Abstract
Marine structures such as ships, oil platforms, and oceanographic systems are permanently at risk of fouling affecting the performance of these structures. Antifouling paints were used to address the concerns associated to marine organisms, but they release toxic materials. This paper presents a new one-step environmentally friendly super hydrophobic coating considering the effect of particle size as well as weight percentage which is feasible in large-scale applications. Firstly, the aluminum flakes are become hydrophobic using silanization method. Then, they are mixed with polyester triglycidyl isocyanurate (TGIC) and are sprayed electrostatically. To assess the fabricated surfaces, effect of mixing time, weight percentage and particle size on the super hydrophobicity are evaluated using scanning electron microscope (SEM), energy-dispersive X-ray spectroscopy (EDS), contact angle measurement, and image processing. This method leads to a super hydrophobic coating with water contact angle about 152°. The super hydrophobic surface with particle size less than 45 µm results in higher resistance to algae settlement providing an excellent antifouling feature. The uniformity resulted from the particle size range 0–45 µm prevents any micro organisms settlement on the surface so that, after sixth day only some tiny algae are appeared on the surface sides.
Introduction
Regarding the ban in 2008 on the use of the application of antifouling (AF) coatings based on organotin compounds, such as tributyltin (TBT) and triphenyltin, the demand for an environmentally-friendly alternative has increased. 1 Use of biocides by considering the half-life of the compounds allowing a perpetuation of the toxic effects into the marine food chain and initiates changes in the early stages of the organisms’ life-cycle. 2 According to the precautionary principle, the strategy of using non-toxic materials is the safest method to adopt. On the other hand, if there is no effective non-toxic alternative, the potential spread of marine invasive species around the world could be ecologically dangerous as toxic paintings are. Hence, it is important to examine the non-toxic antifouling coatings technology potentially in details and its commercial capacity. A wide variety of ways as non-toxic methods are raised to prevent environmental sediments, and one of them is hydrophobic surfaces. A hydrophobic surface is known as the surface with a contact angle greater than 90° and water droplets have a low affinity to be spread on this surface. Then, when a rough surface, particularly at the nanoscale is combined with hydrophobic substances, super-hydrophobic surfaces are formed.3,4 Super hydrophobic coatings have a contact angle of 150° or even more along with low sliding angle lower than 10°. 5 Recently various approaches to create super hydrophobic materials have been developed. These methods include chemical vapor deposition (CVD),6,7 hydrothermal techniques,8,9 sol–gel techniques,10–12 electrospinning method, 13 and lithographically etch.14,15 Generally, micro-nano roughness and low surface energy are two keys features of fabricating super hydrophobic surfaces.16,17 Some of the researches done on the fabrication of these surfaces have focused on the antifouling properties of the surfaces. It was observed that super-hydrophobic surfaces, based on nanometer fumed silica and polydimethylsiloxane (PDMS), resulted in the surfaces became moist after 6 months and this led to short-term connection with bacteria. Reduced resistance against fouling can be attributed to the loss of super hydrophobic properties occurring due to the dissolution of air in the water, in long periods. This is due to degradation of pores that physically remove the air from the liquid-solid surface or reposition of macro molecule layers that chemically and physically modify the surface structure. 18
It was noted that a surface structure known as engineered pillars, ridges and biomimetic topography inspired by the skin of fast moving sharks were replicated in polydimethylsiloxane elastomer. It was reported the reduction of sediments resulting from Ulva Linza spores, a green algae, and the most successful surface (86% reduction of spores) has the highest contact angle 135°. 19 This reduction resulted from the surface topographic width which was smaller than the spores’ diameter. The same conclusion was presented indicating that the topographic height and spacing are more important than hydrophobic effect. 20 Adhesion strength of three common sediments of diatoms, including the adhesion of Amphora-caffeaeformis to the elastomer poly-dimethyl siloxan is stronger than to glass. It has been observed that the diatom often covers the hydrophobic antifouling coatings with low surface energy and binds to it very tightly. 21 Some important conclusions about the role of nano scale roughness in creating an antifouling surface were found and it was examined three hydrophobic surfaces with different roughness. Among these surfaces, the surface including nano-roughness is less sedimentary than the other two surfaces contain nano-roughness on micro-roughness. 22 Formation and testing of novel marine coatings comprising hierarchically wrinkled surface topographies (HWTS) including wrinkles of different length scales (generations) ranging from tens of nanometers to a fraction of a millimeter. The results showed that the HWST coatings remained relatively free of biofouling even after prolonged exposure to seawater (18 months). 23 Both nano- and micro-topographical structures created on the surface of a copper substrate by applying an electroless galvanic reaction between copper and the metal salt which was stable around 6 weeks in a marine environment in Dublin, Ireland. 24 A super hydrophobic surface on a magnesium alloy by a simple immersion process with a solution containing ferric chloride, deionized water, tetradecanoic acid and ethanol was fabricated. 16 The highest water contact angle of this coating was 165° that is considered as a factor indicating resistance to bacteria growth. Also, it was presented that super hydrophobic surfaces via surface-initiated atom transfer radical polymerization of fluorinated methacrylates on poly ethylene terephthalate (PET) fabrics. The obtained super hydrophobic fabrics showed excellent chemical robustness even after exposure to different chemicals, such as acid, base, salt, acetone, and toluene. 25
Generally, some of the surfaces of the super hydrophobic fabrics indicated an excellent antifouling property. Despite the various methods in the production of super hydrophobic coating, some of these methods have the ability to be used as anti-fouling coating. On the other hand, most of the researches which are done to produce super hydrophobic coating limited to producing laboratory samples and based on fairly complex, time consuming and difficult processes that are not cost effective. Also, the surface modifications in nanoscale for sediment control are in the early stage, and it is possible to combine the surface features with nano and micro scales to create a protective agent against sediment species with various sizes. Therefore, fabrication of robust and antifouling super hydrophobic surfaces with a proper durability, simple and economical procedures to be applied on a large surface, is one of the major challenges for researches and industry. In this study, a one-step as well as environmentally friendly process is introduced to fabricate a super hydrophobic surface. Here super hydrophobic aluminum flake particles are combined with polyester triglycidyl isocyanurate (TGIC) using electrostatic spraying technique to fabricate a super hydrophobic surface which is useful for large scale. To this end, effect of different process parameters such as mixing time, weight percentage, and particle size one super hydrophobicity of the surface are studied. In order to assess the fabricated surface, scanning electron microscope (SEM), energy-dispersive X-ray spectroscopy (EDS), contact angle measurement, and image processing are utilized. The results show that the mixed super hydrophobic aluminum flake structures with polyester TGIC provides an anti-fouling feature for surfaces, by enhancing the contact angle and creating a nano-micro structure, to obtain super hydrophobic coatings which are ideal for offshore, shipbuilding, and marine industries.
Materials and methods
In this section, materials, surface treatment of aluminum flake, preparation of polyester/Al flake composite, characterization instruments and antifouling property test are presented.
Materials
Fabricating a super hydrophobic coating requires both polyester resin and cure agent of triglycidyl isocyanurate (TGIC, 6 wt%) which is produced using a D30 twin-screw extruder and is cooled to room temperature. Then the received mixture is grinded to obtain polyester resin powders passing sieves with mesh size 150. For Aluminum flake powder, size range 45–250 μm is picked out from Mehreganchemist company. Aluminum specimens with the size of 2 cm×5 cm×0.5 cm were cut from an AA7075 sheet. Acetone (99%) and trichloromethylsilane (TCMS) with purity of 99% were obtained from Merck Co. For all of the experiments, deionized water with conductivity of 0.1 umho/cm is used if needed.
