Abstract
Background
Understanding care patterns of persons living with HIV prior to diagnosis can inform prevention opportunities, earlier diagnosis, and engagement strategies. We examined healthcare utilization among HIV-positive individuals and compared them to HIV-negative controls.
Methods
Data were from a retrospective cohort from Manitoba, Canada. Participants included individuals living with HIV presenting to care between 2007 and 2011, and HIV-negative controls, matched (1:5) by age, sex, and region. Data from population-based administrative databases included physician visits, hospitalizations, drug dispensation, and chlamydia and gonorrhea testing. Diagnoses associated with physician visits were classified according to International Classification of Diseases chapters. Conditional logistic regression models were used to compare cases/controls, with adjusted odds ratios (AORs) and their 95% confidence intervals (95% CI) reported.
Results
A total of 193 cases and 965 controls were included. Physician visits and hospitalizations were higher for cases, compared to controls. In the 2 years prior to case date, cases were more likely to be diagnosed with “blood disorders” (AOR: 4.2, 95% CI: 2.0–9.0), be treated for mood disorders (AOR: 2.4, 95% CI: 1.6–3.4), and to have 1+ visits to a hospital (AOR: 2.2, 95% CI: 1.4–3.6).
Conclusion
Opportunities exist for prevention, screening, and earlier diagnosis. There is a need for better integration of healthcare services with public health.
Introduction
Despite the advent of combined antiretroviral therapy (ART), many people living with HIV continue to present at an advanced stage of the disease.1–4 In industrialized countries, late presenters (defined as those who present with a CD4+ cell count of <200 cells/mm3 or who progress to AIDS within 1 year of presentation) account for 15–43% of new diagnoses. 4 In Canada, where universal healthcare exists, 39% of reported new HIV cases present at this late stage, 4 while in the province of Manitoba, approximately 27% of new HIV diagnoses made in 2016 were in patients with CD4 count <200 cells/mm3. 5
There are several important implications of delayed recognition of HIV.6–10 First, the longer patients have untreated disease the more opportunities there are for transmission.11,12 Second, healthcare costs correlate inversely with CD4+ cell counts13,14; the annual cost for a patient with a CD4 count of <200 cells/mm3 is approximately double that of a patient with a count that is >500 cells/mm3. 13 Finally, late presentation is known to increase mortality.15–17
The reasons for late presentation are multiple and include both patient- and provider-related factors. Research has demonstrated that people living with HIV often present to healthcare providers at least once in the years immediately prior to their diagnosis.18–22 Duffus et al. 19 observed that late presenters sought care more often than those diagnosed early in the disease. They noted that in 33% of prior visits, a high-risk feature, such as injection drug use was present, and yet testing for HIV was not performed. Shaw et al. 23 found that in the 5-year period prior to their diagnosis, serological testing for other sexually transmitted infections (STIs) was higher among individuals living with HIV, compared to HIV-negative controls. A better understanding of healthcare seeking patterns prior to HIV diagnosis may offer insights into opportunities for HIV prevention, as well as opportunities for earlier HIV testing.23,24
Using population-based administrative and public health databases linked to clinical data, our study aimed to explore the healthcare patterns of individuals living with HIV prior to their initiation into HIV care. By doing so, we aimed to understand missed opportunities for improved screening and earlier diagnosis for those individuals living with HIV, and to identify potential points of contact for prevention and care interventions.
Methods
Study setting and population
We conducted this study as a part of the Missed Opportunities for Diagnoses Epidemiological Study (MODES), a retrospective study using population-based administrative databases linked to HIV clinical data. The two clinical sites of the Manitoba HIV Program (MHP), based in Winnipeg, the capital city of the province of Manitoba, provided care to almost all (i.e., >95%) adults living with HIV in Manitoba. As of 2018, there were approximately 1400 persons living with HIV receiving care through the MHP. 5 Manitoba Health, Seniors, and Active Living (MHSAL) is the single insurer responsible for payment of health services. Information on physician claims, hospital discharge abstracts and pharmaceutical dispensations is contained within population-based administrative datasets, managed and maintained by MHSAL.
Ethical approval was obtained from the Institutional Review Boards (IRB) at the University of Manitoba and the Winnipeg Regional Health Authority. This study was also approved by the Assembly of Manitoba Chiefs’ Health Information Research Governance Committee and the Health Information Privacy Committee of MHSAL.
