Abstract
A hot melt adhesive – mainly used for bonding plastic component in automotive field – was modified with different iron-based particles to give it a reversible behaviour. Mechanical and physical properties of these reversible adhesives were experimentally assessed in the work. The modified adhesives, coupled with electromagnetic induction, are able to guarantee separation of the joints without any damage to the substrates for recycling, reuse or repairing of components. Single lap joint specimens were prepared using epoxy/glass fibres substrates and tests were carried out on neat and modified adhesive with 5% weight of iron and iron oxide. Three different Fe particles size were tested: 450 µm, 60 µm and 1–6 µm. The particles size of iron oxide was 50 nm. Separation was studied using single lap joint specimens under electro-magnetic induction. Experimental results showed that the maximum peak load decreases when the average particles sizes increase. The peak loads of the smallest particles were equal to the ones of the pristine adhesive. The elongation of the adhesives increases when the adhesive is modified with both iron and iron oxide particles. Finally, experimental tests on single lap joints coupled with electro-magnetic induction showed that separation of the substrates is possible using iron oxide particles. Electro-magnetic tests conducted on particles alone, helped to understand that bigger particles are able to overcome the melting temperature of the adhesive but hot-melt adhesives modified with these particles are not able to reach the melting. These tests showed that the number of particles into the adhesive matrix is very important for this kind of tests.
Introduction
In recent years, the use of adhesives in many machinery applications has rapidly increased. Adhesive joints are often preferred to traditional mechanically fastened joints for several advantages. They exhibit a better stress distribution compared to the traditional fasteners.1,2 They can have higher resistance to environmental factors 3 and they are usually preferred to traditional fasteners when joining components made of different materials.4,5Adhesive bonding can be used to join many materials, light composite materials included, and so they represent a potential way to lighten vehicle weight for fuel efficiency and reduction of emission. 6
A wide variety of adhesives is currently available and structural and non-structural adhesives are largely adopted. Thermoset adhesives are generally employed for structural joints but, in the last years, there has also been a significant increment in the use of thermoplastic adhesives. Thermoplastic adhesives are even preferable to traditional thermoset adhesives, in some specific applications, for their ease of application and for their flexibility in joining different kinds of plastic materials, sometimes hard to bond, such as polypropylene (PP). Hot-melt adhesives have been used for both interior and exterior parts. Typical interior components, where HMAs are used, are plastic components such as trim panels, instrument gauge springs, ducts and pipes for air conditioning, sun visor and many other applications that require the fastening of non-structural plastic, wooden and fabric parts. Typical exterior applications for HMAs are plastic bumpers, some skin parts of back door (such as spoiler skin), car plate, external air conditioning ducts, lamp, lamp housing, car skirt and glass roofs. 7 The diffusion of PP based materials is increasing in many industrial fields, especially in the automotive field. 8 and this is a further reason for the increment in the use of thermoplastic adhesives.
Joining light materials, even different, is very important in automotive field, especially for lightweighting. In many cases, these materials, such as composite materials, are even expensive and for this reason, there is an increasing demand of recyclability for economics and environmental factors. For this reason, technologies able to dismantle bonded parts are needed for recovering materials that can be used again (circular economy) or for recycling. Dismantling adhesive joining is possible with the use of different technique. Mechanical cutting, acids, solvents, heating up of interested components are the traditional methods used for the separation of the bonded parts. Unfortunately, these systems can introduce some damages in the bonded parts. Many methods and processes have been proposed that allow the dismantling of the adhesives without any damage,9–16 but there is not an accepted method yet.
One of these methods uses iron oxide nanoparticles, embedded in hot-melt adhesive, coupled to electro-magnetic induction for the separation.9,17,18 In the electromagnetic induction process, inductor is used to increase the temperature of a work piece, usually a metallic component. The inductor works as a primary of an electric transformer and the conductive material as a secondary. 19 In this case, the conductive adhesive is used as the secondary of the transformer. For these reasons, since the inductor generates an alternating magnetic field, there is an increase in the adhesive temperature because of the hysteresis and eddy currents losses of the embedded magnetic nanoparticles. In this way, the adhesive temperature reaches the melting point and then bonding or separation is possible.
In this work, the mechanical properties of a hot-melt adhesive have been investigated by means of single lap joint (SLJ) test. Hot-melt adhesive was modified with three different size (450 µm, 60 µm and 1–6 µm) of iron (Fe) particles with a particle weight concentration of 5%. This percentage has been chosen based on a preliminary analysis with three different weight concentration of iron oxide 3%, 5% and 10%. In that case, the separation of substrates was possible in all the cases and the time was lower with the higher concentration. 5% was chosen because it was a good compromise between cost of iron oxide nanoparticles and separation time. These adhesives have been used to produce SLJ with epoxy-based composite substrates with glass fibres. The idea of this work is to understand whether epoxy-based composite can be joined by hot-melt adhesive and the possibility to bond many internal and external components (cited above) that could be replaced by composite materials. To the authors’ best knowledge, no experimental result is available in literature on the use of HMA to bond composite materials. Furthermore, the sensitivity of these particles to the electro-magnetic field has been investigated. The mechanical and electro-magnetic field effect on the modified adhesives has been compared with hot-melt adhesive modified with the iron oxide particles (nanomagnetite) with a particle size less than 50 nm that exhibits magnetic behaviour under induction heating.
