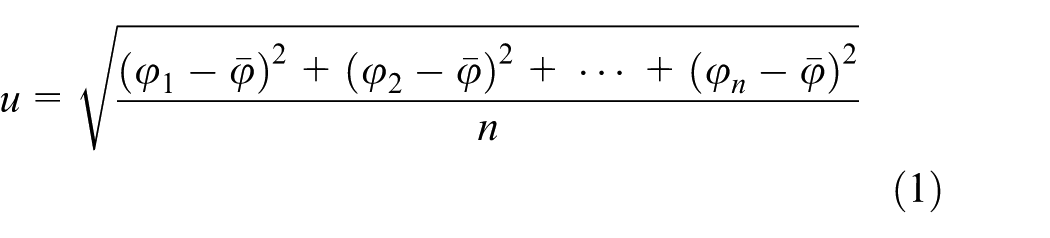Abstract
Non-conventional electrochemical machining in micro-manufacturing has atomic-scale machining accuracy only in theory. By taking full advantage of the material heterogeneity, the micropores which most easily initiate at the surface stoichiometric inhomogeneities for stainless steel can be prospectively obtained with the size kept under control by adjusting the machining parameters. Taking the economy and efficiency into account, a one-step potentiostatic polarization method was established. Optimization of the machining parameters for achievement of microporous structure required by cell adhesive surface on 304 stainless steel in natural 15 wt% NaNO3 solution was confirmed. Based on the potentiodynamic polarization curve, a DC voltage of 5 V in the region of secondary passivation was selected due to the porous secondary passivation film which results in the initiation of large amounts of pores. The effects of the machining time on the pore size, coverage ratio, density, unevenness degree and adhesive cell density on the porous surface were investigated through statistical analysis. The results show that there is a maximum value of machining time, tm, when the surface has a maximum pore density, and minimum pore unevenness degree. Meanwhile, adhesive cell density increases to significant level at tm then levels off over time. Therefore, this approach has been testified possible through the novel use of material microdefects and electrochemical machining to obtain cell adhesive micropores.
Introduction
Stainless steels have been extensively used as implant materials on account of a favorable combination of mechanical properties, corrosion resistance and cost-effectiveness. 1 For successful integration of the implants with peripheral tissue, cell adhesion would be an essential first step. 2 It has been reported that modified surface structure like micro-dimples would significantly improve the cellular adhesiveness and proliferation on contact with the implanted materials.3–6
Non-conventional machining techniques such as electro-discharge machining (EDM), laser beam machining (LBM), electron beam machining (EBM), ultrasonic machining (USM) and electrochemical machining (ECM) have been utilized in micro-manufacturing industries. 7 Compared with other methods, ECM has the advantages of high machining efficiency, absence of a heat-affected layer, lack of residual stresses, cracks, tool wear and burrs, and low production costs. 8 Intensive research efforts have been devoted to processing on micro-scale using ECM.9–11 Byun et al. 12 successfully fabricated a micro-dimple array using a tool electrode of 275 μm in diameter. Others13,14 selected jet ECM which supplies direct electric current with electrolyte jet between the anodic work piece and the cathodic tool to machine surface texturing. Due to the one by one micro-dimple generation, the efficiency has been reduced significantly. In recent years, through-mask electrochemical micromachining (TMEMM) is in widespread use for generating micro-dimple arrays, since all the micro-dimples can be generated at the same time. Madore and Landolt et al. 15 used through-mask electrochemical etching for producing high-precision topographies on titanium, while the photoresist mask has to be removed from the anode workpiece after ECM and cannot be re-used. In view of the reusability of the polydimethylsiloxane (PDMS) mask, Hou et al. 11 fabricated a large-area (PDMS) mask containing micro through-holes to machine micro-dimple arrays with the width 95 μm and depth only 18.9 μm. Nevertheless, now it is still a challenge to further reduce the size of the micro-dimple array and the ratio of etched micro-dimple and mask hole diameter in micro-scale pattern transfer without photolithography of substrates.
