Abstract
Objectives:
The objective of this study was to describe the real-world use and effectiveness of dolutegravir-based regimens (DBRs) in routine clinical practice in the United Kingdom.
Methods:
Retrospective analysis was conducted using data from four National Health Service trusts using Climate-HIV, an electronic case record system. Eligible patients were aged ≥18 years with HIV-1 infection who were prescribed a DBR from December 2012 to March 2018. Outcome measurements were accessed at DBR initiation and at weeks 24, 48 and 96 and the last recorded visit up to the extraction date (last measurement). The primary endpoint was the proportion of patients with HIV-1 RNA <50 copies/mL at Week 48.
Results:
The study cohort included 934 patients; 337 (36%) were female, 414 (47%) were white and 717 (77%) were treatment experienced (TE). The Kaplan–Meier estimated probability of achieving HIV-1 RNA <50 copies/mL at 48 weeks was 96% for treatment-naive (TN) patients and 86% for TE patients. Median times to viral suppression (<50 copies/mL) were 49 and 57 days for TN and TE patients with detectable baseline viral load, respectively, according to Kaplan–Meier analysis. Median follow-up time was 377 days (interquartile range: 131–683). At last measurement, 87% (809/934) of patients remained on a DBR; among those patients, 681 (84%) had HIV-1 RNA <50 copies/mL.
Conclusions:
High levels of virologic suppression and low rates of discontinuation of DBRs were seen in a large, diverse, UK-based population with HIV-1 infection. These findings are broadly consistent with efficacy data from phase III studies.
Introduction
Dolutegravir is a second-generation integrase strand transfer inhibitor indicated for treatment of HIV-1 infection in combination with other antiretroviral (ARV) agents. 1 The clinical efficacy and safety of dolutegravir-based 3-drug regimens (3DRs) have been demonstrated in phase III randomised control trials in treatment-naive (TN)2–5 and treatment-experienced (TE)6–8 patients with HIV-1 infection. Following that, clinical studies with dolutegravir-based 2-drug regimens (2DRs) have demonstrated noninferiority of 2DRs in achieving or maintaining viral suppression compared with dolutegravir-based 3DRs.9–12 As a 3DR, dolutegravir demonstrated superior efficacy in five separate clinical studies versus various other non–dolutegravir-based 3DRs in TN and TE patients.6,8 Dolutegravir has also been shown to be effective in patients with HIV-1 infection with multidrug ARV resistance.6,8 In addition, dolutegravir has a favourable safety profile, few drug–drug interactions and a high barrier-to-resistance profile.8,11,13 As a result of these clinical trials, dolutegravir-based regimens (DBRs) are listed as a preferred treatment option in international guidelines, including those from the US Department of Health and Human Services, 14 the International Antiviral Society, 15 the World Health Organization 16 and the European AIDS Clinical Society. 17
Real-world cohorts are essential to understanding how the results of clinical trials translate to real-world settings and diverse patient populations. Several cohort studies have assessed DBRs in real-world settings. In these cohorts, many patients remained on DBRs because of the high rates of achieving or maintaining virologic suppression and the low rates of discontinuation due to adverse events (AEs).18–22 Herein, we describe the real-world use and effectiveness of DBRs in a retrospective analysis of routine clinical practice in the United Kingdom from the Climate-HIV database.
Methods
Study design
This study is a retrospective analysis of data from the electronic case record system Climate-HIV, a database pooled from four participating hospitals in the United Kingdom (North Middlesex University Hospital National Health Service (NHS) Trust, Nottingham University Hospitals NHS Trust, Birmingham Heartlands Hospital and Homerton University Hospital NHS Foundation Trust). The study time period was from December 2012 to March 2018 (the first records of DBRs in Climate-HIV). Data were collected at the initiation of DBR (index date) and at follow-up time points of weeks 24, 48 and 96 as well as at the last recorded visit. To account for real-world data collection and lag in test data, a window of ±6 weeks around each follow-up milestone (weeks 24, 48 and 96) was used to capture measurements. Patients were followed up until the earliest of last record, death, end of available data or switch/discontinuation of DBR (last measurement). The study protocol was approved by the Integrated Research Application System, which covers necessary approvals from the Research Ethics Committee (Project ID 225435; approved 20 July 2018) and the Health Research Authority (Project ID 225436). Because of the study design and data source, informed consent was not required.
Participants
Patients aged ≥18 years with a recorded diagnosis of HIV-1 infection prior to DBR initiation (index date), who were treated at one of the Climate-HIV–participating hospitals, received their first DBR during the study period and had an HIV-1 RNA measurement recorded within the prior 9 months were eligible for the study. Patients were excluded if they had a diagnosis of HIV-2 infection.
