Abstract
Phase change materials can be the most suitable way to enhance the thermal performance of a heat exchanger, and it is one of the important parameters for various developments in renewable energy and engineering applications for a sustainable future. At the time of phase change, the phase change material can store energy and release that stored energy in the form of heat. It is an eminent candidate for different engineering sectors, including thermal, electronics, civil, and textile. In this research, we used lauric acid as a phase change material and copper oxide, aluminium oxide as nanoparticles experimentally. The characterisation tests such as Thermogravimetric analysis, Fourier Transform Infrared Spectroscopy, Ultraviolet-visible, Thermal Conductivity, X-Ray Diffraction, Field Emission Scanning Election Microscope were carried out on the phase change materials and nano phase change materials. The phase change materials are tested individually to analyse the heat transfer rate of the heat exchanger for a specific period of time at three different inlet temperatures. The thermal conductivity of the phase change material is improved by the addition of copper oxide and aluminium oxide nanoparticles to it. By adding Aluminium oxide and copper oxide nanoparticles to the lauric acid, the heat storage capacity is increased by 16.52%, 38.89% at 60°C, 17.75%, 41.33% at 70°C, and 22.67%, 46.17% at 80°C respectively. Copper oxide is the most suitable nanomaterial to improve the thermal conductivity of low thermal conductive phase change materials, since it has properties like high thermal conductivity, low thermal interface resistance, and high aspect ratio. The heat energy stored in phase change materials is increased due to the addition of nanoparticles. In future, a suitable optimization technique can be employed to predict the optimum nanoparticle weight percentage to get improved thermal performance.
Keywords
Introduction
Heat exchangers are heat transfer devices that allow the flow of energy in the form of heat between two or more fluids at different temperatures. Heat exchangers are mostly used in the process industries, power production, manufacturing industries, electronics, food and chemical industries, refrigeration systems, air-conditioning systems, and space applications.
Phase change materials (PCMs) are mostly used as thermal energy storage materials because they have the property of storing and releasing huge amounts of thermal energy during a phase change (i.e. melting and solidification). Due to environmental benefits and the increase in fossil fuel prices, PCMs are becoming attracted to different engineering applications, such as electronic cooling, building space heating and cooling, and battery thermal management. In this research, we designed a heat exchanger which can exchange heat from the working fluid to PCMs, and conducted experiments to increase the heat storage rate.
BeleenZalba, et al. researched the materials, heat transfer, and applications of phase change materials. The researchers give information about 150 different types of materials that can be used as PCMs, among which 45 are already available on the market. 1 Laura Colla, et al. presented the use of aluminium oxide(Al2O3) nanoparticles to increase the thermal conductivity of paraffin wax, which can be used as a phase change material. They used 0.5% and 1% weight of aluminium oxide nanoparticle and mixed them into two different paraffin waxes. Various properties, like latent heat, conductivity, and many more, are measured to find out the effect of adding nanoparticle to PCMs. An electronic passive cooling device is used to compare the PCM and Nano PCM (NPCM) numerically. They have also used ANSYS simulation software for carrying out simulations and the results are obtained according to their expectations. 2 Ravi Agarwal, et al. synthesized the copper oxide (CuO) nanoparticle using the wet chemical method. They have used copper sulphate and copper acetate both to prepare different copper oxide nanoparticle. The result showed that the nanoparticle prepared from the copper acetate were smaller size and regular comparison, while this was not the case for copper oxide. The Nano fluid was prepared in distilled water by using nanoparticles that were prepared from copper sulphate. Then a thermal conductivity test was conducted on the above sample. There was a rise in thermal conductivity by 40%, which was due to the use of distilled water. When ethylene glycol and engine oil were used in place of distilled water, the rise was only 27% and 19% respectively. 3 Harikrishnan, et al. conducted an experiment to investigate the thermo-physical properties of a mixture of water and glycerol which contains copper oxide nanoparticle which act as a phase change material for thermal storage systems. They took the above mixture in the ratio of 80:20 by weight and used a different mass percentage of nanoparticles. This helped in measuring latent heat and temperature by using differential scanning calorimetry (DSC). The improvement in the thermal conductivity by using a different proportion of nanoparticle is measured by using a laser flash analyzer. Their results show that the PCM can be used in thermal energy storage systems. 4 Kaviarasn, et al. researched the presence of different types of phase change materials and also the different types of nanoparticles present. They have given an idea of the applications of adding nanoparticles to PCMs. According to their research, paraffin wax, hydrated salts, water and bio-based are the most common kinds of PCM used nowadays. The nanomaterials that can be used are copper, aluminium, silicon, carbon nanotubes (CNT), carbon nanofibers (CNF). 5 Muath et al. tried to increase the thermal conductivity of phase change material by adding nanoparticle to it. They have used copper oxide and aluminium oxide as nanoparticles. According to their results, it is concluded that the melting time of PCM is reduced by the addition of nanoparticle to it. Copper oxide showed better results as compared to aluminium oxide. 6 Dsilva, et al. investigated the enhancement of PCMs by adding nanoparticle to it on solar still operations. They used different nanoparticles in different weight percentages for preparing a mixture of NPCM. They used paraffin as the phase change material. Thermal conductivity was increased and the melting point was decreased. Thus, they proved that NPCM can be used as the best solar still. 7 Sivasankaran Harish, et al. prepared lauric acid phase change material and added graphene nanoparticle to obtain NPCM. They used the hot wire method to measure its thermal conductivity. It was found that there was a 230% increase in thermal conductivity due to the addition of graphene nanoparticle. Thus, it proves that the graphene nanoparticle is better than the copper oxide nanoparticle but at the same time, it is very expensive as compared to the latter. They also conducted differential scanning calorimetry to find out the phase change enthalpy and melting temperature. 8 Tayyab-ul-Islam, et al. used copper oxide nanoparticle in four different types of heat exchangers, which are shell and tube, concentric, spiral, and plate types. The copper oxide nanofluid is used in three different concentrations. And the enhancement of the heat exchanger on the addition of nanofluid is analyzed. 9 Xiaowei Fu et al. investigated the chemical structure and thermal properties of the Lauric Acid/diatomite composite by different tests like X-ray diffraction (XRD), Fourier transform infrared (FTIR), Scanning Electron Microscope (SEM), Thermogravimetric analysis (TGA), and the results showed the composite of PCMs have good potential for building applications. 10 Ying Zhang et al. prepared hydrophobic LA/SiO2 PCMs by the one-pot method. The result shows that at the time of increasing volume ratio of methyltriethoxysilane (MTES) to tetraethylorthosilicate (TEOS), the surface of PCMs changed to perfect hydrophobic. 11 Liu Jiesheng, et al. tested the leakage test for Lauric Acid (LA)/Expanded Perlite (EP) composite PCMs. It shows the extreme mass fraction of Lauric Acid controlled in the composite without leakage is 70% of weight. 12 Ahmet Sari, et al. tested the perfect eutectic mixture of Lauric Acid (LA) and stearic acid (SA), and the weight form of the LA-SA eutectic is 75.5%:24.4% at the melting point of 37°C. 13 Chuqiao Zhu et, al., improved the thermal properties of LA/IF composites by using graphene nanoplates (GNP). This shows that graphene nanoplates will be helpful for improving the thermal conductivity of PCMs. 14 Dudul Dasa, et al. increased the thermal conductivity by adding aluminium metal powder (5% of weight) for solar latent heat storage applications. 15 Jagadeeswara Reddy Vennapusa, et al. used 6 different porous supports for the investigation of Caprylic acid (CA) based PCMs composite for the process of packaging in industries. 16 Shuangping Duan, et al. conducted an experimental study on the improvement of the thermal performance of the Trombe wall at a ratio of 45% lauric acid and 55% decanoic acid (DA). 17 Qiang Shena, et al. developed a composite PCM (Lauric acid/modified sepiolite) by the vacuum impregnation method. 18 Mechiri Sandeep Kumar, et al. focused on hybrid nanoparticles Al2O3-CuO for the combination of 75:25, 50:50, and 25:75, and the various volume concentrations of hybrid nanoparticles were tested (about 0.5%, 1%, 2%, and 3%). 19 Sheikholeslamia, et al. tested the charging and discharging units of PCM in a triangular fin embedded with Copper Oxide (CuO) nanoparticle. 20 Al-Waeli, et al. developed a mathematical model for Nano fluid and Nano-PCM for the application of combined PV/T, and the result showed the overall efficiency of the system was improved to 85.7%. 21 Manar Al-Jethelah, et al. investigated the thermal performance of the TES system using scale analysis, experimental analysis, and numerical simulation. The nano-PCMs (CuO), dispersed in coconut oil in different volume fractions of nanoparticle. The computational and experimental findings suggest that adding nanoparticles to PCM improves the melting process significantly. 22 Mohsen Sheikholeslami showed that adding CuO nanoparticles to pure PCM speeds up the solidification process. Furthermore, it is possible to deduce that the maximum rate of solidification is attained for nanoparticle diameters of 40 nm. 23 Swaroop Kumar, et al. concluded that the thermal conductivity, heat flow, and thermal efficiency of PCM improve as the mass fraction/wt percent of nano CuO rises. 24
Though much research has been conducted on PCM, only a limited number of studies have been reported on the estimation of thermal performance of nanoparticles added to Free Fatty Acids (FFA). The Lauric acid is such a FFA that is used in electronic cooling applications, has lower thermal conductivity, but with the addition of copper oxide nanoparticle, the thermal conductivity is increased. Few researches were reported on the combination of Lauric acid with CuO and Al2O3 nanoparticles. This work makes an attempt to characterize and evaluate the heat transfer characteristics of the nanoparticle embedded PCMs.
