Abstract
High-entropy alloys (HEAs) have gained substantial attention over the past two decades, necessitating a comprehensive understanding of their intrinsic and extrinsic properties, including mechanical behavior, creep resistance, phase stability, environmental degradation, etc. Among these, atomic transport mechanisms, particularly along grain boundaries (GBs) play a pivotal role in determining material performance.
This review critically evaluates the “sluggish” diffusion concept, focusing on its validity and applicability to GB diffusion in HEAs. It examines the influences of GB complexions, segregation phenomena, and precipitation processes on GB diffusion behavior in HEAs, comparing them with their counterparts in conventional binary and ternary alloys, both dilute and concentrated. The inherent challenges in accurately characterizing GB diffusion in multi-principal element alloys, given their broad compositional variability and complex microstructures are also highlighted. The contribution of inter-phase boundary diffusion in multi-component alloy systems is also identified and analyzed. Furthermore, the broader implications of GB diffusion on the mechanical and physical properties of polycrystalline HEAs are discussed in terms of their strength, ductility, and degradation resistance.
By consolidating the current state of research on GB diffusion in HEAs and identifying the key research gaps, this review aims to catalyze focused and intensive research efforts into diffusion-related phenomena in HEAs and other compositionally complex alloys. Emphasis is placed on comprehensive understanding the interplay between GB structure, chemistry, and atomic transport phenomena to enable effective GB engineering stategies for these alloys. Insights from such studies will be instrumental in optimizing HEAs for advanced technological applications and in guiding the development of next-generation high-performance materials.
Keywords
Introduction
Internal interfaces, particularly grain and interphase boundaries, play a crucial role in determining the properties and applications of most technologically significant materials.
1
These interfaces govern processing-induced functional modifications and long-term stability.2,3 Often, the impact of interfaces on material’s properties is represented by simply referring to the mean grain size
Internal interface engineering emerges as a key strategy for optimizing the properties and combinations of properties in advanced materials. For example, classical GB engineering, as pioneered by Watanabe,
5
has focused on enhancing the stability and properties of a material by increasing the fraction of low-
The interplay of precipitation kinetics with enrichment at GBs was investigated from thermodynamic perspectives13–15 or accounting for induced structural changes in form of dislocation networks.
16
Devaraj et al.
17
and Kalidindi and Schuh
18
have shown precipitation and segregation to reveal a strong and combined effect on the stability of nano-crystalline alloys. Rajeshwari et al.
19
and Bian et al.20,21 observed a coupled evolution of GB precipitates and the GB shape, with plate-like precipitates at irregular GB fragments and spherical particles at relatively straight interfaces. Moreover, the diffusion properties of high-angle GBs were found to evolve together with the characteristic changes of GB complexions distinguished by the growth of carbide- and
This present review is focused on high-entropy alloys (HEAs), that, more generally, should be termed single-phase multi-principal element alloys, characterized by five or more elements mixed in near-equiatomic proportions. These alloys have attracted significant attention due to their unique structures and associated properties. 22 HEAs often form single-phase crystallographically simple structures at elevated temperatures, attributed to their high configurational entropy. 23 However, these alloys exhibit structural degradation during prolonged annealing at lower temperatures, leading to precipitation and phase decomposition, especially at GBs. 24 Fundamental understanding of the structure – chemistry – property relations, especially with respect to the related atomic transport is imperative and the present work reviews the current state-of-the-art.
Intrinsic chemical disorder and the subsequent interfacial roughening pose formidable challenges in elucidating the GB-mediated plasticity in HEAs. The complex energy landscape of the CoCrFeMnNi HEAs, in conjunction with location-specific perturbations across GBs exposed to different environments, was probed by Li et al.
25
Two distinct modes, collective and random, were discovered, and their partitions are dictated by the activation energy window, external mechanical loading, and local compositions. It was highlighted that Fe disproportionately promotes the collective events and facilitates the slip activities near GBs, while Cr atoms suppress the emission of partial dislocations from GBs.
25
These findings imply promising solutions via synergistic combination of microalloying, heat treatment, and mechanical loading to selectively trigger desired plasticity modes at the deformation stage they are required, and hence to achieve an enhanced tunability of HEAs’ mechanical behaviors.
25
The GB diffusion-controlled deformation mode becomes increasingly relevant in HEAs due to their varied diffusion pathways, altered activation energies, and segregation behaviors. Recent studies26–28 suggest that both the chemical complexity and the GB structure in HEAs can retard or accelerate creep depending on composition and thermal treatment, making GB diffusion-related creep a key consideration in alloy design and performance evaluation. The chemical complexity inherent in HEAs opens new and exciting avenues for GB engineering. By leveraging the diverse spectrum of GB characteristics such as GB character distribution, segregation, phase decomposition, and second-phase precipitation, GB engineering can strategically be applied to enhance a wide range of mechanical, thermal, and corrosion properties in advanced alloys. Recent studies exemplify these opportunities. You et al.
