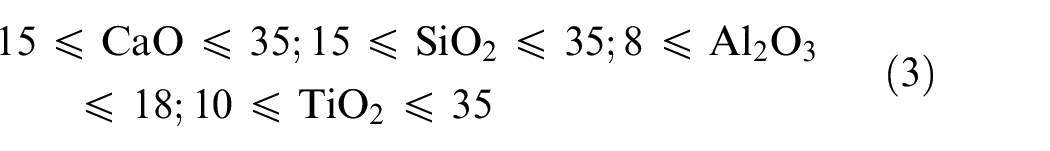Abstract
The bimetallic welds are frequently utilized for pipeline transport system of the nuclear power plants. The occurrences of welding defects generally depend on the filler electrode as well as the electrode coatings during shielded metal arc welding process. This study involves the design of austenitic stainless steel welding electrodes for SS304L–SA516 bimetallic welds. The objective of research work includes the novel design of Al2O3–TiO2–CaO–SiO2 coatings by combining two ternary phase systems using extreme vertices mixture design methodology to analyze the effect of key coating constituents on the weld metal chemistry and mechanical properties of the welds. The significant effect of electrode coating constituent CaO on weld metal manganese content is observed which further improves the toughness of bimetallic weld joints. Various regression models have been developed for the weld responses and multi objective optimisation approach using composite desirability function has been adopted for identifying the optimized set of electrode coating compositions. The role of delta ferrite content in promoting the favourable solidification mode has been studied through microstructural examination.
Keywords
Introduction
The bimetallic welds have applications in the steam generators and reactor pressure vessels of nuclear power plants. These welds represent the weld joints between the austenitic stainless steels and ferritic low alloy steels. The certain characteristics of austenitic stainless steels which include good mechanical behaviour at elevated temperatures make them most suitable to be used in certain sections such as superheaters and reheaters while the use of low alloy steels is preferred in the construction of reactor vessels particularly due to their low cost. The austenitic stainless steels show good weldability through the traditional arc welding processes like shielded metal arc welding (SMAW), submerged arc welding (SAW) etc.
In SMAW process, the design of welding electrode coatings is an area of research. The research work carried out by authors1–6 mainly focused on similar welds using different coatings constituents. Mitra and Eagar7–10 studied the transfer of alloying elements during slag-metal reactions in submerged arc welding process. Pandey et al. 11 analyzed the weld metal chemistry with different welding fluxes for SAW welded joints. The authors12–15 used the statistical design of experiment (DoE) techniques to predict the effect of welding fluxes and process parameters on weld chemistry as well as mechanical properties of similar welds. Zhan et al. 16 developed an intelligent welding procedure qualification based on artificial neural networks. The developed expert system could predict the welding process parameters along with the mechanical properties for a SMAW process.
The important findings of nickel (Ni) based welding consumables for the joining of stainless steels with high nickel alloys are reported by the researchers.17–21 The major drawback of Ni-based welding consumables is their inferior weldability as compared to austenitic stainless steels. Further, the bimetallic welds with these consumables are also not immune to failure. The composition of bimetallic weld is largely dependent on the dilution ratios of base metals and the welding filler wire along with elemental diffusion across the weld.22,23
Lee et al. 24 investigated the electrical and mechanical behaviour of friction welded Cu/Al bimetallic joints during annealing. The researchers applied two kinds of intermetallic compound layers and found the significant increase in electrical resistivity with the increase in thickness of intermetallic compound layers. It was also observed the remarkable decrease in tensile strength of bimetallic joints to near zero at the annealing condition of 773K. Zhang et al. 25 found good shear strength in explosive welded sheets of X65 pipe steel and 2205 stainless steel. The microstructural analysis highlighted the coarse columnar crystal formed in the melted zone near the interface of base materials. Ning et al. 26 examined the laser butt welded Ti-Steel bimetallic joint by using a copper interlayer. The purpose of the interlayer was to reduce the transition zone between base materials. Zhan et al. 27 studied the grain morphology along with the temperature field simulation of dissimilar joint between Ti-6Al-4V and 1050 aluminum alloy using laser welding-brazing technique. The authors also developed a 3D finite element model to find out the temperature field. The authors28–31 explored the mixture design methodology for the design and development of electrode coatings for bimetallic welds. The researchers32–35 investigated the different flux systems to design and develop SAW fluxes for various applications. Khan and Chhibber 36 studied the mechanical behaviour of filler metal on dissimilar welds.
The literature review shows that limited work is published on design of welding electrode consumables for bimetallic welds in SMAW process. There is lack of a scientific approach towards the design of electrode coatings for bimetallic welds involving studies on effect of electrode coating constituents on microstructural evolution and mechanical property enhancement. In the present work, the CaO–SiO2–Al2O3–TiO2 based electrode coatings with austenitic stainless steel fillers have been designed and developed for SMAW process. Total 21 electrode coating formulations have been designed using extreme vertices mixture design methodology and then the effect of various coating constituents on the weld chemistry and mechanical properties of bimetallic welds has been investigated. The paper also examines the role of delta ferrite content during solidification through metallurgical investigations.
Design approach
The extreme vertices mixture design methodology was used for the development of welding electrode coatings. 37 According to this method, for a mixture of k constituents with each constituent having lower limit (α i ) and upper limit (β i ) as constraint, the percentage composition (x i ) of ith constituent and the constrained mixture design can be mathematically represented as follows: 38
The primary requirement for deciding the formulations of electrode coatings is that the composite melting temperature of the coating should be lower as compared to the core wire and the parent materials. So, the percentage compositions of various constituents are widely considered by analyzing the appropriate experimental phase diagrams. Depending upon the relevant previous research, the ranges of electrode coating constituents are further narrowed. 22
In present work, phase diagrams of CaO–SiO2–TiO2 and CaO–SiO2–Al2O3 systems as shown in Figure 1(a) and (b) were used to decide the percentage composition of electrode coating constituents by selecting different points (points marked with X) on these phase diagrams. The ranges of electrode coating constituents (CaO, SiO2, Al2O3 and TiO2) were estimated while taking into consideration the other essential electrode coating constituents in the complete composition and are given below:
and
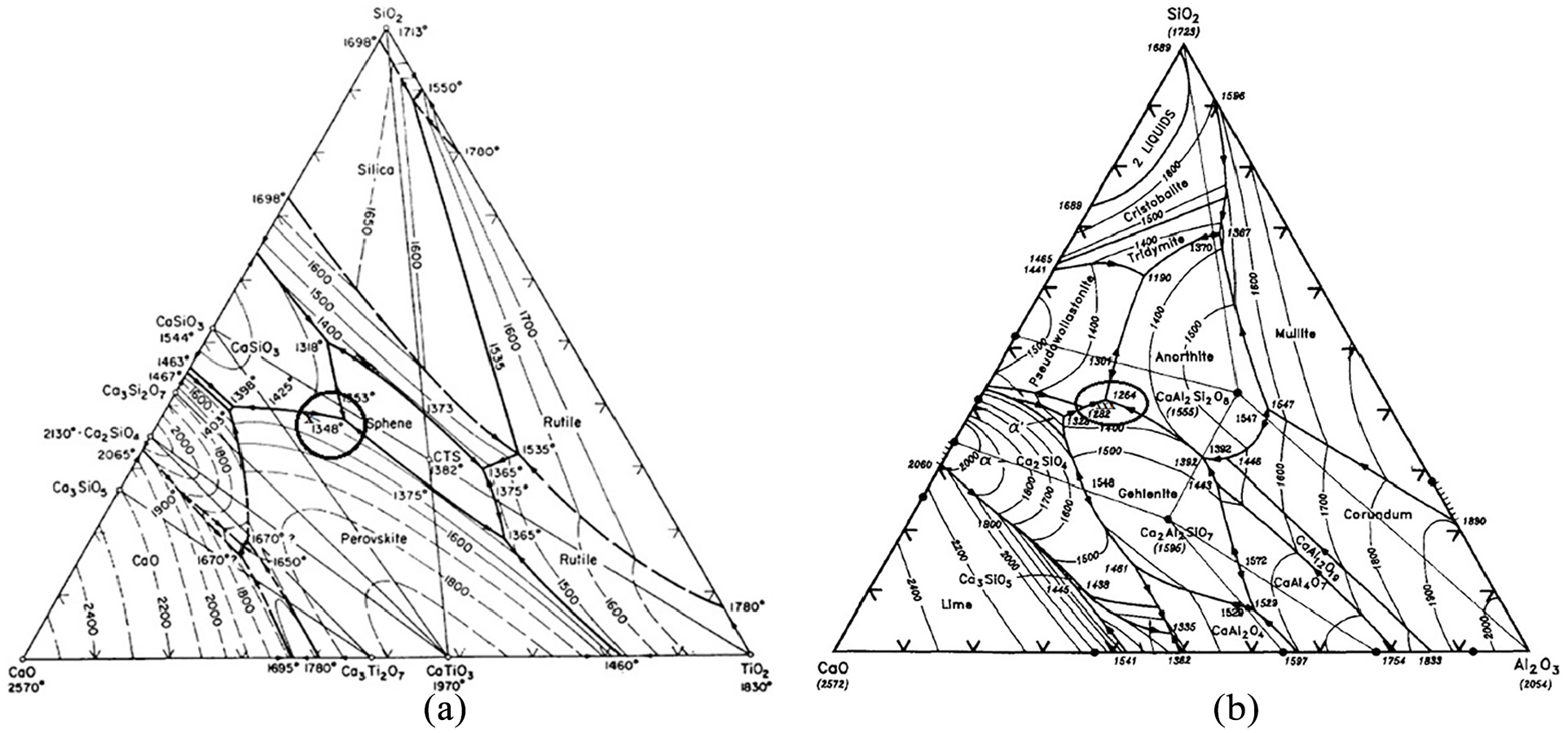
The Table 1 shows the 21 electrode coating formulations finalized on the basis of extreme vertices mixture design method. Each formulation represents the nature of point (vertex or centroid) on the constrained mixture design space as shown in Figure 2.
