Abstract
Magnetically controlled growing rods are used for the surgical treatment of early onset scoliosis. However, one of the key concerns around the use of these rods is the extensive titanium alloy metallosis seen at revision surgery. To quantify the wear causing this metallosis, for the first time the internal wear volumes from magnetically controlled growing rods have been measured. A cohort of 30 retrieved rods was measured, from 18 cases (6 single rods and 12 dual rod constructs) after an average time in vivo of 1120 days. Another pair of rods explanted after 30 days due to infection served as a negligible-wear datum to validate the methodology. From each retrieved rod, the wear volume of the extending bar component and of the outer casing component was measured. This was achieved through geometric measurements of the components, with the geometry of the internal aspect of the outer casings determined using a replicating compound and then measuring the replica. The average wear volume of the retrieved rods was 71.8 (21.2–201.9) mm3 which equated to an average wear rate of 23.4 (6.9–65.8) mm3/year. Of the 32 rods measured, 21 were 90 mm in length and 11 were 70 mm in length. The 90 mm rods showed a statistically significant greater wear than the 70 mm rods (
Keywords
Introduction
The most severe forms of scoliosis in children can lead to respiratory failure and death. This fatal condition, called

(a) An unused MAGEC rod. As indicated by the rule, this is a 90 mm MAGEC rod. (b) Schematic image showing the key internal components of a MAGEC rod. Extending bar and outer casing are indicated.
Although MAGEC rods have been widely adopted internationally, several concerns have come to light. These include fracture of the drive pin,5,6 for which a Field Safety Notice was announced by NuVasive in 2019. 7 Extensive damage to other internal mechanical components of MAGEC rods has also been reported, including the O-ring seal and the radial bearing. 8 Such damage is likely associated with the reduced force output seen in the majority of explanted MAGEC rods 9 and the limited rod growth measured following in vivo use. 10 The design of MAGEC rods has changed over time, with the current iteration, MAGEC X, released in 2018. 11 Unfortunately, MAGEC X was associated with a new failure mode, that of end cap separation, and NuVasive instituted a worldwide recall of MAGEC X rods in February 2020. 12 In April 2021, the CE mark on all MAGEC rods was suspended by the Notified Body, DQS. 13 It was reinstated in November 2021. As part of this reinstatement, the Instructions for Use (IFU) in the EU stated that the maximum implantation time of MAGEC rods should be 2 years; previously it had been 6 years. 14 Given this complicated situation, the UK medical devices regulator, the Medicines and Healthcare products Regulatory Agency (MHRA), commenced an investigation of the risks and benefits of MAGEC rods in April 2020, at which point no further MAGEC rods could be implanted in the UK without the approval of the MHRA. 15 This suspension of implantation lasted almost 4 years until March 2024, 16 a remarkable situation in the regulatory history of orthopaedic implants.
Metallosis has been described as the accumulation of metal particles within tissues. 17 One of the other key concerns around MAGEC rods is the extensive metallosis seen at the time of rod removal.18–21 Metallosis is not seen with traditional growing rods, only with MAGEC rods. 21 It is generally agreed that the metallosis is due to titanium alloy wear particles, given that this is the material primarily used in a MAGEC rod. 21 Raised titanium ion concentrations are seen in children implanted with MAGEC rods.20,22,23 The titanium metallosis is likely the cause of these raised ions. 20 The long-term effects of these titanium alloy ions, and the wear debris which creates them, on the growing child are unknown.24–26
It might be expected that the greater the wear volume of titanium alloy, the greater the risk to the child. Therefore, a fundamental research question is, what volumes of titanium alloy wear can arise from implanted MAGEC rods? By examining and taking measurements from a collection of explanted and disassembled MAGEC rods, this project set out to determine how much titanium alloy can arise from inside a MAGEC rod. Knowing this information may help identify those patients who may be most at risk.
Methods and materials
Wear measurement methodology
When considering the wear of MAGEC rods, fundamentally a MAGEC rod consists of two primary components. These are a titanium alloy extending bar and a titanium alloy outer casing (Figure 2). The titanium alloy is Ti6Al4V. 27 The extending bar (centre of Figure 2) is essentially a stepped solid cylinder with a hole drilled in it; at one end of the hole is a threaded insert. Wear is typically at the threaded insert end of the bar and appears as a polished region on the outer surface, as shown in Figure 2; wear tends to be offset to one side of the extending bar. This offset wear has been explained as a result of off-axis loading in vivo. 8

The outer casing (above) and extending bar (centre) from an explanted, disassembled and cleaned 70 mm MAGEC rod (rod 23, time in vivo 1267 days). At the polished (worn) end of the extending bar, two of the four lugs can be seen.
The outer casing of a MAGEC rod (Figures 2 and 3) is essentially a hollow tube, with three different diameter holes in it. The largest diameter accommodates the rare-earth (neodymium) magnet, the middle diameter accommodates the main (1/4″ nominal outer diameter) section of the extending bar, and the smallest, shortest diameter accommodates the internal groove for the O-ring seal (Figure 3).

Image of outer casing (above) of a disassembled 70 mm MAGEC rod (rod 32). The black component below is a Microset casting made of the internal aspect of the outer casing. The casting indicates, from left to right, the internal groove for the O-ring seal, the central section including the grooves for the lugs of the extending bar and the larger internal diameter to accommodate the magnet.
Where appropriate the ISO standard for retrieval and analysis of surgical implants 28 was followed. MAGEC rods were disassembled as described previously.5,8,9,18 The cutting process can result in ragged edges, if there were any of these then these were removed by turning on a lathe to ensure a square surface. A visual indication is shown in Figure 4.
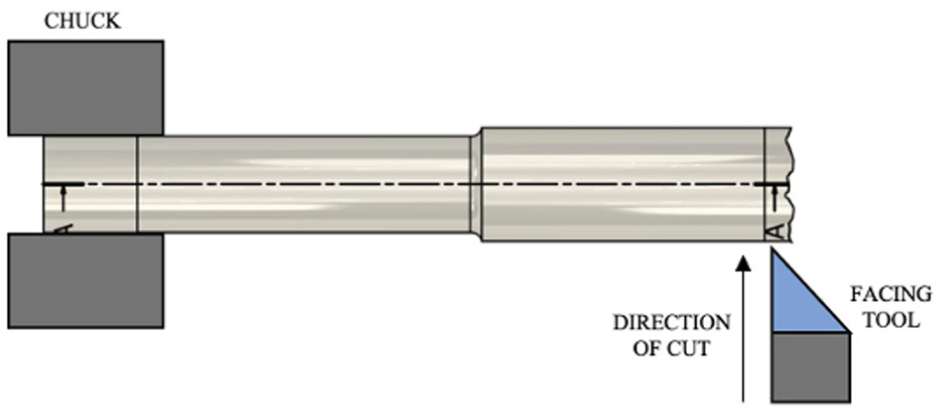
Schematic image indicating facing off process of samples. In this case an outer casing is shown.