Materials and methods
The adherents were bonded with polyolefin-based HMA, Prodas (by Beardow Adams, Milton Keynes, United Kingdom), a copolymer of PP and polyethylene. The chemical, thermal and mechanical properties of the adhesive were studied in a previous work. 20 The impact properties of this modified adhesive are presented in Ciardiello et al. 21 The modified adhesive was prepared by using a hot plate for melting the neat adhesive and by adding 5% weight of iron-based particles with different size. The sizes of the iron particles used in this work are 450 µm, 60 µm and 1–6 µm. The manufacturer of these particles is Good Fellow (Goodfellow Cambridge Ltd, Huntington, UK). Besides, iron oxide (Fe3O4) particles have a size lower than 50 nm and they are produced by Sigma-Aldrich (Saint Louis, United States).
The adherents used for the tests are prepreg (twill 2 × 2 type) E-Glass/Epoxy composite with a mass of 250 g/m2 and resin mass content of 36.5%. Their mechanical properties, material characterization and failure analysis are reported in Herrington and Doucet 22 and Beyene et al. 23 The material supplier is Umeco (UK). The matrix is made with an epoxy resin developed for the automotive sector.22,23 Fourteen prepreg layers were stacked together in order to obtain the laminate with the desired lay-up of 0/90.14. The adherents used for the experimental tests were 100 mm long with cross-section 25 × 2.5 mm.
Following a procedure commonly adopted in the literature for HMAs,7,10,17,18,20 pellets are melted together at 190 ℃, using a hot plate. At 190 ℃, the viscosity of the adhesive is low enough to easily mix the particles into the adhesive by mean of a glass rod for 10 min. The iron and iron oxide particles are added gradually and mixed together with the adhesive.
Mechanical tests were carried out on the SLJ. The geometry of the SLJ is shown in Figure 1.
Single lap joint specimen.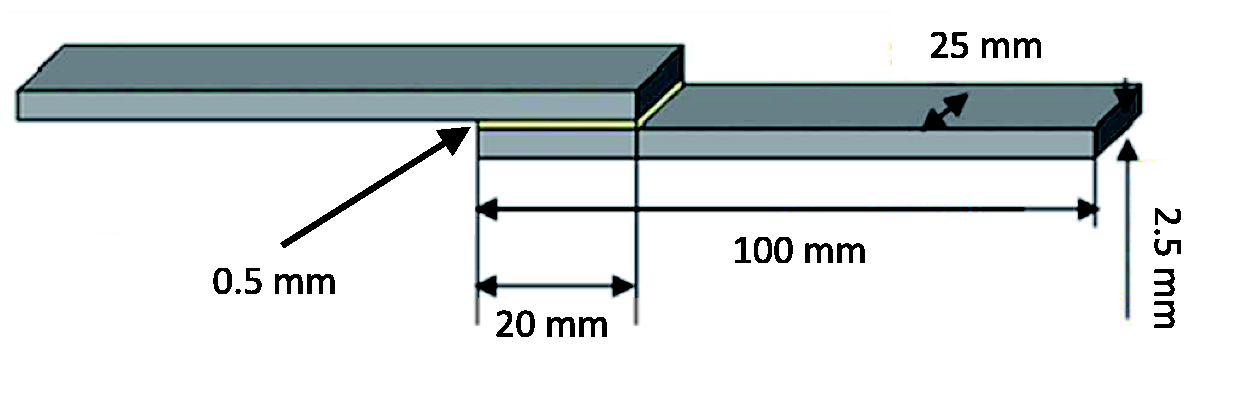
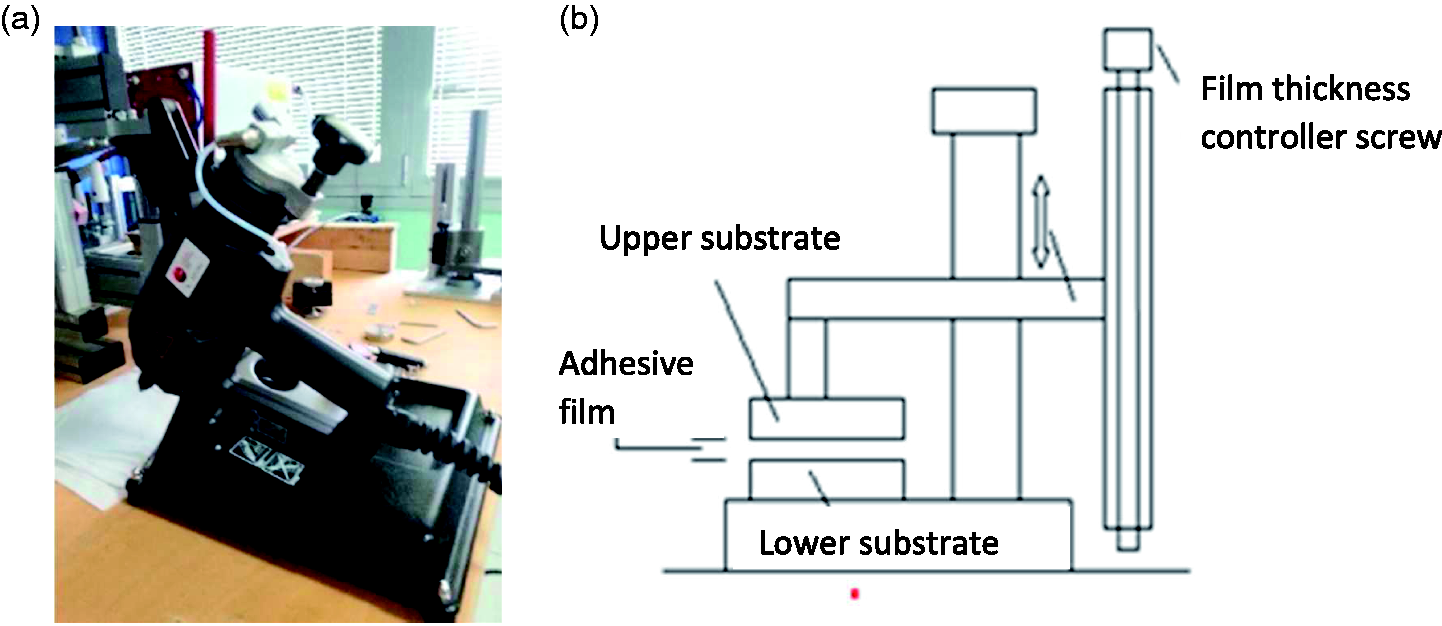
Joint preparation was performed with a hot-melt gun (Figure 2(a)) and an assembly device (Figure 2(b)) which permits to control the thickness of the adhesive joint. Nominal thickness of the adhesive layer is 0.5 mm. As shown in Figure 2(b), a film-thickness controller screw was used to fix the thickness of the adhesive layer at the desired value. Firstly, the adherent (lower substrate in Figure 2(b)) is fixed on the lower base of the assembly device. Then, the HMA at high temperature is uniformly spread over the lower substrate by means of the hot melt gun. An amount of adhesive larger than necessary was used to ensure that the overlap of the lower substrate was completely covered. Then, the upper adherent (upper substrate in Figure 2(b)) was placed on the still melted adhesive. In order to eliminate the adhesive in excess, a weight was placed on the support of the upper adherent. This procedure squeezed out the adhesive in excess until the required adhesive thickness is reached. Tabs were joined to the extremities of the joints, in order to prevent misalignment during the test.
Instrumentation used for the joint preparation: (a) hot melt gun; (b) assembly device.
20
