Abstract
Owing to environmental and disease issues, the use of high-frequency chest wall oscillation (HFCWO) devices in hospitals is consistently increasing. This study proposes a cost-effective actuator-less HFCWO device that utilizes an external wall port utility in hospitals to generate the positive and negative pneumatic pressures required for HFCWO treatment instead of an embedded mechanical actuator. The manufactured prototype with the no-amplification (NO-AMP) setting contained an electric pressure regulator to enable intensity level adjustment and two solenoid valves to enable vibration frequency adjustment, whereas the prototype with the pre-amplification (PRE-AMP) setting contained an additional air reservoir and an air-pressure booster. The prototype device was tuned to output average local maximum values in the pressure waveform similar to a commercial VEST-205 device at an 8–12 Hz frequency and 2–4 pressure intensity levels. In vitro comparative experiments demonstrated that the prototype device showed similar local maximum pressures to those of the VEST-205 (mean absolute pressure difference, <3 mmH2O); in contrast, the proposed device showed significantly higher local minimum pressures than those of the VEST-205 (mean absolute pressure difference, >8 mmH2O). Additionally, the driving sound of the proposed device was 17.0–17.8 dB higher than that of VEST-205. We conclude that the proposed device has the potential to substitute for conventional HFCWO devices under the limited but most frequently used operating conditions, although more detailed modifications are necessary in future studies to improve its performance and clinical usability.
Keywords
Introduction
Approximately 30% of intensive care unit patients experience breathing difficulties and require respiratory support from ventilators. During ventilator treatment, an artificial airway is inserted into the patient’s throat that can disturb ciliary movements in the airway, suppress the cough reflex, trigger long-term secretion accumulation in the airway, and induce lung dysfunction and pneumonia. To alleviate such clinical complications, an airway clearance device such as a high-frequency chest wall oscillation (HFCWO) device is generally used in patients who have difficulty in self-discharging mucus and sputum from the airway due to acute asthma, chronic obstructive pulmonary disease, post-thoracic surgery, cerebral palsy, or other neuromuscular diseases.1,2 During airway clearance treatment, vibrating pneumatic power (5–20 mmH2O) with a frequency of 5–20 Hz is delivered to the body surface near the airway via the HFCWO device, which reduces the viscosity of the secretions, induces mini-coughs, and accelerates the discharge of secretions.3,4 Several studies have demonstrated the clinical efficacy of HFCWO treatment at improving treatment compliance, reducing symptoms of dyspnea, increasing oxygen saturation, improving lung function, increasing secretion discharge, and reducing pneumonia, with no particular side-effects.5–10
Most commercial HFCWO devices, such as the VEST, Monarch, MetaNeb (Hill-Rom Services Inc., Wilmington, DE, USA), MedPulse, SmartVest, (Electromed Inc., MN, USA), CareWay (Korust Co. Ltd., Anyang, Korea), inCourage (RespirTech, Plymouth, MN, USA), BiWaze (ABM Respiratory Care, LLC, Eagan, MN, USA), and RespIn 11 (Resp Innovation, Seillans, France), contain an air-pulse generator in the main body that generates necessary pneumatic power for chest wall vibration treatment. This independent-actuating mechanism is most favorable for out-of-hospital treatment (e.g., home or office). However, the presence of a mechanical air-pulse generator prevents small and cheap manufacturing of the HFCWO device, resulting in a high end-user price. In addition, owing to the recent increase in air pollution (e.g., yellow and fine dust) and the rapid spread of highly infectious respiratory diseases (e.g., influenza and coronavirus disease 2019 (COVID-19)), the requirement for HFCWO devices that alleviate the symptoms of respiratory diseases at a lower price is consistently increasing to improve the reaction capability of hospitals during emergencies such as pandemics.
This study proposes the concept of an actuator-less HFCWO device that utilizes a wall port utility instead of an embedded mechanical air-pulse generator. A prototype device with two different settings was manufactured and comparative in vitro experiments were performed using a commercial HFCWO device.
Materials and methods
Proposed device structure
Figure 1(a) is a schematic of an ideal wall port–equipped actuator-less HFCWO device dedicated for in-hospital use (no bulky tubes between the device and the wall port facility). The device contains neither a mechanical air-pulse generator nor electrical motors inside the case; rather, it is directly connected to the wall port utility (i.e., AIR and VAC ports) near each bed. Figure 1(b) shows a schematic of the moving cart–type wall port–connected actuator-less HFCWO device whose prototype was manufactured in this study that is connected to the wall port utility via AIR-IN and VAC-OUT tubes; it is also connected to a patient-worn jacket or maternity belt via a PAT-OUT tube. A high pneumatic pressure from the AIR port in the wall port utility was consistently supplied to the input port of the prototype device via an AIR-IN tube, which was then stepdown regulated by an electric pressure regulator (P-Regulator) to generate various pressure intensity levels before entering the vacant space inside the jacket/belt via a PAT-OUT tube (during inflation). Simultaneously, the input port of the PAT-OUT tube was connected to the VAC port to discharge air from the jacket/belt via the VAC-OUT tube (during deflation). The frequency of the vibrating air-pulse delivered to the jacket/belt was adjusted using the IN- and OUT-Valves. In this study, we manufactured a moving cart–type prototype device with two different settings: 1) no-amplification (NO-AMP) setting in which air pressure from the AIR port was directly entered into the input port of the P-Regulator (Figure 1(c)); and 2) pre-amplification (PRE-AMP) setting in which air pressure from the AIR port was pre-amplified by an air reservoir and an air-pressure booster and subsequently entered into the input port of the P-Regulator (Figure 1(d)). In both settings, two frequency-adjusting valves (IN- and OUT-Valves) were reciprocally controlled to induce periodic expansion and contraction of the vacant space inside the jacket/belt, which generated a chest wall vibration effect (Figure 2).
