Abstract
Incontinence and toileting difficulties can often be successfully addressed by treating their underlying causes. However, (complete) cure is not always possible and continence products to prevent or contain unresolved leakage or to facilitate toileting are in widespread use. Many people use them successfully but identifying the product(s) most likely to meet individual needs can be challenging and the recently published Seventh International Consultation on Incontinence includes a chapter which draws on the literature to provide evidence-based recommendations to help clinicians and product users to select appropriate products. This paper is based on the same evidence, but reviewed from the different perspective of those keen to identify unmet needs and develop improved products. For each of the main continence product categories it (i) outlines the design approach and key features of what is currently available; (ii) provides a generic functional design specification; (iii) reviews how well existing products meet the requirements of their main user groups; and (iv) suggests priorities for the attention of product designers. It also flags some core scientific problems which – if successfully addressed – would likely yield benefits in multiple incontinence product contexts.
Keywords
Introduction
Everyone with bladder or bowel incontinence or toileting problems would like to be cured but (complete) cure is not always possible. The challenge for those whose problems persist is to discover how to manage them such as to minimise the impact on their quality of life – and that of any caregivers – and this normally involves using technology in the form of continence products to prevent or contain leakage or to facilitate toileting. Even those who are ultimately treated successfully may have to live with symptoms for a time – for example, while they wait for surgery or pelvic floor muscle training to yield its benefits – and they may use continence products temporarily during the waiting period. Others may use them as an adjunct to a treatment that reduces their symptoms without eliminating them. Still, others may use products intermittently, for example, limiting their use to periods or activities associated with troublesome leakage, such as exercising. Finally, some rely on products permanently, either following treatment that has not been (completely) successful or – depending on their co-morbidities, severity of symptoms and personal priorities – because they are not candidates for treatment aimed at cure.
The recently published book from the Seventh International Consultation on Incontinence provides evidence-based clinical guidelines for diagnosing, treating and managing all aspects of incontinence and it includes a chapter on Management using continence products, written by the authors of the current paper. 1 Based on a review of the literature, its primary purpose is to provide clinicians with guidance on selecting those existing products most likely to meet the needs of product users. By contrast, this paper draws on the same evidence to provide guidance for those seeking to develop improved products.
Continence product categories and the general needs of their users
There are many thousands of continence products on the market – as the International Continence Society’s Continence Product Advisor website shows 2 – but they may be classified into a limited number of design categories which can themselves be gathered into major groups depending on whether they are intended to assist with toileting (urine or faeces), prevent or contain leakage (urinary incontinence (UI) or faecal incontinence (FI)), or drain urine from a bladder reticent to (fully) empty itself.
The literature shows that the key considerations for identifying or designing products most likely to meet users’ needs vary in detail between users and between product categories but some characteristics of users (Table 1) and of products (Table 2) crop up repeatedly and it is useful to review them before considering the specific issues relating to each of the main product categories.
User characteristics that commonly arise in identifying – or designing – the continence product(s) most likely to meet the needs of users.
Product characteristics that commonly arise in identifying the continence product(s) most likely to meet the needs of users.

The most obviously pertinent characteristics of people in need of continence products are those that relate directly to their bladder and/or bowel (dys)function (Table 1, first three bullet points). However, other physical characteristics of a user’s body are usually important, too. Different anatomies each present different opportunities and challenges for product choice (as well as product development) and variations in body shape and size between people of the same sex often thwart the preferred one-size-fits-all approach to design. A further raft of user characteristics that are not directly linked to bladder or bowel function may also be important: some products may be ruled out because a user has inadequate mobility, balance, manual dexterity, physical strength, visual acuity or mental acuity to use them effectively. Some people with incontinence or toileting problems are able to look after their own needs but others are dependent on the help of family members (who may themselves have health issues and challenges) or professional caregivers in their own homes or in hospitals or nursing homes. The preferred life-style of product users is also important, as are the environments in which they spend their time, each environment having its own physical characteristics, facilities and challenges. Finally, any two people who are alike in all the characteristics described so far may arrive at different solutions because they have different personal priorities and preferences.
Table 2 provides a list of product characteristics that commonly arise in identifying those products most likely to meet the needs of users, bearing in mind that the needs of a given user may vary with context: given the opportunity to experiment with a range of products, users often opt for a ‘product mix’ rather than using the same one in all circumstances. For example, they may prefer one at home, another at work and yet another on social occasions, each product providing the best match to their needs in its chosen context. 3
The most obvious requirement of any continence product is that it should deliver on its core function of preventing or containing the leakage of urine and/or faeces, or enabling toileting. Further, given the potentially embarrassing, unpleasant and inconvenient consequences of failure, the core function should be delivered reliably. Aesthetics and discretion are also important. Users are understandably keen not to advertise their continence problem by using products that, for example, are visible beneath their clothing, rustle noisily when they move or allow the escape of unpleasant smells. Similarly, potential impact on body image and self-esteem may be important considerations; for example, a product that makes the user look fatter, limits their choice of clothing or has infant-connotations may be rejected. Continence products can be uncomfortable and may cause unintended harm; for example, they may damage the skin through rubbing – causing irritant contact dermatitis – or by failing to keep it dry. They may allow the release of unpleasant odours. Even a functionally perfect product will deliver no benefits to users if it is difficult to access or the price puts it beyond their reach. Finally, the demands of sustainability are receiving increasing attention from product manufacturers, purchasers and users, and there is a drive to eliminate unnecessary packaging, reduce reliance on single-use plastics, and avoid sending used products to landfill or for incineration, where possible.
Incontinence-related skin health and odour
Of the product characteristics listed in Table 2, incontinence-related skin health and odour containment are best considered across all product categories (below) although brief mention will also be made in various product-specific contexts in the sections that follow.
Incontinence and skin health
Skin that is exposed to urine and/or faeces repeatedly or for extended periods is at high risk of developing incontinence-associated dermatitis (IAD) 4 which is coded as irritant contact dermatitis due to incontinence (EK02.22) in the latest WHO ICD-11. 5 Prevalences between 3% and 60% have been recorded for various cohorts of incontinent people. 6 Early clinical signs include erythema, oedema and pain, while later stages are associated with maceration, erosions and excoriations. 7
Sustained contact with urine and/or faeces over-hydrates skin, compromising its barrier function, and making it more susceptible to infection and to penetration by harmful chemicals. Also, over-hydrated skin has an increased coefficient of friction against other materials and it is more easily damaged mechanically, for example by rubbing against a continence product. 8 IAD prevention is helped by good skin care and many cleansing and barrier products are available. 9
Studies to evaluate continence products have often included skin health as an outcome 1 but few have made it their primary focus. Taken together, they indicate that absorbent products10–13 and faecal collection devices 14 reduce the likelihood of IAD development and/or promote IAD healing. When comparing different absorbent products, only minor differences have been reported in terms of skin health,10,12 but there is some evidence that using a product with higher absorption capacity decreases IAD development 11 and promotes IAD healing. 13
The clear, primary message to continence product developers is to avoid over-hydrating users’ skin and avoid exposure to potentially irritant components of products. Rubbing between continence product and skin is also to be avoided. How this is to be achieved may, of course, vary considerably between product categories.
