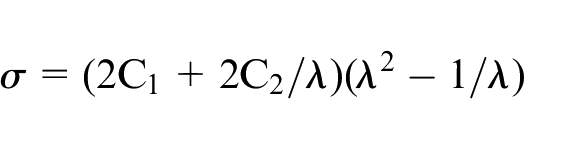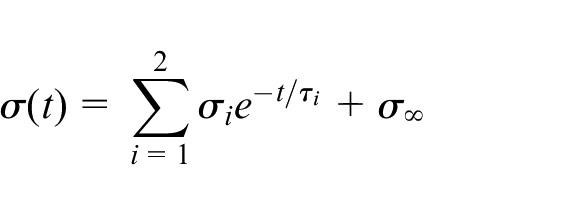Abstract
Articular cartilage (AC) injuries do not heal primarily and large lesions progress to degenerative osteoarthritis. Osteochondral allograft transplantation is an effective surgical treatment but is limited by the lack of donor tissue availability. Fresh allografts can be stored hypothermically up to 28–45 days after which the tissue is no longer viable for transplantation. Vitrification is a method of cryopreservation with the potential to extend the storage time of AC. A specific protocol has been demonstrated to preserve high chondrocyte viability; however, its effect on various mechanical properties of the extracellular matrix (ECM) remains unknown and is the focus of this initial study. Porcine AC was subject to a defined vitrification protocol, using fresh and frozen samples as positive and negative controls, respectively; n = 20 for all three groups. Unconfined compression testing was used to assess mechanical properties of the tissue under rapid load, stress relaxation, and equilibrium conditions. The stress relaxation time constants (modeled with a 2-term Prony series)
Introduction
Articular cartilage (AC) lesions in the knee are common, particularly in young and active patients. Due to its avascular nature, AC injuries do not heal independently and often progress to osteoarthritis. 1 Osteochondral (bone and AC) transplantation has become an established surgical treatment for focal AC defects that restores function and can prevent/delay the need for arthrodesis/arthroplasty. Allografting of fresh osteochondral tissue to treat focal femoral condyle lesions has documented long-term success rates between 70% and 90% at 12 years. 2 The success of this surgery has made it increasingly popular but availability and cost of donor tissue remain the most significant limitations. 3
Currently, allografts are harvested within 24 h of donor death and must undergo a regulatory screening process for contamination and disease which requires approximately 14 days.4,5 Fresh grafts are stored at 4°C while matching for size, contour, and exact location with potential recipients. After 28–45 days of storage, they must be discarded.6,7 Fresh grafts are preferred over frozen grafts (stored at −80°C) because they have higher chondrocyte viability, 7 improved cartilage stiffness, increased matrix content and decreased surface degeneration 6 months after implantation, 8 but are limited by finite storage time.
Vitrification, a form of cryopreservation that uses a high concentration of cryoprotective agents (CPAs) to prevent the formation of ice crystals, 9 has been proposed as a means of prolonging allograft storage time without excessively sacrificing chondrocyte viability. 10 The ability to store AC indefinitely would increase the availability of donor tissue, but the effect of vitrification on tissue mechanical properties has yet to be documented.
Graft survival depends on both chondrocyte and ECM integrity since the cellular component is required for long-term ECM function. 11 Therefore, cryopreservation must attempt to preserve both. Freezing protocols have been shown to cause damage to both cellular and extracellular components, possibly due to ice crystal formation during the freeze-thaw process.12–14 Freezing also affects various mechanical properties of AC, including decreasing aggregate modulus and half-life of stress-relaxation 15 ; as well as decreasing stiffness and peak stress. 16 Vitrification can maintain cell viability, 10 so the objective of this preliminary study was to compare the mechanical properties of fresh, frozen, and vitrified porcine AC, and to guide further investigation. Because vitrification eliminates the formation of ice crystals, we hypothesized that vitrified AC will retain similar mechanical properties to fresh AC and superior mechanical properties compared to frozen AC. This outcome would support the use of vitrification for storing osteochondral tissue samples before transplantation.
