, its uptake is very limited. 31 In low TB transmission settings, 6 months of IPT has shown a long-term protective effect. 38 Furthermore, a recent trial in Côte d’Ivoire demonstrated a sustained reduction of mortality for 6 years by 6 months of IPT in adults living with HIV with high CD4 count. 28 Observational studies in Myanmar and Indonesia reported similar results.39,40 Optimal duration of IPT in high TB incidence settings depends on local TB epidemiology and transmission as well as availability of health infrastructure and acceptance and adherence to treatment. 31
Other effective regimens include daily 3–4 months rifampicin; daily 3 months isoniazid and rifampin; and 3 months weekly isoniazid and rifapentine (3HP). 31 3HP is associated with less hepatotoxicity and higher treatment completion rates than 6–9 months IPT. 31 In a trial in African countries, a single course of 3HP was as effective as two courses of 3HP given annually. 41 Additionally, one month of daily rifapentine and isoniazid was shown to be non-inferior to 9 months IPT. 30 Studies have suggested that rifapentine can be coadministered with efavirenz and dolutegravir without jeopardizing antiretroviral activity.42,43
Most high TB burden countries have adopted TPT in policy though it has not translated into its access at scale. 44 In 2019, data on the coverage of TPT were available only in 21 out of the 38 high TB and TB/HIV burden countries; 13 of them had <50% coverage. 3 This failure is due to health system and operational barriers, 45 in particular fear of creating isoniazid resistance despite lack of evidence, 46 problems with patient acceptability, and concerns about stock-outs and program sustainability. 47 Furthermore, as of June 2020, rifapentine was registered only in 14 countries. 3
Prompt identification and treatment of TB is critical to prevent transmission. Systematic screening for TB should be implemented as a package with TPT. People living with HIV are susceptible to nosocomial TB transmission due to their immunosuppression and frequent exposure to healthcare settings. 13 Nosocomial outbreaks of DR-TB have been reported. 13 Infection prevention and control needs to be strengthened to prevent such tragic events. However, data to evaluate its implementation are lacking. 48 Specific guidance is available elsewhere. 48
People presenting to care with advanced HIV disease
Trials of empirical TB treatment in people living with HIV.
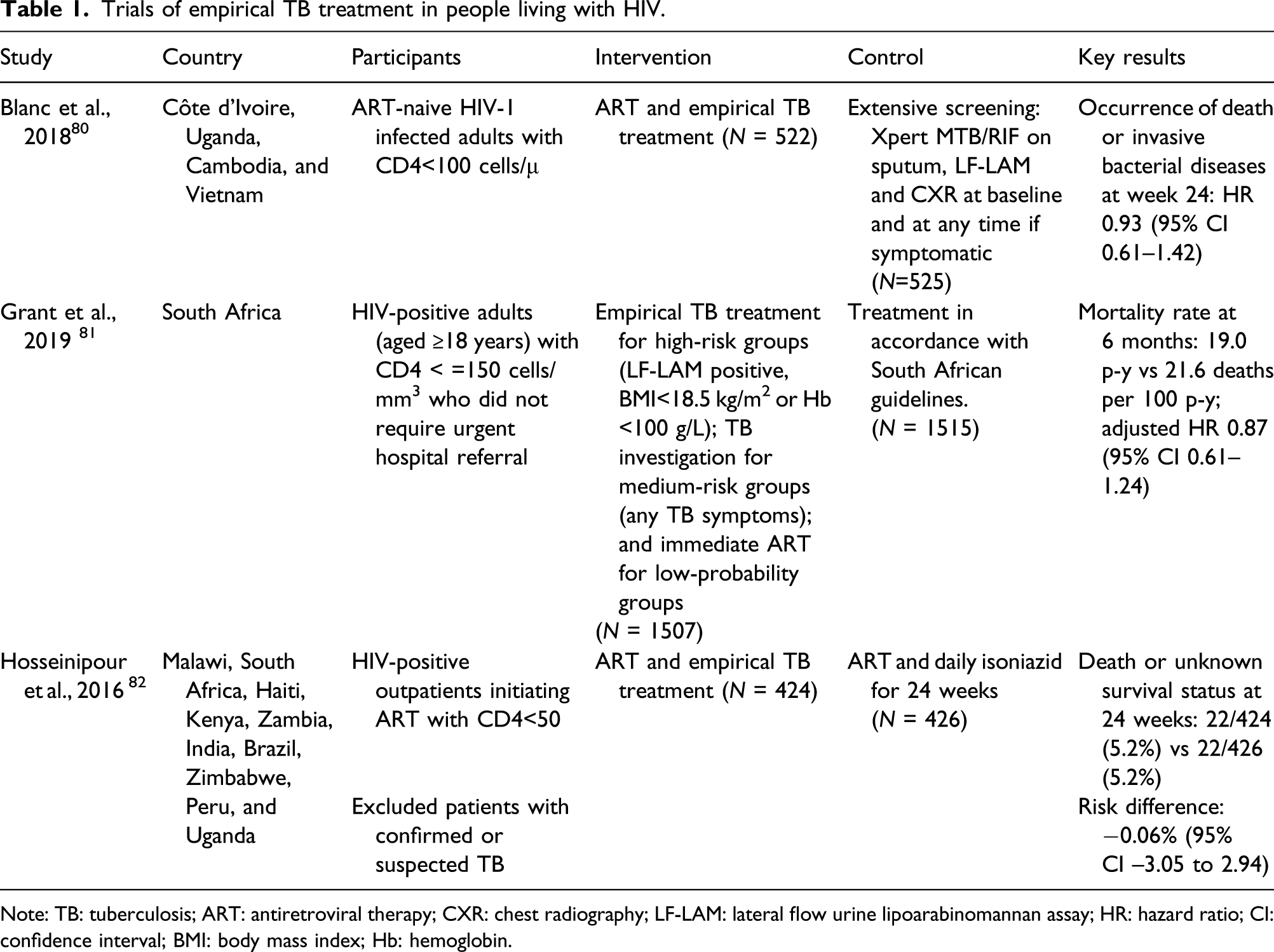
