Abstract
Epoxy resin/cyanate ester copolymer (EC) possesses the advantages of low dielectric constant and low dielectric loss, while the easy flammability of EC restricts its applications. The effect of 10-(2,5-dihydroxyphenyl)-10-hydrogen-9-oxa-10-phosphazephenanthrene-10-oxide (DQ) and copper hydroxystannate (CuHS) on the flame retardancy, smoke suppression, mechanical properties and thermal decomposition behavior of EC was studied. The EC composite containing 7 wt% DQ and 3 wt% CuHS (EC/7DQ3CuHS) passed the UL-94 V0 rating with a limiting oxygen index of 32.0% The peak heat release rate and total heat release of EC/7DQ3CuHS fell by 41.9% and 35.0% respectively, compared with those of pure EC. The total smoke release of EC/7DQ3CuHS was reduced by 36.0% in comparison with that of the EC composite containing 10 wt% DQ. The tensile and impact strengths of the EC composite with 9 wt% DQ and 1 wt% CuHS increased by 16.9% and 19.2%, respectively in contrast with pure EC. The dielectric constant and dielectric loss of the EC composites maintained at relative low values. The as-prepared EC composites have application prospect in electronic packaging industry.
Keywords
Introduction
Epoxy resin (EP) has been widely used in industry and daily life due to its good chemical stability, interfacial adhesion, and mechanical properties.1–3 As a packaging material applied in the occasion of high-frequency electronic devices (i.e. 5G communication equipment), 4 it is challenging for EP to meet the requirements of good flame retardancy, high heat resistance and low dielectric properties.
Cyanate ester (CE) resin contains two or more O-C≡N groups in the main chain, which consists of networks with highly symmetric triazine ring upon curing,5,6 thus shows good mechanical strengths, thermal stability and low dielectric properties.7,8 Therefore, it is promising to integrate the advantages of both thermoset by the reaction between the epoxy groups of EP and the -C≡N groups of CE.9,10 Nevertheless, the flammability of epoxy resin/cyanate ester copolymer (EC) restricts its applications.11,12 It is important to endow EC with flame retardancy while maintaining good mechanical and low dielectric performances as well.
According to the existed literatures, 9,10-dihydro-9-oxa-10-phosphaphenantrene-10-oxide (DOPO) as an effective reactive flame retardant for EP, also exhibited high flame retardant performance in EC. 13 Wang et al. 14 found that the pHRR and THR of the EC composites with DOPO were efficiently reduced while the glass transition temperature (Tg) values of the EC composites declined. Ho et.al 15 synthesized 2-(6-oxido-6H-dibenz(c,e) (1,2)-oxaphosphorin-6-yl)-1,4-benzenediol (DOPO-BQ) and found the EC composite containing 10 wt% DOPO-BQ reached the V0 classification in the UL-94 tests. In our recent publication, 16 we found 10-(2,5-dihydroxyphenyl)-10-hydrogen-9-oxa-10-phosphorophenanthrene-10-oxide (DQ), had as good flame-retardant effect as DOPO in EC and hardly damaged the Tg of the EC composites, whereas the smoke emission of EC significantly increased upon burning. Cao et al. 17 synthesized a copper phenyl phosphate (CuPP) and found that the EC composite containing 5 wt% CuPP achieved V1 rating with a LOI of 30.6% and a high Tg of 221°C, while the smoke emission maintained same level with pure EC. Therefore, the smoke suppression became another concern of EC composites, along with the flame retardancy and the dielectric properties.
In our previous report, copper hydroxystannate (CuHS) smoke suppressant was found to be efficient on the smoke suppression of EP and hardly affected the dielectric properties. 18 Herein, the effects of CuHS and DQ on the fire safety, smoke suppression, mechanical performance and thermal decomposition of EC copolymer were studied in this work.
Experimental
Materials
10-(2,5-dihydroxyphenyl)-10-hydrogen-9-oxa-10 phosphophenanthrene-10 oxide (DQ) was provided by Zhengzhou Alpha Chemical Co., Ltd. Sodium stannate trihydrate (Na2SnO3·3H2O) and copper sulfate pentahydrate (CuSO4·5H2O) were purchased from Sinopharm Chemical Reagents Co., Ltd. Bisphenol A epoxy resin E-51 (epoxy equivalent 195 g ep−1, epoxy value: 0.51) was provided by Shanghai Resin Co., Ltd, and bisphenol A cyanate ester resin (CE, BADCY) was obtained from Wuqiao Resin Company in Jiangsu, China.
Preparation of copper hydroxystannate
Copper hydroxystannate (CuHS) was synthesized by an approach mentioned in our previous publication.18,19 Specifically, 4.8 mmol CuSO4·5H2O was first dissolved with 200 ml deionized H2O in a three-necked flask, and then the solution of Na2SnO3·3H2O (4.8 mmol) in 200 ml deionized H2O was added dropwise to the flask under mechanical stirring. The above solution was reacted at room temperature for 20 h. Afterwards, the products were collected by centrifugation, then washed with deionized H2O and ethanol for more than 3 times. Finally, the products were vacuum dried in an oven under 105°C for 3 days, then ground into powder.
Preparation of ester copolymer composite
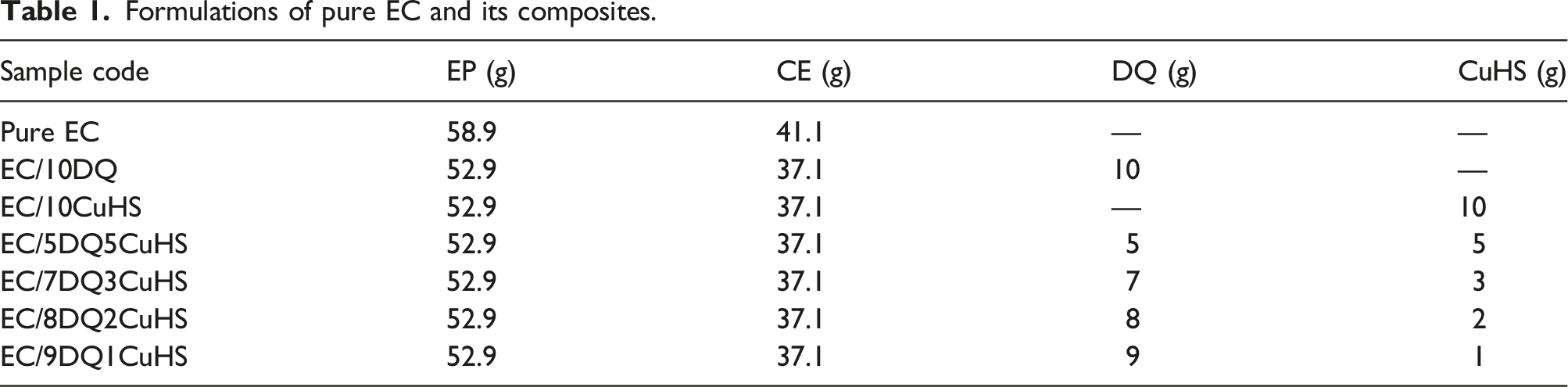
Formulations of pure EC and its composites.
Characterization and measurements
Fourier transform infrared spectroscopy (FTIR) was adopted to characterize the functional groups of the flame retardant and the char after cone calorimetric tests (CCT). The KBr disc samples were scanned on Bruker Hyperion 2000 (Germany) type FTIR apparatus in the range of wavenumber from 400 cm−1 to 4000 cm−1 with the resolution of 1 cm−1.