Wear of an extending bar and of an outer casing was calculated in the same way. First, the components were repeatedly cleaned using an ultrasonic cleaner, as specified in related international standards such as ISO14242, 29 ISO 14243 30 and ASTM F732. 31 Specifically, extending bars and outer casings were placed in an ultrasonic bath (RS Pro Ultrasonic Cleaner 100W 3L, RS Components) along with compatible ultrasonic cleaning fluid. Approximately 1 l of water was added to ensure that the samples were covered fully, along with 200 ml of ultrasonic cleaning fluid to attain a dilution ratio of one part cleaning fluid to five parts water. Each cleaning cycle ran for 15 min at a temperature of 35°C–40°C. Only two outer casings and their corresponding extending bars were cleaned at once, to reduce the possibility of mix-ups and to prevent overloading. The samples were left to air dry then were sprayed with compressed air, before being weighed using an analytical balance with a resolution of 0.0001 g (Kern ABT 220-5DM). In particular, the internal grooves of the outer casings and the cavity behind the threaded insert on the extending bars were susceptible to water entrapment, so thorough drying was crucial. Cleaning was repeated until the mass of the component was unchanged to the nearest 0.001 g.
Next, a geometrically accurate model was created of each individual extending bar and each individual outer casing component in its unworn state. Dimensional measurements were taken using a Quick Scope (Mitutoyo, Kawasaki, Japan) vision measuring system which has a measurement error of around 2.5 µm. 32
The measurements taken on the external aspect of the extending bar are shown in Figure 5 and its accompanying table, Table 1. In each case, three measurements were taken, and an average was calculated.
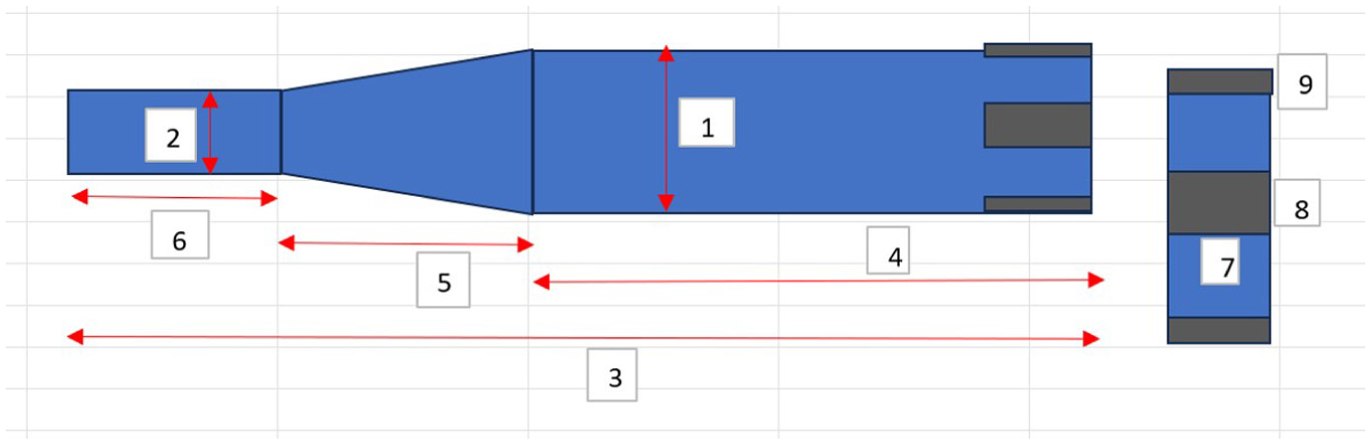
Schematic image showing the various dimensions measured on external aspects of extending bars. Right hand side image is magnified schematic to detail lug region dimensions.
Measurements made on features of external aspect of extending bars.
While the extending bars had a simpler internal geometry compared with an outer casing, namely a blind drilled hole, this geometry was made a little more complex by having a tapped hole at the ‘lug end’ of the extending bar, into which a threaded insert was fitted. For the measurement of the depth and diameter of the blind drilled hole, a coordinate measuring machine (CMM; LEGEX 322, Mitutoyo, Japan) was used. This CMM has a spatial resolution of <1 µm in the area of measurement.33,34
To aid in calculating the mass of a geometric model of the extending bars, the threaded insert was removed from a separate explanted sample, not one of the 32 rods detailed in this study. This allowed the dimensions and mass of a threaded insert to be determined, using the same vision measuring system and analytical balance respectively. From these measurements, a density could be calculated. In addition, material composition of the removed threaded insert was assessed using x-ray fluorescence (Niton XRF GOLD++ (ThermoFisher Scientific, Germany)). From the sample with the removed threaded insert, the depth of the threaded hole in the extending bar was measured.