Participants
Similar to a previously published case-control study, 23 cases were defined as individuals living with HIV, 18 years of age and older, who were newly enrolled in the MHP between 1 January 2007 and 31 December 2011. Recruitment took place from 1 January 2013 to 31 December 2013. From a potential population of 303 persons enrolled in the MHP within the study period, 208 were eligible for the study. Of these 208, 186 consented to participate in the study (consent rate = 76%). An additional 15 patients who were deceased at the time of study recruitment were added to the 186 patients for a final sample of 201 individuals. Ethics board review waived the need for informed consent for these 15 individuals. Of the 201 individuals, 193 were continuously enrolled in the 5-year period prior to case date and included in the final analysis. Case date was defined as the date the case enrolled in the MHP.
Controls were HIV-negative individuals drawn from the general population who were age-, sex- and region-matched to the cases at a 5:1 ratio. Using a previously published Canadian algorithm, 25 controls were deemed “HIV-negative” if they had no existing claims for ICD-9-CM codes [042 or 043] or ICD-10-CA codes [Z20] in physician and hospital claims.
Data sources
Scrambled (i.e., de-identified) personal health identification numbers (PHINs) of cases and controls were used to link the following datasets.
Clinical data
Clinical data were obtained on individuals living with HIV who had provided written informed consent and were abstracted from MHP sites by a research assistant and reviewed by MLB and KK, using a standard data abstraction tool. Information included basic demographic information, date of entry into the MHP, risk factors associated with HIV acquisition, and selected clinical characteristics.
Manitoba health insurance registry
Since 1970, Manitoba Health, Seniors and Active Living (MHSAL) has maintained the Manitoba Health Insurance Registry (“The Registry”) to track individuals registered to receive health services in Manitoba. Date fields for registration, birth, entry into province, and migration in/out of province provide information to track residents for longitudinal analyses, and are updated annually. 26
Physician claims and hospital discharge abstract data
Universal health coverage is provided by MHSAL and computerized records of all physician billing claims and hospitalizations are maintained by MHSAL. Each physician claim and hospitalization includes diagnoses coded using the International Classification of Diseases (ICD) system. For outpatient physician claims, only one diagnosis is entered into MHSAL databases, with diagnoses coded up to three digits using ICD-9-CM. For inpatient hospitalizations, multiple diagnoses can be coded per hospital visit; ICD-9-CM was used up to fiscal year 2004/2005, at which point the coding system was changed to ICD-10-CA. For both time periods, ICD codes were coded to the 5-digit level. The accuracy of these administrative health databases has been demonstrated.27–29
Drug program information network data
Information on all outpatient prescription dispensations is collected in the Drug Program Information Network (DPIN) dataset, which has been online since fiscal year 1995/1996. Within the DPIN, pharmaceutical products are identified by their unique drug identification numbers (DIN). 29
Sexually transmitted infections surveillance data
MHSAL maintains the STI Surveillance Database, which contains socio-demographic, diagnostic, and treatment information on all positive cases of
Measures
With the exception of bacterial STIs, administrative data were searched for any evidence of healthcare utilization in the 2-year period prior to this date for both cases and controls. A 5-year period was used for bacterial STIs, due to the low number of infections recorded.
Physician visits
Physician visits prior to case date were determined using date of physician visit. Only unique visits, based on visit date and ICD-9-CM code were counted. Diagnoses were categorized according to ICD-9-CM chapters, with binary indicators denoting the presence of a diagnosis for any particular chapter. Any inpatient records (i.e., physician claims during hospital stays) were not counted.
Hospitalizations
Hospitalization prior to case date was defined by hospital admission date, and a binary indicator denoting a hospital visit created, with visits lasting more than 24 h included as a hospitalization.
Pharmaceutical dispensations
Dispensation date was used to determine pharmaceutical dispensations prior to case date; using DIN, and based on the work of Marrie et al. and Shaw et al.,31–33 the following prescription categories were created: antibiotics, asthma, diabetes, heart disease, hyperlipidemia, hypertension, respiratory illness, anxiety, bipolar disorder, depression, mood disorders, and schizophrenia.
Chlamydia/Gonorrhea infections
Using the MHSAL STI surveillance data, separate binary indicators were created for CT and GC, indicating at least one positive CT or GC test in the 5 years prior to case date. For all measures and to address confounding due to indication bias (i.e., co-occurrence of healthcare utilization with an HIV diagnosis), a washout period of 180 days prior to case date was used. Thus, for both cases and controls, any utilization within a 180-day period prior to the case date was not counted. Additionally, entry into MHP was chosen as ‘case date’ instead of the date of first positive HIV result because not all individuals living with HIV have a previous positive HIV test on record in Manitoba, prior to their entry into MHP. Analysis of individuals with a previous positive HIV test found that the average gap between a positive result and entry into care was less than 60 days.