Methods
Specimen harvest and preparation
Twenty femoral condyles were harvested from sexually mature porcine hind stifle joints obtained from pigs used for meat consumption from a local deli within 24 h of death. No animals were specially sacrificed for this project. The use of animal tissue for research was approved by the Research Ethics Office at the University of Alberta. These joints were submerged in phosphate-buffered saline (PBS). Three 10 mm diameter osteochondral dowels (OCDs) were cored from each condyle using a handheld coring device. All three OCDs from a single condyle were transferred to the same beaker and cleaned with sterile PBS supplemented with antibiotics (100 units/mL penicillin, 100 µg/mL streptomycin, 0.25 µg/mL amphotericin B) under a sterile biosafety cabinet for 20 min. The OCDs were then placed in Falcon tubes containing 25 mL of sterile Dulbecco’s Modified Eagle Media (DMEM) complete medium [(DMEM complete: Dulbecco’s Modified Eagle Medium F12 (Gibco) supplemented with 10% Newborn Calf Serum (Gibco), 100 units/mL penicillin, 100 µg/mL streptomycin, 0.25 µg/mL amphotericin B (Gibco), and 1 mM sodium pyruvate (Gibco)] and stored at 4°C overnight. The three OCDs from each condyle were randomly assigned to one of three experimental groups (fresh, frozen, and vitrified) using a paired design (n = 20 per group).
Fresh
OCDs in the Fresh Group were kept in DMEM complete at 4°C for approximately 40 h until mechanical testing.
Frozen
OCDs in the Frozen Group were kept in DMEM complete at 4°C overnight, then transferred to PBS and subjected to four freeze-thaw cycles by plunging into liquid nitrogen (LN2) at −195.79°C for 15 min and warming to 37°C using a water bath.
Vitrified
OCDs in the Vitrified Group were kept in DMEM complete at 4°C overnight, then underwent vitrification the following day. The 7-h vitrification process, briefly summarized here, involved the use of several CPAs at increasing concentrations and progressively lower temperatures, followed by rapidly plunging in LN2 and held overnight. 17 All solutions were made in DMEM.
Solution 1: 3 M dimethyl sulfoxide (DMSO) + 3M ethylene glycerol (EG) at 0°C for 90 min
Solution 2: 3 M DMSO + 3M EG + 3M propylene glycerol (PG) at −5°C for 170 min
Solution 3: 3 M DMSO + 3M EG + 2M PG at −10°C for 160 min
After warming to 37°C using a water bath, samples were washed with agitation on an orbital shaker at 180 rpm for 30 min in 25 mL of DMEM at 4°C.
Mechanical testing
Prior to mechanical testing, all OCDs were warmed to room temperature and rehydrated in PBS for 1.5 h. Cartilage was removed from the bony base of the dowel using a surgical blade. Cartilage thickness was measured using a digital caliper at three locations per sample to determine an average initial thickness. When possible, samples were re-punched using the 10 mm diameter core to ensure cross-sectional consistency and to eliminate the effect of the initial coring angle on the samples’ shapes. The cross-sectional area was calculated by taking a photograph of each OCD and analyzing it in a custom MATLAB image processing algorithm to ensure consistency across samples and to determine an accurate cross-sectional area for use in calculations. The software was calibrated using a reference ruler (see Figure 1). Samples were then placed on a non-porous steel disc, within a room temperature PBS bath, for uniaxial unconfined compression testing using the Bose ElectroForce 3200. They were compressed from the top using a non-porous steel disc. The following loading protocol was used: (1) 1 N pre-load held for 120 s, (2) rapid loading at a strain rate of 15%/s to 15% strain, (3) displacement held for at least 1800 s to allow stress-relaxation to an equilibrium state.

(a) Example image of an OCD with a reference ruler used for calibration and (b) an image processing MATLAB algorithm using the reference ruler, and the tracked edge of the OCD (blue) to calculate the cross-sectional area.
