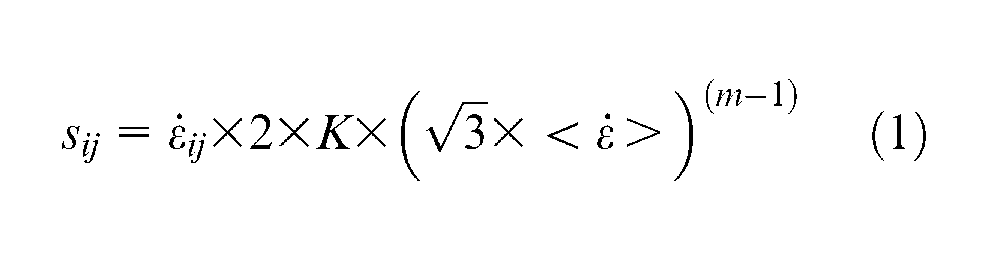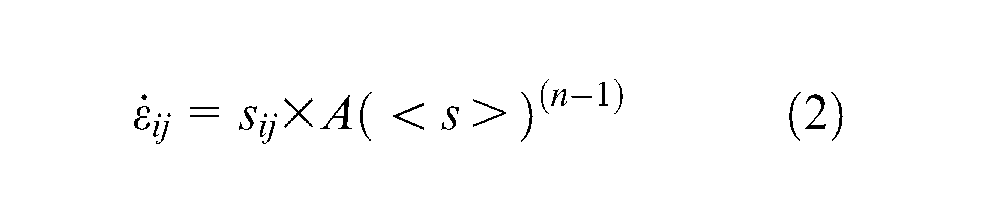Abstract
Diffusion bonding may be used, with superplastic forming, to create a great range of cellular structured materials, with great potential for material efficiency, multi-functionality and lightweighting. However, this potential is generally considered to be constrained by the slowness (hours) of and the overall cost of achieving such solid-state bonds. The work described is part of the basis for the development of a process that can produce good diffusion bonds, in the commonly used titanium alloy Ti64 (Ti6Al4V), in less than 60 s. And so it has the capability of increasing the productivity for diffusion bonding. Localised direct heating can induce high levels of stress, and these thermal stresses can accelerate the diffusion bonding process. The equipment used involved induction heating and required no large (and expensive) hot presses or pressure vessels. Hence, machinery capital cost may be reduced. A sequence of numerical models was found useful to understand the effects of the process parameters on diffusion bonding. Finite element modelling looked at accelerating the collapse of the voids, determined by the relative flatness and roughness of the metallic sheets at different spatial wavelengths. It appears that the localised heating drives the diffusion bonding process in inducing significant stress levels, into the material to be bonded, and thus accelerates the collapse of voids and the achievement of intimate contact. Raising the temperature as much as possible assists the actual diffusion processes required for bonding to occur and is an important aspect of accelerating the process. Therefore, the increase in the rate of bonding and the lower cost of the equipment open up the possibility of much more affordable diffusion bonding and the production of material efficient and functionally useful cellular materials.
Keywords
Introduction
Forms of diffusion bonding (DB) have been used for millennia, most notably to produce the ‘patterned blades’ such as those of the samurai. However, over the last 50 years, there has been more effort to understand it, utilise it and to develop its application.
Metcalfe and Lindemer 1 over 50 years ago summarised what was happening and reviewed the mechanisms involved. The process can be summarised bringing two clean surfaces into close and intimate contact at an elevated temperature to achieve solid-state bonding between the two surfaces. The pressing together of the surfaces provides the intimate contact necessary for the diffusion of material across the surface grain boundaries. This leads to the inter-diffusion of material from the two opposing surfaces.
The solid-state joint so produced is a bond in which there has been no localised melting or scarring and, across the bond, the microstructure is not altered – without any heterogeneous filler material. And if the materials involved are not heterogeneous then, unlike the more common liquid fusion welding, there are no residual stresses.
In addition, such a joint, or diffusion bond, is permanent and the process of diffusion is not reversible under the bonding conditions (unlike those produced in the processes of welding or brazing and soldering). It is a clean bond with no disruption or contamination of the bonded materials and may be indistinguishable from the neighbouring material.
Background and utility of DB
The irreversibility of the diffusion bond is of particular benefit in producing complex structures or structured material as it enables such structured materials to undergo subsequent bonding and forming operations. That is because any subsequent forming or DB operation cannot damage the (diffusion) bonds already formed. The clean nature of diffusion bonds and the lack of residual stresses enable such bonds to have high integrity, and the bonding process has been employed extensively in the aerospace and nuclear industries.
DB combined with superplastic forming is used to construct cellular structured materials. These may have a complex shape and a complex and lightweight, often periodic, internal structure. This is achieved by selective bonding between areas of adjacent sheets. The sheets are then formed to shape and inflated to obtain the internal stiffening structures. The selective bonding is achieved using a pattern of ‘stop-off’ material disposed over the surface so as to inhibit, locally, the DB process.
An alternate manufacturing technique involves DB layers of sheet but each laminate layer or sheet is pre-cut, before stacking, so as to produce a preform that then undergoes a DB procedure. This technique has been commonly used to produce microchannel devices. 2 These techniques of DB of laminates have found application in fuel cell, climate control, micro combustion and solvent separation technologies and for artificial kidneys. 2 This manufacturing technique has also been used to produce high integrity heat exchangers and conformally cooled tooling (for metal casting and forming and also polymer injection moulding – see Wilden et al. 3 )
Advantages of diffusion-bonded structures
The principal drivers for DB are its ability to help manufacture materials or structures with fine and complex detail. As has already been mentioned, this is used in the production of parts and materials based upon the use of DB of sheets into laminates. The applications and subsequent manufacturing operations are different and can be, broadly, considered as applied to the development of microchannel devices or structured (or cellular) materials.
Microchannel devices
The integrity of the diffusion bond enables higher temperature and pressure loading of materials and structures that employ it. Its application in microchannel devices is due to narrow channels that can be fabricated and the consequent improved heat transfer in such channels. This is used to produce consequent improvements in the efficiency and controllability of heat exchangers and chemical reactors. The much-improved surface-to-volume ratios of these devices makes more compact reactors with improved heat flow, compared with macro scale reactors and devices. 2
Resource efficiency and cellular materials
Superplasticity (a condition in which a material may be highly formable and can undergo large deformation) is often associated with DB. It is used in conjunction with DB as an additive process to produce complex shaped parts, with a greatly reduced need for fasteners and fastening operations. This allows concomitant reductions in assembly time, cost and weight. 4
As an additive process, the combination of DB and superplastic forming is resource efficient and is used to produce cellular materials by inflation after selective bonding. The resource efficiency is due to the low waste (due to it being an additive process without material waste due to machining or cutting) and also the material efficiency arising from its cellular structure and potential for multi-functionality of such structured materials.
Much interest in the application of cellular materials follows from the work of Gibson and Ashby. 5 They showed the wide range of functionality and the possible weight (and material) reduction possible of such materials. This interest has also built upon the earlier work of Lakes 6 who considered hierarchically structured materials (as too did Eiffel with his tower) and the opportunities for greater improvements in material efficiency and of the development of new material behaviour.
The appreciation of opportunities for application of DB is seen in terms of the improvement in specific properties as the density reduces7,8 in cellular materials. In addition, Wadley et al. 9 have considered opportunities for improved thermal management as well as vibration control and, also, special improvements for impact/blast energy absorption, in cellular materials.
Cost of DB
The development of cellular materials as structurally efficient lightweight materials has become of growing importance over the last 10 years but the high costs of manufacture 10 have constrained the development and application of such materials. However, although superplastic forming and DB can permit the fabrication of cellular material structures, that could not otherwise be produced, 11 at reduced weight and fabrication costs, there is still a need for these costs to be driven down.
The current common view is that the equipment for DB requires a significant financial investment and that the size and cost of this equipment (large high-pressure and high-temperature ovens) are significant constraints for the production of larger structures. 12 It is also considered that the long times required for diffusion bonding further compound the cost issue and make the process unsuitable for application to mass production. 13 Paul and Lingam 2 have, also, identified a ‘significant barrier’ in the manufacture of microchannel arrays (and their application to improving the scalability and efficiency of chemical reactions) as being the capital cost of the manufacturing equipment, required for diffusion bonding, and the long cycle times to achieve a diffusion bond.
Reducing costs of DB
The costs of DB have been identified as a problem for its increased application. These costs have centred on the machinery required to achieve and maintain the conditions for DB and the costs associated with this machinery and its utilisation.
A brief review of what is done and why and the costs involved is given. This is followed by a review of approaches used to improve the DB process.
Overview of DB processes
The requirements needed to achieve DB were described by Metcalfe and Lindemer 1 and, subsequently, nearly 50 years ago, Garrett et al. 14 described mechanisms and machinery for achieving diffusion bond – including the use of gas pressure loading.
The basic requirement of any DB process is to achieve the required intimacy of contact between the bonding surfaces. This is required to be with an elevated surface temperature. (The bonding temperature is more than half the melting point for the metallic alloy.)
The interfacial voids between the contacting sheets are required to be as small as possible so that the diffusion of material between the surfaces can proceed. This is because diffusion, being a non-linear process, is most adversely affected by the distance to be diffused over. (Thus, a 10-fold decrease in distance may lead to a 100-fold increase in the speed of the diffusion process.)
The numerical models developed and validated by Pilling et al. 15 and Islam and Ridley 16 are able to describe the effect of high pressures in deforming the mating surface so as to reduce the size or eliminate the interfacial voids, over the whole of the area to be bonded. These models then predict the diffusion of the surface material across the much shrunken voids to achieve proper DB. These models include the effect of surface roughness and flatness on the bonding process. They also include the effect of elevated temperatures in reducing the pressures and times required to shrink the voids, as well as the effect on increasing the inter-surface diffusion itself.
Hot isostatic pressing and press bonding
Hot isostatic pressing (‘HIPing’) was originally developed, 17 for use in the nuclear industry, and is the commonly preferred method for DB – especially for large parts for the aerospace community. A HIP vessel is a high-temperature and high-pressure chamber, in which a gas (usually argon) is used to both pressurise and heat the contents of the vessel.
Parts to be bonded must be evacuated and sealed at the periphery or encapsulated inside a soft steel bag – which is also evacuated and sealed. This is so that the hot gas inside the vessel may produce a pressure load on the parts to be bonded – and heat the part too. Upon heating, the (hydrostatic pressure) gas load is transmitted into the parts and so forces the surfaces to greater intimacy.
However, the time to achieve the appropriate conditions of pressure and temperature (for large structures) and to remain at (for the DB to be finished) 18 is long. And this coupled with the large capital investment required for the appropriate machinery incurs high costs. 12
Press bonding, in which a hot forging press is used, appears at first sight, as an attractive and good manufacturing option – in that the press equipment is simple. The commonly used vacuum hot press (VHP) is used for the DB of laminates for microchannel devices. 2 The size of the press soon becomes an issue; however, if large parts are to be bonded as, at a possible bonding pressure of 10 MPa, a 1 m2 sheet would require a force of 10+7 N – or approximately 1000 ton weight.