Study endpoints
The primary endpoint was the proportion of patients with HIV-1 RNA <50 copies/mL at 48 weeks (snapshot analysis). Secondary endpoints included changes in HIV-1 RNA and CD4+ cell count from baseline, mean time to achieve target HIV-1 RNA (<50 copies/mL), treatment failure (switch/discontinuation of DBR) and virologic failure. Switching was defined as patients who switched to a non–DBR-containing regimen but did not include changing of regimens that used identical ARV drugs (e.g., change to/from a single-tablet regimen or generic formulation). Discontinuation was defined as patients who did not receive a subsequent prescription for ARVs for >6 months; this may have been related to death, change to another clinic or lack of engagement (i.e., lost to follow-up). Virologic failure was defined as prior undetectable HIV-1 RNA measurement (i.e., <50 copies/mL) in patients who switched/discontinued a DBR, viral load ≥50 copies/mL and subsequent measurement of ≥200 copies/mL; prior HIV-1 RNA <50 copies/mL measurement and two consecutive viral load measurements ≥50 copies/mL or viral load ≥50 copies/mL with subsequent measurement ≥200 copies/mL and documented virologic failure as reason to switch/discontinue treatment, <1 log decrease after 4 weeks of treatment with a DBR (TN patients only) or viral load >400 copies/mL after 16 weeks of treatment with a DBR (TN patients only). Data for secondary endpoints were collected at last measurement as well as at weeks 24, 48 and 96 from the index date. Treatment persistence, defined as the probability of remaining on a DBR over time, was determined using Kaplan–Meier analysis. The probability of achieving HIV-1 RNA <50 copies/mL over time for both TN and TE patients was also determined using Kaplan–Meier analyses.