Data analysis
AC was assumed to act as an incompressible material under the applied rapid loading rate. 18 The stress-strain behavior in the rapid loading phase was modeled using a Mooney-Rivlin material model, where constants C1 and C2 in the following equation were determined:
Where
Finally, Young’s modulus of the solid matrix was determined from the equilibrium stress values of the curve-fitted 2-term Prony series model (
Results
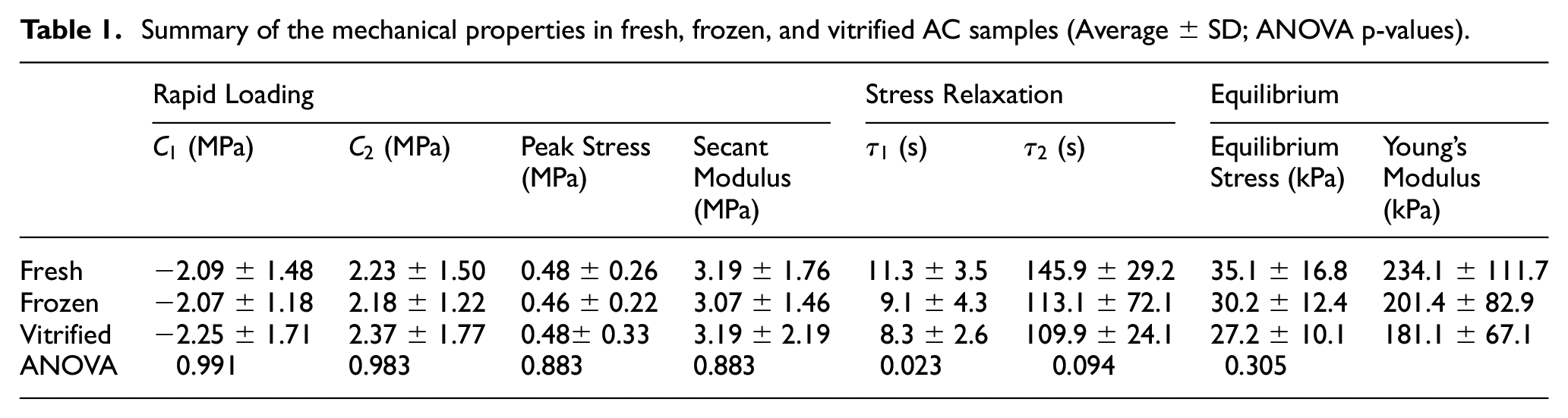
Table 1 shows a summary of the mechanical properties for the fresh, frozen, and vitrified groups reported as average ± standard deviation values. Additionally, the p-values for the repeated measures ANOVAs are shown in Table 1. Three samples (one from each group) were excluded from the analysis because the cartilage was damaged during the coring and bone removal process. Further, one additional frozen sample was excluded from the stress-relaxation and equilibrium analysis because of technical issues with the Bose ElectroForce 3200 during testing.
Summary of the mechanical properties in fresh, frozen, and vitrified AC samples (Average ± SD; ANOVA p-values).
Rapid loading
Using the Mooney-Rivlin model for rapid loading, the average C1 values were −2.09, −2.07, and −2.25 MPa for fresh, frozen, and vitrified respectively (Table 1). The average C2 values were 2.23, 2.18, and 2.37 MPa for fresh, frozen, and vitrified