Abstract
Background
The diagnosis of neurosyphilis is a challenge, and the criteria for deciding when to perform a lumbar puncture are still controversial, especially in people living with HIV with a late latent syphilis diagnosis.
Methods
Retrospective analysis of demographic, clinical, and laboratory data of people with HIV and documented late latent syphilis or syphilis of unknown duration with a cerebrospinal fluid VDRL test.
Results
122 patients were evaluated, of whom 52 had the diagnosis of neurosyphilis. Patients with and without neurosyphilis presented a similar viral load and lymphocyte CD4+ T-cell count. Neurological symptoms (OR 6.4, 95% CI 2.1–22.4; p < 0.01), serum VDRL titers of 1:32 (p<0.01), 1:64 (p = 0.055), and ≥1:128 (p < 0.001) were associated with neurosyphilis. Furthermore, serum VDRL ≥1:32 were associated with (OR 24.9, 95% CI 5.45–154.9; p < 0.001) or without (OR 6.5, 95% CI 2.0–29.2; p = 0.004) neurological symptoms with neurosyphilis; however, VDRL ≤1:16 with neurological symptoms can be associated with neurosyphilis (OR 7.6, 95% CI 1.03–64.3; p = 0.046).
Conclusion
Neurological symptoms, particularly headache, were predictors of neurosyphilis in people with HIV irrespective of their viral load and lymphocyte CD4+ T-cell count in late latent syphilis. A serum VDRL ≥1:32 increased the risk of neurosyphilis in patients with or without any symptoms.
Background
In the last decade, there has been a worldwide rebound in the incidence of syphilis among men who have sex with men (MSM).1,2 Neuroinvasion by Treponema pallidum occurs early in the course of the disease, and it can lead to asymptomatic meningitis that, if untreated, can progress to symptomatic neurosyphilis.3,4 Early forms of symptomatic neurosyphilis include meningitis, stroke, and loss of hearing and vision, while late forms include dementia (e.g. general paresis) and sensory ataxia (e.g. tables dorsalis). In people living with HIV, concomitant uveitis or meningitis may be more common. 5 It has been reported that in all stages of syphilis, cerebrospinal fluid (CSF) abnormalities can be found in 15–30% of the cases.6,7 In many cases, CSF abnormalities were detected without neurological symptoms, leading to the concept of “asymptomatic neurosyphilis” (ANS), which is believed to be the precursor of symptomatic neurosyphilis. The factors involved in the progression from asymptomatic to symptomatic neurosyphilis are unknown but the presence of a low CD4+ T-cell count (≤350 cells/μL) in people living with HIV increases threefold the risk of neurological involvement. 8
The diagnosis of neurosyphilis is challenging. It is based on abnormal serum and CSF tests (i.e. elevations in the CSF white cell count and protein level). The CSF VDRL test (Venereal Disease Research Laboratory test) is specific for neurosyphilis but its sensitivity ranges from 30 to 70%. 9 A considerable proportion of people living with HIV who develop neurosyphilis are asymptomatic.10,11,26 The definition of ANS is extremely difficult and contentious. Most definitions depend on a combination of CSF laboratory tests (protein, cells, CSF treponemal test, and CSF non-treponemal tests) but no consensual definition exists. 12 To the best of our knowledge, the are no studies reporting risk factors associated with neurosyphilis in people living with HIV with late latent syphilis (LLS). 12 For that reason, the objective of our study was to analyze clinical factors associated with neurosyphilis in people living with HIV with a diagnosis of LLS. We hypothesized that VDRL titers ≥1:32 and the presence of neurological symptoms are associated with neurosyphilis.