X-ray Diffractometer (XRD) was used to check the crystallization state of the flame retardant and the char after CCT. The scan was ranged from 5° to 80° at the rate of 5°/min with Cu Kα radiation on a DX-1000 (Dandong, China) type XRD device.
Scanning electronic microscopy (SEM) was utilized to observe the fracture surface of the EC composites and the morphology of the char after CCT. All the samples coated with gold were observed on a Hitachi S-4800 (Japan) type SEM apparatus.
Raman spectra was employed to characterize the graphite extent of the char after CCT. The measurements were conducted on a LabRAM HR Evolution spectrometer under argon laser at 532 nm wavelength with the samples of char powder.
The thermal decomposition behaviors of the EC composites were studied by thermogravimetric analysis (TGA) from 25°C to 800°C with 10°C/min heating under N2. Each sample less than 5 mg was placed into an Al2O3 crucible and tested on a STD Q600 (TA instruments, America) type device.
The glass transition temperatures of the EC composites were measured by Differential Scanning Calorimetry (DSC) analysis on a Q100 (TA instruments, America) type calorimeter from 50°C to 300°C at a heating rate of 10°C/min under N2.
Limiting oxygen index (LOI) was used to evaluate the flammability of the EC composites according to ISO 4589-2:2017 on an HC-2 type device with the specimen size of 100 × 6.5 × 3 mm3.
The UL-94 vertical burning test was adopted to monitor the flammability of the EC composites, and the tests were performed on a CFZ-3 type device according to IEC 60695-11-10:2013 with the sample dimension of 130 × 13 × 3 mm3.
Cone calorimetric test (CCT) was used to analyze the fire parameters of the EC composites upon burning according to ISO 5660. The test was performed on FTT, dual type CCT device with the heat flux of 35 kW/m2 and the specimen dimension of 100 × 100 × 3 mm3.
The decomposition products of the EC composites in the gas-phase were analyzed by TG coupled FTIR (TG-FTIR). The test was conducted on a TG209F3/TENSOR 27 type device with the sample mass of 10 mg exactly for comparison. The heating rate was 10°C/min started from 25°C to 800°C.
The tensile tests of the EC composites were performed on an A DXLL-5000 universal machine according to ISO 844:2004 with the sample size of 80 × 10 × 4 mm3. Each sample was tested for measurements at least for the average.
The impact tests of the EC composites were conducted on a GT-7045-MDL digital machine based on ISO 1209-2:2004 with the pendulum energy of 1 J, and the sample size were 80 × 10 × 4 mm3. Each sample was tested for five measurements at least for the average.
Dielectric properties of the EC composites were tested under the frequency of 1 GHz with the sample size of 25 mm diameter and 3 mm thickness. The measurements were performed on an E4991B Impedance Analyzer (KEYSIGHT).
Results and discussion
Fire behavior of ester copolymer composites
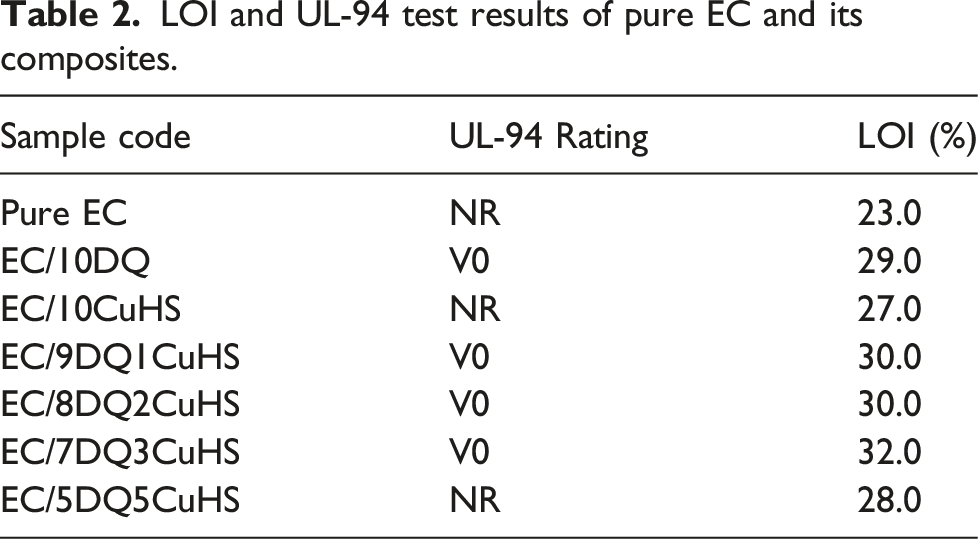
LOI and UL-94 test results of pure EC and its composites.
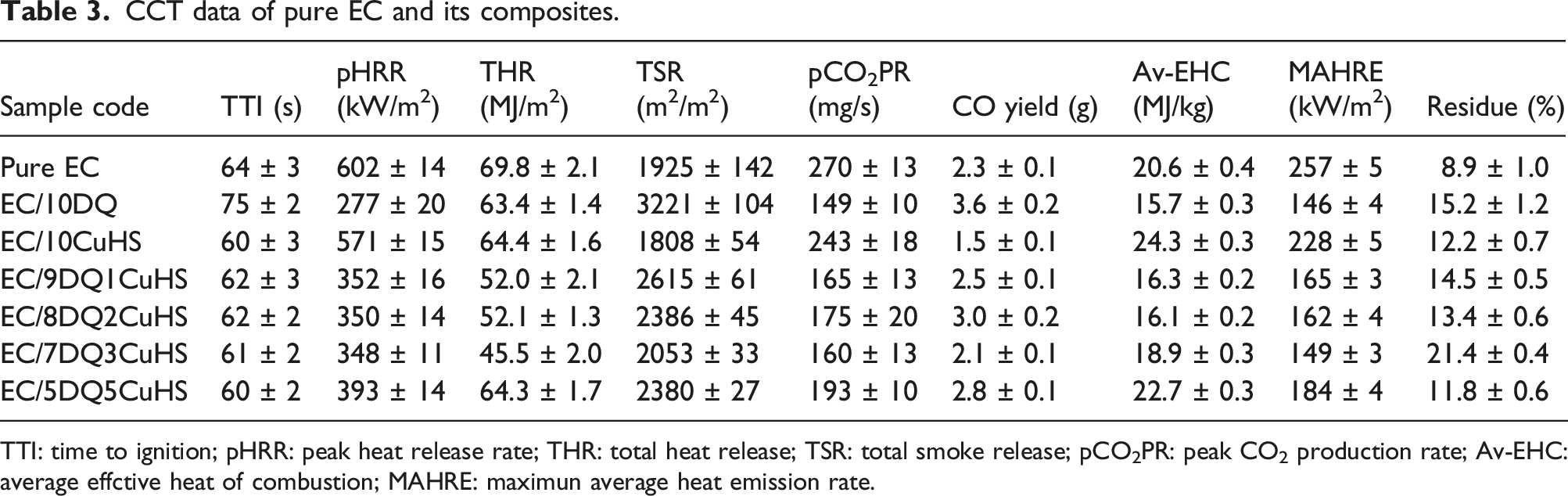
CCT was utilized to study the heat, smoke, and gas release during the burning of EC composites. Figure 1(a)–(b) showed the heat release rate (HRR) and total heat release (THR) curves of pure EC and its composites with the relevant data shown in Table 3. The peak HRR (pHRR) of pure EC was 602 kW/m2. The pHRR of EC/10DQ was reduced to 277 kW/m2, indicating a decrease of 54% compared to pure EC. The pHRR of EC/10CuHS decreased to 571 kW/m2, indicating that CuHS had limited effect on reducing the heat release of EC. The pHRR of EC composites containing both DQ and CuHS was located between that of EC/10DQ and EC/10CuHS. Specifically, the pHRR values of EC/9DQ1CuHS, EC/8DQ2CuHS and EC/7DQ3CuHS were all around 350 kW/m2, while the pHRR of EC/5DQ5CuHS was 393 kW/m2. The THR of pure EC was 69.8 MJ/m2. The addition of 10 wt% DQ or 10 wt% CuHS slightly reduced the THR of EC to 63.4 MJ/m2 and 64.4 MJ/m2, respectively. However, the THR values of EC/9DQ1CuHS, EC/8DQ2CuHS and EC/7DQ3CuHS were significantly lower than those of EC/10DQ and EC/10CuHS. Specifically, the THR of EC/7DQ3CuHS decreased to 45.5 MJ/m2, which was 35% lower than that of pure EC, indicating good