It is well accepted that the occurrence of pitting corrosion should be avoided for ECM processes, as pitting corrosion lowers the surface integrity significantly. 16 However, pits with suitable size/shape are naturally microporous structures required by cell adhesive surface. Stainless steel usually undergoes pitting corrosion. Many studies have shown that pitting most easily initiates at the surface stoichiometric inhomogeneities for stainless steel, such as sulfide inclusions, oxide inclusions, carbide precipitates and the phase boundary. 17 The diameter of the pits usually varies in the range from sub-micrometer to a few micrometers controlled by the applied potential and the polarizing time. Therefore, as an alternative, by taking full advantage of the material heterogeneity, the tiny micropores can be prospectively obtained and controlled during one-step potentiostatic polarization for stainless steel. The work described in this article aims at elucidating the pore propagation laws of 304 stainless steel (304 ss) in NaNO3 solution to optimize the polarization parameters for machining cell adhesive surface.
Experimental procedure
A 304 ss plate with the size of 5 × 5 × 2 mm3 was used in this study. The samples were polished by silicon carbide paper with different grades of roughness (320–5000), followed by ultrasonic cleaning in acetone and deionized water. Then, the samples were dried and cold mounted in epoxy resin with a copper wire connection at the rear. The exposed area of the specimen was 0.25 cm2.
The anodic potentiodynamic polarization of 304 ss was carried out after 10 min immersion in NaNO3 solution at a rate of 1 mV/s with a Zahner Zennium potentiostat. It should be noted the effect of the concentration of NaNO3 solution on the machining quality has been conducted over the concentration range of 5%–20% previously. The results demonstrated that higher electrolyte concentration leads to a higher growing rate of micropores and the higher conductivity (not shown here). In order to investigate the effect of machining time on the machining quality with stable conductivity, a proper growing rate of the micropores should be confirmed so that 15 wt% NaNO3 solution in this study was selected. A standard three-electrode electrochemical cell was used with a platinum mesh electrode as a counter electrode and Ag/AgCl as a reference electrode.
A DC power was used for pore machining and the samples were immersed in the 15 wt% NaNO3 solution with a pure copper plate as a counter electrode. The samples were polarized under a proper potential for 4, 5, 6 and 7 min. The surface morphology was observed using a confocal laser scanning microscope (CLSM, Olympus LEXT OLS4000 3D, Japan). The area of pore mouth and pore depth are two basic indicators to characterize the pore size. The coverage ratio of the pores (φ) represents the percentage of pore mouth areas from the total areas where they locate. The number of pores per unit area (ρ) are used to evaluate the pore density. Herein, φ is available to calculate the unevenness degree (u) of the pore distribution
where n is the number of selected regions for statistical analysis and φ1, φ2, …, φn are the coverage ratio of each region, respectively.
Murine fibroblast L929 cells were cultured for 5 h in the incubator containing three growth factors (500 mL DMEM (Gibco), 56 mL FBS (Gibco) and 5.6 mL (v/v) P/S). After being rinsed with PBS (Phosphate buffered saline), the cells were diluted to 106/mL. Before cell inoculation, the samples polarized at 5 V using DC power for 4, 5, 6 and 7 min were ultrasonic cleaned in acetone and bacteria-free water, followed by heat disinfection at 121°C for 15 min. Then the cells were inoculated on the samples. After 8 h inoculation, the samples were retrieved for further analysis. Ten independent regions with the size of 200 × 200 μm2 on the surface were randomly chosen to analyze the pore parameters and adhesive cell density statistically using ImageJ 1.44p software.