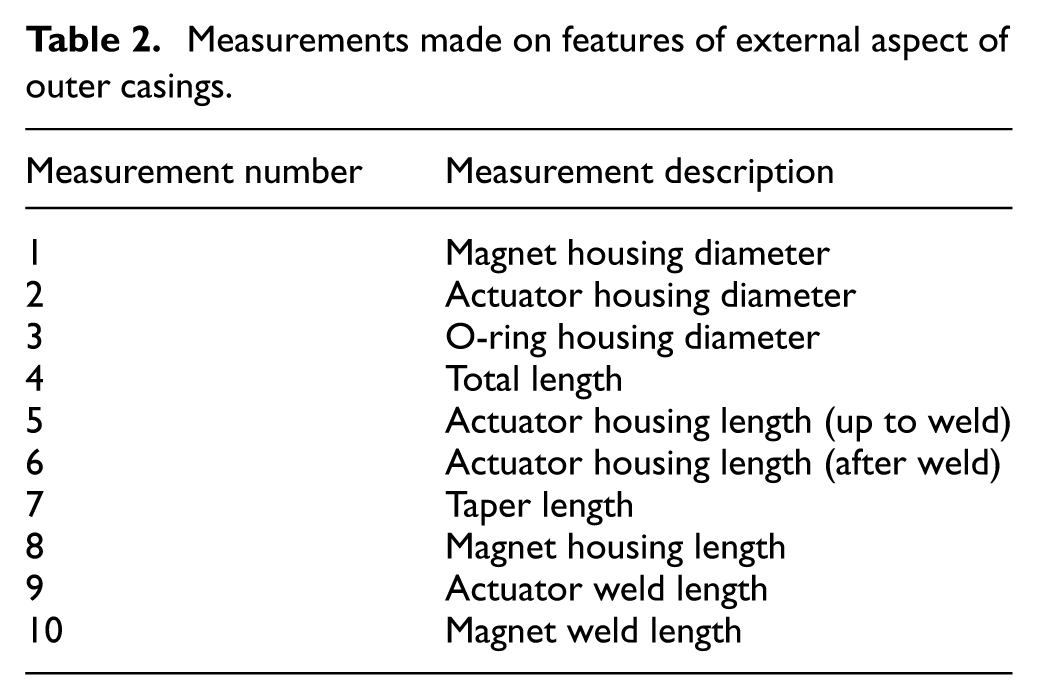
The measurements taken on the external aspect of the outer casing are indicated in Figure 6. Again, for each dimension, three measurements were taken, and an average was calculated. The related measurements are detailed in Table 2.
Schematic image showing the various dimensions measured on external aspects of outer casings.
Measurements made on features of external aspect of outer casings.
To determine the wear of outer casings, the internal dimensions needed to be measured. For this reason, a casting was made with a commercially available replication material, Microset 101, with a claimed shrinkage of <0.1% 35 (Microset, Hinkley, UK). Such a material has been used as a means of reproducing surface roughness features down to ±60 nm, and in an application with hole diameters as small as 0.35 mm. 36 This small size was important as internal features of the outer casing were as small as approximately 0.4 mm.
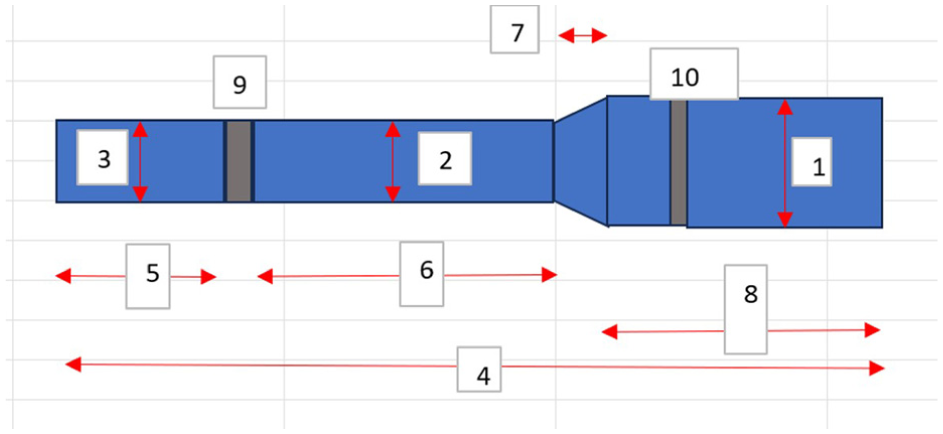
The measurements taken on the internal aspect of the outer casing are indicated in Figure 7. Again, for each dimension, three measurements were taken, and an average was calculated. The related measurements are detailed in Table 3.
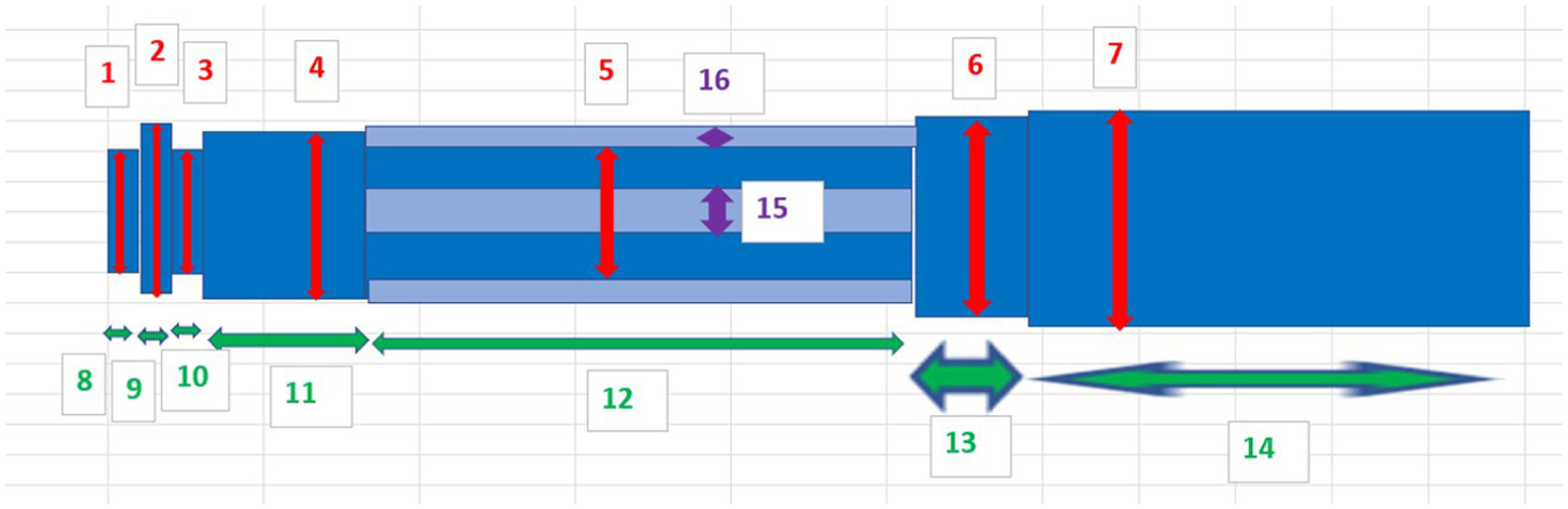
Schematic image showing the various dimensions measured on internal aspects of outer casings.
Measurements made on features of internal aspect of outer casings.