synergistic effect between DQ and CuHS on inhibiting the THR of EC composites. The total smoke release (TSR) curves of the EC composites are shown in Figure 1(c). The TSR of pure EC was 1925 m2/m2. The addition of 10 wt% DQ increased the TSR of EC to 3221 m2/m2, while 10 wt% CuHS reduced the TSR to 1808 m2/m2. Moreover, the TSR of EC/7DQ3CuHS was 2053 m2/m2 and decreased by 36% compared to EC/10DQ, which suggested that an appropriate amount of CuHS effectively suppressed smoke generation caused by DQ in EC composites. Furthermore, the peak CO2 production rate (pCO2PR) of EC/7DQ3CuHS decreased by 41% compared to that of pure EC. Above all, it was found that EC/7DQ3CuHS showed the best comprehensive flame retardant and smoke suppressive performance. CO yield is assessed to reflect the toxicity gas produced during the incomplete burning of EC. Figure 1(d) presented the CO yield curves versus time. EC/7DQ3CuHS showed a 41.7% decreased CO yield compared with EC/10DQ. Maximum average heat emission rate (MAHRE) reflects the hazard of real fire developing, listed as Table 3. The MAHRE of pure EC was 257 kW/m2. The presence of DQ or CuHS both lowered the MAHRE, while DQ was more efficient than CuHS. Moreover, the MAHRE of EC/7DQ3CuHS was significantly decreased by 42.0% compared with pure EC. For assessing the flame-retardant action of DQ and CuHS in EC, the char residue after burning and the average EHC were focused. It was seen from Figure 4f and Table 3 that the residue of EC/7DQ3CuHS was increased to 21.4 wt% from the 8.9 wt% of pure EC, which was due to the generation of protective char. EHC, defined as HRR divided by mass loss rate (MLR). A lower EHC represents the incomplete combustion of the sample, which suggests the flame inhibition in the gas phase. However, a high EHC indicates the flame retardant mode in the condensed phase including charring effect.
20
From Table 3, DQ alone reduced the averaged EHC (av-EHC), while CuHS increased the av-EHC, which means that the metal oxide formed from CuHS promoted the formation of strengthened char of EC. CCT results of pure EC and its composites, (a) HRR, (b) THR, (c) TSR, (d) CO yield, (e) CO2PR, (f) mass loss. CCT data of pure EC and its composites. TTI: time to ignition; pHRR: peak heat release rate; THR: total heat release; TSR: total smoke release; pCO2PR: peak CO2 production rate; Av-EHC: average effctive heat of combustion; MAHRE: maximun average heat emission rate.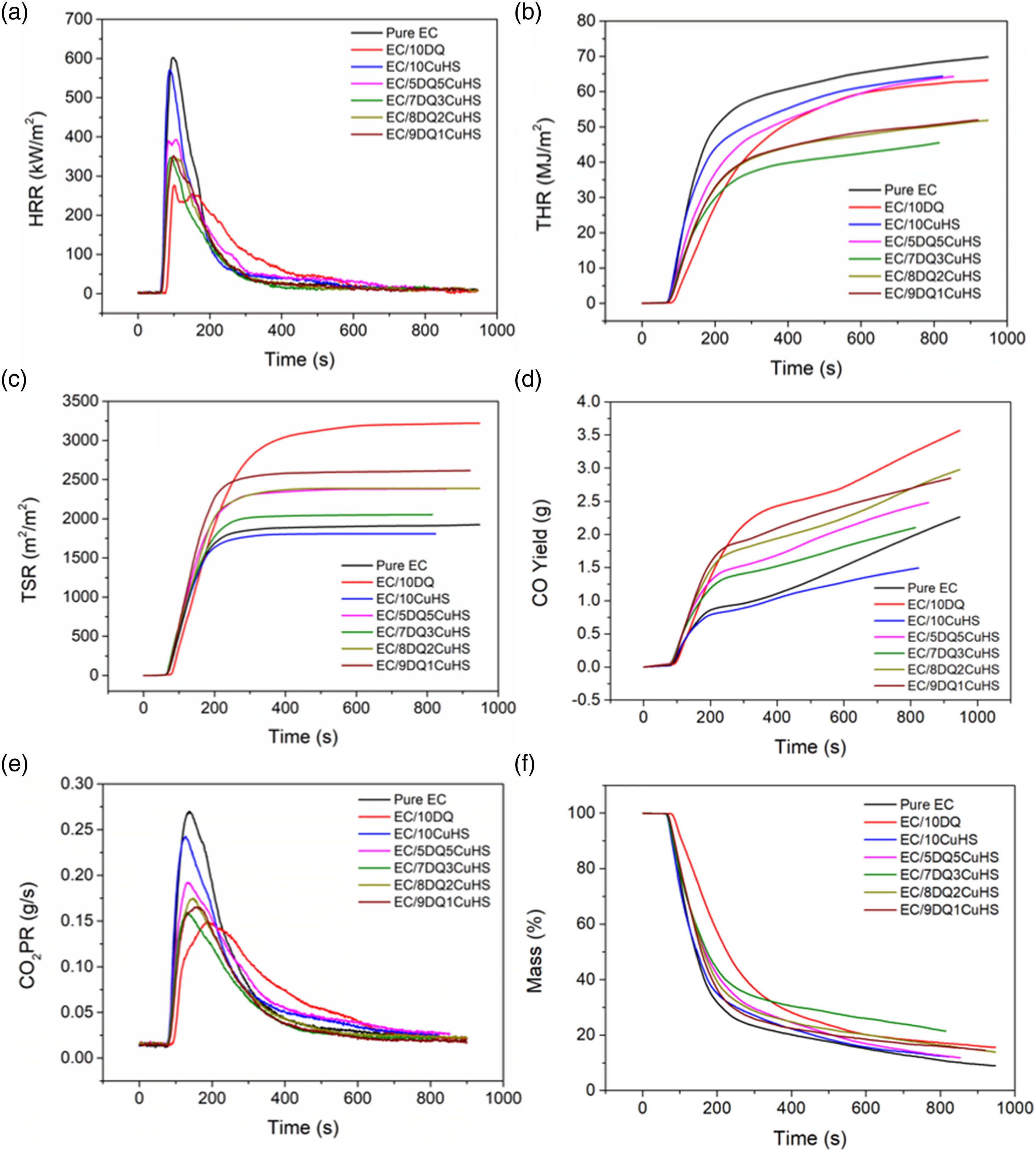
Char analysis
The photos with SEM images of the residual char of EC and its composites after CCT are shown in Figure 2. It was seen from Figure 2(a) that little char residue was left after the combustion of pure EC. The SEM image showed the surface morphology of the residual char was loose with many cracks and pores, indicating that pure EC was almost burnt out. From Figure 2(b), it was observed that the surface of the residual char of EC/10DQ became denser. From the SEM image, the surface morphology was continuous without obvious cracks while there was ash on the surface, indicating that DQ acted both in the gas phase to quench the active radical
21
and in the condensed phase to promote charring. For EC containing 10 wt% CuHS alone (Figure 2(c)), it was found that the surface integrity of the residual char was improved compared to pure EC. However, SEM image of EC/10CuHS showed a fluffy and loose surface, with several pores distributed. For the residual char of EC/9DQ1CuHS (Figure 2(d)), the amount of the char was significantly higher than that of EC/10DQ and EC/10CuHS. The SEM image showed many pores on the surface, indicating that replacing DQ with 1 wt% CuHS hardly improved the quality of the char significantly. From Figure 2(e), it was seen that the amount of the char of EC/8DQ2CuHS was close to that of EC/9DQ1CuHS. From the SEM image, the quality of the char layer of EC/8DQ2CuHS was still relatively loose. From Figure 2(f), it was observed that the residual char volume of EC/7DQ3CuHS was the largest, with a height of nearly 4 cm. In the SEM image, the surface of the residual char was the densest, indicating that the appropriate addition of CuHS promoted the carbonization in the condensed phase during the EC combustion.
