Abstract
The diffusion bonding of 7075, 6061 and 5083 aluminium alloys to AZ31B magnesium was investigated using copper interlayer. An optical microscope along with scanning electron microscopes, equipped with an energy dispersive spectrometry/electron probe microanalysis, was utilized to characterize the microstructure of the joint. The mechanical properties of the joint were also assessed by micro-hardness and shear strength tests. The results indicate the high effect of temperature on the bonding results; so that, with a small change in temperature, severe changes were observed in the bonding results. A temperature range of 475°C–485°C and a minimum duration of 30 min with a low bonding pressure of 0.4 MPa were identified as advisable process conditions. The joint evaluation revealed the formation of CuAl2, Cu9Al4 and Al-Mg-Cu ternary phases on the aluminium-copper side, as well as Cu2Mg, CuMg2 and Al-Mg-Cu ternary phases on the magnesium-copper side in the reaction layer. When increasing the bonding temperature and duration, the amount of intermetallic compounds and, as a result, the mechanical properties of the joints changed. The highest shear strength and micro-hardness, related to the bonding performed at 480°C and holding time of 45 min, were 31.03 MPa and 167 HV, respectively.
Introduction
Aluminium is the second most abundant metal element in the earth’s crust and has a wide range of users in the industry. Aluminium and its alloys have high special strength, good mechanical properties, lightness, good surface properties and excellent corrosion resistance. 1
The high strength-to-weight ratio of magnesium alloys has led to the widespread tendency of the transportation industry to use magnesium alloys.2,3 In addition to lightness, other properties such as good fatigue, dimensional stability and very low permeability to electromagnetic waves have led to the use of magnesium alloys in structures, especially in military structures.4–6 However, their relatively poor corrosion resistance has limited their use, which can be overcome by bonding it to aluminium. Therefore, the widespread use of these two metal alloys has made the dissimilar bonding of aluminium/magnesium metals inevitable.
Intermetallic compounds, which seriously affect the aluminium/magnesium bonding efficiency, are inevitably formed during the bonding process in the bonding zone. Therefore, controlling the formation of intermetallic compounds is necessary to achieve a reliable bond between magnesium and aluminium. By applying diffusion bonding in the vacuum and by carefully adjusting the temperature and holding time, the formation of intermetallic compounds can be controlled.7,8
Diffusion bonding can be done with or without interface layer. Using the interface layer, the different diffusion rates of the two base metals can be adjusted and the negative effects – caused by the difference in thermal properties of the two base metals – can be eliminated. Furthermore, the formation of brittle intermetallic compounds can be partially remedied.9,10 The use of interface layer, in addition to reducing the formation of brittle phases, can also improve the microstructural properties of the joint. 11 Zinc, silver and nickel interface layers have been used in aluminium/magnesium diffusion bonding.12–14 The use of silver, as announced by Wang et al., 14 can lead to a decrease in the formation of brittle phases, resulting in an increased bond strength. In such a way, the shear strength of the bonding was almost doubled by applying the Zn and Ni interlayer.12,15 It has been demonstrated that the mechanical properties of the Mg-Zk60/Al5083 bonding improve with increasing the Zn-interlayer thickness. 16 Another advantage of using an interface layer is an increased intermediate transition zone (ITZ) thickness, 17 which in addition can be used to control the migration of atoms through the interface layer, creating a uniform joint. 18 Moreover, the interface layer is widely applied in metal/ceramic bonding, 19 as it can be a good solution to a non-stable interface in metal/ceramic joining. 20
Although diffusion bonding is an old method that has introduced novel approaches21,22 or modeling 23 in this field, the old process still needs to be studied more experimentally. Therefore, in this research, it is tried to study the diffusion bonding of 5083, 6061 and 7075 aluminium alloys to magnesium (AZ31B) using pure copper interface layer. Bonding is performed in a vacuum condition and the mechanical and microstructural properties of the joint are studied using optical microscope (OM) and scanning electron microscopes (SEM) equipped with energy dispersive spectrometry (EDS) line scan. The Vickers micro-hardness and shear strength test are used to study the mechanical properties of the joint.
Experimental procedures
The chemical analysis of the raw materials is given in Table 1. In order to performing the diffusion bonding experiments, a vacuum tube oven (vacuum capacity of 1 × 10−3 tor, maximum temperature of 1700°C with a 70 mm diameter and 100 mm length cylindrical chamber) was used (Figure 1).

Vacuum tube furnace.
The specimens were first cut by the Charmilles Robofil 290 wire cut machine and, then, ground to 10 × 10 × 5 mm3. In order to remove the oxide layer and surface contamination, the surface of aluminium and magnesium samples was polished by sandpaper No. 1500 and 1000, respectively. Before testing, samples were ultrasonically cleaned in an acetone bath for 15 min and, then, by ethanol 96%, and, finally, were immediately dried by flow of warm air. The polished and chemically treated specimens were stacked in a die and, then, the entire diffusion bonding setup, shown in Figure 2, was inserted into a vacuum chamber. The specimens were heated up to the bonding temperature with a heating rate of 10°C/min and, simultaneously, the required pressure was applied. The copper interface layer with a purity of 99.7% and a thickness of 20 µm was utilized.

Samples fixed in the fixture.
After bonding, in order to check the microstructure of the joint and the base metals, the samples were first cut by wire cut and, then, etched after being polished (at a speed of 500 rpm with alumina powder with an average grain size of 0.3 µm). A solution of acetic acid, picric acid, ethanol and distilled water was used for magnesium side etching; whereas, the Keller’s solution was applied for the aluminium side. 26 The chemical composition of the used etching solutions is shown in Table 2. Evaluation of the microstructure of the samples was performed using the Olympus optical microscope. The VEGA//TESCAN-LMU scanning electron microscope was used to examine the samples and also to characterize the composition of the phases in the joining area. The microscope was equipped with an EDS chemical analysis system as well as a linear analysis of EPMA elements. Map analysis was used to show the distribution of elements in the joint as well as the base metals.
Chemical composition of used etching solutions. 26
Shearing and micro-hardness test was used to determine the mechanical strength of the joints. The shearing test was performed based on the ASTM E229-97 standard instruction. In this sense, samples with a cross section of 9 × 9 mm2 were prepared. Figure 3 shows the fixture used for shearing experiments. The fixture is made of cold drown steel (S.P.K. 110) with a hardness of 700 VCR (HRC 60). To apply force, the SANTAM model STM-50 was used with the loading rate of 0.5 mm per minute. Micro-hardness experiments were performed by the Vickers device with an operating load of 50 g, a loading time of 20 s and at intervals of 20 μm perpendicular to the bonding intersection. The hardness of the base metals – Al 7075 and Mg AZ31 – was measured at 130 HV and 48 HV, respectively.

Jig and fixture designed for shearing test.
In this study; first, it was tried to determine the optimal temperatures for the bonding process. Examination and the study of the binary phase diagrams of copper-magnesium and aluminium-copper revealed that aluminium-copper at 548°C and magnesium-copper at 485°C had a eutectic reaction. 27 Close to the eutectic point, the initial temperature of 510°C and the process duration of 40 min were selected. Bonding in these conditions led to severe deformation of the specimens (Figure 4). With this in mind and in order to carry out perfect joints, it was decided to reduce the temperature and time to 470°C and 30 min respectively, while other conditions were kept constant. In order to further investigate the process, bonding was performed at temperatures of 475°C, 480°C and 485°C. In addition, in order to investigate the effect of process duration, the 40 and 45 min holding times were also considered. The conditions stated in Table 3 for each test were used. In all experiments, the applied pressure was 0.4 MPa.

