Abstract
Q235 steel is a low-cost material widely applied in modern industrial constructions. However, electrochemical degradation is the main drawback for its applications in chloride-enriched environments. To address this problem, NiCrAlMo and NiCrAlMo + 2 wt% nano-CeO2 composite coatings were laser-cladded on Q235 steel substrates. Characterization methods were used to investigate the microstructures, corrosion behaviors, and electrochemical mechanisms of the obtained coatings. The results showed that adding nano-CeO2 is conductive for removing residual gas pores and for refinement of grains, effectively improving the passive and electrochemical properties of the NiCrAlMo coating in both chloride-enriched solutions. Furthermore, the NiCrAlMoCeO2 composite coating exhibits a significantly reduced icorr of 0.4919 µA cm2 and an increased resistance (Rp) of 175,310 Ω cm2 in a neutral solution, demonstrating the good anti-corrosion properties for its application in marine engineering structures.
Keywords
Introduction
Q235 steel is a widely applied material in offshore platforms, ship structures, and pipeline transportation. However, it is susceptible to severe electrochemical attacks when serving in corrosive environments that contain chloride ions, which significantly accelerates the degradation rate. 1 To date, the electrochemical destruction of Q235 steel has caused significant economic loss and safety casualties. Hence, implementing long-term and heavy corrosion prevention is urgent.
Recently, Ni-based coatings have been widely used for preventing corrosion damage on substrates, Tang et al. 2 fabricated a Ni-Co coating by electroplating, which effectively improved the corrosion resistance of X65 steel in a sweet (CO2) corrosion environment. Chen et al. 3 prepared a Ni-Cu-P coating by electroless plating, which showed better corrosion performance than low-carbon steel in a 3.5 wt% NaCl solution. Jiménez et al. 4 demonstrated that a thermal-flame-sprayed Ni-Tec coating had a lower corrosion current and more positive corrosion potential than gray cast iron. Sichani et al. 5 showed that a plasma-sprayedNi-Ti-Al coating exhibited good protection action for AISI 1045 steel.
Despite the many successful cases of Ni-based coatings fabricated on different substrates, it is still difficult to obtain the high anti-corrosion required for Q235 steel in a marine environment. For example, coatings fabricated by electroless plating or electroplating are relatively thin, and unpredictable spallation caused by the pressure of the corrosion products occurs when the substrate is eroded.6,7 Coatings deposited by conventional thermal spraying methods commonly contain minor imperfections of lamellar structures, un-melted and oxide particles, which causes galvanic corrosion on the substrate. 8
Laser-cladding (LC) is an alternative coating approach that may provide solution for the abovementioned problems. Powder materials transported by carrier gas can be melted completely by a high-energy beam, producing a thick coating (thickness varying from hundreds of microns to many centimeters).7,9 The general modified characteristics of the coating can be improved by various particle enhancements.10,11 Zhao et al. 12 showed that stable corrosion-resistant passive film can be formed on electroless plated Ni-P coating by incorporating Mo that have with increased hardness, good thermal conductivity, and superior anti-corrosion resistance. 12 Laszczyńska et al. 13 developed passive films using electrodeposited Ni-Mo alloy coatings enriched with Mo. Mosayebi et al. 14 obtained high polarization resistance of Ni-Mo electroless plated coatings in chloride media. However, to date, few studies have focused on the electrochemical investigation of NiCrAlMo coatings fabricated by LC.
A closer look into the properties of LC coatings is necessary to avoid potential microstructure defects 11 originating from thermal stress and differences in physical and chemical properties of the substrate and the coating materials. Introducing rare-earth oxides of nano-CeO2 is one solution that can improve the fluidity of the molten pool and optimize the coating quality. Gao et al. 15 added the CeO2 in a Ni60-cladding layer, which considerably decreased the number of oxides, pores, and cracks in the composite coating. Wang et al. 16 also improved the coating microstructure and properties by inhibiting cracks and porosities by adding nano-CeO2. Gong et al. 17 reduced the crack sensitivity and wear rate of Ni60A coatings on a TC4 substrate.
Contemporary studies have mainly focused on the microstructure, corrosion, or wear properties of Ni-based coatings with separate deposition of metallic Mo and ceramic CeO2 reinforcements. The effects of simultaneously adding these on the coating electrochemical performance have not been investigated extensively. In this study, NiCrAlMo and NiCrAlMo + 2 wt% nano-CeO2 coatings were successfully cladded on Q235 steel using optimized laser fabrication parameters. The study aimed to investigate the effect of incorporation of nano-CeO2 nanoparticles on the microstructures, corrosion behaviors, and electrochemical mechanisms of Mo-reinforced NiCrAl coating in a 3.5 wt% NaCl solution and a 1 M HCl solution. Furthermore, practical guidelines for the development and industrial application of protective nano-CeO2/NiCrAlMo composite coatings in a marine engineering structure are provided.
Experimental procedures
Coatings preparation
The Q235 steel substrate was supplied by Tianjin Jinshun Steel Co., Ltd and processed into dimensions of 60 mm × 30 mm × 10 mm by wire cutting. Its chemical compositions (wt, %), mechanical, and physical properties are listed in Table 1. The substrate surface was abraded with different meshes of SiC papers, degreased with acetone, and sandblasted by Al2O3 particles before LC. A mixture of NiCrAl and Mo powder was adopted as the starting powder, and the CeO2 (2 wt%) nano-powder (99.99%) was further added as a composite material; all materials were commercially available from Xinzao Alloy Material Co. Ltd. To ensure uniform coating deposition, different powder particles were preliminarily dry-ball milled (ball diameter of 6 mm) at 400 rpm for 3 h, and then dried in an 80°C box-type resistance furnace for 2 h to eliminate moisture.
The chemical compositions, mechanical, and physical properties of Q235 carbon steel.
LC process was conducted using a continuous-wave diode laser system, where the coating powder was transported through its coaxially equipped powder feeding system using high-purity carrier Ar gas. The laser fabrication parameters were power = 1100 W, scanning speed = 3 mm/s, powder feeding speed = 10 g/min, and overlap = 50%, selected based on findings of previous studies18–20 and a series of preliminary experiments.
Microstructure analysis
After LC preparation, the different samples were cut into squares with an area of 10 mm × 10 mm. The surface and cross-section were subjected to mechanical grinding and mirror polishing. The grain size, morphologies, and chemical compositions were then characterized using microscope, scanning electron microscopy (SEM) coupled with energy dispersive spectrometry (EDS), and the dilution (λ) was calculated according to equation (1)21,22:

where s1 and H are the melting area and height of the cladded coating, s2 and h are the melting area and depth of the fusion substrate, respectively. The coating phases were recorded by D/Max-2500 type X-ray diffraction (XRD) using Cu-Kα radiation within 20° -90° identified by MDI-Jade software based on standard PDF cards. Coating microhardness was measured using an HV-1000 type Vickers hardness tester with a load of 500g and a dwell time of 10s to obtain a visual and stable indentation, and the final result was calculated from the average value of 10 different locations on the coating surface.
Electrochemical measurements
The corrosion behavior and electrochemical properties of the substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings in a 3.5 wt% NaCl solution and 1 M HCl solution were evaluated using a CHI660E electrochemical workstation at room temperature. First, stable-state open circuit potentials (OCPs) of the samples were obtained by immersion in corrosion solutions for 30 min; the corresponding EIS data were collected using a low alternating current (AC) amplitude, and the final potentiodynamic polarization (PDP) curves were recorded at a sensitivity of 1 × 10−0.002 repeated in triplicate to ensure accuracy and repeatability of the electrochemical results.