In order to choose a right surface preparation, a preliminary analysis was conducted. Three different surface preparations were tried: degreasing with acetone and hand abrading the surfaces with two different abrasive sandpaper grits, P150 and P50, (and then cleaning with acetone). The P150 grit (lower particles size) gave higher failure load, even though it was just slightly higher than P50, 2%. On the other hand, joints prepared by simply cleaning the substrates with acetone displayed a reduction of the peak load of 12% compared to substrates prepared by abrading with P150. These results are in accordance with Kim et al. 24 Three replications were tested for each treatment. For these reasons, all the surfaces of the specimens used in the experiments were prepared using the grit P150.
Mechanical tests were conducted at a constant displacement rate of 100 mm/min, according to the FCA (Fiat Chrysler Automobile) standard on the hot-melt ahdesive, using an Instron 8801 servo-hydraulic machine. This configuration was used also in literature.7,10,17,20 At least, three joints were tested for each concentration of nanoparticles.
The evaluation of the separation of the SLJs using electro-magnetic induction coupled with iron and iron oxide particles was performed centring the SLJ in a circular copper coil of an inductor, as shown in Figure 3.
Configuration of the induction heating process.
In this configuration, the only part sensitive to the electro-magnetic induction is the nanomodified adhesive because of the metallic particles. The adhesives temperatures were controlled using an infrared instrument Testo 845 (by Testo Spa, Settimo Milanese, Italy). The infrared pointer was pointed on the edge of the bondline of the SLJ. The degree of the thermometer emission was set before the analysis using the manufacturer datasheet. On the right part of Figure 3, the test configuration is shown. For each test, a mass of 0.5 kg was applied to the SLJ in order to submit the joint to a constant load and cause joint separation (by part sliding) when joint adhesive reaches its melting temperature. The time to reach the separation (if it occurs) of the joint was measured. The inductor used for this analysis is Heasyheat by Ambrell, with a maximum power of 10 kW and a frequency range from 10 to 400 kHz. The value of the current was set at 550 A and the value of the frequency of the coil used for this analysis was 275 kHz. The coil used for this analysis is shown in Figure 3. This coil is very useful for preliminary analysis.
Furthermore, tests on the bulk sample of iron and iron oxide particles were performed in order to have a comparison with the increase of the modified adhesives. This test has been conducted using the same values of the electro-magnetic field used for the modified HMA. This test was performed filling a closed small glass container in order to avoid dispersion of the particles in the work environment, as shown in Figure 4. The use of this container was necessary since the metallic particles are attracted by the electro-magnetic field. Figure 4 shows the position of the particles under induction heating. The particles, at the beginning of the test were on the bottom of the container. Once the electro-magnetic field was applied they align on the wall of the container, as shown in the figure. For this reason, the temperature was measured pointing the infrared pointer on one line of the aligned particles.