Surface treatment of Al flake
Surface treatment of Al flake is accomplished in two steps of activation and coating which are explained as followings.
Step 1: Activation
The fabrication process for activation is shown in Figure 1. First, to activate and create the OH group, aluminum flake powder is placed in boiling water for 60 min. Oxide layers are formed on aluminum when it is immersed into water. The formation of these layers is divided into three stages: formation of amorphous surface oxide, dissolution of surface oxide, and finally precipitation of hydrated aluminum oxide (Al2O3+n H2O) which n can vary from zero to three.
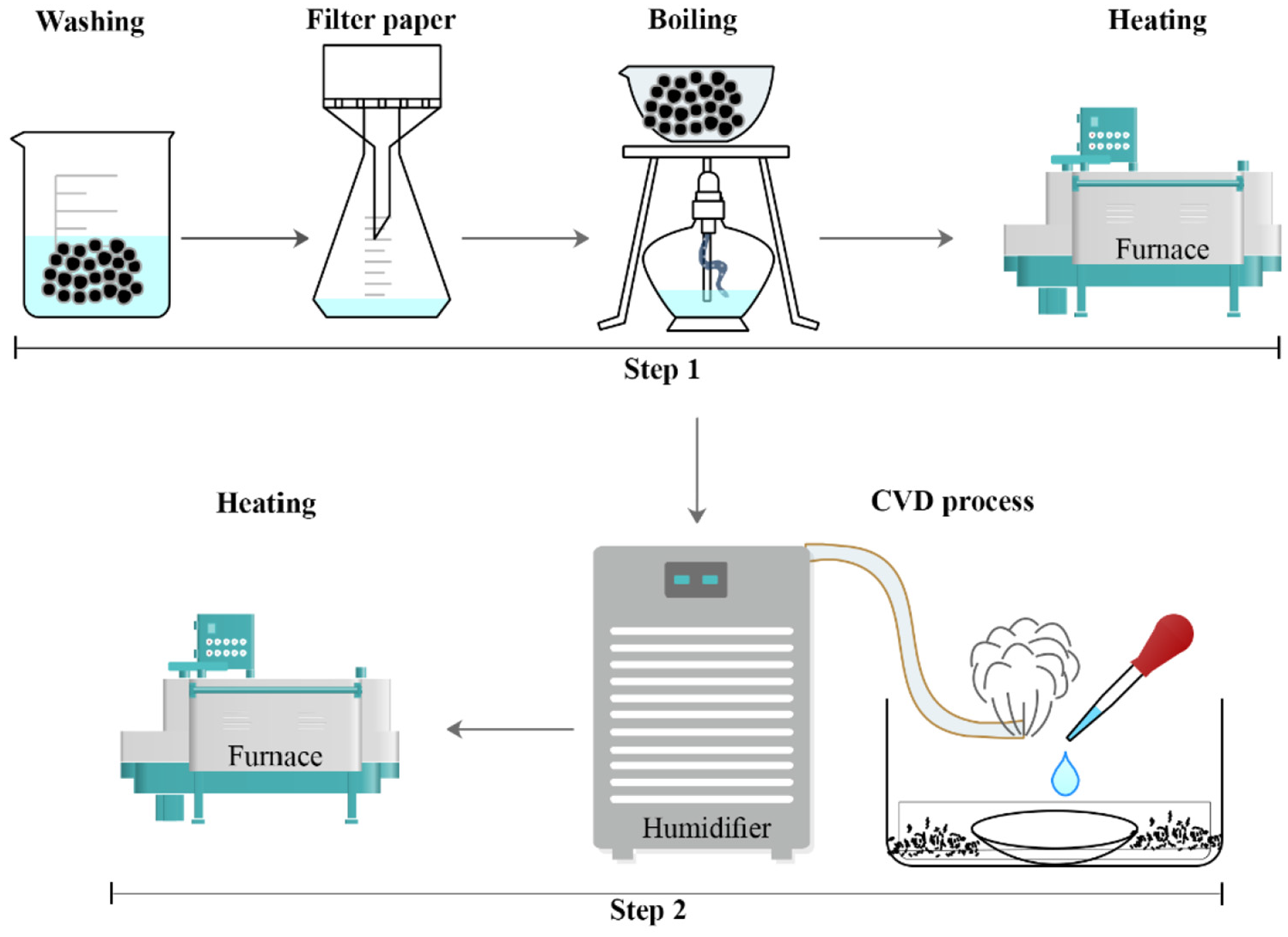
Schematic of preparing hydrophobic aluminum flake powder including two steps of activation and coating.
After the boiling process, aluminum powders were dried at temperature 120°C for 120 min in a radiative furnace and then cooled to ambient temperature. Schematic of the reaction between silane and hydrated aluminum oxide is shown on Figure 2.
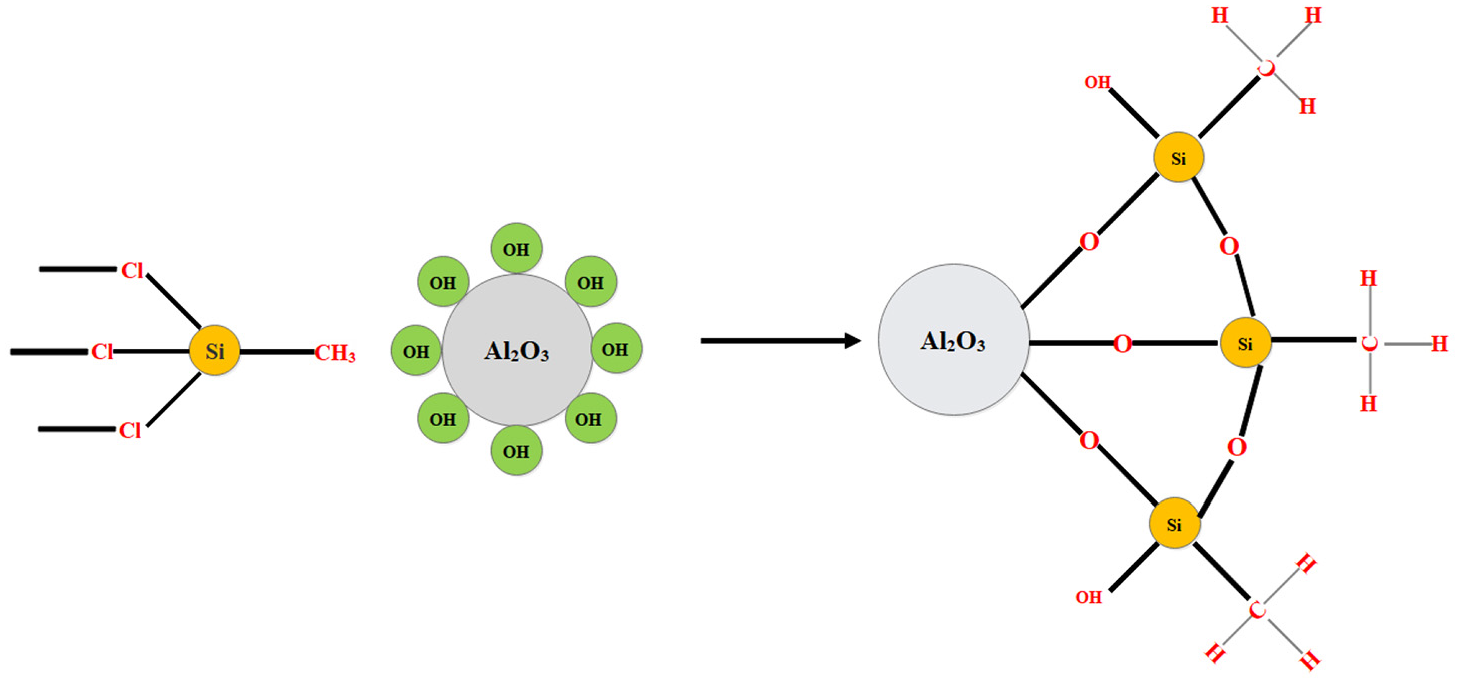
Schematic of surface modification of Al2O3 with TCMS.