In terms of where dimensional measurements were taken on components, the only concerns were to avoid the worn areas, as the whole concept of the wear methodology was to create a model of an unworn component. On the ‘main section length’ (Table 1, Figure 5) of the extending bars this was relatively straightforward as the polished (worn) region was visible. Thus, dimensions (i.e. lug dimensions) were taken on the opposite side to the polished region, given that wear was only on one side of the bar, as explained by off-axis loading. 8 For the internal aspect of the outer casing, no such obvious visual aid as a polished region was available. Therefore, and again based the concept of off-axis loading, 8 measurements of the central section of the casting were taken towards the O-ring end of the casting, rather than the magnet end (Figure 3).
From the geometric model of each unworn component, its volume was calculated in Microsoft Excel. The density of Ti6Al4V was taken as 4430 kg/m3. 37 Knowing the volume and the density, the theoretical mass of the unworn component (Mu) was calculated. This was then compared against the actual mass (Ma).
Thus, Mu – Ma = wear (g)
The wear in grammes could then be converted to a volume (mm3) using the density of Ti6Al4V.
Statistical tests were done using a
Validation work
The main challenge in this work was determining the internal volume of the outer casings. To aid with this, the following validation work was undertaken. Three test pieces were manufactured. Each consisted of Silver Steel, of 10.000 mm ± 0.015 mm outer diameter, 60.000 mm ± 0.020 mm length, with a 6.000 mm ± 0.010 mm hole drilled through. These sizes were chosen as they approximated to the size of the potentially worn aspect of the longest (90 mm MAGEC rod) outer casings (neglecting the region housing the magnet, which would not be subject to wear). Following cleaning in the ultrasonic cleaner, the length of the test piece and diameter of the hole through each test piece was measured using the Quick Scope. A Microset 101 casting of the 6 mm nominal diameter hole of each test piece was then made. After setting, these castings were removed. The diameter and length of the castings was measured using the Quick Scope and compared to the diameter and length of the test piece. The measured length and hole diameter of the test pieces varied on average by a value of 0.0247 and 0.0003 mm respectively. All three castings exhibited shrinkage in both dimensions. The average was 0.0411% and 0.0044% shrinkage in the length and hole diameters respectively, both being below the 0.1% specified by the manufacturer. 35 The difference in dimensions, between the hole and the casting, led to a volumetric difference of 0.868 mm3. This value served as an indication of the accuracy of the wear measurement technique for the internal aspects of outer casings.
To aid validation of the modelling technique, two 70 mm rods retrieved after 30 days due to infection served as a negligible-wear datum. Justification of negligible-wear came from three factors. First, from visual analysis, there was no polished region extending beyond the lugs of the extending bar. Second, when measured across the diameter, between the lugs, the extending bar should be in the range 6.32–6.35 mm. This is the range of unworn diameters based on previous published work 8 and subsequent unpublished data. Third, each of the four lugs are unworn, in that the dimensions of their width are in the range 1.22–1.32 mm.