Position of the particles during the test.
Scanning electron microscope (SEM) analysis were carried out with a Carl-Zeiss EVO50. The electronic high tension used was 20 kV. In order to obtain the best resolution, secondary electron emission signal was used for the Fe3O4 particles and the back scatter signal was used for the Fe particles. The specimens were properly coated with gold in order to have better images.
SEM analysis were also used to evaluate the average size of the particles, since for iron particles of 60 µm and 450 µm, the manufacturer just reported the maximum values of particle size. The average size of the biggest particles is 270 µm with a diameter range between 190 µm and 450 µm. Besides, the average particles size of the iron with a nominal particles size of 60 µm has an average size of 49 µm, from 41 µm to 60 µm. On the other hand, the particles sizes of the other particles have sizes in accordance with the manufacturer data sheets. All the particles used in this work have approximatively a spherical shape.
Results
SLJ tests
Figure 5 displays representative load–displacement curves of unmodified and modified HMA (5% wt). The mechanical performance of the SLJ tests are presented on the same diagram for both the Fe particles (HMA + 450 µm, HMA + 60 µm and 1–6 µm) and the Fe3O4 (HMA + 50 nm that have a particle size smaller than 50 nm). From this point on, HMA refers to unmodified hot-melt adhesive. HMA + 450 µm, HMA + 60 µm and HMA + 1-6 µm refers to the adhesive modified with iron particles that have average diameter of 450 µm, 60 µm and 1–6 µm, respectively. HMA + 50 nm refer to iron oxide particles that have a particle size smaller than 50 nm. Results show that the failure load decreases when the particles size is higher. On the other hand, the modified adhesives present a more ductile behaviour as can be seen by the larger tails of the SLJ test of the modified adhesives.
Representative load–displacement curves of the SLJ test.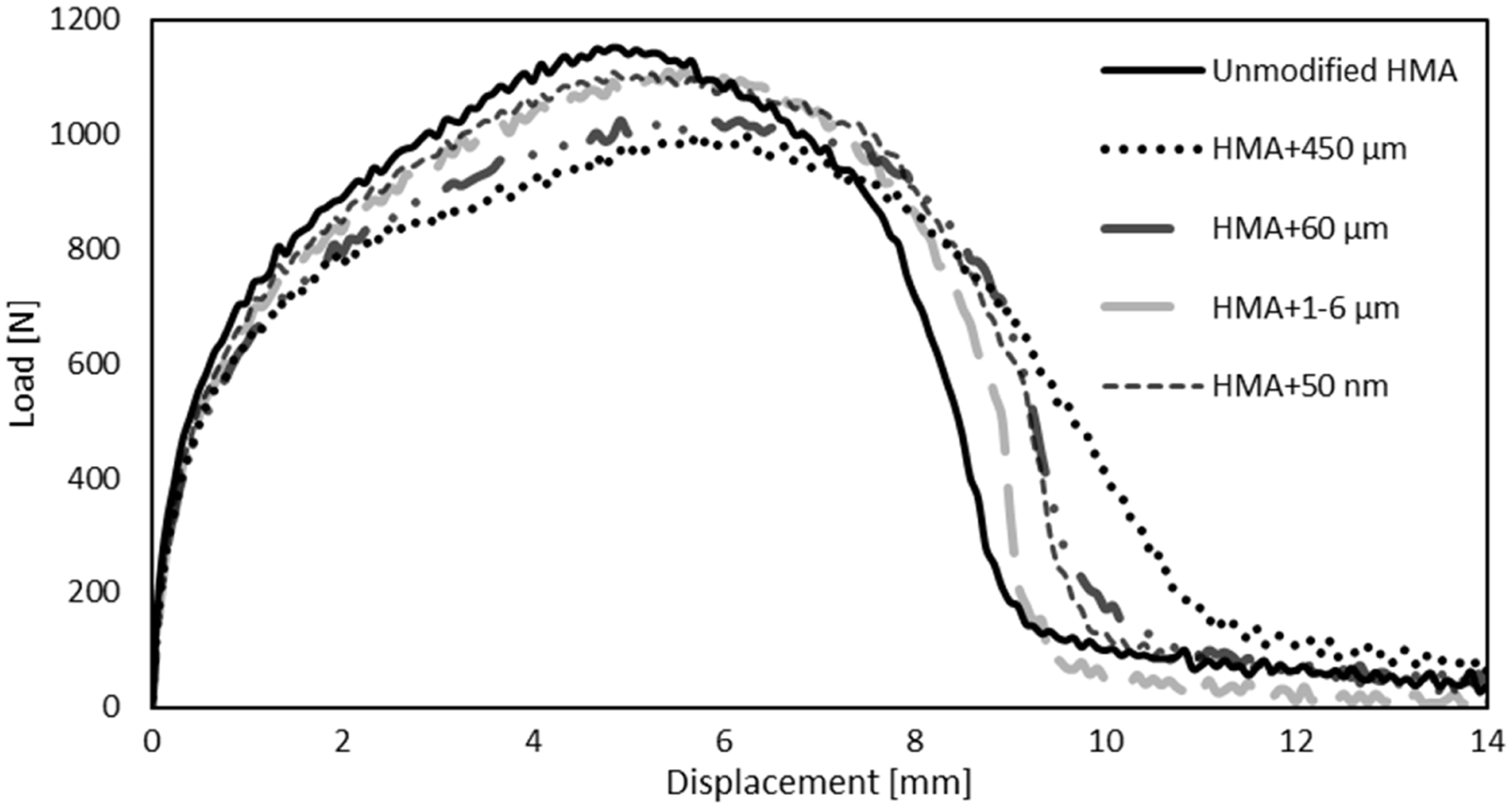
The chart of Figure 6 shows the average shear strength and displacements of the single SLJ tests together with the relative bars errors. The displacement values were measured when the load decreased down to 100 N since this is the load at which all the curves drop. This value has been preferred to the displacement corresponding to the load of zero because the adhesive had very high elongation, in some cases, and so the results can be affected by this behaviour. This diagram confirms that the average shear strength decreases of 8% for the HMA modified with the 450 µm particles and 7% for the HMA modified with the 60 µm particles. On the other hand, the values of the unmodified and modified HMA, with a particle dimension of 1–6 µm, are very close. The same can be said for the Fe3O4 with a particle size less than 50 nm: average shear strength values are very close.