Electrode coating compositions.
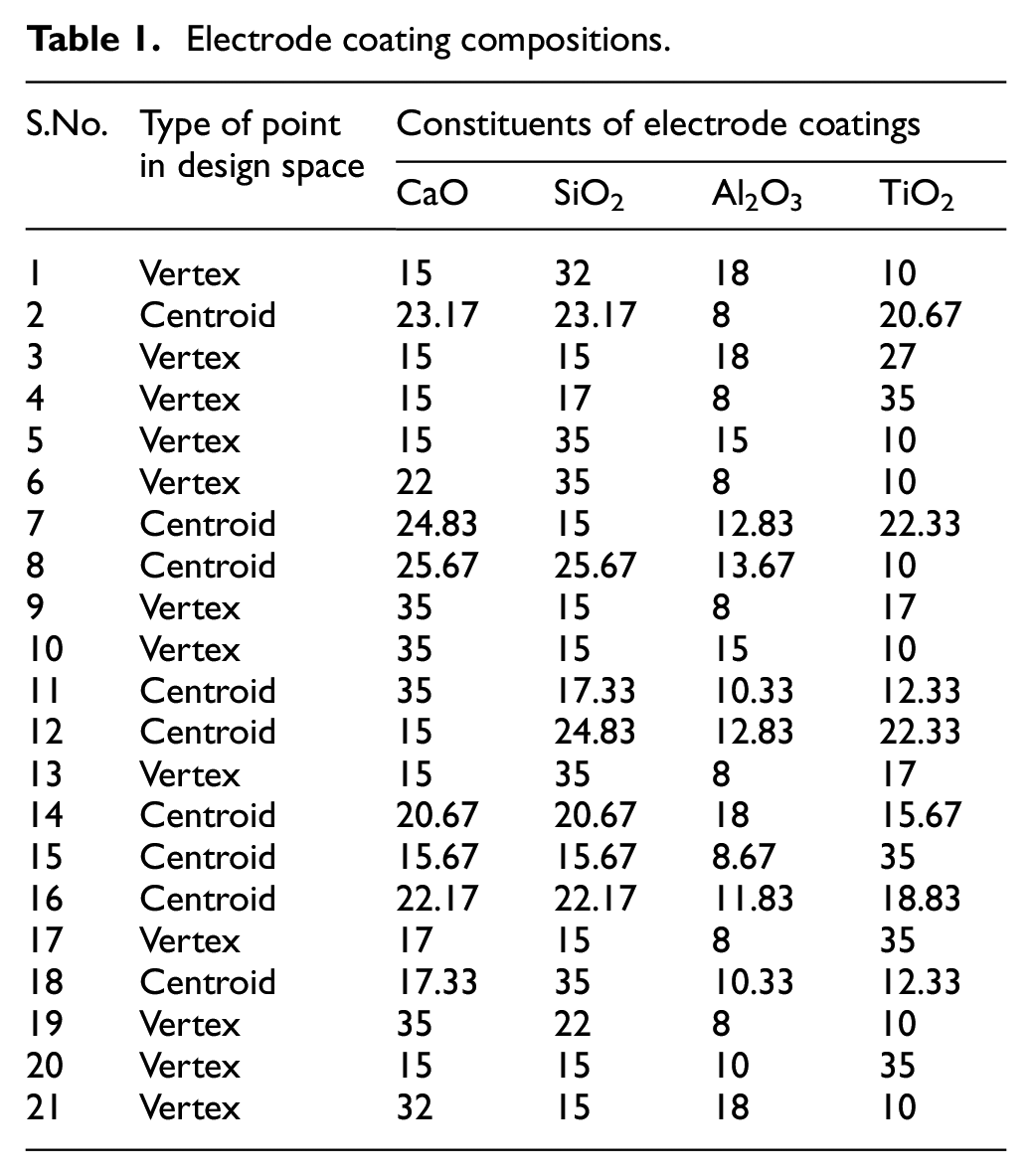
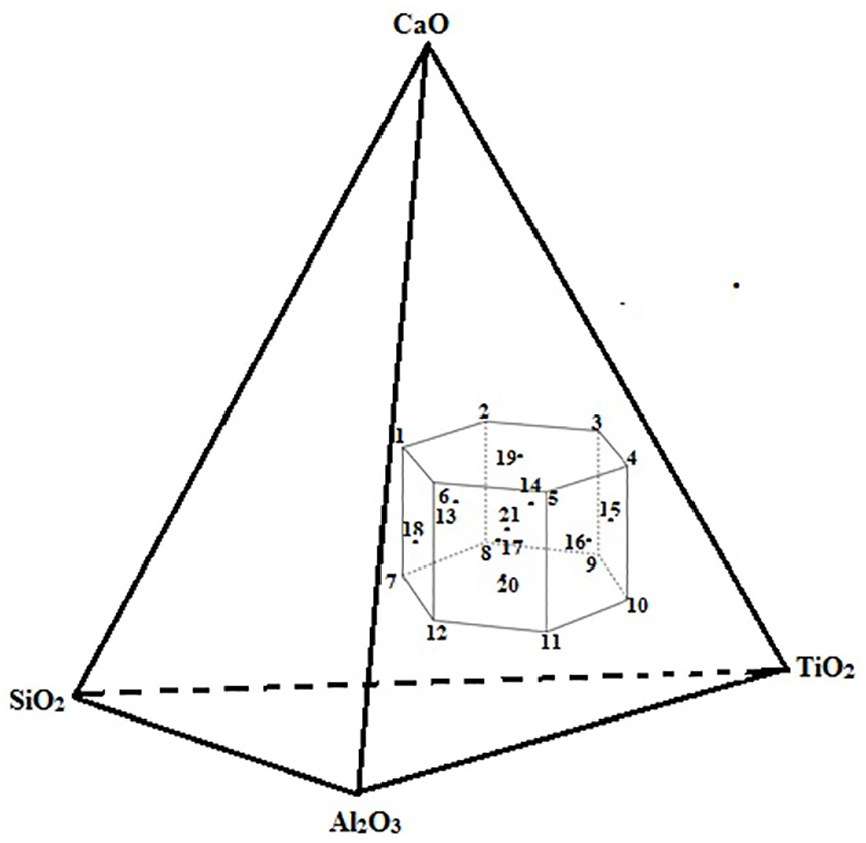
Constrained mixture design space.
Experimentation
Electrode coating formulation
The various electrode coating constituents were weighed as described/formulated in design matrix (Table 1) and then mixed properly with dry mixer. After adding the suitable binder in the dry mixture and followed by further mixing in the wet mixer, the electrodes were extruded using SS308L and SS309L core wires having diameter 3.15 mm and length 350 mm. Each set of electrode coating formulation as given in Table 1 was used to develop two different types of coated welding electrodes that is, one with SS308L core wires and another with SS309L core wires.
For dissimilar welding, the base plates of SS304L and SA516 materials were used and weld coupons of dimensions as described in Figure 3 and Table 2 were prepared using developed coated electrodes. For welding, each set of developed SS309L core wire welding electrodes was used for buttering the SA516 base plates and then SS308L core wire electrodes having same type of electrode coating formulation were subsequently used to make the bimetallic welds by joining the buttered SA516 plates with SS304L plates.
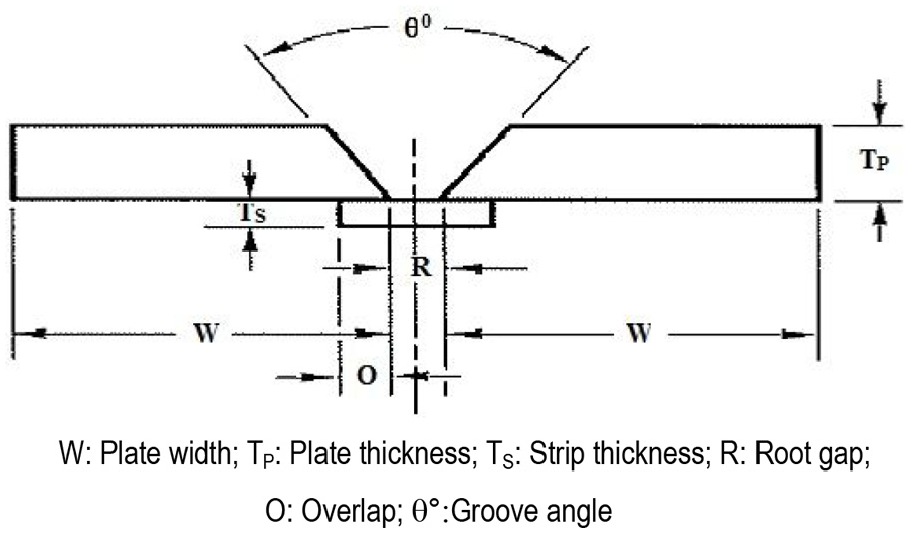
Weld coupon.
Weld coupon specifications.

W: plate width; TP: plate thickness; TS: strip thickness; R: root gap; O: overlap; θ°: groove angle.
Welding parameters
To choose the welding parameters bead-on-plate tests were performed by varying the welding current (80–100 A) whilst keeping the welding speed at approximately 120mm/min. Base plates were butt welded using optimized welding process parameters.
Results and discussion
The chemical composition analysis of base plates along with core wires (Table 3) and of bimetallic welds between SS304L and SA516 base plates (Table 4) was determined using optical emmision spectrometer. The delta ferrite content (δf) which mainly prevents cracking in bimetallic welds during solidification, is represented by ferrite number as given in Table 5. It (δf) was measured using ferritescope and WRC 92 diagram. 41
Elemental compositional analysis of base metals and core wires.