Note: TB: tuberculosis; ART: antiretroviral therapy; CXR: chest radiography; LF-LAM: lateral flow urine lipoarabinomannan assay; HR: hazard ratio; CI: confidence interval; BMI: body mass index; Hb: hemoglobin.
Clinical features
TB signs and symptoms in people living with HIV are variable and depend on the degree of immunosuppression. While pulmonary manifestations are common in less immunocompromised patients, the risk of extrapulmonary TB increases with advanced immunosuppression and presents with various signs and symptoms, commonly including lymphadenopathy and pleural effusion. In a systematic review, 78.9% of HIV-associated TB had at least one of cough, fever, night sweats, and weight loss, and thus WHO recommends TB screening based on those symptoms. 53 However, people living with HIV on ART are more likely to be asymptomatic, and only around 50% have one of the above four symptoms. 54 Chest radiographic findings also depend on the degree of immunosuppression: severely immunosuppressed patients typically present a primary TB pattern (hilar adenopathy, pleural effusion, middle or lower lobe infiltrates). 55 Radiographic findings in those with high CD4 count are similar to HIV-negative individuals, including cavitation. 55
Extrapulmonary TB can affect any organ in the body and commonly involves pleura, lymph node, central nervous system, abdominal organs, pericardium, and bone and often presents as disseminated TB. 56 Diagnosis of extrapulmonary TB is challenging, with most being managed without bacteriological confirmation. The delay in diagnosis increases mortality. 57 New diagnostics including Xpert Ultra and lateral flow urine lipoarabinomannan assay (LF-LAM) offers a great opportunity to expedite diagnosis.
Diagnosis
In people living with HIV, sputum bacillary load is usually low, compromising sensitivity of bacteriological tests.58,59 WHO recommends Xpert MTB/RIF as the initial diagnostic test, given its diagnostic accuracy, rapid turn-around time (2 h), and the ability to identify rifampicin resistance. The test has a much higher sensitivity of 80–90% than sputum smear microscopy. 60 Despite reduced time to treatment and a higher proportion of bacteriological confirmation, evidence for direct clinical benefit had been inconclusive61–63; however, in a recent individual patient data meta-analysis of RCTs, people living with HIV tested with Xpert MTB/RIF had 24% lower mortality than those tested with smear microscopy. 64 A new version of this test, Xpert Ultra, has increased sensitivity by 12% in sputum samples among people living with HIV. 65 Xpert Ultra incorporated a semiquantitative “trace” category to identify paucibacillary TB, which is more common in HIV-positive individuals. 66 The WHO recommends that “trace results” should be interpreted as true positive in people living with HIV. 65