For microchannel devices, VHPs are favoured (because of the relatively small surface area). There are, however, still issues of obtaining pressure uniformity, over the surfaces, due to platen flatness and tolerance and registration errors in positioning the laminates. 2 Such problems become more difficult to deal with as the platen size grows.
Depending upon the materials and geometries to be bonded, the temperatures may, quite commonly, be up to 1000 °C, and the pressures, commonly too, may exceed 100 atmospheres (10 MPa). The bonding times may be 2 or 3 h, and the entire process cycle time may be considerably longer.
Both the cost of the robust containment vessel and the time required to cycle the temperature (of the large mass of the pressure vessel) to operating conditions are issues which increase the cost and limit the production use of DB. 13
Cost reduction work for DB
Work has been done to reduce the time and pressures required for DB with some success. Some of this has focussed on the material state itself and some on improving the bonding process itself.
Material state improvements
It has long been known that reducing the roughness of the surfaces to be bonded and improving their flatness increase the speed of bonding (as quantified in the work and numerical models of Pilling et al. 15 and Islam and Ridley. 16 ). However, there are other approaches to improve the formability of the sheet material (such as reducing the grain size of the material to be bonded), which can reduce the temperature for bonding.
Reduced temperature bonding
Low (comparatively)-temperature versions of materials and DB joining of small sheets18,19 are being developed and tested. This use of lower temperatures reduces the issues of grain growth (during bonding and forming) of material. The reduction in bonding temperature has, also, an effect in reducing the requirements on the tooling and machinery – and hence on the capital costs and the running costs.
However, there are indications 20 that there are lower limits to the temperatures as bonding may be achieved. Sanders et al.13,18 showed low-temperature DB for the production of cellular structures, but that it still required significant bonding times – and hence costs.
Pressure reduction
Hamilton and Ascani 21 patented a process of concurrent superplastic forming and DB. They gives detail, for forming titanium sheet, in which bonding was achieved at much lower normal pressures than those applied when using HIP vessels or VHP for DB.
Subsequently, Sunwoo and Lum 22 examined, experimentally, the concurrent superplastic forming and DB and how beneficial this was in bonding aluminium alloy sheets together. Their aim was to investigate the possibility of disrupting the tenacious oxide layer that exists on the surface of aluminium alloy and how the disruption might facilitate the DB process. They achieved this by in-plane tensile loading of the aluminium sheets and applying a normal loading to press the sheets together. Again, this was achieved with significantly lower pressure loading (<1 MPa) and in relatively short times.
Rayudu et al. 23 demonstrated that a low pressure (0.7 MPa) could produce bonds within an hour. However, the surface roughness (as shown by the gap in non-bonded areas) of the sheets used must have been quite large – as the bond line is given as having a thickness of 20 µm. 23 They showed significant bonding in the regions of large deformation, in the same time, which can be understood, as the stretching of the sheet produces a proportional reduction in the amplitude of the roughness. Thus, significant improvements in reducing the pressures and times to bonding may be achieved by altering the stress state from a pure hydrostatic loading. However, for a more general application, flat, thin sheet material is preferred.
Induction heating to improve DB
Metcalfe and Lindemer 1 refer to Samuel and Samuel 24 as to the possibility of induction heating, in itself, causing an improved rate of DB. 1 However, they discount this in their article. Subsequent applications of induction heating have concentrated on using it to improve the speed of reaching the bonding temperature rather than the ways of increasing the speed of bonding. 25
Need for cost reduction
There are signs of increased need for cost reduction in DB as the advent of friction stir welding has enabled manufacturers to consider seam bonding of (titanium alloy) sheets together to enable fabrication of larger structures. This, hence, in turn requires larger (and more expensive) HIP vessels. 18 In summary, there is a demand for more and larger structures to be bonded, to achieve cellular structured parts, and the technologies to supply appropriate-sized materials have become available. So the critical elements of cost reduction now centre on the time to achieve a diffusion bond and the cost of the machinery to do this.
Methods: numerical and experimental approaches
In DB, the surfaces involved must come into good contact (so the atomic inter-diffusion may so proceed). This must occur over the entire region of the surfaces to be bonded. Thus, the methods to be investigated must reduce the barriers to diffusion and assist in reducing the time to achieve good contact.
Material preparation
In the process of DB, the initial distances, between the two surface interfaces, are reduced by hot forming process (creep or superplastic forming), which collapses the surface voids, formed by the roughness and lack of flatness of the two surfaces involved and then the diffusion process is enabled to take place. So the quicker the initial distances can be reduced, then the faster the bonding, as a whole, may be achieved. The validated models of Pilling et al. 15 and Islam and Ridley 16 demonstrate this.
So, polishing the surfaces, prior to bonding, to reduce the roughness and also achieve flatness, as is commonly done, was undertaken in order to facilitate the bonding process. This was performed on a polishing wheel, to achieve a roughness measure, Ra, of less than 1 µm. Subsequently, the samples were washed in distilled water and methylated spirits, prior to placing in the vacuum chamber.
Material selection
Many metals have a surface oxide layer on them that needs to be removed before metal surface may contact metal surface. In some cases, this may be relatively easily removed chemically or by mechanical cleaning, but in other cases, such as for aluminium alloys, this may not be so easily achieved and special conditions may need to be obtained or special coatings required.
An example of a special condition that assists DB is superplastic forming with diffusion bonding (SPF/DB). In this process, the deformation of the surface breaks up the oxide layer and, as surfaces come into contact, good metal-to-metal contact (and hence bonding) is achieved. (This is commonly performed in a vacuum or inert gas atmosphere – to prevent generation of coating layers on the surfaces involved.)
For the modelling and experiments, reported in this article, a titanium alloy was selected, because of its utility and, also, because the oxide at the surface can be absorbed into the lattice structure of the material at elevated temperatures, as stated by Pilling and Ridley. 26 This absorption reduces the difficulty of bonding titanium alloys and makes it a good and useful material for studying the DB process itself. The material models for the thermal and mechanical modelling use data for Ti6Al4V taken from Elzey and Wadley, 27 Pilling and Ridley 26 and Pilling. 28
Achieving high stresses
Although the process of DB during superplastic forming undoubtedly (see section ‘Pressure reduction’) induces a beneficial stress state in the material being bonded, it involves distortion of the geometry and would be difficult to apply to flat sheet or multiple layers.
In considering the Norton–Hoff viscoplastic equations, often used to describe the material deformation (viscoplastic) in hot forging or hot forming
And reformulating this as
where sij is the deviatoric stress tensor (tensor with hydrostatic component removed);
For titanium, n is 4.3(from Elzey and Wadley 27 ), which gives a significant non-linearity of behaviour due to stress-driven effects. Thus, increasing the Mises stress level by a factor of 10 would, roughly, give a 1000-fold decrease in the effective viscosity and hence a significant increase in the flow rate or strain rate.
Localised heating, in depth, is, however, one way in which stresses may be induced into sheet material, by the mechanism of local thermal expansion. This is described in the patent for the process. 29
Localised heating may be difficult to achieve when a uniform heating in depth is required. This is because, usually, a surface heating method is employed such as, for example, radiant or gas heating. So it is difficult to get heat into the bulk of the material and to the bonding surfaces. (This is due to the time taken for heat to diffuse from the surface and, also, the thermal gradients involved.) Induction heating does, however, induce heat directly, at depth, into materials and hence induces large stresses in materials, without contamination, very speedily.
Local heating apparatus consisting of an induction coil, with axis normal to the sheet surface (Figure 1(a)), was utilised for the bonding experiments. An induction heating coil was used to induce heat locally, but in depth, into the titanium sheets to be bonded. Figure 1(a) shows the induction coil, attached to its power supply transformer, with a white ceramic locator going through its inner diameter. The two titanium alloy sheets were evacuated and placed within a thin flexible container (see Figure 1(b) and (c)) such that it allowed for the close proximity of the coil to the sheets to be heated; the close proximity being required for good coupling of the inductive heating into the sheets.
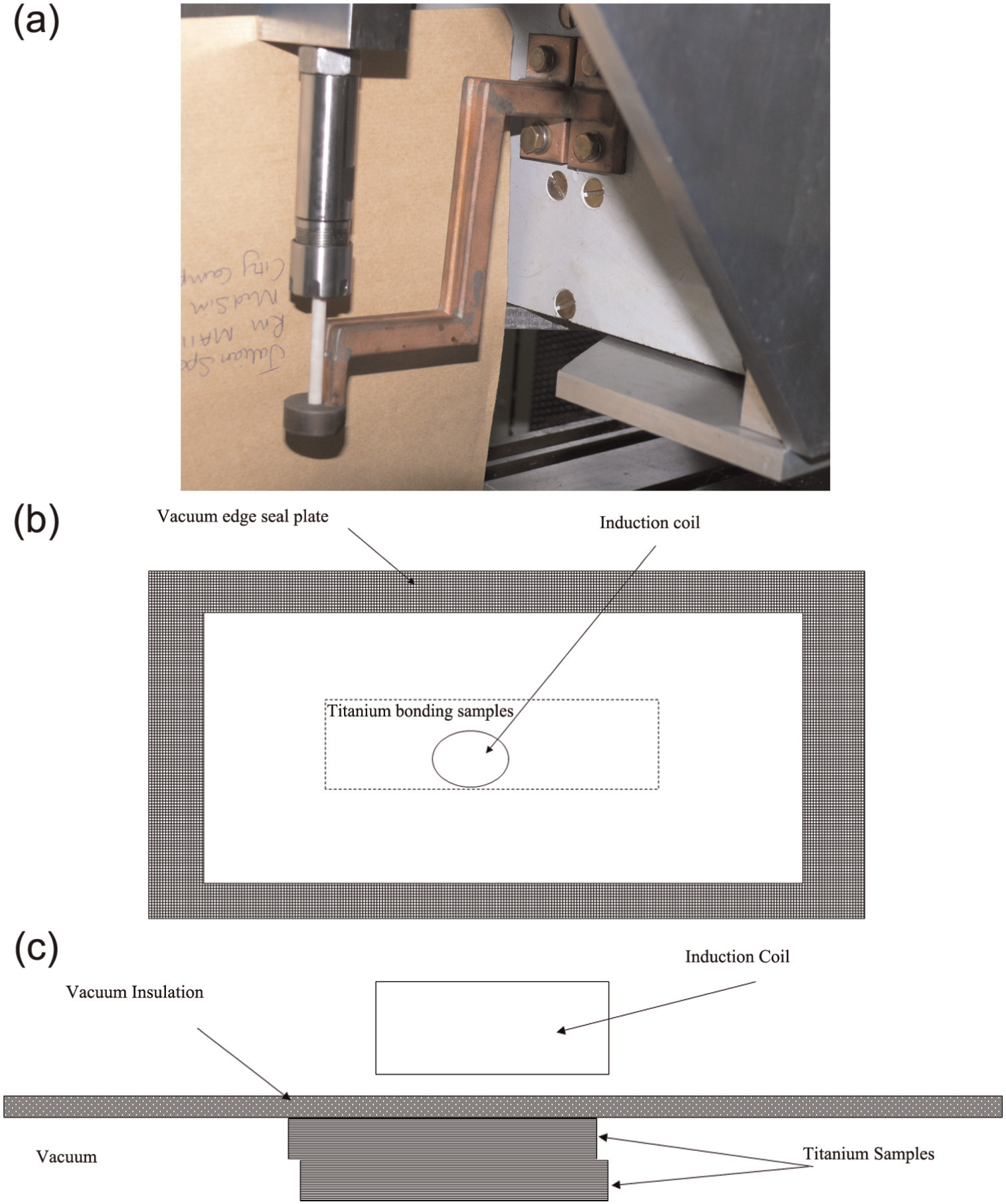
(a) Picture of local heating apparatus; an induction coil, attached to its power supply transformer, with a white ceramic locator going through its inner diameter; (b) plan view of heating and bonding rig and (c) cross section of heating and bonding lay-up
Numerical experiments were undertaken to look at the process of inducing the stresses, in the sheets, and their causes and effects. Numerical simulation, using the MARC finite element software from MSC Software, of the stresses induced and the (creep) stress relaxation involved is a well-used technique that allows useful insight without the difficulties of experimental measurement of transient stresses at high temperatures.
