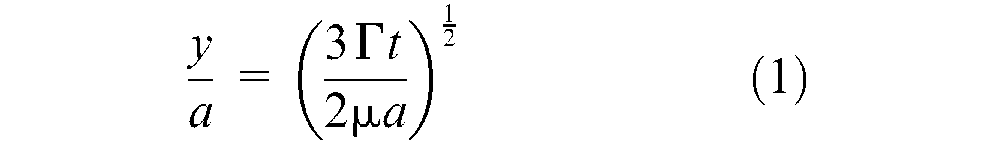Abstract
Previous studies into the use of polymer powders such as Nylon-12 for laser sintering have shown part property degradation caused by repeated heating and cooling. This research investigated the recyclability of a new thermoplastic polyurethane powder in the laser sintering process. The work has novel value as there has been no previous work on processing this material by laser sintering.
A virgin powder sample was retained, along with a sample after each of four laser sintering builds. Each sample’s recyclability was investigated by particle size analysis, differential scanning calorimetry, hot stage microscopy of particle pairs, melt flow index, tensile test sample manufacture and tensile testing.
Upon recycling, no significant trend in the elastomer particle size distribution was identified, and thermal properties were unchanged, as shown by differential scanning calorimetry analysis. Hot stage analysis showed that recycling had no clear effect on sintering time. Melt flow index results showed a small increase in flow rate upon recycling, corresponding to a slight drop in viscosity, suggesting reduced chain length. The outcome of tensile tests showed a minor decrease in strength while elongation at break and Young’s modulus showed a negligible decrease.
The results have shown the possibility of recycling the thermoplastic polyurethane bed powder several times through the laser sintering process without any significant degradation or reduction in properties.
Introduction
In the context of polymers, sintering is defined as ‘the formation of a homogeneous melt from the coalescence of powder particles’. 1 Laser sintering (LS) is an additive manufacturing (AM) process and is an attractive method for producing polymer parts as it requires no tooling. Furthermore, it provides greater design freedom than more conventional methods (such as injection moulding), and is well-suited to applications where the three-dimensional (3D) geometry is complex and/or volumes are low.
LS
LS is a particularly useful AM process as it is versatile enough to allow polymers, metals, ceramics and even some composites to be processed. 2 LS facilitates the building of 3D objects by stacking and consolidating successive layers of powdered material. When processing semi-crystalline thermoplastics, the temperature in the bed is kept just below the melting point of the powder; the CO2 laser heats the powder a small amount to raise its temperature just above the melting temperature, Tm, causing it to melt.
In theory, any material that can be powdered may be processed by LS and, hypothetically, will retain the properties of its bulk form. The unsintered powder surrounding the part in the bed provides support for the component under construction so supports do not need to be built and removed after processing.
However, there are limiting factors; as with most AM techniques, the appearance of a part is enhanced if it is made up of many small layers rather than a few large ones, but this lengthens the time taken to build a component – as a compromise, layers are typically 100 µm thick. Amorphous materials are not as suitable for LS as semi-crystalline polymers as they do not have such a sharp melting point, so the laser is used to raise the powder to its glass transition temperature (leading to poor contact between particles and so poor mechanical properties) rather than fully melt the local area of polymer powder. 3
The applications of LS are widespread in the AM industry; when large numbers are required, manufacture is accelerated further by the ability to make multiple components in a single bed of powder. Complex 3D objects can be directly reproduced from computer-aided design (CAD) designs in a rapid process. 4 There is a range of very attractive qualities of laser sintering, such as the possibility of good mechanical properties and microstructures. There are few restrictions on both form and shape so objects of virtually any geometric complexity can be constructed without tooling. Moreover, human interaction is minimal during the process, materials utilisation is efficient and customisation of components is simple. 5 However, the range of materials that can be processed by LS is still limited – polymers being restricted mainly to nylon-11 and -12, and in reality, it may not be possible for many powders to be consolidated to full density or even to be processed. 6 Properties obtainable in theory will frequently not be achievable once sintering has been carried out, and it can take numerous attempts before optimum processing conditions for a particular material are found.
There is a great deal of interest in elastomers for use in LS; there are some available but there is a need for a wider range of materials and properties. For this reason Elastollan, a thermoplastic polyurethane (TPU) elastomer supplied by BASF Polyurethanes (Derbyshire, UK), for which no previous research into LS has been reported, was selected for this research. The material’s properties, when injection moulded, are encouraging for use in applications such as sports (particularly snow sports) – with notable abrasion and impact resistance, hydrolytic stability and low-temperature flexibility. 7 The possibility of processing this material using LS is of interest owing to the numerous benefits of the process over injection moulding, and identified by material research as being potentially valuable.
Extensive research has been carried out into processing nylon-12 using LS – this was used as a basis of research for this work. Nylon-12 is semi-crystalline and has a sharp melting point. Non-crystalline (or amorphous) polymers are not usually well-suited to LS as their broad softening range renders a complete melt difficult to obtain, thus leading to weak and brittle parts where the part interior is made up of powder particles joined together at points rather than a strong bonding network.
Several consolidation mechanisms may take place during laser heating: liquid phase sintering, partial melting, full melting, chemically induced binding or, although less likely, solid state sintering. (Laser consolidation of polymers usually involves full or partial melting of thermoplastics.) Consolidation of powdered materials in LS depends strongly on initial powder composition, the final desired composition and aspects of the required structure, such as porosity, density and microstructure. 6
The sintering process has been extensively studied for metals and ceramics, where volume and surface diffusion transport mechanisms are important. Polymers diverge from this – the diffusion of organic materials requires high activation energy so viscous flow is the most important transport mechanism for polymeric materials, as outlined by Thümmler and Thomma in 1967. 8
In determining the suitability of a polymer for sintering, the coalescence of pairs of particles on a hot stage microscope can be analysed to estimate rates and mechanisms that may occur during laser heating. This path was followed by Bellehumeur et al. 8 in 1996 – they concluded that particle geometry had no effect on sintering rate but, in general, the rate increased with decreasing melt viscosity. Additionally, they established that incomplete powder coalescence led to high porosity and poor mechanical properties. This suggests that when carrying out research of this nature, various grades of powder must be investigated.
Frenkel derived the first analytical model describing coalescence rate occurring by viscous flow for two identical spherical particles. This model may be applied to hot stage work such as this, which is outlined later in this article.
Recyclability of polymers in LS
Higher viscosity of molten polymers tends to give greater strength when solid owing to increased forces of attraction between the larger molecules and greater energy required to pull them apart. However, previous research by Kruth et al. 6 has shown higher viscosity to result in reduced performance during sintering, as observed in studies of nylon-12. This observation can be taken from Frenkel’s model describing the coalescence rate occurring by viscous flow for two identical spherical particles; sintering rate is inversely proportional to √(µ), where µ is viscosity
a, t, y, and Γ are particle radius, sintering time, sintering neck radius and surface tension of the material respectively.
Nylon-12 tends to ‘degrade’ when it has been heated and cooled several times in the LS process, as long heat exposure leads to chain growth via a polycondensation reaction, a rise in molecular weight and an associated rise in viscosity, adding complexity to process control when recycling bed powder. Consolidation conditions and melting temperature become less consistent, leading to non-smooth, dimpled patterns (‘orange-peel’ texture) on the surface and inferior mechanical properties after several runs. As a result, recycled nylon-12 is frequently combined with large proportions of virgin material (for example, 50%) of intrinsic lower viscosity.