Deformation of bonding sample performed at 510°C and 40 min holding time.
Samples test conditions.
The last number indicates the aluminium alloy type, 7 for Al7075, 6 for Al6061 and 5 for Al5083.
Results and discussion
Macro-analysis
As shown in Figure 4, the first sample test was faced with a large deformation after bonding. Due to the fact that the performing pressure was 0.4 MPa, this large deformation can be considered as being greatly affected by temperature. In addition, a significant 33% reduction in volume was observed after joining. Due to performing the process in vacuum environment, this phenomenon can be caused by high magnesium evaporation. The joint sample performed at a temperature of 470°C and duration of 30 min was shown in Figure 5. As shown in Figure 5(a), the specimens appear to be bonded together, but after several steps of paper sanding, the two pieces are separated. Figure 5(b) shows that a very thin layer of aluminium has diffused into the copper layer.

(a) Al-Mg joint at 470°C and 30 min holding time and (b) this sample after finishing.
The process was also investigated at the temperatures of 475°C, 480°C and 485°C and 30 min. duration (Figure 6). As shown in Figure 6(c), the specimen performed at 485°C was also deformed; while, the amount of deformation and weight loss were much less than the first test operated at the 510°C. According to the results of the joints obtained at temperatures of 475°C and 480°C, the samples were without plastic deformation and the joints were apparently formed (Figure 6(a) and (b)). Further investigations have shown that when temperatures are below 475°C, the joints do not have sufficient strength. Moreover, no acceptable bond was established at 485°C and with increasing the temperature, more deformation and evaporation of samples were observed especially in magnesium side.

Joint samples at 30 min holding time: (a) 475°C, (b) 480°C and (c) 485°C.
Effect of bonding temperature
OM analysis
The optical microscope images of the joints carried out at 30 min duration and temperatures of 475°C and 480°C are shown in Figure 7. Three layers of the copper-transfer zone to aluminium (L1), the middle copper zone (L2) and the copper-transfer zone to magnesium (L3) are formed. Figure 7(a) shows that copper diffused more into aluminium than magnesium; so that, the penetration of copper in magnesium (L3) – in some specific places – is cross-sectional and partial, but in aluminium (L1) it is observed as intermittent strips with the thickness of about 5 µm. According to the Figure 7(b), as the temperature increases from 475°C to 480°C, the thickness of the transfer zone slightly increases and the L1 thickness reaches about 7 µm. It is also observed that only with increasing 5°C in temperature, the amount of copper penetration in magnesium (L3) has increased, significantly. The thickness of this layer is about 10 µm, which shows the effect of temperature on the penetration of copper in magnesium. It is observed that the penetration coefficient exponentially increases with increasing the temperature. 28 In other words, minor changes in temperature cause significant changes in the penetration rate. Therefore, the effect of temperature on the type and thickness of the formed phases is much more than that of time. As the temperature increased, the mobility of atoms will also greatly increase and lead to more penetration and formation of the supersaturated solid solutions with higher thicknesses.

Optical microscopic image of interface bonding at 30 min holding time and (a) 475°C, (b) 480°C.
SEM analysis
Figure 8 shows SEM images of joints performed at 30 min. duration and temperatures of 475°C and 480°C. It is observed that the penetrating layer is formed at the copper-magnesium and copper-aluminium boundaries. As can be seen, the penetrating layer of copper-magnesium in Figure 8(a) is partial; but, in Figure 8(b), the thickness of this layer has increased significantly.

SEM images of joint at 30 min holding time and (a) 475°C, (b) 480°C bonding temperature.
Figure 9 shows the linear analysis of the joints performed at 30 min. duration and 475 and 480°C temperatures. The distribution of the magnesium and aluminium as well as the copper element is shown in this Figure. At all the tested temperatures, changes in the concentration of aluminium and copper atoms in the L1 zone are observed. The L1 zone has two separate parts; the first one is on the side of the copper interface layer and has 17.71 wt.% aluminium and 75.74 wt.% copper. The other part is adjacent to aluminium side and has 47.5 wt.% aluminium, 39.9 wt.% copper and 13.1 wt.% magnesium. The L2 zone remains unchanged and contains only the copper element. L3 region also contains 14.14 wt.% magnesium and 89.07 wt.% copper. Due to the higher concentrations of copper and magnesium, the L3 zone in Figure 9(b) has a wider range than L3 zone in Figure 9(a), which indicates a greater penetration of copper in magnesium at 480°C than at 475°C.
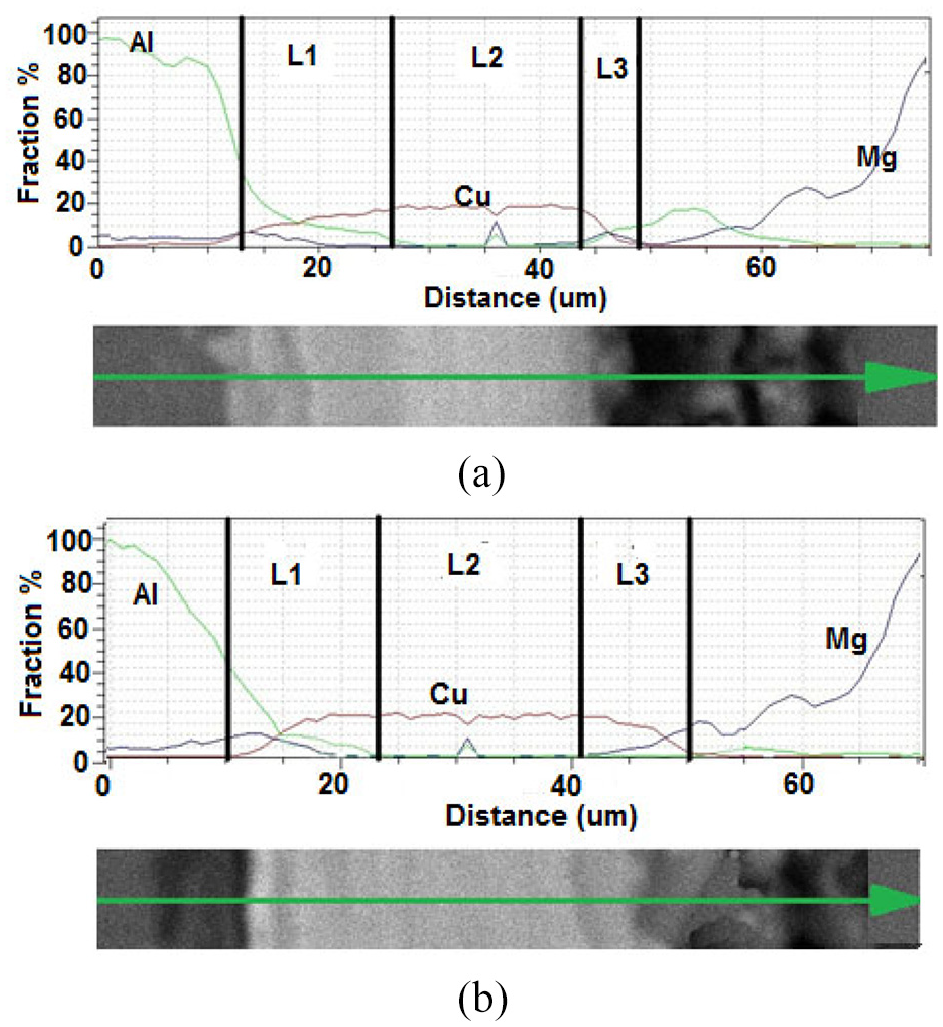
EDS analysis of Al7075/MgAZ31 joint performed at 30 min holding time and (a) 475°C, (b) 480°C boning temperature.
The most important factors that determine the penetration rate of an element are the atomic radius, density and melting temperature of the element. The crystal structure of elements and alloys affects their penetration rate; so that, the atoms penetrate faster in more open structures. The atomic radii of copper, aluminium and magnesium are reported to be 1.35, 1.43 and 1.6 angstroms; respectively. 4 Smaller atoms have a faster penetration rate due to the lower activation energy. Therefore, the rate of penetration and, as a result, the penetration depth of copper in aluminium is higher than that of magnesium. Additionally, as the temperature rises up to 480°C, a decrease in the concentration gradient between magnesium-copper and aluminium-copper atoms is observed in different joint zones. According to Al-Cu binary diagram 27 in the aluminium rich section, after the penetration and increase of copper in aluminium and as a result of an increase in temperature, the solubility of copper in phase α increases and with cooling down to the ambient temperature, the formation of the θ structure (AlCu2) is expected.
Effect of bonding duration
OM analysis
Figure 10 is an optical microscope image of a sample bonded at 480°C for 45 min duration. With an increase in time from 30 to 45 min, the average penetration of copper in aluminium and magnesium reaches more than 10 µm. Figure 11 shows the changes in the thickness of the copper penetration zone in aluminium (L1) and magnesium (L3) with an increasing time at a constant temperature of 480°C.