Incontinence-associated odour
Fear of odour from leaked urine, stool or flatus is a major concern that preoccupies many people with incontinence. Concern about odour can interfere with social relationships, cause people to isolate themselves within their home, or serve as a trigger to seek clinical care.15–17 Caregivers are also bothered by odour. 15 The odour associated with urine leakage is mainly due to the production of ammonia from urea by bacterial ureases, whereas factors that contribute to the odour of faeces (and flatus) include differing states of health and gastrointestinal function, diet composition, the profile of colonic microbes, relative concentrations of volatile gases containing sulphur and hydrogen, and, possibly, short-chain fatty acids or ammonia.18,19 Understandably, there is a demand for products that will mask, contain or – preferably – prevent odour.
Current approaches to addressing malodour can be described as a hierarchy of strategies. A first strategy is reduction/elimination of the source, such as sulphur-containing foods, which when digested release odorous gases. A variety of foods contain sulphur, such as sources of protein (milk, cheese, eggs, meat, lentils, nuts and seeds), cruciferous vegetables (broccoli, cauliflower and kale), allium vegetables (onions, garlic, shallots and leeks), and some whole grains (whole wheat, rye and barley). 20 There is evidence that malodour associated with faeces or flatus may be reduced by ingesting certain probiotics, charcoal mixtures or bismuth subgallate.21–25
A second strategy is to reduce odour from urine/faeces that is – itself – successfully contained. The clinical effectiveness of treating fabric-based continence products with antimicrobial agents to reduce the odour associated with urine in them is unclear 26 but success with a washable bodyworn absorbent product has been reported. 27
Charcoal can absorb odour associated with flatus and charcoal-activated briefs are more effective than charcoal-activated cushions or pads.19,28 The growth of odour producing bacteria can be reduced by lowering the pH of the superabsorbent polymer used in disposable absorbent products. 29 Applying scent to disposable body worn pads does little to mask the odour associated with urine. 30
Another common source of unpleasant odours from contained urine/faeces concerns handheld urinals, bedpans and commodes. They cannot always be emptied straight after use and, also, smells may persist if they are not fully emptied or thoroughly cleaned. 31 It is likely that much of the smell associated with incontinence comes from urine/faeces that has leaked from continence products into clothing, soft furnishings or bedding: this probably explains why penile sheaths – which rely on a good seal with the body – are associated with less malodour than absorbent products.16,32
A third strategy for odour reduction is, therefore, to give freedom from leakage a high priority when selecting products. A fourth strategy is to reduce the impact of any urine (or faeces) that
Although there have been many attempts to address the problem, urine- and faeces-related malodours – feared or real – remain an important challenge in need of urgent attention. A useful account of human olfactory processes has been published and used as a basis for suggesting strategies for tackling the problem. 34
In the sections that follow, the main design categories of products for managing incontinence or toileting are reviewed in turn. Just a limited selection of references is provided, but a more extensive review of the literature is available in the book chapter on which this paper is based. 1 For each product category: (i) the generic design approach and key features of current products are outlined; (ii) a generic functional design specification is provided; (iii) a review of how well existing products meet the requirements of their main user groups is offered; and (iv) priorities for the attention of product designers are suggested.
Handheld urinals and bedpans
Handheld urinals are portable devices designed to allow individuals to empty their bladder in circumstances where gaining access to a toilet is not possible or is inconvenient, often due to their (temporary or long-term) limited mobility or range of joint movement.35,36 Bedpans are designed to contain faeces as well as urine and they usually require caregiver support. 37 A general functional design specification for an effective handheld urinal or bedpan is given in Table 3 while examples of existing designs are shown in Figure 1.
General functional design specification for a handheld urinal/bedpan (with or without assistance from a caregiver).
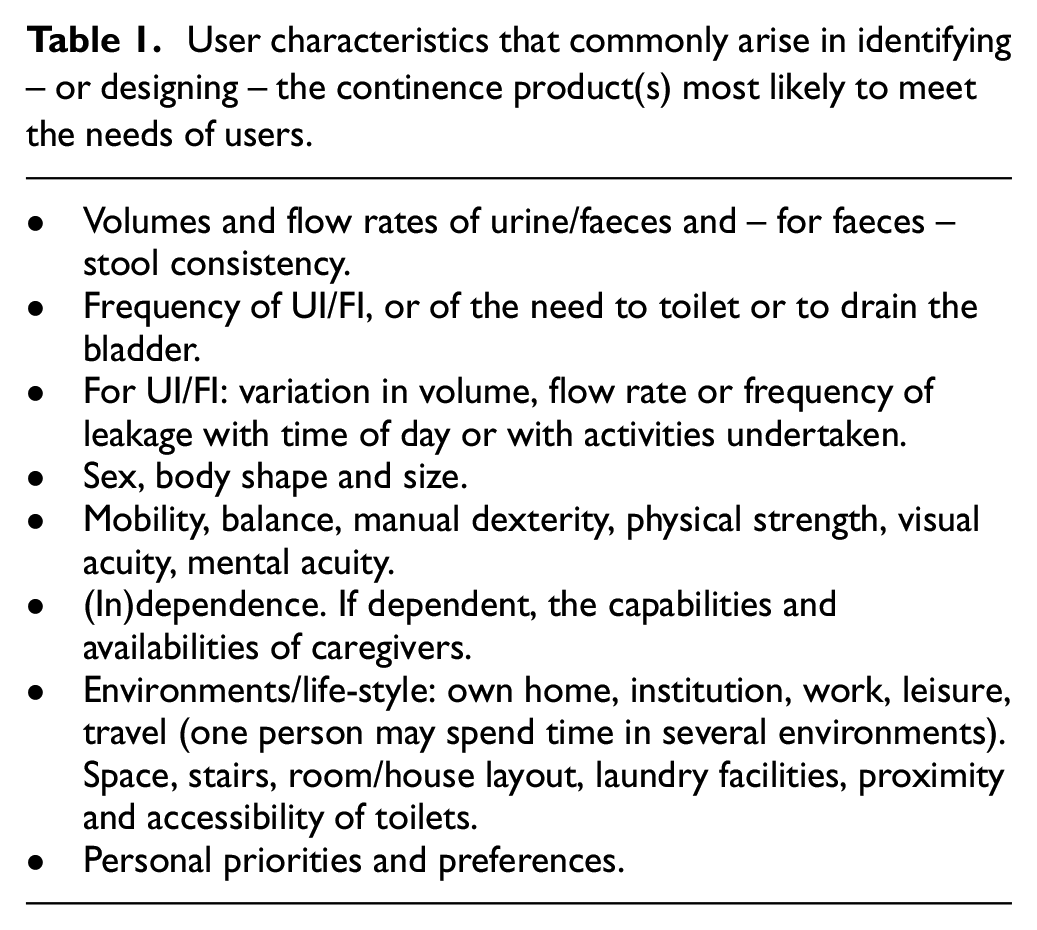
Example female handheld urinal (left) and male collapsible, handheld travel urinal (shown collapsed, and ready for use) (right).