In their study into the recyclability of nylon-12, Dotchev et al. 9 found the most influential parameters for powder aging and degradation were the temperature to which the un-sintered bed material was exposed, and the length of exposure. They found that the melt flow index (MFI – a measure of polymer viscosity) is a sensitive indicator of changes in powder properties and provides a relatively fast and inexpensive method of measuring the magnitude of powder degradation during laser sintering.
In order to keep LS costs low it is beneficial to use the minimum powdered material possible; this may be achieved by re-using the excess material from the LS machine. This material would be heated and re-used several times to maintain low wastage – a particularly important consideration in large-scale manufacturing. Additionally, efficient utilisation of the used LS material is important owing to the high cost of virgin material.
Taking these factors into account, the aim of this research was to understand the effect of recycling on the TPU bed powder in the LS process.
Experimental procedure
Samples under analysis
Virgin powder was sieved and used for an LS build. The remaining powder was removed from the build chamber and re-sieved for use in a subsequent build. Consequently, a sample of powder was retained after successive LS builds.
The samples under analysis in this investigation were:
R0 ‘virgin’ elastomer;
R1 elastomer heated once;
R2 elastomer heated twice;
R3 elastomer heated 3 times;
R4 elastomer heated 4 times;
where ‘R’ refers to recycle number, the number of times the powder has been reused. The elastomer was in the form of black powder. Figure 1 shows the process of obtaining recycled powder from the LS process.
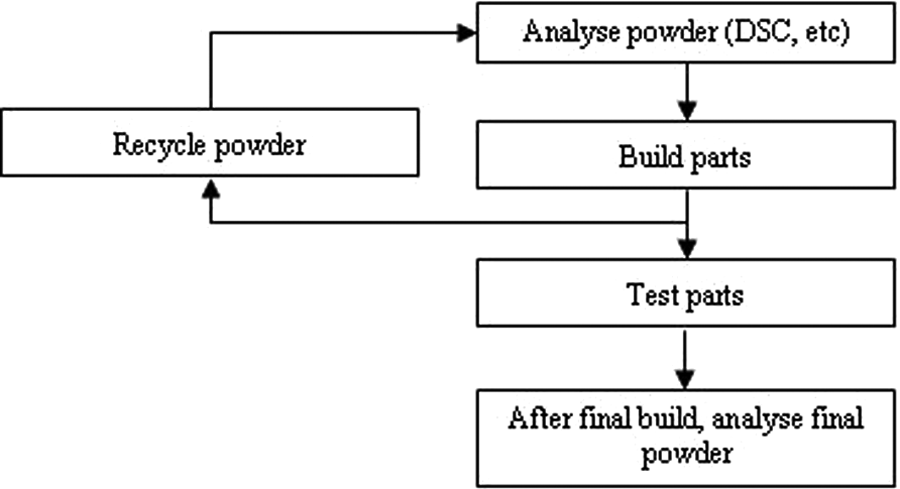
A schematic diagram to show the process of obtaining recycled powder.
Particle size analysis
Particle size analysis was carried out to assess the effect of recycling on the amount of particle size distribution, by using a wide sampling population. This test is important, as particle size (as well as geometry) can influence the build process during LS.
Methodology
A small sample of powder was spread over a glass slide that was then placed under a microscope. The diameter of every particle on several slides was measured using imaging software (Image Pro-Plus). Between 150 and 300 particles were measured for each sample. The measurement chosen was half-way between the maximum and minimum dimensions of the particle – the elastomer powder used was produced for rotational moulding, manufactured by cryogenic grinding-down of pellets. As a result, the particles were not spherical – they were jagged and irregular – so the diagonal was drawn for consistency (see Figure 2).
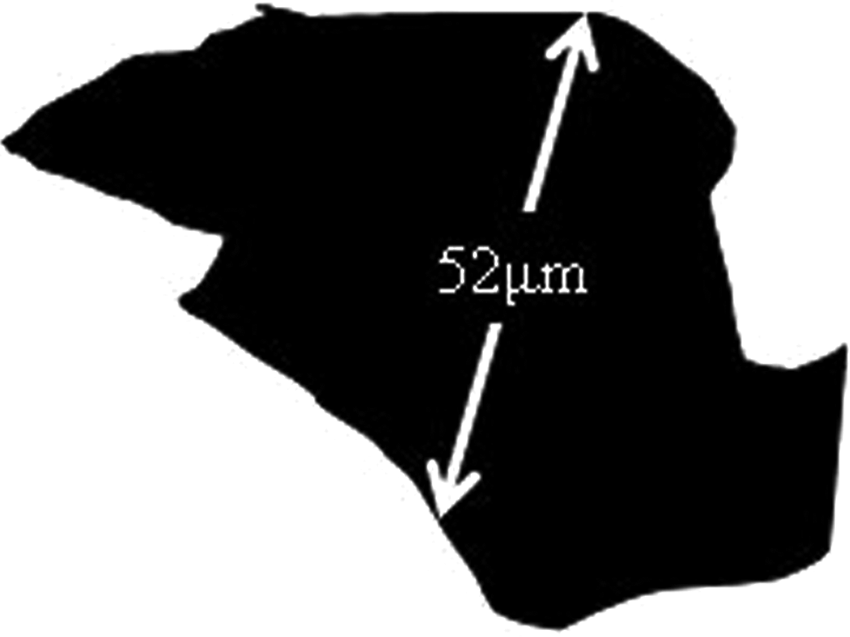
A typical powder particle.
Differential scanning calorimetry
Differential scanning calorimetry (DSC) is the measurement of the change in difference of heat flow to a polymer sample and to a reference sample, while both are subjected to a controlled temperature program. 10 DSC measures the amount of energy absorbed or released by a material sample when it is heated or cooled. The temperature range during which a material melts on heating is recorded, as is the range over which it crystallises on cooling. Results are in the form of the graph shown in Figure 3.
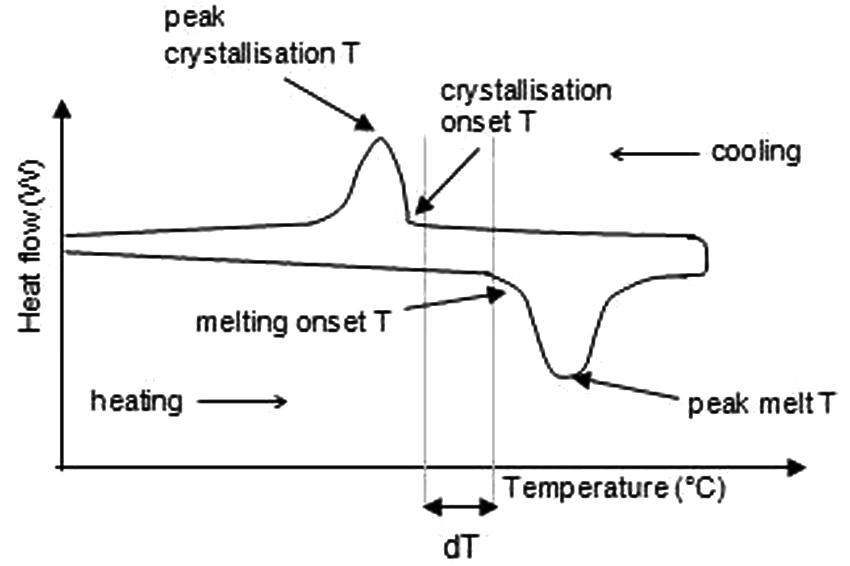
Schematic diagram of a DSC curve.