Statistical analysis
Subpopulations for analysis included TN versus TE. Patients were considered to be TE if they had a record of receiving any prior ARV regimen. Data regarding demographics, ARV regimens, virologic outcomes and reasons for switch were summarised with descriptive statistics. Patients whose final record was extracted before the 24-, 48- and 96-week snapshots were not included in further analysis at these time points. Because of the retrospective non-interventional design of this study, only data that were already recorded in the database could be collated.
Results
Patient characteristics and disposition
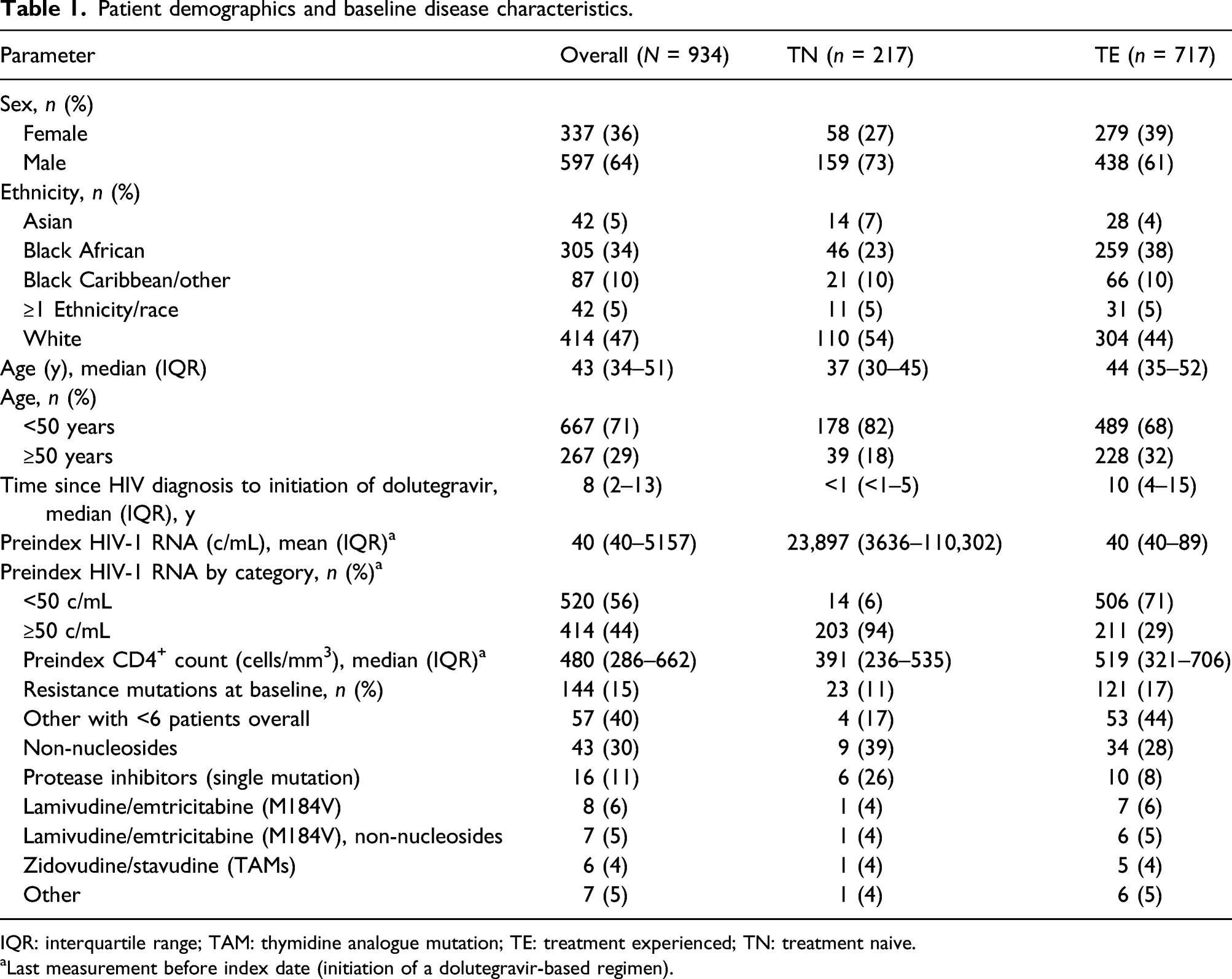
Data for 1204 patients initiating a DBR at a participating site were extracted from Climate-HIV; 270 patients were excluded because they did not meet inclusion criteria (Figure 1). Therefore, the study cohort included 934 patients; of those, 217 were TN and 717 were TE. Of the overall population, 36% (n = 337) of patients were female (Table 1). The median age was 43 years; 71% of patients (n = 667) were aged <50 years. The majority of the population was white (47%); 34% of patients were Black African, 10% were Black Caribbean/other, 5% were of Asian descent and 5% were ≥1 race/ethnicity. Before the index date, median (interquartile range (IQR)) HIV-1 RNA was 23,897 copies/mL (3636–110,302) for TN patients and 40 copies/mL (40–89) for TE patients. The median times from HIV-1 diagnosis (or transfer into the system in the absence of a date of confirmed diagnosis) to the index date were 6 months for TN patients and 10 years for TE patients. For TE patients, the median number of regimens since diagnosis was 4 (IQR: 3–6) and 27% (n = 193/717) had previous exposure to integrase inhibitors. Resistance