According to a UNAIDS survey in 2020, WHO-recommended rapid tests for TB were available in most facilities providing HIV testing and care in 12 out of 30 high TB/HIV burden countries. 67 However, data on the number of HIV-associated TB diagnosed with these tests are lacking. Challenges in the scale of Xpert include lack of knowledge among clinical staff, insufficient support for maintenance, and a high price (at least US$9.98 per cartridge). 68
LF-LAM has emerged as an additional point-of-care test for TB in people living with HIV.69,70 LF-LAM is recommended to assist diagnosis of TB in people living with HIV who have signs and symptoms of TB or a low CD4 count (<200 cells/mm3 for inpatients and <100 for outpatients); and those who are seriously ill. 71 The sensitivity of LF-LAM is highest when CD4 count is low, but it is still around 50% with 84%–98% specificity. 65 The LF-LAM is affordable (US$3.5 per unit) and can be used in peripheral facilities. As of 2019, only 13 of the high TB burden countries had adopted it, and the number of people accessing it is unknown. 3 Treatment initiation guided by LF-LAM showed a reduction of mortality in HIV-positive inpatients, 70 and its use as a screening tool irrespective of symptoms may also reduce mortality. 72 Hence, the adoption and rollout of LF-LAM should be accelerated. Recently, a novel LF-LAM (SILVAMP TB LAM, Fujifilm) has shown improved sensitivity. 73
Diagnosing TB is even more complex in children due to difficulty in obtaining sputum and paucibacillary load. 74 Both culture and molecular studies have a very low sensitivity on induced sputum or gastric lavage in children.74,75 Xpert testing using alternative samples have been explored. In a systematic review, sensitivity and specificity of Xpert using stool samples were 98% and 57% in children. 76 WHO new guidelines recommend Xpert testing in gastric aspirate, nasopharyngeal aspirate, and stool samples not only sputum in children. 65
Diagnostic accuracy of Xpert MTB/RIF in extrapulmonary specimens.
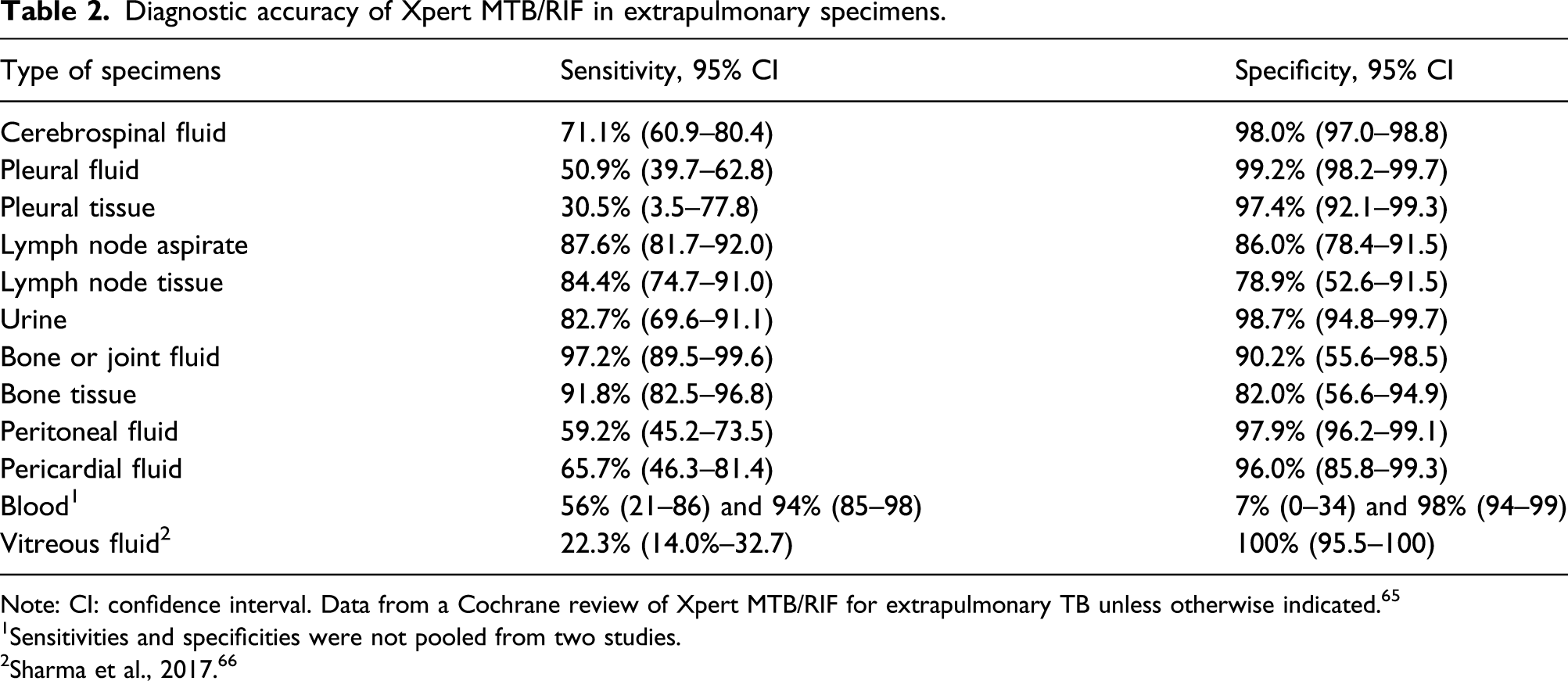
Note: CI: confidence interval. Data from a Cochrane review of Xpert MTB/RIF for extrapulmonary TB unless otherwise indicated. 65
1Sensitivities and specificities were not pooled from two studies.
