Abstract
The effects of varying the alkali metal cation in the high-temperature nucleophilic synthesis of a semi-crystalline, aromatic poly(ether ketone) have been systematically investigated, and striking variations in the sequence distributions and thermal characteristics of the resulting polymers were found. Polycondensation of 4,4′-dihydroxybenzophenone with 1,3-bis(4-fluorobenzoyl)benzene in diphenylsulphone as solvent, in the presence of an alkali metal carbonate M 2CO3 (M = Li, Na, K, or Rb) as base, affords a range of different polymers that vary in the distribution pattern of two-ring and three-ring monomer units along the chain. Lithium carbonate gives an essentially alternating and highly crystalline polymer, but the degree of sequence randomization increases progressively as the alkali metal series is descended, with rubidium carbonate giving a fully random and non-thermally crystallizable polymer. Randomization during polycondensation is shown to result from reversible cleavage of the ether linkages in the polymer by fluoride ions, and an isolated sample of alternating sequence polymer is thus converted to a fully randomized material on heating with rubidium fluoride.
Keywords
Introduction
Composite materials for aerospace applications have traditionally been based on thermosetting matrix polymers such as the epoxies and bismaleimides,
1,2
but in more recent years the potential advantages of thermoplastic matrices (increased speed of fabrication and greater toughness) have begun to be realized,
3
notably with the introduction of long-fibre composites based on semi-crystalline engineering polymers such as poly(1,4-phenylene sulphide)
4
and the aromatic poly(ether ketone)s (PEKs), poly(ether ether ketone) (PEEK)
5
and poly(ether ketone ketone) (PEKK
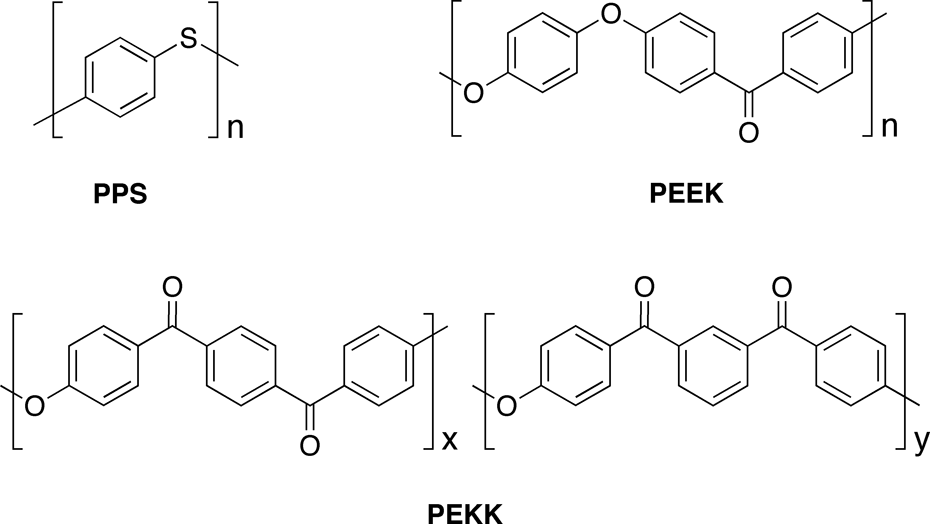
Some thermoplastic aromatic matrix polymers used in carbon fibre composites.
The high crystalline melting points (T
m) of PEKs (typically 340–380°C) result in the retention of significant mechanical strength and stiffness even at temperatures well above their glass transition temperatures (T
gs).
7
However, such T
ms also require correspondingly high composite fabrication temperatures – up to 420°C.
3,8
In the present article, we report a study of a lower melting but still crystallizable PEK matrix polymer (
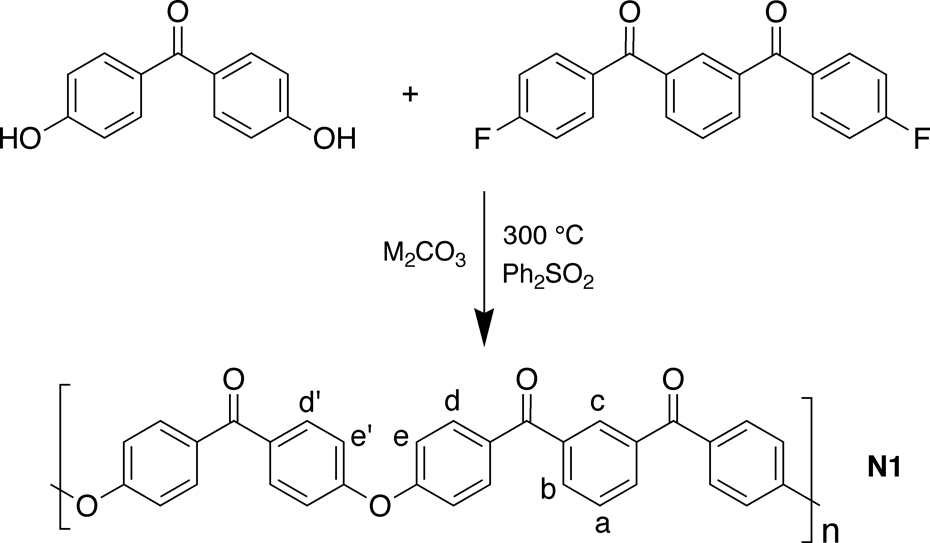
Synthesis of polymer
The polymer that might naively be expected from the above polycondensation would comprise an alternating sequence of two-ring and three-ring monomer residues. A rigorously alternating structure of this type has been obtained from the electrophilic polycondensation of 4,4′-diphenoxybenzophenone with isophthaloyl chloride (Figure 3).