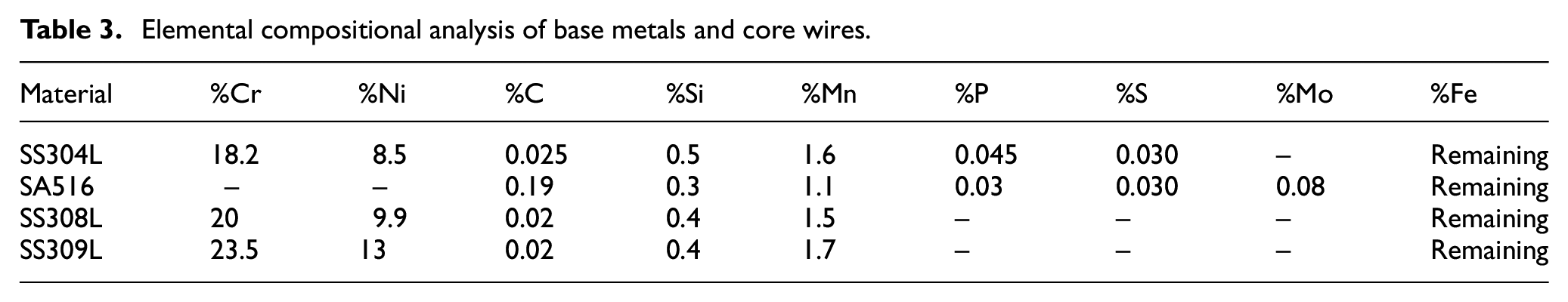
Chemical composition (wt%) of bimetallic welds.
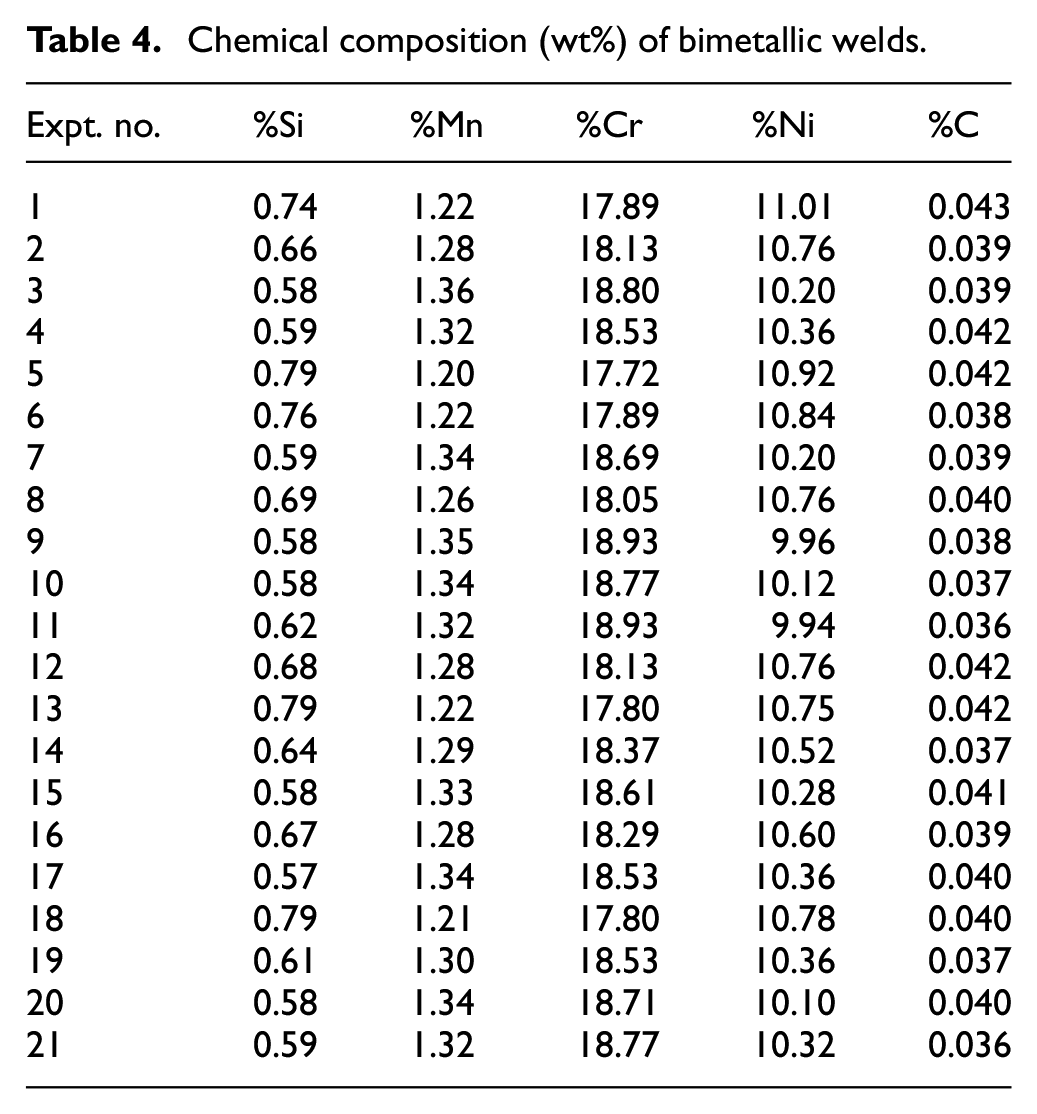
Results of ferrite number (FN).
FA: ferritic–austenitic; AF: austenitic–ferritic.
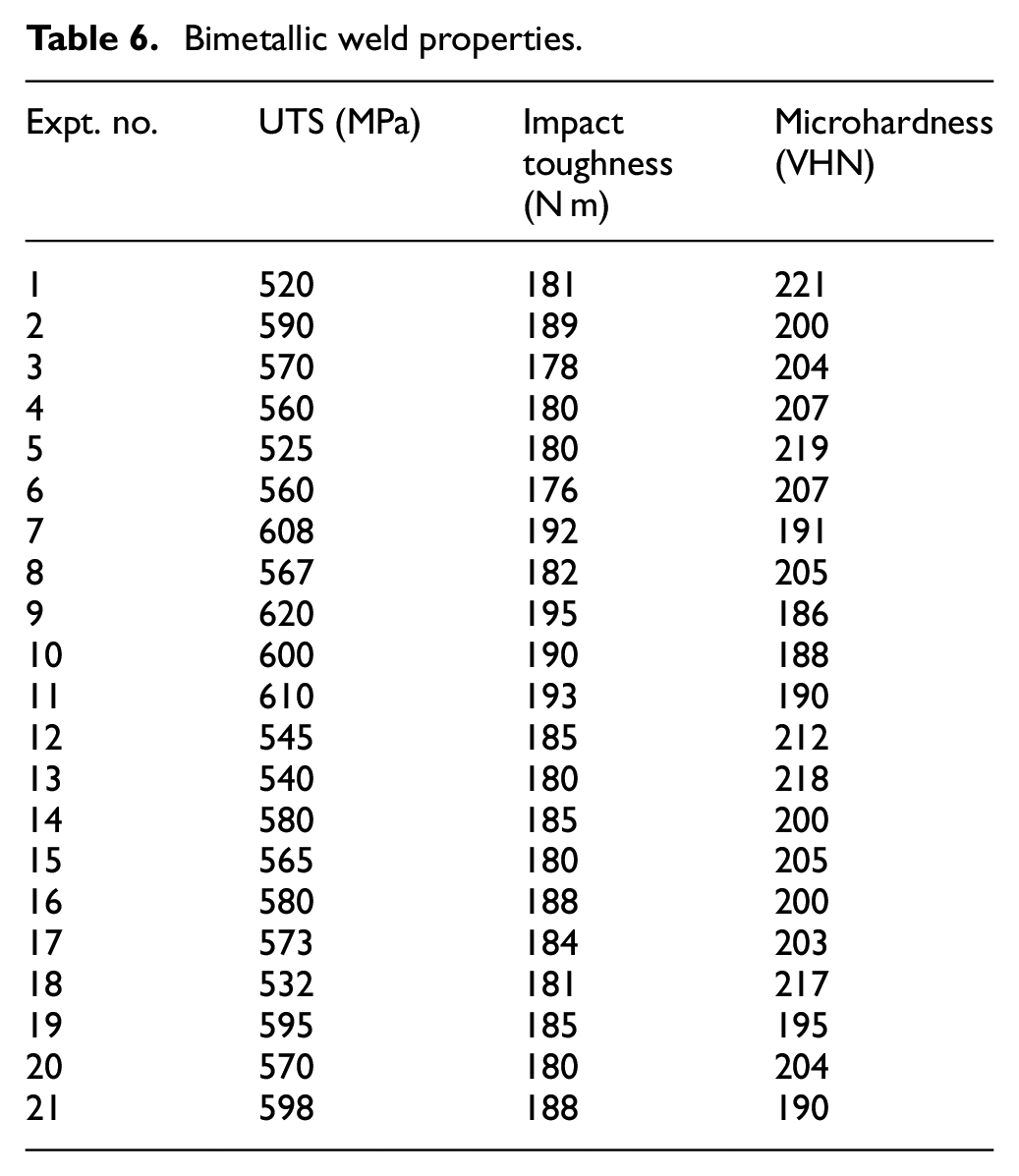
Different samples were cut from the welded coupons for testing of various properties such as tensile test, impact test and microhardness of bimetallic welds. The results of different properties are given in Table 6. The typical microstructures were obtained at 250X magnification from weld metal specimens.
Bimetallic weld properties.