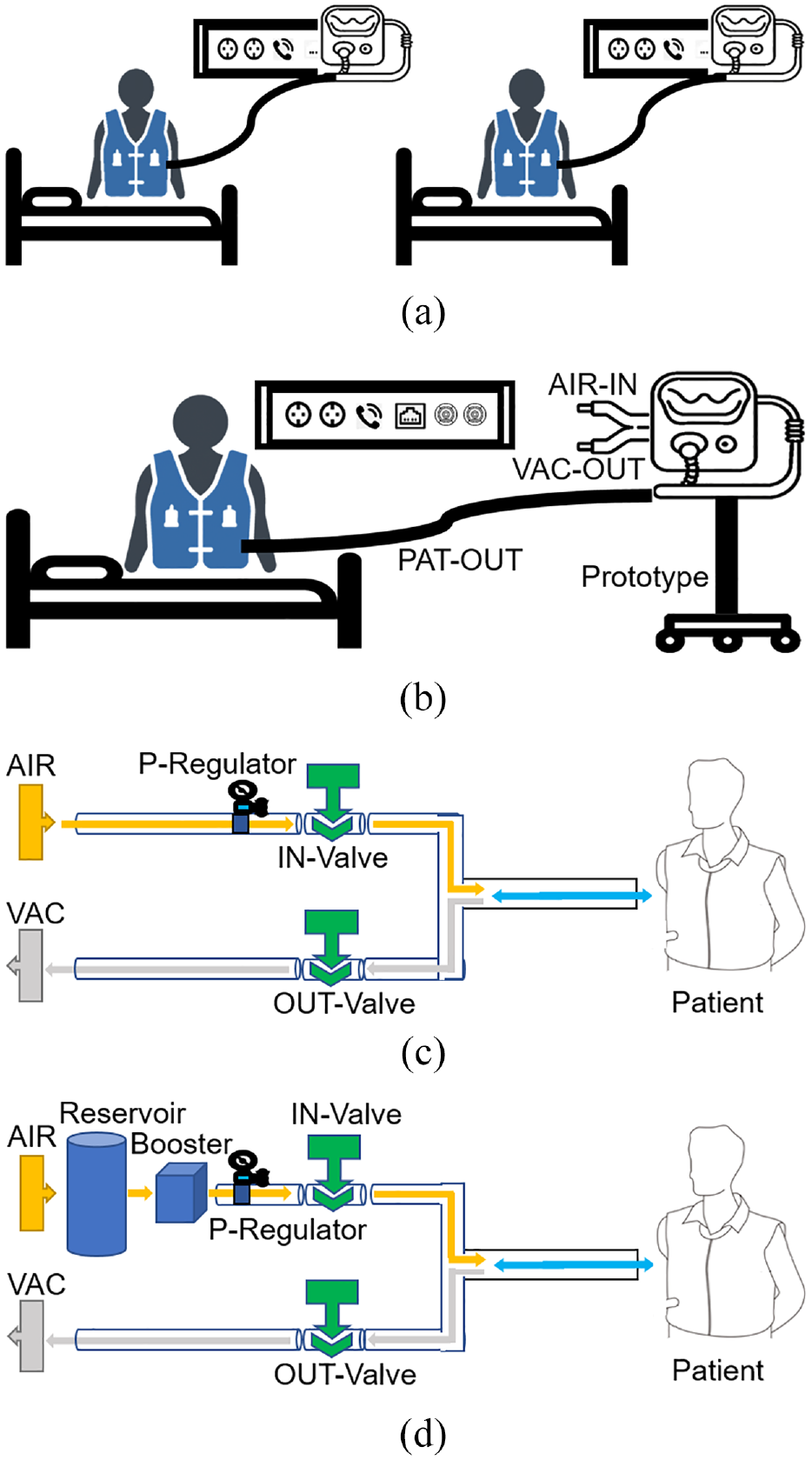
Schematic of the proposed wall port–based high-frequency chest wall oscillation (HFCWO) device for in-hospital use: (a) scheme of an ideal wall port–equipped HFCWO device, (b) scheme of a moving cart–type wall port–connected HFCWO device, (c) scheme of the no-amplification setting, (d) scheme of the pre-amplification setting.
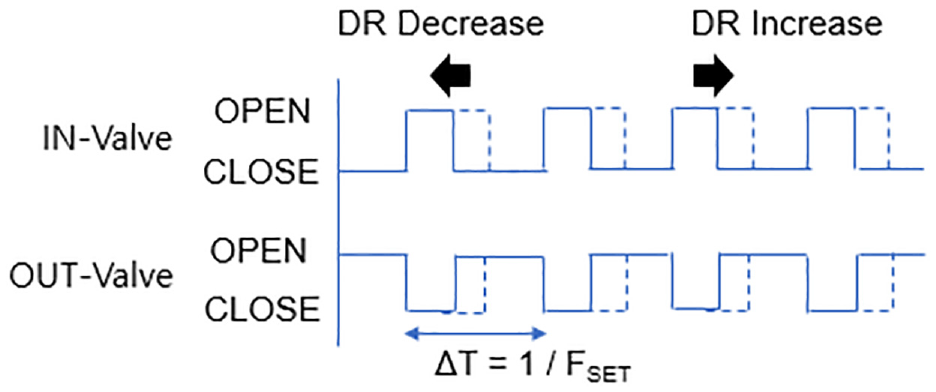
Control scheme of the IN- and OUT-Valves in the prototype device.
Prototype manufacturing
To verify the clinical usability of the proposed concept of a wall port–connected actuator-less airway clearance system as a tool for HFCWO treatment, we manufactured a prototype device in the form of a moving cart considering manufacturing and in vitro evaluation convenience. The prototype device with the NO-AMP setting contained IN- and OUT-Valves (KT37A-01-4G; KCC Co. Ltd., Seoul, Korea) and a P-Regulator (ITV2050-31F2N3; SMC Corp., Yorba Linda, CA, USA) to adjust the vibration frequency and pressure intensity level, respectively (Figure 3(a)). The prototype device with the PRE-AMP setting also contained an additional air reservoir (Medical Oxygen Tank; FP 15 MPa, PH 250 bar, TP 25 MPa) and an air-pressure booster (KPBV06-2A-03-GS; KCC Co., Ltd.) as well as an IN-Valve, OUT-Valve, and P-Regulator (Figure 3(b)).

Manufactured prototype of the moving cart–type wall port–connected actuator-less HFCWO device: (a) no-amplification setting and (b) pre-amplification setting.