Materials and methods:
Phase change material
We have used lauric acid as a phase changing material. Lauric acid [C12H24O2] with a melting temperature of 43-44 °C was purchased from Nice Chemicals Private Limited (India). The lauric acid was in powdered form. It was melted using the heater. Table 1. shows the properties of lauric acid.
Properties of Lauric acid.

Preparation of nanoparticles
Copper oxide nanoparticle are synthesized using a wet chemical method and then they are provided with high temperature to remove any sort of impurities present in them. These can be synthesized by using either copper sulphate or sodium hydroxide mixture or by using copper acetate and sodium hydroxide mixture. This test starts with the addition of sodium hydroxide drop by drop to copper sulphate or copper acetate solution with continued stirring until the pH falls between 6 to 7. A black precipitate is obtained which is separated by using a centrifugal machine. Then it is left for dehydration in the air furnace and then copper oxide nanoparticle is obtained in powdered form. Copper oxide nanoparticle is prepared by using the wet chemical method. 8 gm of copper sulphate is added into 200 mL of water. The solution is allowed to heat up and stir in the stirring machine at a temperature of 40°C for half an hour. Then 4 gm of sodium hydroxide pellets are mixed with 200 mL of water. The same sodium hydroxide solution is added to the copper sulphate solution drop by drop. The pH of the solution is checked at different intervals of time till it comes in the range of 6-7. As a result, a black precipitate of copper oxide is formed. The precipitated solution is then processed through a centrifugal machine which separates the precipitate from the liquid and the precipitate settles at the bottom surface and the liquid is drained out. Then the precipitate collected from the centrifugal machine is allowed to dry in the incubator at 85°C constant temperature for 24 h. The copper oxide granules are collected and transferred to the furnace in a ceramic container and heated up to 450°C for 2 h. Then the sample is ground to obtain the powdered form.
10 gm of Al (NO3)3.9H2O was added to 150 mL of distilled water. The solution is stirred in the stirring machine at room temperature. Then 14 mL of ethanol solution was added drop by drop. The pH of the solution is checked at different intervals of time till it comes within the range. As a result, the colour changed from orange to dark brown. The aluminium oxide granules are collected and transferred to the furnace in a ceramic container and heated up to 500°C for 3 h. The prepared nanoparticles underwent heat treatment for 4 h at 1300°C before cooling to room temperature in the oven.
This research approaches increasing the thermal conductivity of phase changing material by the addition of nanoparticle. Further, the increase in conductivity also results in increasing the heat storage capacity for a particular period. It is because of that, heat can be stored at a faster rate as and when required, for example, during heat losses. Here it is done by performing an experiment that gives temperature as the output and, using that temperature, the heat storage rate is calculated. When heat is lost from the heat exchanger, the PCM stores it on one side and doesn't allow it to escape.
Fabrication of experimental setup
Firstly, the dimensions of the wooden box are fixed and then the wood is cut according to those dimensions using an angle cutter. The pieces are joined together to form a wooden box. Holes are drilled on the two opposite faces of the box by using a drilling machine to insert the copper pipe. Next, we bent the copper pipe using a bender in the turns of the S-form. Digital display temperature sensors are attached to the bottom base of the box to get the temperature differences. The sides and corners of the wooden box are sealed with M-seal and leakage testing was done by pouring water into it.
Figure 1(a) shows the experimental setup used in this work. It consists of the following components.
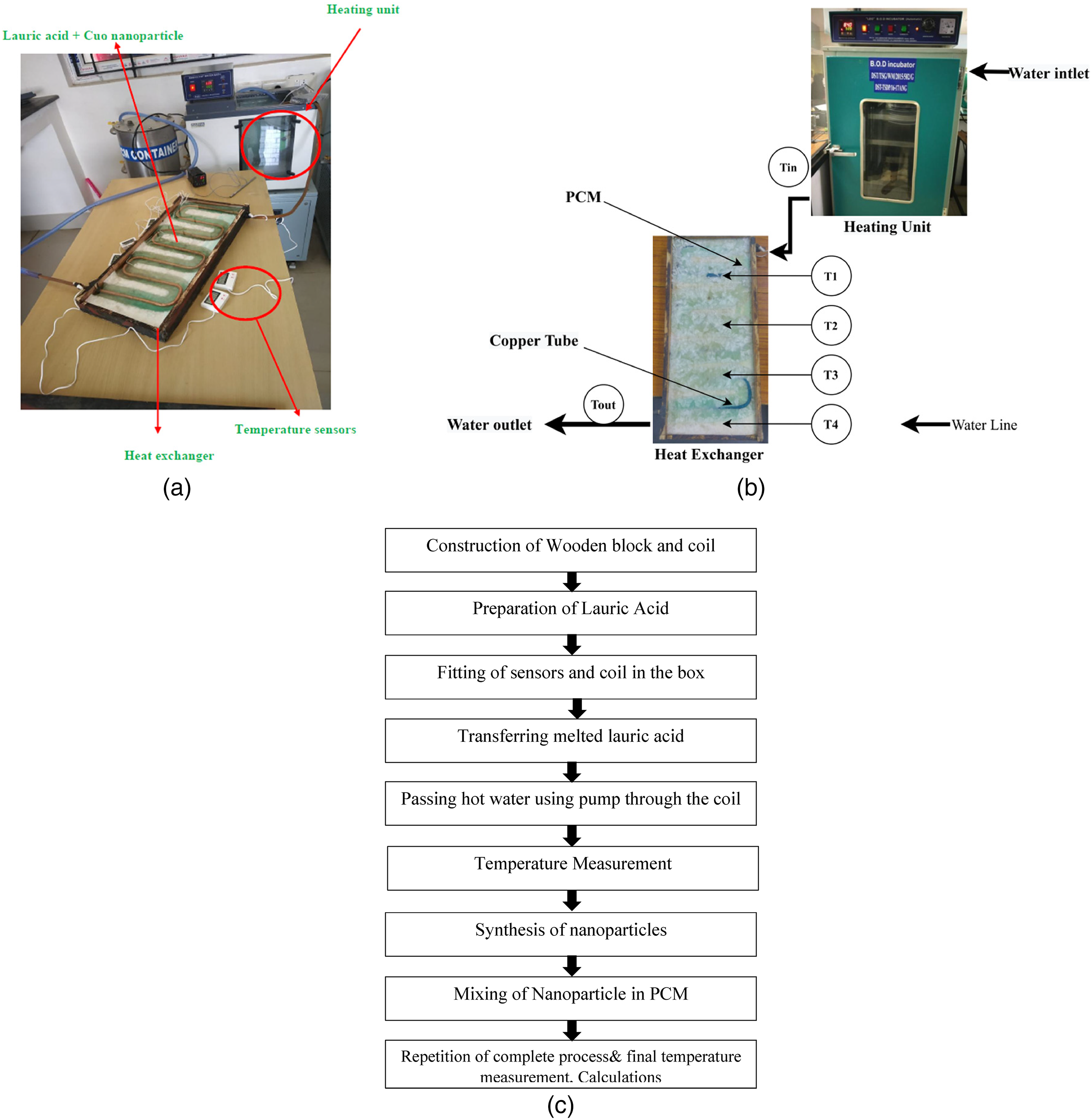
(a) Experimental setup. (b). Schematic diagram of the experimental setup. (c). The flow of working process.