22
For EC/5DQ5CuHS, the quality of residual char was also relatively dense, while a few pores were observed on the surface, indicating that it was less conducive to form thick and dense char when the DQ content was low. SEM images of residual char of pure EC and its composites, (a, a’) pure EC, (b, b’) EC/10DQ, (c, c’) EC/10CuHS, (d, d’) EC/9DQ1CuHS, (e, e’) EC/8DQ2CuHS, (f, f’) EC/7DQ3CuHS, (g, g’) EC/5DQ5CuHS.
The graphitization degree of residual char after CCT of EC composites was investigated by Raman spectroscopy. The integrated area of the D bands divided by G bands (ID/IG) under Raman spectroscopy reflects the graphitization degree of the char.
23
A lower ID/IG value represents a higher graphitization degree.
24
As shown in Figure 3, the ID/IG value of pure EC was 3.8, and the ID/IG of EC/10DQ and EC/10CuHS were 3.49 and 3.54. The order of ID/IG values for the EC composites with both DQ and CuHS was as follows: EC/5DQ5CuHS (ID/IG = 3.23) > EC/9DQ1CuHS (ID/IG = 3.18) > EC/8DQ2CuHS (ID/IG=3.09) > EC/7DQ3CuHS (ID/IG = 2.81). The above results indicated that the char of EC/7DQ3CuHS possessed the highest of graphitization degree, and thus the best quality of the char layer formed after CCT, which effectively acted as a physical barrier to hinder the mass and heat transfer between the gas phase and the condensed phase,
25
therefore achieving the lowest pHRR and TSR in the CCT results for EC/7DQ3CuHS. (a-g) Raman spectra of the char of pure EC and its composites.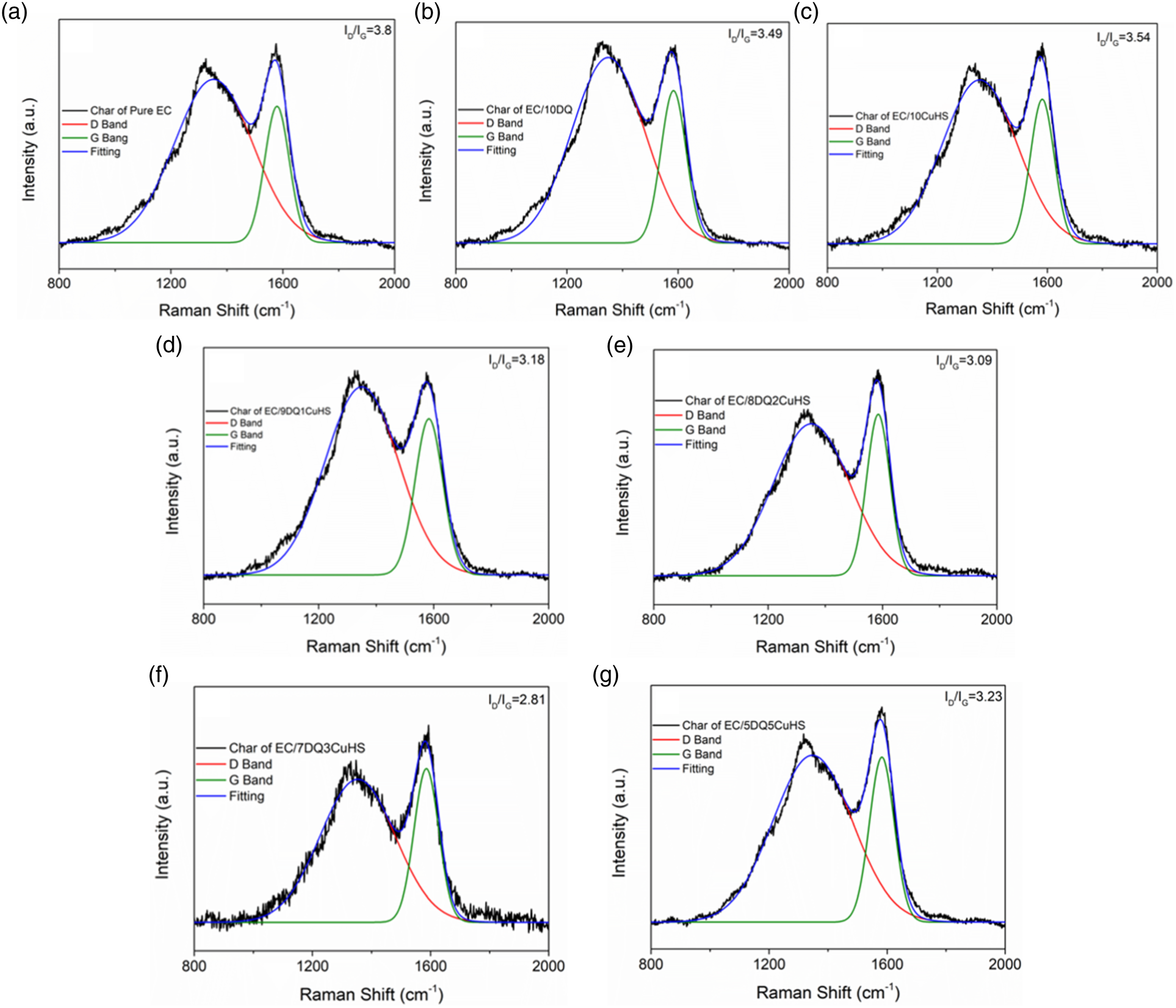
The composition of the residual char of EC composites after CCT was further analyzed by FTIR, as shown in Figure 4(a). It was seen that the char of pure EC only showed two absorption peaks located at 1585 cm−1 and 1228 cm−1 corresponding to C = C and C-C stretching vibrations,
26
respectively. From the spectrum of the char of EC/10DQ, almost no additional absorption peaks emerged, which indicated that DQ mainly played a role in the gas phase.