10,11
The resulting semi-crystalline polymer (
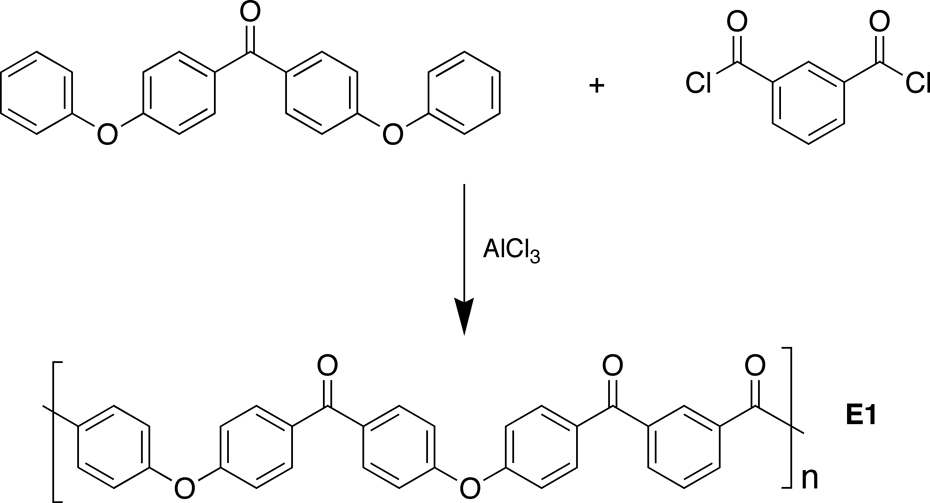
Experimental
Materials, instrumentation and analysis
Monomers, solvents, alkali metal carbonates and other reagents were obtained from Sigma Aldrich (UK) and were used without further purification. Inherent viscosities (η inh) were measured at 25°C with 0.1% w/v polymer solutions in 96% sulphuric acid (H2SO4) using a Schott Instruments CT 52 viscometer (Mainz, Germany). No insoluble gel fractions were present in any of the polymers described. Phase transitions (glass transitions, cold crystallizations and melting points) were identified from the second heating cycles of differential scanning calorimetry (DSC) traces using a TA DSC Q2000 instrument (New Castle, Delaware, USA; 4–12 mg samples, 10°C min−1 under nitrogen atmosphere). A slight excess of alkali metal carbonate was used in each polycondensation to ensure quantitative conversion of the bisphenol to bisphenoxide. Yields of polymers were essentially quantitative and were diminished only by mechanical losses during the milling stage. Proton (1H) and carbon (13C) nuclear magnetic resonance (NMR) spectra were obtained on Bruker Nanobay 400 MHz or 700 MHz NMR