Step 2: Coating
In terms of surface coatings, using silanes is a promising and well-known way. Silanes with hydrolyzable groups such as chloride, reacts with water to form silanols, which then can be coupled with hydroxy groups at the surface, and subsequently polymerize to a silicone layer. Chlorosilanes
In this method, as shown in Figure 1, the surface treatment was carried out in a gas phase in a custom-built reaction chamber. Due to high volatility and quick reaction of TCMS with water, the initiate condition and relative humidity are controlled precisely. Aluminum flake powder was placed in a chamber 20 cm3, while the Petri dish (8 cm diameter) was put in the middle of the chamber vessel. In a standard experiment, humidity was set between 30% and 65% at ambient temperature. 26 Then, 300 µl TCMS was pipetted into the Petri dish, without a direct contact between specimens and liquid. The suitable time for coating is within 6–12 h and typicality is completed about 12 h. Figure 3(a) shows the SEM images of aluminum flake after CVD process. As can be seen in the figure, after the coating process, nano structures on the micron aluminum powders are formed that leads to typical wetting state of Cassie-Baxter. Also, EDS analysis, shown in Figure 3(b), indicates that Si is settled on the aluminum flakes. The nano roughness causes an air layer between droplet and surface and repels the droplet.
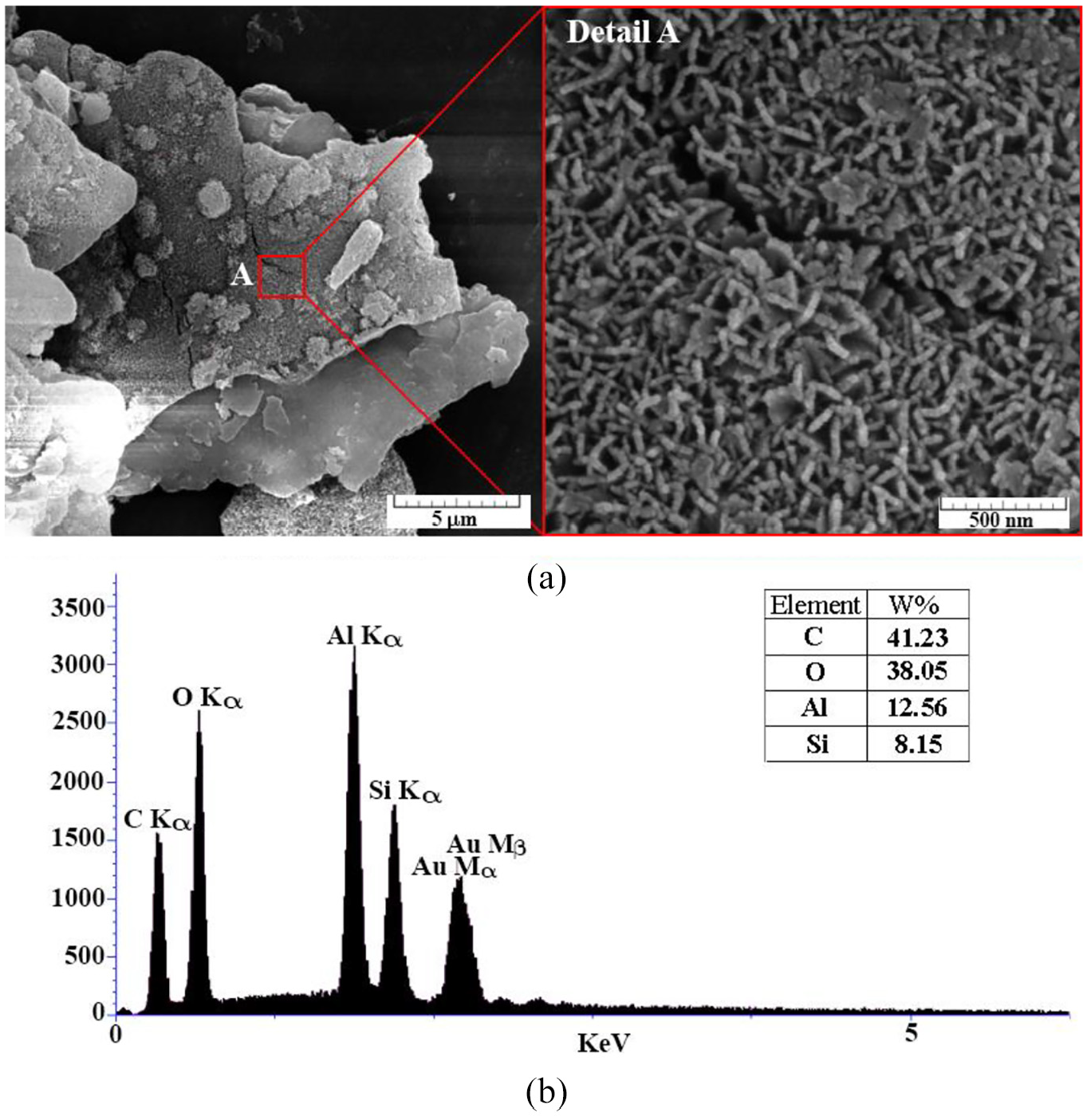
Aluminum flake after CVD: (a) SEM images and (b) EDS analysis.
Preparation of polyester/Al flake composite
In the electrostatic spray, uniformity of combination of resin and superhydrophobic particles, weight percentage as well as particles size in composites are counted as important factors in superhydrophobic surface fabrication. That is why after preparing superhydrophobic aluminum flake particles and sieving, these particles with different sizes and weight percentages were mixed with polyester resin in a tumbler containing ceramic beads (1–2 cm) and rotating at −80 rpm. In this process, mixing time and the ratio of ceramic beads weight to composite is important, to reach a uniform distribution. After obtaining the optimum mixing time, the prepared composite was put in a high-voltage electrostatic gun with 100-kV positive DC voltage.
The distance between the gun and aluminum substrates was 15 cm. The whole substrate was sprayed to get covered by powders. As some of the powders were distributed in the air, substrate could not be coated by them. Hence, more mixed powders should be placed in the electrostatic container vessel in comparison to what is needed for coating. Given amount of powder depending on the coating condition is achieved regarding the experiments which is about 0.05 g/cm2 to cover the whole surface. Then, coated samples put in an oven and warm up at temperature 200°C for 10 min. Figure 4 shows the schematic of the coating process.