mutations were recorded in 17% (n = 121/717) of TE patients. The majority of patients (94% (n = 880)) initiated a once-daily DBR, with abacavir/dolutegravir/lamivudine being the most common regimen (61% (n = 533)). Twice-daily DBRs were initiated in 3% of patients (n = 27), and 5% of patients (n = 42) were taking a 2DR. Disposition of all patients initiating a DBR from December 2012 to March 2018 at four climate-HIV–participating hospitals. AE: adverse event; DBR: dolutegravir-based regimen. Patient demographics and baseline disease characteristics. IQR: interquartile range; TAM: thymidine analogue mutation; TE: treatment experienced; TN: treatment naive. aLast measurement before index date (initiation of a dolutegravir-based regimen).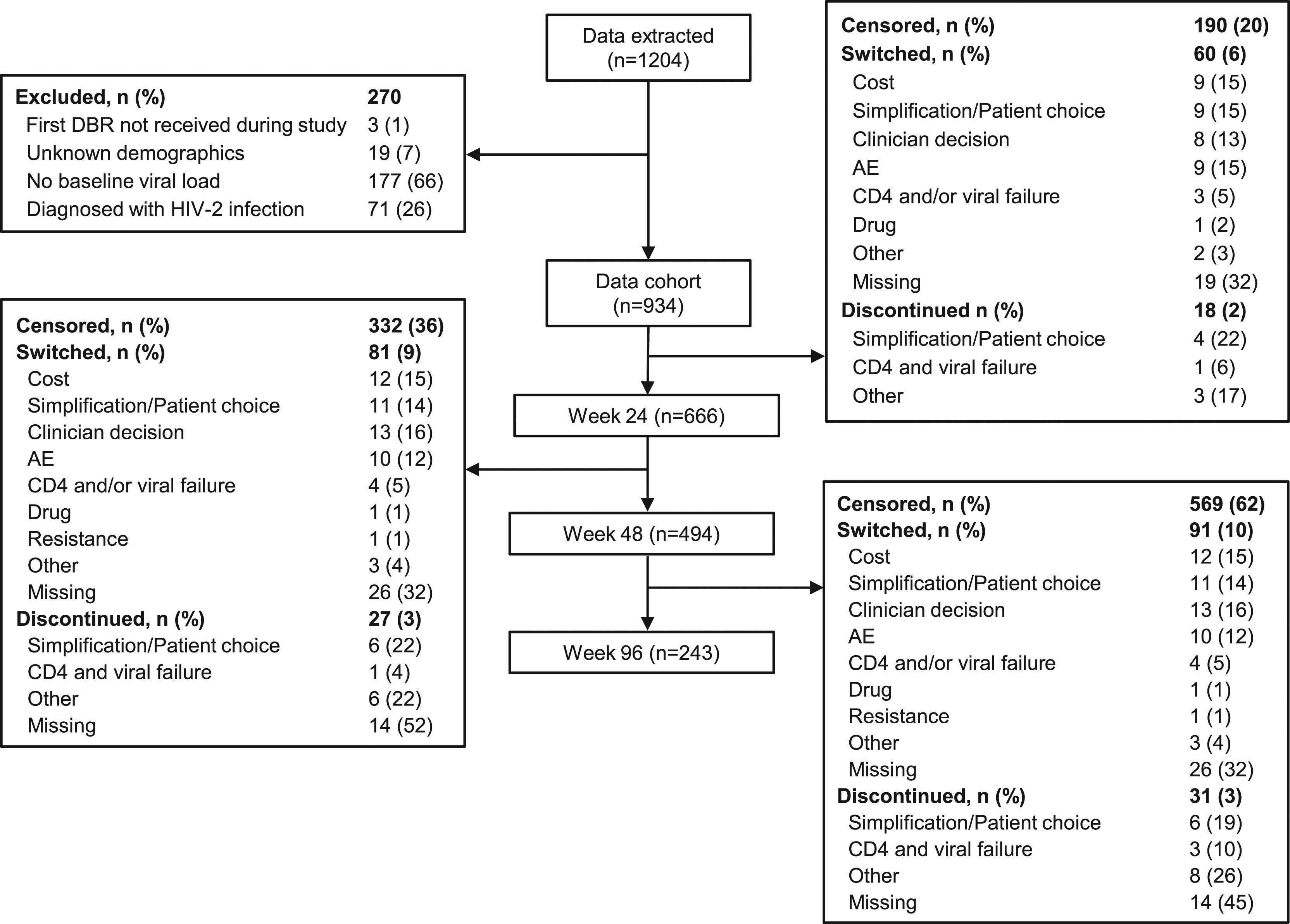
Seven hundred forty-four patients remained in the cohort at 24 weeks; 190 patients were excluded due to their final records being extracted prior to the 24-week snapshot. Of these patients, 90% (n = 666/744) remained on a DBR. Among the patients who remained on a DBR at 24 weeks, excluding those with final records extracted prior to the 48-week snapshot (n = 142), 94% (n = 494/524) remained on a DBR. At 96 weeks, 237 patients were excluded because of premature data and 98% (n = 243/247) remained on a DBR. In addition to time point snapshots, data were collected at last measurement, the earliest last record, death, end of available data or at switch/discontinuation of DBR. The median follow-up time to last measurement was 377 days (IQR: 131–683; Supplementary Table). At the last recorded measurement, 87% (n = 809/934) of patients remained on a DBR, and Kaplan–Meier analysis indicated the rates of persistence of DBRs were 96% for TN and 86% for TE patients over a median