Modelling reduction of voids and bonding
Pilling and Ridley 26 studied isostatic DB and developed models for DB. These included the plastic deformation in the voids and the process of diffusion of grain surfaces and included the effects of grain size (on the plastic flow) and surface roughness. They concluded that the most important effects on the void collapse were due to the plastic deformation, which was required before the proper diffusion of materials around and through grains could occur.
They commented on the importance of including the longer wavelength flatness of the surface when modelling the kinetics of void densification. Figure 2 shows an idealised interaction of two surfaces with different periodicities of roughness and the resultant variation separation of surfaces and initial voids.
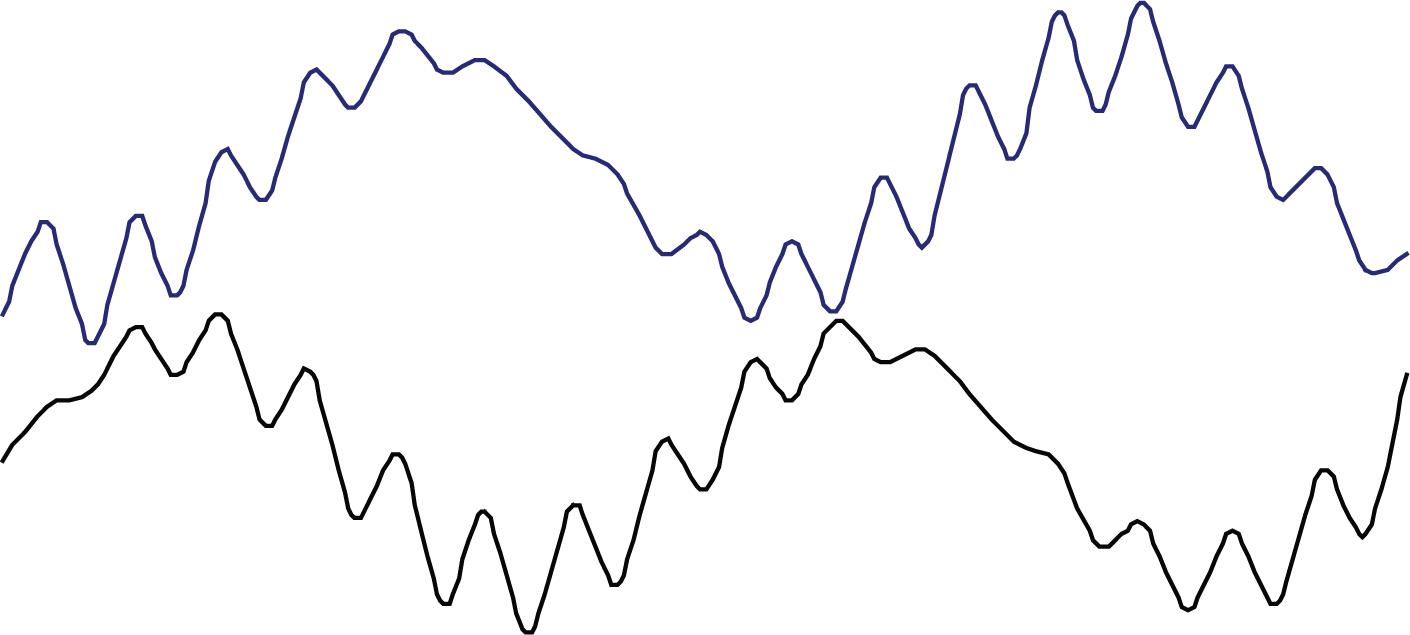
Idealised interaction of two surfaces with different periodicities of roughness and variation separation of surfaces and initial voids.
Takahashi et al. 30 compared different models of void collapse and, for the DB of copper, used finite element modelling of the void collapse with a power law creep material model. They demonstrated that different models were relevant for different constraining conditions and stress states so produced.
In order to deal with the issue of the void collapse (prior to the diffusion processes), numerical simulation (finite element) of the non-isostatic void collapse was undertaken. This included simulations of the stresses, due to localised heating, and the effect of a complex surface topography (including both roughness and flatness) was undertaken. This effect of surrounding material in constraining material and altering the stress state was considered, as too was the effect of the evolution of the temperature field due to the transient conduction into the surrounding material.
The simulated surface consisted of a roughness of wavelength of 3 µm and amplitude of 2 µm superimposed on one of a wavelength of 51 µm and amplitude of 2 µm. This was simulated as part of a 1-mm-thick sheet, and the roughness and its appearance with respect to sheet thickness are shown in Figure 3. The external surface of the sheet was subjected to a gas loading of 1 MPa.
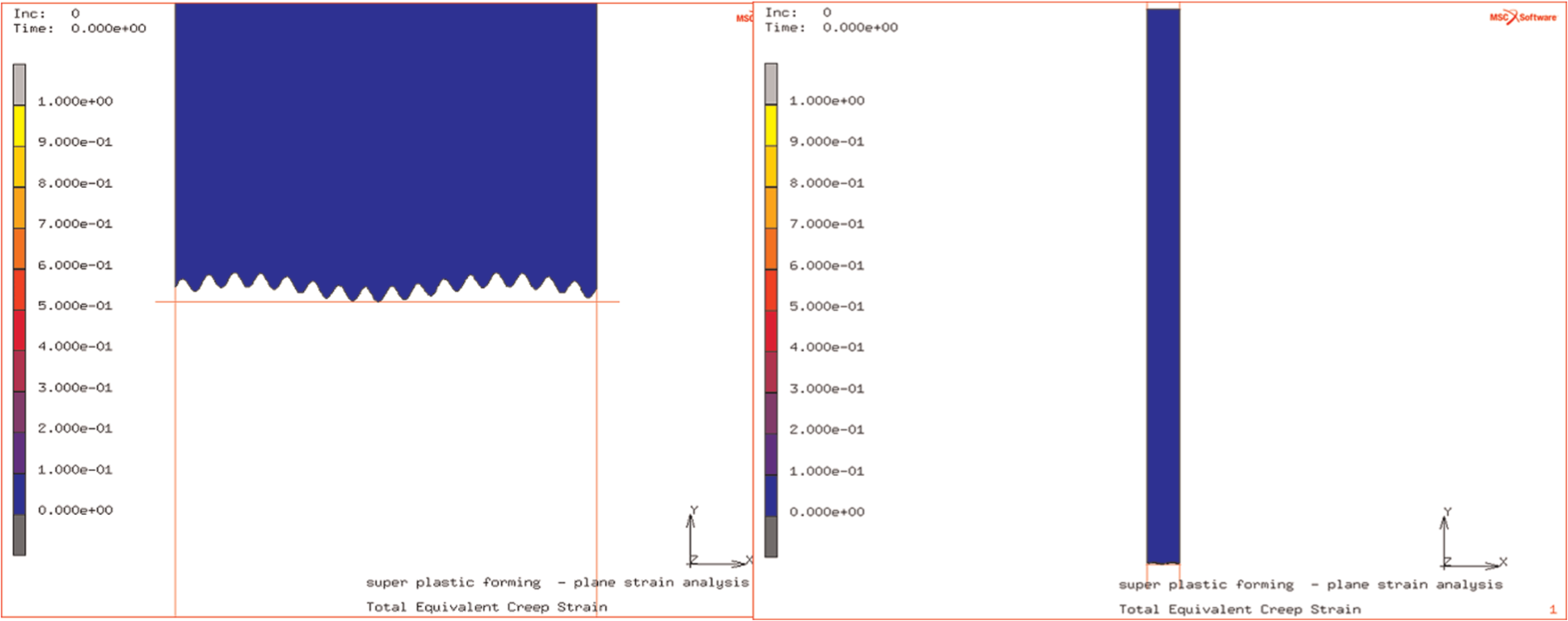
Surface of 1-mm sheet showing superimposed roughness of amplitude 2 µm and of wavelengths 3 and 51 µm.
The effect of localised heating was simulated to correspond to the target conditions that the induction heating experiments used. Thus, a target temperature of 950 °C was used – but so too was a temperature of 900 °C. This later was undertaken in order to investigate the effects relating to the lower temperature being the target temperature (for the same target bonding time of 60 s) and resulting in an unsatisfactory bond.
A somewhat arbitrary time of 600 s was chosen as the time to ramp up to the bonding temperature. This was done in order to give a bound to the effects of the shorter experimental heating time (to attain the bonding temperature) and, also, to look at the initial elastic effects and the build-up of stresses and elastic strains in the sheet and the heated region.
In order to simulate the stress behaviour, during the creep collapse of the inter-surface voids, a subsequent stage of the modelling included the power law creep models for the stress and strains during the 60 s DB period. For the materials, using a power law creep model, the stress exponent (n) had a value of 4.3, and the coefficients of proportionality in the material equations (with stress measured in MPa) were 1.34 × 10−11 and 2.3 × 10−11 at temperatures of 900 °C and 950 °C, respectively.
Three computational models were defined in the following. All were axisymmetric, to simplify the analysis, and all the accuracies were verified by increasing the mesh density and having four times as many elements:
The first consisted of a cylindrical region of the 1 mm plate with a diameter of 10 mm. This corresponded to the heated region in the bonding process – directly beneath the induction coil. This was rigidly constrained at the perimeter in order to find an upper bound on the stresses and deformations induced (mesh of 480 elements, requiring 5 s of CPU).
Another analysis included the 10-mm-diameter heated region as part of the larger 100-mm-diameter sheet. This was a simplified analysis in which no heat flow was modelled and was undertaken to study the mechanical effects of a realistic material, rather than that of a perfectly rigid one. This would correspond to a very rapid heating in which the induced temperature had insufficient time to flow into the surrounding material (mesh of 645 elements, requiring 7 s of CPU).