The range dT is called the ‘process window’– a temperature range over which LS of supercooling polymers is optimum, and warping upon cooling is prevented. This will be different for various polymers; the temperatures at which those transitions occur, and the associated process window, depend on the material. For example, there is a distinction between the process windows of semi-crystalline and amorphous thermoplastics. 6
Methodology
DSC curves were obtained using a DSC Q-200 (TA Instruments, New Castle, USA). The heating range was 25 °C to 220 °C at a heating rate of 10 °C min−1. This range was based on previous DSC tests into the thermal properties of the powder. The nitrogen flow rate was 50 cm3 min−1.
The samples were heated through this range, held isothermal for 1 min and cooled back through the range. Three DSC curves were plotted for each sample. The average sample size was 7 mg of powder, contained in aluminium pans.
An example of a DSC curve plotted is shown in Figure 4, corresponding to R1. The features of interest were the peak melt and peak crystallisation temperatures, and onset melt and onset crystallisation temperatures, in order to determine key thermal properties, such as melting temperature and recrystallisation temperature.
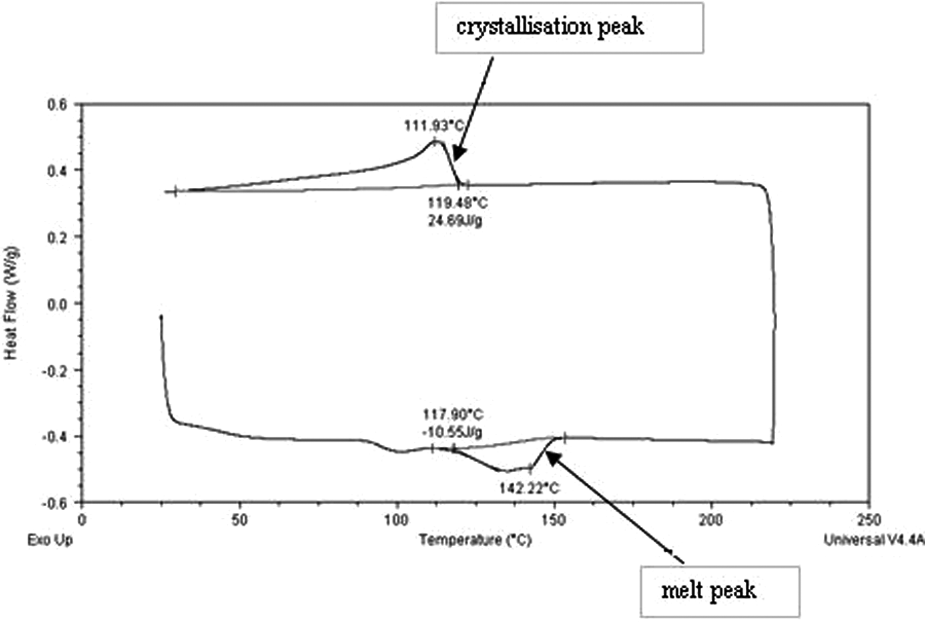
Example DSC curve.
Hot stage microscopy
Having run DSC tests of the material, the temperature range for hot stage microscopy could be selected by reviewing the DSC plots. The temperatures ranged from below the peak melt temperature to above, where the polymer had fully melted.
Methodology
A hot stage microscope was used to study the coalescence of powder particles. Under observation at 50× magnification, the particles were heated from 125 °C to 170 °C, at a rate of 10 °C per minute. This was recorded as a video, and repeated several times for each sample. Once the polymer had melted and begun to flow, the particles resembled the images shown in Figure 5.
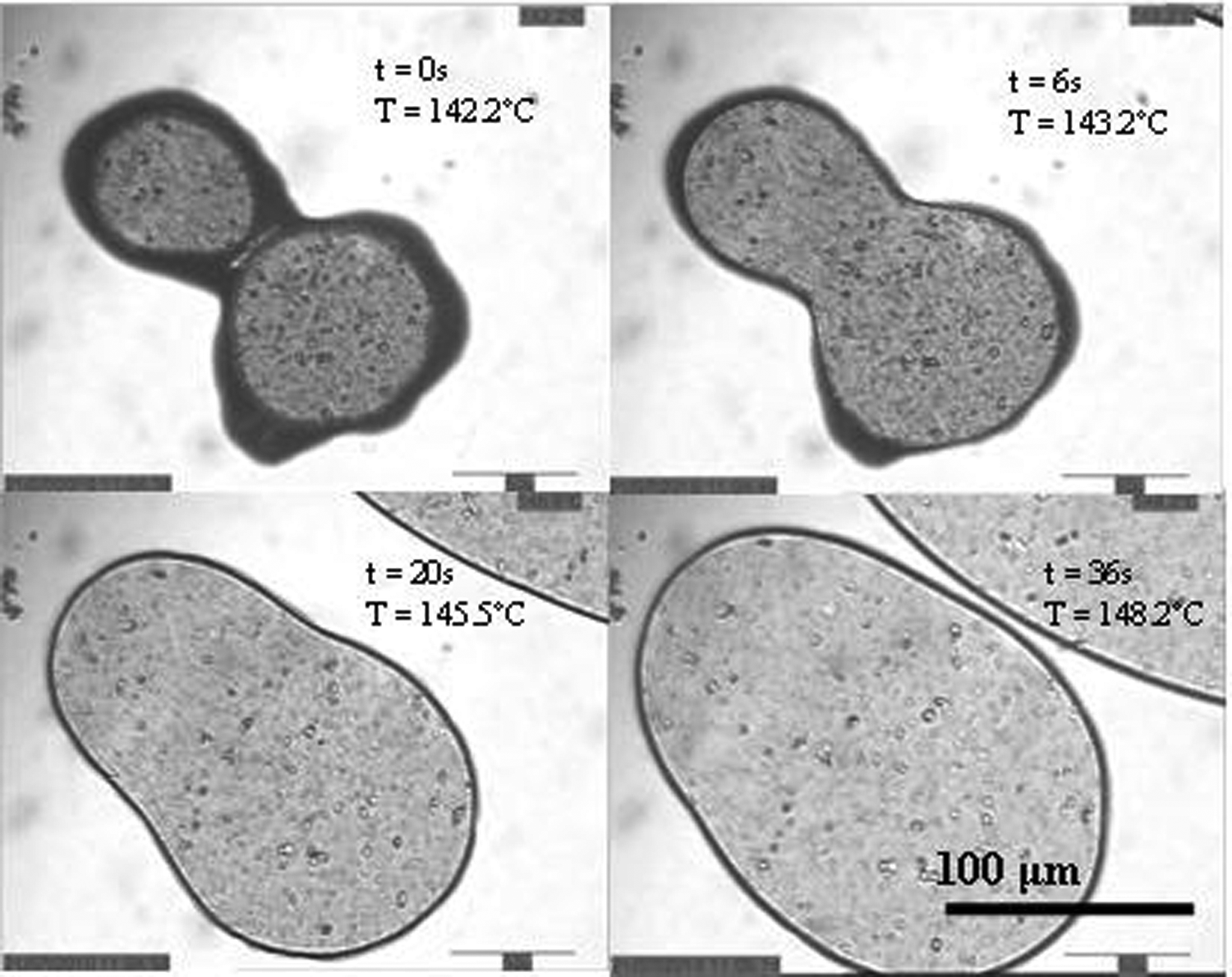
Video micrograph frames showing particle coalescence.