The OCP curves were expressed as a function of the immersion time. The corrosion potential (Ecorr) and current density (icorr) were obtained from PDP curves by the Tafel extrapolation to determine the corrosion behaviors of samples in both solutions. The collected EIS data were represented as plots of Nyquist, impedance modulus |Z|, and phase angle, where the total |Z| was defined by the real part Z’ and imaginary part Z” in the Nyquist diagram using the following equation:

Results and discussion
Characterization of powder feedstock
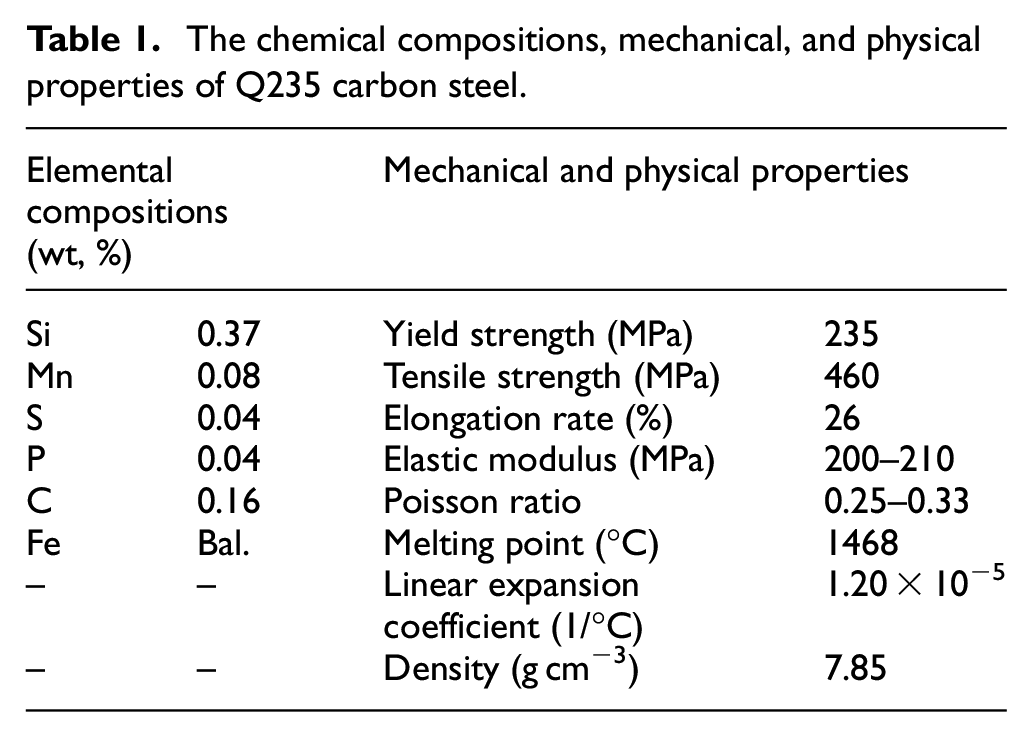
The morphology and EDS analysis of NiCrAlMo powder are presented in Figure 1(a). The diameters of raw NiCrAlMo powder were 45–105 μm, and they were either spherical or elliptical, which ensured good flow and excellent feeding stability for coating deposition. 23 The EDS results demonstrated that the powder was rich in elemental Ni, Cr, and Mo, which constituted 97.67% of the total mass fraction, whereas Al accounted for only 2.33%. No other impurities were detected, indicating the high purity of the original powder.
Morphology and composition of different raw materials for LC: (a) NiCrAlMo powder and (b) NiCrAlMoCeO2 composite powder.
The morphology and EDS analysis of NiCrAlMoCeO2 powder are shown in Figure 1(b). The composite powder also had a spherical or elliptical morphology, while the particle surfaces were relatively coarse due to the collision deformation and adherence of CeO2 nanoparticles after ball mixing. Its content on the Mo particles was characterized, which indicated that the powder surface was primarily composed of elemental Mo, Ce, and O, along with small amounts of Ni, Cr, and Al, demonstrating that the milling process did not change the composition of the powder.
Microstructure characteristics of obtained coatings
XRD, grain microstructure, and microhardness analysis
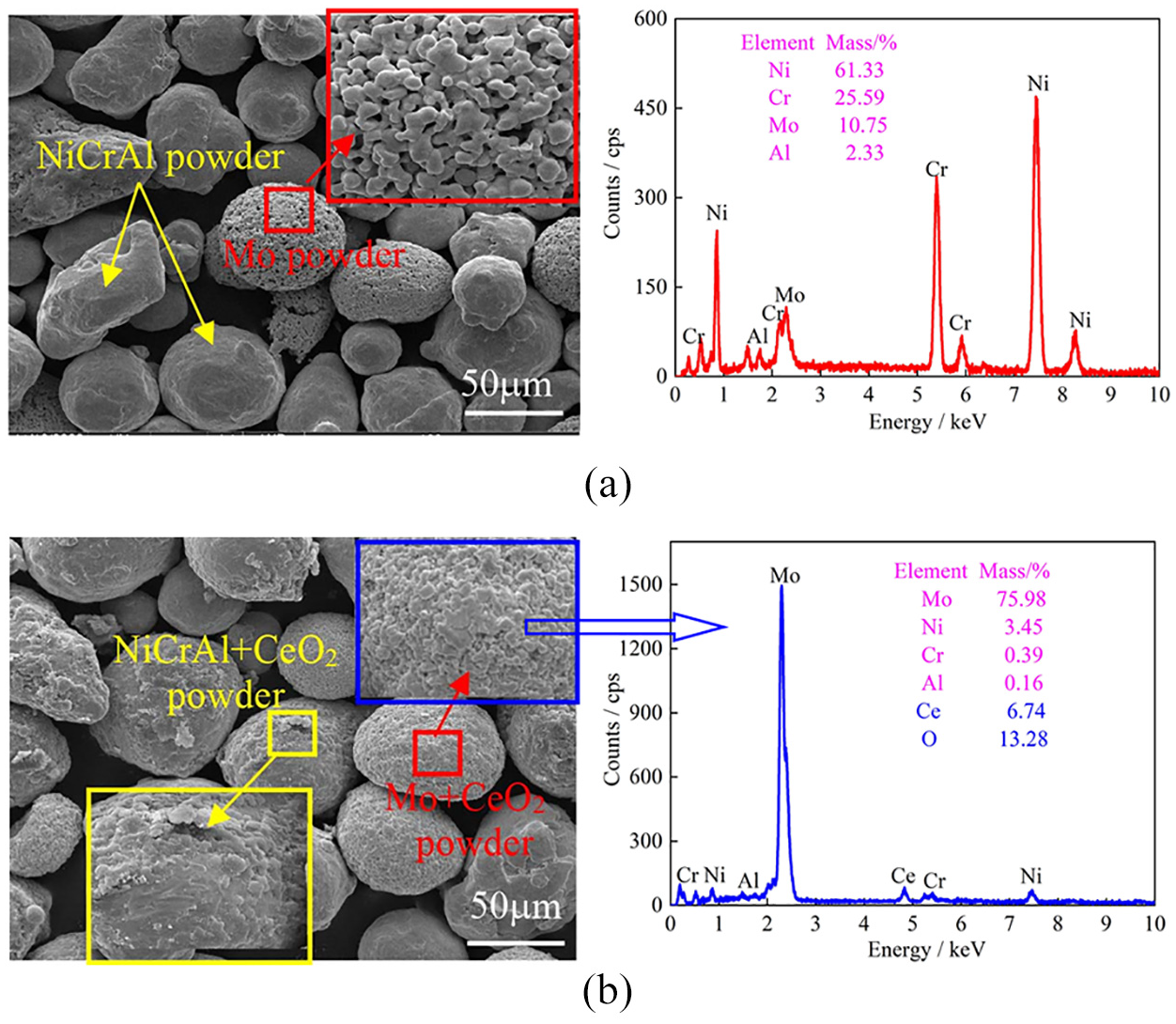
The XRD patterns of the laser-cladded NiCrAlMo and NiCrAlMoCeO2 coatings are shown in Figure 2(a). It was illustrated that the NiCrAlMo coating was composed of FCC and MoNi4, indicating that the Ni had a good atomic affinity with the other elements to form intermetallic compounds. After CeO2 incorporation, the phase constituents were similar, while its diffraction peak was absent, which was due to the additive amount being lower than 5 wt% in the coating.