29
demonstrated that optimizing the GB character distribution, particularly increasing the fraction of
In this review, the recent advances concerning GB diffusion in HEAs is presented, though the presentation is given within a broader context of multi-component alloys. The primary objectives are to elucidate
the extent of ”sluggishness” of grain boundary diffusion in HEAs; the impact of grain boundary complexions, such as segregation, precipitation, on diffusion compared to diluted and concentrated binary or ternary alloys; GB diffusion in HEAs comparatively to other technologically relevant and multi-component alloys like steel, Incononel and superalloys; the challenges associated with assessing grain boundary diffusion across a diverse spectrum of multi-component and multi-principal element alloys.
In compositionally complex alloys, in the presence of multiple phases, the role of phase boundary diffusion becomes crucial, presenting a significant challenge for the reliable determination of the corresponding diffusion rates. The review concludes with an exploration of the impact of grain boundary diffusion phenomena on the mechanical and physical properties of polycrystalline alloys. First, fundamentals of GB diffusion are introduced, illustrated by the impact of GB bi-crystallography, i.e. the interface’s crystallographic parameters, on GB diffusion in simple metals. Addition of a second element, as a residual impurity or solutes on demand, impacts the GB diffusion transport significantly, as it is shortly explained. Then, GB diffusion in multi-principal element alloys is discussed with respect to the GB structure, temperature, impact of configurational entropy, segregation and precipitation. The peculiarity of interphase boundary diffusion in multi-component alloys is outlined. Although the present review is focused on experimental achievements, the computational assessments of structure, chemistry and diffusion along GBs in HEAs are shortly discussed. Current progress and pending issues towards incorporation of the experimental and modelling GB diffusion data into mesoscale, e.g., phase-field models are outlined. Finally, open problems and challenges are highlighted.
Basics of grain boundary diffusion
Already in the 1920–1930s, the observations of a grain size dependence of thorium losses from tungsten thermionic filaments were qualitatively interpreted in terms of GB diffusion in metallic systems.32,33 Though, the first solid proofs of enhanced GB diffusion were provided by autoradiography.34–36 Progress of serial section techniques, rigorous formulation 37 and subsequent solutions of the GB diffusion problems38–40 together with classification of the diffusion regimes in a polycrystalline material 41 boosted the corresponding research. The initial Fisher model, 37 corresponding to a single homogeneous GB slab within a bicrystalline sample, was later developed accounting for a polycrystalline structure of typical samples 42 or specific cases of cylindrical 43 or cubic 39 grains. The original Harrison classification of the diffusion regime in a homogeneous and isotropic polycrystal, i.e. in the case when all GBs feature the same direction-independent diffusion coefficients, was later extended to the real microstructures accounting for a hierarchy of the short-circuit diffusion paths created by internal interfaces44–47 or due to the presence of dislocations in a polycrystal.48,49
Early overviews of GB diffusion in metals were done, e.g., by Peterson 50 or especially with a focus on GB diffusion mechanisms by Balluffi and Mehl. 51 A generalized model which includes the existence of grain boundaries, dislocations, triple junctions in a polycrystalline sample was recently elaborated and analyzed. 52 Recent reviews with an emphasis on the unresolved issues and problems can be found in Refs. Mishin and Herzig, 53 Herzig and Mishin, 54 Divinski and Bokstein, 55 Divinski. 56 In order to document the progress, in Table 1, the main problems/issues mentioned in those reviews are listed and the current progress is commented.
Note that diffusion in multi-component and especially in multi-principal element alloys was not mentioned that time. The present review documents significant progress, especially for pure metals and binary alloys, and some issues can be considered as resolved now (some of this progress relevant to GB diffusion in multicomponent alloys will be documented here). However, the list of open problems is modified, but does not become shorter.