Regression models
The experimental data of various weld responses was used to formulate the regression models in terms of % composition of individual coating constituents and their binary mixtures. The various developed regression models were:

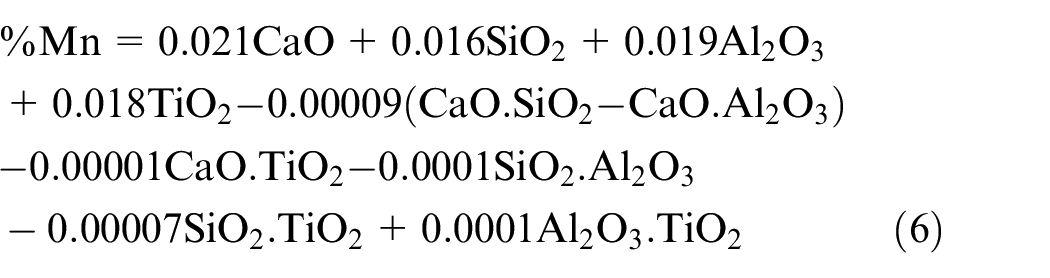




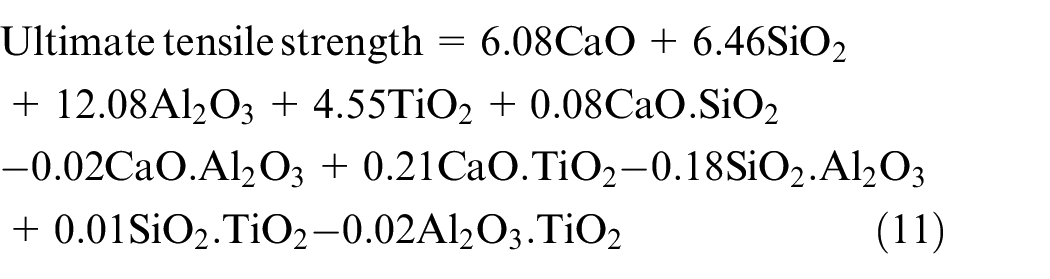

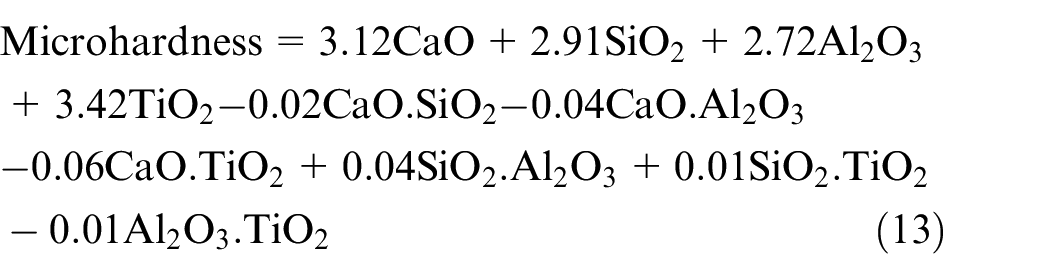
Analysis of variance (ANOVA)
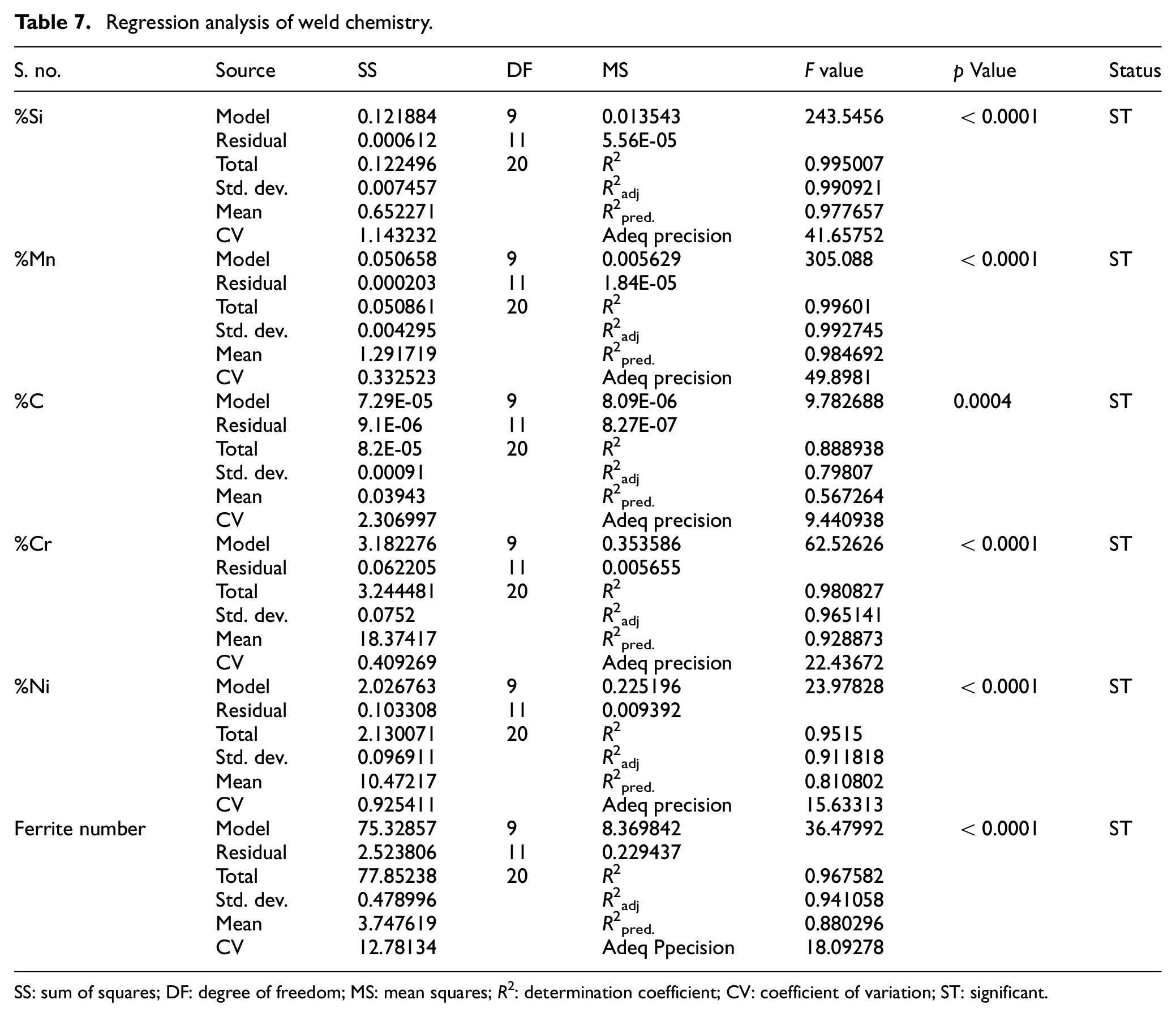
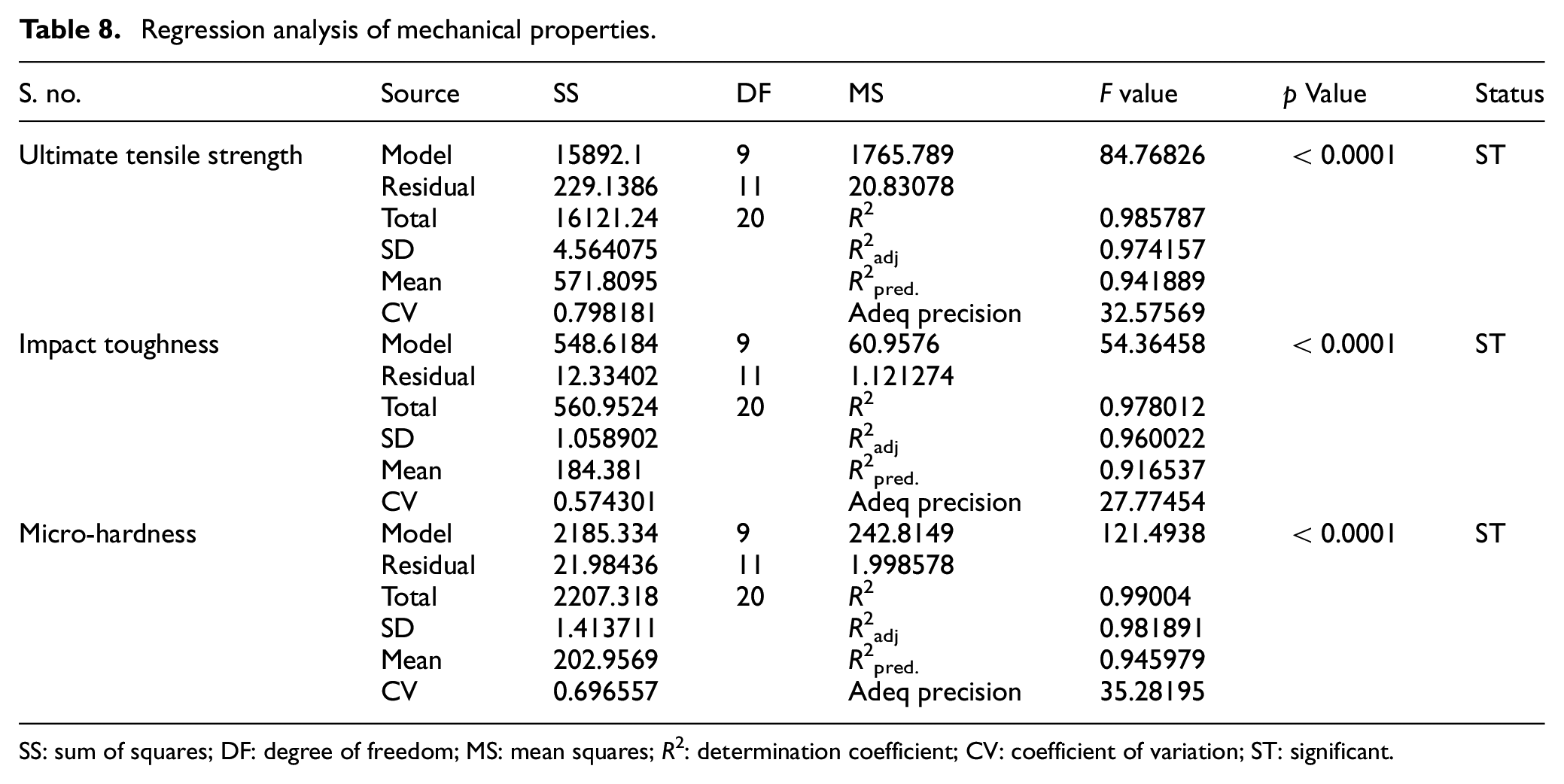
The developed regression models have been analyzed using analysis of variance (ANOVA) that is, t-test and F-test. The significance of various regression coefficients have been tested by comparing their t-values with tabulated t-values at 95% confidance level. The regression analysis of weld chemistry and mechanical properties of bimetallic welds is presented in Tables 7 and 8 respectively. The Tables 9 to 11 depict the t-test analysis for significance of coefficients of regression models of various weld responses. The result analysis of regression models reveals that there are some insignificant terms in the models. These insignificant terms have been removed to improve the models. The summary of these tests recommends the adequacy of developed regression models.