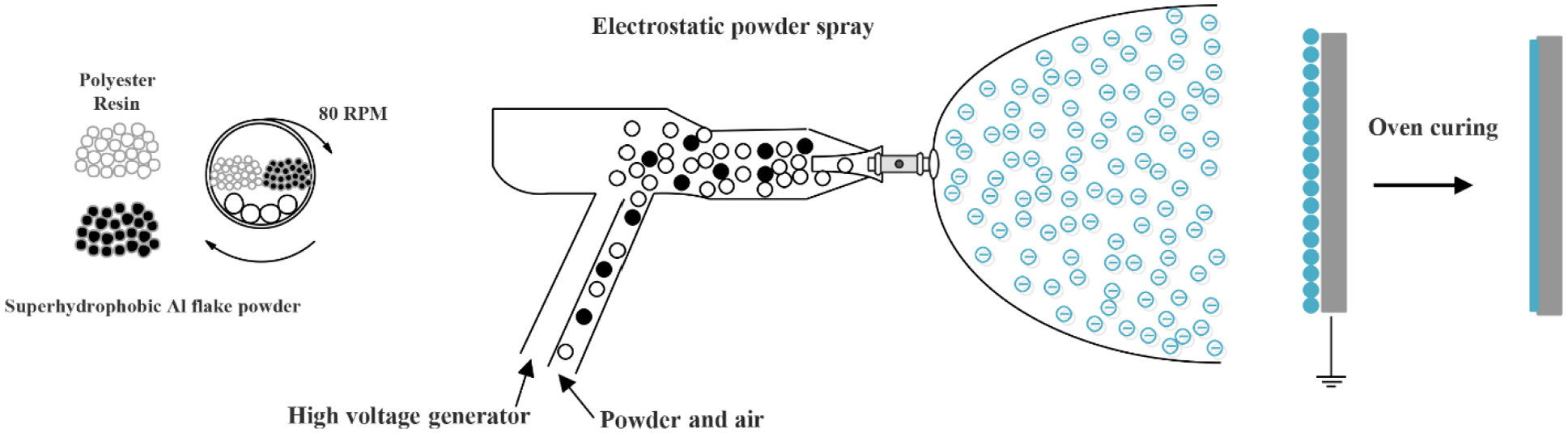
Schematic of the coating process.
Characterization instruments
To measure both static and dynamic water contact angles, a Dataphysics OCA 15 Plus instrument was utilized at room temperature (25 ± 1°C). Amount of 5 µL deionized, double-distilled water was used during the measurements. Number of five measurements were accomplished at five different points to obtain the contact and sliding angles for each sample.
Surface of the samples were analyzed using scanning electron microscopy (SEM) TESCAN VEGA//XMU. A thin Au layer (5 nm) was deposited on the specimens, prior to SEM, to capture high quality images. Additionally, elemental analysis is done using energy-dispersive X-ray spectroscopy (EDS).
Antifouling property test
In order to assess the fabricated super hydrophobic coatings and their resistance to fouling environment, the desired samples were put into an environment containing micro algae. This micro algae in the Walne’s medium was cultivated. To assess the samples, the amount of on the super hydrophobic coatings are measured over time by image processing. It should be noted that the effect of super hydrophobic particles size on the fouling settlement are evaluated.
Results and discussion
Regarding the International Maritime Organization (IMO) roles about utilizing coating compatible with environment and as the latest technology of antifouling coating (foul-release coating) is not efficient enough for static parts, super hydrophobic coatings could be a proper alternative. Despite of different methods used in fabricating super hydrophobic coatings, only some of these methods are feasible in antifouling coating. On the other hand, most of the researches have been done to produce super hydrophobic coatings are limited to lab scales. In this research, a one-step method is presented which is applicable for coating of big surfaces. The combination of the aluminum flakes, in different sizes and weights, with the polyester (TGIC) are sprayed using electrostatic coating. Followings are the results of mixing time, weight percentage of super hydrophobic aluminum flake powders, powders size effects and antifouling properties.
Evaluation of mixing time effects
Uniformity of the combination of polyester resin and aluminum flake in the electrostatic spray process is important when fabricating surface. Mixing time directly affects the dispersion of aluminum flake. To obtain the mixing time range, super hydrophobic aluminum flakes with weight ratio 15% along with some ceramic beads were put into the tumbler. Figure 5 illustrates the SEM images of aluminum flakes and influence of mixing time.

SEM images of aluminum flakes, polyester TGIC and the mixing time.
According to the figure, it can be concluded that the mixing time more than 30 min give rises to more breakage of super hydrophobic particles which is also reported in the literature 27 as well. Further particle breakage leads to lower roughness because when combination of resin and particles cured in the furnace, resin covers the whole surface of particles. Figure 6 illustrates EDS results of super hydrophobic aluminum flakes for different mixing time.
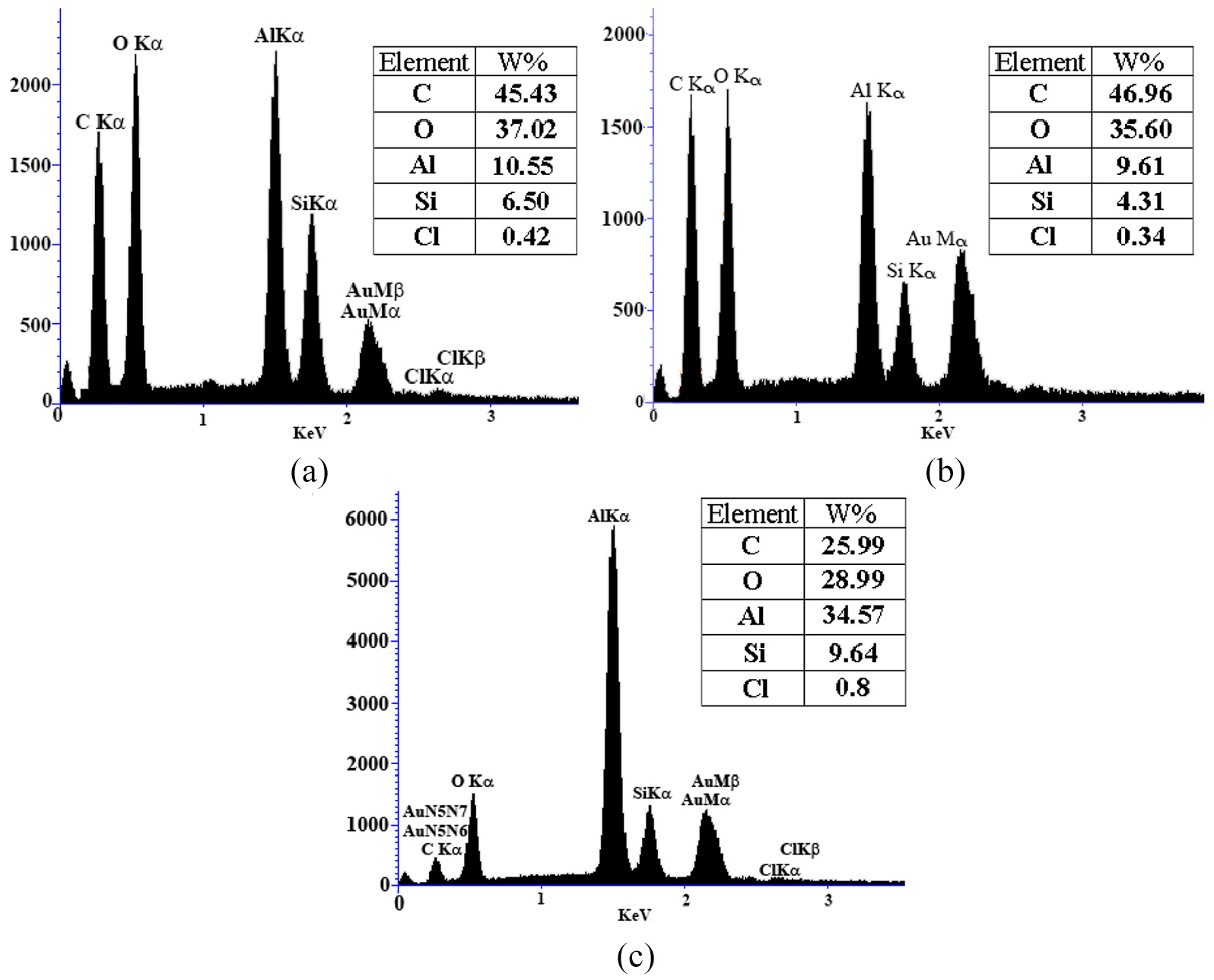
EDS results of the superhydrophobic aluminum flakes: (a) without mixing, (b) mixing time 30 min, and (c) mixing time 60 min.