spectrometers (Billerica, Massachusetts, USA) using polymer solutions in (deuterated chloroform; CDCl3/hexafluoro-2-propanol; (CF3)2CHOH (6:1 v/v) or CDCl3/trifluoro acetic acid (CF3COOH) (6:1 v/v). Mass spectra (electrospray ionization (ESI)) were obtained from 0.1% (w/v) sample solutions in methanol using a ThermoScientific LTQ OrbiTrap XL instrument (Waltham, Massachusetts, United States) equipped with an ACCELA LC autosampler.
Synthesis and characterization
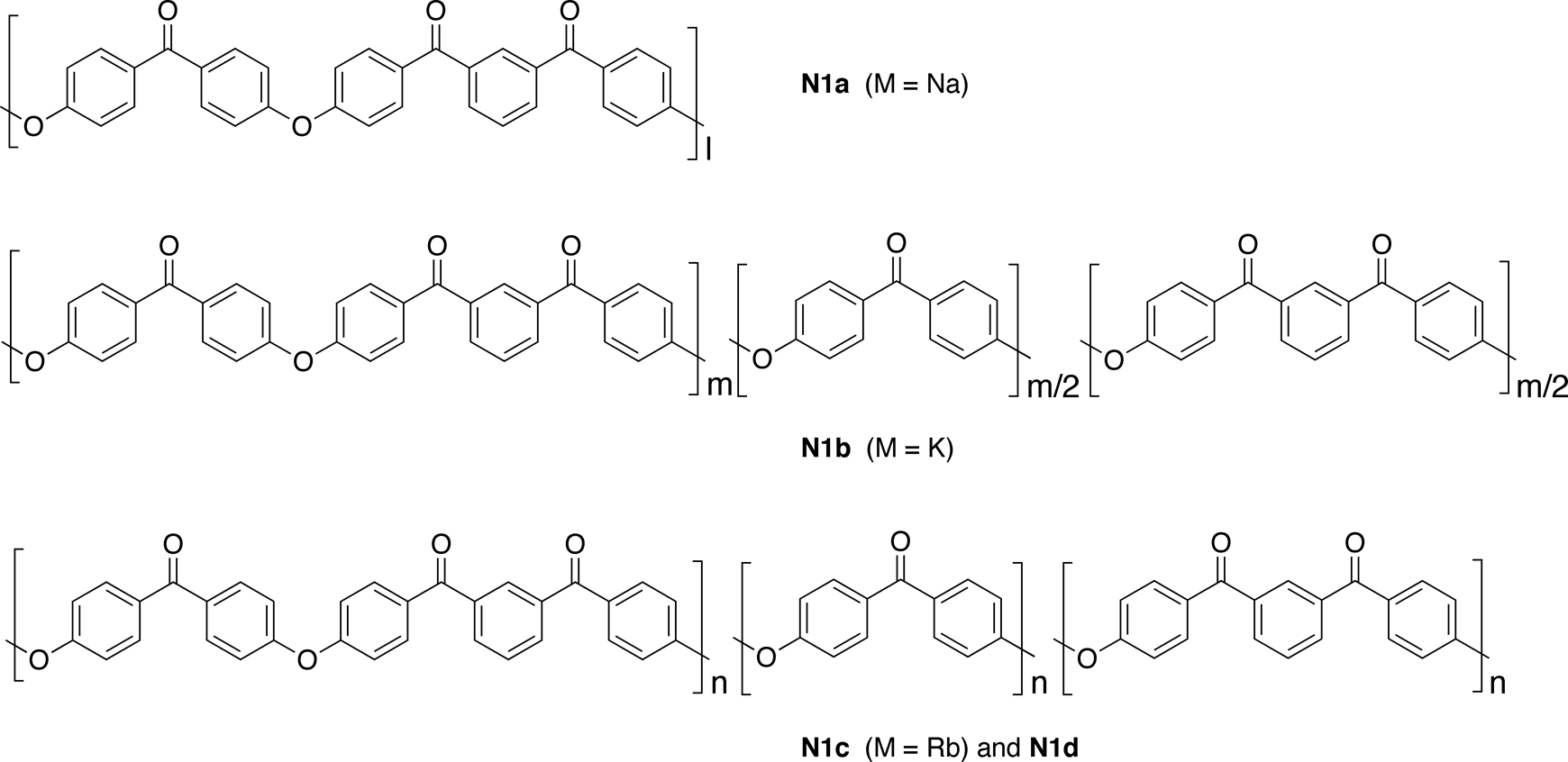
Polymer N1a
A mixture of 1,3-bis(4-fluorobenzoyl)benzene (4.60 g, 14.28 mmol), 4,4′-dihydroxybenzophenone (3.00 g, 14.00 mmol), sodium carbonate (Na2CO3; 1.63 g, 15.4 mmol) and diphenylsulphone (35 g) was heated with stirring to 300°C under argon atmosphere. After 3 h, the polymer solution was poured onto a sheet of aluminium and allowed to cool. The resulting solid was ground to a powder in an ultracentrifugal mill and then stirred in acetone (200 mL) at room temperature for 30 min. The powder was filtered off, washed with acetone and dried. The powder was next extracted with 4× 200 mL of refluxing acetone, and then overnight in a Soxhlet extractor with refluxing acetone. The powder was extracted with 5× 200 mL of boiling water and then finally with 4× 200 mL of refluxing acetone. The resulting, purified material was dried at 110°C under vacuum overnight, affording polymer