A final analysis was undertaken in which the heat flow out of the (inductively) heated region was permitted. This was undertaken in order to appreciate the effects of the time taken to heat to temperature might have on the stress state and the bonding conditions (mesh of 630 elements, requiring 14 s of CPU).
Results
The elastic stresses and the creep relaxation were studied for a localised heated region and a heated region in a plate. Not only the von Mises stresses were studied as they induce the plastic flow but also the stresses normal to the sheet thickness. The stress relaxation allows the elastic stresses to transform into the plastic deformation that is required to drive the collapse of the voids and hence allow the DB to occur. It should be noted that the components of stress and strain for these axisymmetric simulations are as follows:
11 is the radial direction;
22 is the axial direction;
33 is the hoop direction.
Deformation of a simulated surface
The deformation of the contacting surfaces is required in order to eliminate the gross voids and allow the diffusion (for the bonding) to happen. Figure 3 illustrates that there is no thin-shelled bubble being compressed, but the voids are deep seated and small and are remote from the application of the gas loading, conventionally used for DB. The simulation demonstrates how, initially, the applied load flattens the surface – bringing the rough surfaces into a localised contact. Some of the initial deformation at the surface that is occurring as the rough surface comes into contact is shown in Figure 4. In reality, this is the necessary precursor to full void densification – by both mechanical compaction and surface diffusion at the grain size level.
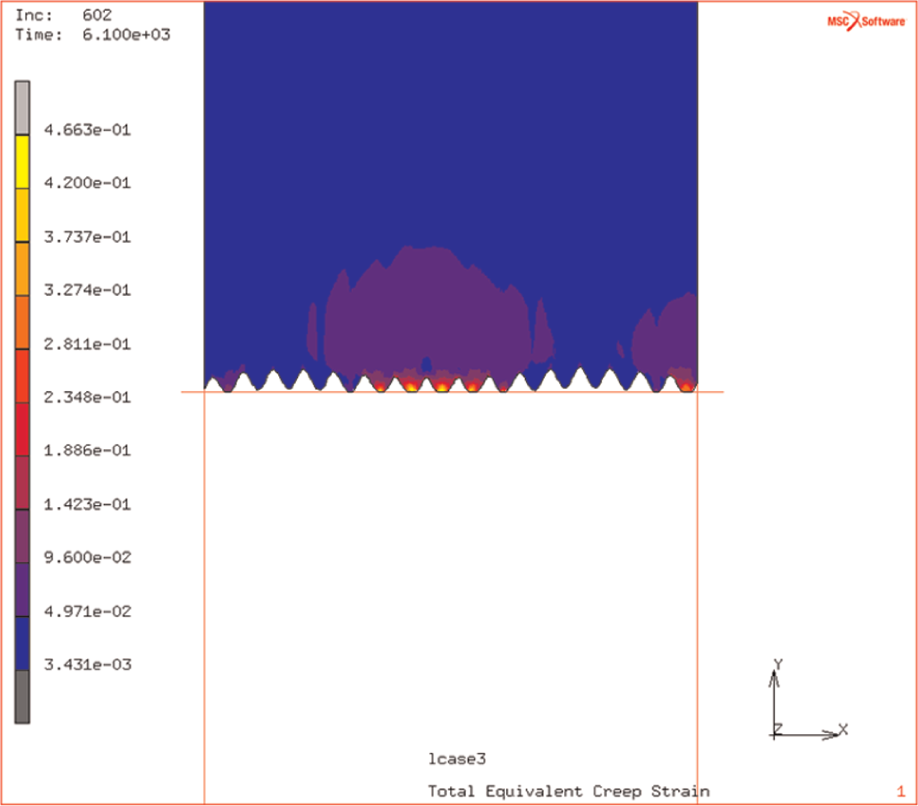
Simulation of rough surface achieving localised contact.
Figure 5 shows a micrograph obtained using a Zeiss scanning electron microscope (SEM), of a partially successful diffusion bond, showing some of the ‘frozen-in’ nature of the surface as the small-scale roughness has been removed by the DB. It is also seen that some of the longer wavelength roughness is still extant. This micrograph was obtained when the material was heated to a higher temperature, after some DB had been allowed to occur (allowing a change in microstructure that inhibited the DB process). It shows that the polished titanium surface was smooth on a local scale but had a very shallow (of the order of 1 µm amplitude) non-flatness superimposed.
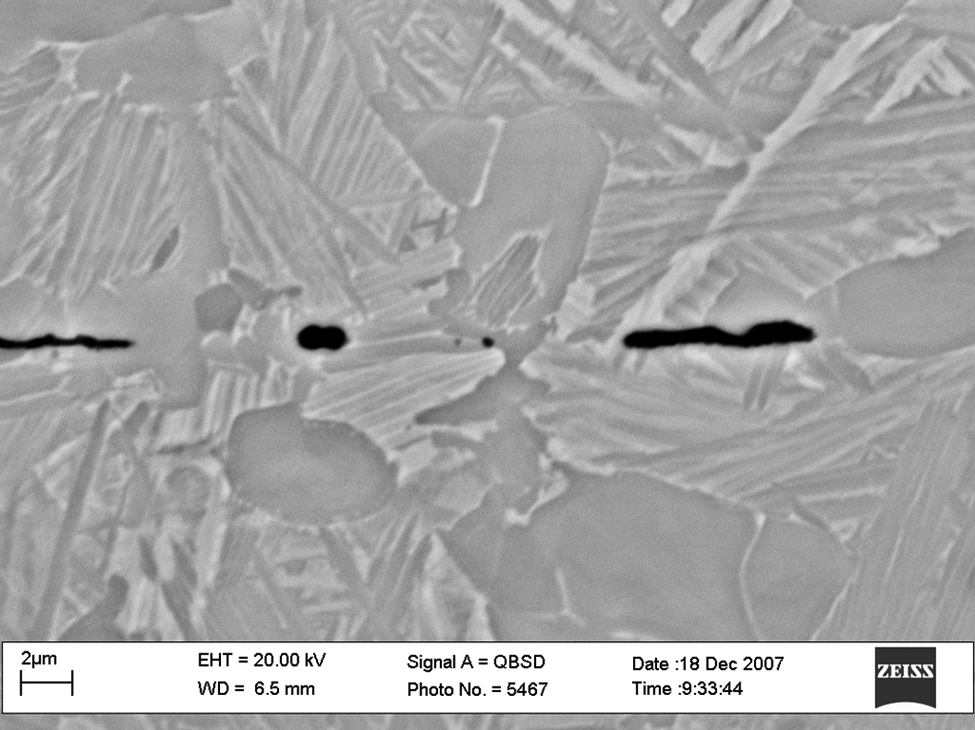
Micrograph of a partial diffusion bond, small-scale roughness vanished but some longer wavelength roughness is extant
Stress induced by heating of isolated constrained material
Due to the localisation of the thermal energy and the possibility of getting rapid heating, it was considered relevant to look at an isolated region in which the heating induced thermal strains and hence thermal stresses. The model was set up to improve the understanding of the effects of the localised heating – not necessarily to measure the induced stresses to be found in the real process. The cylindrical region was constrained in the radial direction – in order to simulate the effects of surrounding colder material, which, being stiffer and stronger, would restrain and hence induce stresses in the heated material. The results in Figure 6(a) show that the thermal expansion induces stress that increases with temperature up to a point, but then the decrease (with temperature) of the elastic modulus starts to dominate and so the induced stresses then decrease. The lack of constraint in the axial direction is responsible for the zero level of the axial stresses.
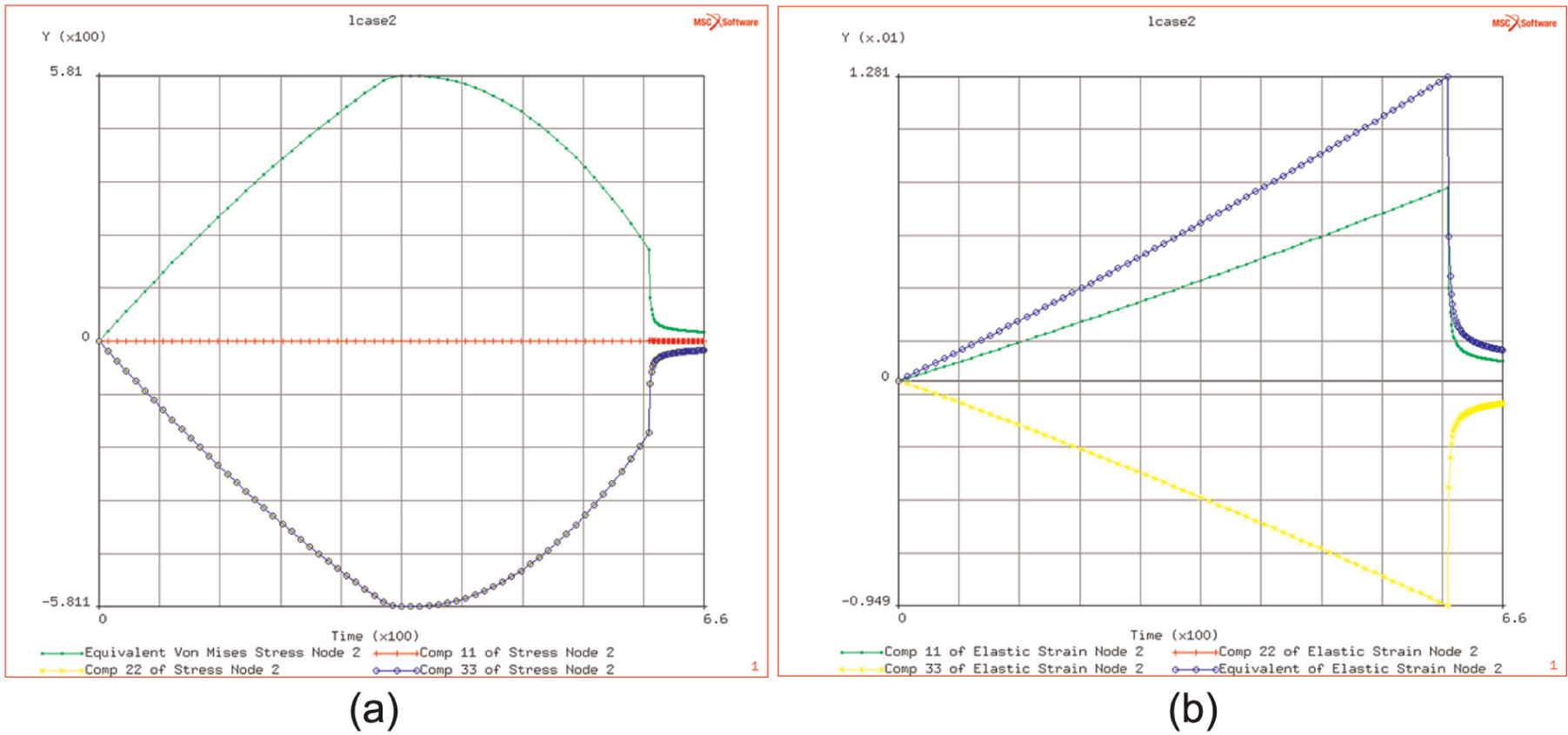
(a and b) History of thermal expansion–induced stress components, as temperature rises, and strain components.