Methods
Study population and laboratory testing
This study was conducted at the HIV Unit of a major university third-level hospital in Guadalajara, Jalisco, Mexico. We retrospectively analyzed the demographic, clinical, and laboratory data from the patients hospitalized between February 2004 and December 2020. Symptoms were obtained retrospectively from the progress notes of hospitalized patients. For the analysis, we sought the presence of neurological symptoms such as altered mental status, gait incoordination, cranial neuropathies, hemiparesis, headache, stiff neck, photophobia, blurred vision or hearing loss; however, only headache, blurred vision, and hearing loss were found in the study participants. All individuals included in the study had documented syphilis by reactive serum treponemal test and non-treponemal test (NTT). The NTT used during the entire study period was serum VDRL. The treponemal test used in all patients was fluorescent treponemal antibody absorption (FTA-ABS). The stage of syphilis was determined according to the Center for Disease Control and Prevention’s (CDC) guidelines 13 and patients with syphilis of unknown duration were considered as LLS. Results of HIV serological tests, plasma HIV-1 RNA viral load, and peripheral blood lymphocyte CD4+ T-cell count were obtained from medical records and the data were used if obtained within 90 days of the lumbar puncture (LP). The limit of detection for HIV-1 considered for the study was <50 copies/mL.
Figure 1 shows a flowchart describing the study design. An LP to diagnose neurosyphilis was performed in all patients who met the following criteria: LLS stage and the presence of any symptoms compatible with neurosyphilis (i.e. neurological, ophthalmological, and audiological),
12
lymphocyte CD4+ T-cell count <350 cell/μL or an VDRL titers ≥1:32. In this study, neurosyphilis was defined as a positive CSF VDRL test, as stated by the 2020 European guidelines.
12