Average shear strength and maximum displacement.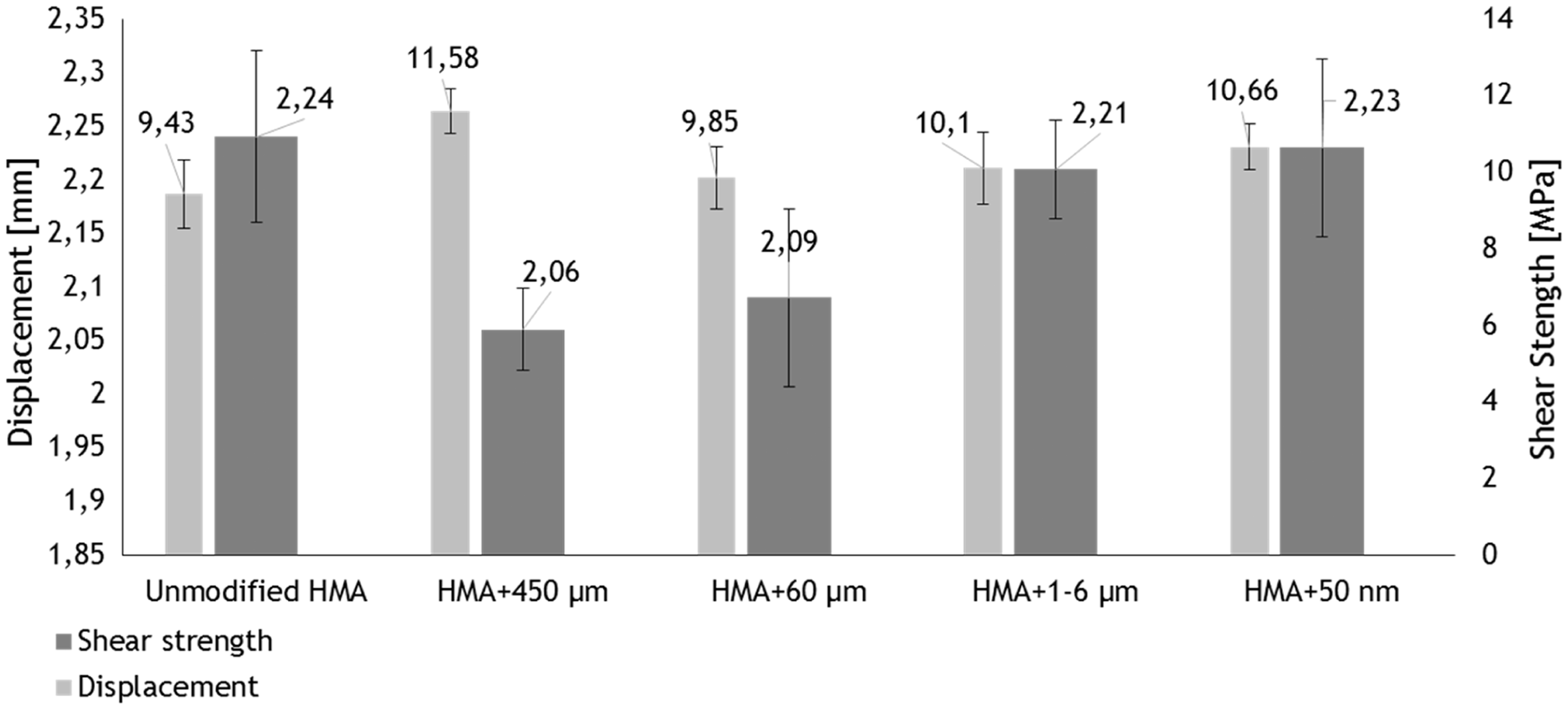
The displacements values (Figure 6) show that the extensions of the modified adhesives are higher. The displacement increases of 22% for HMA + 450 µm, 4% for HMA + 60 µm, 7% for HMA + 1-6 µm and 13% for HMA + 50 nm over the pristine adhesive.
In general, there is a decrease of the maximum shear strength for the adhesives modified with bigger particles, while the load increases for smaller diameter particles. The average elongation of the modified adhesives increases compared to the unmodified HMA in all samples.