Regression analysis of weld chemistry.
SS: sum of squares; DF: degree of freedom; MS: mean squares; R2: determination coefficient; CV: coefficient of variation; ST: significant.
Regression analysis of mechanical properties.
SS: sum of squares; DF: degree of freedom; MS: mean squares; R2: determination coefficient; CV: coefficient of variation; ST: significant.
Analysis of regression coefficients of weld responses (%Si, %Mn, %C) using t-test.
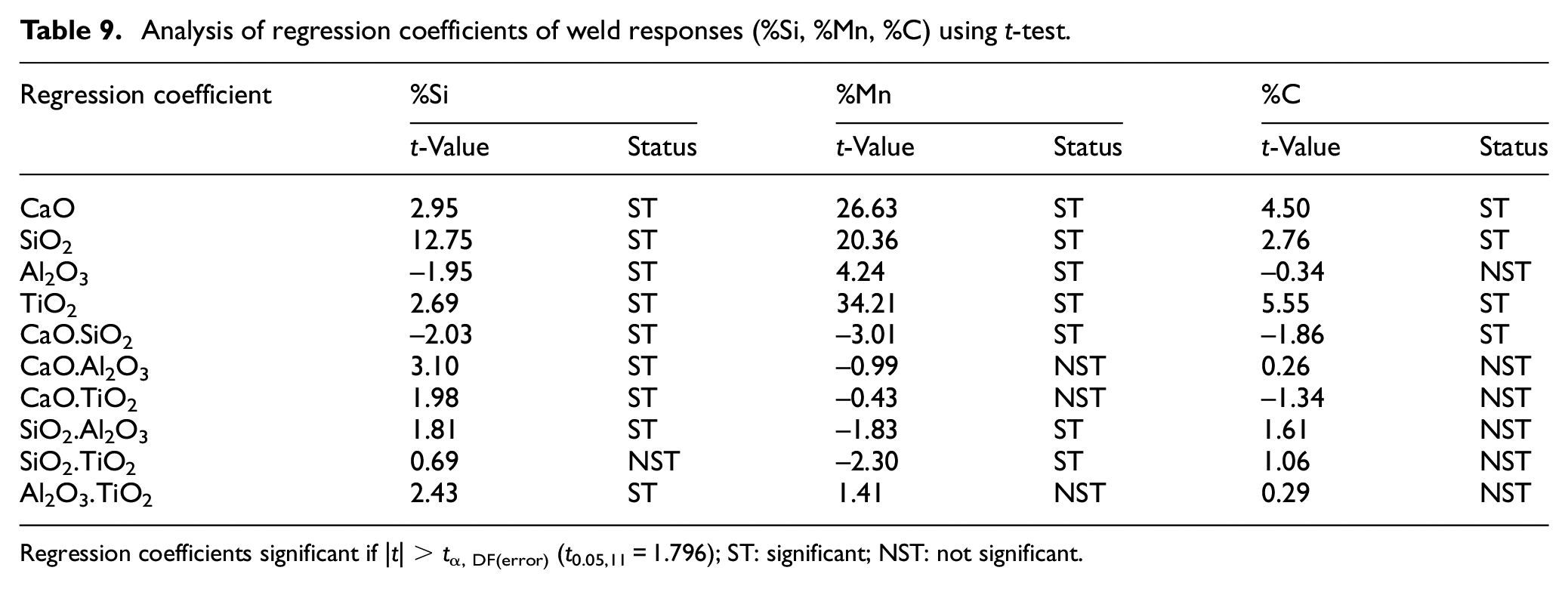
Regression coefficients significant if |t| > tα, DF(error) (t0.05,11 = 1.796); ST: significant; NST: not significant.
The F value of regression model is the ratio of mean square value of model to the mean square value of residual. It basically compares the model variance with the residual variance. 42 Therefore, for F value close to one, there is lesser chance of model significance. At 95% confidence level, the p value less than 0.05 represents the significance of model. The F values and its p values of various weld responses as given in Tables 7 and 8 clearly show the significance of developed regression models.
The coefficient of determination (R 2 ) values of regression models are ≥90% which indicate that predicted values of various weld responses are in close agreement with their actual values as shown in Figures 4(a) to (f) and 5(a) to (c). The coefficient of variation (CV) is calculated as the standard deviation divided by the mean. 42 The low values of CV as given in Tables 7 and 8 indicate the good fit model. The summary of ANOVA recommends the adequacy of developed regression models.
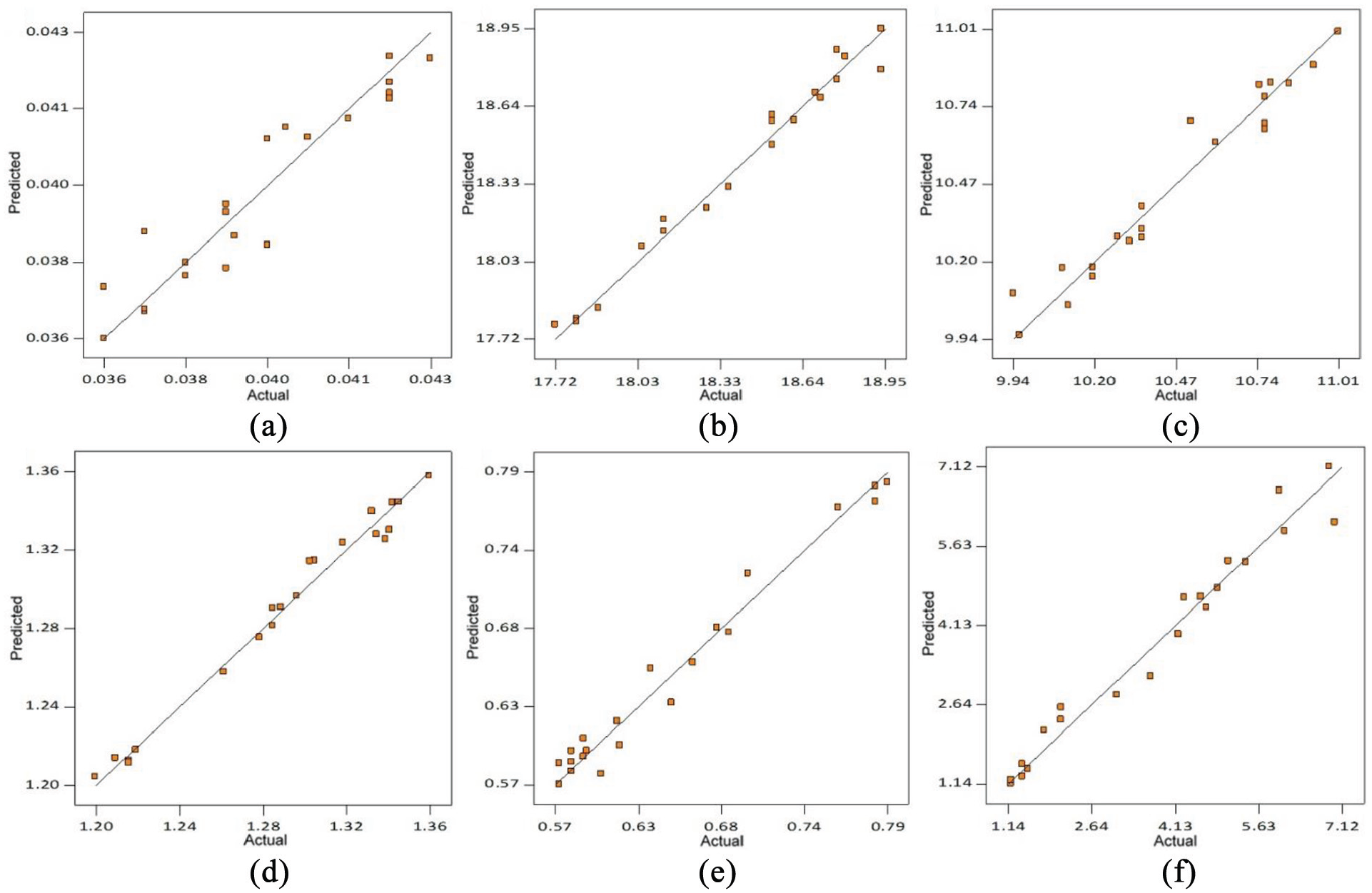
Comparison plots of predicted versus actual weld chemistry of bimetallic welds: (a) %C, (b) %Cr, (c) %Ni, (d) %Mn, (e) %Si and (f) ferrite number.