Optical microscopic image of interface bonding at 45 min holding time and 480°C temperature.
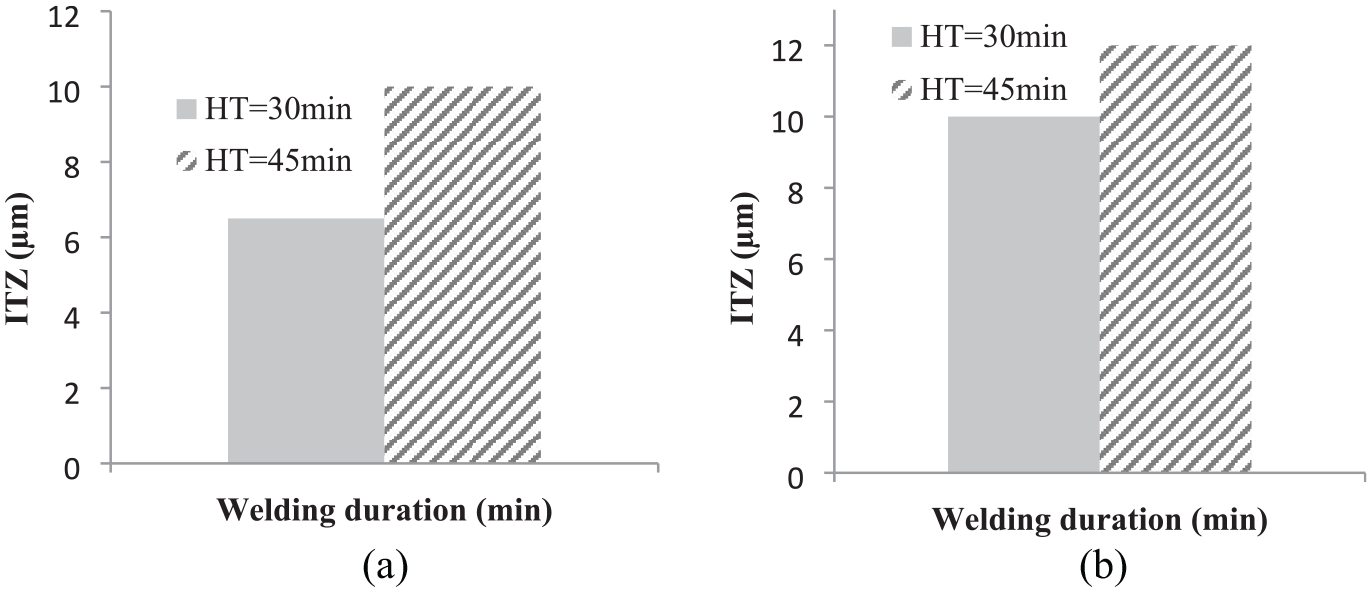
Effect of holding time on ITZ at 480°C in (a) L1 Al/Cu and (b) L3 Mg/Cu.
SEM analysis
Figure 12(a) and (b) show SEM images of joints performed in 30 and 45 min. duration, respectively, while the process temperature was kept at 480°C. As can be seen, the longer the bonding duration is, the more complete the joint would be. In SEM images, the increase in the thickness of L1 and L3 can be clearly seen with increasing the time. In addition, the thickness of the copper-magnesium and copper-aluminium transfer zones increases, resulting in the possibility of the formation of intermetalic compounds. Above the L3 layer, larger sediments are seen in the Figure 12(b). It is thought that the reason for the increase in these sediments is that they take longer time to form. Figure 13 shows the results of a linear analysis of the joints made at a constant temperature of 480°C and holding times of 30 and 45 min. In this sense, with increasing the time, a decrease in the slope of the concentration gradient occurs in zones L1 and L2. At 45 min, the L3 zone contains 19.99 wt.% magnesium, which shows an increase of 5.85% in weight as compared to 30 min. bonding duration. Percentage of copper weight was calculated as 81.09, which is 8.02% less than 30 min. duration.