respectively (Table 1). For both parameters, there were no significant differences between any of the three test groups, as C1 values had an ANOVA p-value of 0.991, while C2 had an ANOVA p-value of 0.983. It should be noted that the Mooney-Rivlin model provided an excellent representation of the experimental data with R2 values above 0.99 for all samples.
Peak stress and secant modulus
The average measured peak stress values were found to be 0.48, 0.46, and 0.48 MPa for fresh, frozen, and vitrified samples respectively (Table 1). The corresponding secant moduli were identified as 3.19, 3.07, and 3.19 MPa for fresh, frozen, and vitrified samples respectively. Again, there were no significant differences between any of the three test groups for these parameters (p = 0.883).
Stress-relaxation
Using the 2-term Prony series to model stress-relaxation, average
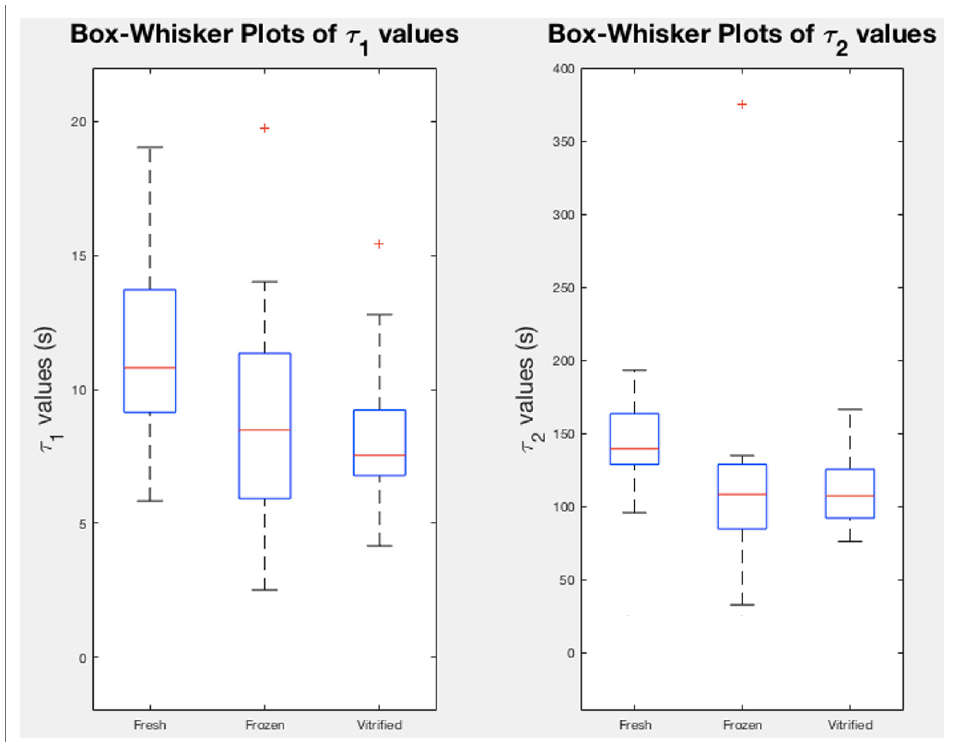
Box-whisker plots of
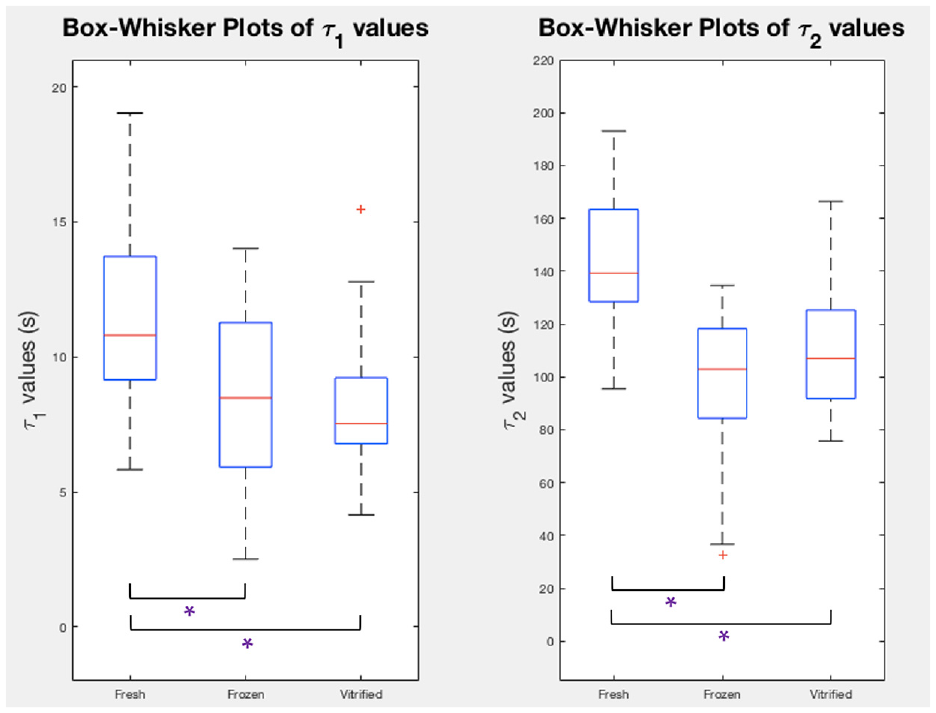
Box-whisker plots of
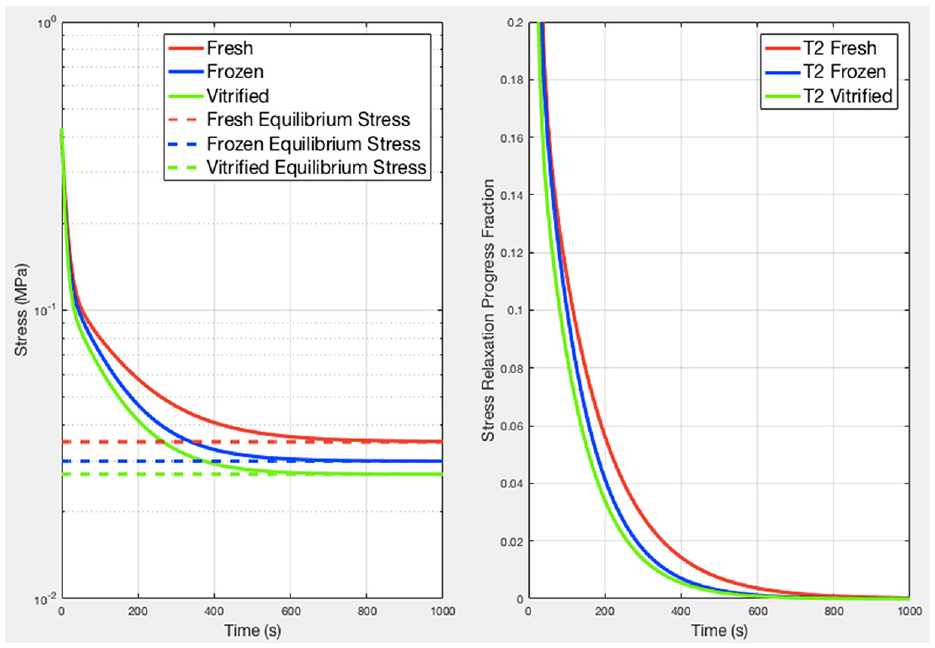
Left: general relaxation trends for each group of samples. Frozen and vitrified curves tend toward their equilibrium values quicker than the fresh curve, indicating a longer relaxation time for fresh samples. Right: progression of the average relaxation curves from peak stress (normalized to 1) to equilibrium stress, showing the percentage of stress from peak to relaxation.