Most handheld urinals are made from moulded plastic, although single-use cardboard designs are available. 38 Some are equipped with handles to facilitate positioning and handling, and some are fitted with a drainage bag to collect urine. Products designed for women are available in various shapes and sizes (some quite small and portable), incorporating an interface opening that is often shaped to fit snugly against the woman’s body. Those intended for men vary less in design, typically having a narrow neck opening to accommodate the penis. Some are fitted with integral non-spill valves intended to prevent the back-flow of urine after use.
It is easier for a handheld urinal to meet the design specification given in Table 3 if the intended user has a penis and existing products for men generally work well. Designing for the female anatomy is more challenging. The use of bedpans is associated with pressure ulcer/injury risk in individuals who have limited mobility or are seriously ill. 39
Priority unmet needs for handheld urinals/bedpans
New handheld urinal designs that better (Table 3) meet the needs of women who are less physically able, and are unable to move to the edge of a bed, chair or wheelchair, or need to use a urinal while supine.
New bedpan designs which are more comfortable; safer to use; less likely to cause soft tissue damage; and increase users’ confidence that they are correctly positioned to avoid spillage.
Commodes and toileting equipment
Toileting (urination and defaecation) is a fundamental aspect of continence and personal care but accessing and using conventional toilets is a complex undertaking that is challenging for people living with a variety of disabilities, including many who have normal bladder and bowel function. Toileting requires core stability, balance, strength, range of movement and cognitive, sensory, perceptual and manual skills to transfer to and from the toilet, adjust clothing, and accomplish wiping/cleansing. A variety of assistive devices exist to help people use conventional toilets which would otherwise present great challenges. Toilet seat raisers, padded toilet seats, grab or support rails, bottom wipers (for gripping and applying toilet paper) and adaptations to clothing are all in common use.
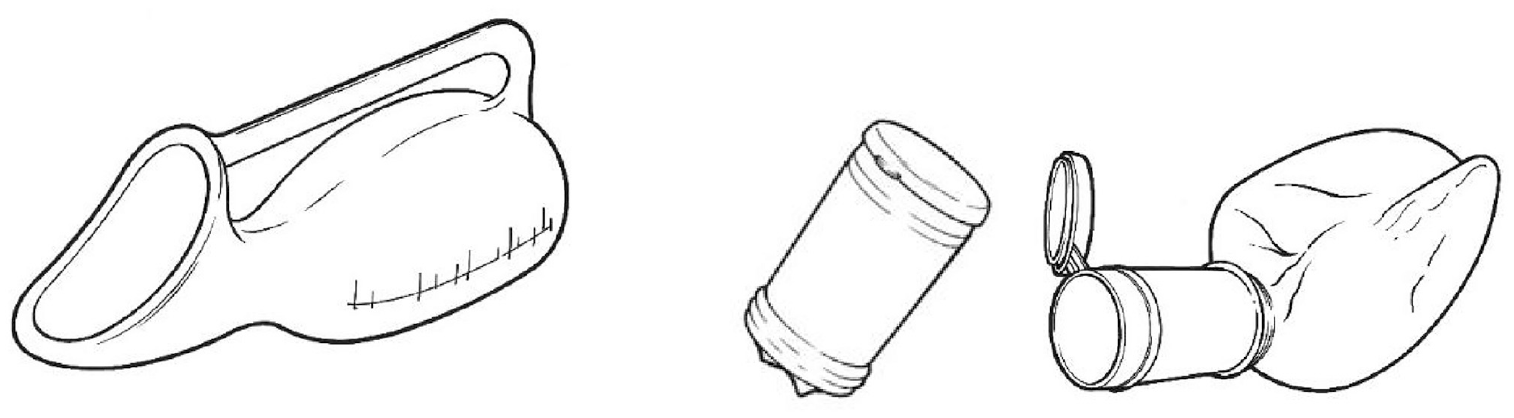
Where an individual cannot access a toilet quickly and safely, adapted alternatives – such as commodes40–42 may be appropriate (Figure 2). In lieu of a toilet, commodes are static or mobile pieces of equipment that comprise a chair-like frame incorporating a toilet seat which holds a removable pan (disposable or washable) positioned to receive urine and faeces. Mobile shower-chairs43,44 are also available. These are waterproof chairs on wheels that incorporate a toilet seat and are designed to be manoeuvred over a conventional toilet once an individual is seated on them. However toileting is to be tackled, the overall process should be achieved against the general functional design specification offered in Table 4.
Chair with commode (left) and toilet frame with integral raiser and grab bar (right).
General functional design specification for a commode, mobile shower chair (with or without assistance from a caregiver, or ancillary toileting equipment such as grab rails).

As with handheld urinals (above) the toileting needs of users with relatively minor physical limitations are generally well met by existing equipment, while satisfactory solutions are harder to find, the greater their disability.
Priority unmet needs for commodes and toileting equipment
The development of validated assessment tools for accurately characterising a user’s capabilities and needs, identifying the product(s) most likely to meet their needs, measuring the effectiveness once implemented, and accurately describing unmet needs for the attention of designers.
The development of commodes with better stability than existing products.
The development of commodes with improved aesthetics for use in home environments, including minimising odour.
Absorbent products
Absorbent products are by far the most commonly used for containing incontinence – especially for women, for whom there are few alternatives. Most absorbent products are bodyworn but some (underpads) are designed to be placed beneath the seated or supine user, and products in either category may be disposable (discarded after a single use) or washable (laundered and reused multiple times). Although each category encompasses many brands, all products fall into one of just a small number of generic designs (Figure 3), which further divide into those intended for male or female anatomy (or both) or children beyond the usual diaper age. Products often come in a range of variants to accommodate different body sizes, and/or different severities of incontinence. Most products are designed to contain only urine but – provided they extend over the anus – they may also work for light FI. Very few products are designed for more severe FI (with or without UI) and those that do, seek to contain faeces rather than absorb it. Despite this diversity of both products and users, priorities for product performance are notably consistent across user groups 45 and Table 5 provides a general functional design specification.