As a preliminary piece of work to validate the concept of using density = mass/volume, and proving that the Quick Scope could provide sufficiently accurate measurements of dimensions from which wear could be calculated, the following was done. Three samples of a 5.000 mm ± 0.015 mm diameter silver steel bar were taken, each cut to a nominal length of 45.000 mm ± 0.020 mm. Each sample was cleaned in the ultrasonic cleaner and dried using the same procedure as described previously. Each sample then had its mass measured three times using the Kern ABT 220-5DM balance. The average became the Mass actual (Ma). The length and diameter of each sample were measured three times using the Quick Scope. Taking these dimensions allowed the mass of the sample to be calculated using density = mass/volume, where a density of the silver steel of 7830 kg/m3 was used. This gave Mass theoretical unworn (Mtu). The Ma and Mtu were compared and a difference of less than the equivalent of 0.1 mm3 was found. Next, the samples were taken, and a slot was machined at one end of the bar. The cleaning and drying protocol was then repeated. The samples were then weighed three times using the Kern ABT 220-5DM balance. Next, the depth and width of the slot were measured using the Quick Scope. Taking these dimensions, alongside the length and diameter of the bar and a density of the silver steel of 7830 kg/m3, allowed a theoretical mass of the ‘worn’ bar (Mtw) to be calculated. Again, the theoretical mass based on density and dimensions, and the actual mass were compared. From this validation work, the difference was always less than 0.1 mm3 wear volume.
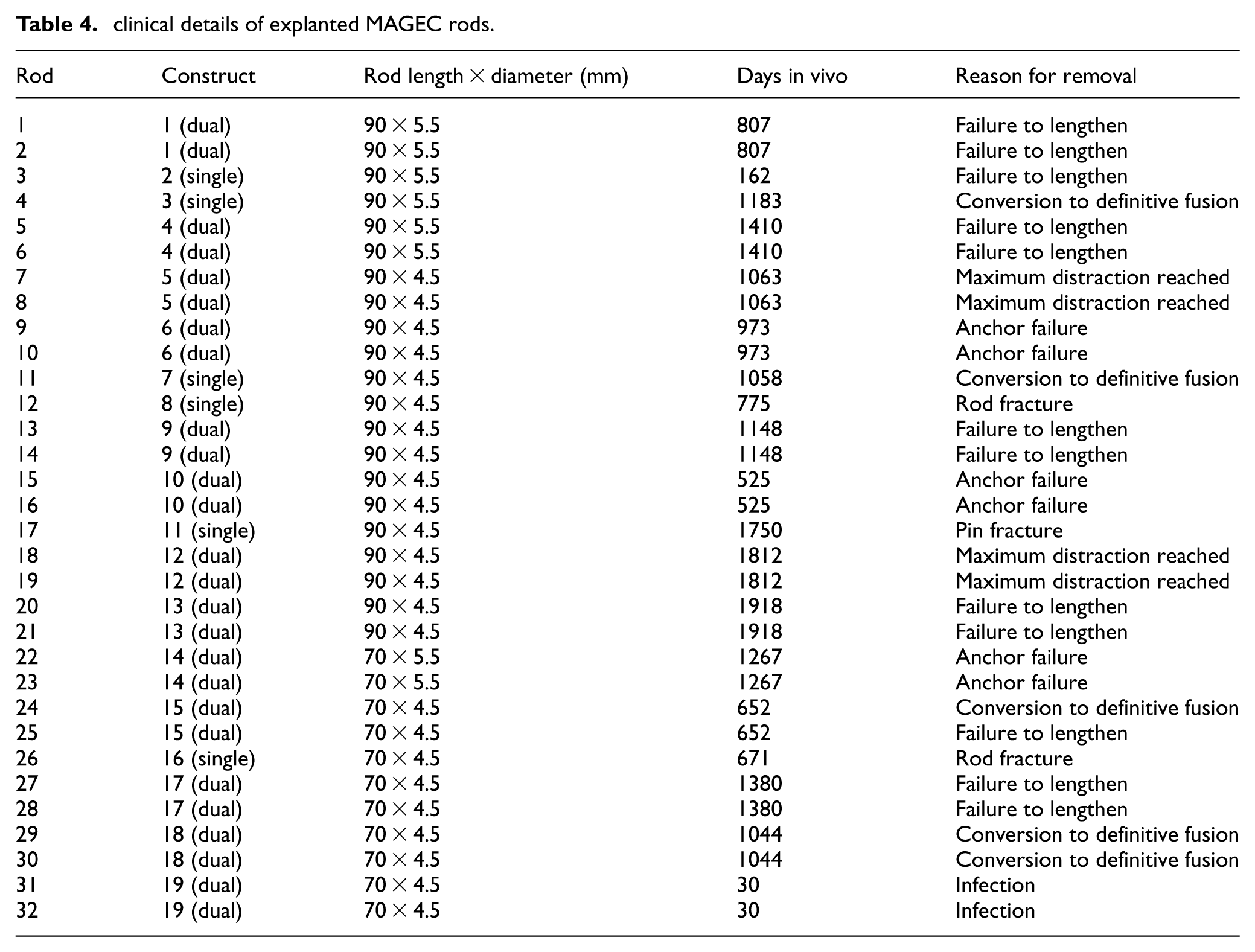
Clinical and explant data
There were 6 single rod constructs, and 13 dual rod constructs analysed, making 32 explanted rods in total which were measured. Of these 32 rods, 21 were 90 mm long, 11 were 70 mm long. Of these 32 rods, 24 were 4.5 mm diameter, 8 were 5.5 mm diameter. Time in vivo was known for all rods. This information, plus the reason for removal, is given in Table 4. Excluding the two rods in vivo for 30 days which served as a negligible-wear datum, the average time in vivo was 1120 days (approximately 37 months) and the range was 162–1918 days (approximately 5.5–64 months). All rods were generation 1.2 or 1.3, as per the description by Tognini et al. 38
clinical details of explanted MAGEC rods.
Results
In total, 32 rods were analysed. In each case, wear measurements were obtained for the extending bar and the outer casing; these were then added together to give the total volumetric wear of a rod. Results are shown in Table 5.
Wear per component and per MAGEC rod. EB = Extending Bar, OC = Outer Casing.
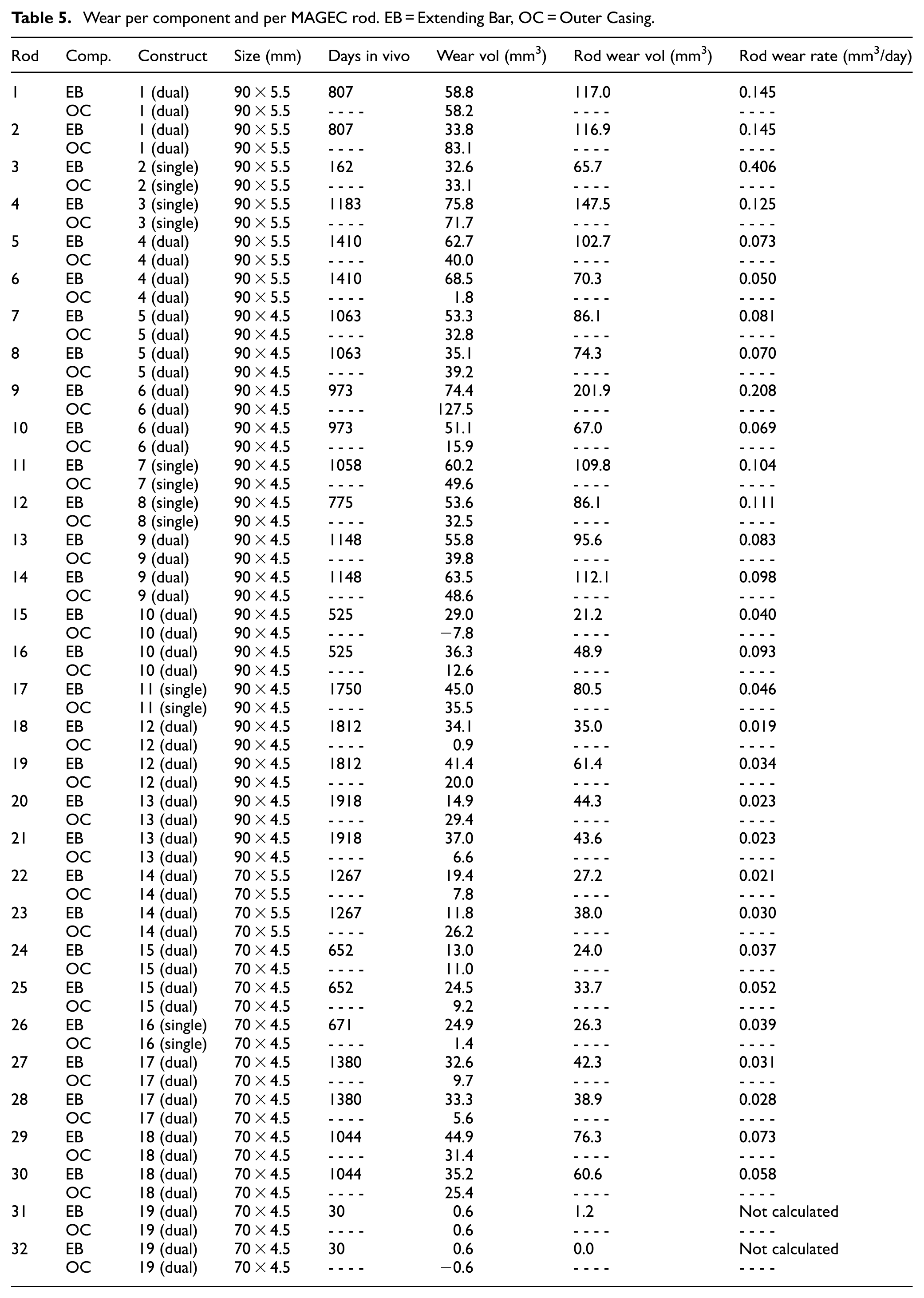
Rods 31 and 32 were from a dual construct that had been in vivo for only 30 days. The rods were removed due to infection. These two rods served as a negligible-wear datum. Aside from these two rods, the average and range data for the wear of extending bars, outer casings, and total rods were 41.9 (11.8–75.8) mm3; 30.0 (−7.8 to 127.5) mm3, and 71.8 (21.2–201.9) mm3 respectively. In turn, again excluding the two ‘datum’ rods, the average and range rod wear rates were 0.080 (0.019–0.406) mm3/day. These equated to 23.4 (6.9–65.8) mm3/year.