Equilibrium stress conditions
The average equilibrium stress values were 35.1, 30.2, and 27.2 kPa for fresh, frozen, and vitrified samples (Table 1). Young’s modulus at equilibrium was determined to be 234.1, 201.4, and 181.1 kPa for fresh, frozen, and vitrified samples. There were no significant differences between any of the three test groups for these parameters (p = 0.305).
Discussion
Osteochondral implantation using allograft tissue is an effective surgical treatment for joint defects, but its practical usage is restricted by limited storage options. Successful cryopreservation of AC would enable long-term storage which would alleviate this limitation. Chondrocyte viability and ECM integrity must both be maintained for successful transplantation.
Various attempts at cryopreservation have been shown to detrimentally affect chondrocyte viability,7,24 reducing or altering the capacity of the chondrocytes to maintain ECM components and leading to extensive graft tissue necrosis and fibrosis. Furthermore, changes in the ECM, such as those that occur with freezing, have been shown to disrupt the mechanical properties of AC.15,16 Ice crystal formation during the cryopreservation process has been proposed as a possible mechanism resulting in chondrocyte rupture and mechanical disruption of the extracellular collagen matrix and proteoglycan network, ultimately leading to graft failure.11,25 Physical damage resultant from ice crystal formation may include the breakdown of aggregating proteoglycans 26 and an increase in open areas in the matrix. 27 Four freeze-thaw cycles were employed to ensure all the chondrocytes within an intact cartilage sample were killed, which is based on previous work in our lab where at least three cycles in LN2 consistently kills all of the cells.
Vitrification transitions a water-based solution into a glass-like solid without ice formation. Therefore, we hypothesized that it may preserve mechanical properties of the tissue. The vitrification protocol used employs stepwise cooling and multi-CPA solutions, which has been shown to mitigate CPA toxicity and maintain chondrocyte viability. 10 However, it has not yet been shown in the literature what effect any vitrification protocol may have on AC mechanical properties.