It is seen, as shown in Figure 6(b), that the axial elastic strain (in the x or 11 directions) increases nearly linearly with linear increase in temperature, whereas the hoop or radial strains are negative due to the constraint on the thermal expansion. However, it does indicate that flow stresses (von Mises) of the order of 200 MPa may be achieved by the constrained or localised heating of the titanium sheet up to a temperature of 900 °C.
Stress relaxation of heated isolated constrained material
The localised heating of the plate induces high stresses but it is seen that these decrease quite rapidly, due to the creep behaviour of the titanium alloy. The stress relaxation is due to the power law creep and induces a tensile creep strain, of the order of 1.7%, in the axial direction and compressive ones in the hoop and radial directions (of half the value of the axial creep strain) (see Figure 7).
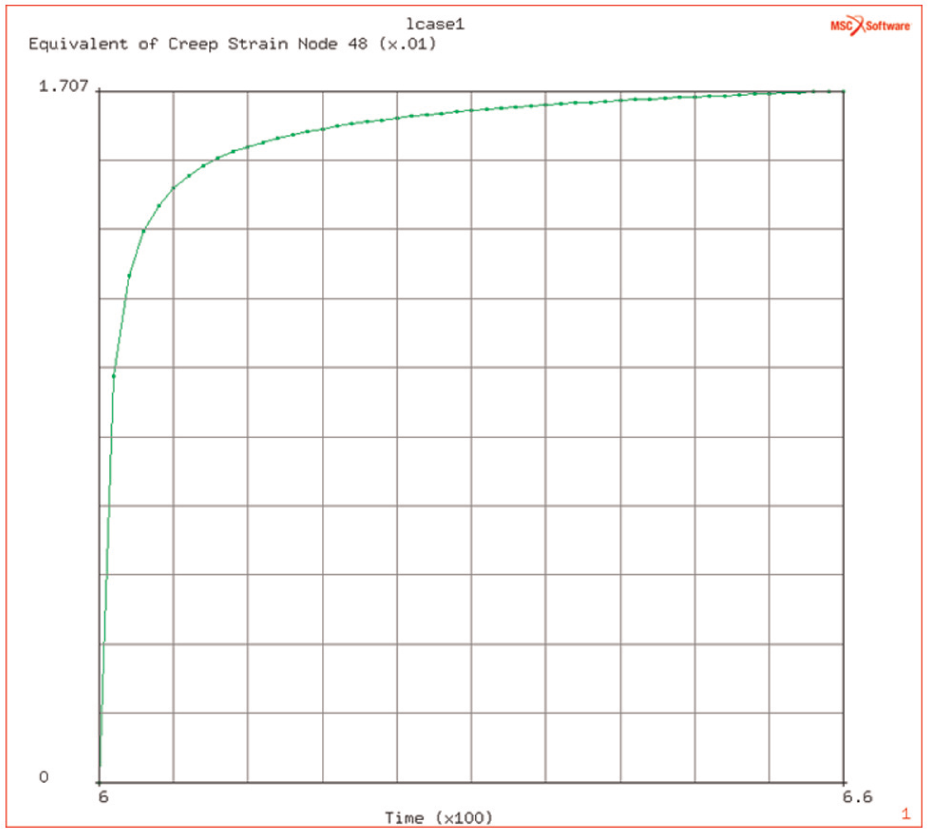
(Equivalent) creep strain evolution during dwell at elevated temperature.
The study of this effect at different temperatures, over the range of DB temperatures and during the 60 s long period of the DB, indicates that the originating flow stress (i.e. that at the end of the ramp up to attain the DB temperature) is affected by the temperature (over the range of 900 °C–950 °C) and decreases from 290 down to 200 MPa as the temperature increases from 900 °C to 950 °C (see Figure 8(a) and (b)).
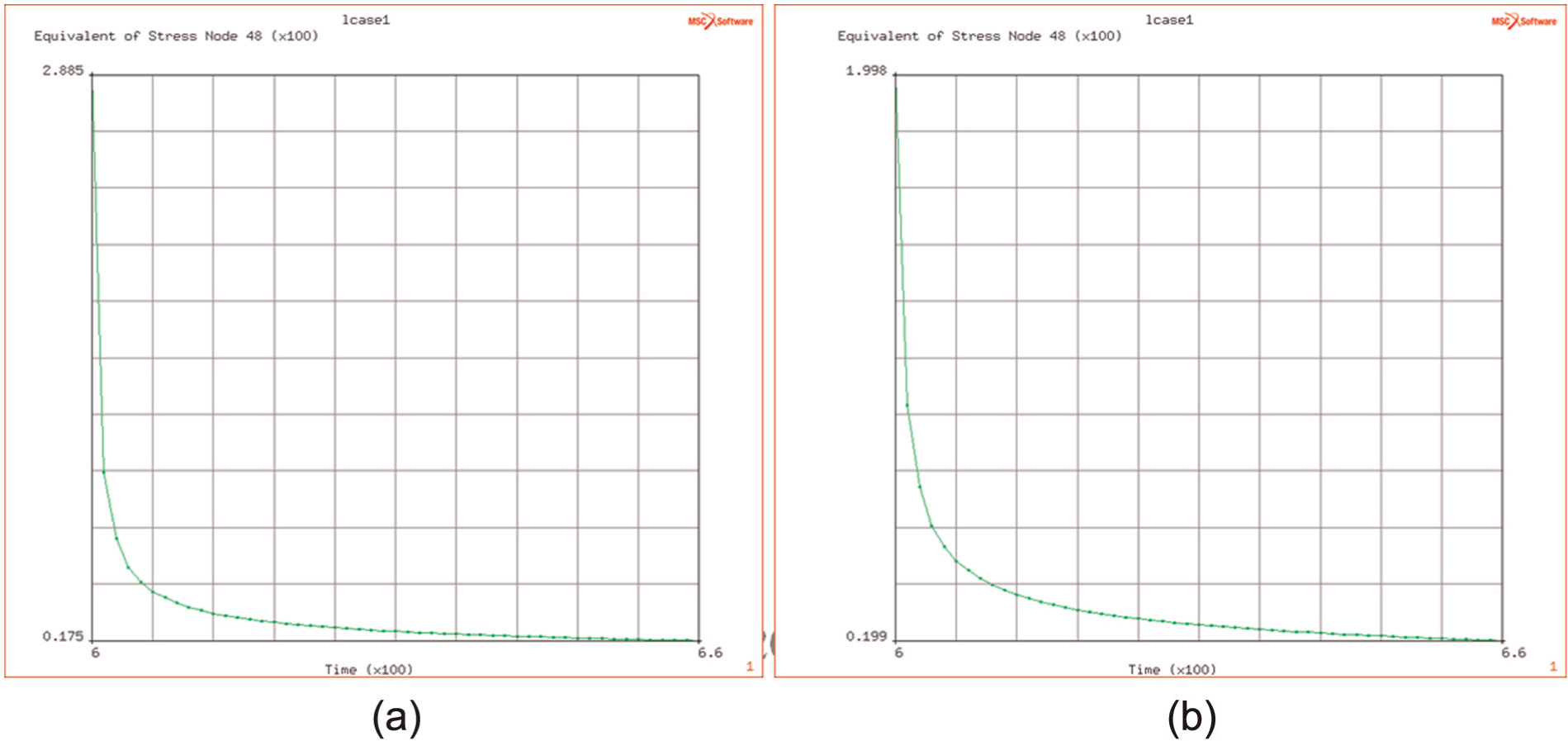
(a and b) Decay of (thermally induced) equivalent stress at 900 °C and 950 °C.
The temperature significantly affects the creep relaxation time of induced flow stresses. The time taken to reduce from 90% of initial level to the final levels (of 20 and 18 MPa, respectively) shows a 50% increase from 5 to 9 s at the higher temperature.
Stress induced by localised heating of a plate (mechanical analysis)
Surrounding the heated region by an annular plate of outer diameter of 100 mm leads, as expected, to a reduction in the flow stress in the heated region. This is due to the compliance of the surrounding plate. Some higher stresses are observed at the periphery of the heated region, due to the thermal gradients between the hot and the cold regions. The peak stress, in the heated region, is reduced from 581 to 170 MPa (see Figure 9(a) and (b)), and the elastic strains, in the heated region, are significantly reduced in the heating phase, with the effective elastic strain being reduced from 1.3% to 1.0% (see Figure 10).
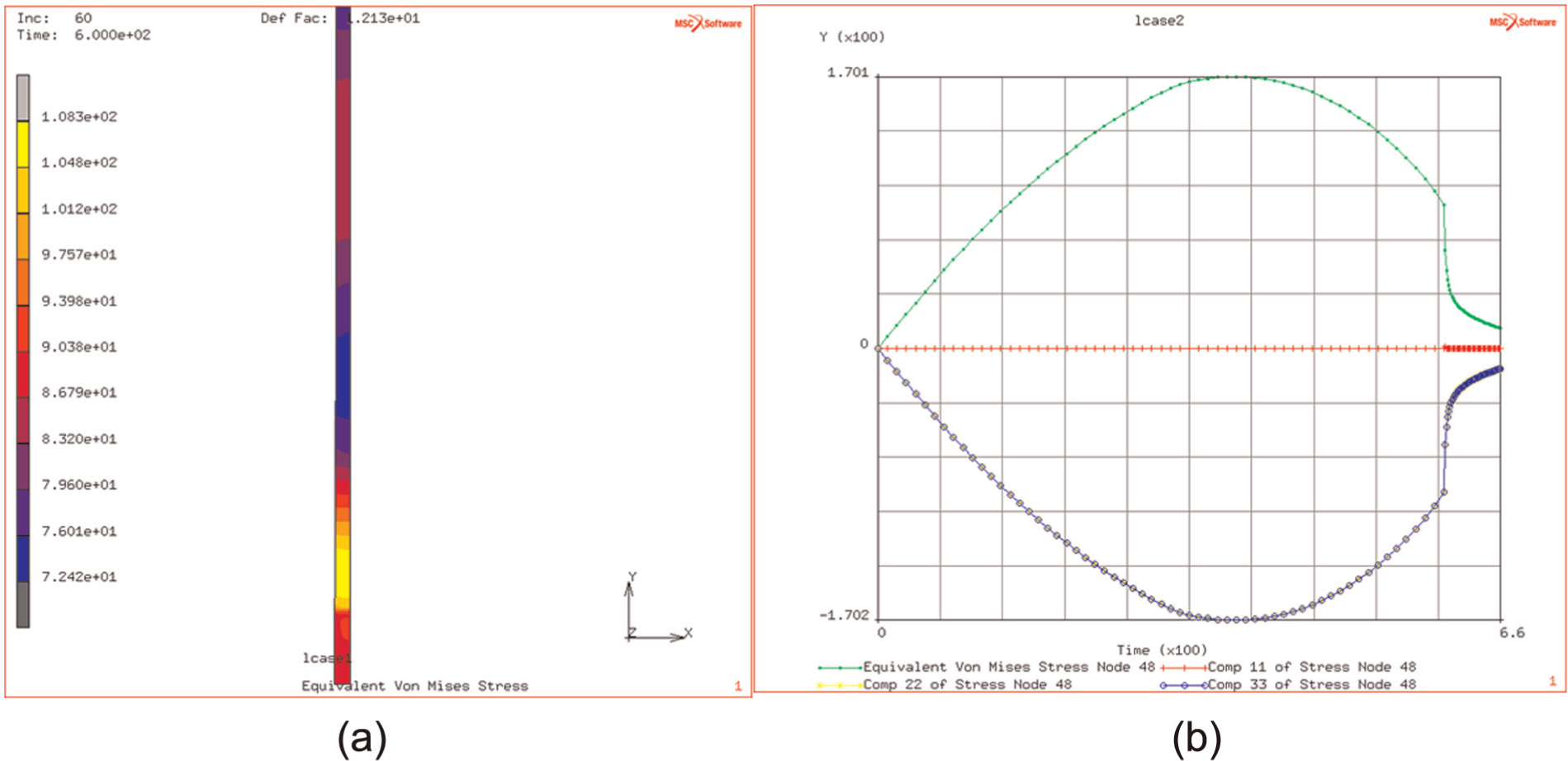
(a and b) Contour of stress in heated plate (prior to relaxation) and history stress, thermally induced during heating and relaxation.