Polymer N1b
This polymer was obtained using the procedure described for polymer
Polymer N1c
The same procedure as described for polymer
Polymer N1d
Polymer
Polymer N1e
The same procedure as described for polymer
Bisphenol 2
A solution of 4-hydroxy-3-methylbenzoic acid (3.00 g, 19.7 mmol) and o-cresol (2.27 g, 21 mmol) in a mixture of trifluoromethanesulphonic anhydride (3.36 mL, 20 mmol) and trifluoromethanesulphonic acid (30 mL) was stirred under nitrogen for 18 h. The solution was then added dropwise with stirring into 800 mL of cold water. The precipitate was filtered off, washed repeatedly with boiling water, then with a 0.01M sodium hydroxide solution, and finally with cold water until a neutral filtrate was obtained. The solid was dried in a vacuum oven at 90°C for 5 h to afford bisphenol
Polymer 3
A mixture of bisphenol
Results and discussion
Polycondensation of 4,4′-dihydroxybenzophenone with 1,3-bis(4-fluorobenzoyl)-benzene, in diphenylsulphone as solvent, at 300°C in the presence of an alkali metal carbonate M
2CO3 as base (Figure 2; M = Na, K, or Rb), afforded high-molecular-weight PEK
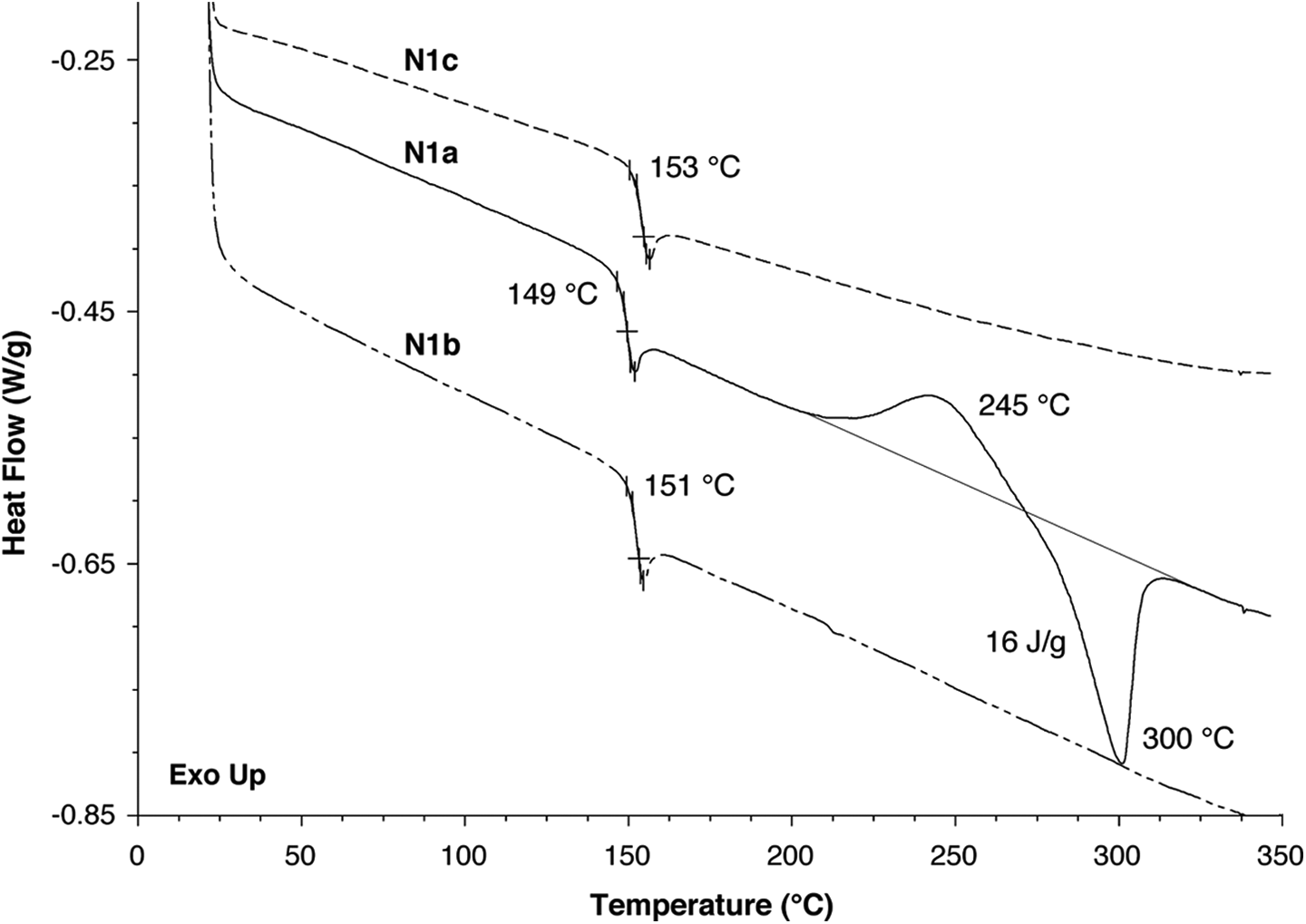
DSC scans (2nd heating, 10°C min−1) for polymers
It seemed possible that the observed variation in crystallizability between the three polymers could result from differences in their sequence distributions since transetherification with sequence randomization is known to occur during the nucleophilic synthesis of aromatic polyethers in which both monomer residues in the chain are activated towards nucleophilic attack adjacent to the ether linkage.
12,13
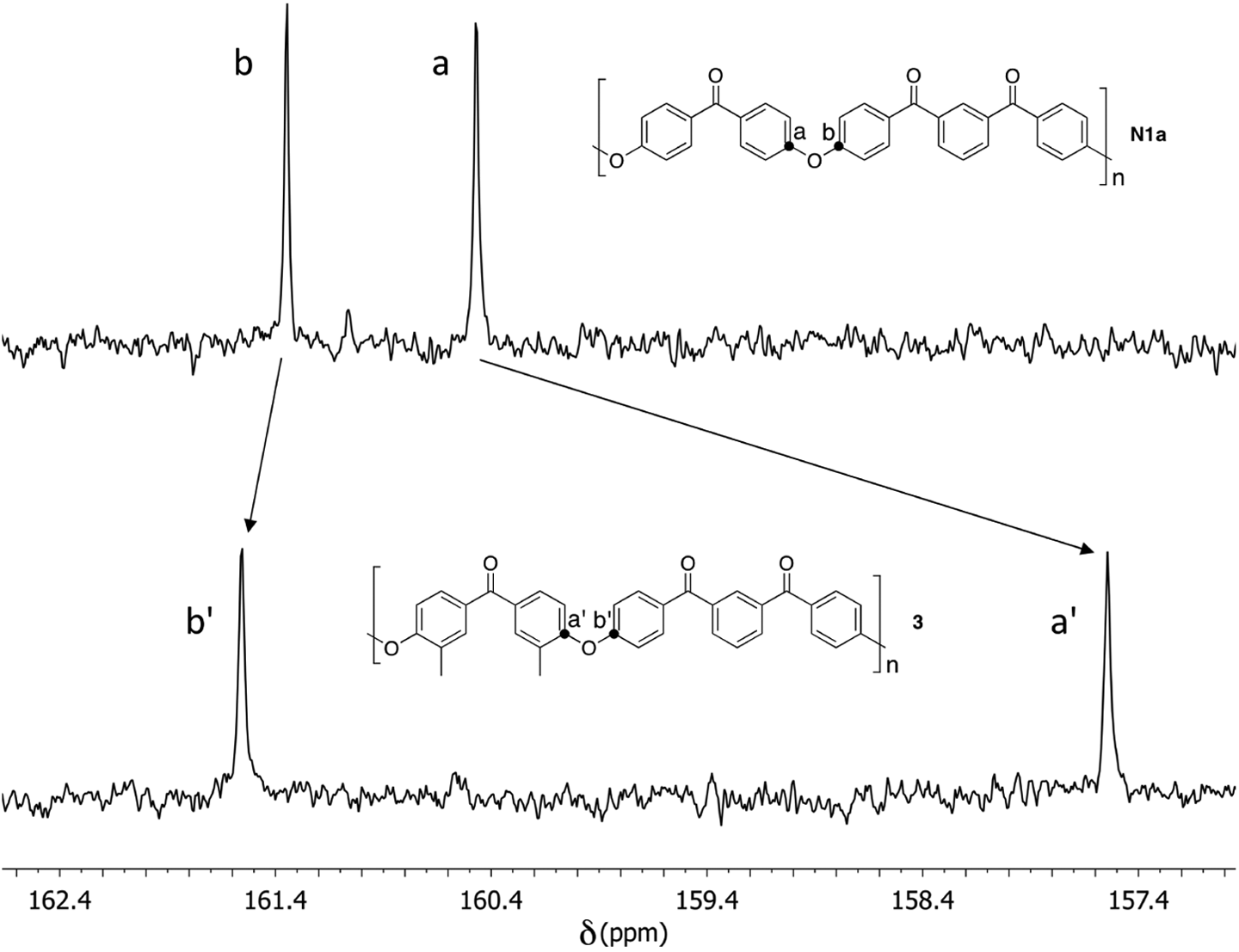
This possibility was confirmed by 13C NMR analysis (Figure 5), which showed useful diagnostic resonances in the range δ = 160–162 ppm, corresponding to the aromatic carbons attached directly to ether oxygens. Polymer
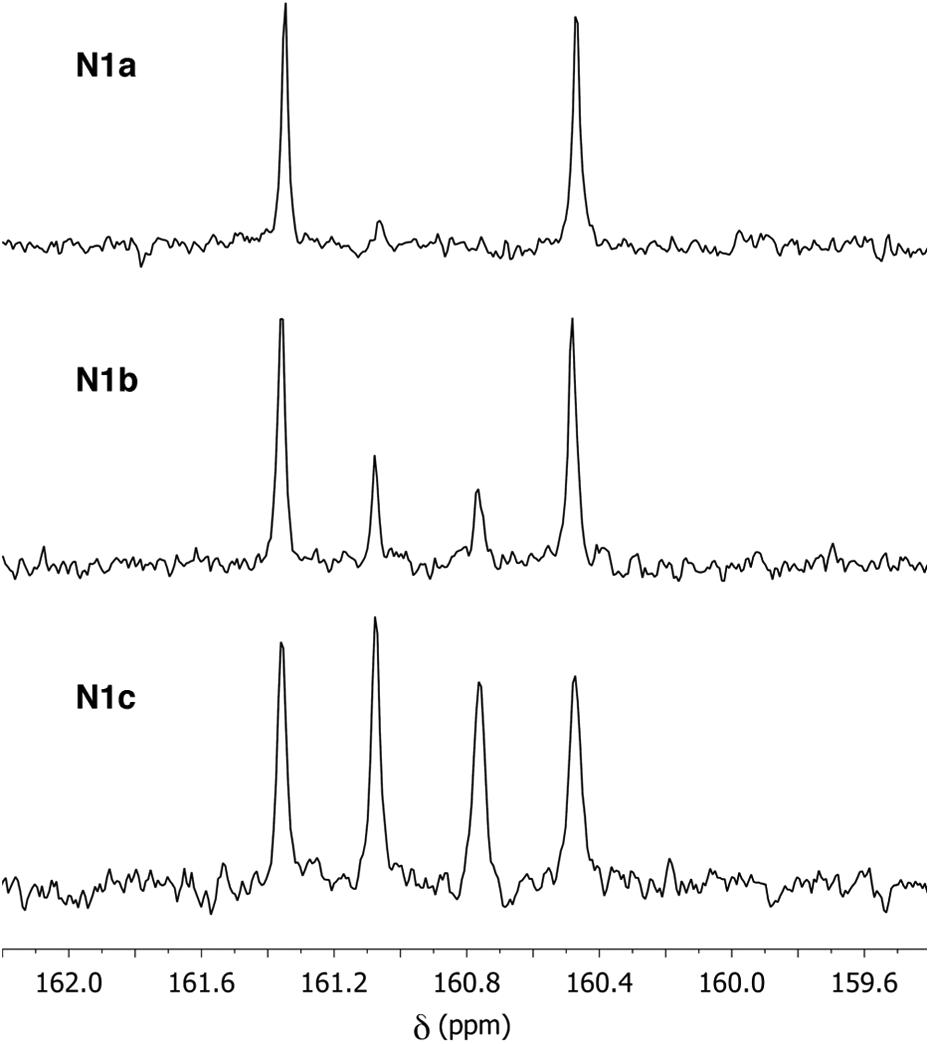
13C NMR resonances in the C–O–C region for polymers
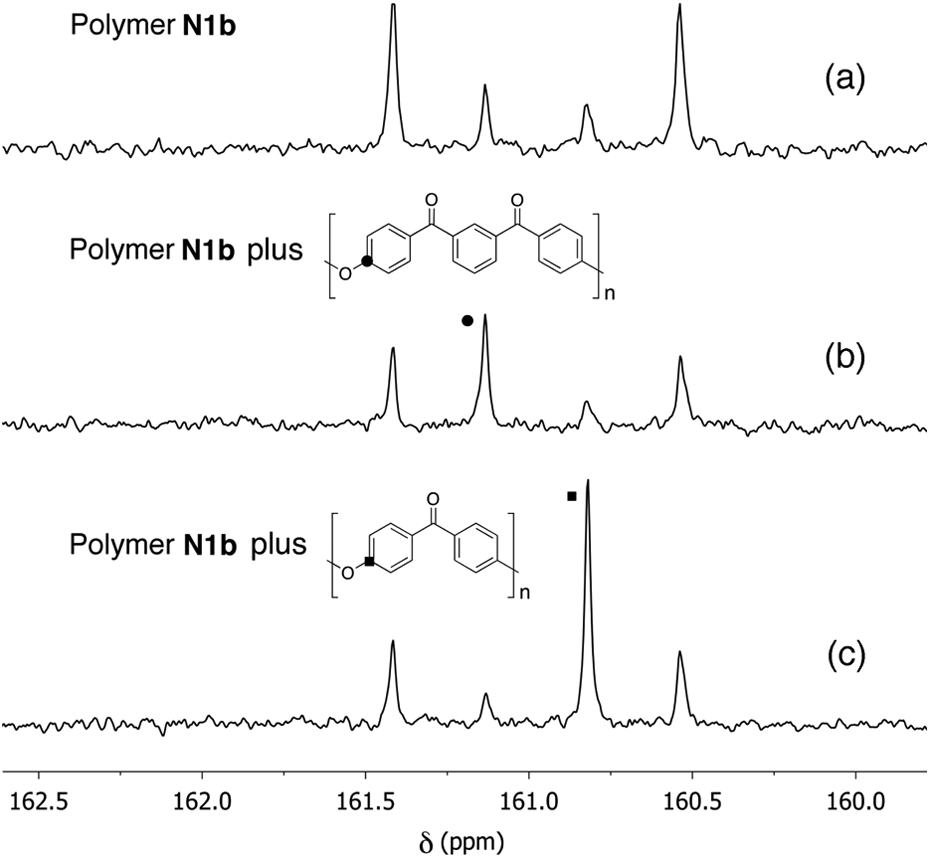
In order to aid the assignment of 13C–O–C peaks to the specific two-ring and three-ring residues, a dimethyl-substituted polymer (
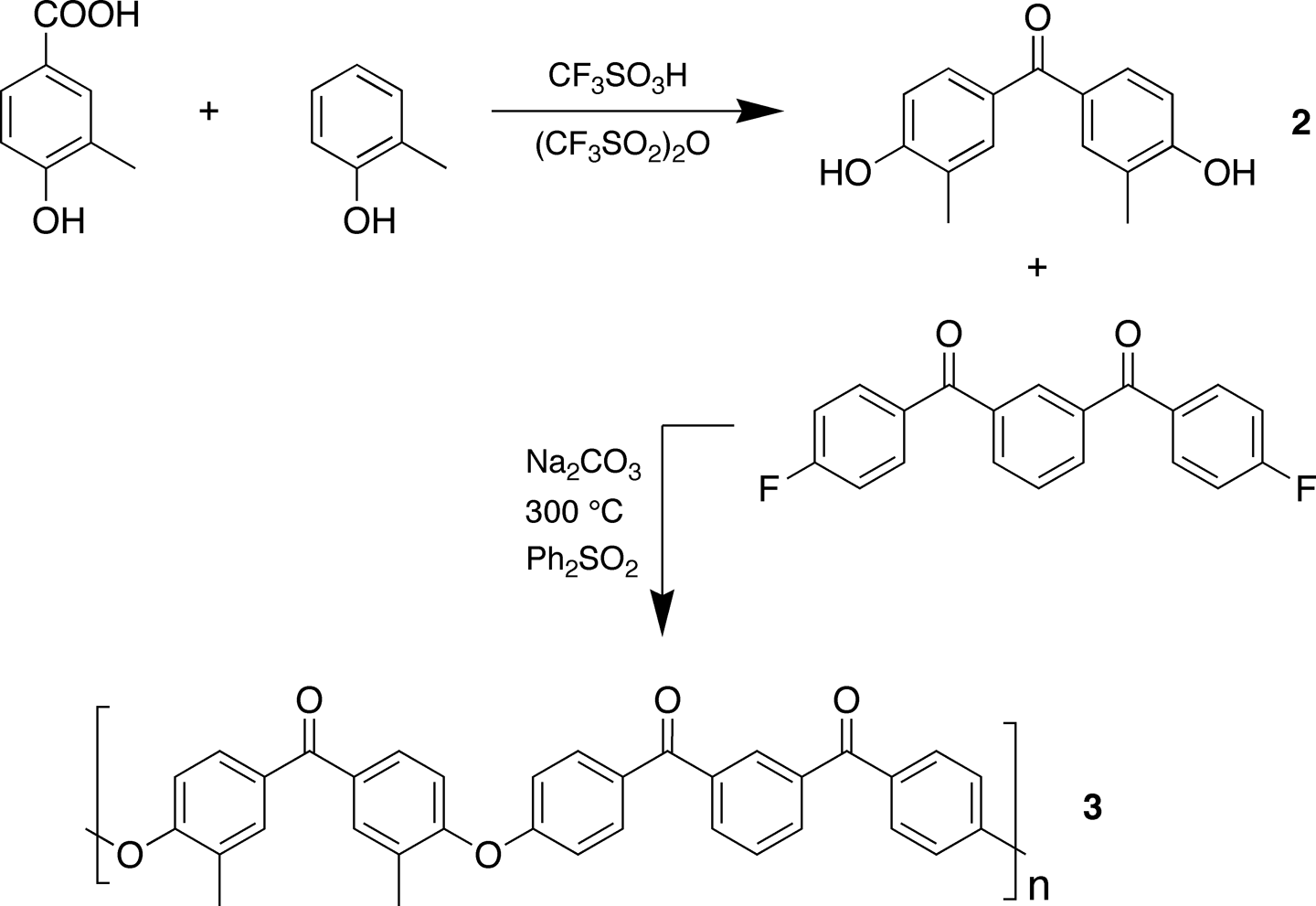
Synthesis of bisphenol
In the 13C NMR spectrum of polymer
13C NMR resonances in the C–O–C region for polymers N
The 13C NMR spectra of polymers
13C NMR resonances in the C–O–C region for (a) polymer
The 13C NMR spectrum of polymer
Representations of the chain sequences in polymers
A number of possible mechanisms have been proposed for transetherification during the synthesis of aromatic PEKs, but all depend on reversible, nucleophilic cleavage of the ether linkages (Figure 10). Candidate nucleophiles in the system include the carbonate and fluoride anions, and indeed potassium carbonate has previously been shown to induce a small degree of sequence randomization in an aromatic PEK, albeit requiring very high reaction temperatures (340°C) and long reaction times (6 h).
15
The fluoride ion can be a very strong nucleophile in dipolar aprotic solvents,
16,17
but its effectiveness in the present context would depend both on the solubility of the fluoride salt involved and on the extent of pairing with its counterion in solution. The larger the counterion, the weaker the ion pairing and the more soluble the salt, so RbF should be much more effective than sodium fluoride, with potassium fluoride somewhere in between (r
ionic = 1.16, 1.52, 1.66 Å for 6-coordinate Na+, K+, and Rb+, respectively).