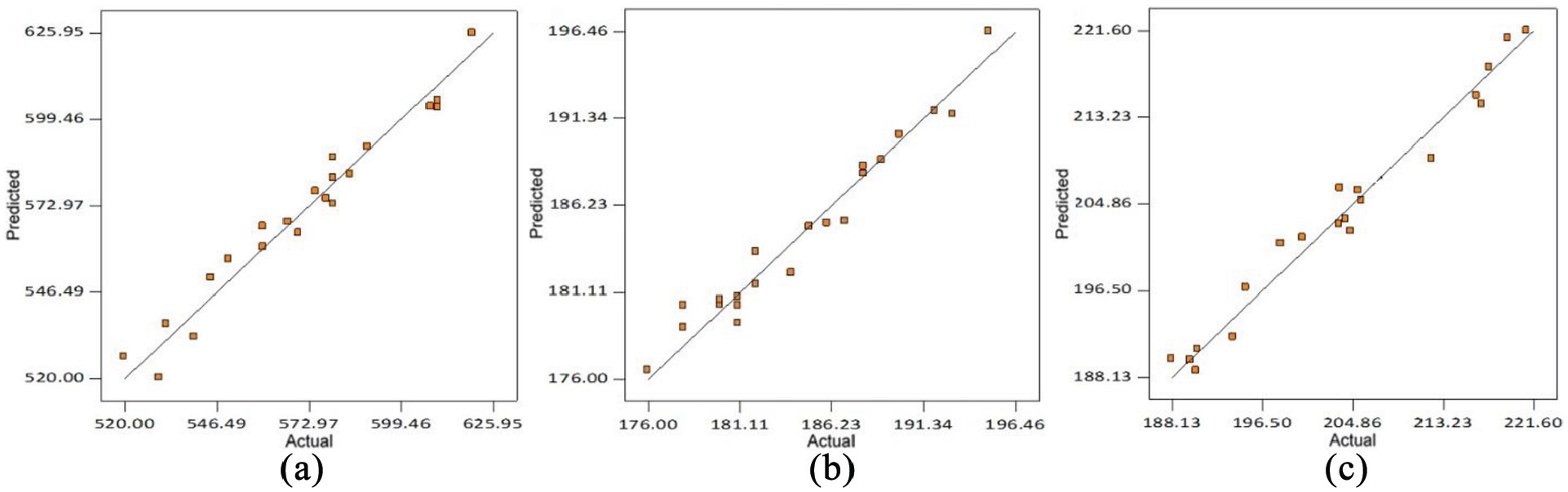
Comparison plots of predicted versus actual mechanical properties of welds: (a) UTS, (b) impact toughness and (c) microhardness.
Role of electrode coating constituents on weld chemistry
The results show that percentage of silicon in weld gets significantly increased with the increase in SiO2 content of coatings. SiO2 gets dissociated into silicon and oxygen during slag-metal reactions and thereby raises the silicon content in the weld. 43 Al2O3 tends to decrease the weld metal silicon content as it reacts with the fluorides present in the electrode coatings to form AlF3 compound which further reacts with silicon according to the following reaction: 44
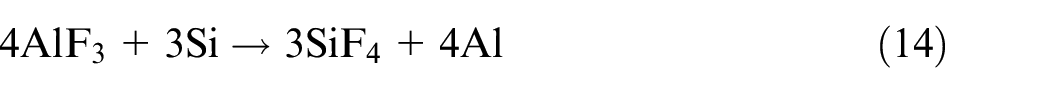
The coating constituents CaO and TiO2 show increasing effect on weld metal silicon content. The effect of binary mixture CaO.SiO2 is to decrease the silicon content as the CaO has a tendency to decrease the SiO2 activity in slag-metal reactions and thus decrease the silicon content in weld composition according to the following reaction: 4


The individual effect of all the constituents of electrode coatings that is, CaO, SiO2, Al2O3 and TiO2 is increasing on the manganese content of weld metal. The transfer of manganese content in weld is given by Pandey et al.: 11

The CaO increases the activity of MnO during slag-metal reactions thereby shifting reaction to the left side which is also confirmed by Sharma and Chhibber. 33 The effect of TiO2 could be explained from the fact that it decreases the viscosity of slag during slag-metal reactions and hence promotes the transfer of manganese to weld. All the binary mixtures of SiO2 show the reversing trend on manganese content of weld metal due to the increase in the concentration of MnO in slag resulting in the lower level of manganese content in the weld. 7
The individual electrode coating constituents except Al2O3 show the increasing effect on the carbon content of weld metal. CaO.SiO2 shows the single significant decreasing effect on weld metal carbon content whereas the interaction effects of other binary mixtures are not significant as shown in Table 10. The regression analysis reveals that the electrode coating constituents and the binary mixtures (CaO.SiO2, CaO.TiO2, SiO2.TiO2) increase the nickel content of the weld metal while the interaction effect of binary mixture Al2O3.TiO2 is significantly decreasing.
Analysis of regression coefficients of weld responses (%Cr, %Ni, ferrite number) using t-test.
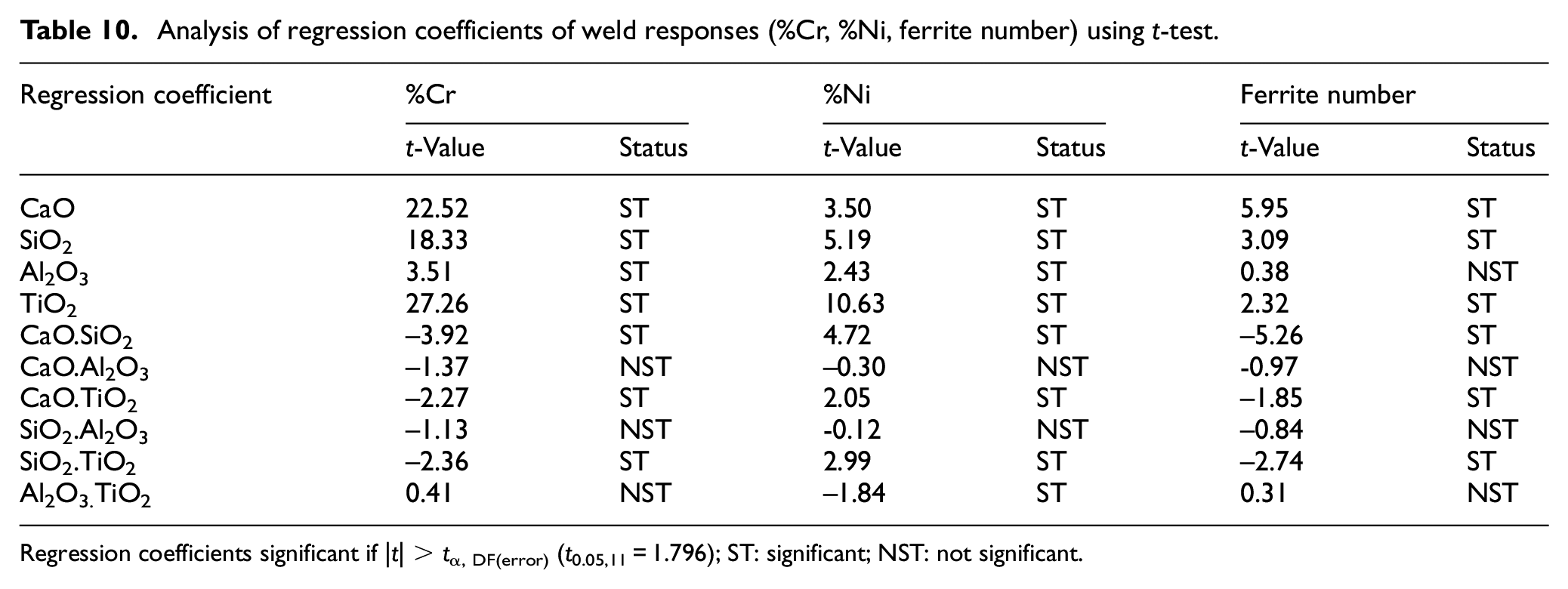
Regression coefficients significant if |t| > tα, DF(error) (t0.05,11 = 1.796); ST: significant; NST: not significant.
Role of electrode coating constituents on solidification behaviour
Electrode coating constituents promote the delta ferrite having vermicular morphology in welds as compared to sigma phase as is clear from the microstructures (as explained later in the microstructure analysis section of this paper). The results of ferrite number as given in Table 5 show that the solidification modes are either ferritic–austenitic (FA) or austenitic–ferritic (AF).
Role of electrode coating constituents on mechanical properties of weld
The regression analysis reveals that all electrode coating constituents have significant increasing effect on the ultimate tensile strength of weld (Table 11). The electrode coating constituents tend to promote inclusions in the form of metal sulfides and oxides in weld which may further assist in the ferrite nucleation at the interface between austenite matrix and inclusions. The plasticity of the material gets improved with the finely dispersed spheroid-shaped inclusions. Al2O3 tends to deoxidize the weld pool, thereby increasing the UTS of the weld. This result is in agreement with the research work reported by Jindal et al. 38 The effect of CaO.SiO2 is to increase the UTS as the SiO2 activity is reduced due to the formation of SiO44− complex ion. 4 Therefore, the excessive dissociation of SiO2 gets reduced and the amount of dissolved oxygen remains limited. The binary mixture SiO2.Al2O3 shows significant decreasing effect on ultimate tensile strength due to their combined increasing effect on O2 in the weld.
t-Test analysis of regression coefficients of mechanical properties.