SEM image of joint performed at 480 °C temperature and (a) 30 min, (b) 45 min holding time.
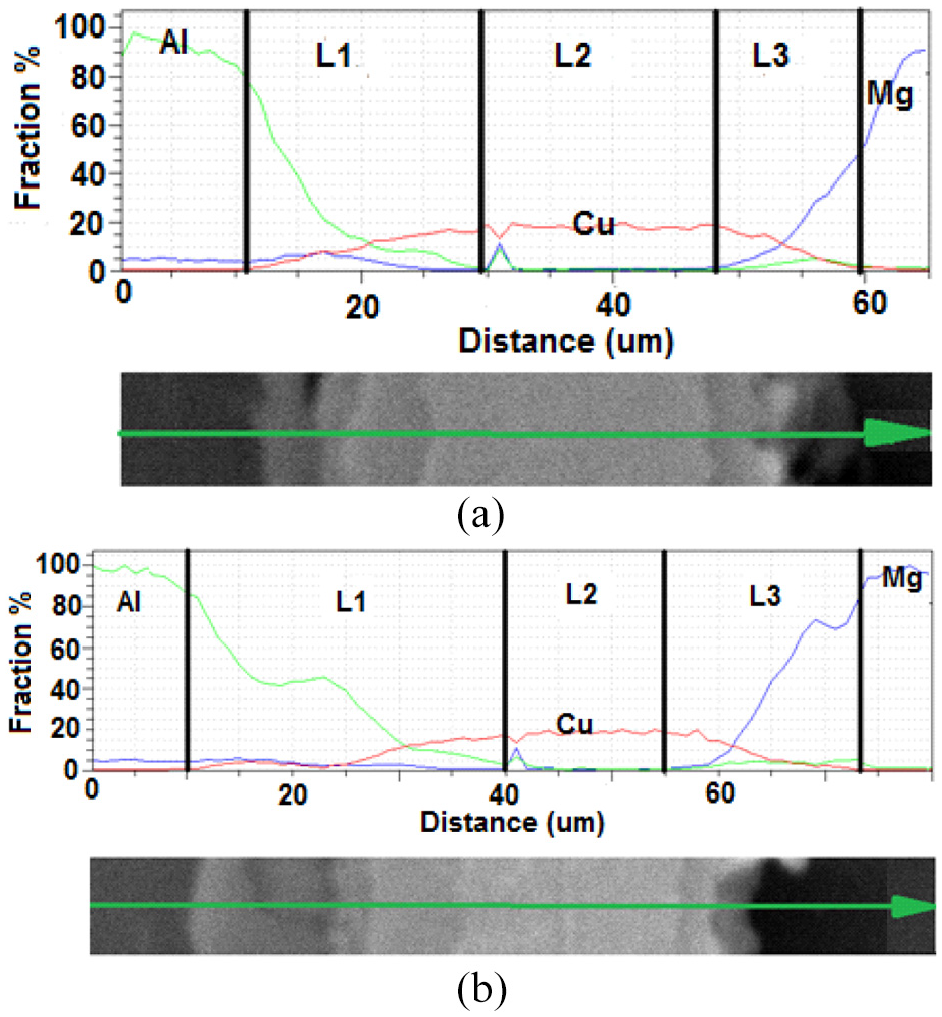
EDS analysis of Al7075/MgAZ31 joint performed at 480 bonding temperature and (a) 30 min, (b) 45 min holding time.
Point analysis of the joints
Figures 14 and 15 show the results of plane analysis of the aluminium alloy 7075 and magnesium AZ31 joints performed at 480°C for 30 and 45 min holding time, respectively. As can be seen, copper penetrates both aluminium and magnesium alloys and, as it approaches these two alloys, its concentration decreases gradually. Copper is also more diffusible to aluminium. This may be due to the weakening of the molecular bonds on the aluminium side and the easier penetration of the copper atoms towards the aluminium.

Element map analyses for the sample joint at 480°C and 30 min.

Element map analyses for the sample joint at 480°C and 45 min.
Figure 16 shows the SEM image joints performed at 30 min holding time and 475°C and 480°C. As can be seen, the amount of sediment formed in the AZ31 magnesium alloy also increases with increasing the bonding temperature at a constant time. Analysis of these sediments is also shown in Figure 17. These deposits in AZ31 magnesium include magnesium (47.18 wt.