The lauric acid which is melted is poured into the heat exchanger and allowed to freeze at room temperature. The setup consists of the heat exchanger (Figure 1(b)) connected through a pipe to the pump that is immersed deep in the heating unit having water as the heating element. The water from the heating unit is passed through a pipe to the copper coil and the process lasts for 30 min. Figure 1(c) shows the flow of the working process.
The lauric acid is melted and the temperature below different turns of the copper coil is noted using temperature sensors. Nanoparticles in powdered form are mixed into the melted lauric acid and the mixture is allowed to stir in a stirring machine for 1 h. The mixture is again allowed to mix in the sonicator for proper dissolution. The above steps are repeated again for the above prepared lauric acid and nanoparticle mixture and the temperature readings are again noted for this mixture.
Analysis methods
The thermal conductivities of Lauric acid and NPCMs were tested at room temperature using a Decagon Devices Inc (KD2 Pro Thermal Properties Analyzer), and the measurements were repeated five times to ensure the correctness in results. A thermogravimetric analyser is used to conduct the TGA test (SDT Q600, TA Instruments, USA). A thermogravimetric analyser continuously measures mass that changes with the temperature changes over time. Mass, temperature, and time are regarded foundation measures in thermogravimetric analysis, from which a variety of other data may be generated. By creating an infrared absorption spectrum, Fourier Transform Infrared Spectroscopy (FTIR) detects chemical bonds in a molecule. The composition of PCMs is revealed by FTIR spectra (IRAffinity-1, Shimadzu, Japan). The most common purpose of this test to identify unknown materials and validating new material composition. SPECORD (210 plus) was used to monitor the Ultraviolet-visible (UV-Vis) test The purpose of this test was to identify, characterise, and explore nanomaterials, since the optical characteristics of nanoparticles are highly dependent on their size, shape, concentration, aggregation state, and refractive index near the nanoparticle surface. X-Ray Diffraction examination (Bruker D8 Advance, Germany model) was used to determine the crystal size of nanoparticles. The scattering angle 2θ was maintained between 20 to 80 degrees. The average crystallite size (D) was derived from the line broadening using Debye-Scherrer's equation D = 0.9λ/Wcosθ, where W is the full width of half maximum (FWHM) and λ is the wavelength of the X-ray. 25 A Field Emission Scanning Electron Microscope(FESEM) was used to examine the morphology of CuO and Al2O3 nanoparticles (FEI QUANTA 250 FEG, Thermo Fisher) and Energy Dispersive Spectroscopy (EDS) test was carried out to identify the material composition and dispersion rate in the same.
Data reduction
The capacity of heat stored in Phase Change materials is calculated by using the following expression (1),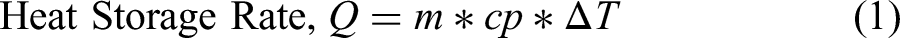
m = mass of the phase change material (kg)
Cp = Specific heat of the phase change material (kJ/Kg.K)
ΔT = Temperature gradient (°C)
The temperature gradient is measured using the following expression (2),
t2 = Average temperature of the phase change material (°C)
t1 = Initial temperature of the phase change material (°C)
Here, t2 is calculated by taking the average temperature of PCM. Because in the wooden box, we have fixed four temperature sensors in four different places. The temperature (t2) is calculated by using the following expression (3),
T1, T2, T3, T4 = Temperature of the phase change material (°C)
Uncertainty analysis
Uncertainty analysis was done by using Moffat, R. J.
26
method. By using the following expression (4), the experimental uncertainty was determined for various parameters.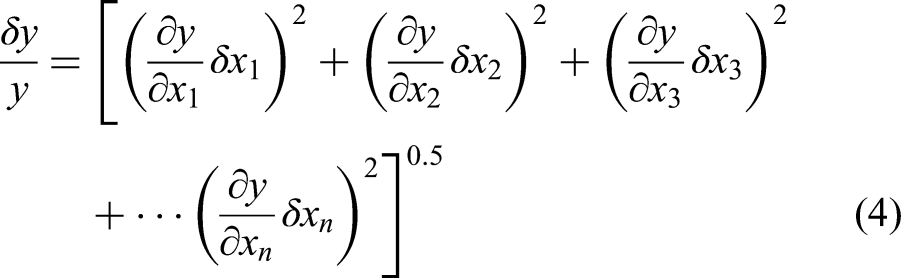
Results and discussions
TGA test result
The sample of lauric acid phase changing material in powdered form was given for the Thermogravimetric analysis test This test is performed to get information about the properties of lauric acid like absorption, adsorption, and thermal decomposition. The temperature at which the test is carried out is 800°C at a rate of 20°/minute. The result of the test is obtained in the form of a graph which is known as a decomposition graph, as shown in Figure 2. The decomposition of lauric acid starts at 120.84°C and the complete decomposition occurs at 355.37°C. Using the graph (Figure 3), the weight loss up to 120.84° is calculated as 3.131%. The main decomposition took place in the temperature range of 120–256°C. Thus, the test results confirm the suitability of lauric acid to be used as a phase changing material in the experiment.