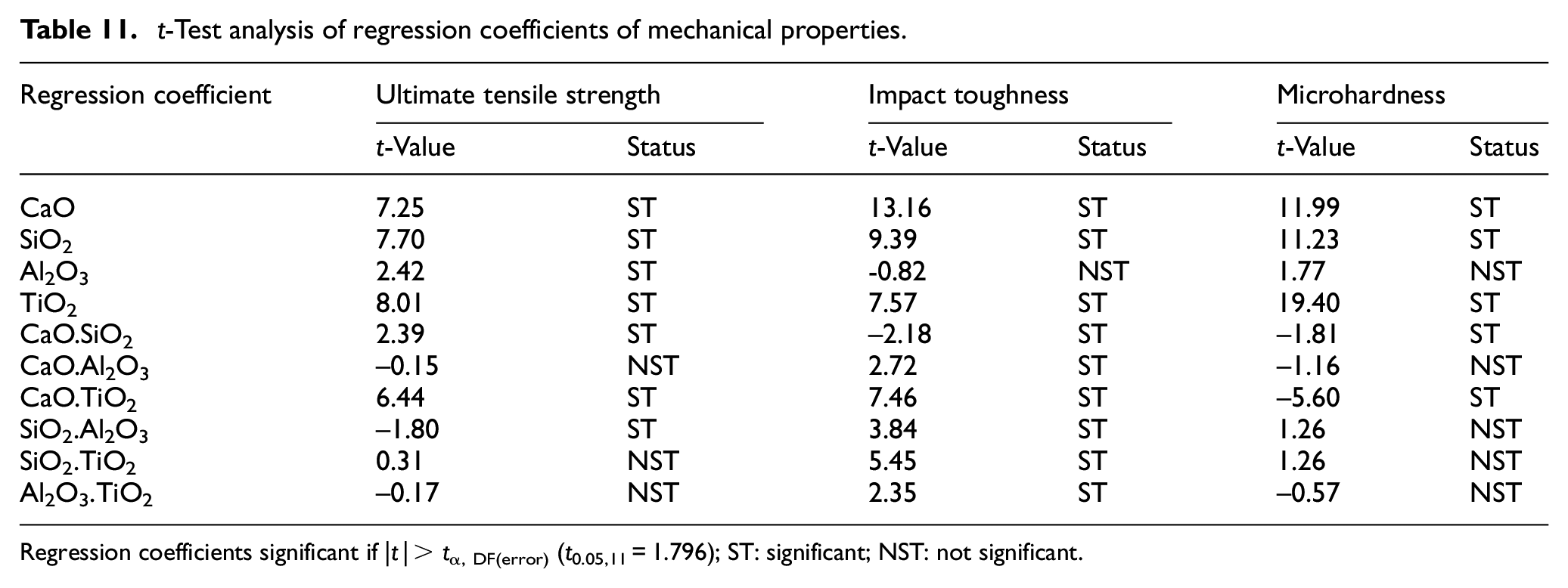
Regression coefficients significant if |t |> tα, DF(error) (t0.05,11 = 1.796); ST: significant; NST: not significant.
The basicity index of electrode coatings is increased with increase in CaO constituent which further increases the impact toughness of welds as also reported in literature. 45 The TiO2 constituent shows an increasing effect on the impact toughness of bimetallic welds. Kohno et al. 46 highlighted the grain refining property of TiO2 as the primary cause for the increase in impact toughness of welds. The interaction effects of all binary mixtures except CaO.SiO2 show the increasing trend on impact toughness. It is observed from regression analysis that the electrode coating constituents CaO, SiO2 and TiO2 increase the microhardness of weld while the effect of Al2O3 is not significant.
Contour plots
The contour surface plots of predicted values of chemical composition and mechanical properties of bimetallic welds for varying amounts of electrode coating constituents with constant TiO2 = 18.83% are shown in Figures 6(a) to (f) and 7(a) to (c), respectively.
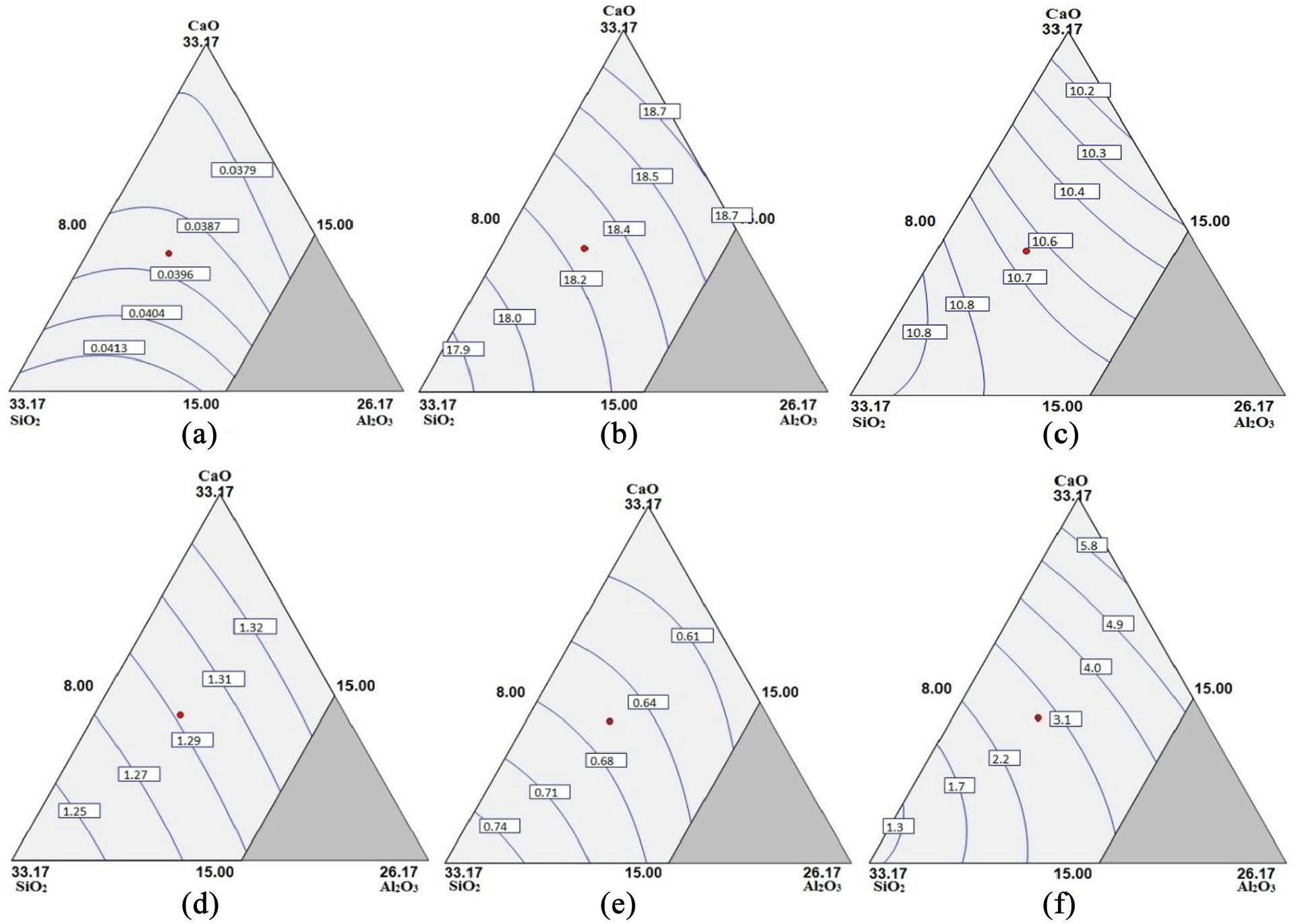
Contour surface plots for: (a) %C, (b) %Cr, (c) %Ni, (d) %Mn, (e) %Si and (f) ferrite number.
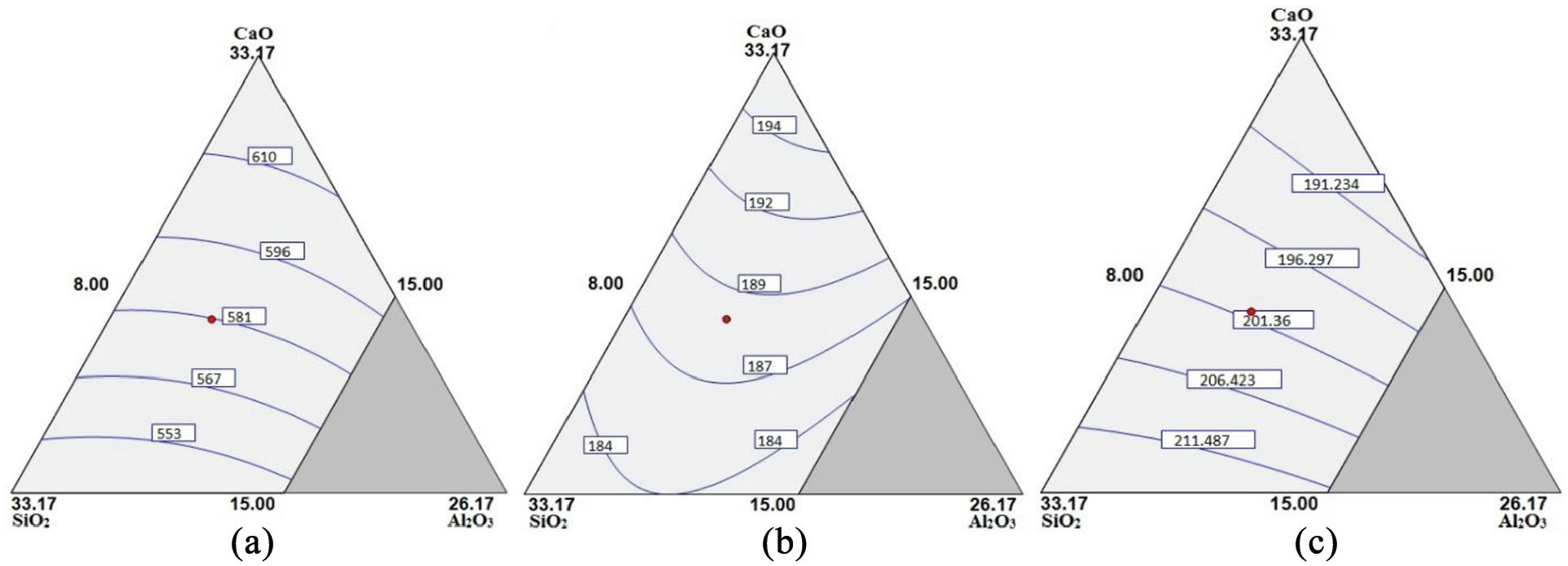
Contour surface plots for: (a) UTS, (b) impact toughness and (c) microhardness.