Parallels may be drawn between these micrographs and the DSC plots; as can be seen on the first image, the polymer was able to flow at 142.2 °C, corresponding to a peak temperature on the DSC plot. By the time full coalescence had taken place at 148.2 °C, the peak was levelling out again.
The measurements a1, a2 and y (as shown in Figure 6) were taken at appropriate intervals using imaging software, and used later on in the Frenkel analysis section of this article.
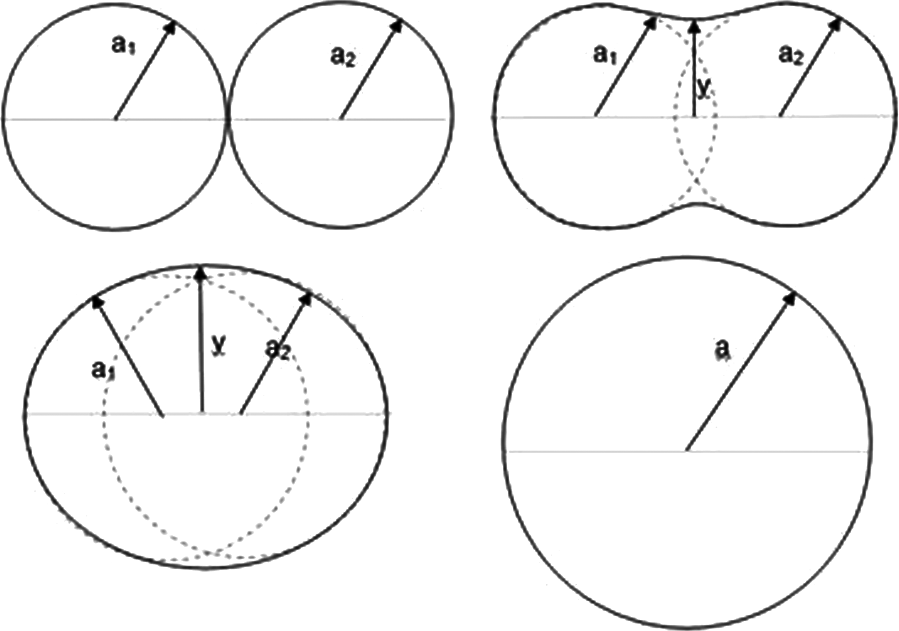
Schematic diagram of particle coalescence.
MFI
In order to investigate any effect of recycling on molecular weight properties, MFI was employed to measure the samples’ viscosity, acting as an indicator of changes in molecular weight.
Methodology
The molecular flow rate (MFR) for each sample was determined using ASTM standard D1238 (the melt flow rate of a thermoplastic material). The units of measurement are grams of material per 10 min (g/10 min), based on the measurement of the mass of material that extrudes from the die over a given period of time. 11
The elastomer was held at 160 °C under a mass of 2.06 kg. A range of parameters were tested and these were identified as being most suitable. The MFR was measured for each sample.
Mechanical properties
The mechanical properties of tensile test specimens made by LS from the different samples of powder (R0 – R3) were tested to determine whether or not recycling has an effect on the tensile properties of the elastomer. Elongation at break, Young’s modulus and tensile strength were measured.
Methodology
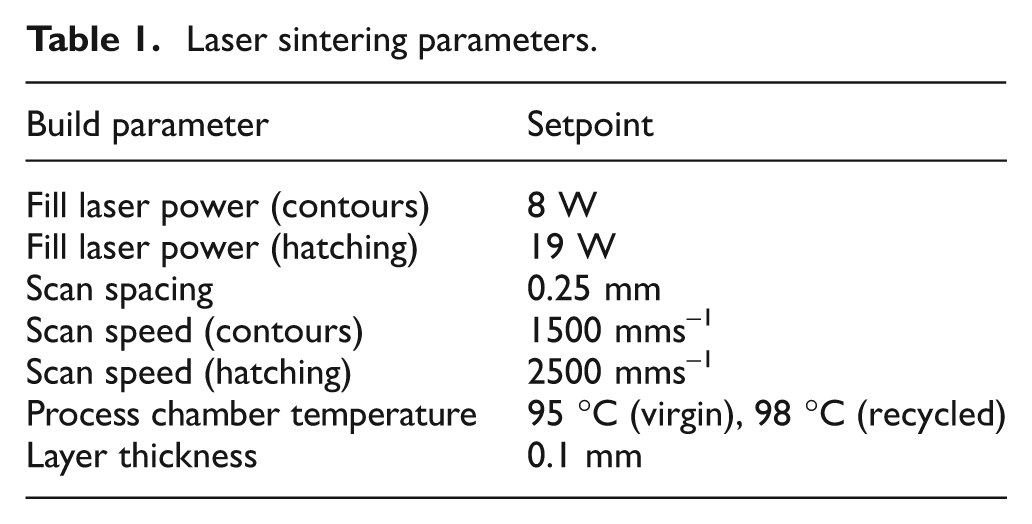
Tensile test specimens were produced by LS on an EOS Formiga P100 system (EOS GmbH, Munich, Germany). Three specimens were produced, in the YX direction and according to British Standard BS EN ISO 527-2:1996, 12 for each level of recycling. Table 1 shows the parameters used for part production.
Laser sintering parameters.
In order to test the tensile properties, a Zwick testing machine (Zwick Testing Machines Ltd., Herefordshire, UK) with long travel contact extensometer was used. The Young’s modulus was measured using a 1 mm/min strain rate between a strain of 0.05% and 0.25%, after which the tensile strength and elongation at break were measured at 5 mm/min.
Results and discussion
Particle size analysis
A histogram was plotted for each sample to show the distribution of particle sizes, and normalised with respect to frequency (number of particles measured) for purpose of comparison (Figure 7).