A popular embedded board (Arduino Uno with MCP4725; Arduino Corp., Somerville, MA, USA) connected to a custom-made sensor/valve/switch interfacing board was used to control the manufactured prototype. The voltage range of control input of the P-Regulator was 0 V (minimal output pressure – 0% of AIR port pressure) to 10 V (maximum output pressure – 100% of AIR port pressure) in device specification; in contrast, the voltage range of analog-out from the controller (at 12-bit digital-to-analog conversion (DAC)) was 0 V (DAC register = 0) to 3.3 V (DAC register = 4095). Therefore, an additional voltage amplification circuit was inserted between the DAC-OUT port of the controller and the control input port of the P-Regulator (i.e., 0% of the AIR port pressure was discharged via the PAT-OUT tube when DAC-OUT = 0 V, and 100% of the AIR port pressure was discharged via the PAT-OUT tube when DAC-OUT = 3.3 V). The prototype has two operation modes: 1) EXPERT-MODE (for technical engineers), in which the operator can adjust the control input of the P-Regulator within 1–10 V (in 0.1-V increments), adjust the on/off frequency of the IN- and OUT-Valves within the range of 5–20 Hz (1-Hz increments), adjust the duty ratio of the IN- and OUT-Valves to 10–90% (0.01% increments), and adjust the device operating time within 1–30 min (1-min increments), respectively; and 2) USER-MODE (for clinicians), in which the operator can only select the vibration frequency and the pressure intensity level (similar to most air power–driven HFCWO devices on the market). When further fine-tuning of the control parameters is required during use, a trained examiner (from the company or the hospital’s engineering department) switches the device to the EXPERT-MODE, carefully adjusts the appropriate control parameters for each step of vibration frequency and pressure intensity level on the USER-MODE, switches the prototype back to the USER-MODE, and returns the device to clinicians for HFCWO treatment use. The clinician can then adjust the device’s vibration frequency and pressure intensity level as in most conventional HFCWO devices.
In vitro evaluation
To compare the performance of the manufactured prototype with a commercially available HFCWO device with an internal air-pulse generator, we investigated the list of HFCWO devices in our hospital; then, among the three candidates (VEST-105 (Hill-Rom), VEST-205 (Hill-Rom), and CareWay (Korust)), we chose a VEST-205 with maternity belt by considering the worldwide prevalence of each device and the number of each device in the hospital. The maternity belt was wrapped around the chest of the mannequin, and a pressure sensor (RA30P; Marbledex Co. Ltd., Seongnam, Korea; sensing range = 5 gf to 4 kgf; sampling resolution = 10 bits) was attached to the surface of the mannequin to measure the degree of vibration force delivered to the airway (Figure 4). The sensor attachment position was determined according to the guidance of a clinical expert at our hospital, while the pressure sensor was pre-calibrated using standard weights. First, the pressure levels of the AIR and VAC ports in the wall port utility of our hospital were measured using a commercial pressure sensor (C9557 Pressure Metter; Comark Ltd., Norwich, UK) at 41,860 mmH2O for the AIR port and −8240 mmH2O for the VAC port. Second, to acquire the reference pressure levels that were utilized during the fine-tuning process in EXPERT-MODE, VEST-205 was connected to the maternity belt via two PAT-OUT tubes, and the measurements of the mannequin-attached pressure sensor were recorded for 60 s with a sampling frequency of 100 Hz during repetitive operations under various operating conditions. In this study, we recorded data for the vibration frequencies of 8–12 Hz and pressure intensity levels of 2–4, which are the most frequently used in HFCWO treatment for adult patients, by referring to the advice of a clinical expert and the recommendation of a guideline document from a device manufacturer: (1) vibration frequency, 8–14 Hz according to patient tolerance/comfort; (2) pressure intensity level, adjusted at 1–6 levels according to patient tolerance/comfort; and (3) operation time, adjusted up to 20 min per treatment. 11 Third, the VEST-205 was disconnected from the air tubes, and the prototype with the NO-AMP setting was connected to the wall port utility and maternity belt. The device was then switched to EXPERT-MODE, and the values of the control parameters were carefully adjusted to identify the optimal parameter settings that represented the average values of the local maximum most similar to those from VEST-205 under the same conditions. Subsequently, the USER-MODE code for the NO-AMP setting was fine-tuned based on EXPERT-MODE measurements. For accurate device fine-tuning, we implemented additional code to enable a duty ratio calculation and control input measurement in the control program of the prototype. Fourth, the prototype with the PRE-AMP setting was connected to the wall port utility and maternity belt, the outlet pressure of the air-pressure booster was set to 90 psi (63,276 mmH2O), and the same processes (in step 3) were repeated to implement the USER-MODE code for the PRE-AMP setting. Fifth, the prototype was switched to USER-MODE and repeatedly operated at 8–12 Hz vibration frequencies and 2–4 pressure intensity levels. The measurements of the mannequin-attached pressure sensor were recorded for the NO-AMP and PRE-AMP settings and compared with those for the VEST-205.

Experimental setting for the prototype with no-amplification setting.
Results
Table 1 presents the operational specifications of the VEST-205 and the prototype manufactured by USER-MODE. The prototype could stably control and maintain a vibration frequency of up to 14 Hz and a pressure intensity level of up to 6. When the parameters exceeded this limitation, the intensity of the pneumatic power delivered to the mannequin-attached pressure sensor slowly decreased with time during continuous operation, which may have been due to the positive/negative pressure levels of the wall port utility in our hospital and the narrow internal diameter of the utilized solenoid valves (i.e., IN- and OUT-Valves).
Operational specifications of the VEST-205 and the manufactured prototype in USER-MODE (common for no-amplification and pre-amplification settings).