Finally, the fracture failure mode was analysed by visual inspection. The failure mode was both adhesive and cohesive for all the specimens. In fact, Figure 7 displays that the cohesive zone is larger for the basic HMA and it is slightly lower for the modified HMA. It seems that the presence of iron and iron oxide reduces the cohesive area and this causes a lower maximum strength. It is worth to highlight that there were no deformations on the specimens and the rotation around the edge of the overlap was insignificant during the tests.
Failure modes for the unmodified and modified adhesive.
Experimental results of the induction heating process on the modified adhesives
Figure 8 shows the temperature–time curves for the modified HMA under electro-magnetic induction. The temperature was measured on the edge of the SLJ. The diagram shows that the rapidity of the heating process increases with the increase of the particles size. The maximum temperature reached after 240 s for the HMA + 450 µm, HMA + 60 µm and HMA + 1–6 µm are 43 ℃, 53 ℃ and 68 ℃, respectively. This could be due to the eddy current losses that are proportional to the area of the considered particles. On the other hand, the temperature of the iron oxide particles, HMA + 50 nm, increases with a completely different trend: it reaches around 210 ℃ in 60 s. This difference could be due to the magnetic behaviour of the iron oxide particles that increases the particles losses and consequently the temperature of the adhesive.
Temperature profiles of the HMA modified with iron and iron oxide particles under induction.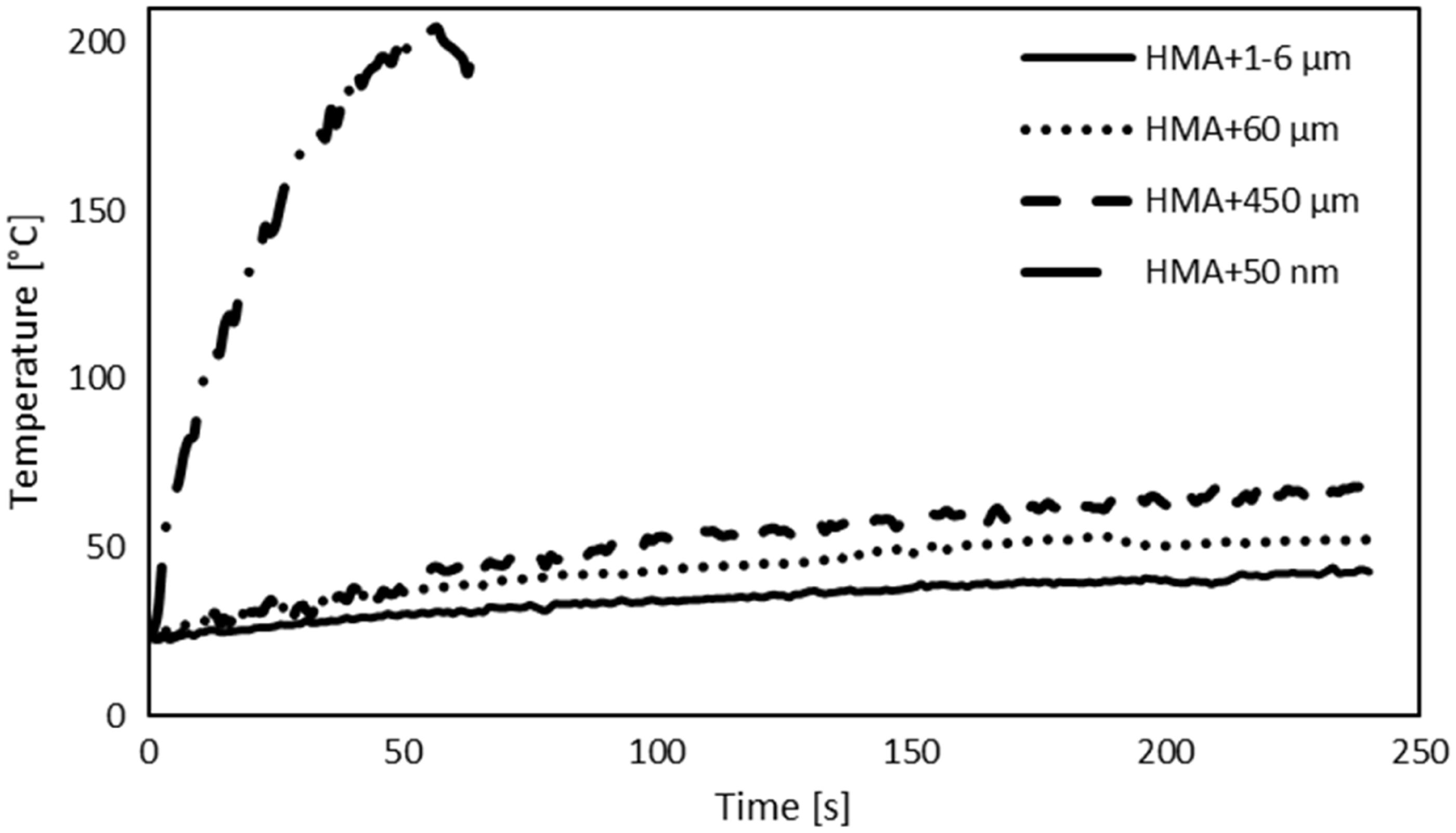
Separation tests were tried according to the configuration presented in ‘Materials and methods’ section. No separation occurred with the adhesive modified with iron particles. Figure 8 evidences that these modified adhesives had a maximum temperature of 78 ℃, that is very low compared to the melting temperature of this HMA, 160 ℃. The adhesive modified with iron oxide, HMA + 50 nm, reached the melting point of the adhesive and separation of the joint occurred after 105 s with a limited scatter of 2 s.