Results and discussion
Typical potentiodynamic polarization curve
A typical potentiodynamic polarization curve of 304 ss in 15 wt% NaNO3 solution is shown in Figure 1(a). In the anodic branch, spontaneous passivation is present until the applied potential is up to 1.2 V (vs Ag/AgCl). Then, current density increases due to the emerging of oxygen evolution. It is observed that there is a decrease of current density at 1.5 V (vs Ag/AgCl) following with a short current plateau and then the current density increases again at 1.8 V (vs Ag/AgCl). According to Song et al., 18 such behavior is related to the secondary passivation during oxygen evolution. Secondary passivation film always shows a porous structure which results in the initiation of large amounts of pores in this potential range. As the polarizing potential continues to increase, the increasing rate of the current density slows down as shown in the inset curve in Figure 1(a), which indicates that the whole surface almost dissolved simultaneously. The evolution of the current density with the machining time was recorded under the DC voltage of 5 V for 1 min and is shown in Figure 1(b). The current density is stabilized at ∼0.088 A/cm2, which is comparable with the current density in the region of secondary passivation in the potentiodynamic polarization curve. Meanwhile, it reveals that the localized dissolution and passivation is relatively balanced. Pistorius and Burstein 19 argued that under higher applied potential, the growth of the pits is diffusion controlled with the presence of a salt film on the surface. It is expected that the pore shapes are homogeneous due to the independence of the current density in the pits with the applied potential. Thus, a DC voltage of 5 V is selected as a proper potential to further induce pores.
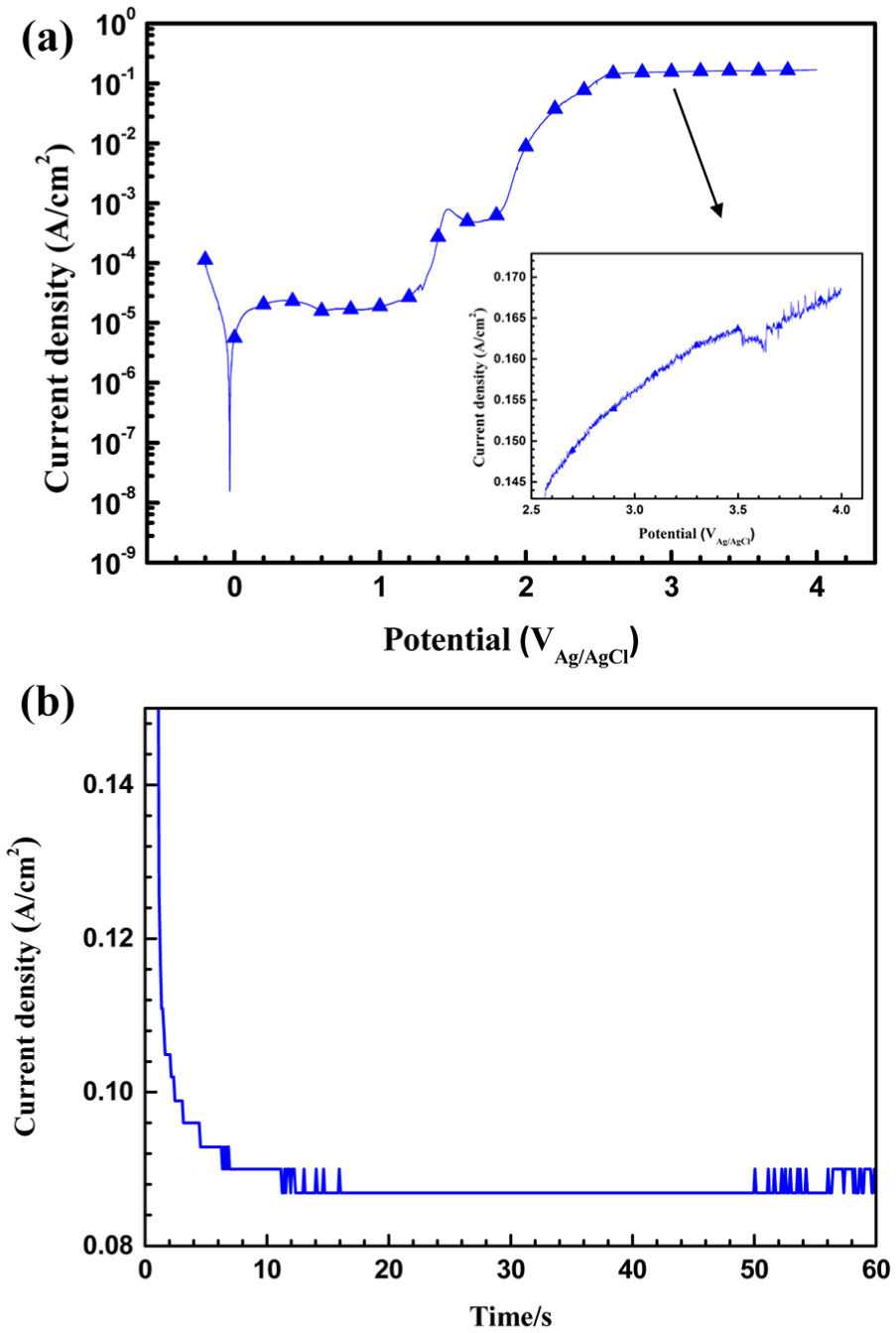
(a) The typical potentiodynamic polarization curve of 304 ss and (b) the current density signatures recorded at 5 V using DC power in the solution of 15 wt% NaNO3.