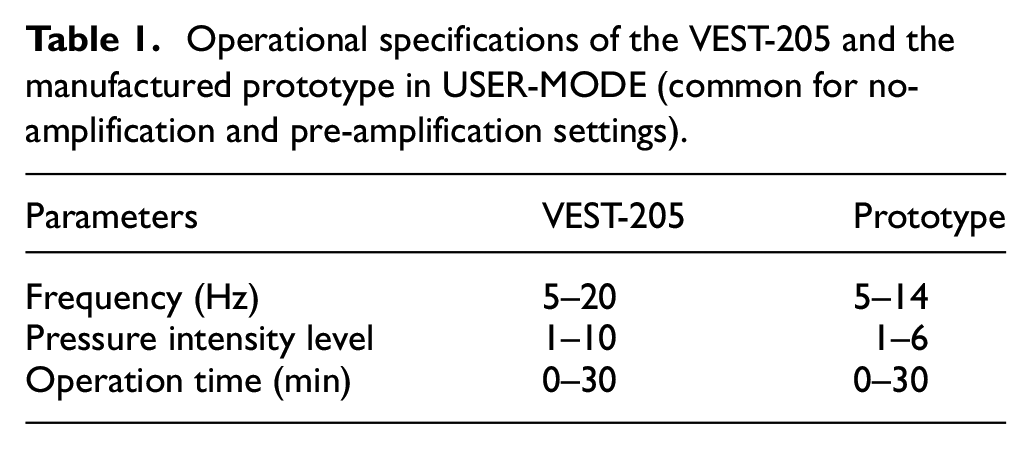
Table 2 presents measurements taken by the mannequin-attached pressure sensor (i.e., local maximum and local minimum) when the mean local maximum measurements of the two devices were most similar to each other during the parameter fine-tuning process in EXPERT-MODE at vibration frequencies of 8–12 Hz and pressure intensity levels of 2–4 (VEST-205 versus the prototype with NO-AMP setting in USER-MODE). Table 3 shows the same measurements for the VEST-205 versus the prototype with the PRE-AMP setting in USER-MODE. Under each control condition, sensor measurements were recorded for approximately 1 min, and then the values of 400 local maximum and 400 local minimum for each recording were used to calculate the mean and standard deviation. In both amplification settings, the duty ratio of the valve control signal increased as the vibration frequency increased. The values of the voltage level of DAC-OUT port (i.e., the control input of the P-Regulator) for the NO-AMP setting were generally 0.5–0.7 V higher than those at PRE-AMP setting, implying that the internal parameter settings of the proposed wall port–connected HFCWO device require fine-tuning at the installation (purchase) stage to fit the various pressure levels of the AIR and VAC ports of individual hospitals. The range of variations in local maximum pressure (on average) of the prototype device was 13.21–17.57 mmH2O for the NO-AMP setting and 12.94–18.12 mmH2O for the PRE-AMP setting. Moreover, the range of variations in mean local minimum pressure of the prototype device was 8.52–15.16 mmH2O for the NO-AMP setting and 9.69–14.56 mmH2O for the PRE-AMP setting. Compared to the measurements from the VEST-205 (average local maximum, 10.59–18.03 mmH2O; average local minimum, 0.28–1.78 mmH2O), the prototype device showed substantially higher local minimum values than the VEST-205 for both amplification settings.
Local maximum/minimum values (mean ± standard deviation; in mmH2O) measured by the mannequin-attached pressure sensor when the mean local maximum of the two devices were the most similar.
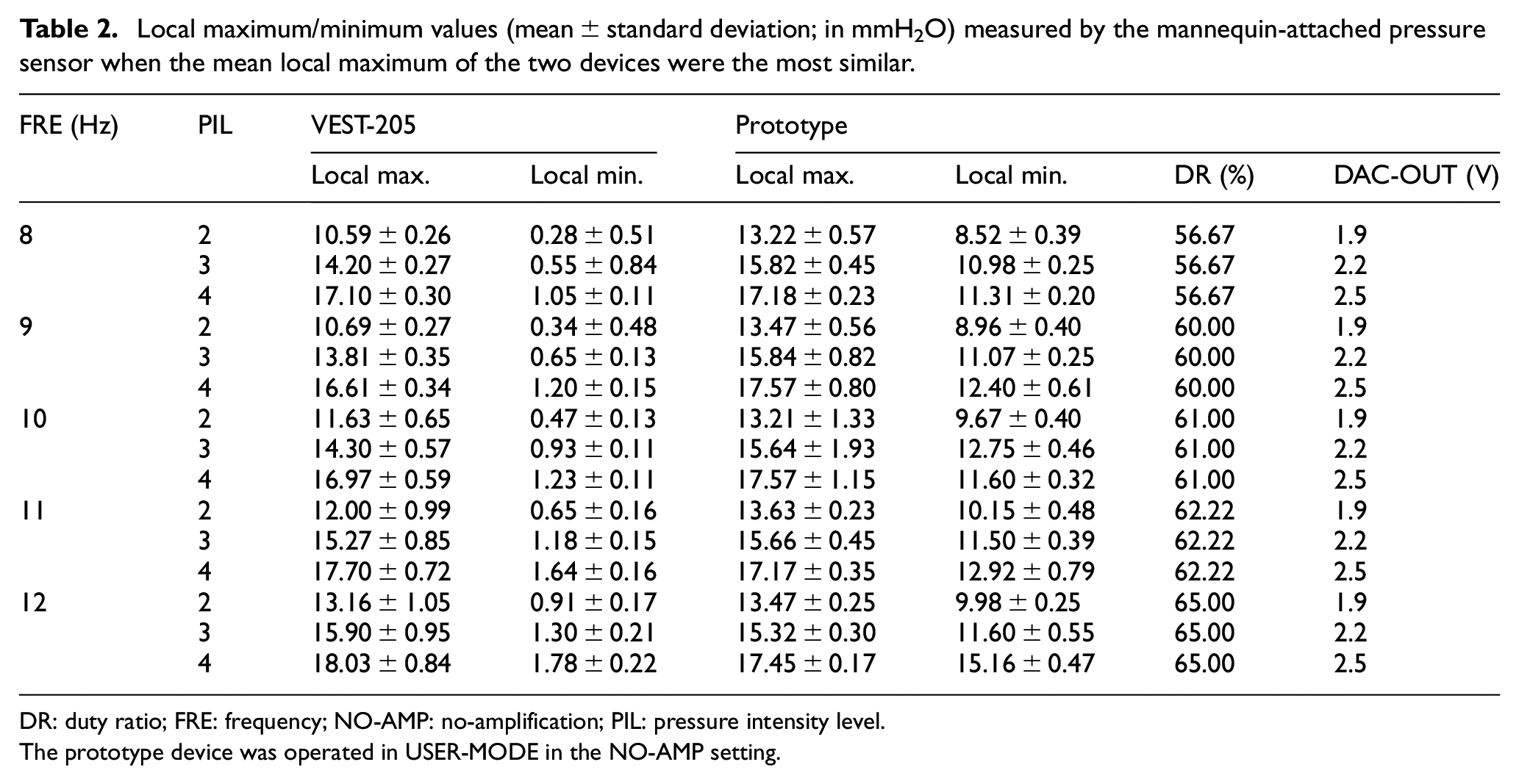
DR: duty ratio; FRE: frequency; NO-AMP: no-amplification; PIL: pressure intensity level.
The prototype device was operated in USER-MODE in the NO-AMP setting.
Local maximum/minimum values (mean ± standard deviation; in mmH2O) measured by the mannequin-attached pressure sensor when the mean local maximum values of the two devices were the most similar.