27
For EC/7DQ3CuHS, the absorption peak at 1105 cm−1 belonged to Sn-O bending vibration,
19
and the broad absorption peak around 638 cm−1 corresponded to the stretching vibration of Cu-O,
28
which were similar to the peaks appeared in EC/10CuHS. (a) FTIR and (b) XRD results of the char of pure EC and its composites.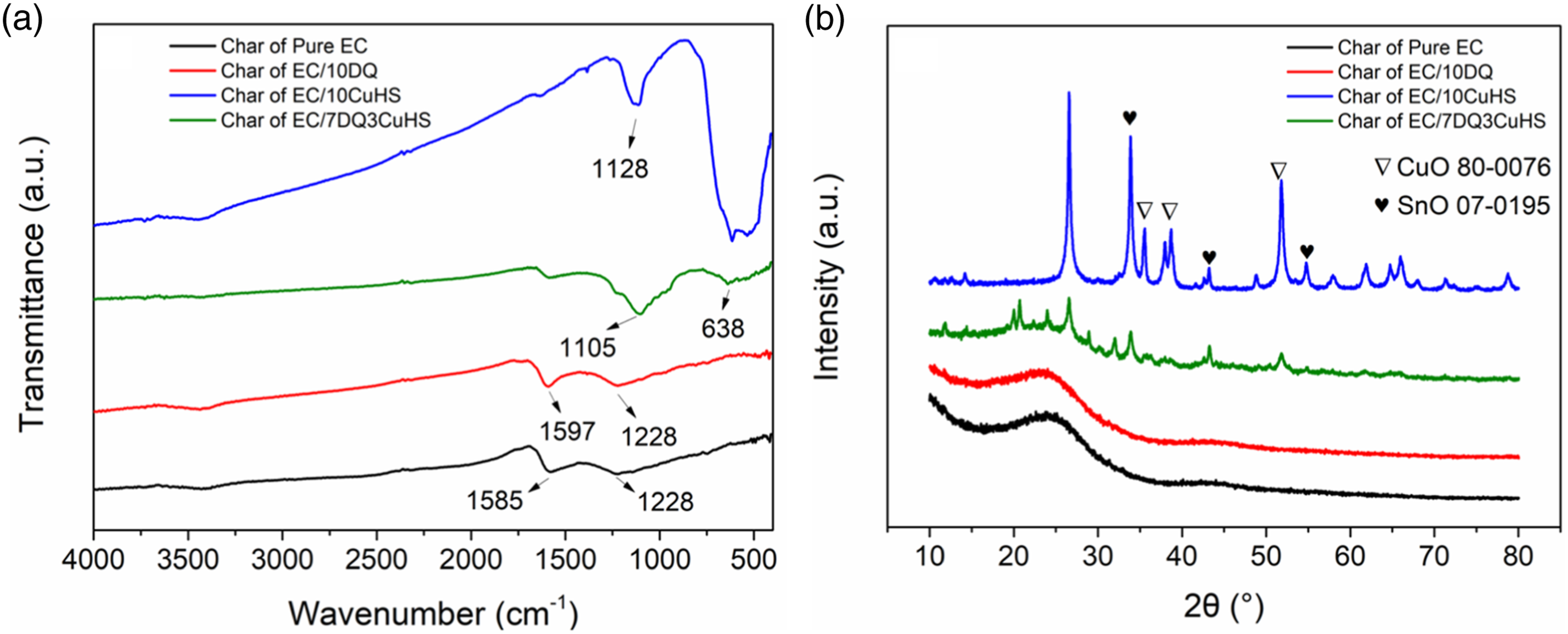
In order to further confirm the presence of elements in the residual char, XRD was utilized to analyze the crystalline composition in the char, as shown in Figure 4(b). The residual char of pure EC and EC/10DQ only showed a broad peak at around 25°, corresponding to the (002) crystal plane of graphite. In the residual char of EC/10CuHS and EC/7DQ3CuHS, the major crystalline components were SnO (JCPDS No. 07-0195) and CuO (JCPDS No. 80-0076).29–31 The metal oxides in the residual char played an important role in improving the density of the carbon layer as physical barrier.
The synergistic mechanism between DQ and CuHS on the flame retardancy and smoke suppression of EC composites was as follows. During combustion of EC composites, DQ mainly played a role in the gas phase. The phosphorus-containing free radicals generated from DQ terminated the combustion chain reaction by capturing the active free radicals, 32 while the H2O released by CuHS decomposition played a role in diluting combustible gases and cooling.18,33 In the condensed phase, the metal oxides formed by the decomposition of CuHS acted as a physical barrier on the EC surface, which improved the quality of the char layer and effectively prevented mass and heat transfer between the gas phase and the condensed phase. 34
Thermal decomposition behavior
The thermal decomposition behavior of DQ was studied by TG, and the results are shown in Figure 5(a). The decomposition of DQ mainly occurred between 310°C and 520°C, during which the P-C and C-C bonds from DQ broke and volatile substances were released.
35
After 520°C, the decomposition rate was relatively slow, corresponding to the further oxidation of the residue. The T-5wt% of DQ was 342.9°C, and its Tmax was 424.1°C, indicating its high thermal decomposition temperature. In addition, the char residue of DQ at 800°C was only 1.5 wt%, indicating its poor carbonization ability and mainly action in the gas phase.