Comparison on XRD, grain microstructure, and microhardness analysis of the obtained coatings: (a) XRD diffraction patterns, (b) partial enlarged peak, (c) grain size of NiCrAlMo coating, (d) grain size of NiCrAlMoCeO2 composite coating, and(e) Microhardness.
Meanwhile, both Ni-based coatings exhibited the (111) preferred orientation shown in Figure 2(b), and the grain size was evaluated by the Debye-Scherrer equation, which decreased from 9 to 5 µm after CeO2 incorporation shown in Figure 2(c) and (d). Due to the refinement and dispersion strengthening mechanisms provided by CeO2,24–26 the average microhardness of NiCrAlMoCeO2 composite coating was ∼1.5 times that of the NiCrAlMo coating, as shown in Figure 2(e).
Morphologies and elemental composition on coating surfaces
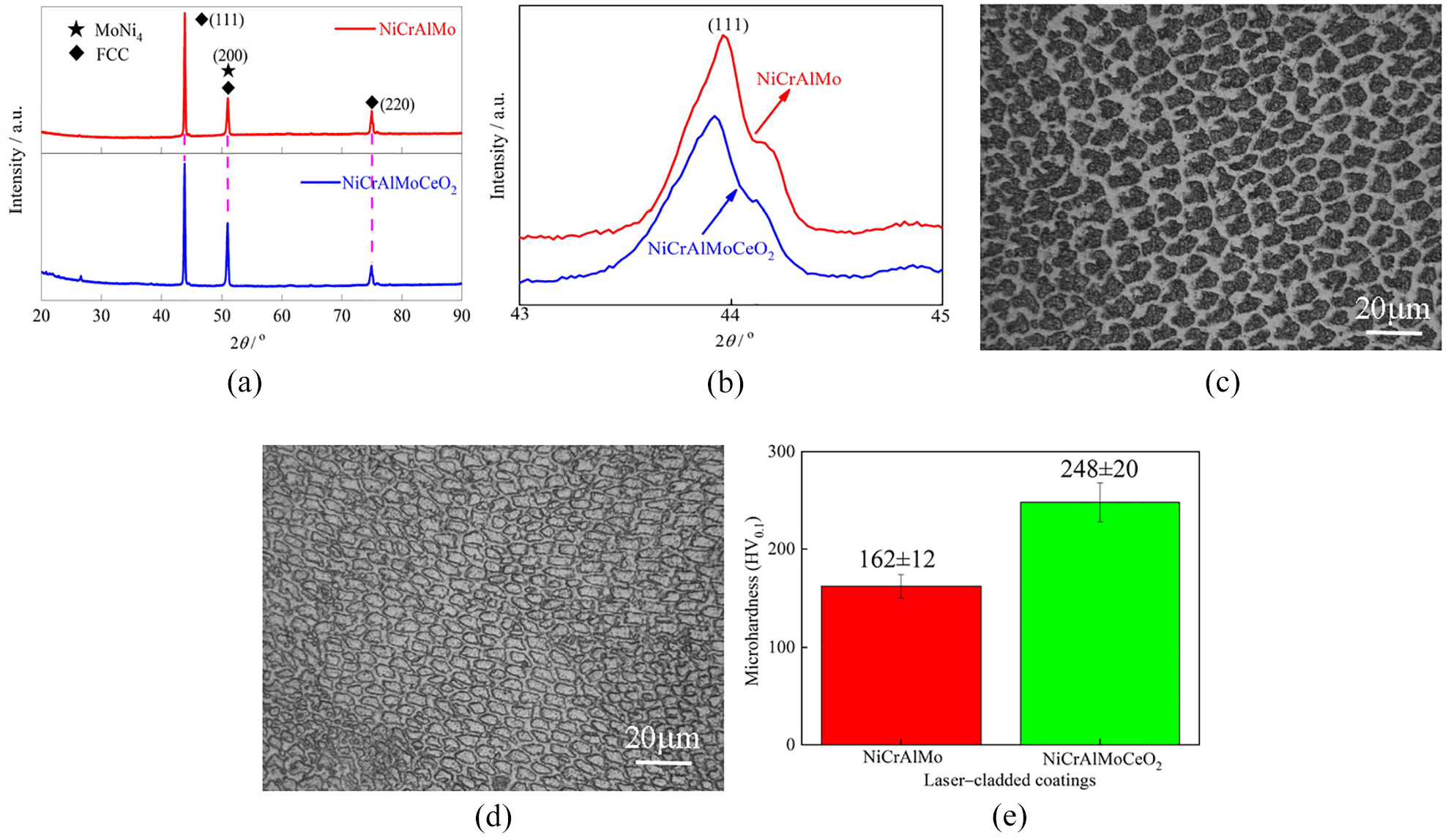
The morphology and EDS analysis of the laser-cladded NiCrAlMo coating surface are shown in Figure 3(a). There were no visible cracks, semi-molten particles, or burning loss traces, but the microstructure had gray and white contrast regions. The EDS results revealed that the Ni, Cr, Al, Mo, and Fe were the main constituents of gray area 1, while the local bright area 2 was identified as the Mo-rich zone due to its sharp increase in the concentration of 30.92 wt%. The Ni, Cr, Al, and Mo originated from the powder feedstock, while the Fe element was diluted from the substrate by the high-temperature laser melting and deviated from its nominal composition. Additionally, a few pores were observed as dark contours due to the CO, CO2, and other gases trapped on the coating. 27 During LC, the mobility of the molten-pool without a rare-earth element was usually poor, which led to the overflow rate of gases being slower than the coating solidification rate (104–106 K s−1) and sealed to form the pore defects. 21
Morphologies and EDS spectra of the obtained coating surfaces: (a) NiCrAlMo coating and (b) NiCrAlMoCeO2 composite coating.
The morphology and EDS analysis of laser-cladded NiCrAlMoCeO2 composite coating surface are shown in Figure 3(b). Compared to the previous coating surface, the pores and Mo-rich area were eliminated, indicating that the CeO2 incorporation had a positive effect of purifying and modifying the microstructure. 28 Explained by the fact that the CeO2 was conductive to improve the absorption rate of powder materials to laser irradiation energy, which was equivalent to increase the laser heat input for indirect coating heating. 21 As a result, the convection and stirring effect in the molten pool was promoted, 24 and the cooling and solidification time of liquefied molten metal was further prolonged, 27 which promoted the uniform distribution of coating elements, facilitated the gases and impurities to escape from the molten pool, and eventually led to the structure organization to be more uniform. Furthermore, the coating surface was mainly composed of elemental Ni, Cr, and Mo, which accounted for 93.81% of the total mass fraction indicated by the EDS results of area 4. The additional element Al remained low due to its low mass fraction in the powder, while the Fe content increased slightly similar to that on NiCrAlMo coating.