For the threaded insert used inside each extending bar, this was determined to be aluminium bronze alloy 630. The internal thread was 6–80 UNF 2B, the external thread was 10 56 UNS-2B. The depth of the threaded hole in the extending bar was measured to be 7.747 mm.
Results showed that 90 mm MAGEC rods were associated with greater wear volumes than 70 mm MAGEC rods, 85.0 mm3 compared with 40.8 mm3 respectively (

Composite image of three explanted extending bars. Top image, one of the negligible-wear datum components from pair of 70 mm MAGEC rods (rod 32). Central image, another extending bar from a 70 mm MAGEC rod (rod 27, 32.6 mm3 of wear, 1380 days in vivo). Bottom image, an extending bar from a 90 mm MAGEC rod (rod 4, 75.8 mm3 of wear, 1183 days in vivo).
While the average wear of a rod from a single-rod construct (86.0 mm3) was greater than that from a dual rod construct (68.3 mm3), this was not statistically significantly different (
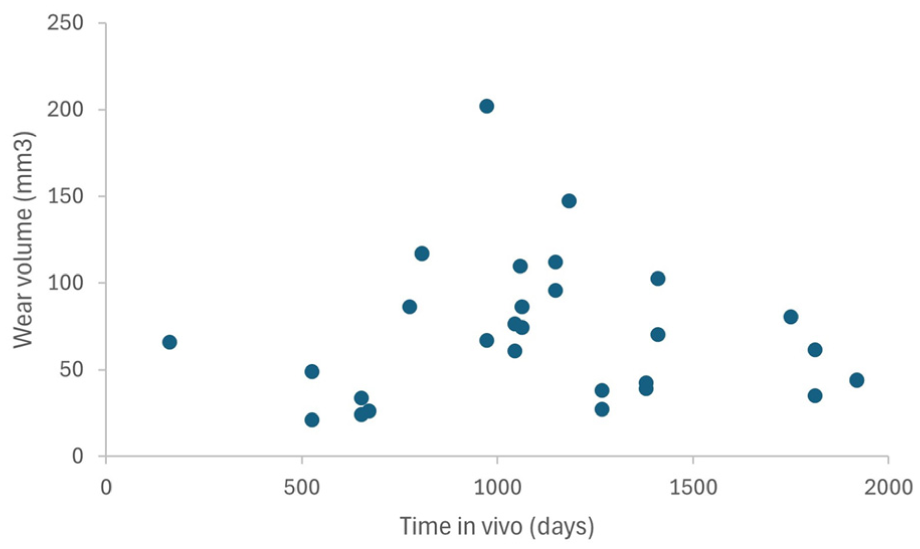
Figure 9 shows a graph of rod wear volume against time in vivo.
Discussion
For the first time, wear volumes from the internal aspects of explanted MAGEC rods have been reported. For 30 rods, excluding the two rods removed at 30 days due to infection, the average wear volume was 71.8 mm3 and the average wear rate was 23.4 mm3/year. On this basis, for dual rod constructs, which are far more common than single-rod constructs, 9 this means that approximately 47 mm3/year titanium alloy wear debris is generated.
To put these wear rates into context, a wear rate of 25.6 mm3/year has been reported for cross-linked polyethylene (XLPE) total hip replacements, based on radiographs of 105 hips. 39 A wear rate of 25 mm3/year has been reported for ultra-high molecular weight polyethylene (UHMWPE) total knee replacements, based on measurements from 10 explanted Press Fit Condylar (PFC) designs. 40 Now, while they give context to the most common orthopaedic implants, these are polyethylene wear volumes, so it may be more appropriate to look to wear of metals used in orthopaedic implants.
It has been shown that wear rates of above 2.3 mm3/year (approximately two pin heads 41 ) from the articulating surfaces of a metal-on-metal resurfacing hip is sufficient to lead to a wear related failure and thus a revision operation. 42 In fact, the amount of metal debris from a taper-trunnion surface of a modular hip which can lead to revision is even less, with a median of 0.4 mm3/year being reported from a cohort of 104 metal-on-metal hips revised for wear related failures. 43 Clearly the titanium, aluminium and vanadium of the Ti6Al4V alloy used in MAGEC rods is different to the cobalt, chromium and molybdenum used in metal-on-metal hips, but the high volumes of titanium alloy measured are notable.
A counterargument to concerns regarding titanium alloy wear debris may be that such alloys are successfully used in millions of implanted hip and knee replacements, without any negative clinical implications from titanium debris. While this is true, in these applications the titanium is static, and its surface is not designed to provide an articulation. Where such an articulation of the titanium surface occurs, while it may be unintended, the clinical consequences can be substantial. A 2011 case report described renal failure in a patient with a worn through titanium acetabular shell.
44
The authors commented ‘
While there is established evidence of toxicity related to wear of CoCr, such evidence is less strong with titanium alloy. That said, some authors claim to have provided ‘
It has been known that titanium alloy does not make a good bearing material since its inception. For example, in 1958 Miller and Holladay reported titanium’s lack of wear resistance.
50
In 1993, having studied the impact of CoCr and Ti wear debris on tissues, Haynes et al. noted that the potential of titanium wear should be minimised.
51
Thus, the importance of minimising titanium alloy wear has been known for several decades. In a 2011 paper which considered the titanium, aluminium and vanadium which constitute many titanium alloys used in the human body, the authors stated ‘
Moreover, much of the history of joint replacements in the human body has recorded the clinical consequences of high wear and has thus, for very good reasons, been concerned with minimising wear. From the poly tetra fluoro ethylene (PTFE) used in the first Charnley hips, which was replaced with the harder wearing UHMWPE, which in turn was replaced by XLPE, a reduction in wear volumes has resulted in more successful orthopaedic implants.