follow-up period of 362 and 385 days, respectively (Figure 2). Switches and discontinuations infrequently occurred, with 13% of patients switching or discontinuing a DBR (n = 125; n = 22 TN and n = 103 TE). Switching off a DBR was more common among TE patients (11%) than TN patients (6%), with a median time (IQR) to switch or discontinuation of 101 (30–248) and 124 days (63–242), respectively. For TN patients, the most common reason for switching when recorded was clinician decision (n = 5 (36%)), whereas cost reduction (n = 13 (16%)) was the most common for TE patients. Adverse events collected from the Climate-HIV database as reasons for switch included those that were gastrointestinal (n = 4), renal (n = 3) and central nervous system (n = 2) in nature; skin rash (n = 2); osteopenia (n = 1) and fatigue (n = 2). Kaplan–Meier analysis of time to switch/discontinuation. DTG: dolutegravir.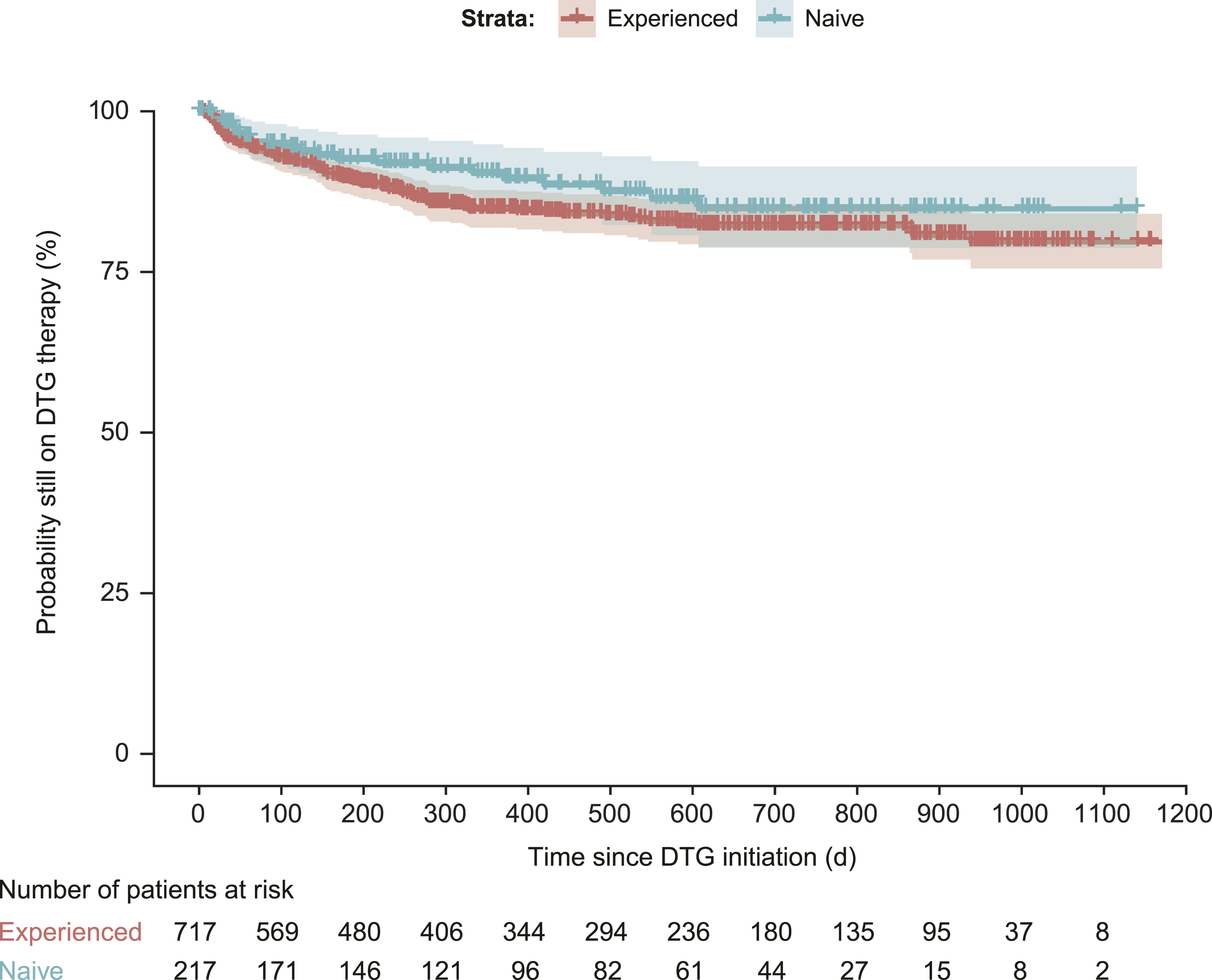
Clinical outcomes
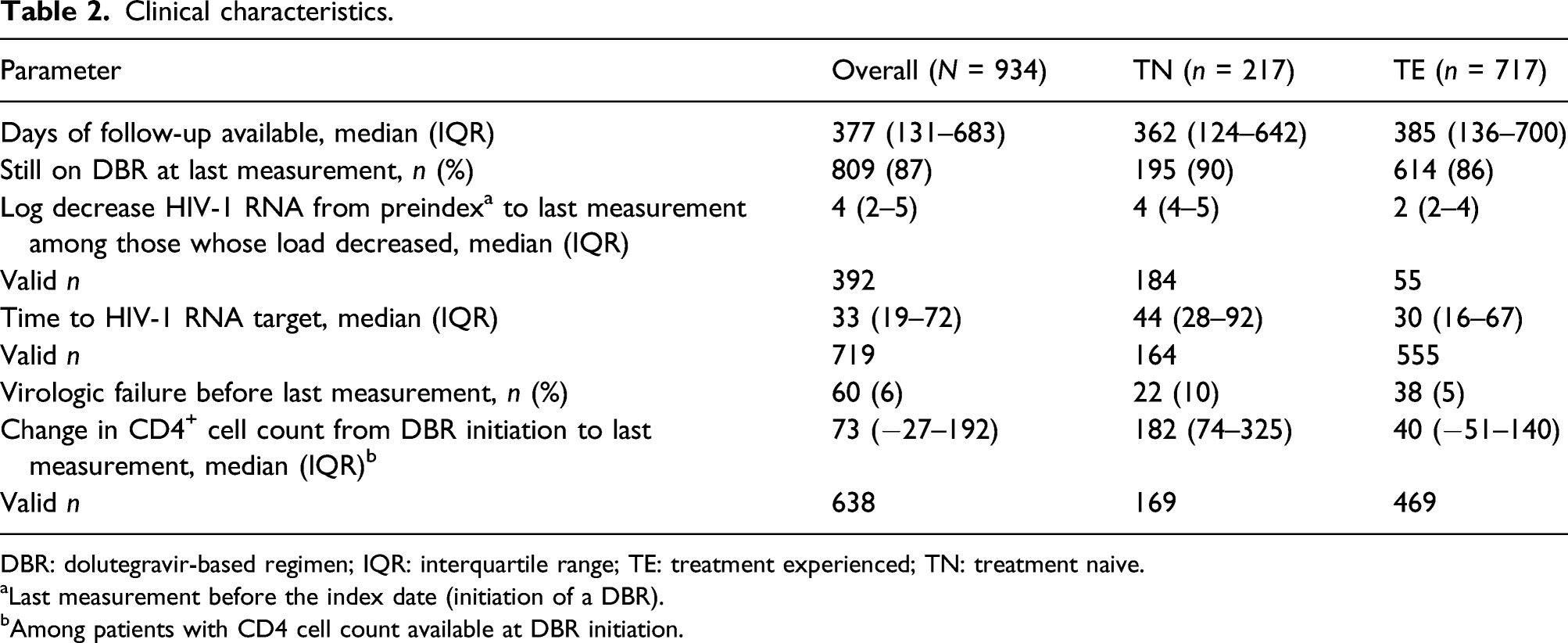
In patients with HIV-1 RNA ≥50 copies/mL at the index date, Kaplan–Meier analysis indicated that the probability of achieving HIV-1 RNA <50 copies/mL at 48 weeks was 96% for TN patients (Day 350) and 86% for TE patients (Day 351; Figure 3). Median times to achieve HIV-1 RNA <50 copies/mL were 49 days (IQR: 40–63) for TN patients and 57 days (IQR: 40–75) for TE patients according to Kaplan–Meier analysis. Of the 809 patients who remained on a DBR at last measurement, 84% (n = 681) had HIV-1 RNA <50 copies/mL (Table 2); this finding was generally similar across subgroups. At last measurement, median (IQR) decreases in viral load were 4.43 log10 copies/mL (3.74–5.10) for TN patients (n = 184) and 2.5 log10 copies/mL (1.7–4.0) for TE patients (n = 208). Median (IQR) increases