To avoid misclassification bias, the group without neurosyphilis was composed of patients who presented a negative CSF VDRL and a negative CSF FTA-ABS. For the data analysis, we included patients with complete clinical data. The group classified as without neurosyphilis was mostly asymptomatic (90%) and only seven patients (10%) presented with symptoms unrelated to syphilis. The seven symptomatic patients without neurosyphilis had diagnose unrelated to neurosyphilis that explained their symptoms: two patients reported with headache had a final diagnosis of meningeal cryptococcosis, three patients with blurred vision had a diagnosis of ocular toxoplasmosis uveitis (2) or tuberculosis (1) and two patients with hypoacusis had drug-induced ototoxicity. These diagnoses, plus a negative CSF VDRL and negative CSF FTA-ABS test, make the possibility of neurosyphilis unlikely. Therefore, this group of individuals with a negative VDRL and FTA-ABS was considered as “low probability” of neurosyphilis.14,15 The CSF FTA-ABS test is highly sensitive and in the absence of specific neurological symptoms, neurosyphilis is very unlikely to be present with a negative CSF FTA-ABS test.14,15 Flowchart of the retrospective study design. PLWHIV, people living with HIV; FTA-ABS, fluorescent treponemal antibody absorption test; VDRL, venereal disease research laboratory; ELS, early latent syphilis; LLS, late latent syphilis; CSF, cerebrospinal fluid.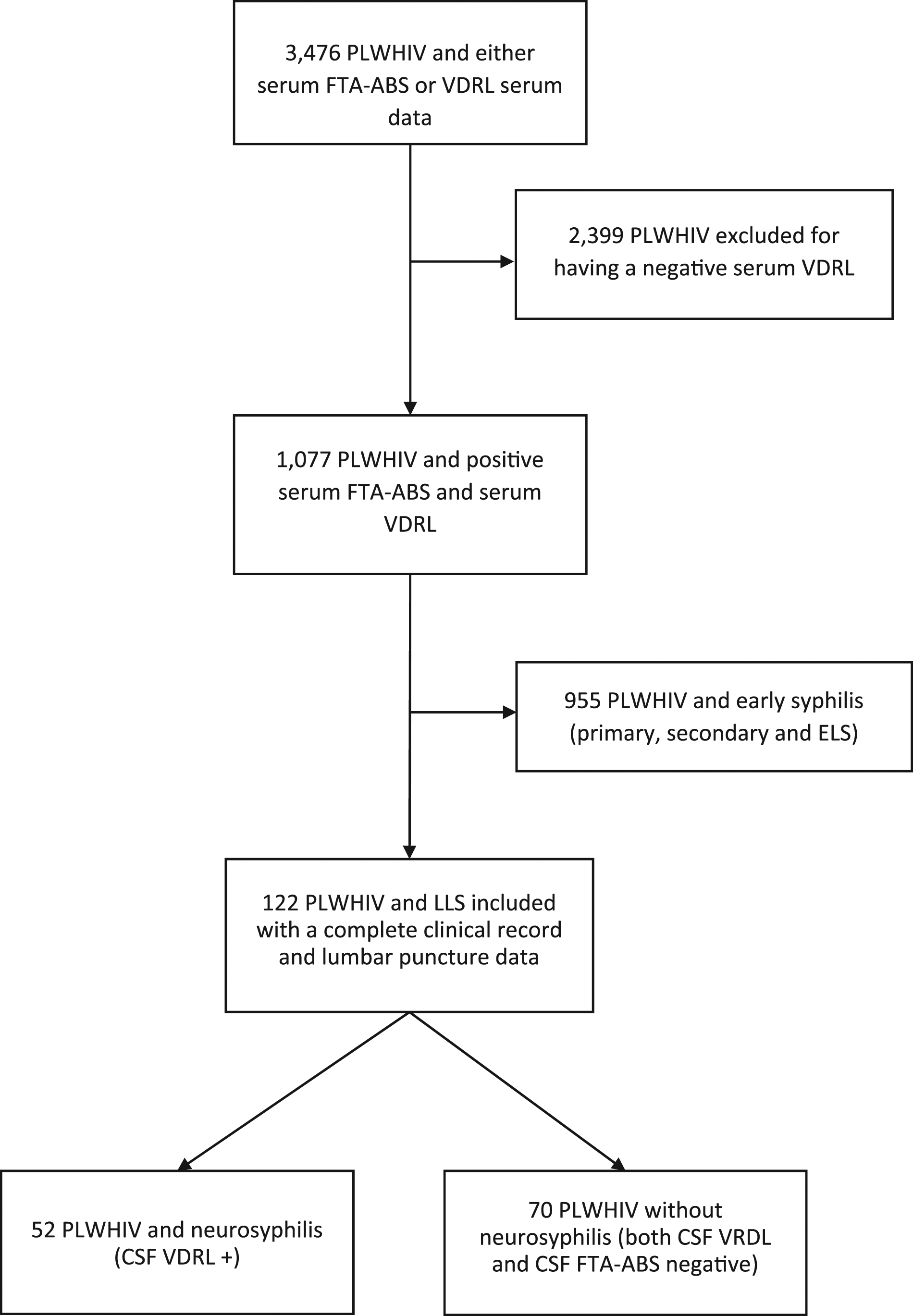
Statistical analyses
Demographic data are presented as continuous and categorical; Student’s t-test and the Mann–Whitney test were used to detect the differences in numerical variables depending on their distribution. In addition, we compared categorical data between groups with X2 and Fisher’s exact test. To assess the relationship between clinical variables and neurosyphilis, we used univariate and multivariate logistic regression. In the first model we included variables we considered to be clinically relevant based on previous studies and we used a backwards elimination approach eliminating those variables with non-significant p-values. Goodness-of-fit and model performance were assessed with the Hosmer–Lemeshow test and c-statistic (area under the receiving operating curve), respectively. All statistical analyses were performed in the R statistical program version 1.3.1073 (Vienna, Austria). Packages used for calculations included “ResourceSelection” and “DescTools.” A two-sided p-value <0.05 was considered as statistically significant.
Results
Demographic, clinical, and laboratory characteristics between groups.