Unconfined compression testing, as is commonly used in the literature to assess cartilage mechanics,22,28–30 was used to assess the effects of freezing and vitrification on the mechanical properties of porcine AC. No significant differences in mechanical parameters (secant modulus and Mooney-Rivlin material constants) between fresh, frozen, and vitrified samples were documented during the rapid loading phase. This result is partly consistent with the work of other research groups. Groups using indentation techniques to assess the unrelaxed (analogous to rapid loading phase of our study) and relaxed (analogous to equilibrium loading phase of our study) properties of cartilage have not found differences in either property between frozen or cryopreserved tissue and fresh tissue. 31 Unconfined compression testing of bovine femoral condyle cartilage disks similarly produced no detectable changes in biomechanical properties after a single freeze-thaw cycle to −20°C. 30 Black et al. also using indentation techniques, determined that the unrelaxed shear modulus of lapine femoral cartilage subjected to freeze-thaw was within the range of normal (i.e. fresh) and did not increase, unlike cartilage stored for increasing lengths of time. 32
In this study, the only significant difference was in stress-relaxation phase data between the three groups. A significant difference in the
The altered time constants are evidence of increased permeability of the ECM. In the case of frozen samples this may be a result of ice formation disrupting or damaging the solid phase matrix such that porosity is increased and fluid flow is facilitated, as previously proposed by others. 15 While some partial ice formation cannot be ruled out in the case of vitrified samples, it is unlikely considering the CPA concentrations were sufficiently high to achieve vitrification (8 M). Jomha et al. demonstrated that a DMSO concentration of 6 M or more permitted complete vitrification, absence of ice, and minimal matrix disruption. 27 Our hypothesis that vitrification would protect against mechanical alterations was premised primarily on the avoidance of matrix disruption resultant from ice formation. That there were mechanical alterations in the stress-relaxation response post-vitrification, requires explanation. The osmotic stress of vitrification (repeated swelling and shrinking), which results from exposure to CPA solutions of different osmolalities, could adversely alter matrix structure and composition. 11 For example, if stress secondary to osmotic flow was sufficient to fragment proteoglycans or the collagen network, matrix permeability could increase and manifest as a shorter relaxation time, as seen here. The osmotic pressure within the ECM has been shown to be primarily due to excess ions attracted to the fixed negative groups of glycosaminoglycans. 33 If, for instance, the highly polar S=O group of DMSO 34 were to draw ions out of the ECM during CPA loading and these were not replaced by ions circulating in the DMEM, this mechanism may also have limited the osmotic pressure resisting fluid exudation. Another possibility is the rapid flow of water out of the matrix upon initial exposure to the high concentration of CPAs (without intervening sequential increases in CPA concentrations to manage the flow) may have caused physical damage to the matrix components. To confirm an explanation for both frozen and vitrified groups will necessitate further research. It should be noted that the decrease in relaxation time constants between the groups represents a small difference that may only become apparent with constant, prolonged loading, as opposed to the more rapid cyclic loading of normal walking. The actual impact of these differences in behavior will be investigated with a transplant study in future work.
From a clinical perspective, these results raise questions, to be expected for a pilot study. The majority of the mechanical results for both frozen and vitrified grafts were not different from the fresh grafts, but Pallante et al. 8 has shown that frozen grafts deteriorate over 6 months after transplantation due to matrix disorganization. It is known that frozen grafts do not have live chondrocytes to restore graft integrity, which could be possible in the vitrified grafts. Further, the fresh and frozen conditions used in this study do not mimic clinical practice. The clinical standard for “fresh” would be after storage for 21–28 days at 4°C in some maintenance media as compared to the 40 h of storage here. The frozen grafts clinically are typically frozen at approximately 1°C per minute compared to rapid immersion in LN2 in this study which is approximately 60°C per minute. 35 Slow freezing causes much larger ice crystal formation which can impart larger structural damage on the cartilage matrix than smaller ice crystals from rapid freezing. Thus, the mechanical differences noted here may not be equivalent to those that cause graft deterioration in other published clinical studies and it is difficult to know whether the one difference noted in this study has clinical implications. Testing a fourth group of experimental samples, prepared by slow freezing and thawing cycles, was not included within the study but would be beneficial to assess in future study designs. The intent of this study was not to closely mimic clinical storage protocols, but instead to act as a first step in evaluating mechanical differences between storage methods generally. For this reason, experimentally simple methods of freezing and vitrification were employed. Future studies will continue with investigations designed to address some of these clinical questions.
The present study investigated the changes in mechanical properties of the AC tissue under rapid loading and stress-relaxation with vitrification and freezing compared to fresh tissue. It was found that both freezing and vitrification maintain the mechanical integrity of AC under rapid loading and at equilibrium conditions, but may result in faster stress-relaxation compared to fresh tissue. Thus, our hypothesis was only partially correct and, as such, next steps will include investigation of the effect of osmotic stress and water movement on ECM properties during vitrification and more closely reflect clinical practice. Optimization of the vitrification protocol may improve the results. Performing histology after mechanical testing would also be of value to assess the effect of testing on the cellular and extracellular components of the samples, keeping in mind that long mechanical testing may alter the histology of the samples. Furthermore, the clinical significance of this difference in stress-relaxation behavior between fresh and vitrified AC requires exploration with transplantation studies. To our knowledge, this is the first study that endeavors to compare the mechanical properties of fresh, frozen and vitrified AC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Edmonton Orthopaedic Research Committee. J.H. is funded by the Alberta Innovates Summer Research Studentship (SRS). I.W. is funded by the Natural Sciences and Engineering Research Council (NSERC). K.W. is funded by the Li Ka Shing Sino-Canadian Exchange Program between University of Alberta and Shantou University. Samples were obtained from Delton Sausage & Deli in Edmonton, Alberta.