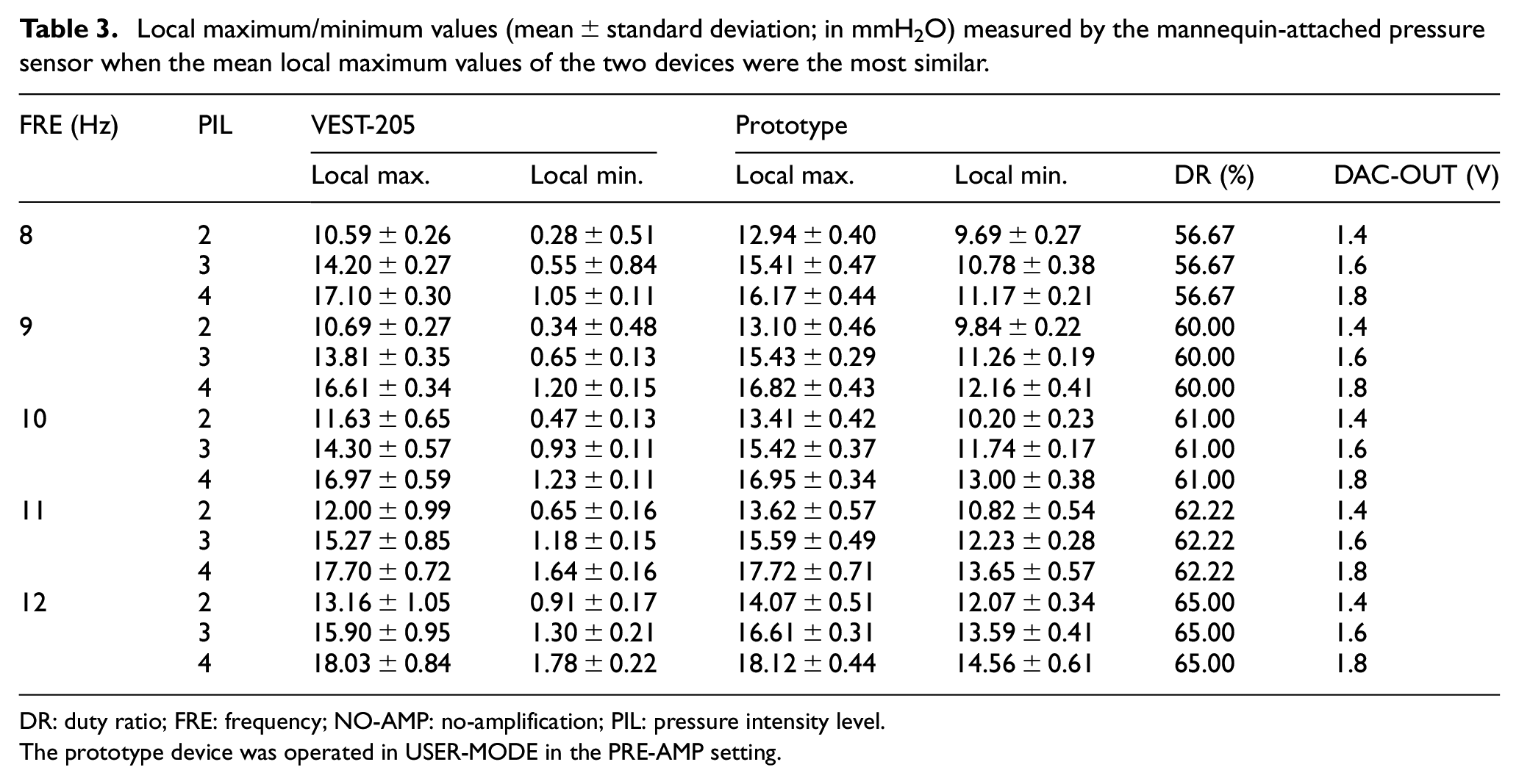
DR: duty ratio; FRE: frequency; NO-AMP: no-amplification; PIL: pressure intensity level.
The prototype device was operated in USER-MODE in the PRE-AMP setting.
Table 4 presents the differences in the mean local maximum and minimum pressure values of the VEST-205 and prototype device (USER-MODE) at vibration frequencies of 8–12 Hz and pressure intensity levels of 2–4 (Tables 2 and 3). Under all operating conditions, the pressure differences at the local maximum between the two devices did not exceed 3 mmH2O; more specifically, the absolute pressure difference at the local maximum was minimal (0.08 mmH2O) at frequency/intensity = 8/4 and maximal (2.78 mmH2O) at frequency/intensity = 9/2 in the NO-AMP setting; in contrast, the absolute pressure difference at the local maximum was minimal (0.02 mmH2O) at frequency/intensity = 10/4 and 11/4 and maximum (2.41 mmH2O) at frequency/intensity = 9/2 in the PRE-AMP setting, respectively. However, the local minimum values of the prototype device were always significantly higher (8.24–13.38 mmH2O in the NO-AMP setting, 9.41–12.78 mmH2O in the PRE-AMP setting) than those of the VEST-205.
Mean pressure differences in local maximum and local minimum (in mmH2O) values for the VEST-205 versus prototype device in USER-MODE.