16
(a) TG and DTG of DQ, (b) DSC of pure EC and its composites, (c) TG and (d) DTG curves of pure EC and its composites.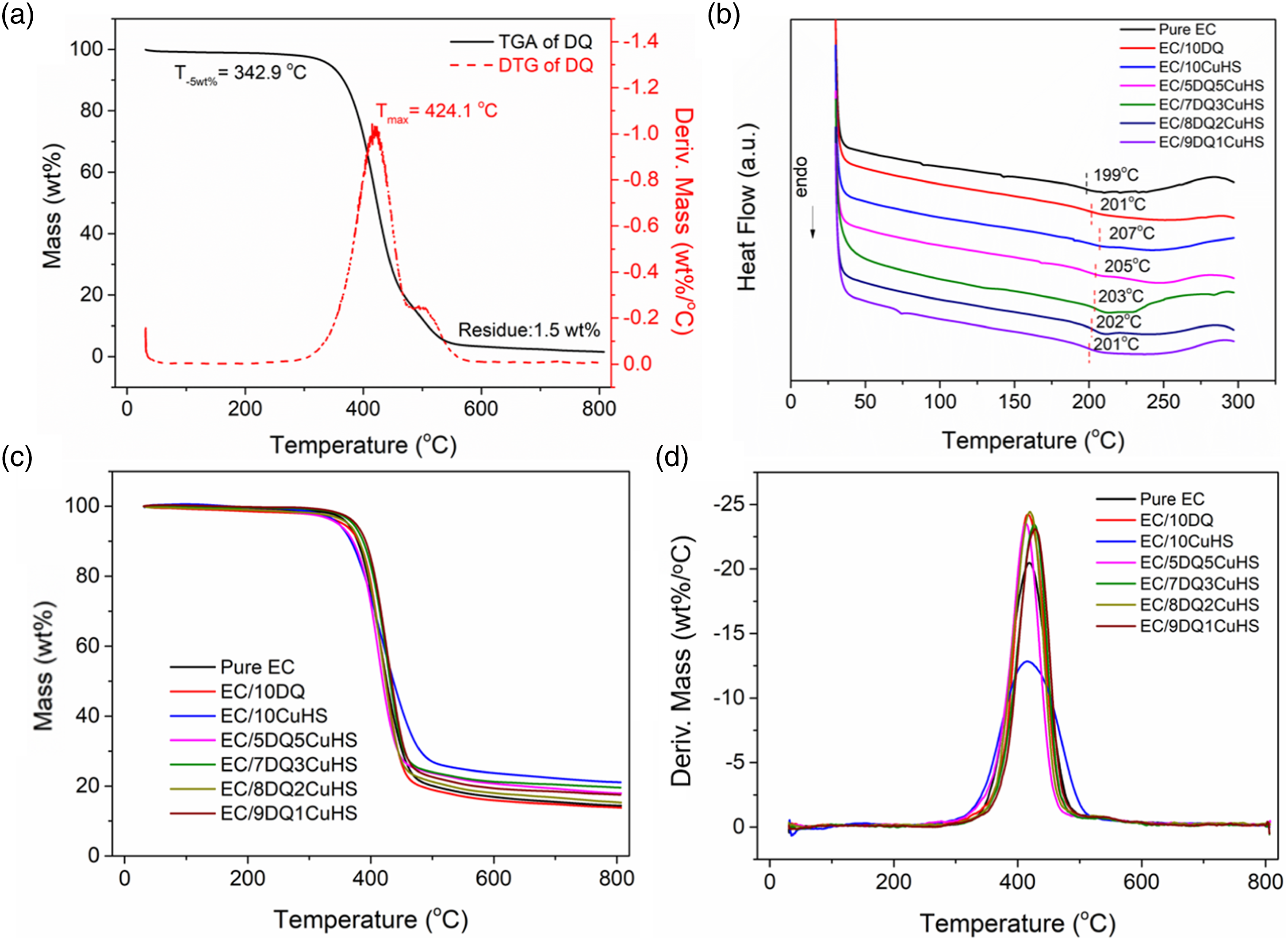
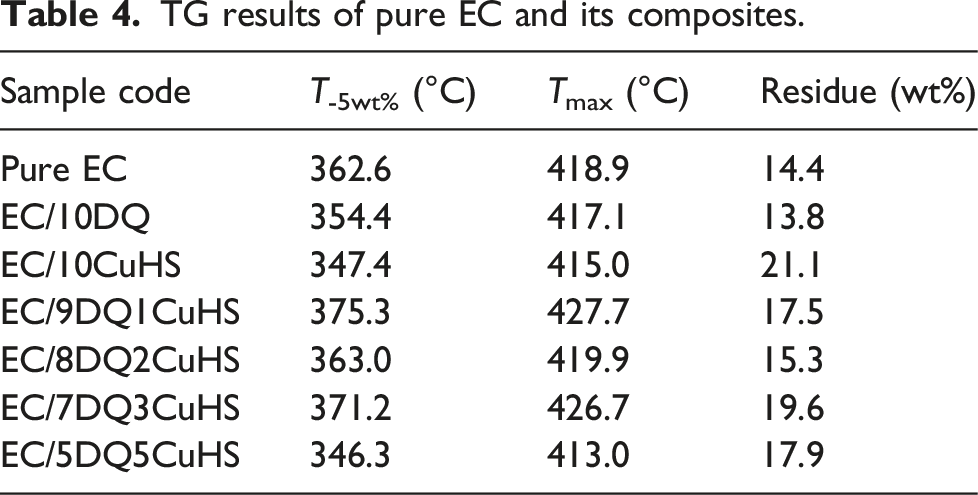
TG results of pure EC and its composites.
The glass transition temperature (Tg) of EC composites was studied by DSC, and the DSC curves are shown in Figure 5(b). The Tg of pure EC was about 199°C, and the Tg of EC/10DQ increased to 201°C. This is mainly due to the introduction of rigid benzene ring structures from DQ. 15 The Tg of EC/10CuHS was 207°C, while the Tg of the EC composites with both DQ and CuHS was located in between. Moreover, the Tg values of EC composites increased with the incremental CuHS content. Above all, both DQ and CuHS improved the Tg of EC composites, thus met the application occasions demand for high initial thermal resistance.
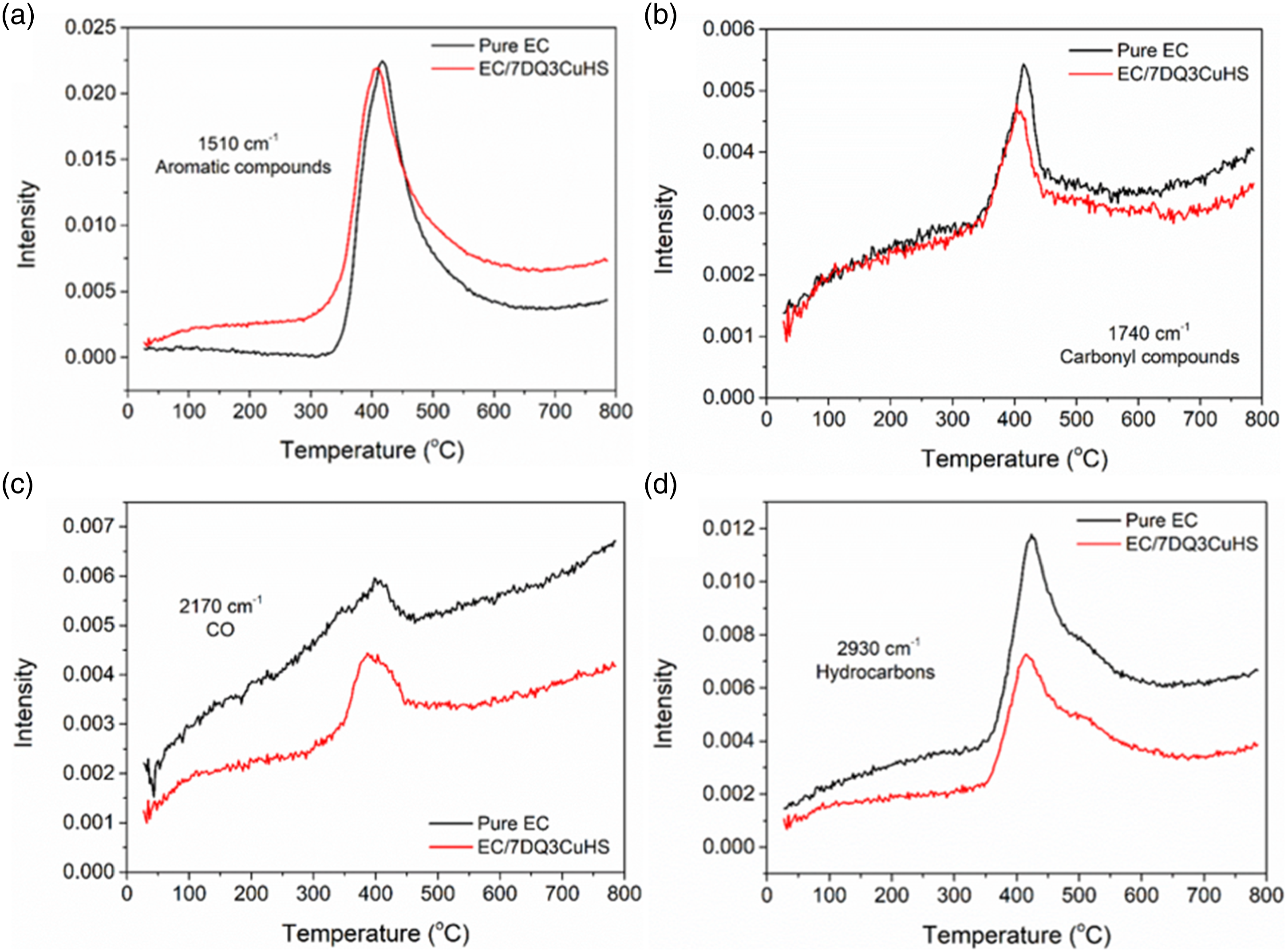
TG-IR was used to further analyze the decomposition products from pure EC and EC/7DQ3CuHS in the gas phase. According to the three-dimensional infrared spectra and the spectra of decomposition products at different temperatures (Figure 6(a)–(d)), the major decomposition products from pure EC and EC/7DQ3CuHS included NH3 (∼3251 cm−1), hydrocarbons (2960 cm−1), carbon dioxide (2360 cm−1), carbon monoxide (2170 cm−1), carbonyl compounds (1740 cm−1), aromatic compounds (1510 cm−1) and compounds containing C-OH bond (1328 cm−1, 1260 cm−1 and 1180 cm−1). Figure 7 provides intuitive comparison of the relative absorption intensity of major decomposition products between pure EC and EC/7DQ3CuHS as a function of temperature. The relative absorption intensities of carbonyl compounds, CO, and hydrocarbons decomposed from of EC/7DQ3CuHS were significantly lower than those of pure EC within 800°C, while the absorption intensity of aromatic compounds (smoke precursors) from EC/7DQ3CuHS