As can be achieved from the EDS analysis, calculating the ratio of Si to Al shows that using many ceramic beads leads to particle wear and amount of Si on the aluminum flake particles which is shown in Figure 7. As can be seen from the figure, ratio of Si/Al decreases over the mixing time so that by enhancing the time from 30 to 60 min, this ratio goes down further as Si layers settled outside of aluminum flakes are worn out further while contacting with ceramic beads.
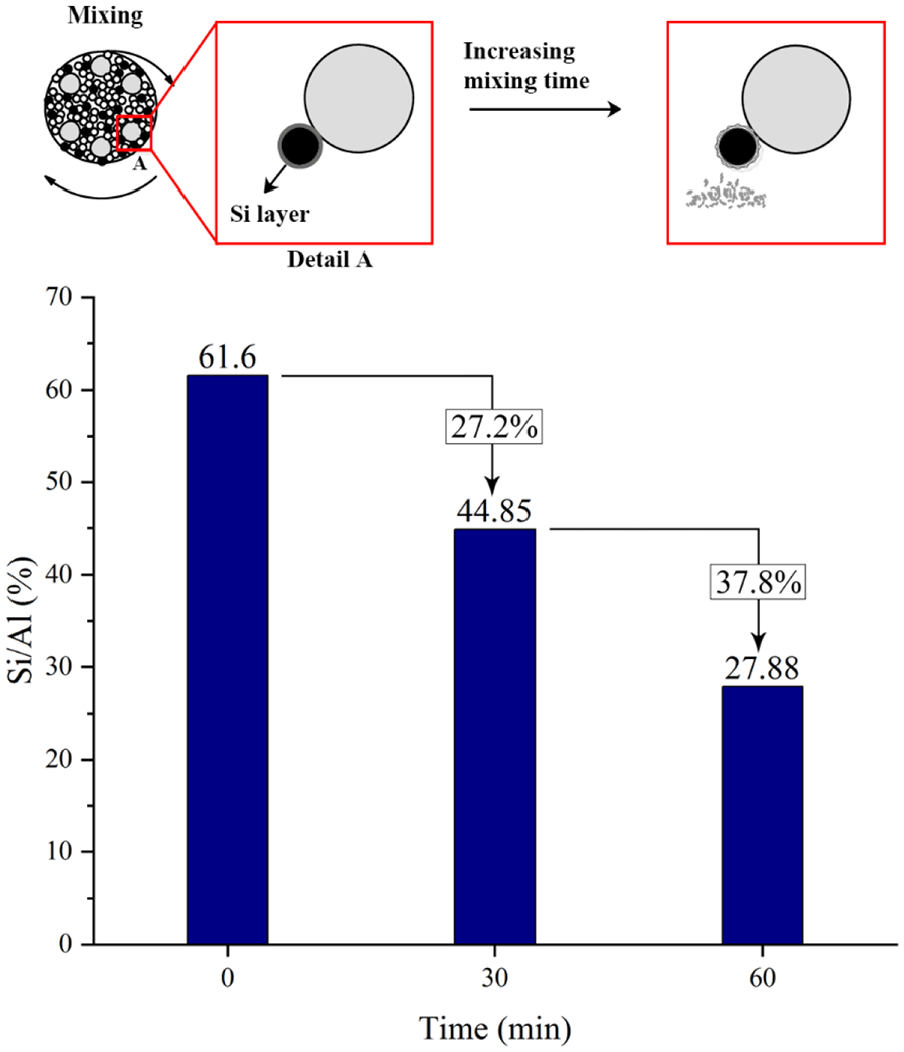
Effect of the mixing time on the ratio of Si to Al.
When TCMS is coated by the CVD process on the aluminum flakes, a layer of Si is settled on the surface which makes the surface hydrophobic. Hence, decreasing the ratio of Si/Al results in losing the Si from the surface and reducing the hyrophobicity.
Effect of super hydrophobic aluminum flake powders weight percentage
Influence of super hydrophobic aluminum flake powders weight percentage on the contact angle is shown in Figure 8. In the figure, weight percentage of aluminum flake equal to zero means only-resin coated surface. As can be seen in the figure, with the increase of hydrophobic aluminum flake powders, surface contact angle increases. Over increasing percentage of the hydrophobic particles leads to less coating adhesion resulted from a phenomenon which is known as resin marbles. 28 The interaction between superhydrophobic nano-textured aluminum flakes and molten polyester produces what is called “resin marbles”. As aluminum flake is hydrophilic naturally, aluminum particles will be hydrophilic and can be wetted by water droplets. If the aluminum particles become functionalized with hydrophobic silanes, the particles will not be wetted by water and become hydrophobic and this phenomenon is called water marbles. According to the water marbles, when resin is combined with the hydrophobic particles and then is sprayed on the surface and cured subsequently, because of the large amount of hydrophobic particles in the composition of aluminum flakes and polyester, hydrophobic particles covers the resin and prevent it from being melted.