Dilution ratios, morphologies, and line scans on coating cross-sections
The total thickness of the fabricated NiCrAlMo and NiCrAlMoCeO2 coatings was ∼1 mm, while the dilution ratio calculated by equation (1) was respective 13.9% and 17.5%, respectively. The dilution rate of a coating between 10% and 20% is acceptable, and it was beneficial for forming strong bonds with the substrate and maintaining the required coating performance.
The cross-section morphology of the NiCrAlMo coating is shown in Figure 4(a). The coating was deposited homogeneously with a clear bonding line to the substrate, but a few pores were observed, which are ascribed to the trapped gas bubbles prior to the rapid coating solidification.21,29 Moreover, the coating elements Ni, Cr, and Mo had a higher content than Al and decreased gradually to the substrate with a diffusion layer thickness of ∼23 µm; in contrast, the Fe content clearly increased. It was previously shown that the gradient layers of mutual elements resulted from the convective diffusion between the coating and the substrate, which led to the formation of excellent metallurgical bonds.30,31
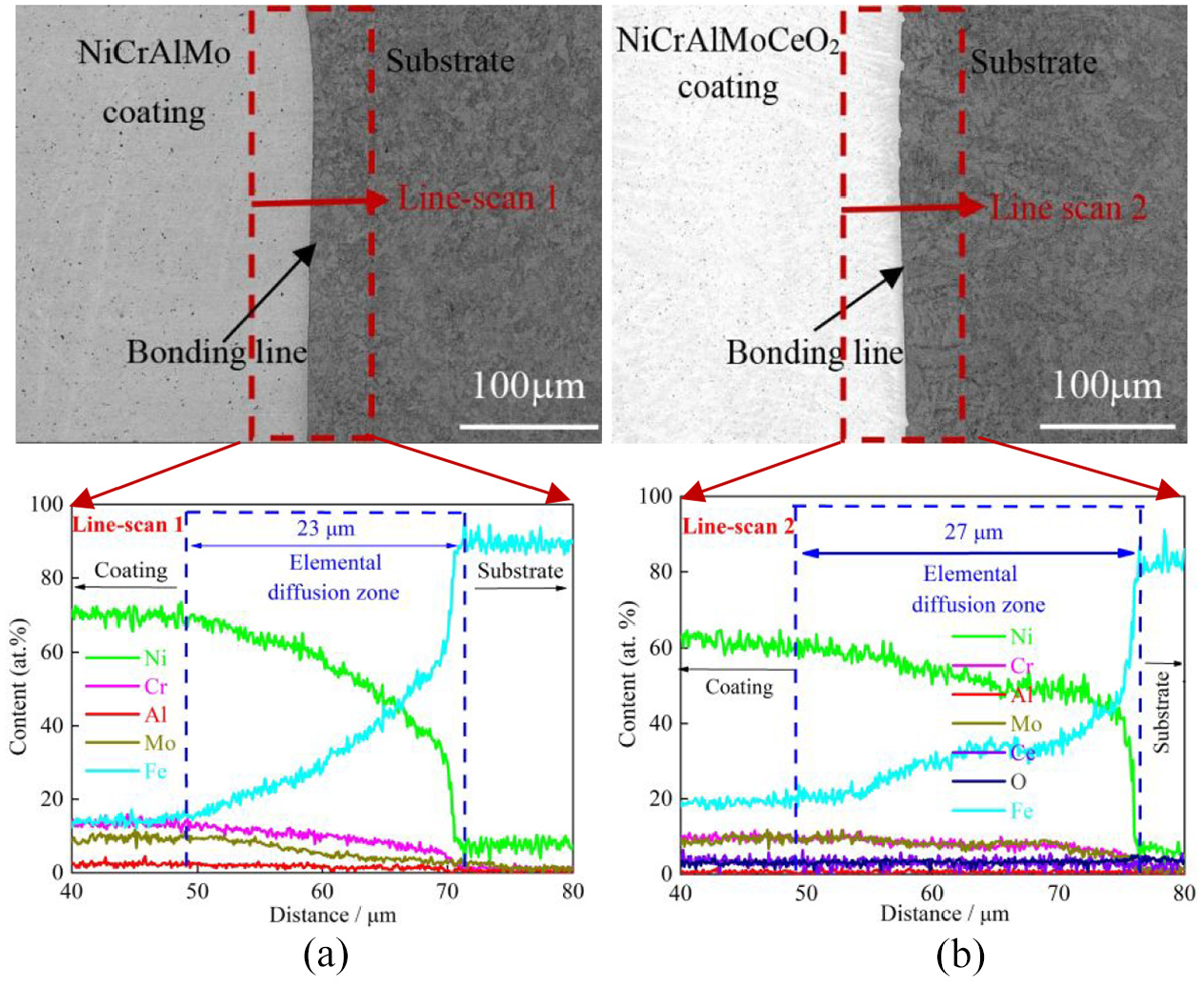
EDS line-scan across the obtained coating cross-sections: (a) NiCrAlMo coating and (b) NiCrAlMoCeO2 composite coating.
The cross-section morphology of the NiCrAl MoCeO2 composite coating is shown in Figure 4(b), which shows a good metallurgical bond without apparent pore defects. Compared to the elemental contents of the NiCrAlMo coating, Ni and Cr decreased, while Fe increased, presenting greater interfacial diffusion thickness of ∼27 µm. The increased element interdiffusion was also due to the indirect increase in laser heat input by CeO2, which not only enhanced the fluidity of the coating elements, but also enhanced the thermal impact of the coating droplets on the substrate.21,32 Consequently, the convection circulation center of the molten pool moved down, and the substrate melting depth increased, which allowed more Fe to enter into the molten pool, resulting in greater dilution than the NiCrAlMo coating. This was consistent with pore reduction on the coating surface, where the larger dilution resulted in a more liquid phase, which relatively decreased the solidification rate of the melt and provided more time for the floating of the gas bubbles. 21
Overall, the metallurgical bond was obtained for two types of laser-cladded Ni-based coatings due to the interfacial diffusion layer, while the CeO2 co-addition contributed to the elimination of surface pores, unevenness, and segregation by increasing the element flowability.
Corrosion behaviors
The corrosion behaviors of the substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings in a 3.5 wt% NaCl solution and 1 M HCl solution were investigated using OCP and PDP measurements. As depicted in Figure 5(a) and (b), the OCP of the substrate had the lowest values in both solutions, which demonstrated the active dissolution during whole immersion. After being covered with NiCrAlMo and NiCrAlMoCeO2 coatings, the OCP curves all had different degrees of positive shifts, clearly showing various passivation responses once immersion in the corrosion medium. For the NiCrAlMo coating, the potential decreased rapidly at initial immersion, indicating the quick dissolution of oxides and the resulting activation behavior in both solutions. However, the potential of the NiCrAlMoCeO2 composite coating increased with minor fluctuations, suggesting the occurrence of spontaneous passivation. Thereafter, the OCP of the NiCrAlMoCeO2 composite coating in the 3.5 wt% NaCl solution changed slightly, while that in the 1 M HCl solution, it gradually moved to the more negative position, Hence, better passivity stability was achieved in the neutral solution.