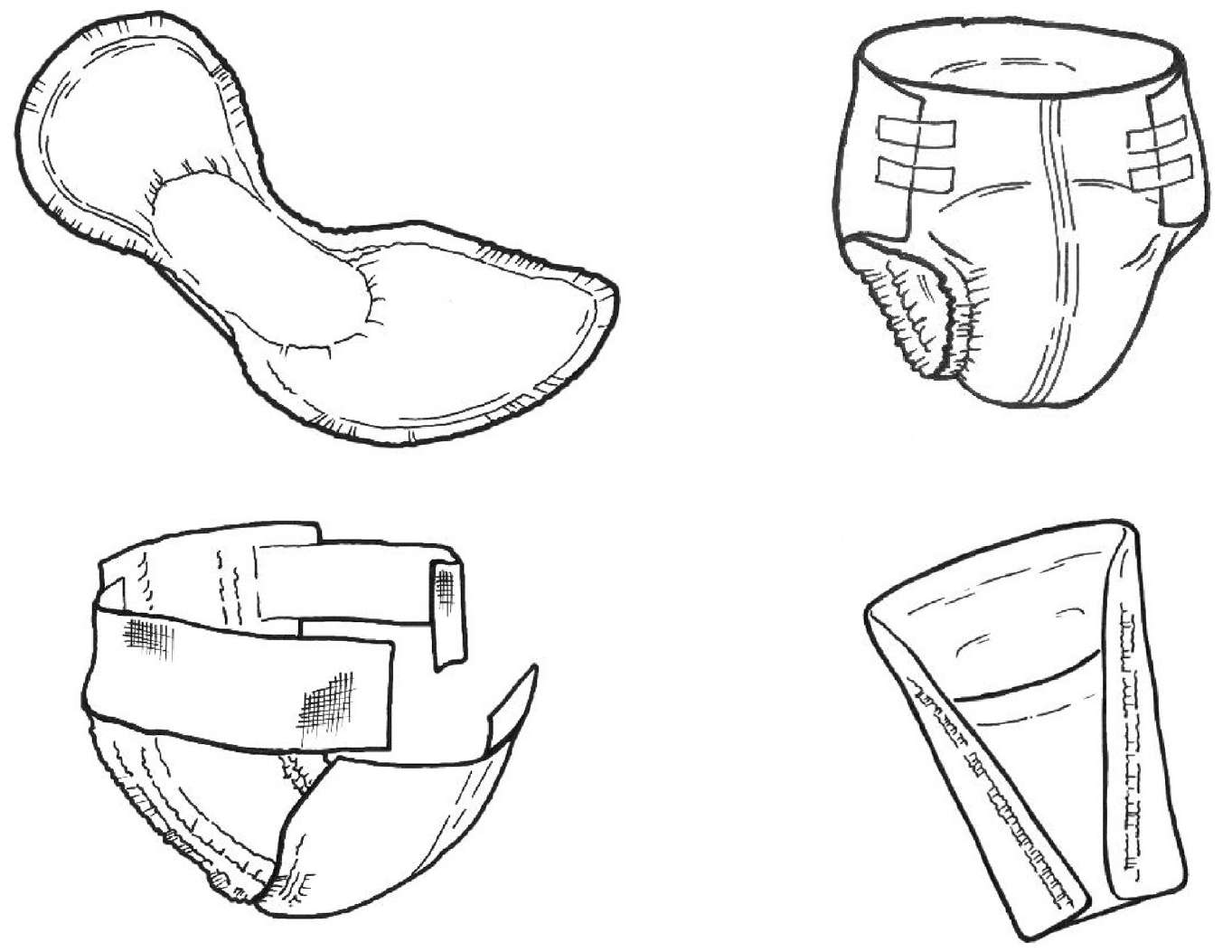
Disposable, bodyworn absorbent products for moderate/heavy incontinence: shaped pad (top left), all-in-one (top right) and belted pad, shown open (bottom left). Male pouch (bottom right).
General functional design specification for an absorbent incontinence product (with or without assistance from a caregiver).

Unsurprisingly, the smaller the volume of urine leakage experienced and the more mobile, dexterous and independent the person is, the easier it is to identify an absorbent bodyworn product that successfully meets their needs (Table 5). Those with heavier incontinence are generally less well served, especially if they have reduced mobility and manual dexterity and rely on assistance for product changing. The ability of disposable products to contain urine without leakage and to protect skin from damage has steadily increased in recent years 46 but further improvements would significantly increase the quality of life of many millions of users worldwide.
Because disposable bodyworn products for moderate-heavy UI usually account for the lion’s share of incontinence product expenditure for healthcare organisations that buy in bulk, there have been considerable efforts to develop laboratory test methods that can predict the leakage performance of products, so avoiding time-consuming, expensive user evaluations to inform buying decisions. There is an international standard 47 test method which was developed for this purpose and it is widely used internationally to assist with purchasing choices. It was clinically validated with products available up to about 2007 but there is evidence that it is blind to some features of modern products known to confer user benefits. 48
Washable absorbent bodyworn incontinence products have been available for many years; indeed, they predated the disposable products that have now largely replaced them. They can be cheaper – on a per-use basis – than disposables but their performance is generally inferior, especially with regard to containing urine without leakage and to comfort. 45 The burden of washing and drying them between uses often falls on products users or their carers, which can be problematic. Those washable products intended for heavier incontinence are particularly disappointing in these respects, although products for light UI that have been fashioned to resemble regular underwear are appreciated by some for their aesthetics. An increasing awareness of the environmental impact of product manufacture, use and disposal has provoked a renewed interest in washables among both users and manufacturers. However, designing new reusable products that match the performance of their single-use counterparts is challenging.
Many disposable products designed to contain UI also cope adequately with light FI (faecal smearing) but none deal effectively with heavier FI, especially if stool is fluid, and even more especially if the FI is combined with UI, bathing the skin in a highly corrosive mix.
The improvement of underpads – disposable or washable – is not a high priority since their use is declining.
Priority unmet needs for absorbent products
The development of a robust, clinically-validated alternative to ISO 11948-1 47 for measuring the absorption capacity of single-use, bodyworn absorbent products for heavy UI.
The development of washable, bodyworn absorbent products for heavy UI with improved leakage performance, that wash and dry well and are aesthetically acceptable.
The development of single-use, bodyworn absorbent products that will contain heavy FI effectively – with or without associated UI.
The development of absorbent products with a lower impact on the environment in terms of their manufacture, use and disposal.
Bodyworn urine collection devices
Bodyworn urine collection devices are primarily available for men and they come in two broad design categories, generally referred to as sheaths3,49–51 and bodyworn urinals. Sheaths (sometimes called condom catheters, uridomes, Texas catheters or external catheters) fit closely over the penis, much like a contraceptive condom (Figure 4). They receive urine as it leaves the body and convey it – via an integral tube at the distal end – to a drainage bag (worn on the thigh or calf, or suspended from the waist) where it is stored until emptying is convenient.
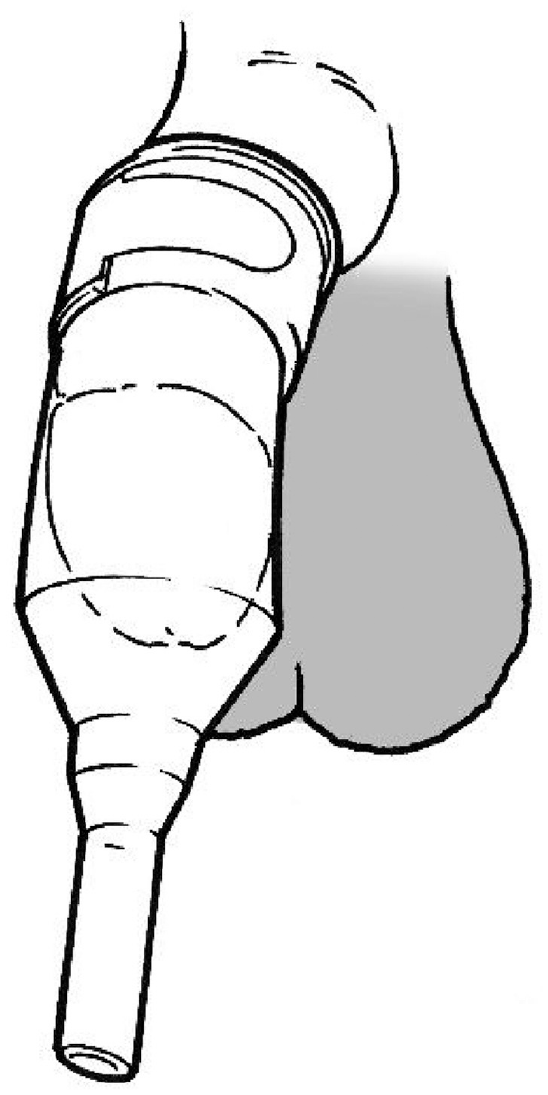
Sheath without integral adhesive, secured with external fixation.