in CD4+ count among patients with CD4 counts available at the index date were 182 cells/mm3 (74–325) for TN patients (n = 169) and 40 cells/mm3 (−51–140) for TE patients (n = 469). Overall, virologic failure occurred in 10% (n = 22/217) of TN and 5% (n = 38/717) of TE patients. Of the 60 patients (6%) experiencing virologic failure, 91% (n = 20/22) of TN patients and 87% (n = 33/38) of TE patients remained on a DBR at last measurement and 68% (n = 15/22) of TN and 61% (n = 23/38) of TE patients had HIV-1 RNA <50 copies/mL at last measurement. Six percent (n = 8/125) of patients who switched or discontinued had a resistance test recorded during follow-up. Among the tested patients, no nucleoside reverse transcriptase inhibitor or integrase strand transfer inhibitor emergent resistance was observed. Kaplan–Meier curves for the probability of achieving undetectable viral load (<50 copies/mL) for (a) treatment-naive and (b) treatment-experienced patients with detectable viral load at dolutegravir initiation. Clinical characteristics. DBR: dolutegravir-based regimen; IQR: interquartile range; TE: treatment experienced; TN: treatment naive. aLast measurement before the index date (initiation of a DBR). bAmong patients with CD4 cell count available at DBR initiation.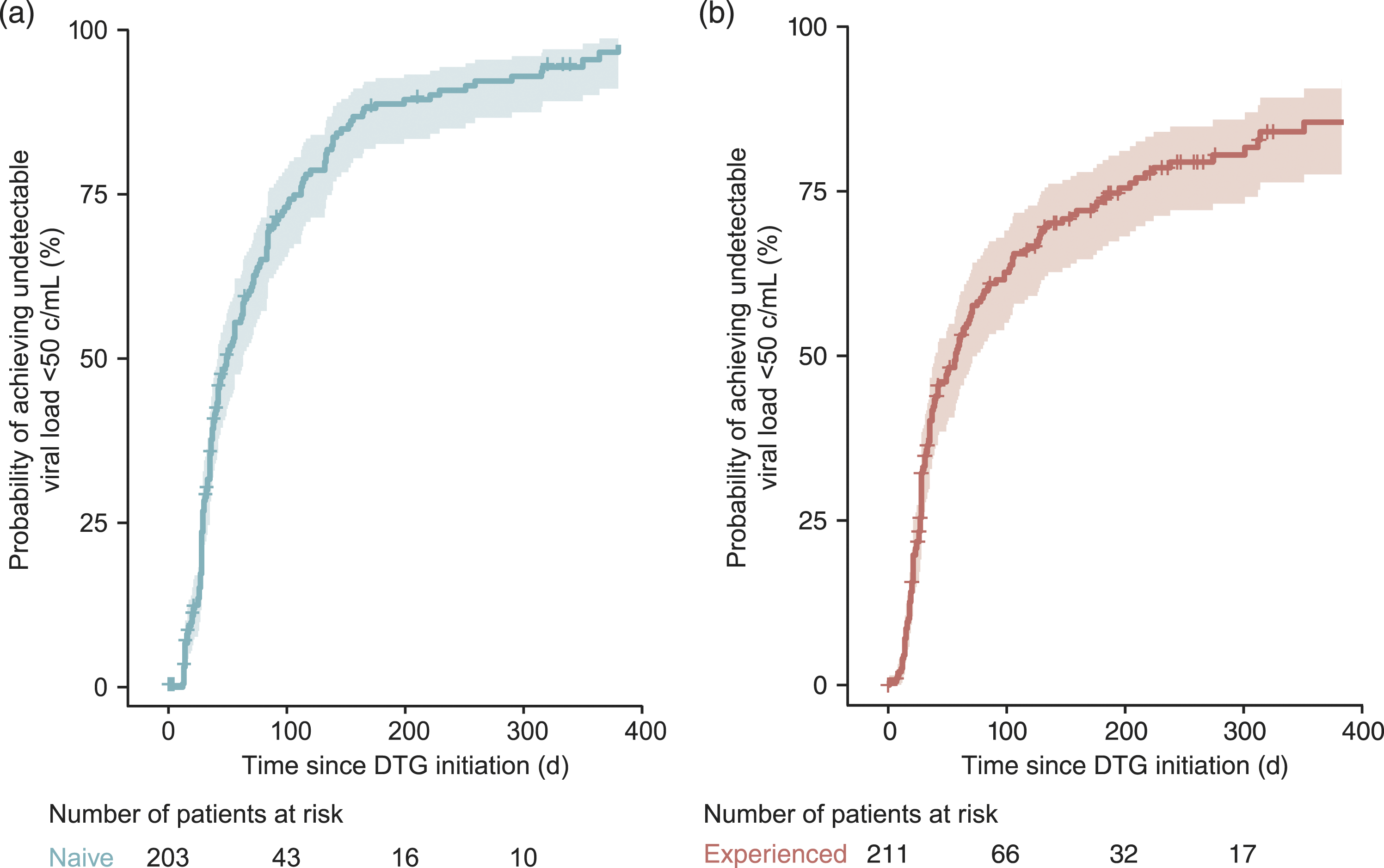
Among those who remained on dolutegravir at 48 weeks, similar proportions of patients aged <50 and ≥50 years had undetectable viral load (46.9% (n = 169) and 45.5% (n = 61), respectively; 48.9% (n = 176) and 46.3% (n = 62) had no viral load data at 48 weeks, respectively). There were also no substantial differences in proportion of patients experiencing virologic failure (4.4% (n = 29) of those aged <50 years and 4.9% (n = 13) of those aged ≥50 years).