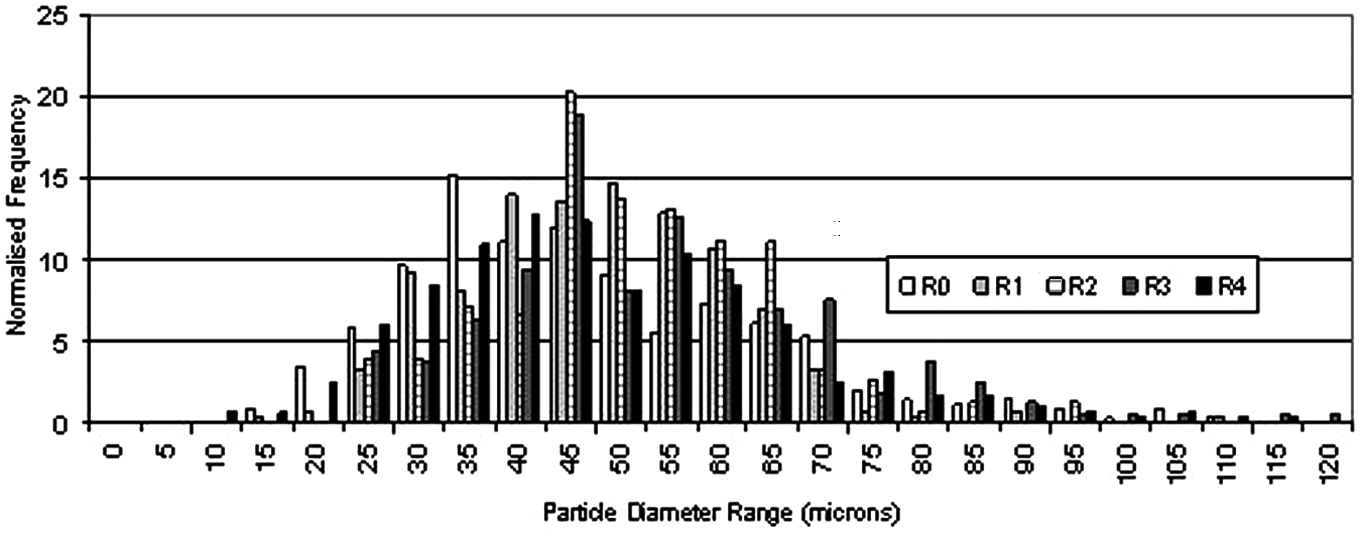
Normalised particle size distribution results for all samples.
Figure 7 shows that there is no apparent change in particle size as a result of recycling. Additionally, there is no evidence of small particles disappearing upon repeated recycling, indicating that solid state sintering of non-laser processed samples does not occur. Figure 8 further confirms that there is no apparent change in average particle size or in particle size distribution.
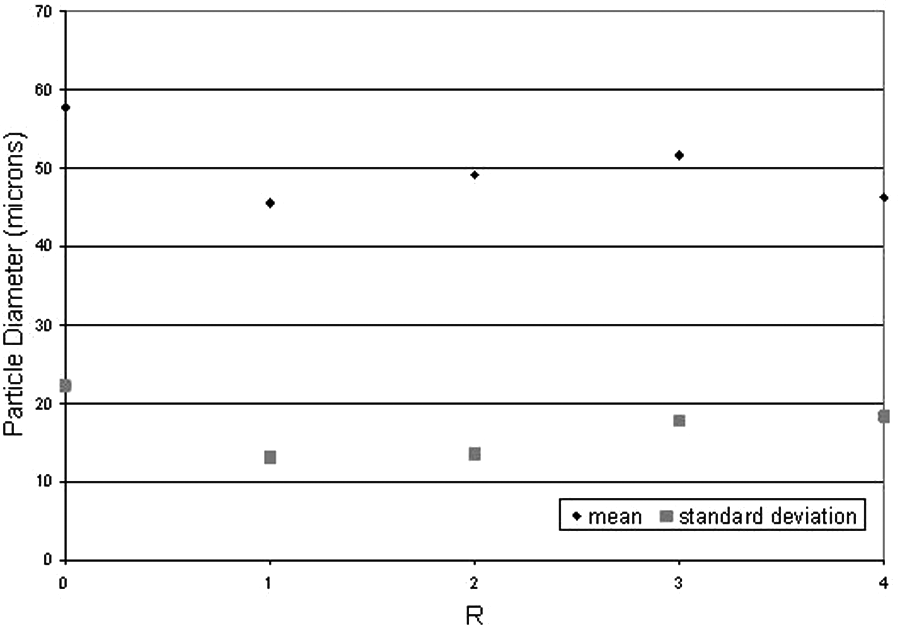
Graph comparing mean and mode particle sizes for all samples.
The mean particle diameter remained between 45 µm and 52 µm with a very small increase but no significant trend caused by increasing R, and the standard deviation remained relatively constant (see Figure 8).
As the bed temperature lay below the melting point of the elastomer, it can be assumed that a very small amount of solid state diffusion took place, if any. Having been heated several times, it would be expected for particle sizes to increase where smaller particles fuse together if solid state diffusion were occurring. This does not appear to take place.
DSC
Figure 9 displays the DSC results for all five samples and demonstrates the proximity of peaks to each other.
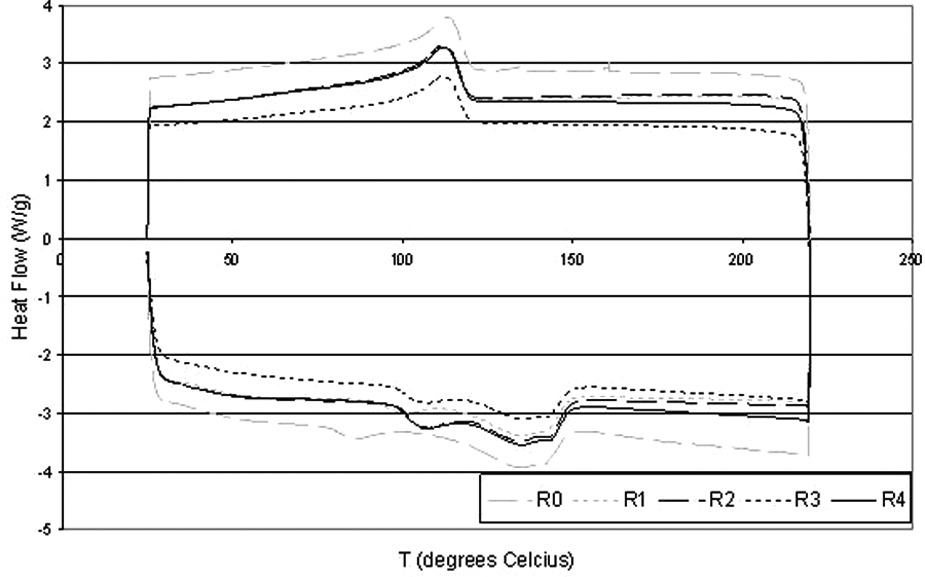
DSC results for all samples.
The process window is of particular interest in the context of this investigation. As this material is an elastomer, the melting temperature range is large. Consequently, the melt peak for each sample was very broad, leading to overlap of the melt and crystallisation onset temperatures in each case (and a negative process window).
In order to determine differences between samples more clearly, the process window was taken to be the difference between melt peak and crystallisation peak temperatures: instead of using melt/recrystallisation onset temperatures, the peak temperatures have been presented. These results are plotted as a function of times recycled in Figure 10.