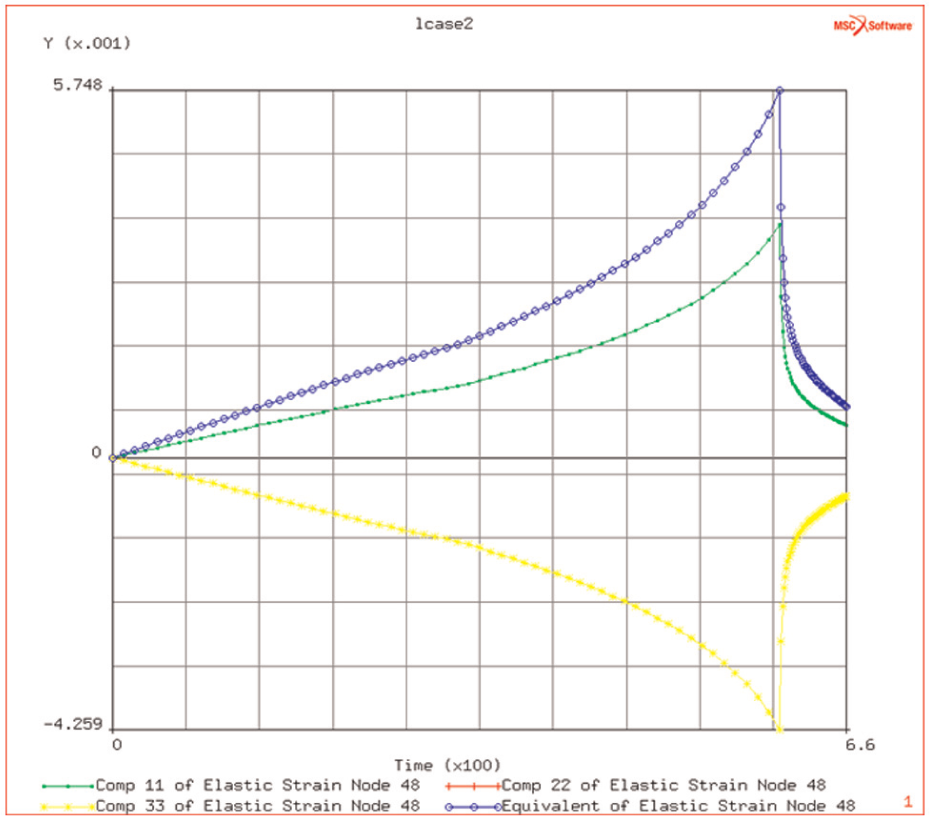
Elastic strain due to heating history and (creep) relaxation phase.
Stress relaxation in locally heated plate (elasto-creep mechanical analysis)
The localised heating of the plate induces high stresses but it is seen that they decrease quite rapidly, due to the creep behaviour of the titanium alloy. The stress relaxation, due to the power law creep behaviour of the material, induces a tensile creep strain, of the order of 1.0% (see Figure 11(b)), in the axial direction and compressive ones in the hoop and radial directions (of half the value of the axial creep strain)
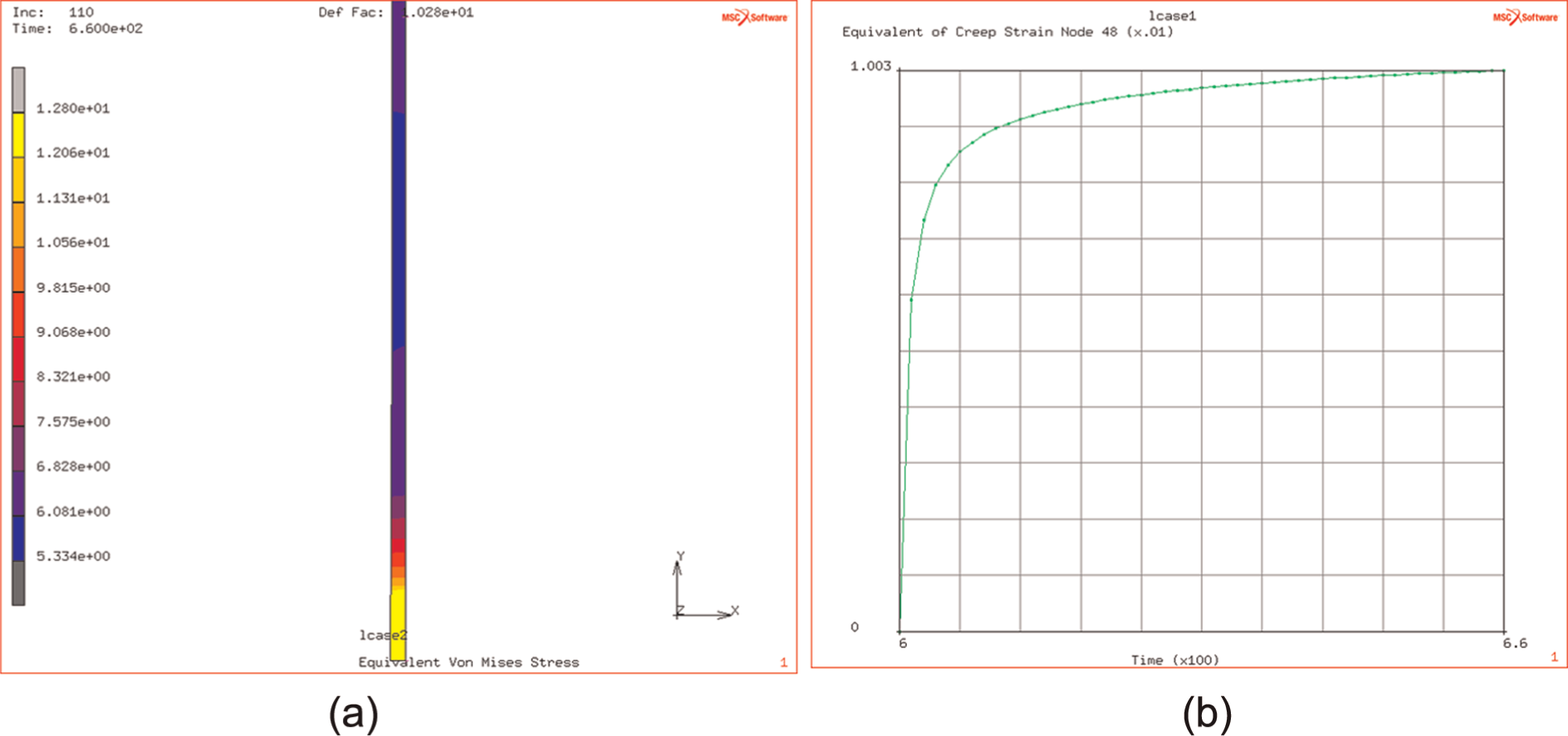
(a and b) Final stress distribution and creep evolution at bonding temperature.
The study of this effect at different temperatures, over the range of DB temperatures, indicates that the originating flow stress (i.e. that at the end of the ramp up to DB temperature) is affected by the temperature (over the range of 900 °C–950 °C) and decreases from 214 down to 160 MPa as the temperature increases from 900 °C to 950 °C. This is illustrated in Figure 12(a) and (b).
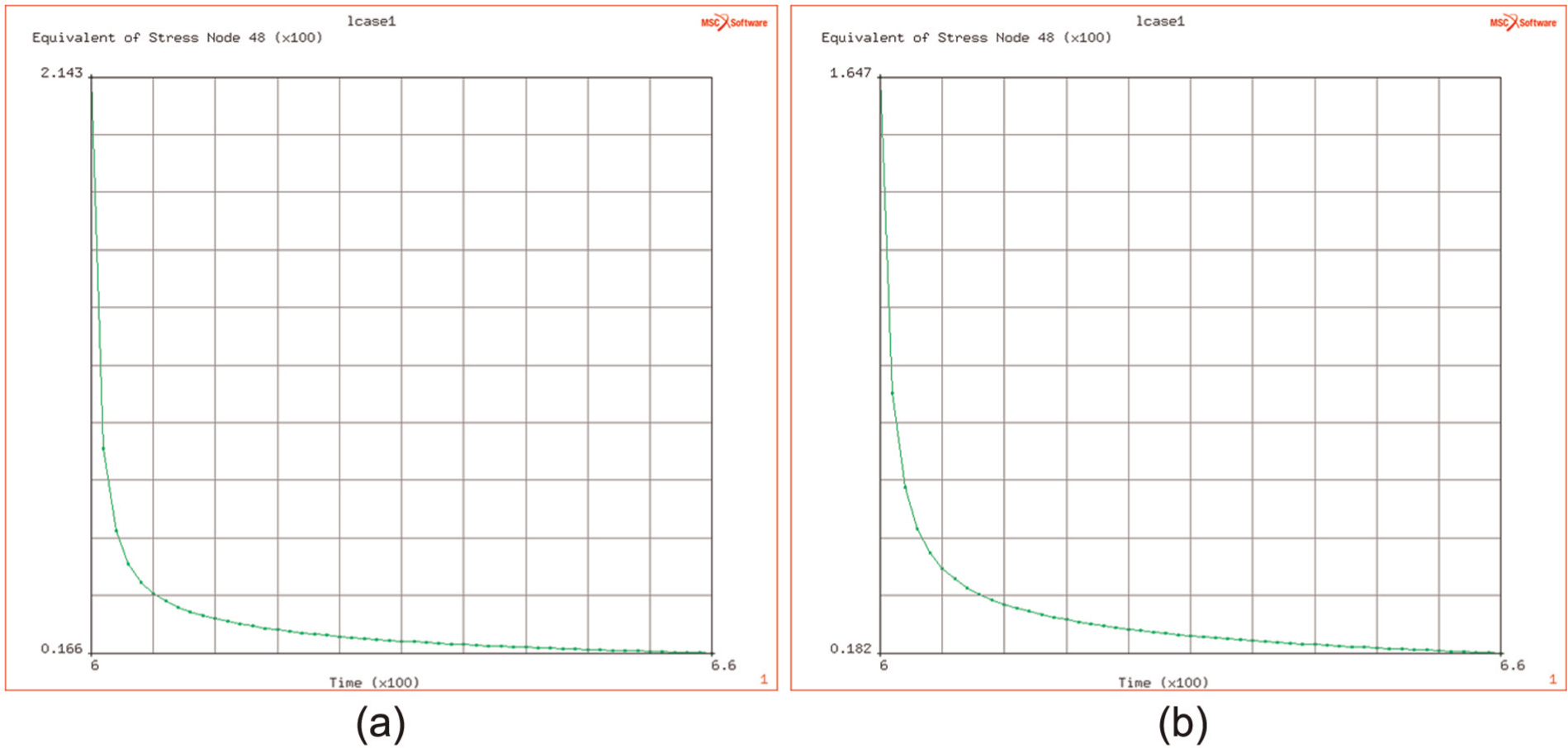
(a and b) Relaxation of stress at temperatures of 900 °C and 950 °C, after heating phase.