%), aluminium (38.12 wt.%), manganese (13.24 wt.%) and copper (1.47 wt.%). Figure 18 also shows the results of EDS analysis of sediments on the 7075 aluminium side. Evidently, the sediments on the 7075 aluminium side contain aluminium (70.78 wt.%), iron (20.93 wt.%), copper (4.76 wt.%), zinc (1.30 wt.%), magnesium (1.21 wt.%) and manganese (1.03 wt.%).

SEM image of Al7075/MgAZ31 joint obtained at 30 min holding time and (a) 475°C, (b) 480°C.
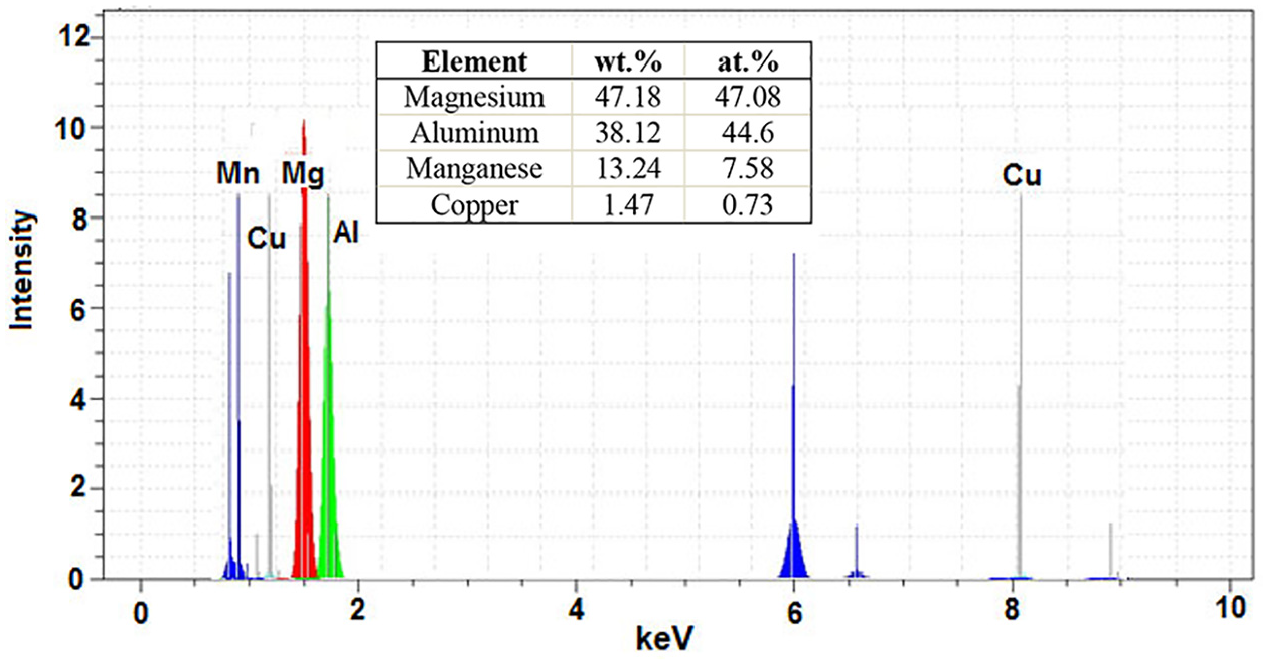
EDS spectra from precipitate point marked by letters A on Figure 16(b).
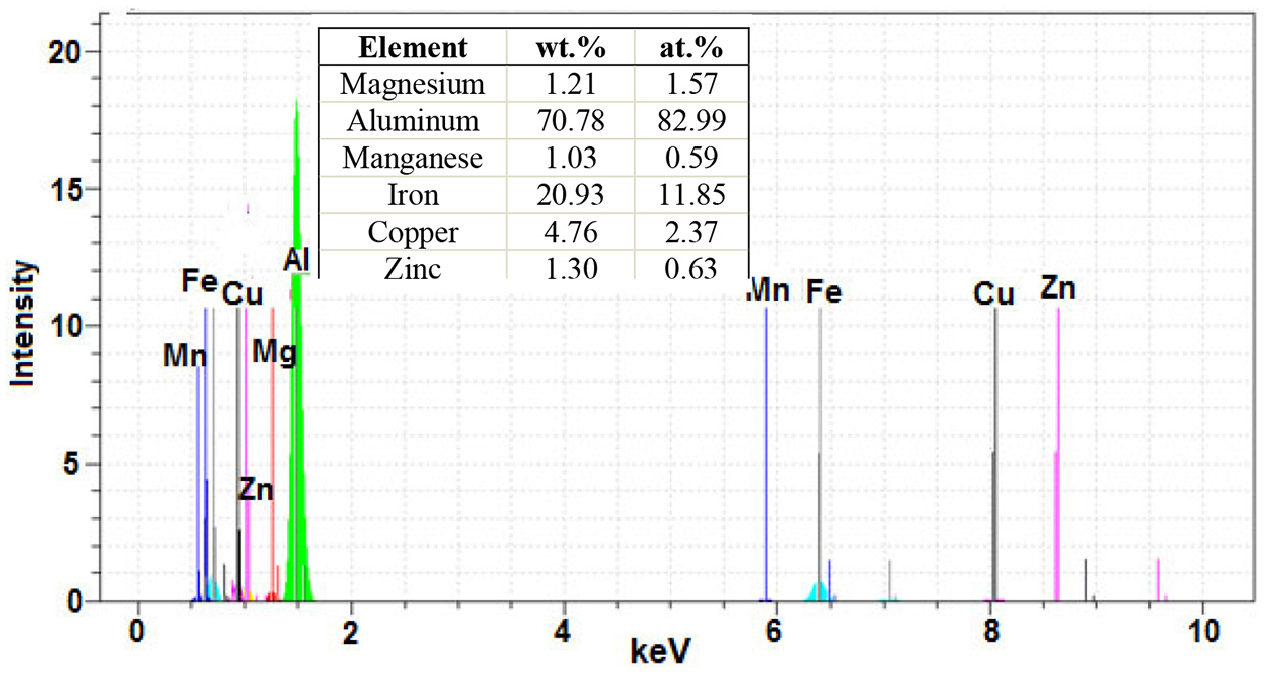
EDS spectra from precipitate point marked by letters B on Figure 16(b).
Figure 19 shows the different bonding zones formed in the joining of Al 7075/Mg AZ31B performed at 480°C and 30 min. In the aluminium side, two zones are identified, one towards aluminium and the other towards copper. Phase CuAl2 is formed on the aluminium side and phase Cu9Al4 is formed on the copper side.