2Sharma et al., 2017. 66
Treatment
Drug-susceptible TB treatment
The treatment regimen for drug-susceptible TB does not differ by HIV status. Treatment should be given daily rather than intermittently due to a higher risk of failure. 80 ART substantially improves outcomes and should be given to all. Extension of treatment beyond 6 months is not required because of HIV infection. Six months of treatment is associated with a higher risk of failure than an extended treatment only in the absence of ART. 80 Some experts recommend an extension in certain forms of extrapulmonary TB (e.g., central nervous system). High-dose rifampicin (15 mg/kg) given for the first 2 months has shown a reduction of mortality in people living with HIV with CD4 count <100 cells/mm3 without increased risk of hepatotoxicity. 81 A recent RCT demonstrated a non-inferiority of a 4-month regimen (including high-dose rifapentine, isoniazid, moxifloxacin, and pyrazinamide) to the standard regimen, which included a subgroup of HIV-positive participants. 82 In another RCT, in children (including HIV-positive children) with minimal TB, shortening the continuation phase into 2 months was non-inferior. 83
The management of extrapulmonary TB is similar in HIV-positive and -negative individuals. Corticosteroids have been shown to reduce mortality from TB meningitis in HIV-negative individuals, but data are limited for people living with HIV. 84 For pericarditis, a systematic review suggested that steroid may reduce deaths in HIV-negative patients, but no benefit was found among HIV-positive participants. 85 Data are very limited in those on ART.
Drug-resistant TB treatment
Recommended treatment regimens for DR-TB are the same for HIV-positive and -negative individuals. 86 Shorter treatment regimens now recommended by the WHO are promising to improve outcomes. 86 In an RCT, mortality was higher among HIV-positive participants given 9–11 months regimens than long-term regimens; however, this was not statistically significant. 87 In programmatic data from South Africa, a shorter all-oral bedaquiline-containing regimen resulted in higher success rate than a shorter regimen including an injectable agent (73% vs 60%), with a similar effect observed in HIV-positive participants. 86 In the Nix-TB trial, 6 months of bedaquiline, pretomanid, and linezolid resulted in treatment success in 91% of HIV-positive participants who had extensively drug-resistant TB or treatment-intolerant or nonresponsive MDR-TB. 88 ART should be started as soon as possible irrespective of drug resistance status, although interactions and shared toxicity between ART and MDR-TB drugs may complicate management. For example, close monitoring of QT prolongation is needed when delamanid and/or bedaquiline is coadministered with protease inhibitors.
Timing of antiretroviral treatment
WHO recommends ART should be started as soon as possible within two weeks of initiating TB treatment, regardless of CD4 count. In 2019, 88% of notified HIV-positive TB patients worldwide received ART. 3 Starting ART during TB therapy reduces the risk of death by around three-fifths. 89 TB and HIV co-treatment is complicated by interactions between some anti-TB drugs, notably rifampicin, and several commonly used antiretroviral drugs. Detailed information is available elsewhere (https://www.hiv-druginteractions.org/).
Treatment of IRIS
IRIS can present as new TB diagnosis in a patient recently started on ART (unmasking TB), or as paradoxical IRIS, worsening of symptoms after initiation of TB treatment. The risk of IRIS increases with decreasing CD4 count at ART initiation. 90 Studies have shown that mortality due to TB-IRIS is low (0.8%) except for TB meningitis 91 ; however, morbidity remains significant. Nevertheless, ART should be started as soon as possible within the aforementioned time frame. Immediate initiation of ART in TB meningitis was not found to reduce mortality but was associated with severe adverse events. 92 The optimal timing of ART in TB meningitis is uncertain, and it needs to be initiated with caution.