18
This is fully consistent with our experimental results for sequence randomization in the synthesis of
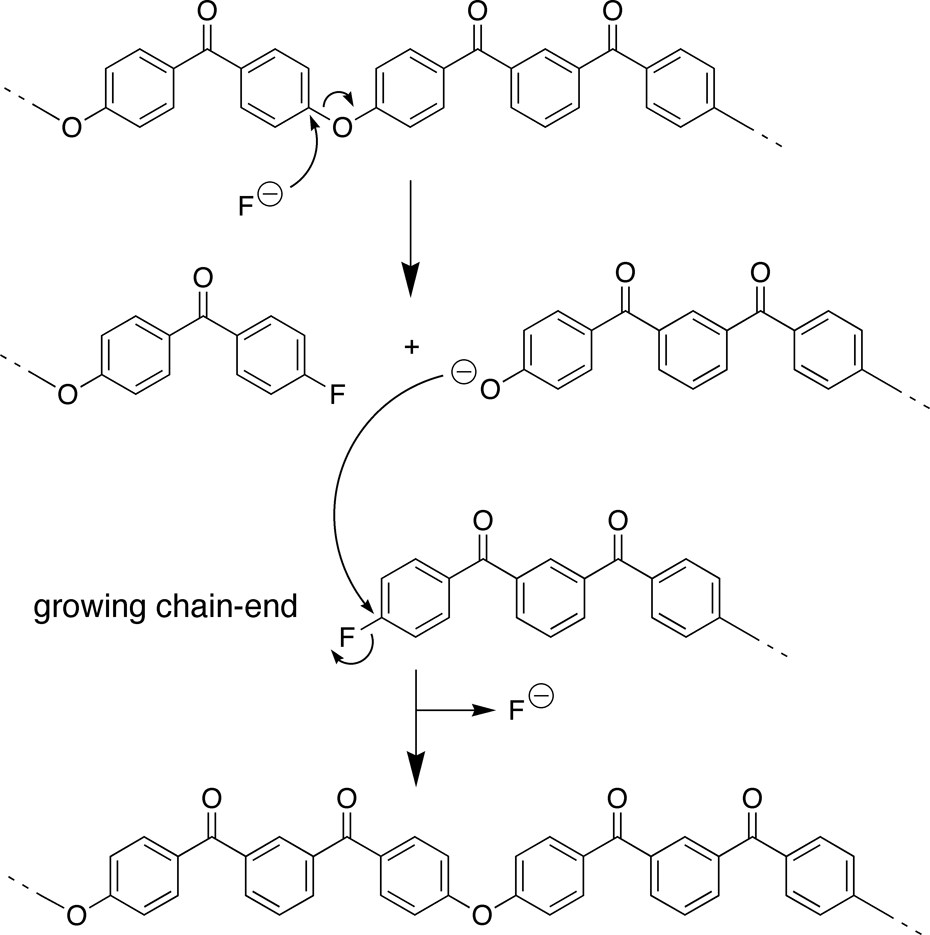
Partial mechanism for fluoride-catalysed sequence randomization in the synthesis of polymer
In the present work, sequence randomization catalysed by fluoride ion was demonstrated conclusively by treatment of the alternating polymer
As a final test of the proposed mechanism for sequence randomization, the polycondensation shown in Figure 2 was carried out using lithium carbonate as base. The extremely low solubility of lithium fluoride in organic solvents
19
should strongly inhibit fluoride-catalysed transetherification and indeed, as shown in Figure 11, resonances arising from sequence randomization were scarcely discernable in the 13C NMR spectrum of the resulting polymer (
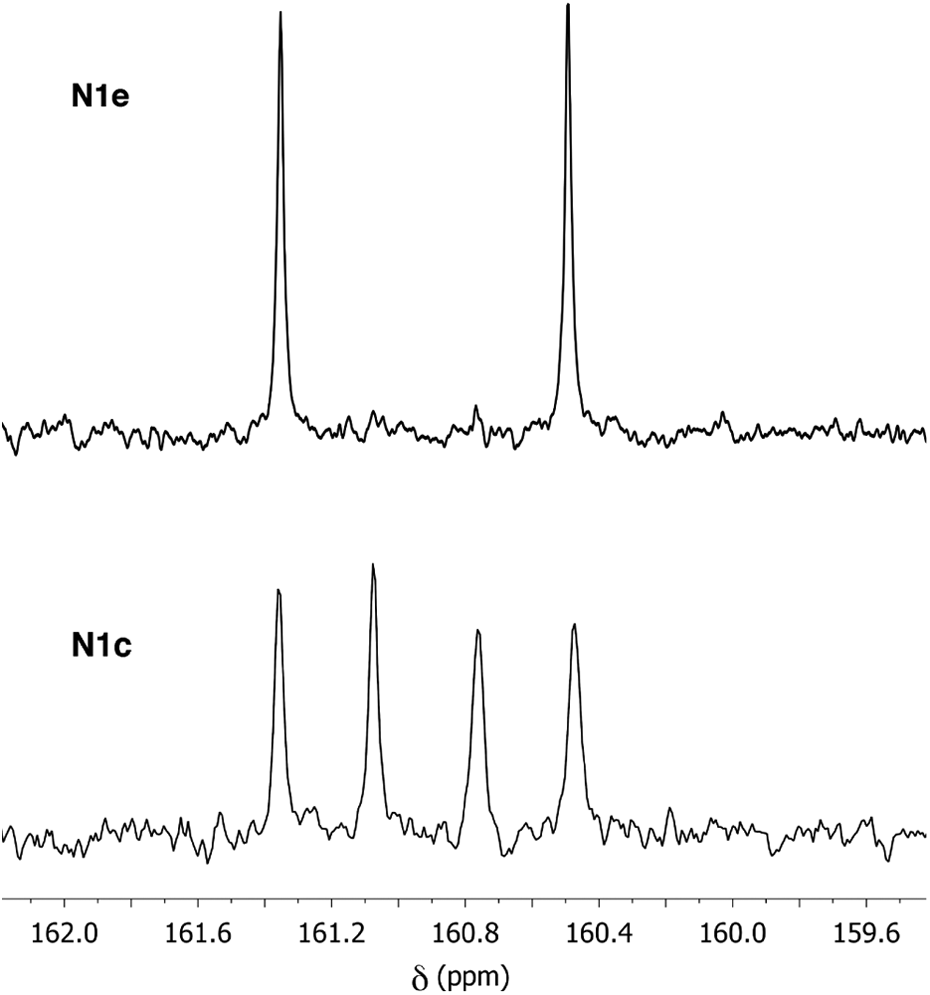
13C NMR resonances in the C–O–C region for polymers
As shown in Figure 12, polymer
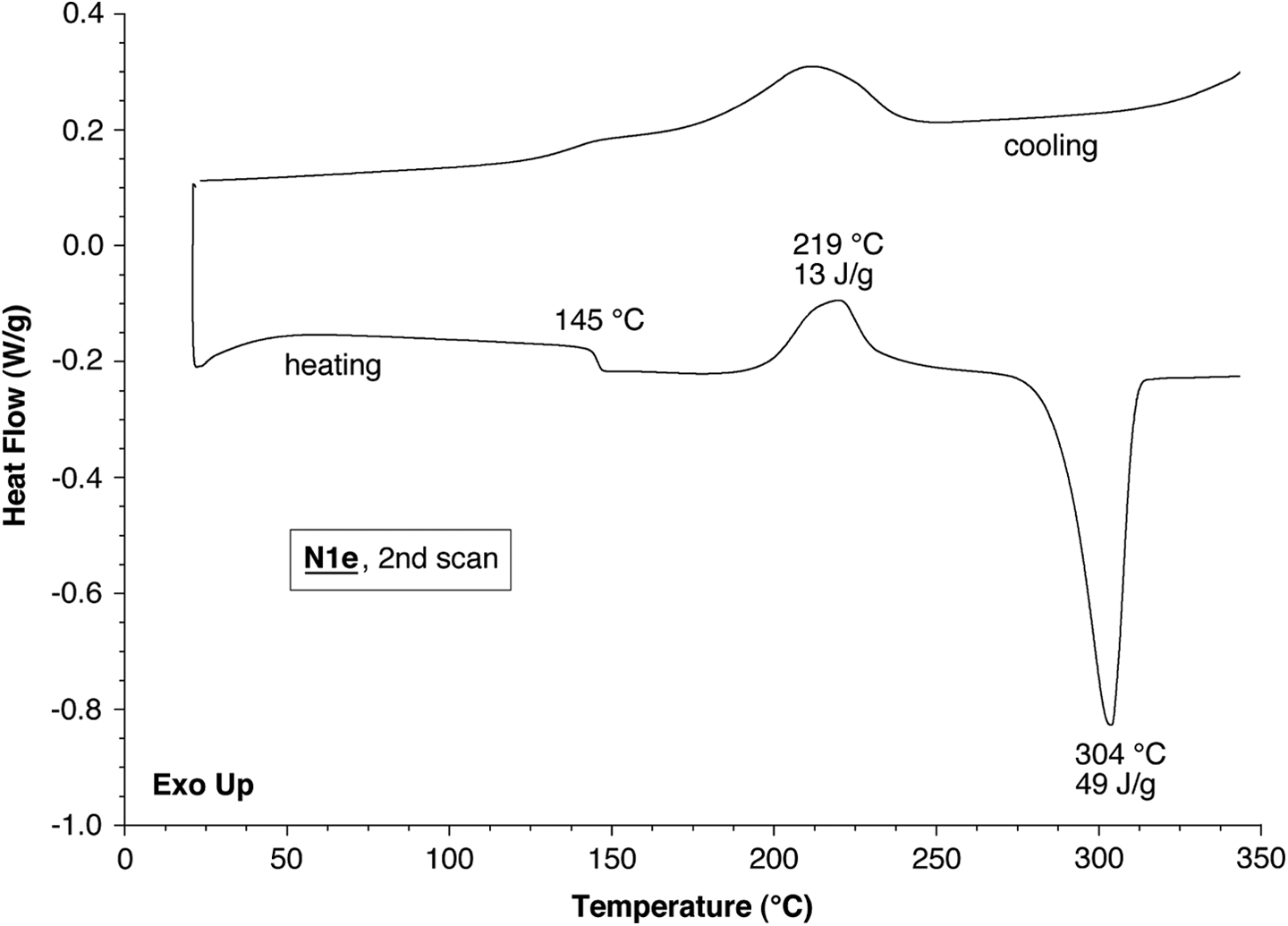
DSC scans (2nd heating and subsequent cooling scan, 10°C min−1) for polymer
An intriguing observation is that, although DSC analysis customarily discounts the first heating scan, a consistent feature of the first (but not subsequent) DSC heating scans for polymers
Conclusions
Sequence randomization, via transetherification, during the nucleophilic synthesis of an aromatic PEK involving fluoride displacement from a bis(4-fluoroaryl)ketone can be controlled by varying the alkali metal cation (Li+, Na+, K+ or Rb+) present during polycondensation. The degree of transetherification increases with the ionic radius of the alkali metal involved, and a proposed mechanism in which fluoride ions reversibly cleave the growing polymer chain is substantiated by a direct demonstration of sequence randomization in the presence of RbF. The crystallizability of the polymer from the melt declines markedly as the degree of sequence randomization increases, although crystallization of the more highly randomized polymers from solution in diphenylsulphone affords an unusually low-melting crystalline phase whose nature remains to be established.
Footnotes
Authors’ Note
Underlying data for this article may be requested from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Cytec Aerospace Materials and the UK Engineering and Physical Sciences Research Council, who jointly funded a PhD studentship for K.J.C.L. through the University of Reading.