Statistical methods
Only individuals who were continuously registered (i.e., no evidence of out-migration) and had a first registered date at least five years prior to the study period with the MHSAL registry for the duration of the study period were included in the study. Conditional logistic regression was used to test for the association between HIV status and the likelihood of each measure of healthcare utilization; odds ratios (ORs) and 95% confidence intervals (95% CIs) are reported. All analyses were performed using Stata 13 (College Station, TX).
Results
Selected characteristics of participants.
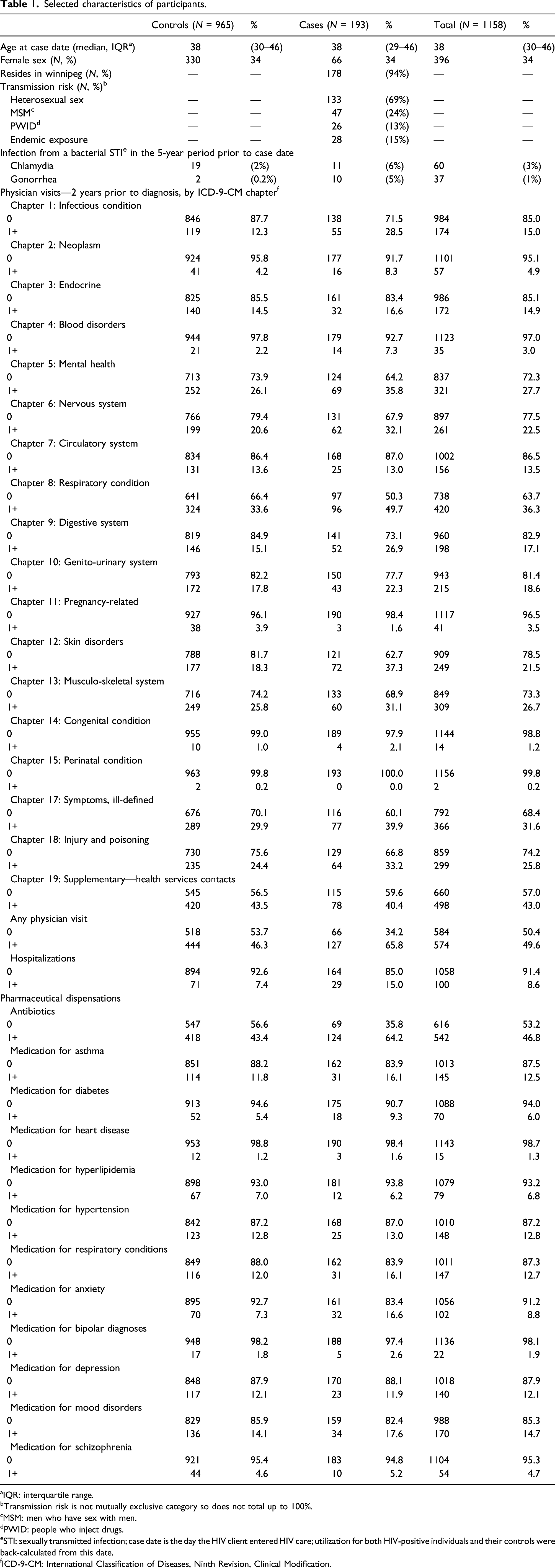
aIQR: interquartile range.
bTransmission risk is not mutually exclusive category so does not total up to 100%.
cMSM: men who have sex with men.
dPWID: people who inject drugs.
eSTI: sexually transmitted infection; case date is the day the HIV client entered HIV care; utilization for both HIV-positive individuals and their controls were back-calculated from this date.
fICD-9-CM: International Classification of Diseases, Ninth Revision, Clinical Modification.