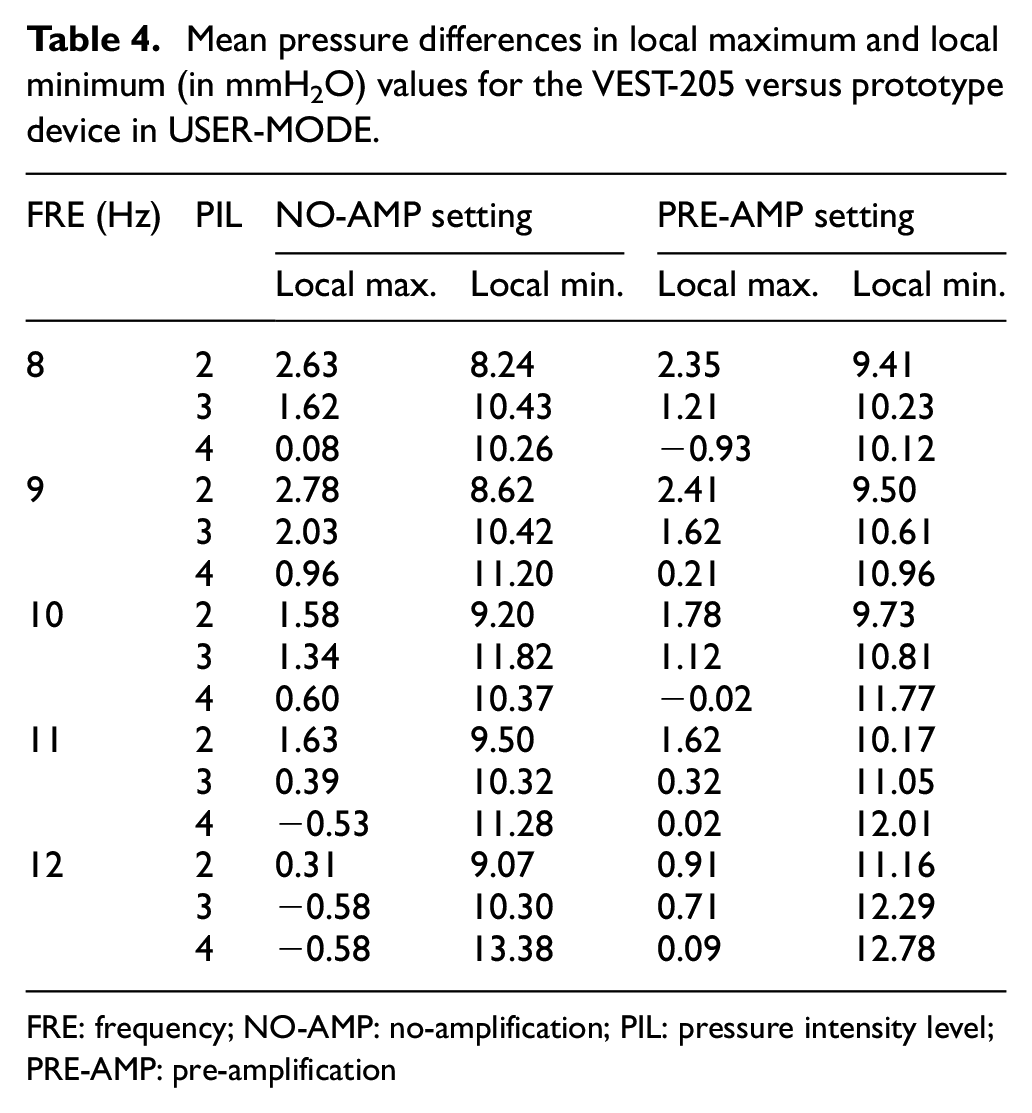
FRE: frequency; NO-AMP: no-amplification; PIL: pressure intensity level; PRE-AMP: pre-amplification
Table 5 presents the results of the non-parametric Wilcoxon signed-rank test for local maximum/minimum values between the [VEST-205 versus prototype with NO-AMP setting], [VEST-205 versus prototype with PRE-AMP setting], and [prototype with NO-AMP setting versus prototype with PRE-AMP setting] conditions in USER-MODE because the sensor measurements (400 local maximum and 400 local minimum for each frequency/intensity condition) did not follow a normal distribution based on the Kolmogorov-Smirnov test. For the local maximum value, the VEST-205 and the prototype device showed statistically significant differences (p < 0.05) in all conditions except for frequency/intensity of 11/4 (p = 0.664) and 12/4 (p = 0.253). In addition, there were statistically significant differences between the NO-AMP and PRE-AMP settings of the prototype device under all conditions except for frequency/intensity of 9/2 (p = 0.850), 9/3 (p = 0.845), 11/4 (p = 0.804), and 11/3 (p = 0.163). For the local minimum value, there were statistically significant differences between the two devices and between the NO-AMP and PRE-AMP settings of the prototype device under all conditions (p < 0.05).
Results of non-parametric Wilcoxon signed-rank test (p-values) for local maximum/minimum values in USER-MODE.
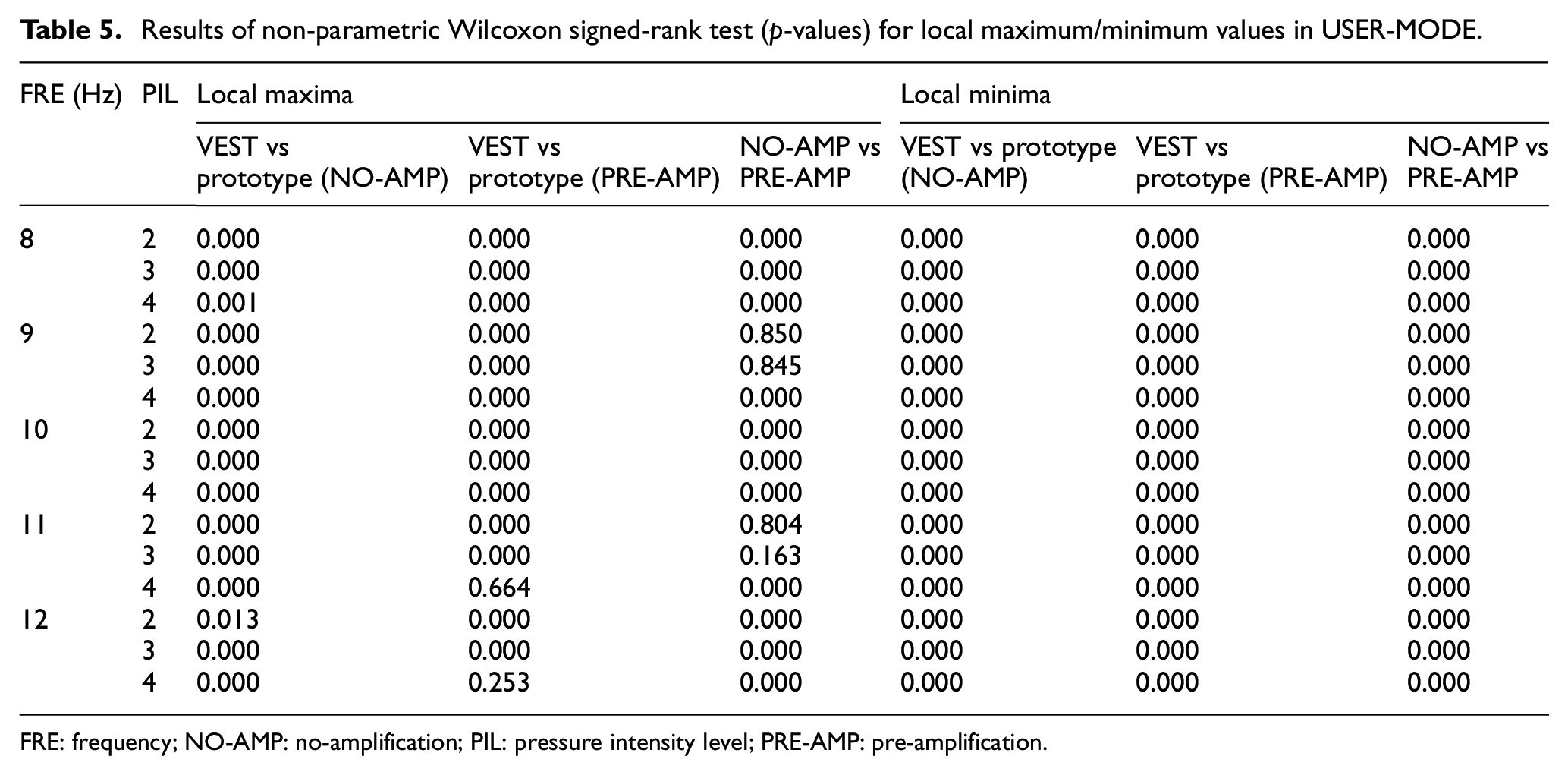
FRE: frequency; NO-AMP: no-amplification; PIL: pressure intensity level; PRE-AMP: pre-amplification.