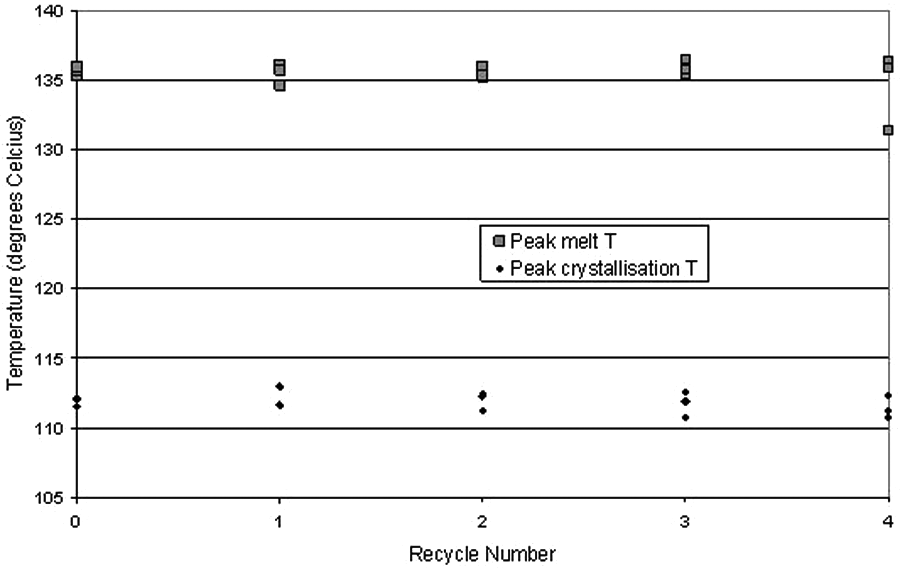
Graph of process window results for all samples.
Figure 10 shows that there was no obvious change to peak melt temperature or peak recrystallisation temperature, and thus there was no change to the process window as a consequence of recycling. The results were repeatable, giving convincing evidence that recycling the elastomer powder has little effect on its thermal properties. Changes in melting and recrystallisation temperatures would have been significant as the energy required to sinter material would have been affected. These results enable easier processing, as with a constant process window, LS parameters do not have to be altered before every build.
Hot stage microscopy
Initial relationships between y/a and time suggested the polymer followed the Frenkel model, but the elastomer particles did not always start at the same distance from each other on the slide, so did not exactly fit the stages shown in Figure 6. Consequently, in these cases the y/a ratio went above 1, where the particles coalesced into an ellipse rather than a circle. When this happened the measurements were cut off at 1.
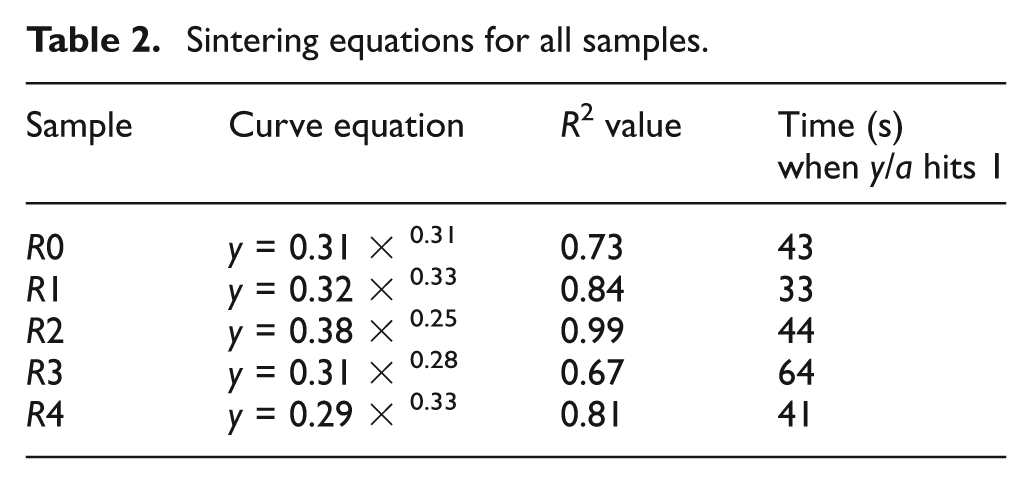
From these measurements, graphs of y/a against t were plotted for each sample. All the curves for each R-value were combined on one graph (see Figure 11), giving an average necking relationship for the sample (equations shown in Table 2). This enabled the time taken for the y/a ratio to reach 1 to be found, giving a rough comparison of sintering time between the samples. The graph of sintering time against R (Figure 12) shows wide scatter, but indicates that recycling has no significant effect on sintering time.
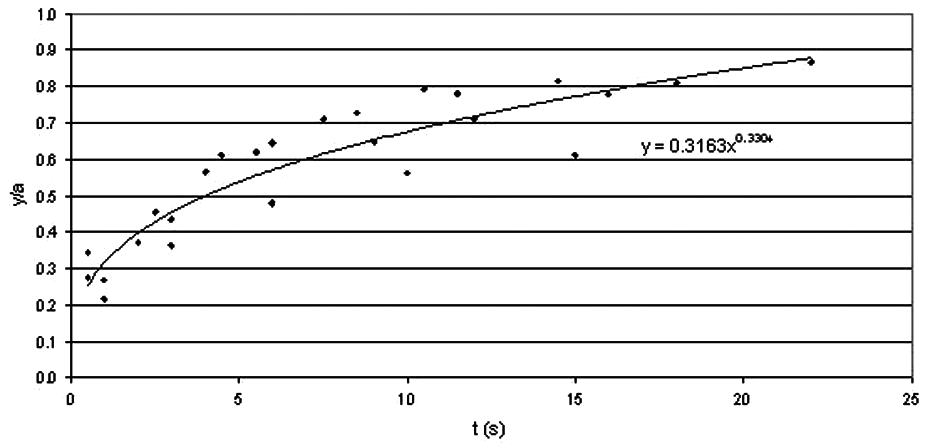
Graph of average sintering rate for R1.
Sintering equations for all samples.
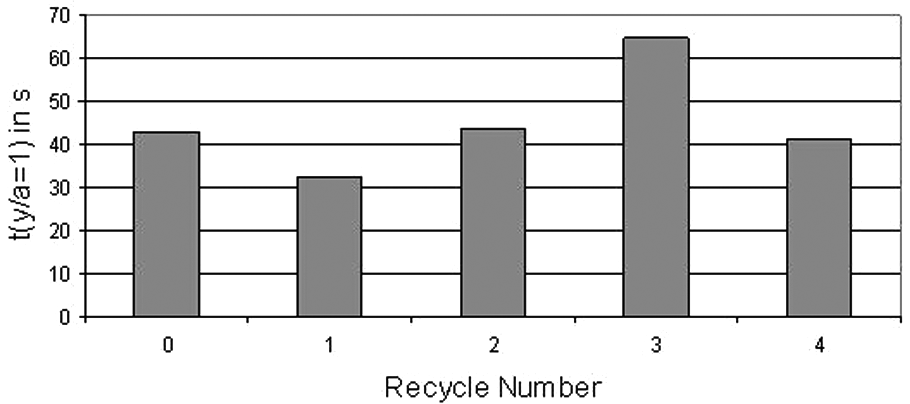
Graph comparing time to sintering completion (y/a = 1) of all samples.
Further work may show the time for particle coalescence during LS would not vary with R. Hot stage microscopy is excellent as an initial approximation but is not accurate enough for detailed analysis. Additionally, the parameters are orders of magnitude smaller than those involved in LS (during which, heating happens in microseconds) so the temperature and heating rate would have to be scaled-up dramatically to model the processes occurring during LS. This was beyond the scope of this research.