Physician visits
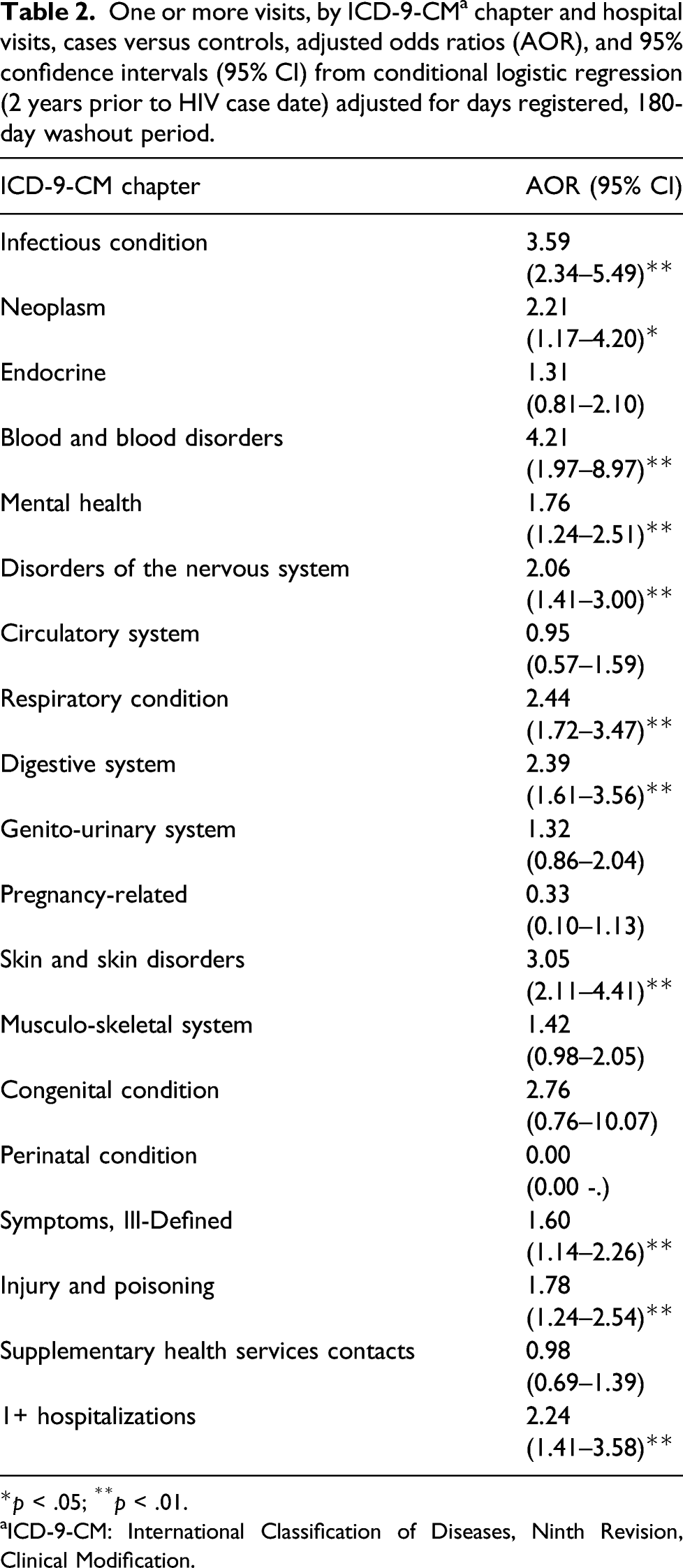
One or more visits, by ICD-9-CM a chapter and hospital visits, cases versus controls, adjusted odds ratios (AOR), and 95% confidence intervals (95% CI) from conditional logistic regression (2 years prior to HIV case date) adjusted for days registered, 180-day washout period.
*
aICD-9-CM: International Classification of Diseases, Ninth Revision, Clinical Modification.
Top five most frequent Chapter 1 and Chapter 4 diagnoses from physician visits, HIV-positive participants (2 years prior to HIV case date), and 180-day washout period.

aInternational Classification of Diseases, Ninth Revision, Clinical Modification.
Hospitalizations
Hospital visits were also significantly associated with HIV status; those with one or more hospital visits in the 2-year period prior to case date were at over twice the odds of being HIV positive (AOR: 2.2, 95% CI: 1.4–3.6; Table 2).
Prescription use
One or more dispensations, by type of prescription, cases versus controls, adjusted odds ratios (AOR), and 95% confidence intervals (95% CI) from conditional logistic regression (2 years prior to HIV case date) adjusted for days registered, 180-day washout period.

*
Bacterial Sexually transmitted infections
Bacterial infection and HIV, odds ratios (OR), and 95% confidence intervals (95% CI) from conditional logistic regression.