Sheaths are usually made from latex or silicone rubber and supplied in a range of diameters (typically 20–40 mm, in 5–10 mm increments). One-piece systems include integral skin adhesive to secure them to the penis shaft while in two-piece systems the adhesive is applied to the penis skin as a spray or comes as a separate strip. A band on the outside of the sheath may also be used (with or without adhesive between sheath and skin) to achieve fixation (Figure 4). The section of the sheath between the end of the penis and the start of the drainage tube may become twisted or kinked, or the internal walls may stick to one another between voids. This can lead to (partial) blockage and sheaths often include design features aimed at addressing these problems. Some variants are designed to accommodate a retracted penis (common in older men): they may be shorter or have a wider adhesive seal. One design intended for men with a retracted penis is applied to the glans rather than the shaft of the penis. Some sheaths are provided with an applicator which aims to make application easier or reduce manual contact with the penis. Sheaths are typically changed every 24 h. Table 6 provides a general, functional design specification for an effective sheath.
General functional design specification for a sheath.

Though many men use sheaths successfully, it is clear from the literature that selecting an appropriate product, fitting it effectively and using it successfully is challenging and often not achieved. Much of the consequent leakage, skin damage and discomfort that has been reported has been attributed to lack of training for caregivers but may also be a consequence of lack of user-focussed design. The literature on sheath use is quite sparse and there is a paucity of published evidence for the efficacy of many design features intended to address well-known problems.
Products in the second category of bodyworn urine collection devices for men are usually called bodyworn urinals (BWUs) and they are usually made of latex. They typically comprise a ring-shaped opening or cone-shaped component which is worn (loosely) around the penis, held firmly against the pubis by means of a belt and straps, and attached to an integral urine collection bag (Figure 5).
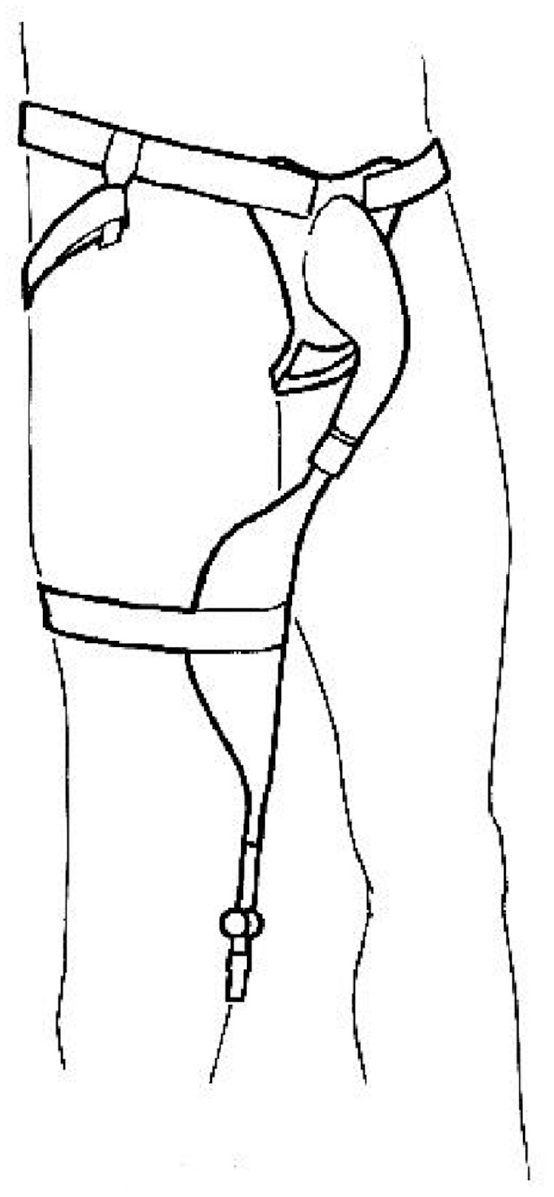
Bodyworn urinal with leg bag.
Bodyworn urinals (BWUs) are more substantial collection devices than sheaths and they are designed to be washed and reused multiple times and worn for extended periods. Fewer men use them than sheaths, but they may be a good option for those with a retracted penis. Dribble containers are a subset of BWUs and they involve holding a drainage bag or other container over the penis using a much lighter structure than the flanges used in BWUs. They are often disposable. Unsurprisingly, a general functional design specification for an effective BWU (Table 7) overlaps considerably with that for a sheath (Table 6).
General functional design specification for a bodyworn urinal.

BWUs are not widely used but they do deliver significant benefit to a minority of men, especially as part of a mix in which each product works best in different circumstances. 3 The literature on BWUs is even more sparse than that on sheaths but it is clear that existing designs are generally unpopular with (potential) users and poor aesthetics pose a particular challenge.
Finally, numerous attempts have been made to design bodyworn urine collection devices for women but very few have reached the market and none is widely used. The female anatomy makes it much harder to meet requirements (Table 7) in an aesthetically acceptable way for women than it does for men. However, there is growing interest in addressing the needs of severely disabled or immobile women (and men) using a bodyworn cup or pad containing a moisture sensor which activates a pump when needed to draw urine away to a suitably-sited receptacle. 52
Priority unmet needs for bodyworn urine collection devices
The development of improved sheath designs which better meet the requirements listed in Table 6.
The development of improved sheath designs focussing on the particular needs of men with a retracted penis.
The development of validated assessment tools for accurately characterising a sheath user’s capabilities and needs, identifying the product(s) most likely to meet their needs, measuring the effectiveness once implemented, and accurately describing unmet needs for the attention of designers.
The development of improved bodyworn urinal designs for men and women which better meet the requirements listed in Table 7 and have improved aesthetics.
Urine drainage bags
Urine drainage bags aim to provide discreet, temporary storage of urine and can be attached to either an indwelling catheter or a sheath. They are generally made from PVC, PVDF (less noise from rustle) or PE and fall into two main categories. First, bodyworn bags generally have a lower capacity (usually ≤500 ml) and are designed to be worn under clothing on the leg (calf or thigh) or suspended from the waist. They are mainly used during the day and are secured using straps or other support devices. Second, there are large capacity bags (usually ≥2000 ml) – commonly called night bags – which are designed to provide additional drainage capacity overnight or for people who are permanently in bed. They are usually held in a position away from the body on either a floor-stand or a hanger that hooks onto the bed or chair frame. They may also be connected to a bodyworn bag to form a link system for overnight use. Bags are generally changed every few days: 3–20 days is reported. An example urine drainage bag - used in combination with a sheath - is shown in Figure 6. Table 8 provides a general, functional design specification for an effective urine drainage bag.