53
Empirically, it might be expected that the greater the wear volume, the more negative the consequences on the human body. Historically, this dose dependent effect has been seen with UHMWPE
54
and CoCr
55
used in artificial hip joints. It could apply to titanium too. In 2004, based on their study of wear debris from hip and knee replacements, Daley et al. wrote ‘
At 23.4 mm3/year, the wear rates from explanted MAGEC rods are high, but they are comparable with another titanium alloy growing rod system. Reporting on six growth guidance rods (LSZ-4D, CONMET, Moscow, Russia) plus associated fixtures from three cases, a wear rate of 12.5 mm3/year has been offered.
57
In a sister paper, the same authors reported on their cohort of 25 patients implanted with the growth guidance rods and stated ‘
Results showed that 90 mm MAGEC rods were associated with greater wear volumes than 70 mm MAGEC rods, 85 mm3 compared with 40 mm3 respectively (
From Table 5, there are several points to highlight. For the two rods which served as a negligible-wear datum, rods 31 and 32, the overall rod wear was determined to be 1.2 and 0.0 mm3 respectively. These values help to indicate the accuracy of the wear measurement methodology. For the outer casings, a few of the wear volumes are low. In one case, the outer casing of rod 15, a negative wear volume of 7.8 mm3 has been calculated. In the cases of the outer casings of rods 6, 18 and 26, the wear volumes are 1.8, 0.9 and 1.4 mm3 respectively. These are lower than would be expected and are likely an underestimate of the true wear. As an explanation, it is possible that wear debris has been trapped and adhered to the internal surfaces. Despite repeated efforts at cleaning, this recalcitrant debris remained in place.
A 2022 paper measured the wear volumes from the growth marks of 44 explanted MAGEC rods. 59 The maximum wear volume from a single rod was 0.15 mm3.59 Clearly, this is far less than the maximum reported in this paper (202 mm3). However, this over-100-fold difference helps to show the gulf in magnitude between the external wear and the internal wear of MAGEC rods. The internal wear volumes from MAGEC rods are substantial and should not be ignored.
Limitations
Limitations of the validation methodology might relate to the use of replication material and the inevitable shrinkage. Yet the replica manufacturer claims this shrinkage is less than 0.1% 35 and the validation work reported in this paper shows it was 0.0411% and 0.0044% over 60 and 6 mm respectively. Compared with, say, the in vitro wear testing of hip 29 and knee 30 prostheses, or screening wear testing of biomaterials, 31 where test rigs and international standards exist, the wear measurement methodology applied in this work is not traditional. Much explant analysis work starts with a lack of knowledge of the manufacturer’s dimensions of components. Researchers frequently need to ‘work backwards’ from unworn surfaces to be able to identify worn surfaces and thus calculate wear. This approach has been used successfully to measure the wear of explanted femoral heads and acetabular liners of metal-on-metal hips,34,60 explanted trunnions of total hip replacements 61 and the polyethylene components of explanted total knee replacements. 40 In each of these cases, wear from external surfaces was measured, so that access to the surfaces was relatively straightforward. This means that a straight stylus can be used, and attached to the SP-25 scanning head of the CMM that was used to create point clouds from which wear can eventually be calculated. 34 The volumetric wear within the internal taper of femoral heads has also been successfully achieved using a CMM, 62 but in such cases the internal taper is relatively wide and shallow, (typically around 12 mm diameter at the base and 15 mm deep 63 ) so that access for the ruby probe at the end of a straight stylus of the CMM is straightforward. In the case of explanted MAGEC rods, a method for measuring the wear on the internal surface of the outer casing had to be determined. With an overall length of 90 mm, an internal diameter of 6.5 mm and localised internal features including an O-ring groove of less than 1.2 mm width, the latter meaning that a star probe needed to be used, rather than a straight probe, meant that no suitable probes for the SP-25 scanning head of the CMM existed. 64 Hence another non-destructive wear measurement method had to be found, namely the use of replicating compound with the ability to reproduce surface features such as 0.35 mm holes 36 to create a casting which accurately reproduced internal features. The casting was then measured using the Mitutoyo Quick Scope (with an accuracy of 2.5 µm32). For consistency, the same Quick Scope was used with the external features of the extending bars, and the external features of the outer casings. The context of this work is important. Explants bring challenges such as an unknown initial mass, so that a researcher cannot simply weigh an explant and thus determine the wear. In the case of MAGEC rods, there are multiple internal components, so these had to be removed and the two key components in terms of titanium alloy wear, extending bar and outer casing, thoroughly cleaned prior to weighing. Determining the internal wear volumes of explanted MAGEC rods has not been done previously, likely in part because it is so challenging and a non-traditional approach, yet one based on engineering fundamentals, was taken.
The use of replicas to measure wear from explanted orthopaedic components has been used previously. In the 1990s Hall et al. used replica material to determine the wear volumes from explanted polymeric acetabular cups. 54 That procedure related to the relatively simple sub-hemispheric shape of acetabular cups, whereas the internal aspect of the outer casing of a MAGEC rod is far more complex in shape. Going back even further in time, and reported in 1978, Charnley himself produced replicas of the internal aspect of retrieved acetabular cups to measure the wear. 65 In this case it was a 2D measurement as he wished to compare explants with X-rays. 65
It may appear that the use of the 30-day explants as a negligible-wear datum is questionable. However, as shown in Figure 8, the extending bar which had been in vivo for 30 days was in an almost pristine condition. The visual contrast with the worn extending bars shown in Figure 8 which had been in vivo for 1380 and 1183 days respectively, should be clear. Moreover, as described in the Methods and Materials section, the criteria for negligible wear were not only visual, but based on the dimensions of specific aspects of the extending bars which served to indicate whether any wear had taken place or not.
Clearly there are aspects of tribology (roughness, hardness, friction) which are related to the wear process. However, the fundamental aim of the paper was to quantify, for the first time, the wear volumes arising within explanted MAGEC rods. This has been done. An investigation of other aspects would need to be the basis of separate papers, particularly as such information appears to be unavailable in the scientific literature.