Melt flow rate
Figure 13 displays the flow rate values for each sample. The results suggest a small overall increase in flow rate with increasing R, corresponding to a drop in viscosity. This is contrary to predictions based on nylon-12, where viscosity increased on recycling owing to chain growth and an associated increase in molecular weight by a polycondensation reaction. Seemingly this effect did not occur during recycling of the elastomer, or if it did, there was a simultaneous reaction occurring, acting to reduce chain length.
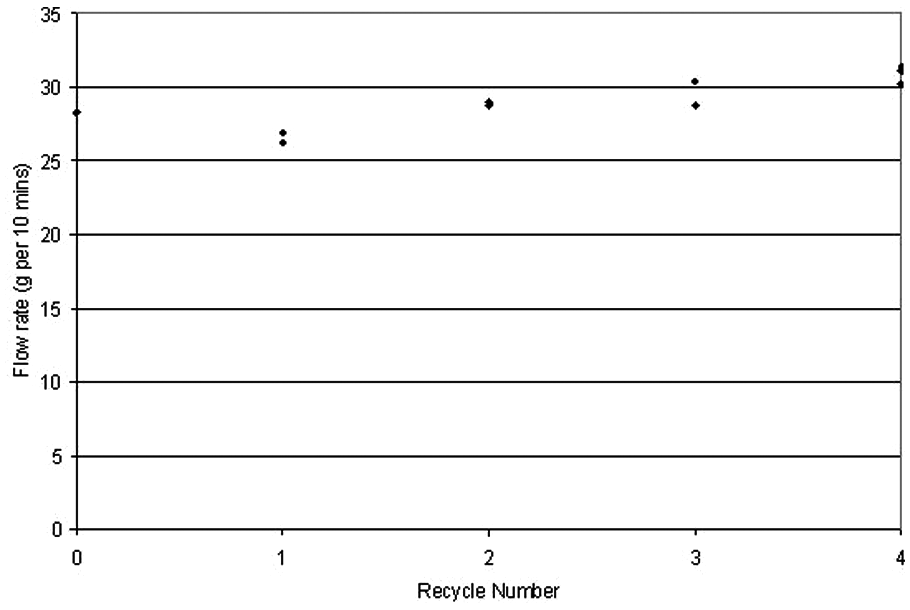
Graph comparing flow rate for all samples.
This decreasing viscosity would not be expected to have a significant effect on the sintering properties of the elastomer – as outlined in Frenkel’s equation, sintering rate increases with decreasing viscosity – though shorter chain length may imply slightly poorer mechanical properties.
Mechanical properties
Figures 14, 15, 16 and 17 show the results of the tensile tests. Figure 15 shows elongation at break values ranging from 104% to 160% with an average of 137%. These results are consistent with, and in many cases higher than, other reported values for LS elastomers. However, reported values for injection moulded Ellastollan are considerably higher, in the range 420% to 700% (S-series). 7 The elongation at break results show no clear trend with recycle number. This implies constant ductility on recycling.
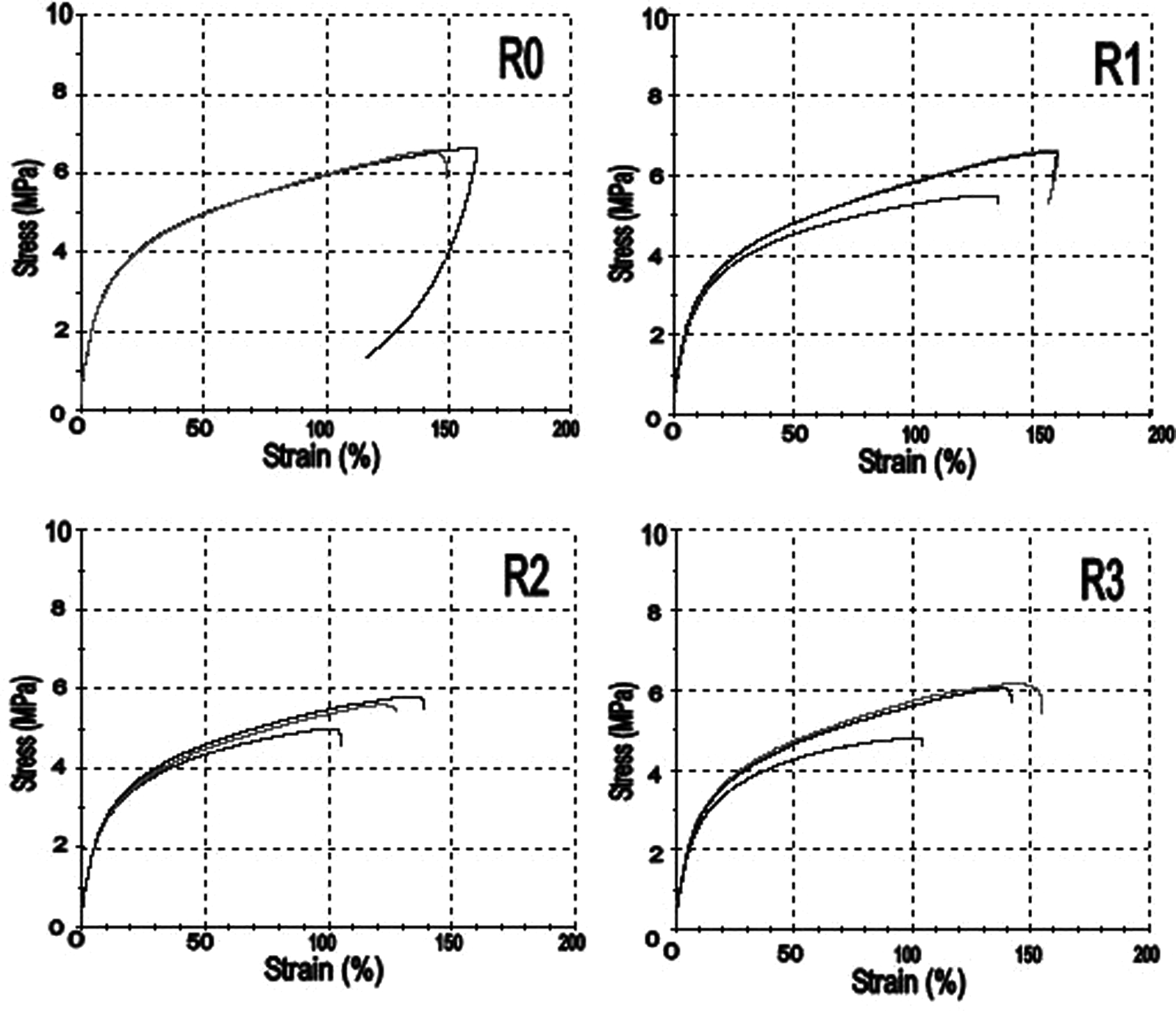
Tensile test graphs for all stages of recycling.