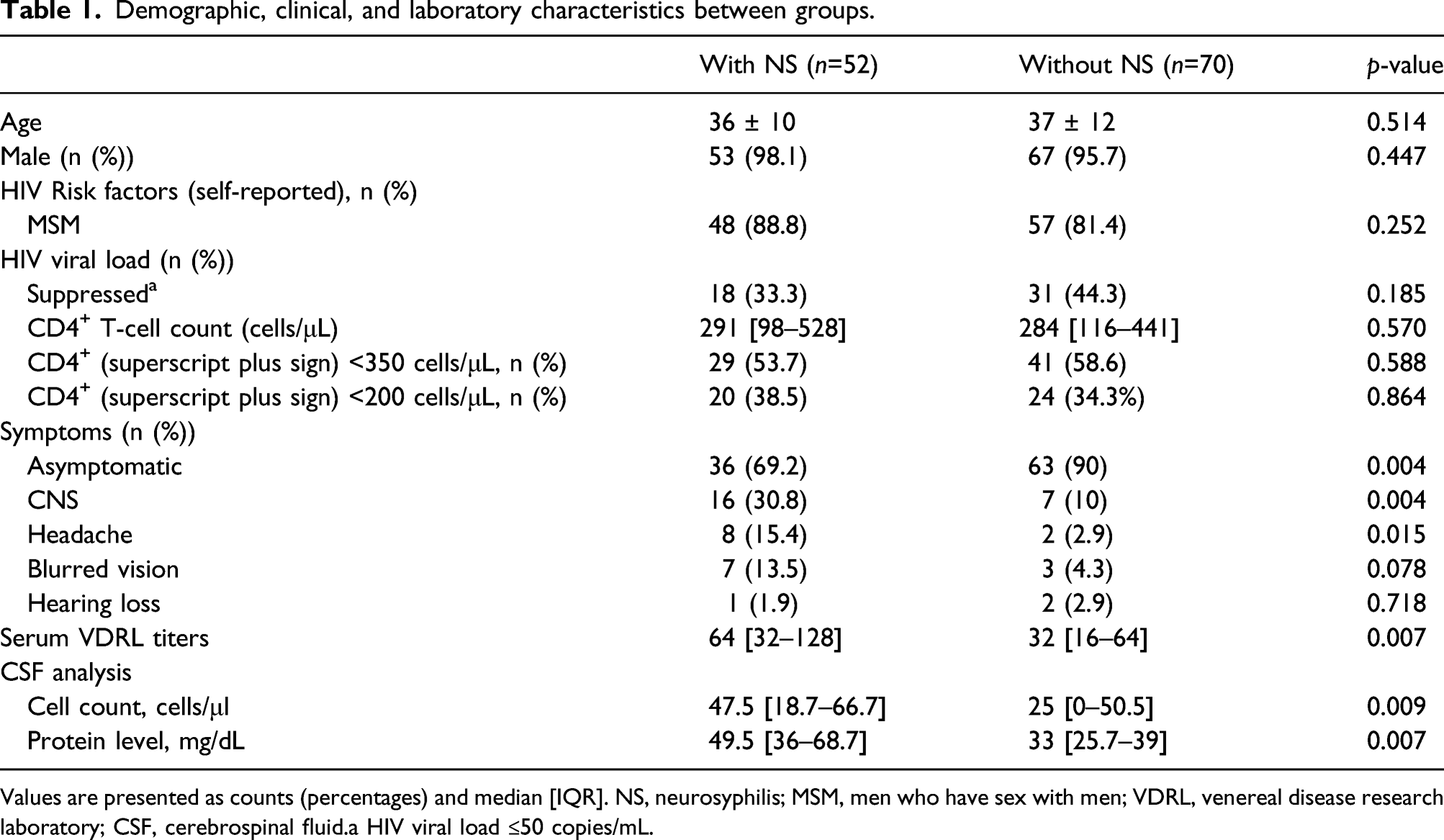
Values are presented as counts (percentages) and median [IQR]. NS, neurosyphilis; MSM, men who have sex with men; VDRL, venereal disease research laboratory; CSF, cerebrospinal fluid.a HIV viral load ≤50 copies/mL.
Univariate analysis of risk factors associated with neurosyphilis.