One-step potentiostatic polarization by DC power
Figure 2 shows the typical microporous morphologies of 304 ss plate polarized at 5 V using DC power for 4, 5, 6 and 7 min observed by CLSM. Figure 3(a)–(c) presents the evolution parameters of the pores. The variation of mean pore mouth radius over time is depicted in Figure 3(a). The fitted slope n of the mean pore radius with machining time decreases from 1.2 to 0.27, while the mean pore depth linearly increases with the slope of 0.83. It indicates that when the pores grow to a certain size, with the localized acidity maintained inside the pore, the growth rate in depth direction become faster and dominates. It should be noted that the deeper pores tend to intensify the local stress field and hence reduce the load carrying capacity of the component. 20 It is clearly seen in Figure 3(b) that φ increases nearly linearly with machining time, whereas ρ increases first from 4–6 min and then decreases at 7 min. The coalescence of pores occurs as ρ increases which cutting down the following number of the pores; u decreases first then increases at 7 min as depicted in Figure 3(c). One possible explanation is that large amounts of newly generated small nucleation sites emerge and coalesce. Meanwhile, the preceding large pores continue growing.
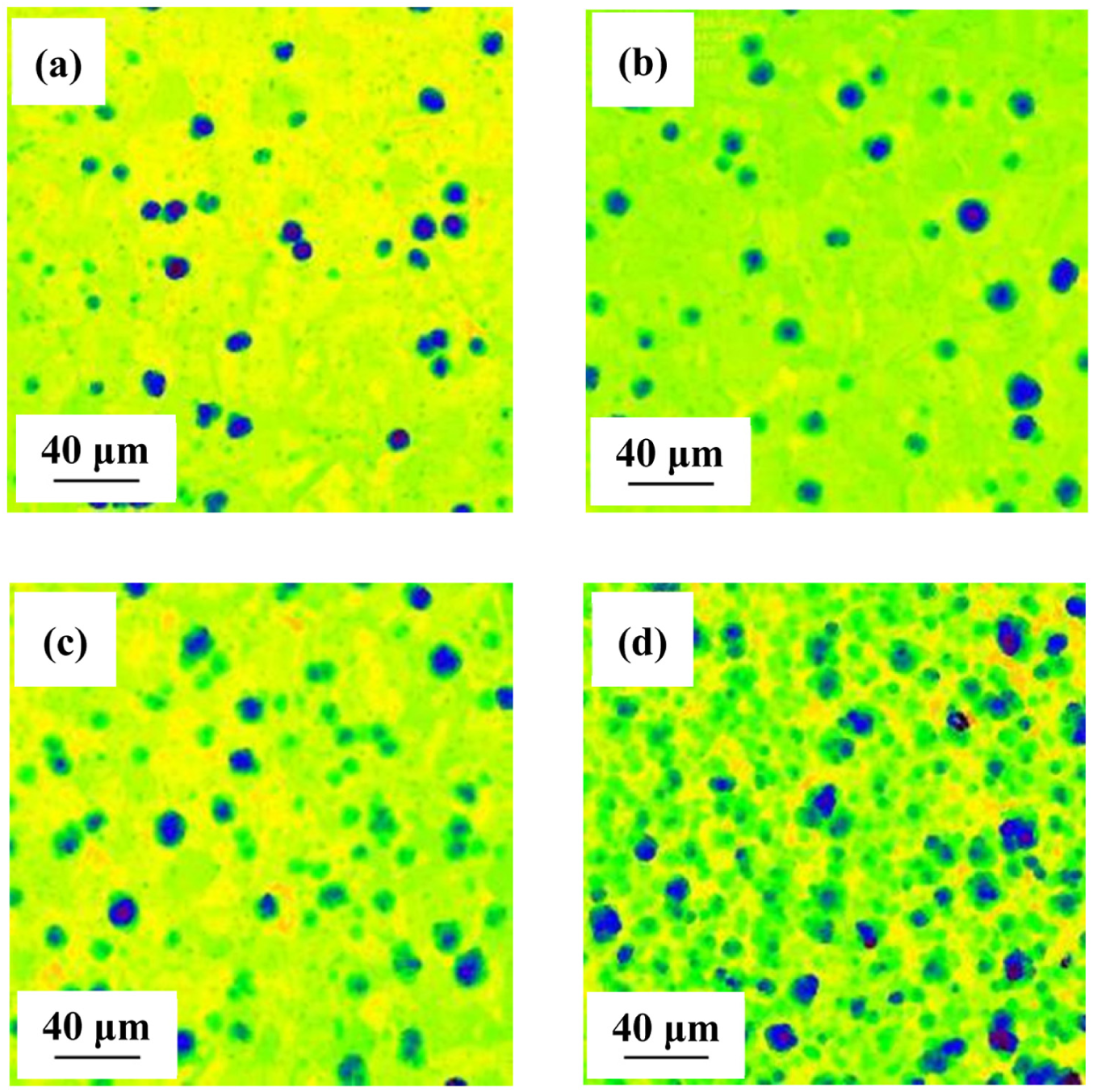
The typical microporous morphologies of 304 ss plate polarized at 5 V using DC power observed by CLSM for (a) 4, (b) 5, (c) 6 and (d) 7 min.
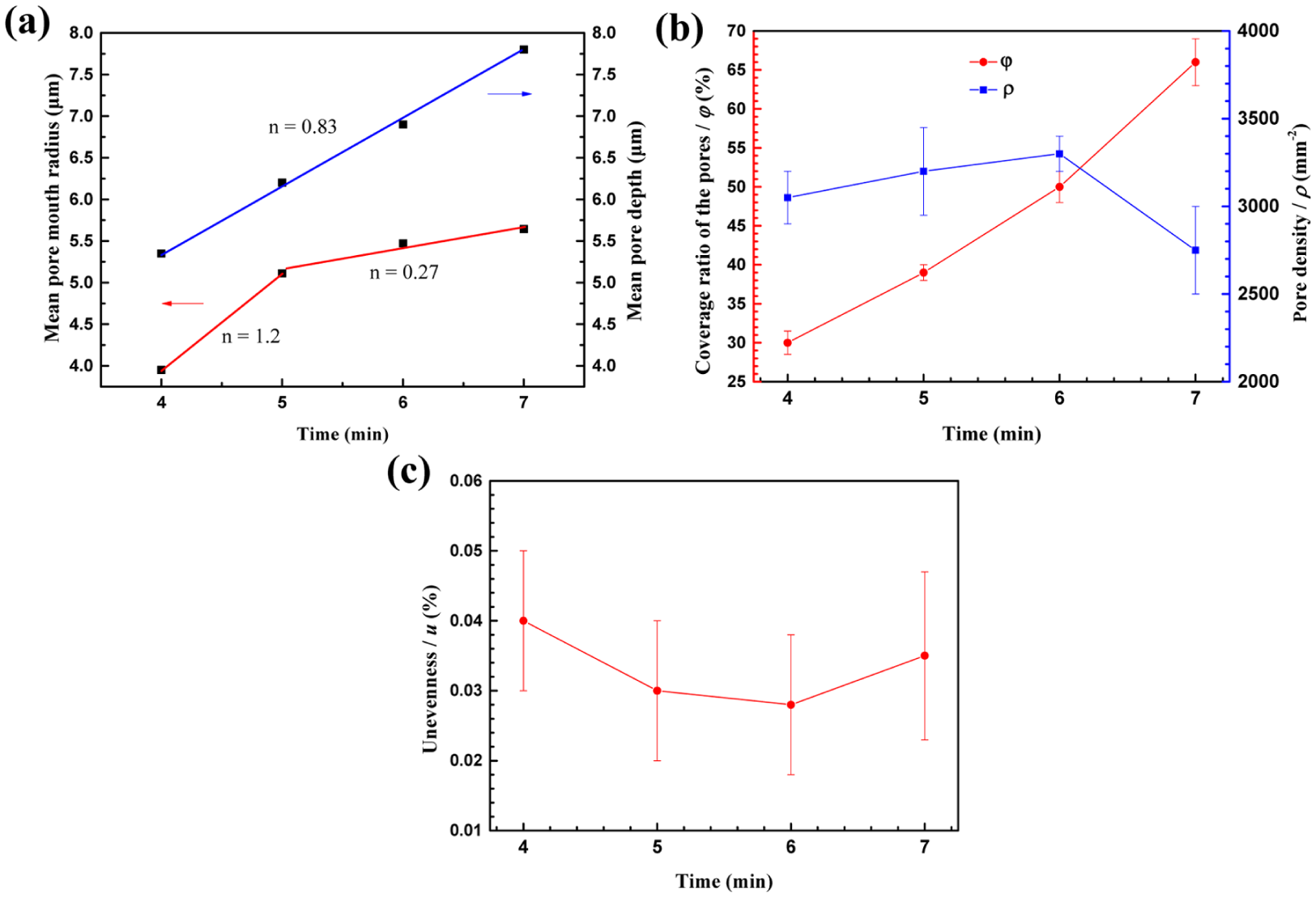
The variation of (a) mean pore mouth radius and depth, (b) the coverage ratio (left) and density (right) of the pores and (c) unevenness degree of the pores.
Cell adhesion on the microporous surface
Cell adhesion experiments show that pores exhibit excellent ability to promote cellular adhesiveness and spreading on the surface of the samples. In Figure 4(a), after culturing for 8 h, the adhesive cell density increases with the polarizing time first from 4 to 6 min with a multiple of 1.8, while no obvious increase from 6 to 7 min. Figure 4(b) and 4(c) is the morphologies of the microporous surface polarized for 4 and 6 min with adhesive cells. In consideration of the sufficient number of pores after 6 min polarization, the inhibition of the cell movement by the pores is significant. A maximum value of the machining time tm of 6 min is proved as the optimal time for machining cell adhesive surface.