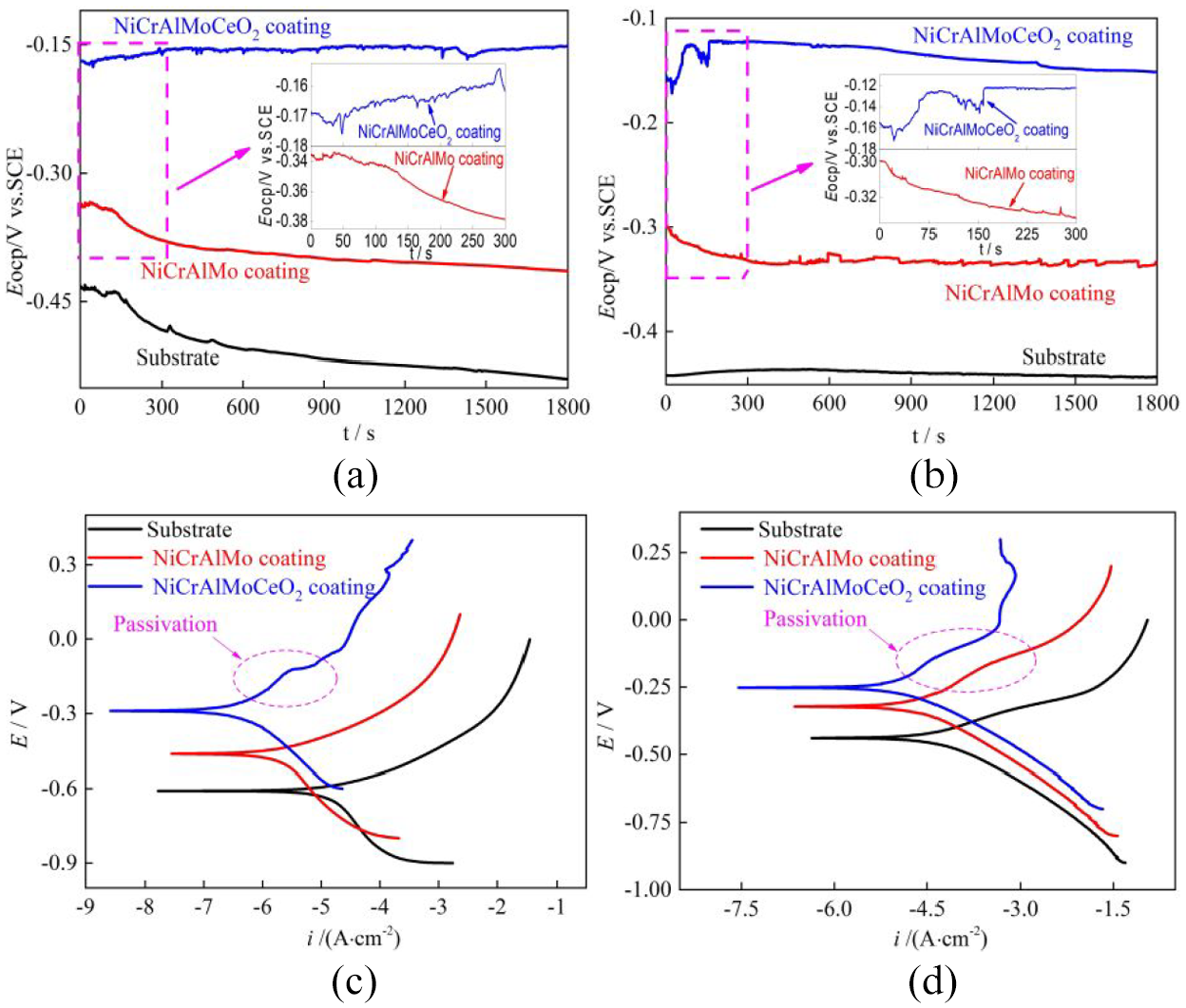
OCP and PDP curves of the substrate, laser-cladded NiCrAlMo and NiCrAlMoCeO2 coatings in different solutions:(a) OCP in 3.5 wt% NaCl solution, (b) OCP in 1 M HCl solution, (c) PDP in 3.5 wt% NaCl solution, and (d) PDP in 1 M HCl solution.
The PDP curves were used to further investigate the passivation effectiveness of the substrate and coatings in both solutions. As shown in Figure 5(c), the anodic and cathodic branches of the coatings in the 3.5 wt% NaCl solution moved to the positive direction relative to the substrate, while the current density of the NiCrAlMo coating increased gradually with no obvious passivation phenomenon. In contrast, the NiCrAlMoCeO2 composite coating exhibited anodic passive behavior with a pitting potential (Epit) of −0.12 V and a passive current density (ipass) of ∼10−5.5 A cm−2. As shown in Figure 5(d), the PDP curves of the NiCrAlMo and NiCrAlMoCeO2 coatings in the 1 M HCl solution were more electropositive than that of the substrate, while the passivation region on the latter had a higher Epit of −0.13 V and a lower ipass of ∼10−4.5 A cm−2. As a result, the thermodynamic tendency and the corrosion kinetics of the substrate significantly decreased under the effects of the two types of coatings, while the passive property of the NiCrAlMo coating in both environments was further improved by co-addition of CeO2.
The final Eocp, Ecorr, and icorr of the substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings are shown in Figure 6. The Eocp and Ecorr were relevant to the corrosion activity of the samples, where the higher values implied more positive chemical stability. 33 Figure 6(a) and (b) show that the Eocp and Ecorr of the Ni-based coatings in both solutions were more positive than that of the substrate, while the NiCrAlMoCeO2 composite coating possessed the highest value. Generally, higher values indicated the slow permeation of the corrosion solution through the coating, which is associated with the effective barrier of the protective oxide film. The more negative values of the NiCrAlMo coating and the substrate demonstrated their higher susceptibility to corrosion. The dissolution extent of the coating was closely associated with the icorr, and the slower corrosion reaction rate was accompanied by a smaller icorr. 33 As seen in Figure 6(c), the substrate had the largest icorr, followed by the NiCrAlMo and NiCrAlMoCeO2 coatings; therefore, the corrosion progress on the latter was the slowest. Moreover, compared with the value in the acidic solution, the icorr of the NiCrAlMoCeO2 composite coating in the 3.5 wt% NaCl solution decreased by two orders of magnitude, which further indicated its better passivation inhibition in a neutral solution.
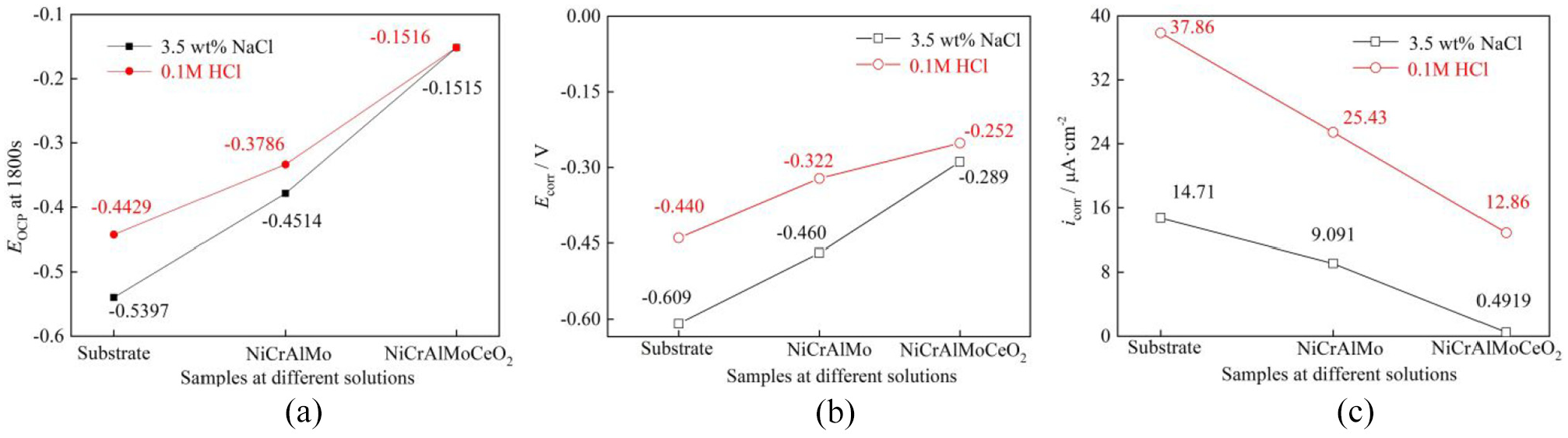
OCP and PDP parameters of the substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings: (a) Eocp at 1800 s,(b) Ecorr, and (c) icorr.