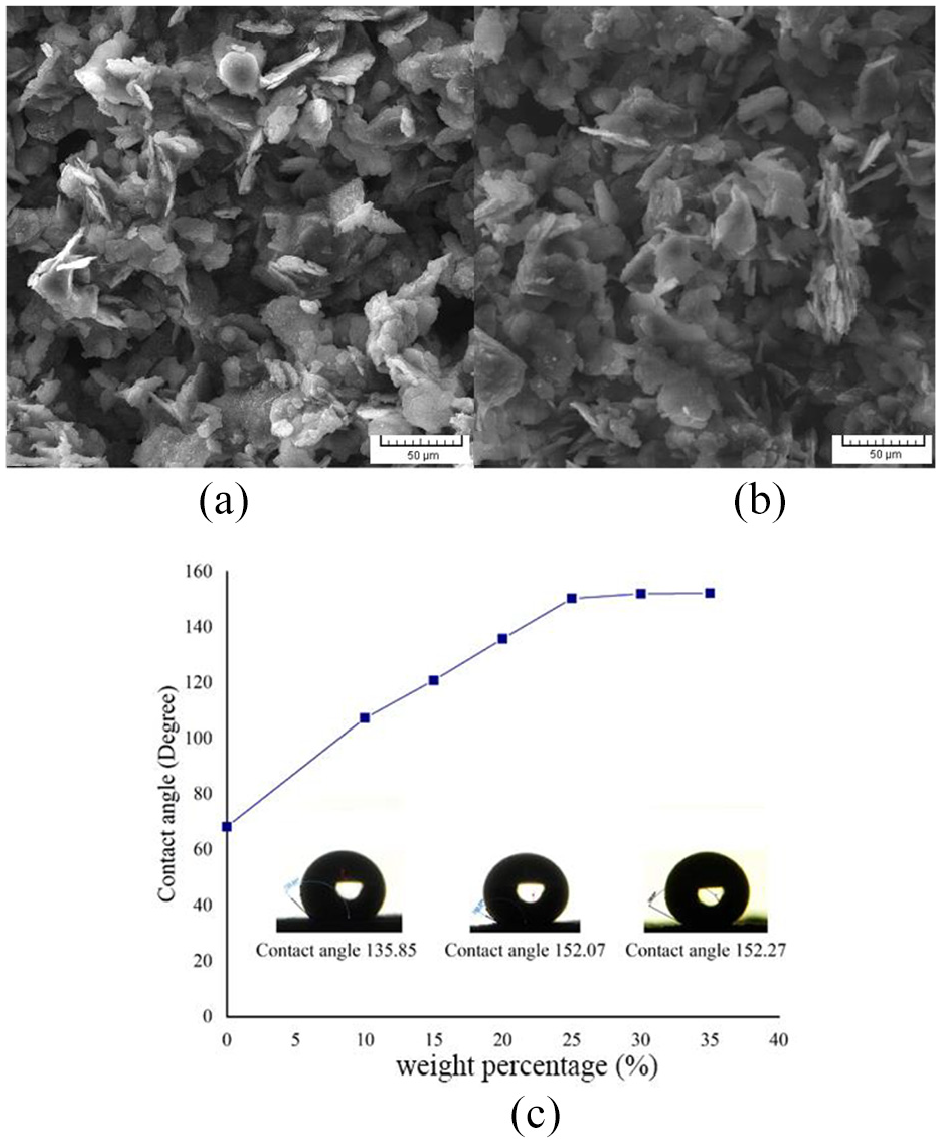
(a) SEM images of aluminum flake weight percentage 25%, (b) weight percentage 35%, and (c) shape of water droplet (5 µL) on the surface of coating.
Superhydrophobic/superhydrophilic nano-textured silica particles. As shown, maximum percentage of aluminum flake particles with size 45–250 µm are between 30 and 35 wt.%.
Effect of super hydrophobic particles size
As mentioned in the previous sections, aluminum flake weight percentage 30–35% and mixing time 15 min were chosen. In the electrostatic spray process, super hydrophobic particles size must be smaller than resin particles to make a stable super hydrophobic surface. In order to analyze the effect of the particles size on the surface super hydrophobicy, three size ranges 0–45, 45–106 and 106–250 µm are considered. According to Figure 9, increase in the particle size leads to increase in the slipping angle and reduce in the contact angle. As a matter of fact, the more particle size become, the more distance between two peaks of roughness, and water can penetrate into the structured surfaces. Thus, when water droplets meet the surface, they conform to the surface. As a result, surface structure changes from Cassie-Baxter to Wenzel. Additionally, the results from SEM which are shown in Figure 10 confirms that by increasing in the super hydrophobic particle size, more holes appear on the surface-shown with red cycles- and it shows the less particle size becomes, the more area of surface are covered by the particles.
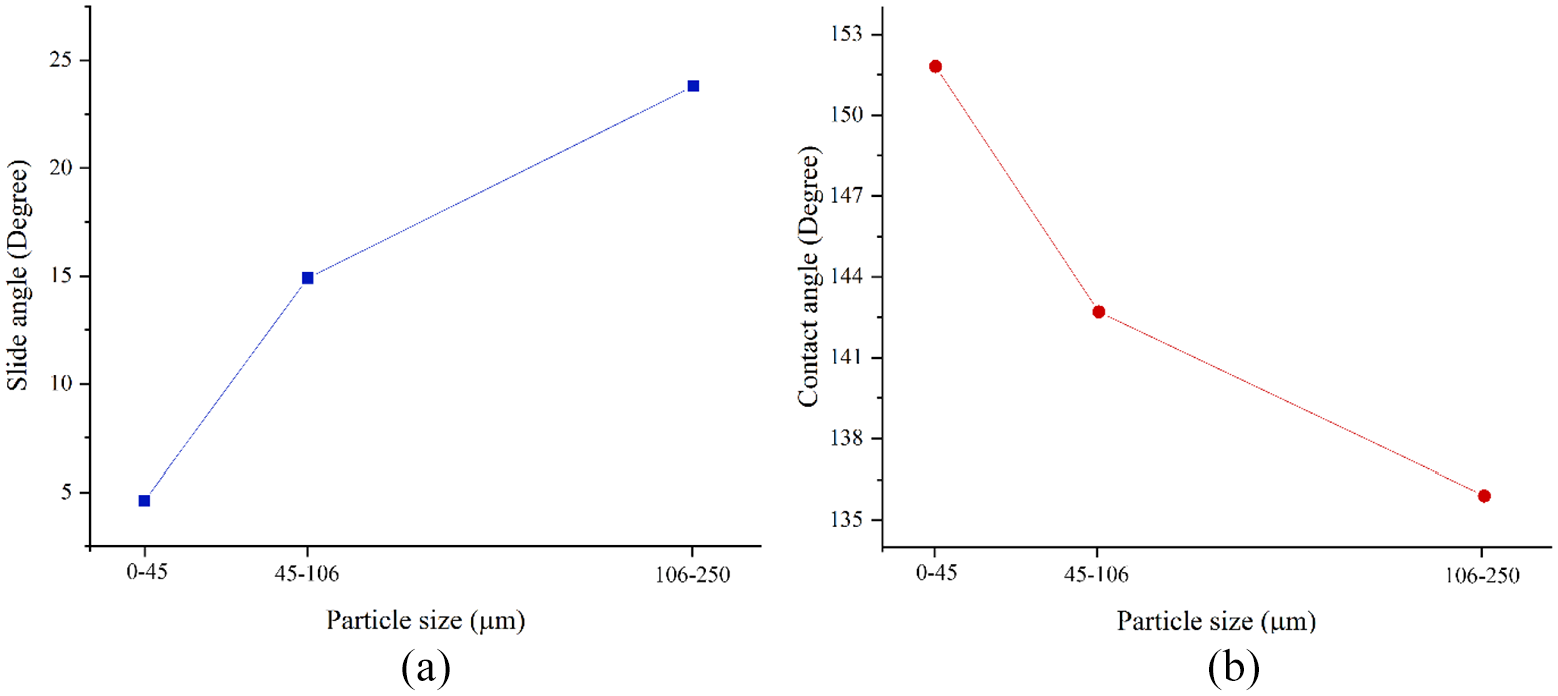
Effect of the superhydrophobic particle size: (a) influence on the slide angle and (b) particle size effect on the contact angle.

SEM images of super hydrophobic coating in different aluminum flake particle sizes: (a) 0–45 µm, (b) 45–106 µm, and (c) 106–250 µm.
To better show the surface roughness and its effect on wettability, Figure 11 is presented. As can be seen, increasing the particle size results in the surface structure changes from Cassie-Baxter to Wenzel meaning more spaces between particles, comparing Figure 11(a) with (b). These spaces facilitate surface wetting by droplets.