was slightly higher than that of pure EC, which was due to the volatile aromatic compounds generated from decomposition of DQ entering the burning zone, corresponding to the increase of smoke release in CCT. Nevertheless, CuHS mainly acted in the condensed phase and showed little effect on the decomposition products in the gas phase. TG-FTIR curves of (a) pure EC, (b) EC/7DQ3CuHS; absorbance of decomposition products of (c) pure EC, (d) EC/7DQ3CuHS under various temperatures. Intensities of major decomposition products of pure EC and EC/7DQ3CuHS, (a) aromatics, (b) carbonyl compounds, (c) CO, (d) hydrocarbons.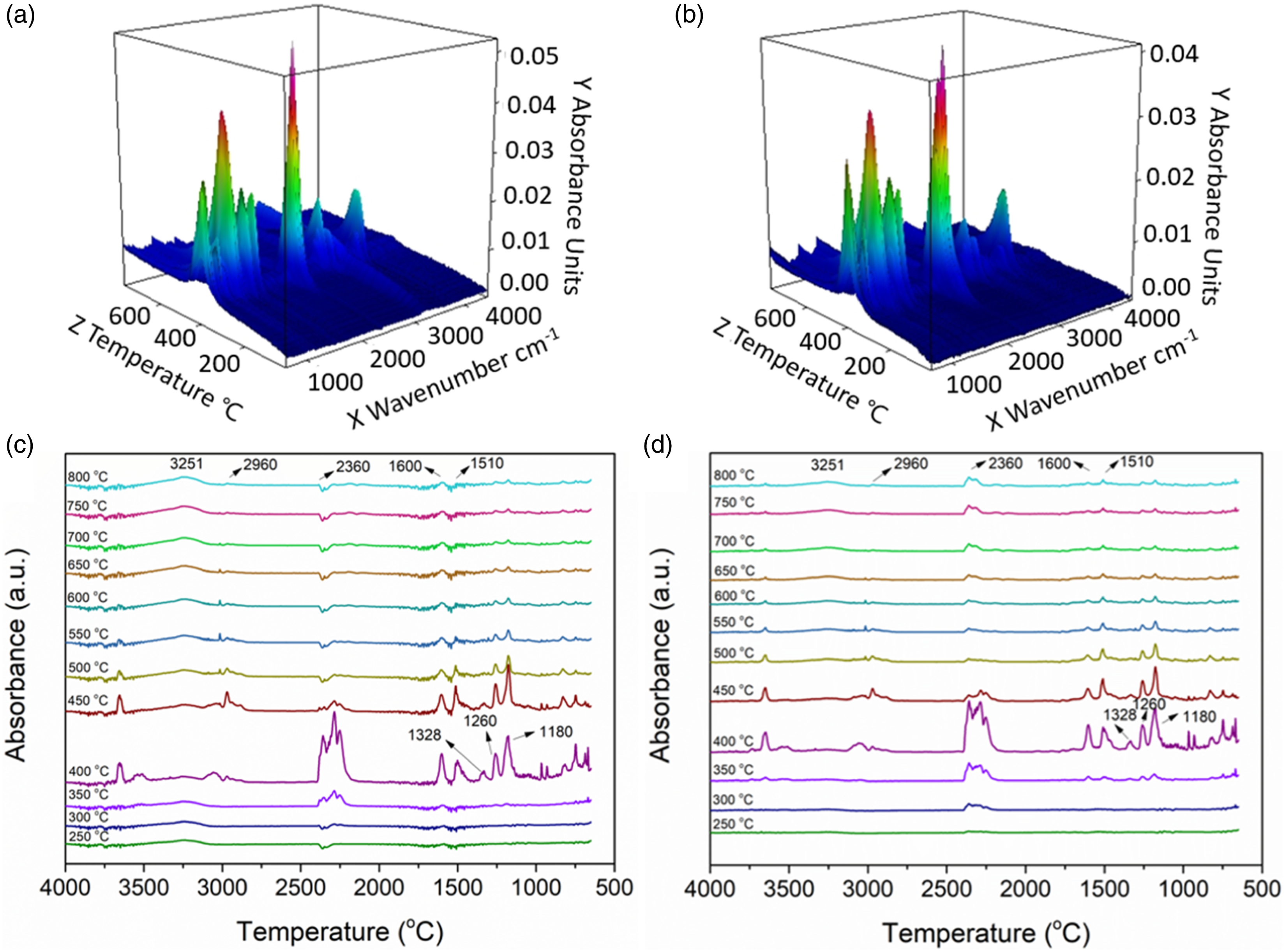
Mechanical properties
The mechanical properties of pure EC and its composites are shown in Figure 8. The tensile and impact strengths of pure EC were 47.2 MPa and 12.5 kJ/m2, respectively, while those of EC/10DQ were increased to 51.0 MPa and to 13.8 kJ/m2, respectively. The tensile and impact strengths of EC/9DQ1CuHS were further increased to 55.2 MPa and 14.9 kJ/m2, which were 16.9% and 19.2% higher than those of pure EC. As the amount of CuHS gradually increased to 2 wt% and 3 wt%, the tensile and impact strengths of EC composites gradually decreased, while still higher than those of pure EC. The tensile and impact strengths of EC/10CuHS were 40.7 MPa and 7.7 kJ/m2, respectively, which were 13.8% and 38.4% lower than those of pure EC. Above all, it was found that the combination of small amount of CuHS (<3 wt%) with DQ improved the tensile and impact strengths of EC composites. The addition of DQ increases the number of rigid triazine rings in EC composites, which improved the mechanical strengths of EC composites.
36
In addition, a small amount of CuHS was uniformly dispersed in the EC composites, which showed reinforced effect and inhibited the crack propagation of the EC composites under external forces.