SEM image of Al7075/MgAZ31 joint obtained at 30 min holding time and 480°C.
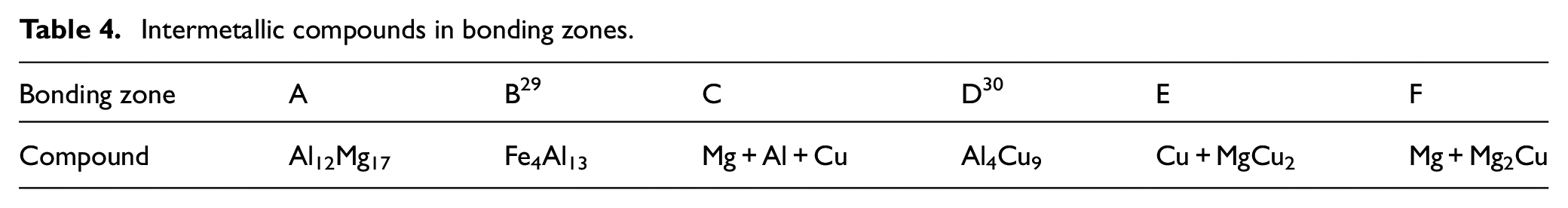
Figure 20 shows the analysis of the zones marked in Figure 19. According to the EDS analysis and considering the weighted percentage of zone C, which includes 40.26 wt.% aluminium, 41.06 wt.% copper and 18.15 wt.% magnesium, in addition to CuAl2 phase, there is a possibility of forming three Al-Mg-Cu phases in this zone. Zone D also has 14.91 wt.% aluminium and 84.4 wt.% copper, which can be regarded as phase χ (Cu9Al4). Point E contains 12.23 wt.% magnesium and 87.77 wt.% copper. Due to the weighted percentage obtained and the binary magnesium-copper diagram, the formation of Cu2Mg phase is expected. Point F also has 70.04 wt.% magnesium, 8.69 wt.% aluminium and 22.28 wt.% copper. This zone, like C and in addition to the CuMg2 phase, has the three Al-Mg-Cu phases. The formation of intermetalic compounds is an important issue in bonding dissimilar metals because the strength of the joint is significantly affected by these compounds. In this study, regarding the use of different Al-Cu–Mg ratios, provided by EDS analysis, and also Al/Cu and Mg/Cu binary diagram in addition to the previously reported studies (applying XRD and EDS analysis simultaneously), the possibility of intermetallic compounds’ formation in different bonding zones is shown in Table 4.
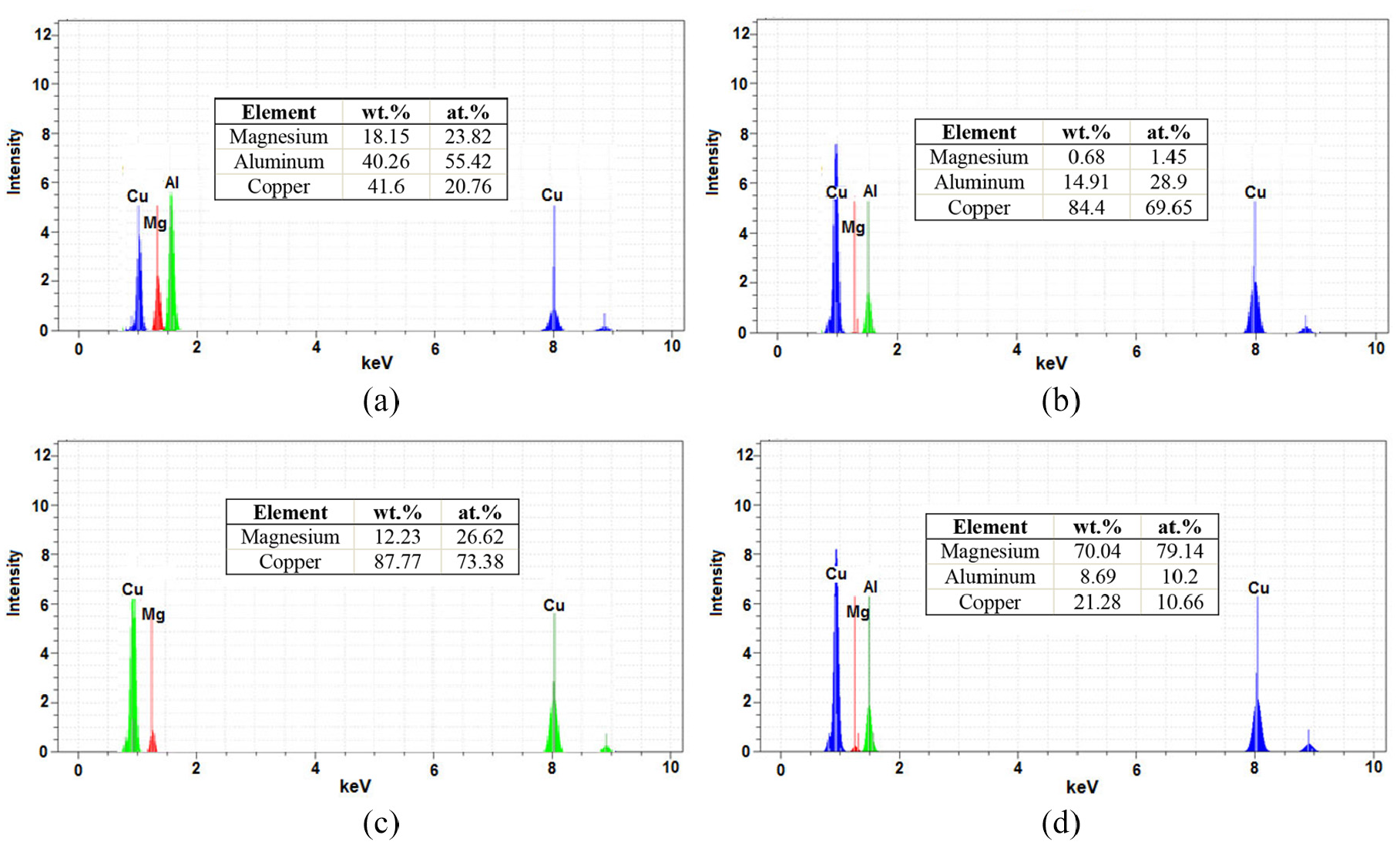
EDS of joint at 480°C and 30 min at point: (a) C, (b) D, (c) E and (d) F on Figure 19.
Intermetallic compounds in bonding zones.
In contrast to phase changes in molten state, homogeneous nucleation will rarely occur in solid state phase changes, but heterogeneous nucleation occurs in places such as impurities, boundaries and dislocation areas. According to the theory of diffusion, the formation of the initial phase in the dissimilar metals bonding will not be immediate at a certain temperature, but it needs a latent period of time. Internal diffusion in the intersection occurs to a certain extent in the latent period. 31
Mechanical evaluation
Micro-hardness
Figure 21 shows the micro-hardness profile within the bonded joints obtained at a duration time of 30 min and temperatures of 475°C and 480°C. The general distribution of micro-hardness at different temperatures on the aluminium and magnesium side is similar and the amount of micro-hardness suddenly increases in the diffusion zone. Due to the complete lack of copper penetration and the presence of pure copper, the reduction of hardness is observed in the central line of the joint. It can be concluded that the hardness in the bonding zone at the 480°C is 158 HV and; at the 475°C, it is about 100 HV. A decrease in temperature leads to a decrease in the formation of intermetallic phases. As a result, the micro-hardness is reduced.