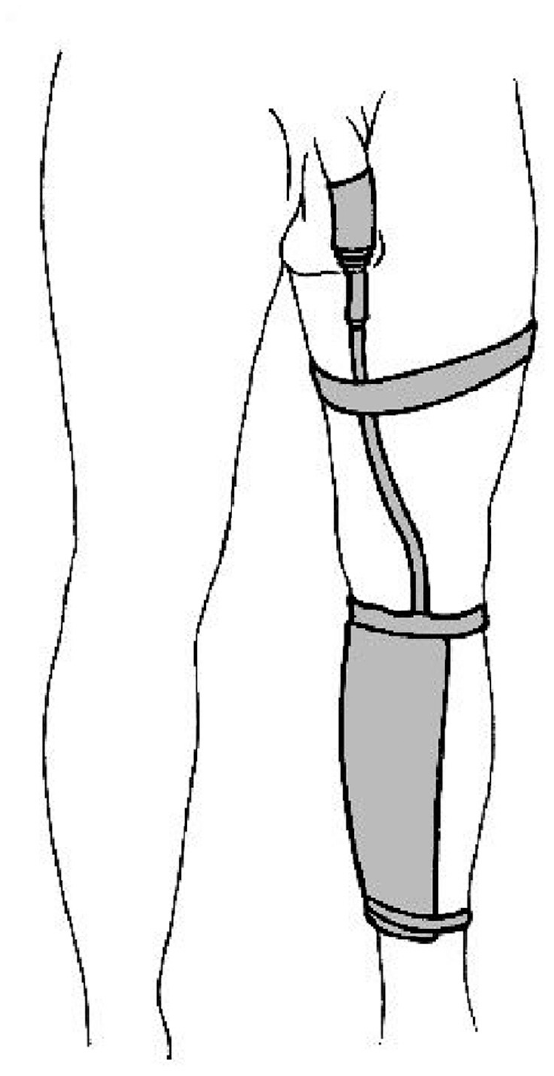
Example of a bodyworn urine drainage bag attached to a sheath and secured on the calf.
General functional design specification for a urine drainage bag.

A limited number of evaluations of urine drainage bags have been published.53–55 Urine drainage bags generally work well, and the popularity (or otherwise) of many features is a matter of individual needs, capabilities and preferences. For example, a user with arthritic fingers may need a larger, easier to operate tap despite the disadvantage of poorer discretion beneath clothing. Preferences on where (calf, thigh or waist) and how (latex or fabric straps or a belt) to secure bodyworn bags vary widely. Loops in the tube feeding a drainage bag can produce air locks (called dependent loops in the literature) which can increase resistance to flow enough to provoke leakage elsewhere in the system. 56
Priority unmet needs for urine drainage bags
New urine drainage bag designs that offer improvements regarding the ergonomics of putting on, emptying, removing and cleaning, as well as comfort and discretion (Table 8).
A solution to the problem of dependent loops.
Mechanical devices for women with UI
Mechanical devices for women with UI are designed to prevent stress urinary incontinence (SUI); that is, leakage that accompanies increased bladder pressure following coughing, sneezing or physical activity. They are positioned in the vagina, in the bladder, or in/over the urethra (Figure 7). 57
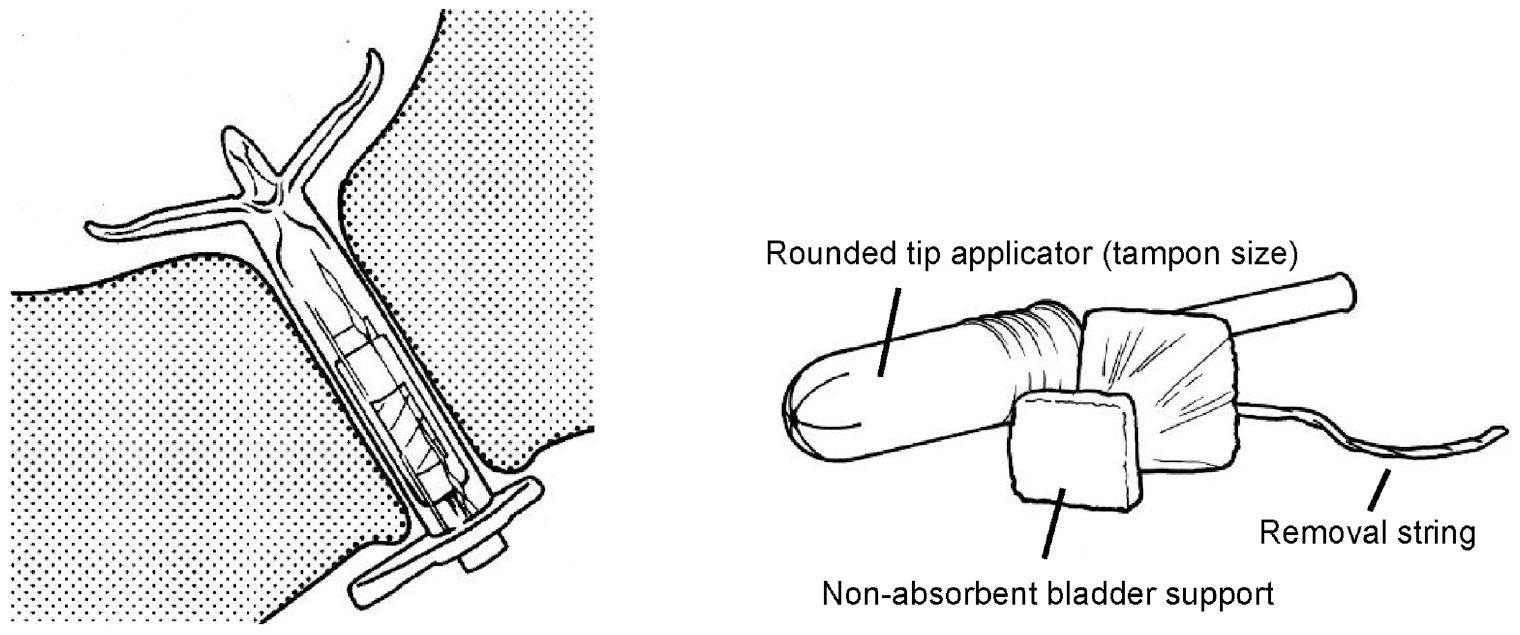
Examples of mechanical devices for women with UI: InFlow Intraurethral Valve-Pump (by Vesiflo, USA), in position (left) and Poise)/Impressa (by Kimberly Clark, USA) intravaginal device (right).
Internal vaginal devices are the most common, and they include tampons and pessaries along with products designed specifically to address SUI. They aim to prevent leakage at source by supporting the bladder neck via the vagina, which sits immediately posterior to the urethra. An intravesical device is also available which adopts a different strategy to leakage prevention at source, reducing the magnitude of the transient bladder pressure rises that accompany coughing and the like. 58 Made from a highly compressible material, its volume decreases substantially when the bladder pressure rises, so reducing the magnitude of the momentary challenge to the urethral closure mechanism. Urethral devices increase resistance to urine leakage by occluding the urethral meatus (external urethral devices) or the urethra itself (internal urethral devices). Some devices of the latter category are intended to be worn for a short period, during activity (like physical exercise) that is likely to provoke leakage. Others are designed to be worn for longer periods and include a valve that can be opened for bladder emptying using a magnet. Table 9 provides a general, functional design specification for an effective mechanical device for women with SUI.