Figure 9 displays the temperature–time curves for the bulk samples of iron and iron oxide particles under electro-magnetic induction.
Temperature profiles of the iron and iron oxide particles under induction.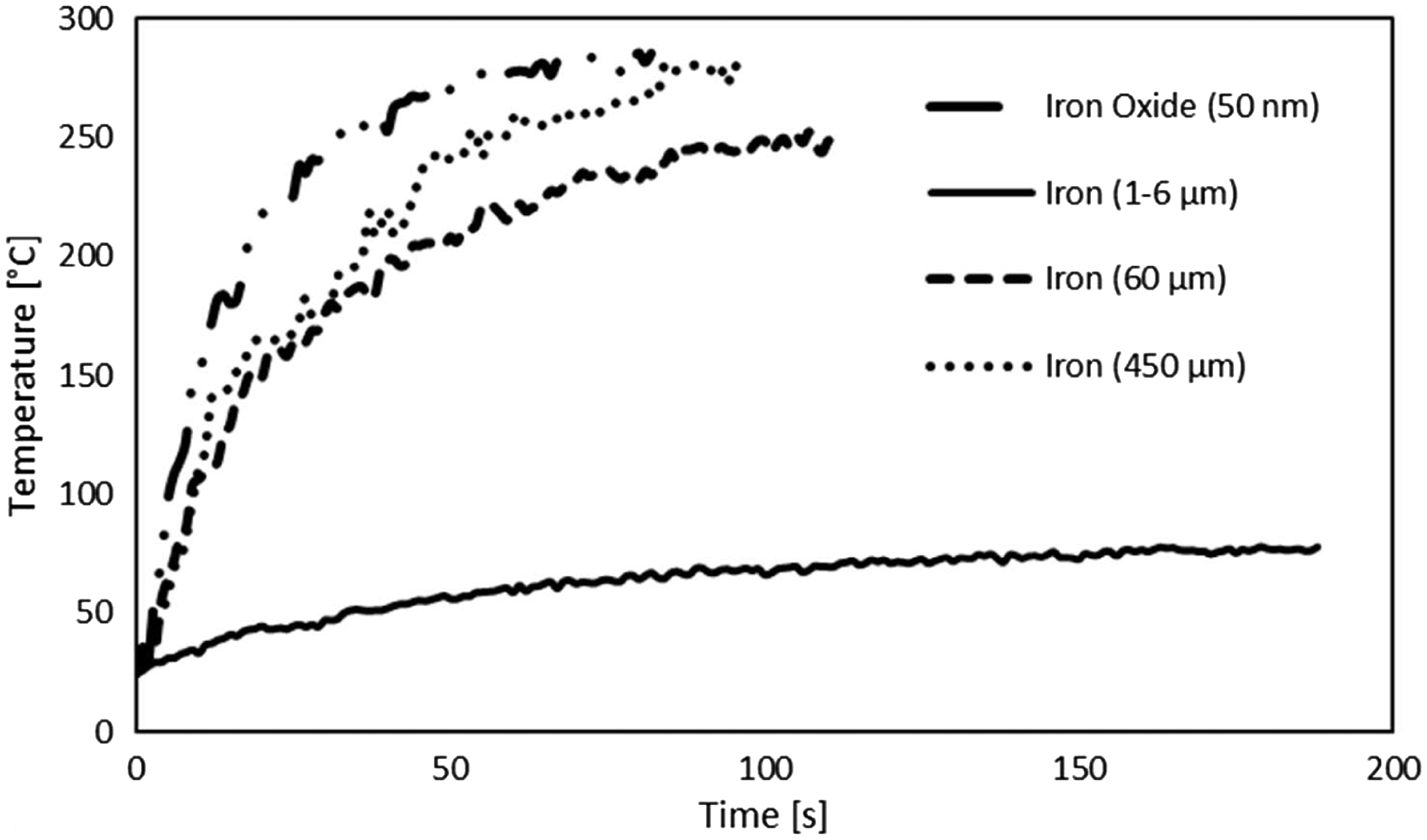
As expected, the temperatures of the bulk samples were higher than the modified HMA due to the different thermal conditions. In fact, in the last case, particles touch each other and so they increase rapidly their temperature. On the other hand, particles embedded in the matrix reached lower temperatures since they have to transmit the heat to the adhesive. The iron oxide particles reached a temperature of 287 ℃ after 75 s. Iron particles with 450 µm diameter heated up to 280 ℃ in 100 s. Iron particles with 60 µm diameter reached a maximum temperature 252 ℃ in 115 s. Finally, the iron with the lower particle diameter, 1–6 µm, reached the lower temperature of 78 ℃ after 190 s. It is worth to note that iron particles with an average diameter of 60 and 450 µm reached a temperature much higher than the melting temperature of the HMA but the adhesive modified with these two particles did not reach the melting point. This can be attributed to the lower number of particles into the adhesive matrix, since higher are the particle diameters lower is the number of the particles into the HMA for equal weight concentration. Thus, when we have particles of 60 and 450 µm, the low thermal conductivity of the adhesive together with the low number of particles in the adhesive impedes the increase of the temperature up to the melting point. The number of the particles inside the matrix is so relevant since particles represent the thermal source for this application. On the other hand, the temperature of the iron oxide particles and iron with lower particles size, 1–6 µm, is closer to the maximum values of the modified HMA.