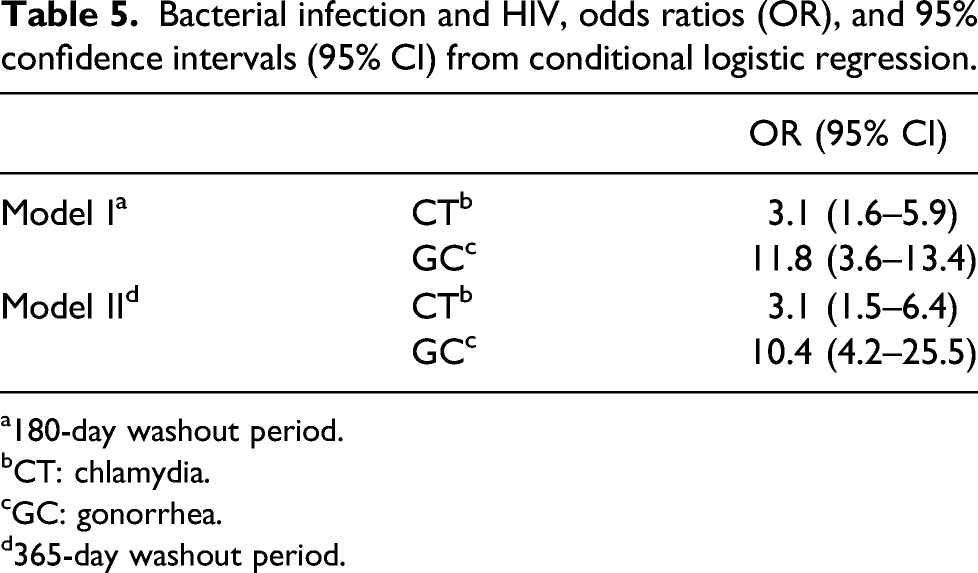
a180-day washout period.
bCT: chlamydia.
cGC: gonorrhea.
d365-day washout period.
Discussion
Building on earlier work demonstrating the higher likelihood of serological tests among individuals eventually diagnosed with HIV,
23
our results demonstrate that healthcare utilization was higher among those who were subsequently diagnosed with HIV, compared to their controls. Our analyses also reveal that utilization
With respect to the association between diagnoses of blood and blood disorders and subsequent HIV, the MHP gets referrals for patients presenting with lymphadenopathy, anemia and/or thrombocytopenia; disorders associated with HIV.34–36 A more precise predictive decision-making tool incorporating these disorders in clinical practice may lead to earlier diagnoses of HIV. In describing the co-occurring epidemics of HIV, and substance use, Singer et al. were one of the first to place HIV within a “syndemics” framework,37–39 recognizing that risk and vulnerability of individuals to HIV occurs in environments of harmful social/structural conditions. Thus, these harmful conditions contribute to poorer health outcomes, including HIV40,41; the higher use of healthcare services of cases in our study aligns with this perspective.
Our findings can contribute to frameworks used to examine missed opportunities for HIV diagnoses.
23
Several factors have been proposed to explain the heterogeneity in missed opportunities for HIV diagnosis. For example,
More research to understand the structural and practice level issues contributing to gaps in prevention and care services may help to optimize HIV prevention.
47
For example, in Manitoba, responsibility for follow-up of individuals testing positive for CT or GC, including partner notification, is left to the discretion of the testing practitioner, who
At the same time, individual-level behavioral factors are also likely at play 51 ; strengthening community-based prevention efforts, such as outreach to high-risk individuals and other priority populations, and which focus on health promotion, a more enabling environment, and addressing other needs such as housing, mental health and addictions issues may also reduce gaps in prevention.52,53 Our findings illustrating the higher likelihood of dispensations for mental health conditions among individuals with HIV supports prioritization of mental health services. Community organizations and primary and public healthcare have historically been underfunded in Canada. 44
Strengths and limitations
Our study had a number of strengths, including the population-based nature of the MHP, as well as empirical data on healthcare utilization. Although we sampled only a portion of the total MHP population available, the socio-demographic characteristics of our sample did not differ substantially from other publications using the entire MHP population. 54 Our control group allowed for comparisons to the general population.
Our study had some limitations. CT/GC incidence is likely underestimated as data collected by MHSAL is based on a passive surveillance system. However, since populations at higher risk for HIV and other STIs are less likely to access care, this underestimation should have driven differences between cases and controls to the null. Case date was defined as the date of MHP entry; this date was chosen instead of the date of first positive HIV result as not all HIV-positive individuals had a previous positive HIV test on record in Manitoba prior to their entry into MHP. This gap between first positive HIV test and entry into MHP care could have potentially biased rates of CT/GC incidence and testing; however, among those with a previous positive HIV test, the average gap between a positive result and entry into care was less than 60 days. Thus, our 180-day washout period was meant to err on the side of caution. Controls were assumed to be HIV-negative; but are more accurately referred to as not known to be positive.
In conclusion, our results demonstrate distinct patterns of healthcare utilization amongst HIV-positive individuals, prior to their HIV diagnosis. Stronger efforts to integrate the work of public health personnel, community organizations, and general practitioners are needed in the Canadian context to improve HIV prevention efforts, as well as support earlier HIV testing and linkage to care for those who test positive.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Gilead Foundation. SYS is supported by a Canada Research Chair in Program Science and Global Public Health (Tier II).