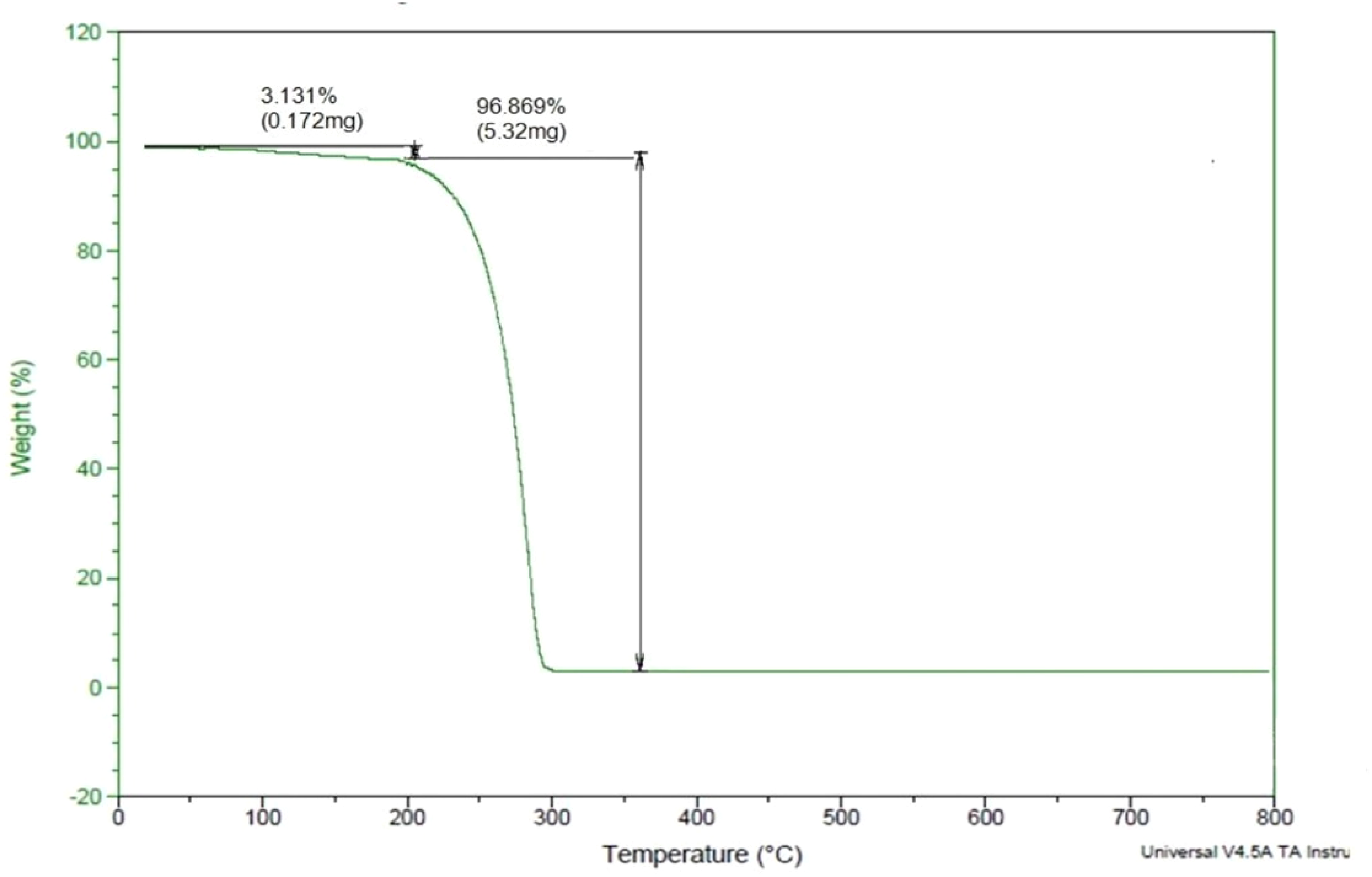
TGA graph of lauric acid.
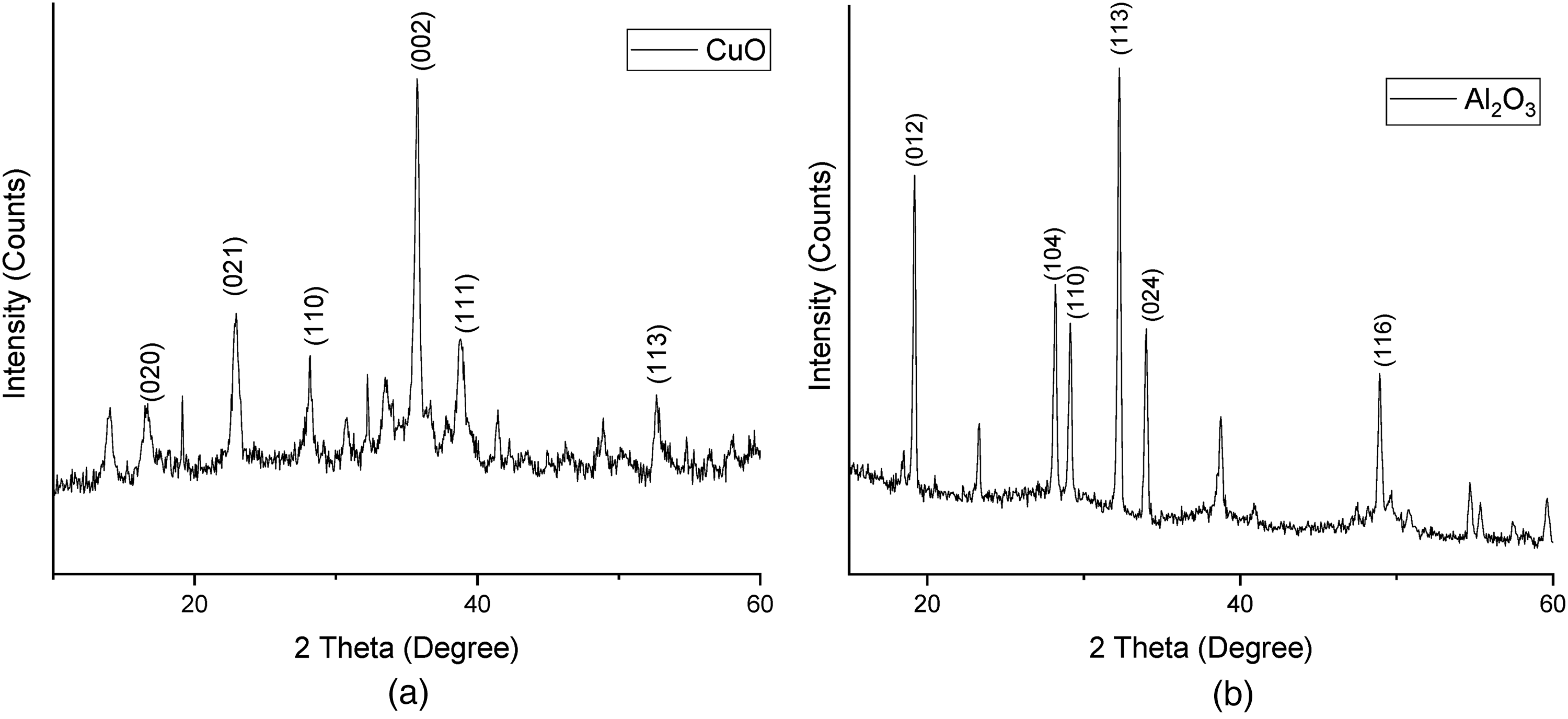
(a). XRD Pattern of CuO Nanoparticles. (b). XRD Pattern of Al2O3 Nanoparticles.