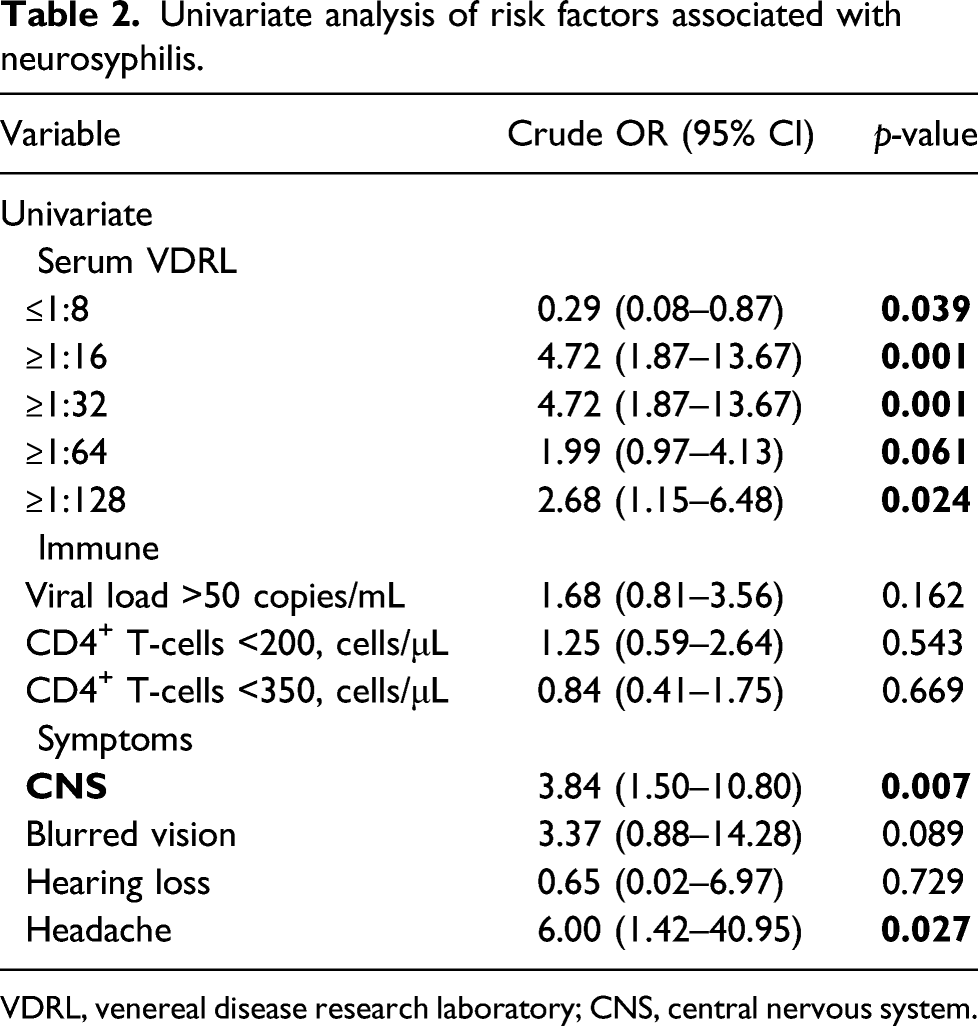
VDRL, venereal disease research laboratory; CNS, central nervous system.
Multiple logistic regression for neurosyphilis.

CNS, central nervous system. C-statistic=0.74. Hosmer–Lemeshow goodness-of-fit test, x2=1.36, df=8, p=0.98. (High p-value means good fit)
Association between VDRL titers and the presence of symptoms with neurosyphilis.