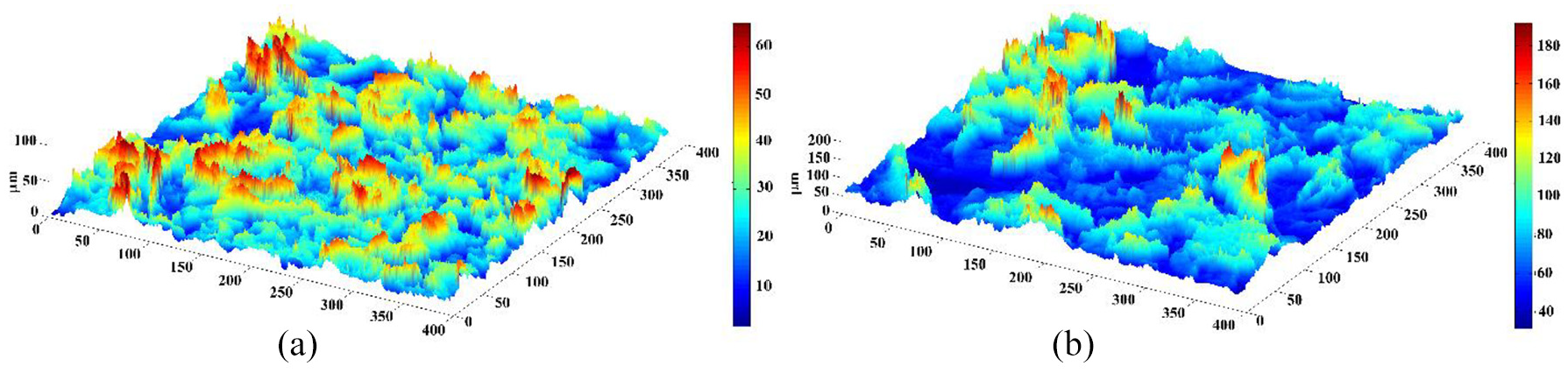
The topography of coated surface: (a) particle size 0–45 µm and (b) particle size 106–250 µm.
Antifouling properties
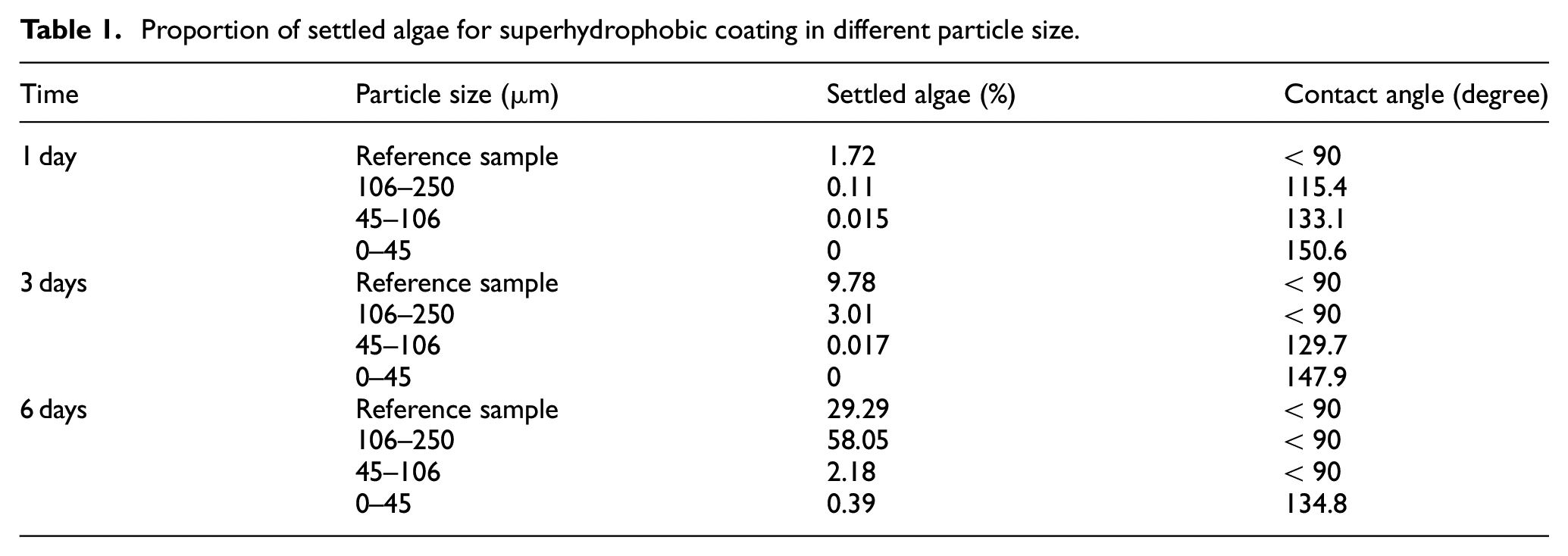
To assess the super hydrophobic coatings in the algae environment and evaluating the coatings performance, three super hydrophobic coatings with different sizes along with a reference surface-without any coating- are compared. Results show algae adhesion to the super hydrophobic coatings with their contact angles versus day which can be found in Table 1. According to the Figure 12 and the Table, for super hydrophobic coatings with sizes 106–250 µm after 1 and 3 days, less algae are formed on the surface compared with the reference samples. These coated surfaces after 3 days lose their super hydrophobicity features since air molecules are dissolved and replaced by micro organisms as they are rougher than the reference samples, hence more algae are settled on the surface over time. Regarding Figure 12, super hydrophobic coatings with sizes 45–106 µm although their hydrophobicity properties were kept in the first and third days, as their super hydrophobic particles size are bigger than the micro organisms one, small areas of the surface are covered by these micro organisms. This is attributed to the height topography and distance between particles are more important than hydrophobicity. 20 The hydrophobic surface loses its hydrophobicity in the sixth day and acts as a rough surface which is more susceptible than a smooth surface-reference sample- to algae settlement, so micro organisms were stuck on the surface. Super hydrophobic surfaces with particle size range 0–45 µm results in better features as the particles could cover the surface uniformly. This uniformity prevents any micro organisms settlement on the surface and as can be seen from the figure, after sixth day only some tiny algae are appeared on the surface sides.
Proportion of settled algae for superhydrophobic coating in different particle size.