In an RCT in South Africa, prednisone given 4 weeks after initiation of ART reduced the risk of TB-related IRIS by 30% without increasing risk of cancer and severe infection. 93 Steroids are also shown to reduce the morbidity of TB-IRIS and are commonly used to treat IRIS. 94
Pregnancy and HIV-associated TB
TB is one of the common causes of morbidity and mortality in women of reproductive age.95,96 TB prevalence in pregnant and postpartum women in HIV-prevalent countries is more than three times higher than low HIV burden countries. 97 Risk of adverse birth outcomes is increased for HIV-positive pregnant women with TB. Risk of mother-to-child transmission of HIV has been reported to increase 2.5-fold, 98 while perinatal mortality has been reported to increase sixfold. 99 The coverage of ART among pregnant women was 85% globally in 2019 while a wide variation existed by region. 25
Symptom screening has a lower sensitivity in HIV-positive pregnant women than nonpregnant individuals. 54 A recent trial showed increased case detection by universal sputum testing by Xpert in HIV-positive pregnant women. 100 Strategies to expedite the diagnosis of TB among pregnant women are urgently needed.
WHO recommends TPT regardless of pregnancy status. 36 A recent clinical trial reported more frequent adverse pregnancy outcomes among women with HIV given IPT during gestation, 101 yet observational studies reported a protective effect. 31 The conflicting results might be explained by bias in observational studies and differences in study populations since the RCT appeared to include women who were less at risk for TB than observational studies. It should also be noted that loss to HIV care after delivery tends to increase, 102 which can result in a missed opportunity to receive TPT. Therefore, benefits and harms need to be weighed and discussed with pregnant women to decide timing to initiate TPT. Currently, data on the coverage of TPT among pregnant women are lacking.
Program implementation
In both HIV and TB programs, delays and difficulties in accessing services are important causes of patient loss to care and associated mortality.103–105 Decentralization of care, long practiced in TB control programs, is now accepted as a central way to delivering ART to increase coverage and retention in care. 106
Integration of TB and ART services in settings with a high burden of both diseases is another key strategy for improving outcomes. 107 In South Africa and Uganda, full integration of TB and HIV services reduced time to ART initiation and increased its uptake.108,109 Other documented benefits include reduced mortality, 110 reduced delays in providing cotrimoxazole, 111 reduced waiting times, 112 and reduced loss to follow-up between antenatal, TB and HIV services. 113 This latter example illustrates the importance of integrating TB and HIV services in other services where both diseases are commonly identified. 110 While the risk of nosocomial transmission is often raised as a concern for the integration of TB and ART services, TB transmission occurs mostly from undiagnosed TB, and thus, effective management of both conditions can reduce the risk. 114 In HIV-prevalent settings, decentralizing MDR-TB treatment and care facilitates integrated management of both MDR-TB and HIV and has been found to increase initiation of MDR-TB treatment and reduce time to treatment and is predicted to substantially reduce mortality.61,115
Engagement of community health workers can improve outcomes for both HIV and TB,116,117 and in settings with a high prevalence of coinfection, it makes sense to integrate TB and HIV services at the community level. 118 Many countries in sub-Saharan Africa employ TB/HIV counselors to support joint activities. Recent reductions in donor support combined with a lack of national prioritization threaten the sustainability of TB/HIV lay counselors. 119
Funding gaps remain large for both TB and HIV. In 2019,US$18.6 billion was available for the HIV response, far below the target (US$26.2 billion) set by UNAIDS for 2020. 25 US$13 billion per year is required for TB program (and US$2 billion for research), while only a half was available in 2019. 3 These gaps would result in enormous health and social toll that could have been averted. Investment in the TB and HIV response would bring a significant economic return, US$42 and US$6.4 for every US dollar spent, respectively.120, 121 Benefits from the investment extend to other diseases, as has been shown in the COVID-19 pandemic where platforms for TB and HIV program were leveraged to accelerate the response. Commitments made at the United Nations High-Level Meetings are being tested.
Conclusions
Notwithstanding progress in rollout of TB and HIV services, substantial gaps remain in the prevention, diagnosis, and treatment of HIV-associated TB. Innovations, such as shorter preventive and treatment regimens, Xpert Ultra, LF-LAM, and new MDR-TB treatment regimens, offer an immense opportunity to bring HIV-associated TB under control. They must be rapidly adopted and scale up supported by adequate funding with robust monitoring of the uptake. Further research should aim to achieve durable TB protection, non–sputum-based point-of-care TB diagnosis, and more effective treatment of drug-susceptible and resistant TB across populations.
Footnotes
Acknowledgements
We gratefully acknowledge valuable inputs to our manuscript from Sabine Hermans Amsterdam Institute for Global Health and Development.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