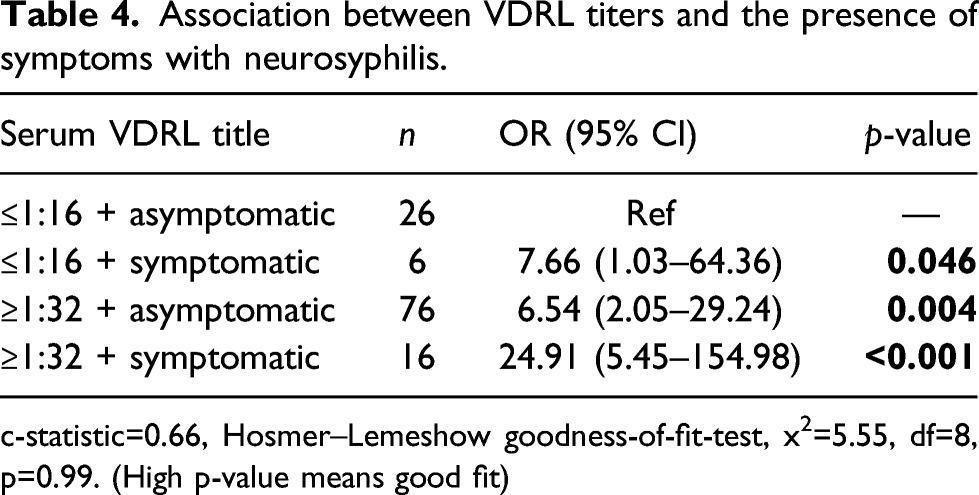
c-statistic=0.66, Hosmer–Lemeshow goodness-of-fit-test, x2=5.55, df=8, p=0.99. (High p-value means good fit)
Discussion
We retrospectively analyzed clinical factors associated with neurosyphilis in people living with HIV with LLS. In the neurosyphilis group, ANS accounted for a considerable number of cases, as consistently reported in previous studies.10,11,17,18,26 In addition, approximately 84% of the patients with neurosyphilis were MSM, representing a high-risk group for HIV and syphilis. Moreover, we found that even low VDRL titers (e.g. ≤1:16) can be associated with neurosyphilis in the presence of neurological symptoms, while in the absence of symptoms, only serum VDRL ≥1:32 was an independent predictor of neurosyphilis. Remarkably, the combination of serum VDRL ≥1:32 plus any neurologic symptom (headache, blurred vision, hearing loss) had at least 5 times greater odds of neurosyphilis (OR 24.91, 5.41 to 154.98; p<0.001). As with other bacterial infections in CNS, Treponema pallidum can cause an infectious arteritis that may affect any vessel in the subarachnoid space surrounding the brain or spinal cord and result in ischemia, thrombosis, and infarction. 16 Thus, the presence of headache is a manifestation of CNS invasion to the parenchyma and blood vessels. Headache by itself appeared to be the symptom with the strongest association—among all neurological symptoms—with neurosyphilis in our studied population.
In our population, we found a median CSF cell count of 25 cells/μl in those without neurosyphilis. In this group, only 44.3% of patients were virologically suppressed, and the median CD4+ T-cell count was 281 cells/μl. Early in the HIV pandemic it was reported that a high proportion of people living with HIV have CSF abnormalities. 20 Spudich et al. 21 found a median WBC of 15 cell/ml in people living with HIV—without CNS infection—who were asymptomatic and non-virological suppressed. In addition, Marra et al. 22 found an association between pleocytosis due to HIV infection—with detectable HIV serum viral load—and CD4+ T-cell count >200 cells/μL. These are characteristics seen in our group without neurosyphilis, as 55.7% of patients were not virologically suppressed. Finally, two patients in the group without neurosyphilis had the diagnosis of cryptococcal meningitis, which could have contributed to the pleocytosis.
Based on the great proportion of ANS in the population, not performing an LP in asymptomatic patients would miss a considerable number of neurosyphilis diagnoses; hence, the absence of neurological signs or symptoms should not discourage a complete evaluation to rule out CNS involvement. In people living with HIV, current European 12 and Canadian 19 guidelines recommend the performance of an LP in patients with a CD4+ T-cell count of <350 cells/μl or a rapid plasma reagin (RPR) titer of ≥1:32 23 . Libois et al. 24 concluded that LP could be restricted to people living with HIV and syphilis and present neurologic manifestations or an RPR titer of ≥1:32. The POLCA Cohort study showed that individuals with serum VDRL titers ≥1:32 were positive for neurosyphilis in 84.6% of the cases. 25 Furthermore, a study by Ghanem et al. 26 found these previously mentioned criteria with a CD4+ T-cell count of <350 cells/μL to be the most reliable in identifying ANS with a sensitivity of 100%. Our results are in line with these previously mentioned studies. All patients with symptoms, particularly neurologic symptoms (e.g. headache), are at increased risk of neurosyphilis, as shown in other studies,17,27 and an LP should be performed to make the diagnosis. However, CDC guidelines since 2010 recommend against LP unless neurologic symptoms are present, stating that CSF examination in absence of neurologic symptoms is not associated with improved clinical outcomes.1,13
Despite the above, guidelines have recognized ANS as a nosological entity,12,28,29 and there is concern regarding that some individuals living with HIV may not be able to clear the spirochete from the CNS. 30 Moreover, recent evidence showed that cognitive impairment was common in individuals with syphilis at high risk of CSF-defined neurosyphilis who did not report neurological symptoms. Considering pre-treatment cognitive performance, Davis et al. 31 reported that the risk of cognitive decline was significantly lower in those with CSF pleocytosis treated for neurosyphilis compared to those without CSF pleocytosis and not treated for neurosyphilis. A follow-up of individuals in the same study showed that those with syphilis experienced 40% cognitive decline in the year after syphilis diagnosis.