Figure 5 shows the measurements of the operation sounds of the two devices (VEST-205 and the prototype in the NO-AMP setting) using a commercial sound level meter (TES-1357; Mire-SC Co. Ltd., Gunpo, Korea; frequency weighting = mode A) during operation (frequency range, 8–12 Hz; pressure intensity levels, 2 and 4). During the measurements, a sound level meter was placed in front of the HFCWO device at a distance of 15 cm. Measured sound levels of the VEST-205 were in the range of 54.1–56.2 and 55.9–57.5 dB at pressure intensity levels 2 and 4, respectively; in contrast, those from the prototype device were in the range of 72.1–73.6 and 72.9–75.1 dB at pressure intensity levels 2 and 4, respectively.
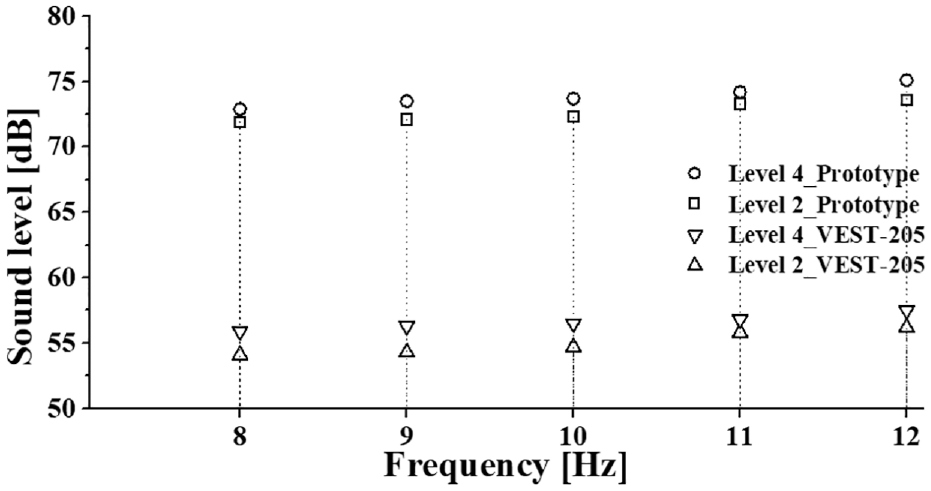
Measured operating sound levels for the VEST-205 versus prototype device win the no-amplification setting.
Discussion
With increasing environmental problems (e.g., air pollution) and the emergence of highly infectious diseases (e.g., COVID-19), the clinical prevalence of respiratory diseases and frequency of HFCWO treatments is consistently increasing.12,13 According to a recent market surveillance report, the estimated global market size for airway clearance devices was USD 579 million in 2022 and is projected to exceed USD 946 million by 2032 (compound annual growth rate = 4.9%). 14 In addition, subjects with chronic respiratory diseases, such as chronic obstructive pulmonary disease and asthma, are particularly vulnerable to environmental pollution. 15 Furthermore, several studies have reported that the risk of cross-infection in hospitals is a serious public health concern, especially during pandemics; thus, appropriate infection control is very important for hospitals.16,17 However, most hospitals cannot procure sufficient numbers of HFCWO devices owing to financial constraints; therefore, time scheduling of available devices is used to cater to the needs of all patients, but this approach is associated with long waiting times and the potential risk of cross-infection between patients with highly infectious respiratory diseases. Hence, there is a demand for more cost-effective HFCWO devices that can be exclusively utilized by individual patients during hospitalization. For example, according to a report by Ansaripour et al., 18 the cost of HFCWO treatment is more than twice that of conventional chest wall physical therapy, implying the need to reduce the end-user price of HFCWO devices. Efficient financial expenses, expansion of treatment capability, and reduction in the risk of in-hospital cross-infection are all important factors for most hospitals; therefore, the need to develop low-cost HFCWO devices is high. Here we manufactured a prototype HFCWO device that utilizes an external wall port utility in a hospital to generate the pneumatic power needed for HFCWO treatment instead of utilizing an internal air-pulse generator. In vitro comparative experiments of the VEST-205, one of the most widely used HFCWO devices in the clinical setting, demonstrated that the prototype device could generate almost equivalent chest wall vibration power (i.e., average local maximum) to that of the VEST-205 under the most popular operating conditions (i.e., 8–12 Hz vibration frequency, 2–4 pressure intensity level), implying that the proposed device can substitute for commercial HFCWO devices with internal air-pulse generators in limited (but frequently used) operating settings in hospitals. Although the prototype device could not meet the overall operational specifications of commercial devices (Table 1), it showed sufficient pneumatic power in popular frequency/intensity settings for most critical inpatients, the primary focus of the current study.
In addition to conventional air power–driven HFCWO devices, which contain a mechanical blower that can independently generate pneumatic power for HFCWO treatment in any location (i.e., hospital, home, or office), there is another type of small and light HFCWO device called the AffloVest. 19 Such motor-driven, jacket-assembled HFCWO devices can eliminate the need for a mechanical air-pulse generator by implementing its function using multiple electric motors that can improve patient mobility during HFCWO treatment, making them more suitable for out-of-hospital use than conventional HFCWO devices with mechanical air-pulse generators. However, in hospitals, improving patient mobility during HFCWO treatment is not an important concern because most critical patients cannot move freely and lie in bed almost fulltime. To enable hospitals to improve their reaction capability for the spread of environmental and medical crisis that require more frequent HFCWO treatments, increasing the number of catered HFCWO devices despite a limited budget is most important. The proposed actuator-less HFCWO device contains neither a mechanical air-pulse generator nor electric motors; rather, it utilizes wall ports near the patient bed that are already installed in most hospitals; thus, they require no additional budget for setup. Therefore, it is possible to substantially reduce the device’s manufacturing cost. As a result, the end-user price of the device can be decreased to a reasonable level, which can reduce the financial burden on hospitals to enhance their reaction capabilities.