14
(a) Tensile strength, (b) impact strength of pure EC and its composites.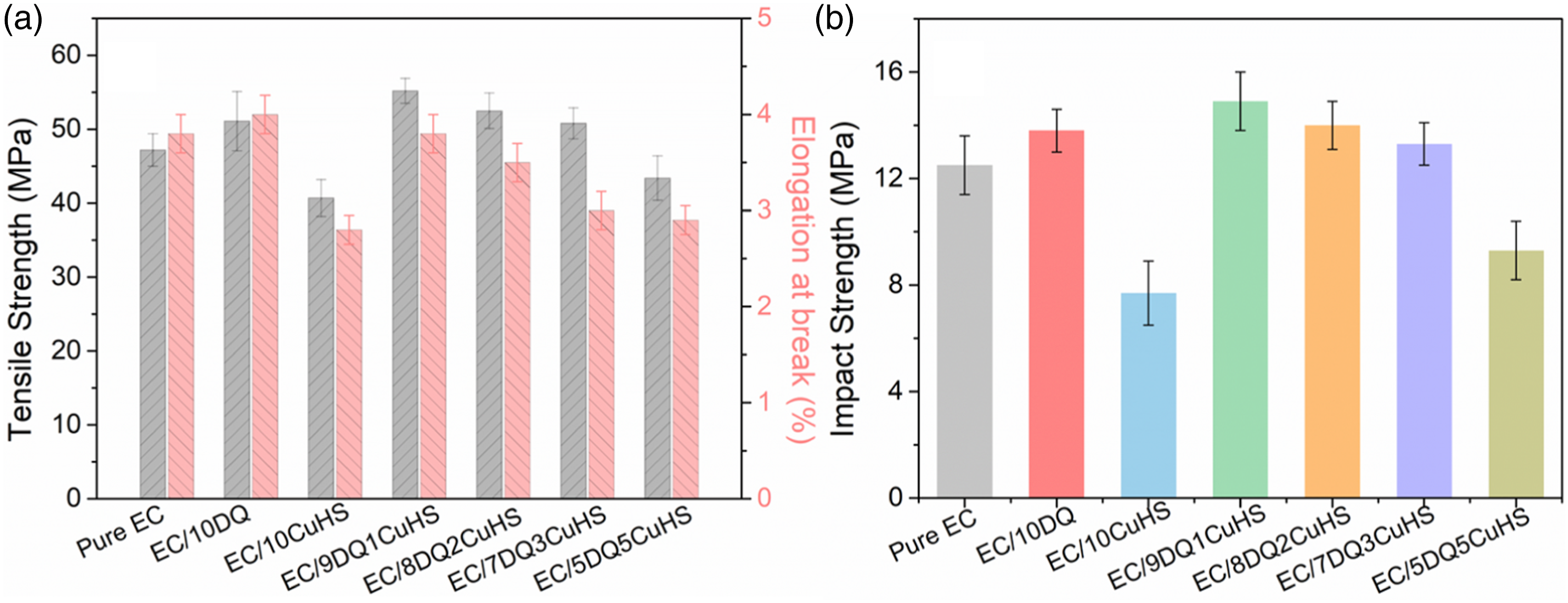
The morphology of the fracture surfaces of the EC composites after tensile tests was observed by SEM, shown in Figure 9. It was seen from Figure 9(a) that few protuberances and wrinkles were distributed on the relatively smooth fracture surface of pure EC, indicating the typical brittle fracture of homogeneous EC.
37
For EC/10DQ, the protuberances and wrinkles on fracture surface were significantly increased compared to pure EC, and the depth of the cracks was significantly increased (Figure 9(b)), indicating more energy consumed for crack propagation under external forces.
38
For EC/10CuHS (Figure 9(c)), there were fewer protuberances and wrinkles distributed on the surface and the cracks were less deep than EC/10DQ. In addition, some particle aggregates were observed on the fracture surface of EC/10CuHS, indicating that the addition of 10 wt% CuHS was difficult to disperse uniformly in the EC matrix, therefore affecting the mechanical strengths of the EC composites. As CuHS gradually replaced DQ, the protuberances and wrinkles on the fracture surface of EC/9DQ1CuHS (Figure 9(d)), EC/8DQ2CuHS (Figure 9(e)), and EC/7DQ3CuHS (Figure 9(f)) showed irregular shapes and gradually concentrated distribution of cracks. It was mainly due to the strong interface interaction between CuHS of a small amount and the EC matrix, altering the direction of crack propagation under external forces.
39
In addition, the aggregates extent of particles on the fracture surface gradually increased with the incremental CuHS content. For EC/9DQ1CuHS, the CuHS particles were dispersed evenly in the EC matrix, corresponding to the highest tensile strength. From the SEM image of EC/5DQ5CuHS (Figure 9(g)), it was seen that the protuberances and wrinkles density and crack depth were reduced, which was due to the less DQ dosage. Moreover, more CuHS aggregates were distributed on the surface led to stress concentration in the EC composites under external forces, resulting in a decrease in tensile strength. SEM images of the fracture surfaces of pure EC and its composites, (a) pure EC, (b) EC/10DQ, (c) EC/10CuHS, (d) EC/9DQ1CuHS, (e) EC/8DQ2CuHS, (f) EC/7DQ3CuHS, (g) EC/5DQ5CuHS.
Dielectric properties
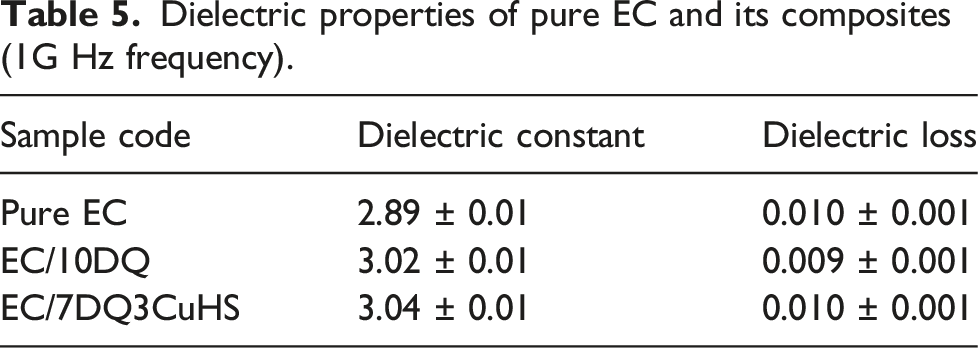
Dielectric properties of pure EC and its composites (1G Hz frequency).
Conclusions
In the present work, the effects of the combination of DQ and CuHS on the flame retardancy, smoke suppression, mechanical properties, thermal decomposition properties and dielectric properties of EC composites were investigated. Compared with pure EC, the pHRR and THR of the EC composite containing 7 wt% DQ and 3 wt% CuHS (EC/7DQ3CuHS) were fell by 41.9% and 35.0%, respectively. Compared with EC/10DQ, the TSR of EC/7DQ3CuHS was decreased by 36.0%. In addition, EC/7DQ3CuHS passed V0 classification in the UL-94 VB tests with a LOI of 32.0%. The tensile and impact strengths of EC/9DQ1CuHS were increased by 16.9% and 19.2%, respectively compared with pure EC. The dielectric constant and dielectric loss of EC composites remained at a relatively low values, which showed application prospects in the field of microelectronics.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21975185).