General functional design specification for a mechanical device for women with SUI.

There have been numerous attempts to develop mechanical devices for women with SUI and, although several have reached the market, few have been subjected to independent, long-term, user trials of reasonable size. Trials usually report high drop-out rates and a high incidence of discomfort. It is clear that not all women support the design concept from an aesthetic point of view. 57 However, it is equally clear that a minority of potential users are benefitting. It is, therefore, unsurprising that the European Association of Urology recommends that physicians should not routinely offer intravaginal devices as a treatment for incontinence. 59
Priority unmet needs for mechanical devices for women
New designs that meet requirements (Table 9) better than existing designs, preventing urine leakage without causing tissue damage. (However, it would be unwise to devise new designs until the (non) efficacy of existing products is established and the reasons for success and failure are better understood, including their long-term value, effects and safety.)
Mechanical devices for men with UI
Male mechanical devices (also called penile clamps or penile compression devices) are designed to prevent urine leakage by compressing the penile urethra using a clamp or a peri-penile strap (Figure 8). Table 10 provides a general, functional design specification for an effective device.
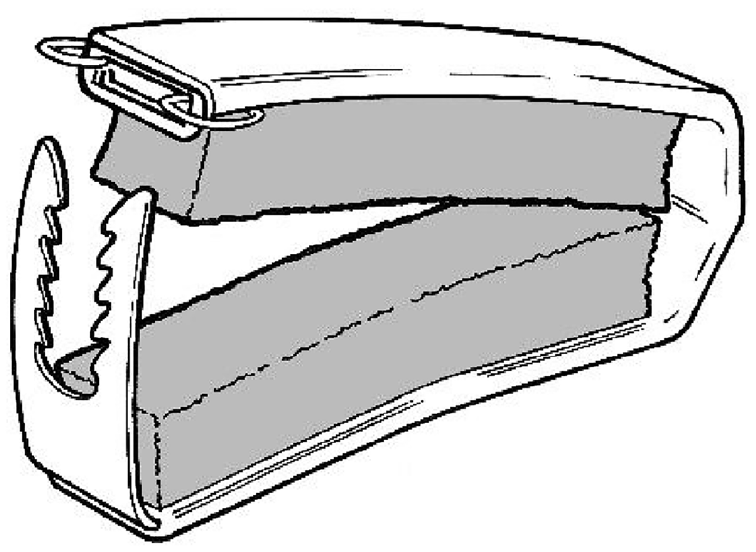
An example penile clamp.
General functional design specification for a mechanical device for men with UI.

Although a range of devices is available, there has been little research on their safety and efficacy. 60 There is clearly potential for products to cause tissue damage and penile ischaemia, and so they should be used with caution. The clinical opinion is that they should be released at least every 2 h and are unsuitable for use overnight or when sleeping. 3 Although they cannot be used for protracted periods, some men find them very useful for limited periods involving activities likely to provoke leakage (such as sport or dancing), as part of a mix of products in which each works best in different circumstances. 3
The core challenge in designing mechanical devices is how to occlude the urethra without causing soft tissue damage and some useful work has been done to measure the impact of various device designs on penile blood flow. 61
Priority unmet needs for mechanical devices for men
New designs that meet requirements (Table 10) better than existing designs, preventing urine leakage without causing tissue damage.
Indwelling catheters
Indwelling urinary catheters are used to drain the bladder and they may be inserted via the urethra (most commonly) or suprapubically (Figure 9). In almost all designs, a balloon near the proximal end of the catheter – just below the drainage eyes – is inflated after insertion to anchor it in place, and deflated before removal. Catheters are generally supplied in different lengths (shorter for women; longer for men) and a range of outside diameters (measured in Charrière: 1Ch = 1/3 mm) and balloon sizes. Those for short-term use are commonly made from latex, while those for long-term use (> 30 days) are usually made from silicone rubber which – being stiffer than latex – can provide a larger drainage lumen for a given outside diameter and overall catheter stiffness. Catheters are usually connected to a urine drainage bag for continuous drainage. Alternatively, they may be fitted with a tap (catheter valve), enabling urine to be retained in the bladder until emptying is convenient. Table 11 provides a general, functional design specification for an effective indwelling catheter.
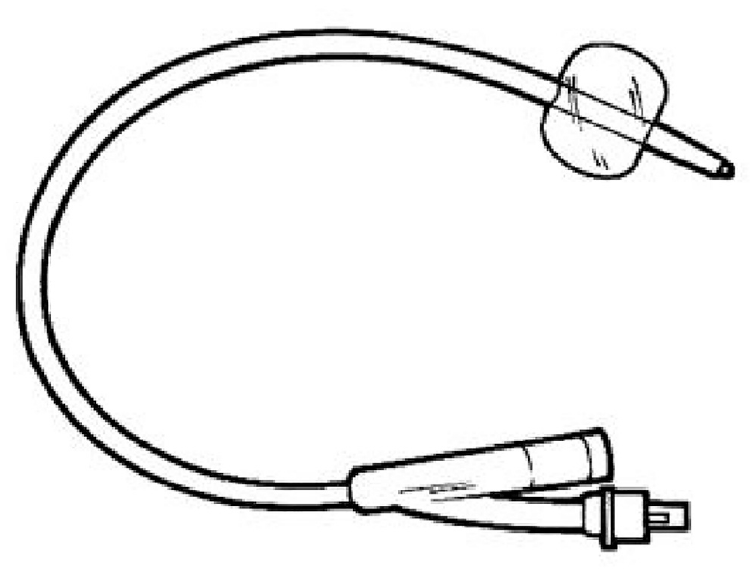
An indwelling catheter with balloon inflated.
General functional design specification for an indwelling catheter.

Indwelling urinary catheters have some major, well-documented shortcomings that have proved remarkably resistant to efforts to overcome them. 62 Accordingly, experienced clinicians have concluded that catheters should not be inserted as a continence measure until all other reasonable management and treatment strategies – such as toileting, containment products and medication – have been ruled out. 63 The two biggest – and mutually confounding – problems are catheter-associated urinary tract infection (CAUTI) and catheter blockage. The probability of individuals with indwelling urinary catheters developing microbe colonisation (asymptomatic bacteriuria) increases by ~3%–8% per day, to 100% by day 30, 64 and recurrent urinary catheter blockage caused by encrustation occurs in 40%–50% of long-term catheterised patients. 65 There have been numerous attempts to address these problems by using, for example, novel materials, coatings and surface treatments, and catheter washout / maintenance solutions, resulting in new introductions to the market. However, their efficacy has never been confirmed when large, carefully designed evaluations have been conducted.
The list of failed attempts to solve these problems is discouragingly long but the need remains and, given that indwelling catheters are the most commonly used incontinence devices after absorbent products, the potential benefits of any success are enormous.
Priority unmet needs for indwelling catheters
New designs/materials which resist the microbial biofilm formation that leads to CAUTI and blockage in current designs.