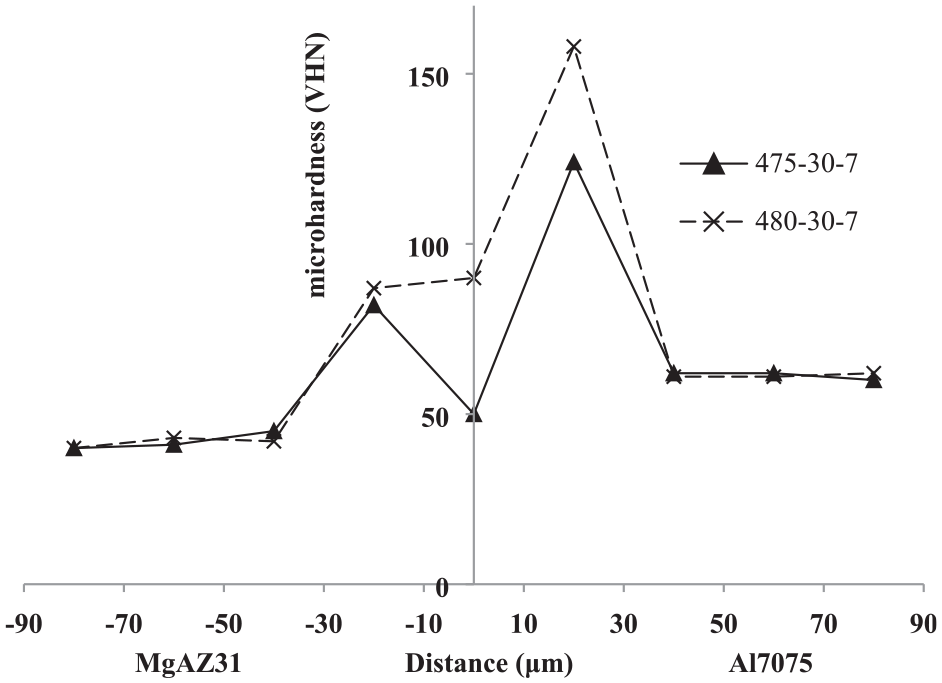
Microhardness profile peripendicular to the intersection of Al7075/MgAZ31 joint performed at 30 min, 475°C and 480°C.
Figure 22 shows the micro-hardness profile of the process completed at 480°C and 30 and 45 min. durations. In this sense, the hardness in the bonding zone is 167 HV at 45 min and 158 HV at 30 min. After comparing the effect of process temperature and process duration on the micro-hardness, it can be concluded that increasing the time can increase the formation of intermetallic compounds and; thus, increase the hardness.
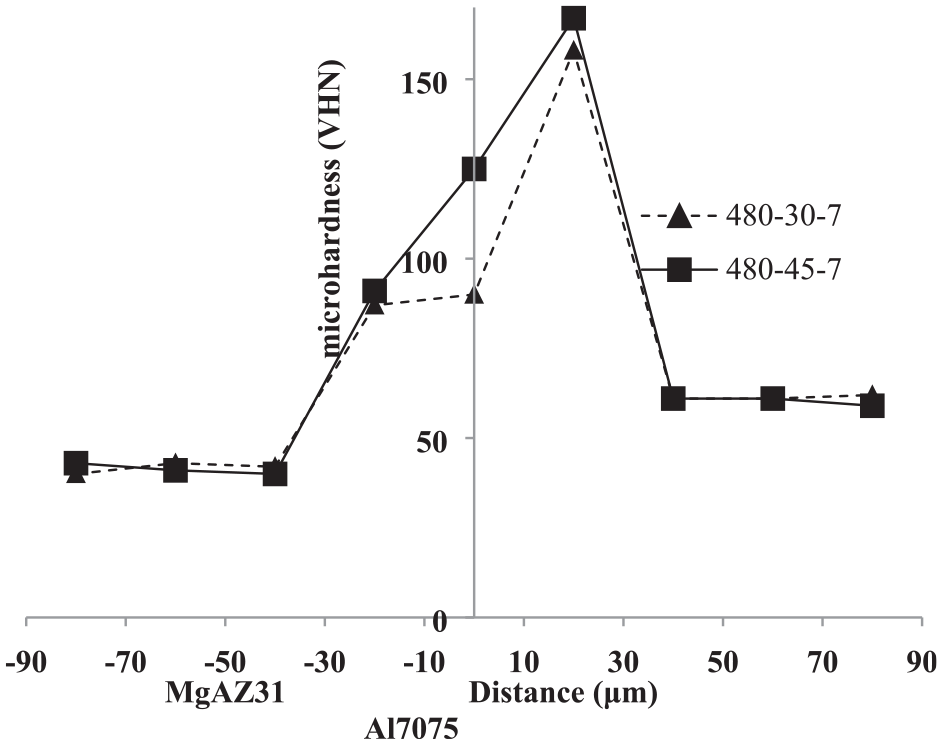
Microhardness profile peripendicular to the intersection of Al7075/MgAZ31 joint performed at 480°C and 30 min and 45 min.
Shear strength
Figures 23 show the force-displacement diagrams of Al7075/MgAZ31 joint obtained at 475°C, 480°C and 30 min. duration. As can be seen, with increasing the temperature from 475°C to 480°C, the shear strength of the joint increases from 13.02 to 16.33 MPa. Due to the constant pressure and time, increasing the bonding temperature leads to more penetration of elements and, then, results in increasing the shear strength.
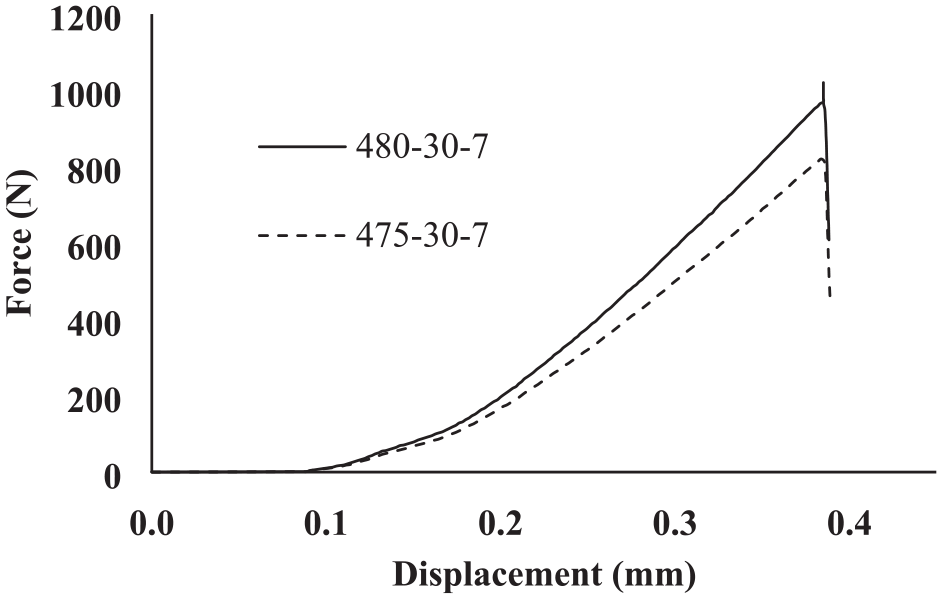
Force-displacement diagram for Al7075/MgAZ31 joint performed at and 480°C, 475°C and 30 min.
Figures 24 show the force-displacement diagrams of Al7075/MgAZ31 joint obtained at 30 and 45 min durations and 480°C. As the time increases from 30 to 45 min, the strength of the joint increases from 16.33 to 31.03 MPa. Due to the increase in time and the resulted diffusion, the strength increases significantly. However, it is thought that increasing the time to higher values, due to the formation of more intermetallic compounds, will lead to a decrease in shear strength.
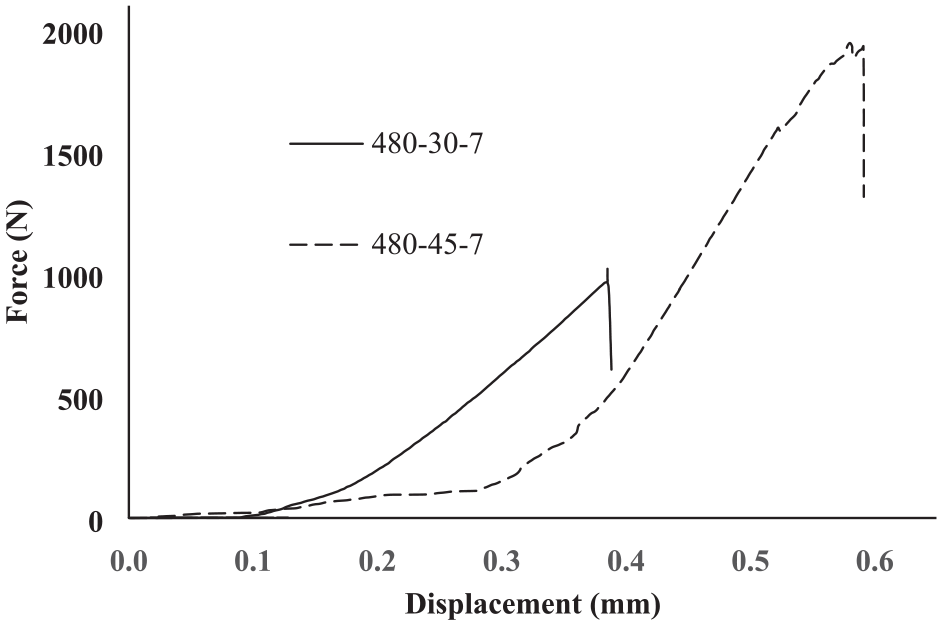
Force-displacement diagram for Al7075/MgAZ31 joint performed at 30, 45 min and 480°C.
Comparison between Al7075, 6061 and 5083 bonded to MgAz31
Figures 25 and 26 show the optical microscope images and SEM images of the components made of magnesium alloys AZ31 bonded to 6061 and 5083 aluminium alloys at 480°C and 45 min, respectively. According to the SEM images shown in Figure 26(a) and (b), it can be concluded that the amount of copper diffusing in 5083 and 6061 aluminium alloy is almost the same as the value seen in aluminium alloy 7075. Analysis of the points marked in Figure 26(b) are shown in Figure 27. Based on these results, it is expected that the chemical composition of the intermetallic phases formed in the bonding of Al5083 and Al6061 alloys is similar to that of Al7075 alloy.