Discussion
In this retrospective analysis of real-world clinical data, DBRs showed high levels of virologic suppression and low rates of discontinuation in a large, diverse, UK-based patient population with HIV-1 infection. At the last recorded measurement (median follow-up time of 377 days), 87% of patients remained on DBR and 6% of patients were classified in this study as having virologic failure; however, the majority continued DBR, and no recorded resistance to nucleoside reverse transcriptase inhibitors and integrase strand transfer inhibitors was recorded among patients who switched or discontinued treatment. One reason for switch was documented as ‘resistance’ by the clinician; however, no new emergence resistance was observed in documented notes (Table 2). Discontinuing and switching ARV regimens were infrequent (10% and 3%, respectively); the most common reasons for switch were clinical decision and cost reduction rather than AEs. The probability of achieving HIV-1 RNA <50 copies/mL at 48 weeks was 96% for TN patients and 86% for TE patients.
The real-world data in this heterogeneous population, in which >30% were women and more than 50% identified as non-white, are reassuringly consistent with phase III studies, the results of which show that dolutegravir is an effective and well-tolerated treatment option in both TN2–5 and TE patients.7,8,11 These results also agree with previously published real-world clinical data that show a high number of TN and TE patients remaining on DBRs with high rates of virologic suppression as well as low discontinuations due to AEs.18–22 For example, a retrospective study of patients in Australia showed that the probability of patients remaining on a DBR after 1 year was 95%, with few discontinuations due to AEs. 19 Similarly, our results demonstrate that the probability of remaining on a DBR in a large, heterogeneous, UK-based patient population with HIV infection after approximately 1 year was 90% for TN patients and 86% for TE patients.
These results are also consistent with real-world evidence from DBR studies. In an observational multinational cohort of 490 patients with HIV-1 RNA <50 copies/mL while treated with efavirenz or a ritonavir-boosted protease inhibitor for ≥6 months and switched to a DBR, 32 (6.5%) patients experienced a detectable viral load and 44 (9.0%) patients switched or discontinued therapy by 1 year after the switch. 23 Use of dolutegravir in all TN (n = 22) and TE (n = 98) individuals at a single London HIV centre from 2014 to 2015 was associated with low rates of failure to achieve HIV-1 RNA <50 copies/mL and few discontinuations throughout the median follow-up period of 5.5 months 24 These studies demonstrate how improvements in HIV therapy have led to effective and safe treatment options for both TN and TE patients, paving the way for dolutegravir-based 2DRs. Randomised control trials with dolutegravir-based 2DRs have demonstrated high rates of efficacy, high barrier-to-resistance and good tolerability profiles in both TN and TE patients.9–12 Real-world data studies on dolutegravir-based 2DRs are emerging to reflect this finding in those clinical trials.25–31 Although they were observed in a small number of patients in this study, real-world data on dolutegravir-based 2DRs from 2018 onwards report larger patient cohorts (332–1374 patients in 2020).27–31 The high rate of efficacy demonstrated in randomised control trials and observed in real-world data for dolutegravir-based 2DRs led to an increasing number of patients being switched from dolutegravir-based 3DRs and maintained on dolutegravir-based 2DRs. This seems to be more apparent because people living with HIV infection are ageing and are taking treatment for longer periods. 32 Compared with the general population, individuals living with HIV are at increased risk of comorbidities and polypharmacy.33–35 Polypharmacy has been associated with increased risk of adverse health outcomes.36,37
The ongoing COVID-19 pandemic has caused substantial worldwide disruptions to healthcare systems, resulting in the reduced frequency of face-to-face consultations and routine monitoring at many clinics providing care to people living with HIV. 38 It is reassuring that the observations in this large cohort broadly mirror the efficacy and tolerability of DBRs in randomised control trials.