The temperature significantly affects the creep relaxation time of induced flow stresses. The time taken to reduce from 90% of initial level to the final levels (of 18 and 16 MPa, respectively) shows a 50% increase from 6 to 9 s at the higher temperature.
Stress induced by transient localised heating of a plate (thermo-mechanical analysis)
The simpler computer models form a progression from localised heating and mechanical effects – assuming a static nature to the temperature distribution – that is, neglecting the diffusive flow of heat out from the localised region of heating. Removing these limiting constraints and applying a model including the transient thermal effects will alter the temperature distribution and the thermally induced stresses. The flow stress, in the heated region, is reduced to 90 MPa and the peak flow stress, at the edge of the heated region, is reduced to 108 MPa, at the end of the heating ramp (see Figure 13(a) and (b)).
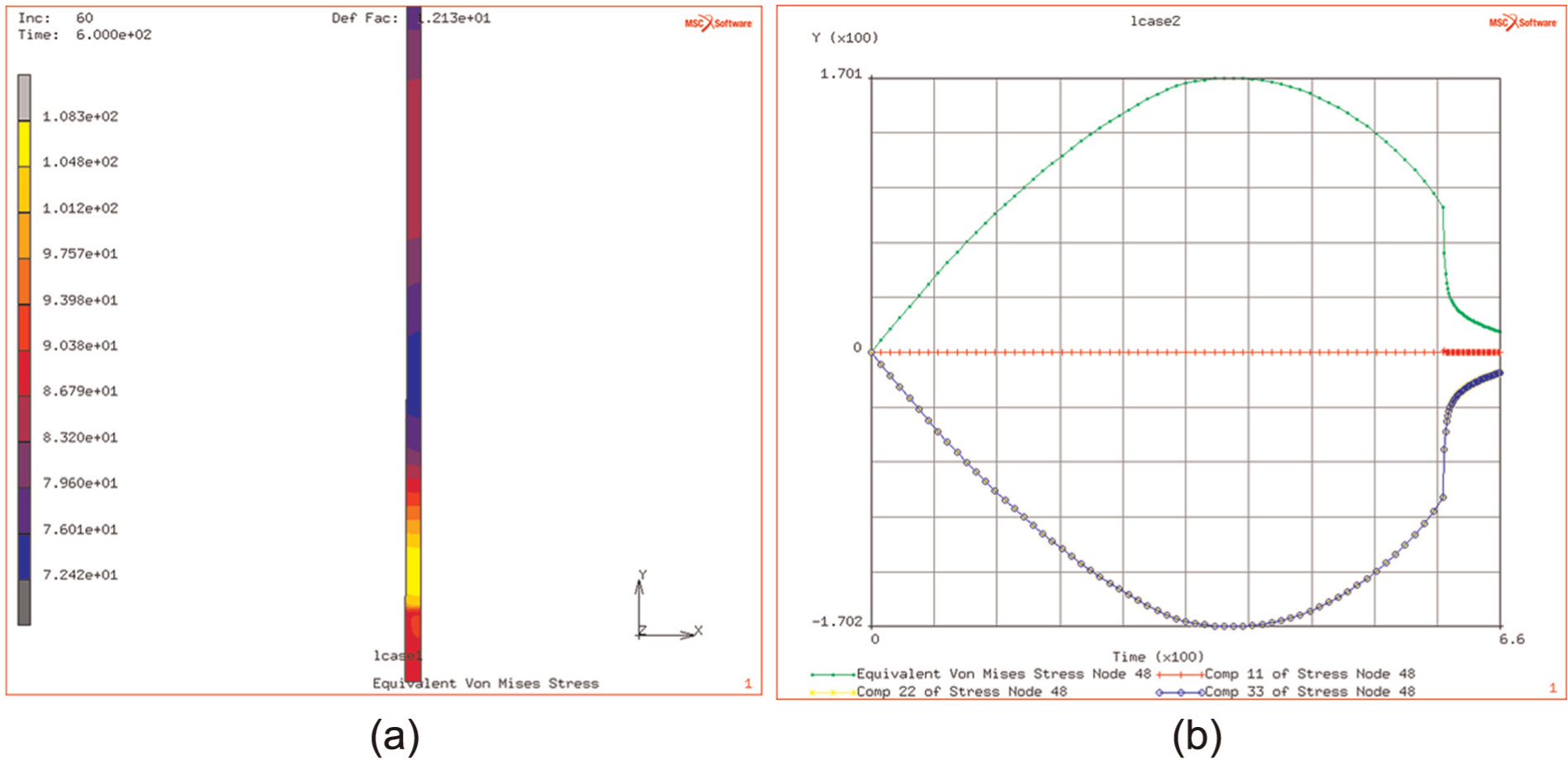
(a and b) Distribution of thermally induced stress, at the end of heating regime and history of stress evolution.
The effective elastic strain in this is reduced to 0.6%. This demonstrates the, as expected, further reduction in the stress levels in the plate due to the transient thermal heat flows into the surrounding material, reducing the temperature gradients and hence the induced stress levels.
Stress relaxation by transient localised heating of a plate (thermo-mechanical analysis)
The localised heating of the plate induces high stresses but it is seen that they decrease quite rapidly, due to the creep behaviour of the titanium alloy. The stress relaxation, due to the power law creep behaviour of the material, induces a tensile creep strain, of the order of 0.4% (see Figure 14(b)), in the axial direction and compressive ones in the hoop and radial directions (of half the value of the axial creep strain).
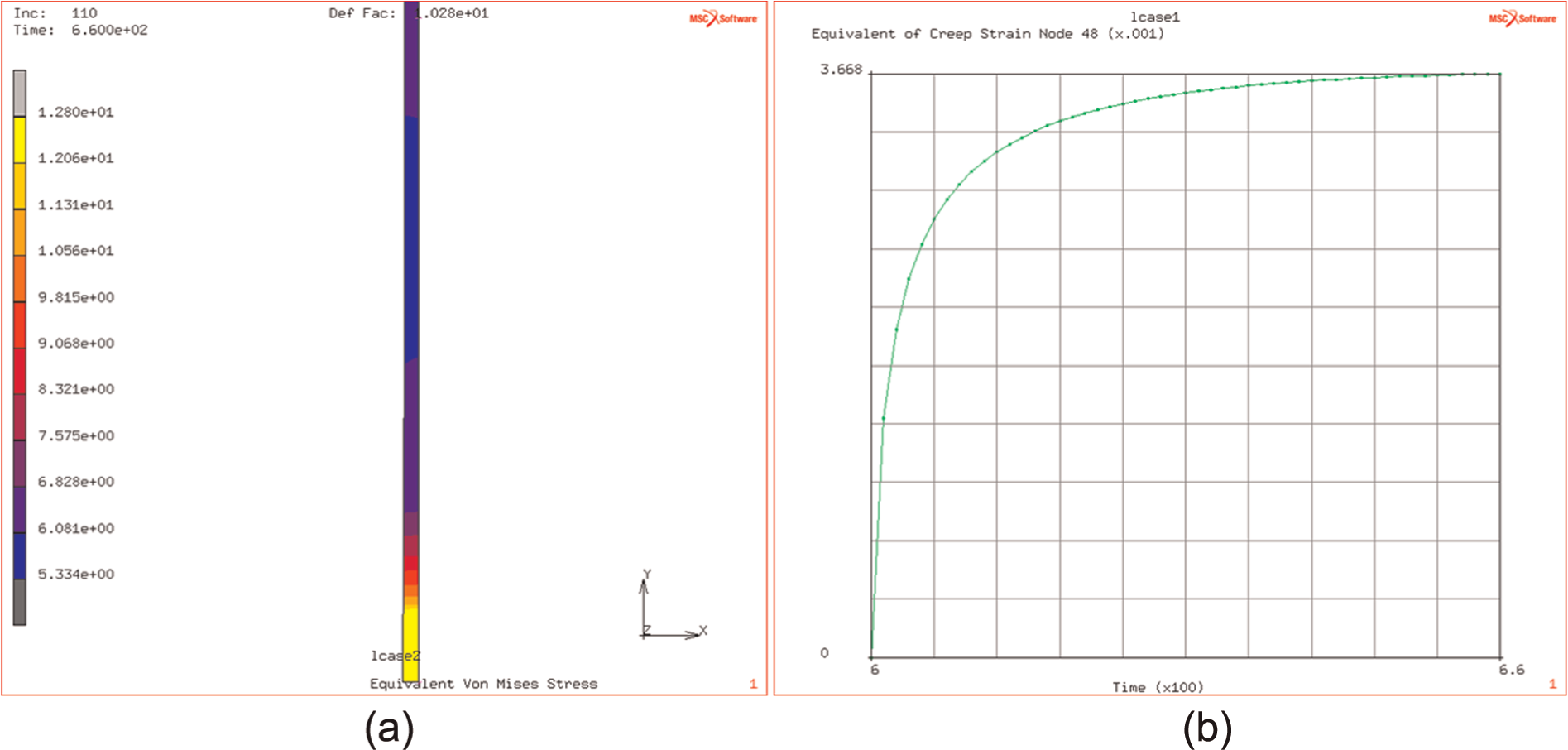
(a and b) Stress distribution after 60 seconds of relaxation (bonding) in heated plate and increase of creep strain in the stress-relaxation bonding phase.