Electrochemical mechanisms
The electrochemical mechanisms of the substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings in 3.5 wt% NaCl solution and 1 M HCl solution were studied via the electrochemical impedance spectrometry. As shown in Figure 7(a) and (b), the Nyquist semicircle was in an incomplete and depressed characteristic, which illustrates the “dispersion effect” arising from surface roughness and heterogeneity. 34 The semicircle diameter of the Ni-based coatings was almost greater than that of the substrate, while that of the NiCrAlMo coating was further increased by dozens of times after CeO2 incorporation. As presented in previous studies, the Nyquist semicircle was proportional to the corrosion mitigation ability, and the larger diameter revealed that it is a better corrosion barrier for the NiCrAlMoCeO2 composite coating. 35 This was similar to the order of their impedance modulus at low-frequencies (i.e. |Z|f→0), where the higher magnitude of 105 Ω cm2 was obtained for the NiCrAlMoCeO2 composite coating, while the lower range of 102∼104 Ω cm2 was obtained for the NiCrAlMo coating and the substrate. In general, the increase in |Z|f=0.01HZ also suggested improvement in electrochemical stability. 36 Furthermore, the NiCrAlMoCeO2 composite coating had a relatively higher and broader peak distribution at medium and low frequencies, which verified its better passive protection of the corroded surface. The order of electrochemical properties in both solutions was NiCrAlMoCeO2 coating > NiCrAlMo coating > substrate.
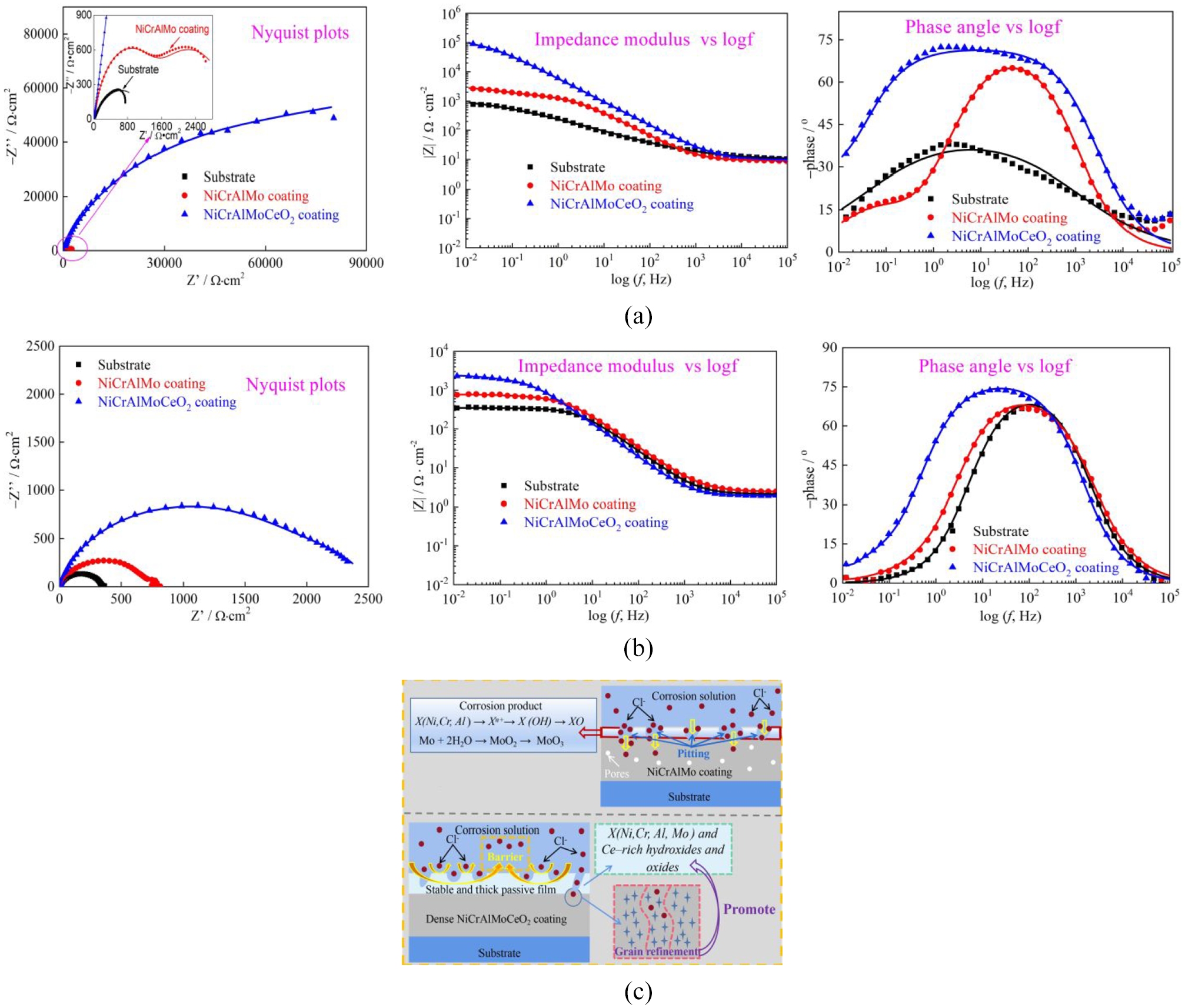
Nyquist, impedance modulus, and phase angle analysis of the substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings: (a) in 3.5 wt% NaCl solution, (b) in 1 M HCl solution, and (c) schematic diagram of electrochemical mechanisms.
The schematic diagram of coating electrochemical mechanisms is presented in Figure 7(c). During the electrochemical period, the four-electron oxygen process was the main cathodic reaction on the sample surfaces:
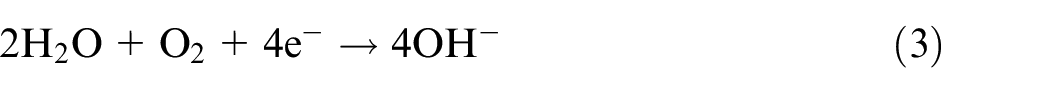
For the NiCrAlMo coating, the free ions of X (Ni, Cr, and Al) were released and contacted with OH− to form the oxide layer, as shown in equations (4)–(6):


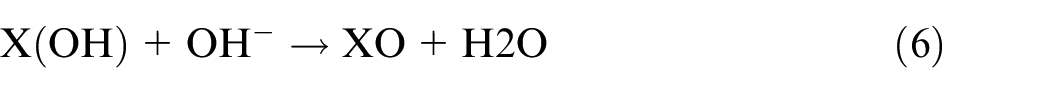
Mo was also dissolved into the various oxidized forms of MoO2 and MoO3 according to equations (7) and (8) 14 :
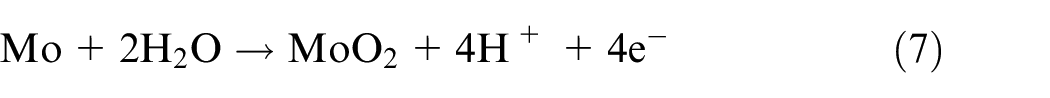
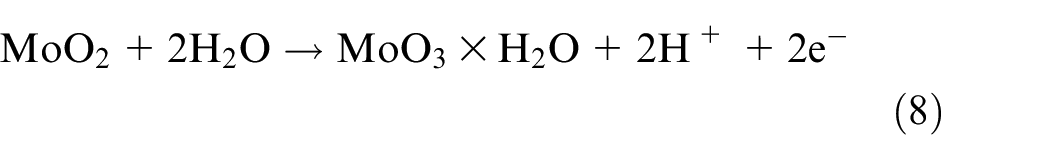
The formed composite oxide layer adhered on the coating surface was beneficial toward reducing the contact area between the coating and the corrosion solution, which reduced the corrosion tendency and restricted the dissolution to a certain extent.