Intermittent catheters
Intermittent catheterisation (IC) involves passing a catheter into the bladder to drain urine and then removing it until emptying is next needed. The catheter may be a simple open-ended tube or – more usually – the proximal end may be closed and drainage eyelets provided in the catheter shaft adjacent to the tip. Catheters are supplied in different lengths (shorter for women; longer for men) and a range of outside diameters. They are usually made from PVC. Insertion is often facilitated by applying a gel to the shaft surface but many are supplied with an integral, hydrophilic, lubricious coating. A variety of ancillary devices is available; for example, mirrors to help women locate their urethral meatus, and catheter holders for those with poor manual dexterity. IC catheters may be designed for single use or – less commonly – for use multiple times, cleaning between uses. Multiple-use catheters could reduce catheter waste and cost, but their equivalence with single-use catheters (in terms of urinary tract infection (UTI) risk) has not been established. 66 Policy regarding single-use versus multiple-use of IC catheters varies between countries. Table 12 provides a general, functional design specification for an effective IC catheter (and ancillary devices).
General functional design specification for an IC catheter (and ancillary devices).

Many people use IC very successfully: IC catheters are small and unobtrusive and, unlike many other continence devices, do not have to be worn continuously. However, some who could – in principle – benefit from IC, are reluctant or unable (due to poor manual dexterity or eyesight) to perform the technique themselves, unwilling to accept the procedure from a caregiver, or not offered the opportunity to try. 67 IC avoids many problems associated with indwelling catheters, but UTI remains an issue for many users. 63
Many companies have given careful thought to the ergonomics and aesthetics of IC catheters and their use, offering – for example – insertion aids and unobtrusive packaging.
Priority unmet needs for intermittent catheters
Reusable catheters and associated cleaning materials and methods that perform as well as single-use coated catheters in terms of insertion (i.e. smooth/low friction), ease of use and incidence of infection.
Products and devices for preventing or managing faecal incontinence and its sequelae
Products rely on a variety of – often ingenious – approaches to preventing or managing faecal incontinence (Figure 10).

Examples of products for preventing FI: Peristeen anal plug (by Coloplast, Denmark) (left) and Renew anal insert, with applicator (on the end of the finger) in place (by Renew Medical, UK) (right).
Anal plugs, anal inserts, and vaginal inserts aim to prevent faecal leakage, and may be suitable for community-living, physically mobile individuals68–70 (The vagina sits immediately anterior to the rectum and vaginal inserts occlude the rectum by pushing on the dividing wall.). Rectal catheters, long and short rectal tubes, and non-balloon-based intra-rectal sheaths redirect faeces from the rectum, collecting it in an external drainage bag for patients confined to bed in hospital or long-term care settings due to illness or functional disability.71,72 Peri-anal pouches and various types of absorbent pads (see above) collect or absorb faecal leakage.73,74 Table 13 provides a general, functional design specification for an effective device for preventing or containing FI.
General functional design specification for a device to prevent or contain FI.

People with FI vary enormously in their product needs, depending – in particular – on the quantity and consistency of the faeces to be contained, the nature of any rectum-related comorbidities, and whether or not they are ambulatory. Most published trials report that the products evaluated had delivered substantial benefit to some users while presenting serious limitations to others, making them ineffective or unacceptable. For devices placed in the rectum or vagina, discomfort and soreness68,69,75,76 are often reported along with difficulties with insertion and a tendency to fall out. Adverse effects of bowel catheter systems such as bleeding and pressure ulceration have been reported.77,78 Reported limitations with anal pouches are difficulty in applying them and maintaining their seal with skin, and damage to the peri-anal skin.73,79
Published trials of existing products are often small, uncontrolled and short-term and so the data they yield do not provide a robust and comprehensive basis for devising improved designs. Nevertheless, some common limitations of existing designs are well established (see above) and improved designs that address them for the different subsets of users from the highly heterogeneous FI population have the potential to substantially enhance their quality of life.
Priority unmet needs for products and devices for preventing or managing faecal incontinence and its sequelae
Perianal pouches that are easier to apply, maintain adherence and remain in place for an extended period, reduce odour, and divert faeces away from the skin to avoid skin damage.
A single-use perianal pouch that can be self-applied and removed easily.
An anal plug or insert that can be worn and retained for longer periods with greater comfort.
Conclusions
Most of the needs highlighted above are specific to particular continence product categories or user groups, but some – relating to skin health or odour or sustainability, for example – apply across product categories. Some scientific problems – if successfully addressed – would likely yield benefits in multiple contexts. For example, all continence products (in common with many other medical and non-medical devices) share an interface with soft tissues, usually the skin but sometimes the urethral, rectal or vaginal wall. A better understanding of the biomechanics of such interfaces, taking account of pressure and shear stress distributions, friction and the confounding effects of urine would probably provide useful insights for product designers. Similarly, a better understanding of how biofilms develop on the surfaces of catheters and other polymer-based products that are bathed in urine for sustained periods might suggest ways of disrupting their actions, so addressing the blockages and colonisation by bacteria that they often cause. The urea in urine gradually decomposes yielding ammonia which can contribute to odour, to catheter blockage and – if the urine is on the skin – to a rise in skin pH, leading to skin damage. Accordingly, finding ways to acidify urine or reduce ammonia formation in excreted urine, and better protect the skin from the effects of exposure to alkaline pH is likely to be helpful.
Finally, the heterogeneity of the population of continence product users gives rise to some challenges that – though ubiquitous – are easily overlooked. It is not surprising that people with different kinds and severities of incontinence or toileting problems can vary enormously in their product needs and priorities but – less obviously – this may also be true of different people managing a similar problem. A product that works very well for one may be rejected by another with similar bladder or bowel challenges but having, for example, different mobility, dexterity or lifestyle (Table 1). Indeed, the same individual may find that their needs are best met by using different products in different contexts. That’s why any clinician aspiring to help patients identify suitable products and use them effectively needs to be skilled in considering the wider contextual issues (Table 1). Likewise, any designer aspiring to develop successful continence products needs to factor those same issues into their work. Frustratingly, this may mean that what, initially, appeared to be a sizeable, homogenous user group fragments – possibly in a commercially disappointing way – into multiple subgroups whose varying needs are hard to reconcile in a single design.
Evidence of this heterogeneity is widespread across the continence product literature and one of the great needs in the field is for validated tools – a range might be needed, covering different product categories – that can be used to identify the key characteristics of both users and products and suggest promising matches worth trying. An ideal tool would also reveal why a particular product was (not) able to deliver what an individual needed, and suggest important target properties for future designs. The same tool might also help with evaluations of existing products. Published evaluations invariably report how (un)popular products were with users but they don’t always identify shortcomings sufficiently well to assist designers. For most product categories, clarifying which combinations of user and product characteristics lead to success and rejection – and why – would be enormously beneficial.
Although incontinence and toileting problems are seldom life-threatening, they do erode the quality of life of many millions of sufferers and caregivers. Improved continence products have delivered substantial benefits to many in recent decades but there remains significant room for improvement. It is hoped that this paper will help highlight unmet needs, challenging and encouraging designers to further innovation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