Microstructure analysis
The microstructures of various weld specimens are shown in Figure 8(a) to (c). The microstructures of specimens indicate the ferritic–austenitic (FA) as the primary solidification mode and the presence of austenite with varying amount of delta ferrite (δf) of vermicular ferrite (VF) type. The microstructure of weld specimen (coating formulation no. 1) shows the presence of some large size inclusions (I) on grain boundaries in Figure 8(a). This electrode coating formulation contains higher amount of SiO2 and Al2O3 (Table 1) and their binary interaction promotes the large size inclusions which adversely affects the mechanical properties of the weld (Table 6). The delta ferrite may also transform to lathy ferrite (LF) in addition to vermicular ferrite as shown in the weld microstructure of coating formulation no. 2 (Figure 8(b)). This weld specimen has better mechanical properties as given in Table 6 due to the presence of small size spherical shaped inclusions (size 0.4 µm) inside the grains. This can be attributed to relative higher amount of TiO2 and CaO in the electrode coating formulation which further assists in promoting the small size inclusions in weld. The inclusions of intermediate size 2 µm were observed in the microstructure of weld specimen of coating formulation no. 7 as shown in Figure 8(c). During SMAW process, the slag-metal reactions control the interaction of various electrode coating constituents with molten weld pool which further result in the variation of elements transfer in the weld. The variation in weld chemistry leads to the different mechanical properties and microstructures.

Weld zone microstructures obtained from: (a) experiment no. 1, (b) experiment no. 2 and (c) experiment no. 7.
Validation of models
To validate the different regression models for various weld properties, three coating compositions were randomly chosen to confirm or validate with the experimental results. Table 12 shows the error percentage for different weld properties which is approximately ≤5% in most results.
Error percentage for different properties.
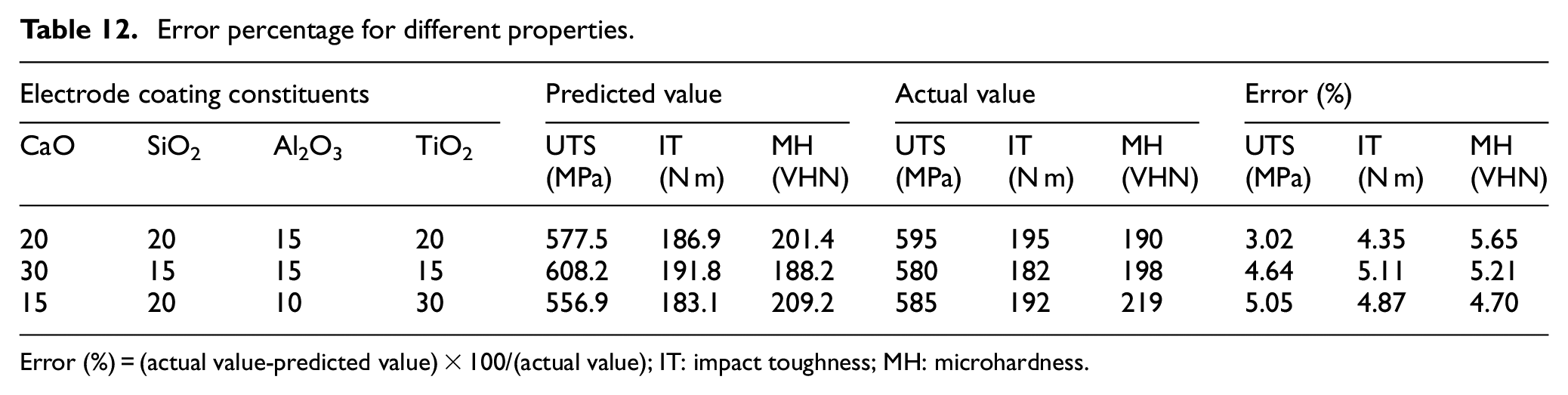
Error (%) = (actual value-predicted value) × 100/(actual value); IT: impact toughness; MH: microhardness.
Optimisation methodology
Derringer and Suich 47 proposed a multi objective optimisation methodology in which the simultaneous optimisation of several output responses is reduced to a single response optimisation problem with the help of composite desirability. The desirability function transforms the response variable into a value (on 0 to 1 scale). The composite desirability combines the individual desirabilities and highlights the relative importance of responses. Depending upon the nature of the output response, the different weights are assigned in this optimisation method.48,49 Table 13 shows the three multi objective optimized solutions with equal importance to mechanical properties along with the target value of ferrite number. The target value of ferrite number was chosen in accordance with Bystram. 50 The graphical demonstration of desirability of optimized solution no. 2 is shown in Figure 9. It clearly indicates that the desirability of all the electrode coating constituents along with ferrite number is one while the composite desirability of complete solution is 0.87.
Optimisation of different properties.
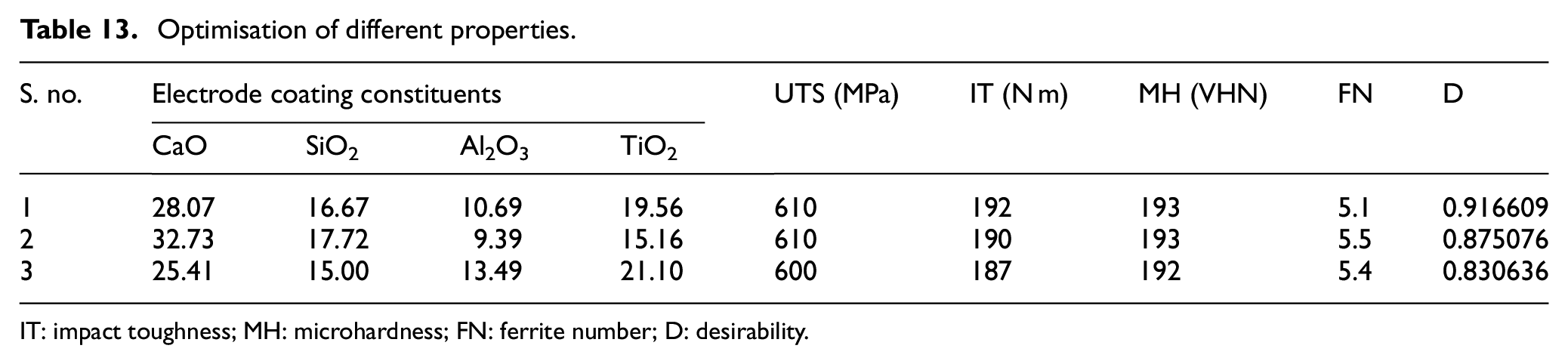
IT: impact toughness; MH: microhardness; FN: ferrite number; D: desirability.
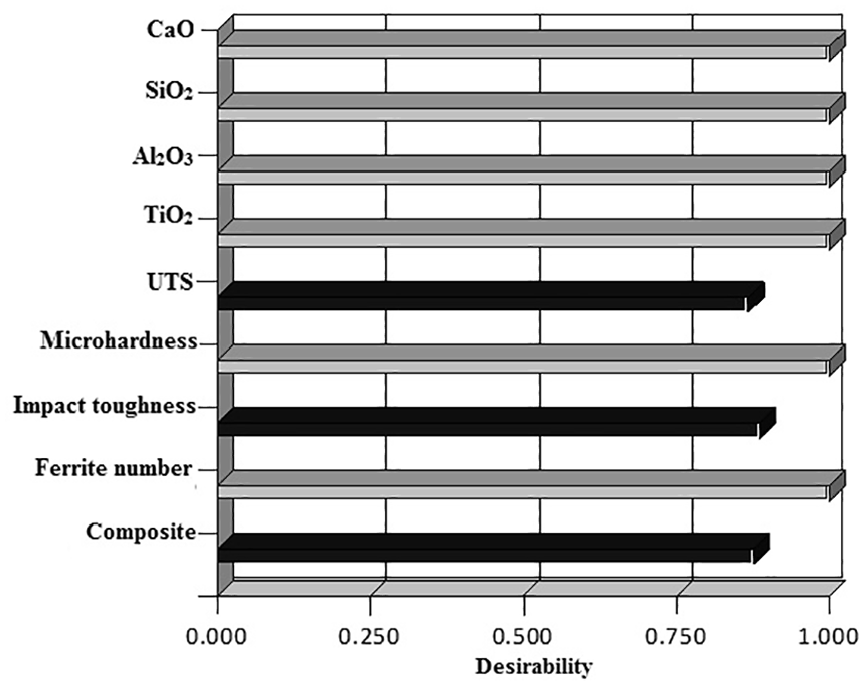
Graphical demonstration of variation of desirability of optimized solution.
Conclusions
Welding electrodes have been successfully developed based on combined CaO–SiO2–Al2O3 and CaO–SiO2–TiO2 phase diagrams using extreme vertices design method for bimetallic welds. SiO2 increases the weld metal silicon content whereas the tendancy of Al2O3 is to decrease it.
TiO2 promotes the transfer of manganese to weld by decreasing the viscosity of slag during slag metal reactions. Al2O3 releases O-2 ions that react with carbon thereby reducing its composition. CaO tends to increase the manganese content of the weld by increasing the activity of MnO. The interaction effect of coating constituents on the weld metal chromium content is decreasing.
The mechanical properties of bimetallic welds significantly depend on the linear and binary interaction effects of electrode coating constituents. Binary effect of CaO.SiO2 increases whereas SiO2.Al2O3 significantly decreases the weld strength, while CaO and TiO2 content increase the weld toughness. The significant increasing effect of electrode coating constituents CaO, SiO2 and TiO2 on microhardness has been observed.
The microstructures of the weld samples reveal the presence of inclusions of varying size and the delta ferrite (with vermicular morphology) at the dendrite cores. Higher amount of SiO2 and Al2O3 and their binary interaction promote large size inclusions which drastically reduce the mechanical properties of the welds.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.