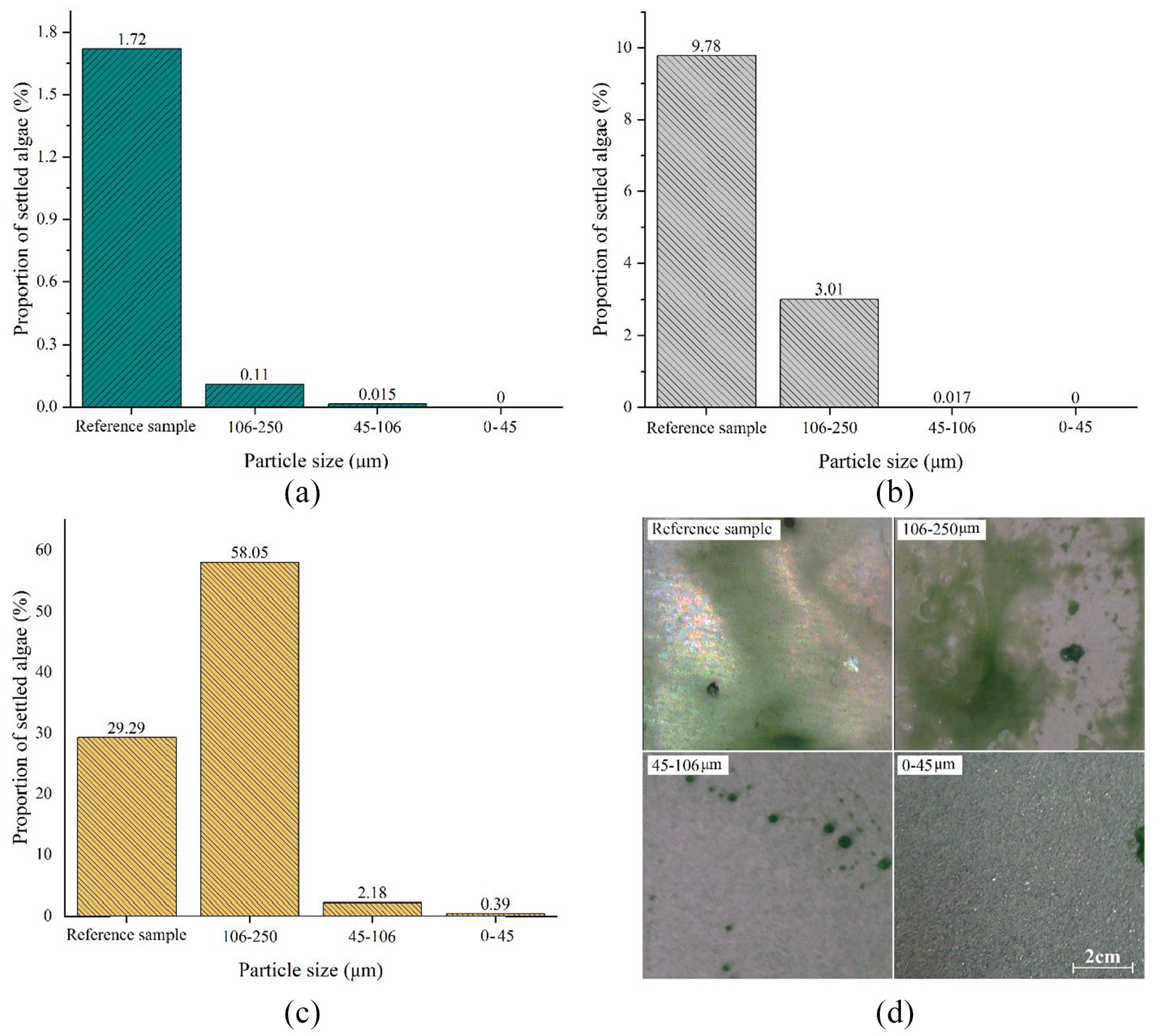
Performance of the fabricated super hydrophobic coating over time: (a) first day, (b) third day, (c) sixth day, and (d) settled algae on the super hydrophobic coating after sixth day.
Schematic of the particle size effect on the antifouling properties is shown in Figure 13. When silane vapor is settled on the aluminum flake particles, a nano structure on the micro particles are formed. Spraying particles through electrostatic spray gives rise to some ups and down on the surface. The more particle size, the more distance between roughness peaks and if this distance becomes bigger than micro organisms, the surface hydrophobicity is eliminated. According to the experimental results shown in Figure 12 and what is shown in Figure 13, it can be concluded that less distance between surface peaks results in harder penetration of micro organisms. Also, nano structures formed on the micro particles surface prevent surface destruction, but since the micro organisms size and the distance are almost the same, these organisms covers the surface irregularly although the surface hydrophobicity were kept regarding the measured contact angles.
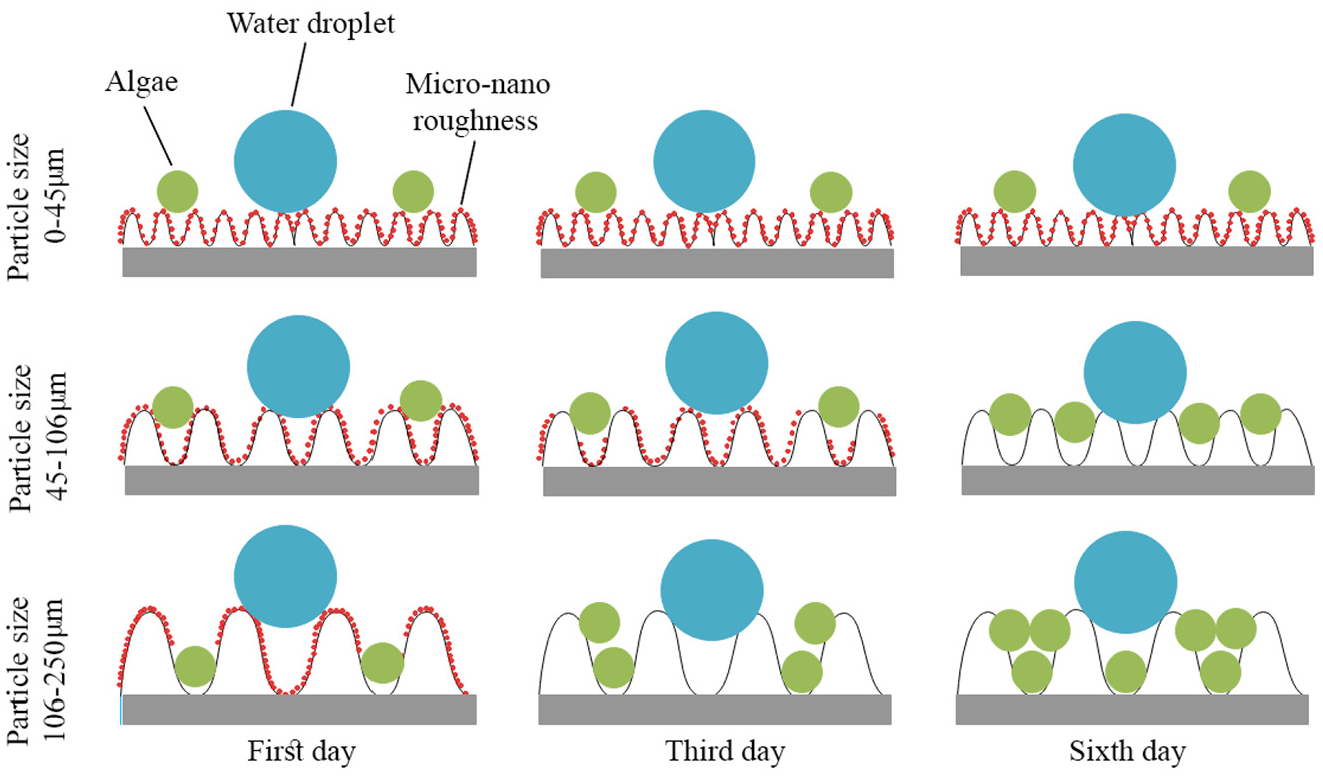
Schematic of particle size effect on the antifouling properties.
Conclusion
This research presents a new one-step environmentally friendly super hydrophobic coating, applicable in large-scale. According to the results, following conclusion can be drawn.
The new fabricated super hydrophobic coating shows a considerable feasibility in large-scale applications.
Increasing the mixing time results in breakage of Si layers and in turn reducing the particle hydrophobicitiy. The best trade-off for mixing time is the time lower than 30 min.
When the amount of hydrophobic aluminum flake powders enhances, surface contact angle increases, but it should not be more than 30–35%.
Coatings with particles smaller than 45 µm result in lower sliding angle and higher contact angle as they cover the whole surface.
The less particle size becomes, the less algae settles on the surface. Particle size lower than 45 µm leads to the most resistance to the algae settlement.
Footnotes
Acknowledgements
Authors would like to appreciate Dr. Saadat-bakhsh and Neda Faal Nazari for their kind supports.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