Characterization of nanoparticles
The powder X-ray diffractometer is used to characterise nanoparticles in the 2 theta range of 20° to 60°. The formation of CuO and Al2O3 nanoparticles has been confirmed using X-ray diffraction. The XRD patterns show that the material is crystalline with a monoclinic structure in nature. 2θ = 16.68°, 23.11°, 28.13°, 35.47°, 38.71°, 52.65° correspond to the characteristic reflection of (020), (021), (110), (002), (111) and (113) for copper oxide (Figure 3(a)). 27 Peaks of Al2O3 (Figure 3(b)) are 2θ = 19.18°, 28.22°, 29.14°, 32.26°, 34.1°, 48.9°, which correspond to the characteristic reflections of (012), (104), (110), (113), (024), (116), planes with monoclinic structures, and are well matched with JCPDS (Joint Committee on Powder Diffraction Standards) data (file No. 89-5895, 901-5131 for CuO and Al2O3 respectively). 28
The produced CuO and Al2O3 nanoparticles emerged as spherical and near spherical shapes in Figure 4(a) and (b), respectively. The particle diameters were determined to be between 54 nm and 74 nm for Copper Oxide and 26 nm and 53 nm for Aluminium Oxide. The spherical shape of the nanoparticles may provide sufficient surface area for heat interaction, which might help to speed up the heat transfer rates of composite PCMs during the melting and solidification processes. Figure 4(c) and (d) show the findings of Energy Dispersive Spectroscopy (EDS) for CuO and Al2O3 nanoparticles, respectively.
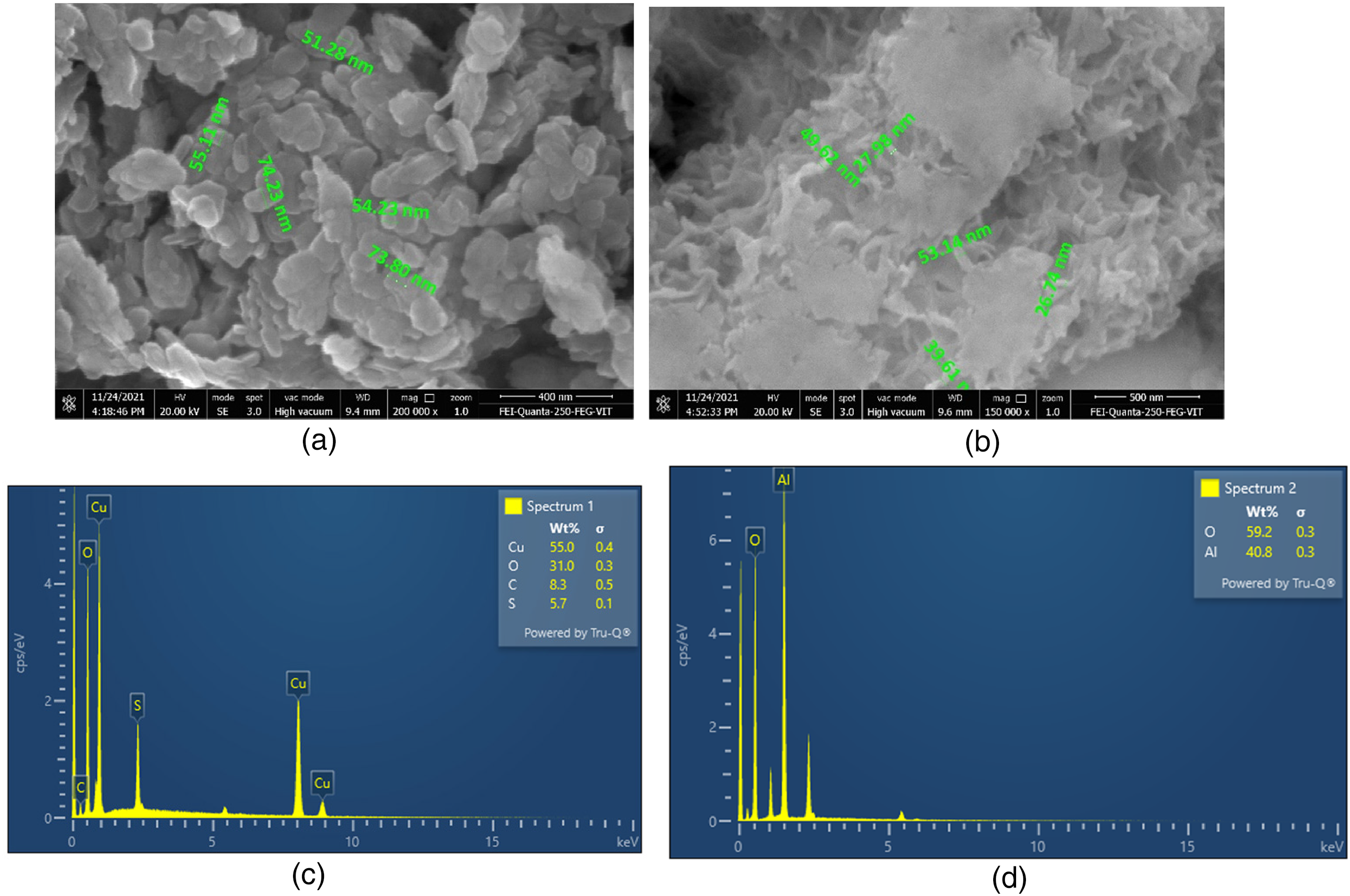
(a). FESEM image of CuO nanoparticle. (b). FESEM image of Al2O3 nanoparticle. (c). EDS Result of CuO nanoparticle. (d). EDS Result of Al2O3 nanoparticle.
The UV test identifies various characteristics of the sample, for example, reflectance, adsorption, in the ultraviolet and visible spectrum(UV-vis). And the output is obtained in the form of a graph, as shown in Figure 5(a). The graph shows the wavelength absorption of the copper oxide nanoparticle. The sample shows maximum absorption at 222 nm and 252 nm for CuO and Al2O3 nanoparticles respectively. In the graph, the peak shows the inter band transition from deep level electrons in the valance band.
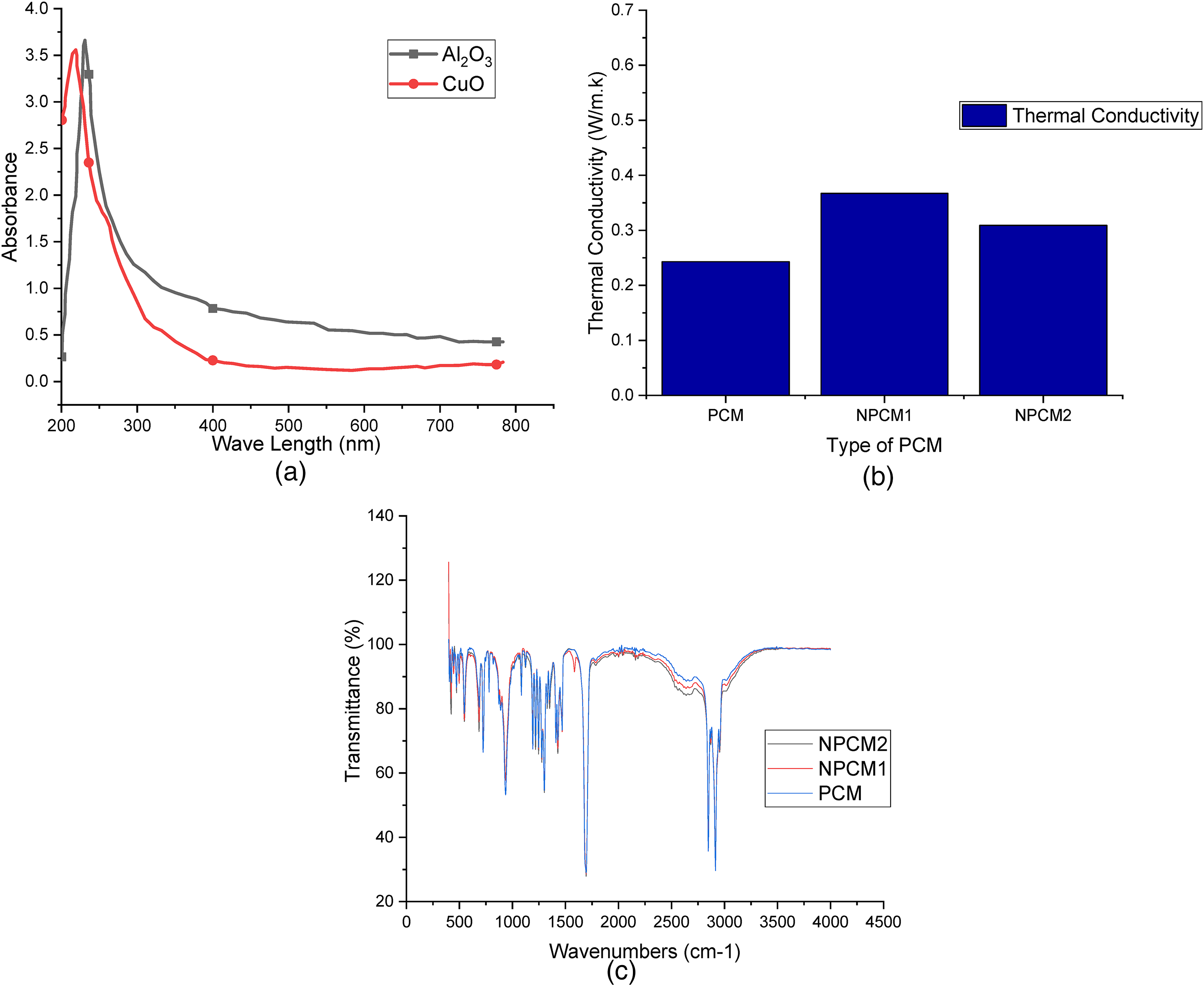
(a). Wavelength v/s Absorption graph of nanoparticles. (b). Thermal Conductivity of PCMs. (c). FTIR results of PCMs.