Databases like the Climate-HIV database that collect data on treatment outcomes in patients with HIV-1 infection provide valuable information for addressing evidence gaps not likely to be assessed in randomised control trials.37,39 Climate-HIV at the time of study design was the only single-point database with detailed patient-level data for individuals with HIV and was shared across four large separate NHS trusts with broad patient demographics. Climate-HIV was also the only database with single-point access as it was linked to the electronic patient record system so CD4+ cell count and viral load records were up-to-date. Such evidence is important for evaluating the real-world implementation of new treatment guidelines or interventions.39,40 Thus, the dissemination of HIV-1 treatment outcomes data from databases, including the real-world DBR clinical outcomes reported in this analysis, is critical for informing the real-world success of HIV-1 treatments.
This study has several limitations. Data are from four specialist clinical practices in England, so they may not be generalisable to other populations. Patients were followed up until the earliest of last record, death, end of available data or switch/discontinuation of DBR (last measurement). However, many patients’ follow-up periods ceased before time point milestones, particularly at 48 and 96 weeks. At 48 and 96 weeks, 602 (64%) and 365 (39%) patients remained in the cohort, respectively, whereas 332 (36%) and 569 (61%) patients, respectively, were excluded due to their final record extraction occurring before the snapshot. This limits the conclusions that can be made regarding these endpoints. Kaplan–Meier curves provide a more reflective analysis of events with the time-to-event curves. More effective treatment, earlier initiation of ARV therapy and better tolerability have led to less need for repeat measures of treatment success markers such as HIV-1 RNA value and CD4+ count. In turn, these were reflected in the number of missing HIV-1 RNA values and CD4+ counts at the study milestones. For example, at weeks 24 and 48, a total of 56% and 48% of patients, respectively, did not have their HIV-1 RNA measurement recorded during the time period (assessed within a ±6-week time frame). Future studies may consider using time-varying analyses, which may be more appropriate for real-world settings because they take into account multiple measurements recorded within the time frame prior to milestones. 41 In addition, because of the retrospective non-interventional design, we could only collate data that were already populated in the database. Data from the Climate-HIV database indicate specific AEs as reasons for switch or discontinuation; however, a full safety profile was unavailable for these patients. For example, ‘Reason for start ART regimen’ is listed in a drop-down menu in Climate-HIV, but there is no further information that differentiates the reason to start one ART instead of another choice. Furthermore, in Climate-HIV, reasons for switch or discontinuation of a DBR were often marked as missing by clinicians, thereby limiting the conclusions to be drawn from these data. At the beginning of the study, patients were classified as TN or TE based on the presence of any prescribed ARV within their Climate-HIV record before DBR initiation. However, several individuals who were transferred from another clinic may have been initiated on DBR before entry into the Climate-HIV database.
Despite these limitations, the results of the present study suggest that DBR is an effective and tolerable treatment option for heterogeneous populations in both TN and TE patients based on achieving undetectable HIV-1 RNA and the low rates observed for discontinuing or switching therapy.
Supplemental Material
sj-pdf-1-std-10.1177_09564624211027099 – Supplemental Material for Using Climate-HIV to describe real-world clinical outcomes for people living with HIV taking dolutegravir-based regimens
Supplemental Material, sj-pdf-1-std-10.1177_09564624211027099 for Using Climate-HIV to describe real-world clinical outcomes for people living with HIV taking dolutegravir-based regimens by Chinyere Okoli, Achim Schwenk, Matthew Radford, Melissa Myland, Stephen Taylor, Justine Barnes, Ashini Fox, Alison Darley, Fiona Grimson, Iain Reeves, Sajid Munshi, Adam Croucher, Naomi Boxall, Alistair Paice, Jean van Wyk and Paul Benn in International Journal of STD & AIDS
Footnotes
Acknowledgements
All listed authors meet the criteria for authorship set forth by the International Committee of Medical Journal Editors. Editorial assistance was provided under direction of the authors by Sherri Damlo, ELS, of MedThink SciCom and was funded by ViiV Healthcare.
Author’s note
Data included in this manuscript have been previously presented at HIV Drug Therapy Glasgow; October 28-31, 2018; Glasgow, UK
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C Okoli, A Paice, J van Wyk, and P Benn are employees of ViiV Healthcare and own stock in GlaxoSmithKline. M Radford was an employee of ViiV Healthcare at the time of the study. A Schwenk, J Barnes, A Darley, and S Munshi have nothing to disclose. M Myland and F Grimson are employees from IQVIA and have received grants from ViiV Healthcare. S Taylor has received grants from ViiV Healthcare, Gilead, and Merck. A Fox is employed by Nottingham University Hospitals NHS Trust, which received a grant from ViiV Healthcare before the conduct of the study to update clinical HIV databases. I Reeves has received grants from ViiV Healthcare. A Croucher is employed by the Jonathan Mann Clinic, Homerton University Hospital NHS Foundation Trust, which received a grant from ViiV Healthcare during the conduct of the study to update local patient records. N Boxall is employed by and has received personal fees from IQVIA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by ViiV Healthcare.
Data availability statement
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