In terms of assessing the validation methods, the use of density = mass/volume, with the balance, cleaning method and Quick Scope, was shown to work for a relatively simple shape where external measurements could be taken, namely the silver steel bar with a slot cut in it, to less than 0.1 mm3. It was shown that shrinkage of Microset 101 is so small that the internal aspects of a surrogate 90 mm outer casing would have an error of <1 mm3. Admittedly these surrogates did not have four internal slots, but if the bore diameter could be reproduced with sufficient accuracy, it was assumed that the same would apply to the internal slots. The use of the CMM for the central hole of the extending bar is a more conventional method. A simple volumetric calculation of a hole of 58.42 mm depth (associated with the longer extending bar used in a 90 mm MAGEC rod) and 3.912 mm bore, with a measurement ‘error’ of 1 µm would be associated with a volumetric error of less than 1 mm3. There is a limitation that just one threaded insert was measured, and just one depth of thread that the insert fits in. More were not measured as a decision was made not to destroy further retrieved samples; there was a desire to preserve them for potential future research. Also, based on the overall results, the measurements of the threaded insert and its tapped hole appear to be credible.
A limitation in relation to the samples analysed might be that only non-MAGEC X rods were measured, given that MAGEC X is the latest iteration of the magnetically controlled growing rod. However, the earlier version of MAGEC rods have been implanted into many children, and many remain in children to this day. In addition, when MAGEC X was recalled in 2020, the manufacturer offered the older version in its place. As such, the MAGEC rod design assessed in this paper could have been implanted as recently as 2021. Moreover, it is possible that some rods are left within children after spinal growth is complete, as questions around the need for removal have been raised.
25
Furthermore, metallosis is as common in MAGEC X (73%
11
) as in earlier versions of MAGEC rod (79%
18
) and thus these volumetric wear results may be applicable to MAGEC X too. A recent study evaluated the FDA’s adverse event database regarding MAGEC rods.
4
While exact data from the manufacturer is not available in the public domain, it was estimated that 12,000 MAGEC rods may have been implanted up to the date of data evaluation (30 November 2023).
4
The wear data presented in the current paper may be of value to this large cohort. Moreover, if the end cap of a MAGEC X comes off, which has been reported in 24% of cases,
11
then there is little to prevent wear debris egress. Another paper reported a 7% rate of endcap loosening.
66
It was postulated that the difference between 7% and 24% was explained by the latter MAGEC X rods have a longer time in vivo (42 vs 15.5 months).
11
Another paper analysed the FDA database of adverse events for MAGEC X, and found that ‘
A possible limitation in relation to the Results is that titanium alloy wear debris morphology and number was not quantified. However, the aim of this work was, for the first time, to quantify titanium alloy wear volume. At an average rod wear of 72 mm3, this has now been done. Previously, a mean size of 3.36 microns for titanium alloy wear debris from inside MAGEC rods was offered. 21 Assuming an average particle to be spherical and having a radius of 1.68 microns, gives an average wear particle volume of 2 × 10−8 mm3. Therefore, the approximate total number of titanium alloy wear particles from a MAGEC rod can be determined from 72 mm3/2 × 10−8 mm3 = 36 × 10 8 , or 3.6 billion particles per rod.
Another limitation is that the wear volumes measured from the explanted MAGEC rods may not equate to the amount of wear that ends up in the child; some may remain within the rod. However, given the substantial damage that is seen to many O-ring seals of MAGEC rods,8,18 as well as the likelihood of off-axis loading negating the action of the seal, 8 it is appropriate that the potential of all the wear debris ending up in the child’s tissue is considered. Moreover, the high prevalence of metallosis reported in multiple publications associated with MAGEC rods18–21 implies that leakage of titanium wear debris past the seal is substantial. This leakage remains extensive with MAGEX X rods too. 11
Additional clinical data was available for many of the explanted rods. These data include age at insertion, sex, diagnosis, height at insertion, weight at insertion, number of lengthenings, frequency of lengthenings, age at removal, reason for removal, height at removal, weight at removal, rod growth (mm) and force output (N). Given these multiple clinical factors, they will be the topic of a separate, clinically focussed paper. As indicated in Figure 9, there was no strong relationship between time in vivo and volumetric wear of a rod. While this may seem counterintuitive, there are multiple clinical factors that explain the apparent lack of a wear versus time relationship. One is the lack of activity data; some children with scoliosis requiring growing rod surgery will be far more active than others, some of whom will be wheelchair bound. Loading, and thus wear according to boundary lubrication theory, 53 will vary widely between the children, and such loading will likely increase over time. In a dual-construct arrangement, where one rod is fitted on the concave aspect of a scoliotic spine, and the other on the concave side, it is likely that one rod will be associated with greater loading than the other. Indeed, this difference in wear between two rods in dual-rod construct is seen most strongly with rods 9 and 10 (Table 5).
A further limitation regarding the clinical data might be viewed as the lack of blood titanium ion concentrations associated with the rods. Such data was not available from the clinical centres who kindly supplied the explanted MAGEC rods. Also, while blood ion levels give an indication, the true issue is likely to be the local concentrations of titanium alloy in tissue around the spine. As a parallel, from some work on metal-on-metal hips, median blood Cobalt was 0.7 µg/l, while the median Cobalt in the synovial joint surrounding the metal-on-metal hip was 113.4 µg/l. 67 In 2014, an EU wide review of metal-on-metal implants said that they should be avoided in women of child-bearing age. 68 While this review was concerned with implants based on CoCr, there have been similar concerns with titanium.22,26 In addition, it is known that metal ions, including those of titanium, can cross the blood-brain barrier with potential neurotoxic effects. 69
Conclusions
For the first time, the wear volume arising within explanted MAGEC spinal rods has been determined. Based on measurements of 30 explanted rods with a mean time in vivo of 1120 days, the average wear from a rod was 71.8 mm3 (range 21.2–201.9 mm3), which equated to an average wear rate of 23.4 (range 6.9–65.8) mm3/year. Given that dual rod constructs are more commonly employed than single rod constructs, so an average of 46.8 mm3/year can be estimated for dual rod constructs. Single-rod construct wear (86.0 mm3) was greater than of a rod in a dual-rod construct (68.3 mm3), but not significantly (
Footnotes
Acknowledgements
The author thanks the many spinal centres which have contributed to this and related research by supplying explanted MAGEC rods. Several Stage 4 engineering undergraduate students from the School of Engineering at Newcastle University contributed to this work (Emma Hindhaugh, Sophie Lord, Patrick Mowbray, Hannah Reece, Ted Ross, Gregor Stewart, Jamie Curson, Thomas London, Zaigham Mobsshar) and their input is greatly appreciated.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author thanks the British Scoliosis Research Foundation (BSRF), now Scoliosis Support and Research (SSR) for their generous funding which allowed this work to take place.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