Characterization of nano phase change material (NPCMs)
PCMs with superior thermal conductivity are chosen for the latent heat storage systems for enhancing energy storage and release rate capabilities. CuO and Al2O3 nanoparticles were employed to improve Lauric acid's thermal conductivity. The impact of Copper oxide and Aluminium oxide nanoparticles on the thermal conductivity of Phase change materials are shown in Figure 5(b). The KD2 Pro Thermal Properties Analyzer was used to take individual measurements of thermal conductivity for PCM and NPCMs at room temperature (29°C). Thermal conductivity values for PCM, Copper Oxide nanoparticles mixed PCM (NPCM1), and Aluminium Oxide nanoparticles mixed PCM (NPCM2) were 0.243 W/mK, 0.367 W/mK, and 0.309 W/mK, respectively.
The results of the tests show that nanoparticles improve the thermal conductivity of NPCMs. When compared to Lauric acid, the thermal conductivity of the PCMs containing CuO and Al2O3 nanoparticles was increased by 51.05% and 27.16%, respectively. In comparison to NPCM2, the thermal conductivity of NPCM1 increased by 18.77%. Better thermal conductivity might be achieved by increasing the mass fraction of nanoparticles. 29
The findings of Fourier-transform infrared spectroscopy (FTIR) demonstrate that nanoparticles dispersion was uniform throughout the PCM. Based on the FTIR spectrum graph, no new peak was discovered, as shown in Figure 5(c). The FTIR spectrum graph revealed that there was no chemical reaction as a result of the components being mixed together. As seen in the graph, there was only a physical contact between the lauric acid and the CuO and Al2O3 nanoparticles.
Validation of the experiment
The experiment results were validated using the energy balance equation. The outlet water temperature is taken from the temperature sensor, and that temperature value was compared with the results obtained from the expression (7).
Total Amount of Heat Supplied = Amount of Heat Stored + Amount of Heat Rejected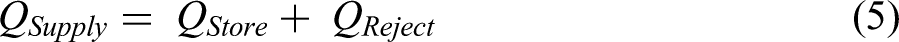
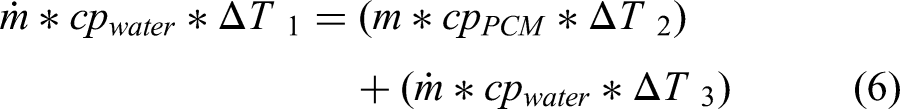
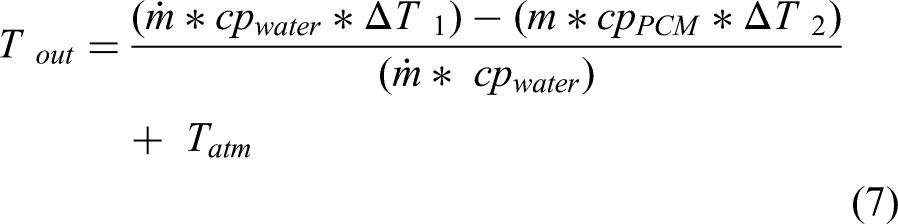



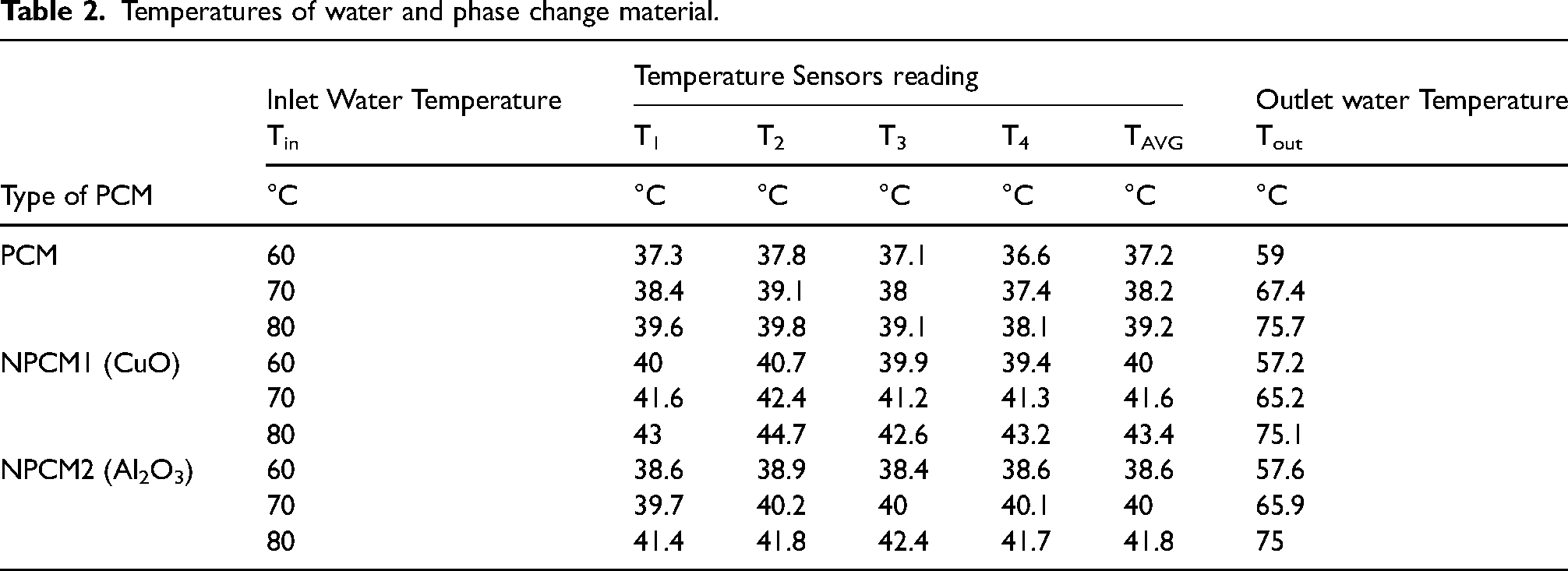
Where, m = mass of the PCM (2.5 kg), ṁ = mass flow rate of water (0.28 kg/s), Tin = Inlet water temperature (60°C, 70°C, 80°C), Tout = Outlet water temperature, Tatm = Atmospheric temperature (30°C) and TPCM = Phase Change Material temperature. The inlet water temperatures(Tin) were varied from 60°C to 80°C. The TPCM is calculated from the expression (3). The experimental outlet temperature of the water is shown in table 2. Figure 6(a) shows the comparison of the experimental and theoretical results.