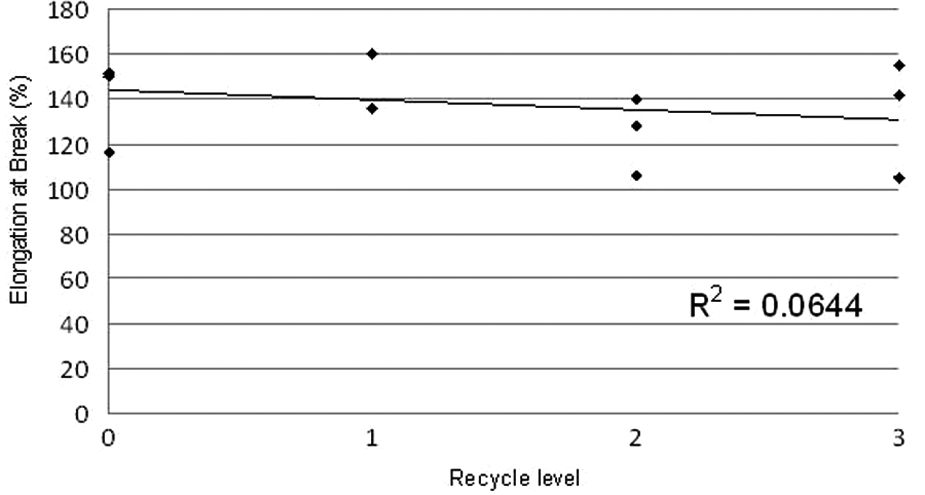
Graph of elongation at break for all samples.
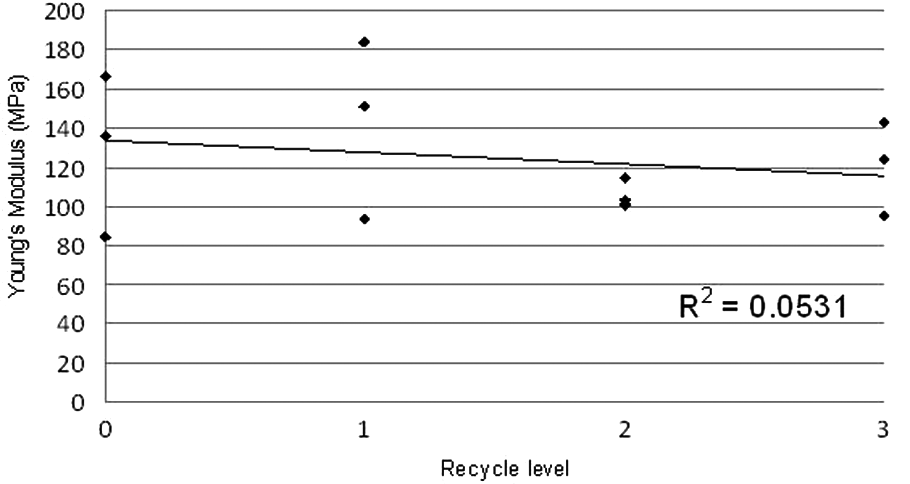
Graph of Young’s modulus for all samples.
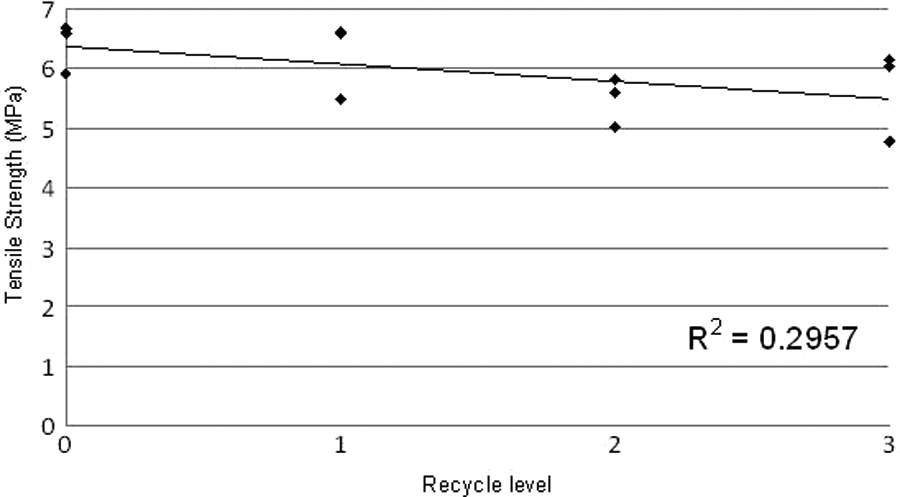
Graph of tensile strength for all samples.
Figure 16 shows that Young’s modulus ranged from 84 MPa to 183 MPa with an average of 125 MPa. As expected, these results are considerably lower than those for LS nylons (which can be as high as 2.3 GPa1 3 ) but they are comparable with values reported for other LS elastomers. As with the elongation at break data, the Young’s modulus results show no clear trend as a consequence of recycling. This implies constant stiffness on recycling.
Figure 17 shows tensile strength values ranging from 4.8 MPa to 6.7 MPa with an average of 5.9 MPa. These results are comparable with values from other LS elastomers but considerably lower than reported results for injection moulded Elastollan, which range from 27 MPa to 43 MPa (S-series). 7 The values for tensile strength indicate a slight reduction as a consequence of repeated recycling, although the spread of results is large compared with changes in averages. This graph shows a small decrease in tensile strength on recycling.
Figures 14 to 17 suggest a slight overall decrease in mechanical properties, but the amount of variation is relatively small. The regression coefficient (R2) value is negligible in all cases apart from Figure 17, tensile strength, where the value is small (0.2957). Therefore, it is not possible to conclude that recycling does cause a reduction in properties. The results for tensile strength show a possible influence of recycling on properties. The MFR results showed a slight increase in MFR that might be explained by a reduction in molecular weight of the polymer owing to thermal cycling. If this has occurred, it could cause a slight reduction in tensile strength as has been observed. However, a reduction in molecular weight would also be expected to result in a reduction in elongation at break, but this was not observed.
Conclusions
DSC analysis of the elastomer samples has shown no effect of recycling on the thermal properties of the powder. This reflects upon the build parameters: there was no large shift in melting temperature so machine temperatures may remain constant from one build to the next, removing the need for the adjustment of parameters.
Hot stage analysis has shown that recycling had no obvious effect on sintering time, contrary to the small overall increase in flow rate with increased recycling shown in MFI analysis results, corresponding to a slight drop in viscosity. These MFI results suggested reduced chain length, and this corresponds to the minor decrease in strength in the outcome of tensile test results, as does the negligible decrease for the elongation at break and Young’s modulus.
It has also been identified that there is no significant trend in the elastomer particle size caused by increased recycling, suggesting the absence of solid state sintering. The implications of this result are constant spreading and layer deposition, and as a result there is no need to adjust layer thickness between builds. Maintenance during the sintering process is simpler and it is easier to remove powdered material from parts during part breakout.
The results have shown that it is possible to recycle the elastomer bed powder several times through the LS machine without any significant degradation or reduction in properties, an encouraging result for the elastomer in the upcoming future of LS. This is particularly important once the cost of raw materials is taken into account, and is important from an environmental stand-point.
Footnotes
Appendix
Acknowledgements
Professor Mike Caine, Andrew Woolley, Ray Owens, Dr David Hitt and Dr David Ross, Loughborough University and the ENSURE scholarship scheme.
Funding
The project was funded by ENSURE scholarships: Student Undergraduate Research Experience.