In a previous study, 20 it has been showed that the HMA used in this paper is suitable for PP substrates. In this work, it was shown that this hot-melt adhesive can be used also for epoxy-based substrates. It was demonstrated that higher particle sizes gave worse results in terms of mechanical performances. The maximum load decreases by 8% for the adhesive modified with a particle size of 450 µm and 7% for the adhesive modified with particles of 60 µm. The HMAs modified with lower particle sizes, iron with 1–6 µm and 50 nm give better results in terms of maximum load, that are very close to the unmodified adhesive.
Finally, the adhesive modified with the iron particles was not able to reach the melting temperature of the adhesive, while the HMA modified with the iron oxide reached the melting temperature in a relatively rapid time. This could be due to the different physical behaviours of the two particles. In the case of the iron oxide (magnetite), the magnetic behaviour increases the losses and this causes a rapid increase of the adhesive temperature.
SEM analysis
The SEM analysis was used to study the dispersion of the Fe and Fe3O4 particles inside the adhesive matrix. The presence of the Fe particles with a diameter of 450 µm can be also evaluated by visual inspection since they are bigger. The visual inspection showed a homogenous distribution of the particles inside the matrix. SEM analysis showed the presence of few Fe particles for the HMA + 450 µm and HMA + 60 µm on the adhesive surface. As the particles density is constant, adding the same weight of particles their number will be different: much higher for the smaller ones. For these reason, SEM analysis was able to display just few particles when HMA + 450 µm and HMA + 60 µm were analysed. Figure 10 shows the Fe particles of 450 µm in the adhesive matrix. The higher magnification on the right side, displays the interaction between particles and matrix that seems to be very good.
SEM analysis of HMA + 450 µm. SEM: scanning electron microscope.
Figure 11 displays the SEM analysis of HMA + 60 µm. Also, in this case, there are just few particles visible in the adhesive that can be located as the white spots in the black matrix. The lower magnification image on the left side shows a small part of the particles, since they are well embedded in the adhesive. The higher magnification image, on the right side, displays a bigger Fe particle.
SEM analysis of HMA + 60 µm. SEM: scanning electron microscope.
Figure 12 shows the particles with a diameter of 1–6 µm. Again, the Fe particles are recognisable by the white spots in the matrix. The number of the particles increases, since the size is smaller. Particles seem to be well dispersed in the adhesive matrix.
SEM analysis of HMA + 1–6 µm. SEM: scanning electron microscope.
Figure 13 shows the dispersion of Fe3O4 particles inside the HMA. The little white spots are the nanoparticles. The distribution seems to be uniform, the dimension of nanoparticles seems to be in accordance with the data sheet of the manufacturer.
SEM analysis of HMA + 50 nm. SEM: scanning electron microscope.
Conclusion
The aim of this work was to evaluate the mechanical behaviour of hot-melt modified adhesive when epoxy-based substrates are used. Furthermore, to understand whether iron and iron oxide nanoparticles can be used in order to separate substrates for repairing and recycling. The motivation of this study is due to the increase of the use of composites in automotive and aerospace applications and to the necessity to replace or repair in case of need. In previous studies, 20 it was demonstrated that this adhesive give good results for PP substrates. In this work, it was shown that this adhesive gives good mechanical performances also for epoxy-based substrates.
Primarily, the effect of the particles diameters was evaluated using the iron particles and comparing the results with the pristine adhesive. The HMA modified with the particles with higher diameters gave worse results in term of peak load. The reduction of the peak load was 8% for the adhesive modified with particles of 450 µm and 7% for the HMA modified with particles of 60 µm. The adoption of the smallest iron particles gave the same mechanical results of the pristine adhesive. Therefore, the load peak decreases with the increase of the particles size. On the other hand, iron particles did not allow the separation of the adhesive since they were not able to reach the melting temperature of the HMA.
Electro-magnetic tests conducted on particles alone, helped to understand that bigger particles are able to overcome the melting temperature of the adhesive but hot-melt adhesives modified with these particles are not able to reach the melting. This is because for equal weight, the low thermal conductivity of the adhesive together with the low number of particles in the adhesive impedes the increase of the temperature up to the melting point. The number of the particles inside the matrix is very important since particles represent the thermal source for this application.
The use of the iron oxide gave better results in term of heating and mechanical properties. This kind of particles embedded in the hot-melt adhesive allows the separation of the joint with an average time of 105 s. The mechanical properties are similar to the pristine adhesive in terms of peak load while the elongation is higher. This represents a good result, given that this adhesive, already used in automotive applications, can give the same mechanical characteristic introducing the possibility to separate the joint when needed for recycling, repairing or reuse of expensive materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