Optical microscopic image of interface bonding at 45 min holding time and 480°C bonding temperature for: (a) Al5083/MgAZ31 and (b) Al6061/MgAZ31 joint.

SEM image of: (a) Al5083/MgAZ3, (b) Al6061/MgAZ31 joint obtained at 45 min holding time and 480°C.
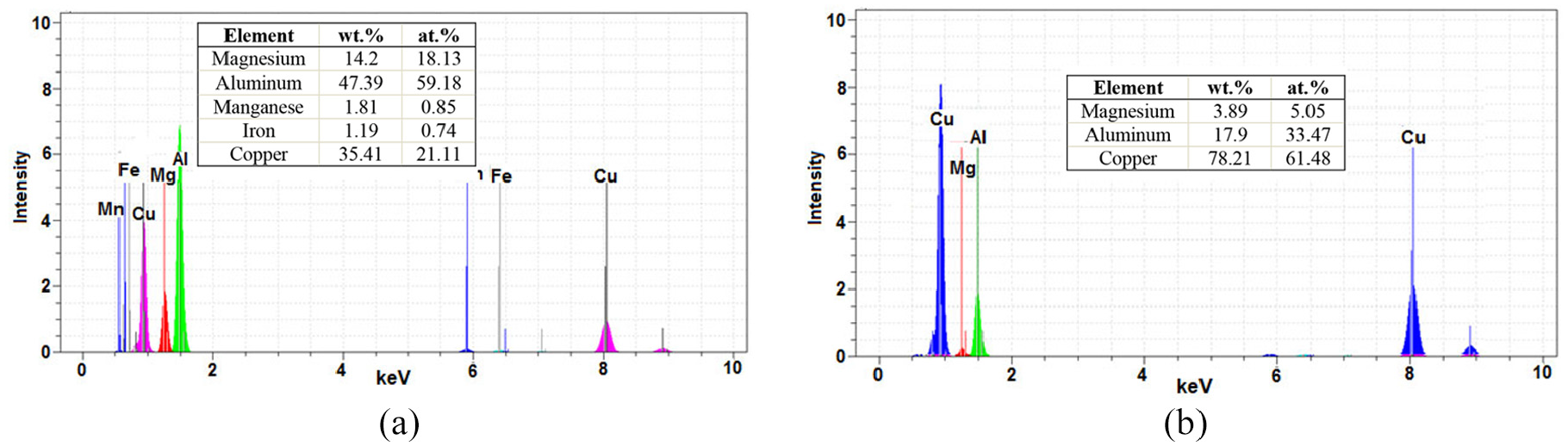
EDS spectra from Al6061/MgAZ31 joint performed at 480°C and 45 min at point marked by letters: (a) C and (b) D on Figure 26(b).
In terms of comparing the mechanical properties of the obtained joints, Figure 28 shows the micro-hardness results related to three aluminium alloys (7075, 6061 and 5083) bonded to the MgAZ31. As can be seen, there is no significant difference between the micro-harnesses obtained between different aluminium alloys bonded to Mg-AZ 31. Following the study of mechanical evaluation, the force-displacement diagrams of Al7075, Al6061 and Al5083/Mg-AZ31 joints performed at 480°C and 45 min bonding duration is also shown in Figure 29. The highest strength was related to the 7075 aluminium alloy, which was equal to 31.03 MPa. The other obtained strengths were 25.51 MPa and 30.76 MPa for 6061 and 5083 aluminium alloys, respectively. Accordingly, it can be concluded that the mechanical properties obtained in Al5083, Al6061 and Al7075 alloys bonded to MgAZ31 do not show a significant difference between the obtained results. The only difference is related to the highest shear strength of the Al7075/MgAZ31, which is also due to the Al7075 alloying specification. Sheng et al., 32 in Al 6061-O bonding to Mg AZ31 at 440°C temperature, 30 min duration and 140 MPa pressure, achieved the highest shear strength of 37 MPa. In this regard, the hardness was also obtained in the diffusion zone equal to 240 HV. Moreover, Fernandez et al. 33 in Al-6061-T6 bonding to Mg AZ31-B at 430°C, 30 min time and 13.148 MPa pressure, achieved the highest shear strength equal to 49.39 MPa.
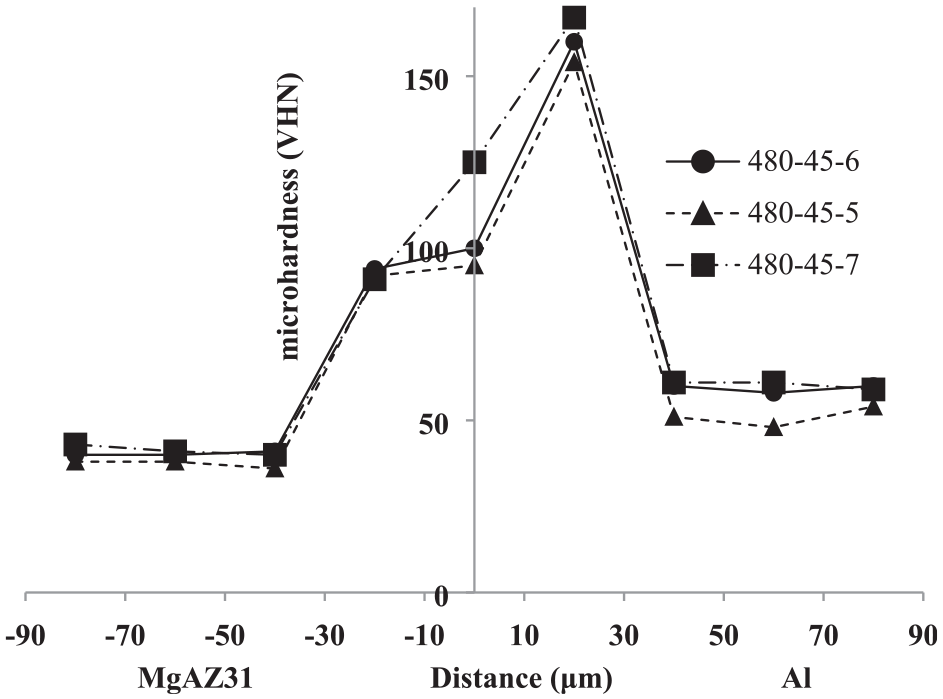
Microhardness profile peripendicular to the intersection of Al7075, Al6061 and Al5083/MgAZ31 joint performed at 480°C and 45 min.
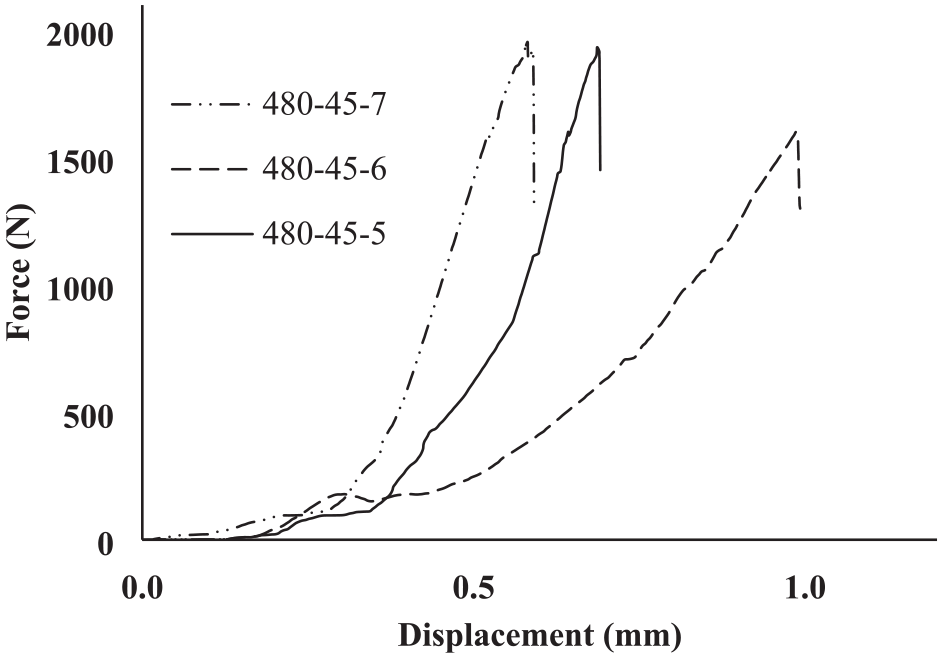
Force-displacement diagram for Al7075, Al6061 and Al5083/MgAZ31 joint performed at 45 min and 480°C.
Conclusions
In this study, 5083, 6061 and 7075 aluminium alloys were successfully bonded to AZ31 magnesium with pure copper interface layer. Besides, it was without the slightest macroscopic deformation. According to the conducted research, the following results can be presented:
In all cases except the conditions in which the temperature was 470°C and the duration time was 30 min, a bond was formed. The most appropriate process conditions are temperature range 475°C to 480°C and minimum duration of 30 min. Bonding will not occur at temperatures below 475°C, and at temperatures above 480°C the work will be subject to large deformation.
The bonding processes of 5083, 6061 and 7075 Al alloys to AZ31B Mg were similar. The only difference is related to the obtained mechanical strength, which is due to the alloying structure. The highest mechanical strength is related to 7075 Al/AZ31 Mg alloy bond.
According to the results, increasing the temperature and time led to increasing the thickness of different intermediate layers in the transition zone.
Scanning electron microscopy results indicate the formation of CuAl2, Cu9Al4 and Al-Mg-Cu ternary phases on the aluminium-copper side, as well as Cu2Mg, CuMg2 and Al-Mg-Cu ternary phases on the magnesium-copper side in the transition zone. Additionally, as the temperature increases, the amounts of these compounds also increase.
Temperature has a great effect on the micro-hardness; so that, the micro-hardness of the performed joint at 475°C and 30 min duration was equal to 100 HV, while with only 5°C increase in temperature, the hardness increased to 158 HV. Unlike temperature, the bonding duration has a large effect on shear strength, in such a way that, with increasing the duration from 30 to 45 min, the amount of shear strength increased from 16.33 to 31.03 MPa (at 480°C bonding temperature).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.