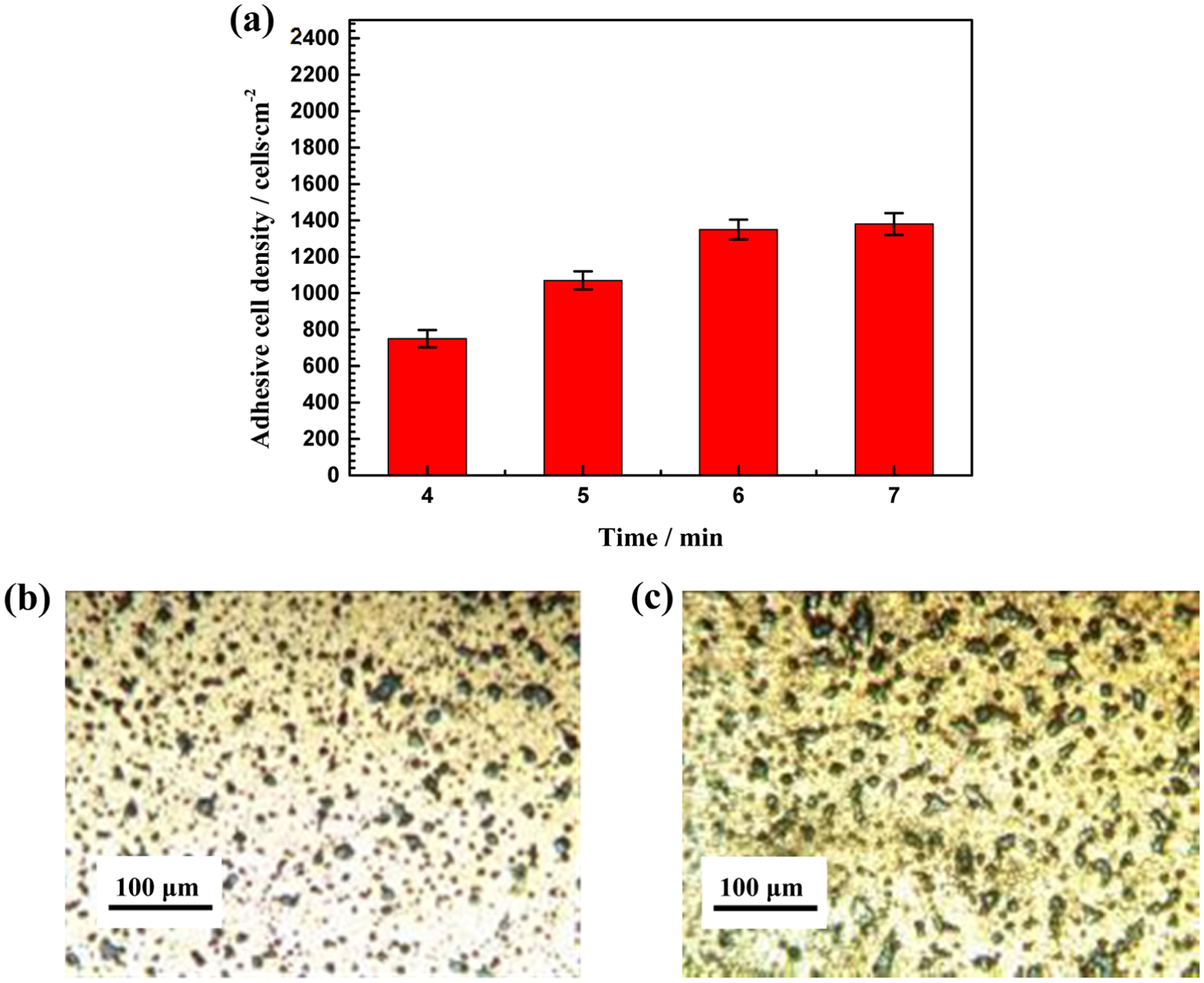
(a) The adhesive cell density with machining time and the microporous surface polarized for (b) 4 min and (c) 6 min with adhesive cells.
Conclusion
In this study, we have established a one-step potentiostatic polarization method to obtain microporous structure on 304 ss plate in natural NaNO3 solution. With a proper electrolyte concentration of 15 wt% and an DC voltage of 5 V applied in the region of secondary passivation, the effect of machining time on the machining quality with a proper growing rate of the micropores can be effectively investigated. The results show that the pore size, coverage ratio, density, unevenness degree and adhesive cell density vary with the machining time, which affect the cellular adhesiveness on the porous surface. Through statistical analysis, it is concluded that the pore shape varies from shallow disk to occluded pit due to the change of the increasing rate of the pore mouth area and pore depth with the increasing of machining time. There is a maximum value of machining time, tm, when the surface has a maximum pore density, and minimum pore unevenness degree. Meanwhile, adhesive cell density increases to significant level at tm then levels off over time. Therefore, an optimum machining time at which the microporous structure is most favorable for cell adhesion has been confirmed. One-step potentiostatic polarization is proved an economical and efficient method to make cell adhesive micropores.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the financial supports by the National Natural Science Foundation of China (Grant No. 51701047), Nansha District Science and Technology Program 2016GG022 and Guangdong Natural Science Foundation of China (2017A030310587).