During document surveillance, we found a US patent that commented on a wall port–based HFCWO mechanism similar to the currently proposed device (NO-AMP setting). 20 However, that device adopted a passive air ejection mechanism similar to that of the VEST-205, whereas the proposed device adopts an active air ejection mechanism using a wall VAC port. This difference in the air ejection mechanism did not significantly affect the HFCWO treatment; however, the structure of the proposed device (i.e., using AIR and VAC ports simultaneously) can also be applied to more extensive respiratory treatments that require positive and negative pressures. For example, the proposed device can be modified into a cough-assisted device that alternately uses positive expiratory pressure and negative inspiratory pressure. 21 In future studies, we plan to expand the current study to develop an additional wall port–based hybrid treatment device that can be used for both HFCWO and cough-assisted treatments.
Several modifications must be made to the current prototype model to improve its clinical usability. First, as shown in Tables 2 and 3, the internal parameter settings of the proposed device (i.e., duty ratio of the valve control signal and control input of the electric pressure regulator) require fine-tuning before initial use to fit the wall port pressures (AIR and VAC) of individual hospitals since they can vary among facilities. However, this initial fine-tuning process may be difficult for clinical staff; therefore, it is necessary to automate the initial fine-tuning processes using additional sensors and auto-fitting software so that the proposed device operates constantly regardless of an individual hospital’s pressure settings. Second, as shown in Figure 5, the driving sound of the prototype device is 17.0–17.8 dB higher than that of the VEST-205. Because HFCWO treatment generally continues for 10–20 min, it is necessary to reduce the driving sound of the proposed device to minimize patient inconvenience and even the potential risk of hearing damage. To achieve this, the overall structure of the prototype device should be further optimized to reduce superfluous noise during operation. For example, we assume that the primary cause of the high operating sound of the current prototype is the small internal diameter of the commercial solenoid valves (IN- and OUT-Valves) used in this study. To further reduce the operating sound of the prototype device, it will be necessary to design and manufacture more optimized valve structures in future studies (e.g., by enlarging the internal diameter and minimizing the overall size). Third, in the comparative study, we utilized a maternity belt that is generally used with the VEST device series in hospitals that contains two orifices for simultaneously connecting the two PAT-OUT tubes. During the VEST’s operation, two identical PAT-OUT tubes were connected to the maternity belt via these orifices, whereas during the prototype’s operation, only one PAT-OUT tube was connected to the maternity belt and the other orifice was sealed to minimize unwanted passive air discharge during the device’s operation. The structure, size, and tube interface of the dedicated maternity belt (or jacket) must be modified to improve the clinical usability of the prototype device. Fourth, in this study, we manufactured a moving cart–type device as the first prototype (Figure 1(b)). Further studies will be necessary to implement a smaller and lighter prototype of the actuator-less HFCWO device that can be directly attached to a wall port without bulky AIR-IN and VAC-OUT tubes (Figure 1(a)).
To validate the clinical effectiveness of the proposed actuator-less HFCWO device, it will be necessary to further evaluate the clinical effects of the proposed device in terms of subjective patient feelings during long-term repetitive HFCWO treatments. For example, as shown in Table 4, the local maximum pressures of the prototype device in USER-MODE can be tuned to levels similar to those of the VEST-205 (mean absolute pressure difference, <3 mmH2O) by adjusting the control parameters in EXPERT-MODE. However, the local minimum pressures of the prototype device were significantly higher than those of the VEST-205 in both pre-amplification settings (mean absolute pressure difference, >8 mmH2O). Because the local maximum pressure (i.e., expansion of vacant space in maternity belt) induces a short burst of expiratory air flow and the local minimum pressure (i.e., contraction of vacant space in maternity belt) induces air flow in the inspiratory direction, such relatively high local minimum pressures of the prototype device may affect the efficiency of viscosity reduction and discharge acceleration of secretions in the airway during HFCWO treatment. In addition, relatively high local minimum pressures can affect patients’ subjective feelings (e.g., constriction or stiffening) during treatment. To identify the clinical effects of relatively high local minimum pressures of the prototype device on the efficiency of viscosity reduction and discharge acceleration of secretions in the airway and the patients’ subjective feelings during HFCWO treatment, future well-designed ex vivo, in vivo, and clinical experiments are required. In addition, the operating sound of the prototype device was 17.0–17.8 dB higher than that of the VEST-205 (Figure 5). To evaluate the clinical effects of the relatively high operating sound of the prototype device on patient discomfort during HFCWO treatment, additional questionnaire surveys targeting patients must be administered after the treatments. Furthermore, in the current study, we set the time delay between the closing of the IN-Valve and the opening of the OUT-Valve to 0 (Figure 2). However, when a few people in our team wore the maternity belt and experienced chest wall vibrations using the prototype device for several seconds, all commented that the feeling of chest wall vibration was somewhat different as the time delay varied between the two valves; more specifically, the feeling of chest compression was slightly increased while the feeling of chest vibration was slightly decreased as the time delay between two valves was not 0. We did not verify the concrete effect of the time delay between the two valves on the power of chest wall vibration or the performance of airway clearance; these factors require evaluation in future studies.
This study had some limitations. First, we manufactured a prototype device in the form of a moving cart considering the convenience of implementation and in vitro evaluations. However, to improve clinical usability, the prototype should be more diversified; for example, a wall port–equipped form (Figure 1(a)) or a bed-equipped form. 20 In future studies, more diverse prototypes should be manufactured and their performances evaluated. Second, as shown in Figure 5, the operating sound of the prototype device was higher than that of the VEST-205 owing to structural limitations. To reduce the operating sound of the prototype device, it will be necessary to improve the structure of the internal air valves to reduce the flow resistance by increasing the diameter of the overall air pathway through the device.
Conclusions
In this study, we proposed a novel wall port–based actuator-less HFCWO device and evaluated its performance through in vitro comparative experiments using a commercial HFCWO device with an internal air-pulse generator. Based on the experimental results, we conclude that the proposed device has the potential to replace the VEST-205 in limited but frequently used clinical settings, although several modifications and upgrades are necessary to improve its clinical usability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Research Foundation of Korea (NRF) Grant funded by the Korean Government (MSIT) (No. NRF-2022R1A2C1008862). This study was also supported by a 2022 research grant from Pusan National University Yangsan Hospital.