The study of this effect at different temperatures, over the range of DB temperatures, indicates that the originating flow stress (i.e. that at the end of the ramp up to DB temperature) is affected by the temperature (over the range of 900 °C–950 °C) and decreases from 110 down to 90 MPa as the temperature increases from 900 °C to 950 °C. This is illustrated in Figure 15(a) and (b).
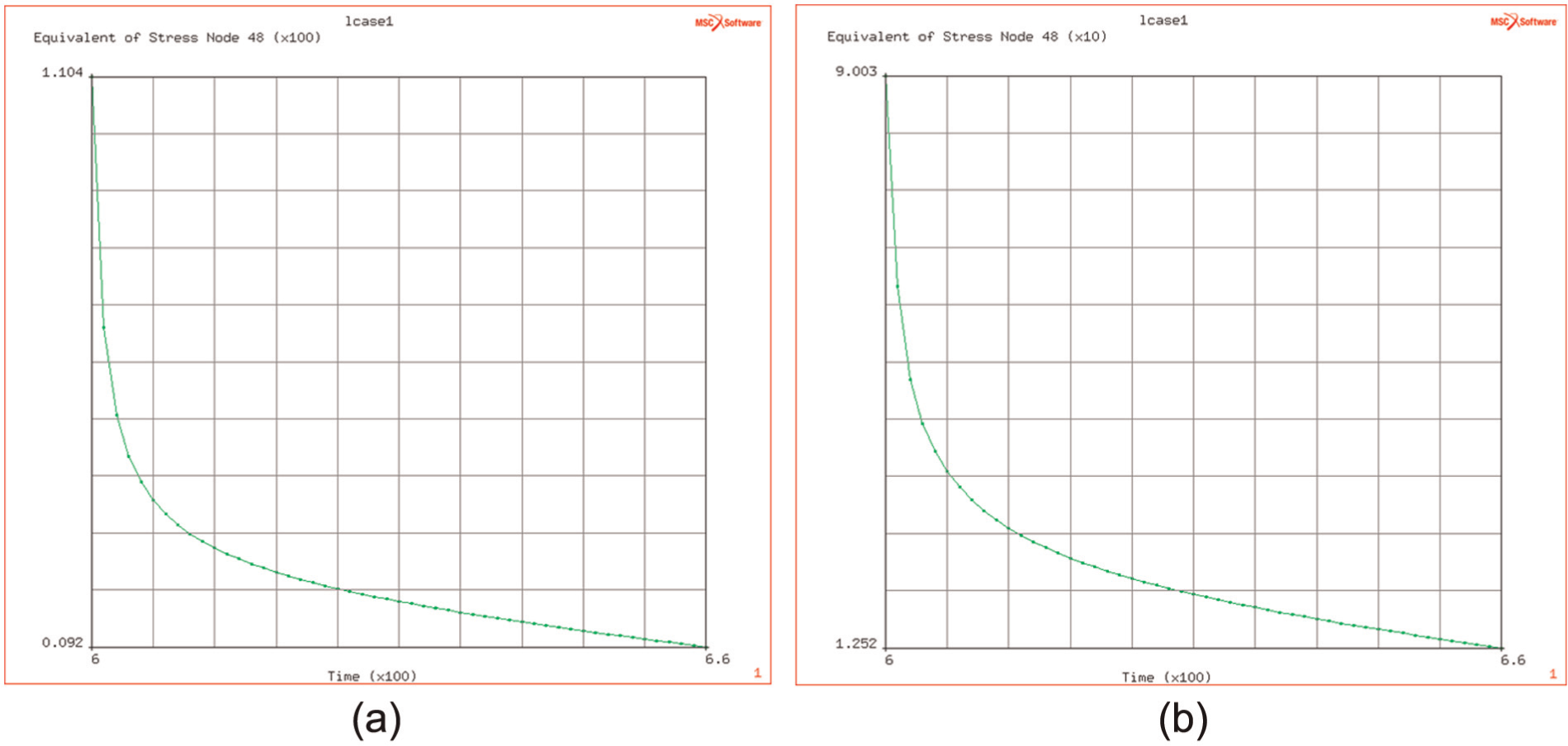
(a and b) Effect of bonding temperature on rate of stress relaxation during bonding (after heating to desired bonding temperatures of 950 degC and 900 degC respectively).
The temperature does not significantly affect the creep relaxation time of induced flow stresses. The time taken to reduce by a factor of 0.9 from initial level to the final levels (of 12.5 and 12.5 MPa, respectively) shows only a small increase from 27 to 28 s at the higher temperature.
SEM micrographs of bonds and voids
The successfully diffusion-bonded specimens were bonded at a temperature just below 950 °C as lower temperatures, such as 900 °C and 920 °C did not produce a good bond in the 60 s period of the DB cycle (the bonding period as opposed to the ramp up to and down from temperature). As can be seen from the micrographs (Figure 16), a good bond was obtained and a fine microstructure was maintained. It can be seen that it is difficult to assess the original line of the surfaces, and it requires the matching of the grain structure (at the edge of micrographs) and the presence of the occasional defect to so do. The inset micrograph (which is part of the montage) illustrates two groups of small defects/voids: each of size approximately 1 µm and periodicity of 50 µm apart; this is consistent with a periodicity of about 50 µm of the long wavelength roughness or flatness.

Montage of micrographs (obtained from a scanning electron microscope) showing line of diffusion bonding and defects.
Other micrographs obtained from specimens heated above 950 °C, Figure 17, show long wavelength roughness effects more clearly. This is because the higher temperature results in a phase change and much larger grains of beta-phase titanium alloy Ti64. The larger grains are more resistant to creep deformation and hence, effectively, ‘freeze in’ the surface structure. It is easily seen that the larger grains are of the order of 50 µm size, and that the voids/defects still remain – with some detail of the flatness or waviness observable.

Micrograph of ‘overheated’ bond – showing coarse-grained structure and longer wavelength roughness.
Although the larger grains enable easier tracing of the diffusion bond, as it is easier to match up a montage of micrographs of such coarse-grained structure, it is generally not desirable to so treat the material. This is because such microstructure is not suitable for subsequent superplastic forming operation and the mechanical properties of beta-phase titanium are not desired.
Discussion
The physical experiments demonstrate that, with localised heating, it is possible to diffusion bond thin sheet material quite rapidly (in under a minute). Previous studies of DB (notably Pilling 28 ) confirm that the need for the understanding of the nature of the surfaces to be bonded and their mechanical deformation is a necessary precursor to the diffusion processes required for the bond.
This is because diffusion processes generally speed up as the inverse square of the distance over which the diffusion is to occur. So reducing the distance by a factor of 10 will speed up the process a 100-fold. The numerical modelling of a simulated surface (with short and long wavelength aspects to the surface topography) demonstrates that the reducing of the long wavelength proportion of the surface amplitude variation is required before good void collapse/dissolution can occur. This is confirmed by the micrographs that have a ‘frozen-in’ structure and show a witness of nature of the voids collapsing. It is seen from the simulation results that localised heating can induce significant stresses and plastic (creep) deformation in a thin sheet (1 mm) of the Ti64 titanium alloy.
Such stresses are hard to measure in the environment and situation of the bonding process, and the numerical modelling provides a useful insight into the stress levels and behaviour. Evolving the numerical experiments from an ideal situation of rigidly constrained and heated material to a more realistic dynamic heating of a titanium plate allows this. The numerical modelling demonstrates that considering the localised heated region in isolation with a simulated ‘rigid material’ constraint produces a stress field, in the heated region, that is very similar, and not much higher (200 MPa as opposed to 170 MPa) than when the constraint is supplied by colder material.
When transient heat flows are included, as part of a fully coupled thermo-mechanical model, into the simulation, then the stresses drop substantiality to around 90 MPa. This indicates that if the initial heating to bonding temperature is slow (of the order of 600 s), then a more detailed numerical model is required to maintain good accuracy of the stress field.
It is noted that the lower stress field found in the fully coupled analysis is subject to a faster decay (due to creep stress relaxation) but this is a function of the power law creep material properties of the titanium alloy. This leads to a final stress field (at the end of the DB regime) of between 20 and 12 MPa for a bonding temperature of 950 °C. This is still a good stress level for superplastic forming and DB. The relative proximity of the stress levels at different temperatures suggests that these global stresses themselves do not make a great difference in the diffusion processes except in so much as to bring a great preponderance of the surface into intimate contact, so required for good and fast diffusion processes to occur.
The intimate contact then drives the localised small void (of the order of grain size or less) collapse, and the diffusion processes then produce the bond. This is posited because the DB was achieved with a time of 60 s at a temperature of approximately 950 °C and not at lower temperatures such as 920 °C. The conventional DB models, such as Pilling, 28 deal with this temperature sensitivity aspect.
It should be noted that induction heating can heat very quickly and, if that is required, then the effects of the transient heat flow on reducing stress levels will be greatly reduced – leading to the simpler (mechanical) modelling being satisfactory for studying or simulating the process.
Future work
Further work could measure the initial surface flatness and roughness and look at the ways of measuring it during the bonding process. This is required for a better understanding of the nature of the bonding process and the phases (void collapse and material diffusion) and could be well supported by more detailed numerical simulation of simulated surface topographies under appropriate stress conditions. Although workers such as Wu et al. 31 have recently undertaken surface measurements, and corresponding simulations, this has involved bonding times of thousands of seconds, rather than the 60 s bonding time used in this work.
And this reduction in bonding time should facilitate the development of experiments to study the void collapse in isolation from the diffusion processes themselves. It is anticipated that future work will include the development of improved techniques for monitoring the temperatures and heat flows during the process and controlling them. It is hoped to have obtain access to specimens bonded by the classic HIP processes and to perform both mechanical and metallographic examinations of the bonds.
Conclusion
It has been shown that localised direct heating can induce high levels of stress, and that these thermal stresses can accelerate the DB process – as atmospheric loading effects are, by comparison, trivial. This leads to good diffusion bonds in the titanium alloy Ti64 in less than 60 s – without the use of expensive pressure vessels or presses. This opens up the possibility of much more affordable DB and production of material efficient and functionally useful cellular materials. A sequence of numerical models was found useful to understand the effects of process parameters on DB. Some modelling looked at accelerating the collapse of the voids, defined by the relative flatness and roughness of the metallic sheets at different spatial wavelengths. It appears likely that the localised heating drives the DB process as a totality but that raising the temperature as much as possible assists the actual diffusion processes required for bonding to occur and is an important aspect of accelerating the process. The acceleration of the process will be such that there is insufficient time for significant grain growth to occur. The use of ‘overheated’ material, with its effective ‘frozen-in’ voids, has been found useful to assist in observing and understand the evolution of the surface topography of the titanium sheets during the DB.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was funded by the Midlands Aerospace Alliance, UK and they supported the dissemination of the research as part of their Aerospace Technology Exploitation Programme (ATEP).