The error percentage was calculated using the expression (11). For this experiment, the error percentage is within 5%. This error may occur because of heat loss.
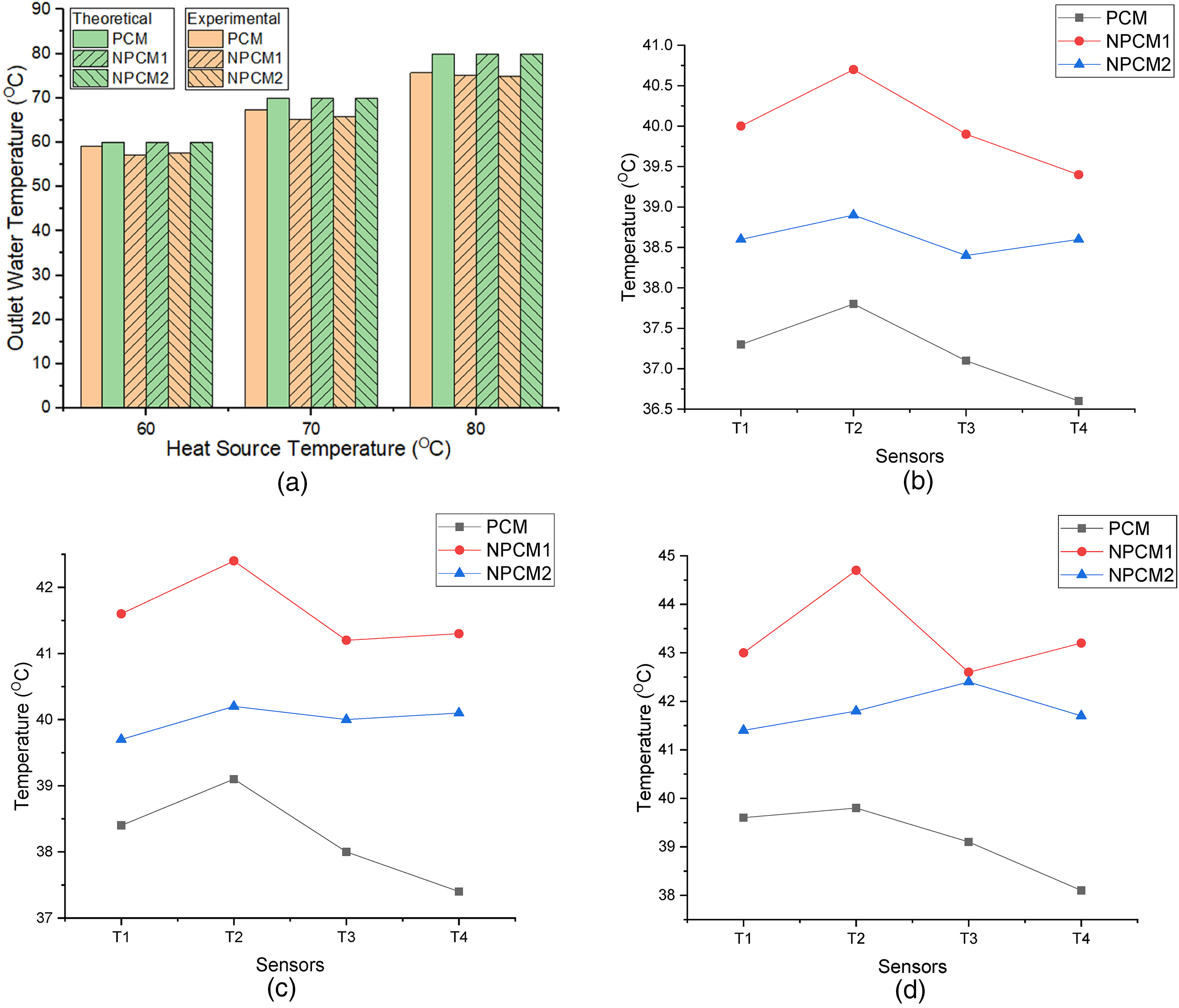
(a). Outlet Water Temperature (Experimental vs Theoretical). (b). Inlet Temperature at 60°C. (c). Inlet Temperature at 70°C. (d). Inlet Temperature at 80°C.
Temperatures of water and phase change material.
Heat storage capacity of PCM
Lauric acid is taken as a storage material in this case. The heat storage capacity is calculated using the expression (1). Here, heat is supplied by the water at three different temperatures likely 60°C, 70°C and 80°C for 30 min respectively. Table 2 shows the temperature readings of PCM. The average temperature reading of PCM in the wooden block at 60°C, 70°C, 80°C is 37.2°C, 38.2°C, and 39.1°C respectively. The temperatures are measured by using temperature sensors, and the readings are plotted in Figure 6(b) to 6(d) at 60°C, 70°C and 80°C respectively.
Using the expression (3), the average temperature of PCM is calculated. The specific heat of the lauric acid is 2.27 kJ/kgK, and the volume of the wooden block is calculated by using the dimension of it. The volume of the wooden block is 0.00288 m3. The initial temperature of lauric acid, t1 = 30°C. Table 2 shows the temperatures of water and phase change material. The amount of heat stored in PCM at 60°C, 70°C, 80°C is 40.57kJ, 46.35kJ, and 51.56kJ respectively.
Heat storage capacity of NPCM1
Copper Oxide nanoparticles mixed PCM (NPCM1) is taken as a storage material in this case. CuO nanoparticles are mixed with lauric acid. The heat storage capacity is calculated using the expression (1). Here, heat is supplied by the water at three different temperatures likely 60°C, 70°C, and 80°C for 30 min respectively. Table 2 shows the temperature readings of Nanoparticles Phase Change Material (NPCM1). The average temperature reading of NPCM1 in the wooden block at 60°C, 70°C, 80°C is 39.4°C, 41.3°C, and 43.2°C respectively. Using the expression (3), the average temperature of NPCM1 is calculated. The temperatures are measured by using temperature sensors. The initial temperature of lauric acid, t1 = 30°C. Table 3 shows the heat storage capacity of NPCM1. The amount of heat stored in NPCM1 at 60°C, 70 °C, 80°C is 56.35kJ, 65.51kJ, and 75.37kJ respectively. The amount of heat stored in NPCM1 is increased by 15.78kJ, 19.16kJ, 23.80kJ compared with PCM at 60°C,70°C, and 80°C respectively. And the heat storage percentage increased by 38.89%, 41.33%, and 46.17% respectively.
Heat storage capacity of phase change materials.