After the incorporation of nano-sized CeO2 particles, the electrochemical property of the NiCrAlMo coating was effectively improved, indicated by the larger Nyquist semicircle diameter, Zf=0.01HZ, and higher phase angle peak. This improvement was associated with increased surface quality, improved grain refinement, and better passivation protection.
First, the porosity of the NiCrAlMo coating surface not only provided more local sites for preferential pitting to take place preferentially but also led to the abundant corrosion of galvanic cells between defects and uniform regions, which increased the corrosion activity and accelerated the destruction rate. 37 The added CeO2 modified and purified the coating microstructure, minimizing the number of active sites and microcells, which significantly increased chemical stability for the corrosion surface.
Second, the circuitous path length of the aggressive ions to the substrate was also extended by reduced crystallite size, 38 and the simultaneously increased grain boundaries with well-dispersed Mo element provided more nucleation sites for the formation of corrosion products, which resulted in a more uniform and dense protective film on the coating surface. 39
Third, the oxide film was further thickened by Ce-rich hydroxides and oxides obtained from equations (9) to (12)40–42:
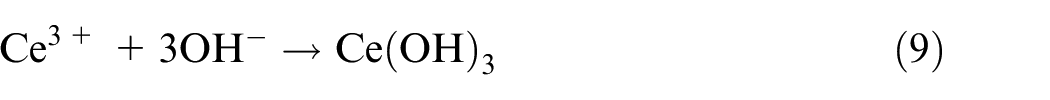
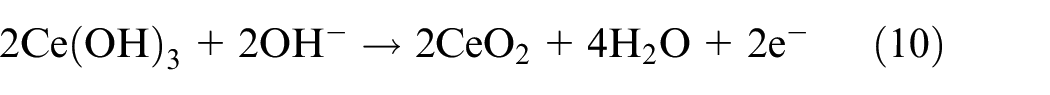
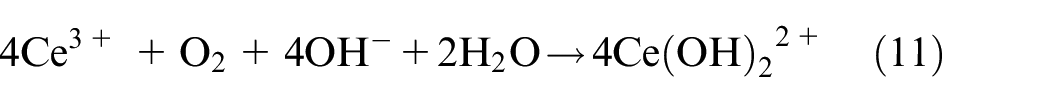
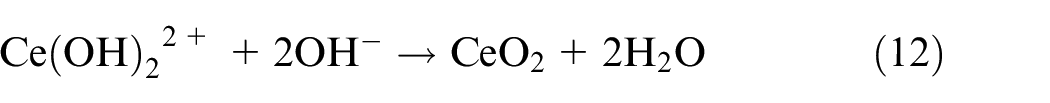
As a result, the active inhibitory, shielding, and blocking functions of the NiCrAlMoCeO2 composite coating in both solutions resulted from the co-contribution of the modified microstructure and compact passivation film by insertion of the CeO2 nano-particle, which effectively inhibited electrolyte penetration and reduced the electrochemical dissolution or migration for the coating cations into the solutions.
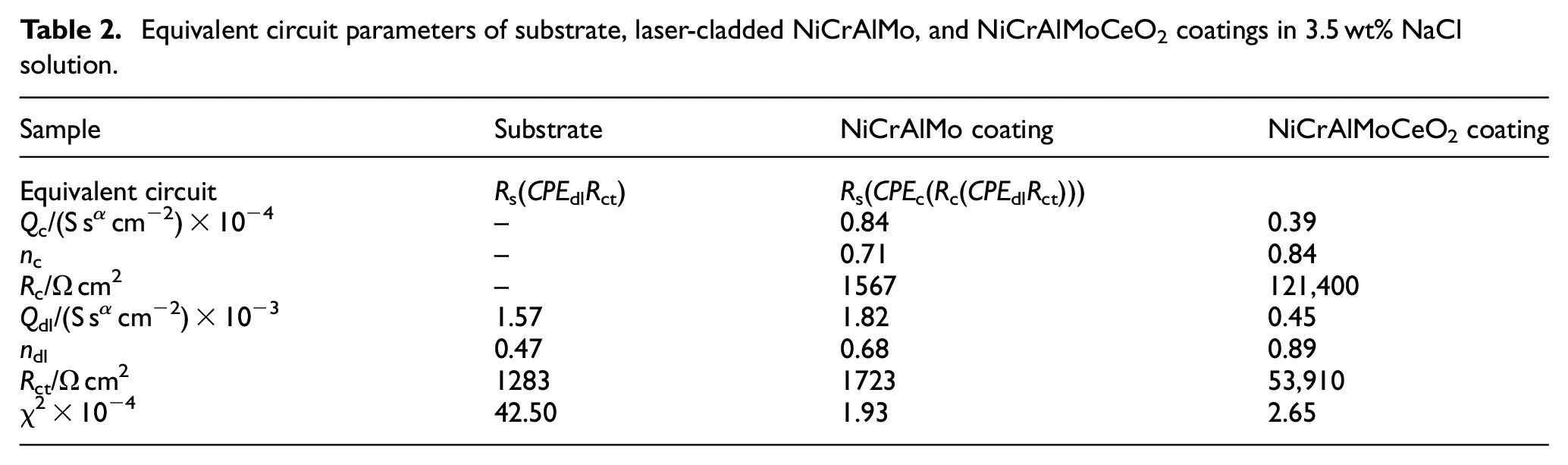
Two widely adopted EC models quantitatively characterized the electrochemical performance of coatings and substrates shown in Figure 8(a) and (b). The fitting results are summarized in Tables 2 and 3. Lower Q and higher n represent smaller susceptibility and better homogeneity, while the Rct and Rc denoted the resistance toward the transportation of corrosive ions and the charge transfer across the substrate, which was mainly attributed to the passivation contribution difference. 43 The values from Table 2 show that the substrate had the largest Qdl of 1.57 × 10−3 S sα cm−2, smallest n2 of 0.47, and lowest Rct of 1283 Ω cm2, which indicated its fastest electrochemical degradation rate. Both Qc and Qdl from the NiCrAlMo coating and NiCrAlMoCeO2 composite coating exhibited a similar change, which decreased from 0.84 × 10−4 to 0.39 × 10−4S sα cm−2 and 1.82 × 10−3 to 0.45 × 10−3 S sα cm−2, respectively. Meanwhile, the corresponding n1 and n2 of the NiCrAlMoCeO2 composite coating were also larger, which justified its higher capacitive property and better corroded surface stability. It was also seen that the Rc and Rct of NiCrMoAlCeO2 composite coating were 121,400 and 53,910 Ω cm2, which were far larger than those of the NiCrAlMo coating. Therefore, the oxide film formed on the NiCrAl MoCeO2 composite coating was more uniform and less defective, which provided higher barrier protection.
Equivalent circuit parameters of substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings in 3.5 wt% NaCl solution.