Data regarding progression in LLS are lacking and some experts recommend CSF assessment in asymptomatic people living with HIV with LLS and lymphocyte CD4+ T-cell count <350 cell/μL or RPR titers ≥1:32, especially for those who present serological failure (i.e. failure of anti-treponemal titers to decline four-fold or greater after treatment or if there is a documented four-fold increase after initial decline) or serofast (i.e. the persistence of low antibody titer of an NTT 1–2 years after treatment of early syphilis), and in those given an alternative treatment (e.g. tetracyclines) for late syphilis. 12 Due to the high rate of ANS in our sample, we considered that asymptomatic patients with a serum VDRL titer ≥1:32 should undergo an LP to diagnose neurosyphilis. Our results are especially useful in a clinical setting where VDRL is the only available NTT.
Our study has several limitations. First, the retrospective analysis from a single academic center may lead to a potential selection bias. Also, some subcategories with a small sample size—such as VDRL ≤1:16 and ≥1:32 and symptomatic—yielded wide confidence intervals and should be interpreted carefully. Second, a considerable number of our patients were categorized as LLS due to their unknown disease duration. This could misclassify some patients with early latent syphilis as LLS. Third, LP was performed based on either CD4+ T-cell level <350 cell/μL, serum VDRL titers, and the presence of symptoms; using either criterion limited our ability to detect CD4+ T-cell differences between in those with and without neurosyphilis. However, a strength of our study is that neurosyphilis was diagnosed with the gold standard (i.e. VDRL in CSF), and the group without neurosyphilis had both negative CSF VDRL and CSF FTA-ABS plus an alternative diagnosis of their symptoms which makes the possibility of neurosyphilis extremely unlikely. Thus, the analysis was made with true positives and true negatives. Although our sample was not randomly obtained from the medical records, the patients included in the study are an heterogeneous sample of individuals referred to our hospital from the first contact clinics and second-level hospitals across the state of Jalisco, which improves its external validity.
Conclusion
The presence of neurological symptoms, particularly headache, was associated with neurosyphilis in people living with HIV with LLS. In the absence of symptoms, a serum VDRL ≥1:32 showed an increased risk of neurosyphilis in our population.
Key messages
• In people living with HIV and late latent syphilis, the presence of neurological symptoms were predictors of neurosyphilis irrespective of their viral load and CD4+ T-cell count. • A serum VDRL ≥1:32 increased the risk of neurosyphilis in patients with late latent syphilis with or without any symptoms. • Asymptomatic NS is still a topic of debate that considering the recent evidence, should be sought, and treated.
Footnotes
Acknowledgements
We thank the medical staff, research assistants, and the nursing department for their invaluable help that made this study possible.
Author Contributions
PMA, LGH, VVRH, FAL, SZQ, MAZ, and JAV designed the study. LAGH and JAV supervised the study. PMA, GAAS, LAQF, ICS, KRS, and JCVL performed data curation. GAA and PMA conducted the formal analysis. All authors contributed to results interpretation. The original draft was elaborated by PAM, GAAS and MAZ, while the rest of the authors constantly reviewed and edited the article for important clinical content. All authors approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient Consent Statement
Given the retrospective nature of this study, patient consent was not required before data collection. The University’s local ethics committee approved the study and allowed for data collection and analysis.
Availability of Data
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