Heat storage capacity of NPCM2
The aluminium Oxide nanoparticles mixed PCM (NPCM2) is taken as a storage material in this case. Al2O3 nanoparticles are mixed with lauric acid. The heat storage capacity is calculated using the expression (1). Here, heat is supplied by the water at three different temperatures likely 60°C, 70°C, and 80°C for 30 min respectively. Table 2 shows the temperature readings of Nanoparticles Phase Change Material (NPCM2). The average temperature reading of NPCM2 in the wooden block at 60°C,70°C, 80°C is 38.6°C, 40°C, and 41.8°C respectively. Using the expression (3), the average temperature of NPCM2 is calculated. The temperatures are measured by using temperature sensors. The initial temperature of lauric acid, t1 = 30°C. The amount of heat stored in NPCM2 at 60°C,70°C, 80°C is 48.6kJ, 56.35kJ, and 66.63kJ respectively. The amount of heat stored in NPCM2 is increased by 8.03kJ, 10kJ, 15.1kJ compared with PCM at 60°C, 70°C, and 80°C respectively. And the heat storage percentage increased by 16.52%, 17.75%, and 22.62% respectively.
The variation is due to the change in the type of the Phase Change Material (Organic/Inorganic), the melting point of the PCM, Volume of PCM, heat source temperature, process timing, the concentration of nano particles. Table 3 shows the comparison of the present work with the existing work that is done with paraffin wax. It is clear from table 3 that the addition of nano particles enhanced the thermal characteristics of the base PCM in both cases.
The experiment was conducted on different types of nanoparticles in PCM for a heat exchanger. The results are shown in Figure 7. It is observed that the heat storage capacity is increased by 38.89% at 60°C, 41.33% at 70°C, and 46.17% at 80°C by using NPCM1, and 16.52% at 60°C, 17.75% at 70°C, and 22.62% at 80°C by using NPCM2. The amount of heat stored in NPCM1 is increased by 7.75kJ, 9.16kJ, 8.74kJ at 60°C, 70°C, and 80°C respectively compared with NPCM2. Thus, by adding nanoparticles to PCM, the thermal conductivity of lauric acid PCM increases. Copper Oxide nanoparticles added to PCM (NPCM1) give better results compared with the remaining two PCMs. Because of copper's good thermal conductivity. Thus, the performance is improved and the efficiency of the heat exchanger is increased. The thermal conductivity and heat storage rates are depending on the amount of nanoparticles added to the base PCM. The addition of CuO and Al2O3 nano particles increased the thermal conductivity from 0.243 W/mk to 0.367 W/mK and 0.243 W/mk to 0.309 W/mK respectively. Since the thermal conductivity increases the heat storage rate is also expected to increase & vice versa. The addition of nano particles enhances the thermal conductivity of pure Lauric acid by 1.5 times and 1.27 times respectively. It is in accordance with the previous research works.30,31
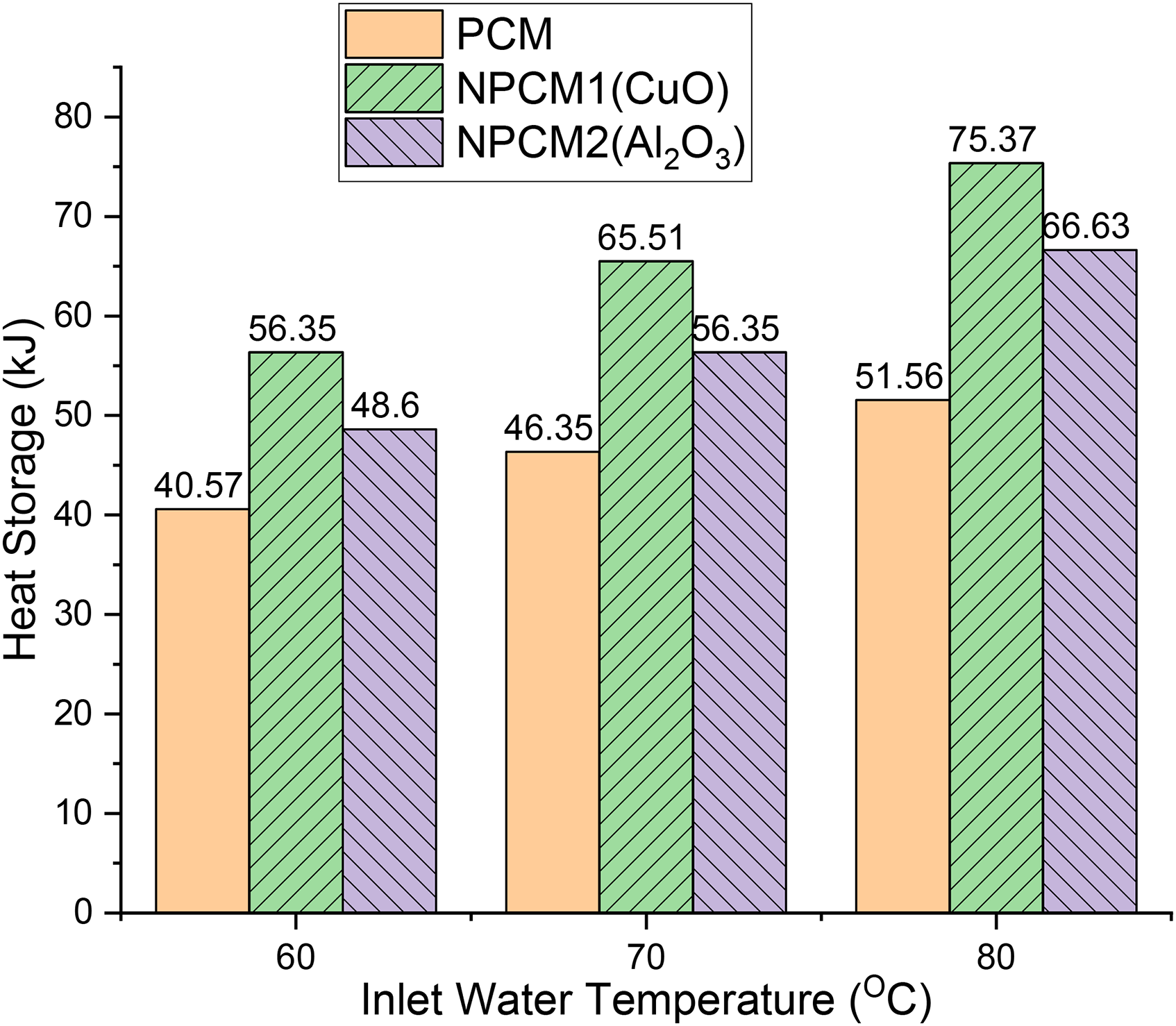
Heat storage vs inlet water temperature.
Conclusion
In this research, we have conducted experiments to find out the heat storage capacity of a heat exchanger using phase change material and nanoparticles. First, only lauric acid is used in the heat exchanger and then nanoparticles are added to it. In all three cases, temperature readings are noted and heat storage capacity is calculated. It is observed that the heat storage capacity of the heat exchanger is increased by 38.89% at 60°C, 41.33% at 70°C, and 46.17% at 80°C by using NPCM1, and 16.52% at 60°C, 17.75% at 70°C, and 22.62% at 80°C by using NPCM2. Hence, it is concluded that the thermal conductivity of phase change material is increased by adding nanoparticles to it, which shows that with the addition of nanoparticles, more heat energy can be stored for a particular period of time as compared to using only lauric acid. Thus, the performance is improved and the efficiency of the heat exchanger is increased. As nanoparticle addition to phase change material results in higher energy storage, it can be used in industrial heat exchangers which have high energy losses. Using PCM can prevent heat losses by storing the heat at a greater rate and for a short period of time. This stored heat energy can be used for different purposes. This also finds application in areas like space stations, food and agriculture, marine, chemicals and petrochemicals, HVARC, etc.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