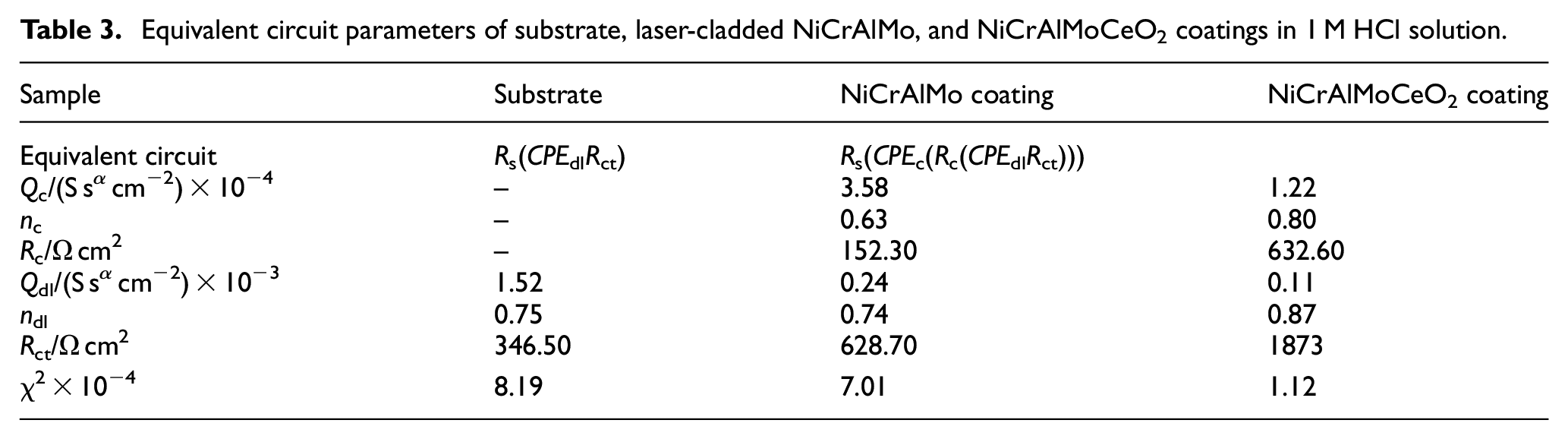
Equivalent circuit parameters of substrate, laser-cladded NiCrAlMo, and NiCrAlMoCeO2 coatings in 1 M HCl solution.
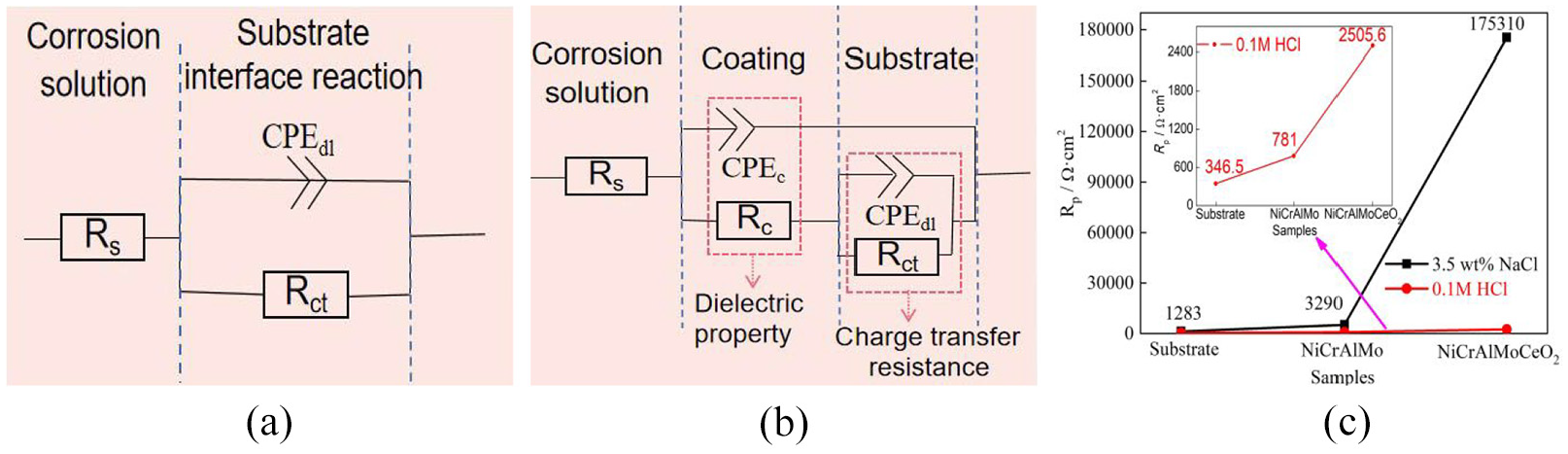
EC models for the substrate, laser-cladded NiCrAlMo and NiCrAlMoCeO2 coatings in different solutions: (a) EC model for substrate, (b) EC model for coatings, and (c) Rp/Ω cm2.
The corresponding electrochemical parameters of all the samples in the 1 M HCl solution are listed in Table 3. Similar to the results in the 3.5 wt% NaCl solution, the NiCrMoAlCeO2 composite coating had the smallest Qc and Qdl of 1.22 × 10−4 and 0.11 × 10−3 S sα cm−2, and the largest n1 and n2 of 0.80 and 0.87 among the three investigated samples. Furthermore, the corresponding Rc and Rct were 632.60 and 1873 Ω cm2, significantly larger than the NiCrAlMo coating and substrate. Hence, the oxide film of the NiCrAlMoCeO2 composite coating was more protective, which corresponded to its highest passivation region in the PDP curve.
The corrosion mitigation efficiency of the NiCrAlMoCeO2 coating in both solutions was envisaged by the polarization resistance, Rp, which was calculated as the sum of Rc and Rct. By comparing the values in Figure 8(c), it was known that the Rp of the NiCrAlMoCeO2 coatings in the 3.5 wt% NaCl solution was ∼70 times than that in the 1 M HCl solution, implying its better anti-corrosion prevention for the substrate in neutral solution. Due to the corrosion product films being less stable and having relatively higher solubility in acidic solutions,44,45 which eventually showed better passivation stability and more effective anti-corrosion prevention in the 3.5 wt% NaCl solution.
Conclusions
The following conclusions of this study are summarized below:
The laser-cladded Ni-based coatings formed a good metallurgical bond with the substrate, while the surface quality and microhardness of the NiCrAlMo coating are improved by nano-CeO2 incorporation.
The electrochemical performance of the substrate in the 3.5 wt% NaCl solution and 1 M HCl solution was enhanced by the NiCrAlMo and NiCrAlMoCeO2 coatings, while the latter has a better passive property, which is mainly attributed to the densification and homogenization of the microstructure and the formation of Ce-rich oxides.
The icorr of the laser-cladded NiCrAlMoCeO2 composite coating in the 3.5 wt% NaCl solution was two orders of magnitude smaller than that in the 1 M HCl solution, and the corresponding Rp was several times larger, which shows the improved electrochemical resistance in the neutral solution.
As the actual amount of salt concentration of seawater was approximately 3.5 wt%, the laser-cladded nano-CeO2/NiCrAlMo coating presents a great anti-corrosion application prospect for Q235 steel in a marine environment. Future work recommended studying the mechanisms of salt-spray corrosion, microbial corrosion, and corrosion-wear for this composite coating.
Footnotes
Author contributions
Chen Haixiang: Conceptualization, methodology, Software, Data curation, Writing-Original draft preparation, Writing, and Investigation. Wang Kun: Supervision, Validation, Reviewing, and Editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 51875413) and the Natural Science Foundation of Shanghai (No. 21ZR1467700) and the Fundamental Research Funds for the Central Universities (No. 22120210443).