Nowadays, large-scale MD simulations provide direct atomistic insights into enhanced GB diffusion in different classes of materials.58–67 Furthermore, in-situ TEM investigations provided direct observations of enhanced atomic transport within the grain boundaries. 68 Though, this is not a general statement, as it was found, e.g., that Au GB diffusion in polycrystalline Si is slower than the corresponding bulk diffusivity.69,70
The fundamentals of GB diffusion in bi- and poly-crystalline materials can be found in the famous book of Kaur, Mishin and Gust
4
and subsequent developments with respect to GB diffusion in materials with a focus on different hierarchies of the short-circuit diffusion paths For example, the following chains of microstructure elements, like dislocations
Grain boundary bicrystallography
A plain grain boundary, as an interface between two crystals, is characterized by the following macroscopic and microscopic parameters, see Figure 1:
Matrix of rotation between the two crystalline coordinate systems, for instance, the axis GB plane inclination with respect to a chosen direction in a particular grain, Vector of atomic translation of one grain with respect to another,
The set of parameters
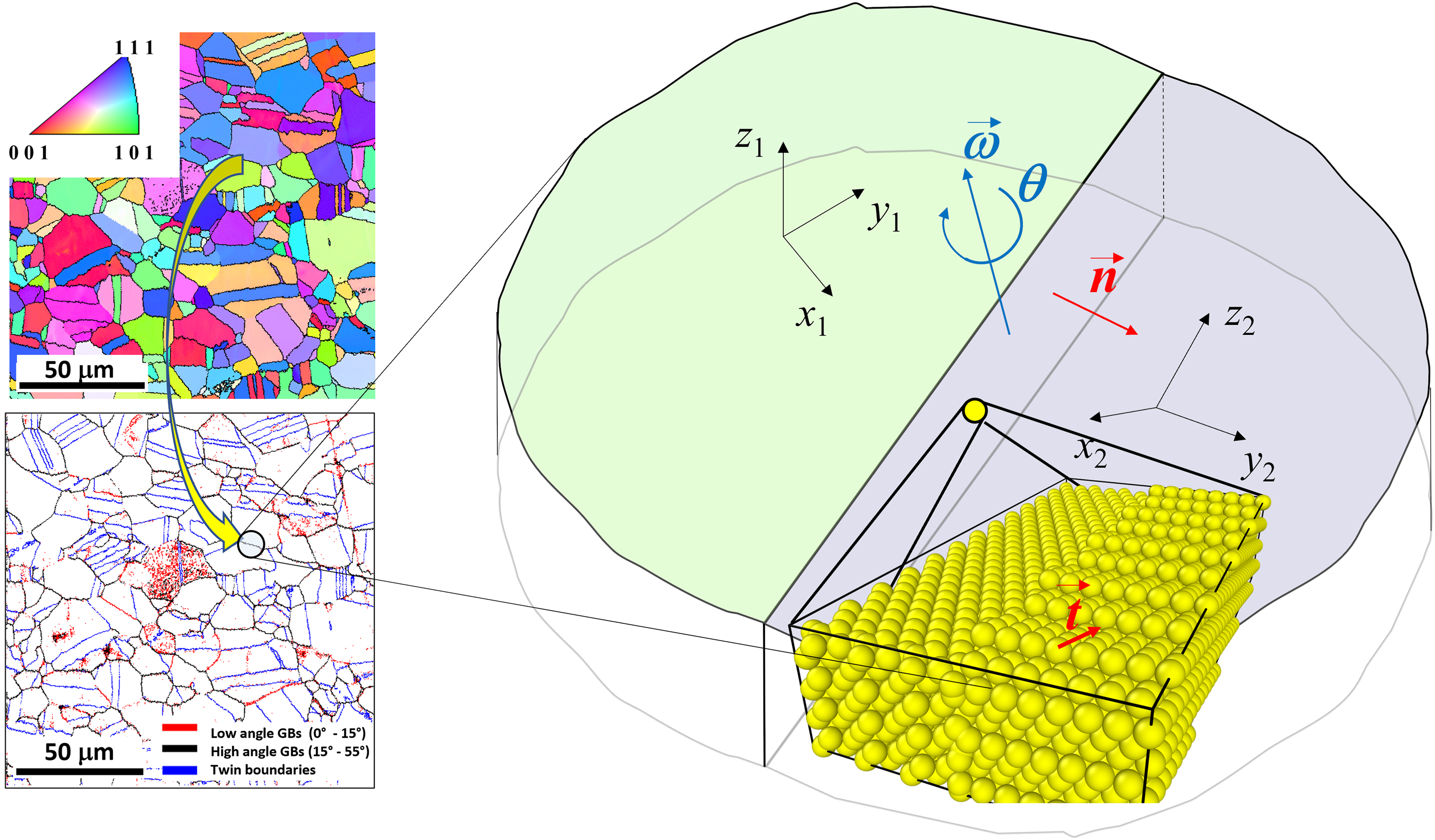
An example of electron back-scatter diffraction (EBSD) analysis (top-left panel) of microstructure, corresponding GB character distribution (bottom-left), and the GB bi-crystallography for arbitrary selected flat segment of a high-angle grain boundary (right panel): two neighboring grains, the atomic positions in which are defined by the two (orthogonal for cubic grains) coordinate systems
In dependence on the given set of the parameters Low-angle grain boundaries with High-angle GBs with Special (low sigma) GBs with (specific) values of Pure tilt GBs with Pure twist GBs with GBs of a mixed type; Symmetric and asymmetric GBs.
Generally, GB structures, GB energy, and GB diffusivity depend on all these parameters, but in a nontrivial way.81–83 Since the number of parameters is large, already the presentation of the GB properties of polycrystalline materials as a function of these five parameters is nontrivial.84,85
Measurements and analysis of grain boundary diffusion
Tracer sectioning experiments
Typical GB diffusion experiments are destructive, performed using suitable tracer elements (radioisotopes), and consist of following main steps (for further details see, e.g.,Gärtner et al.
86
):
sample preparation and characterization; microstructure equilibration (annealing at the temperatures of the intended GB diffusion experiments for at least double duration); plane-parallel polishing; proper annealing for stress removing; tracer deposition and diffusion annealing treatment; reduction of the sample diameter to remove potential artifacts of surface/lateral diffusion; plane parallel sectioning and measurements of radioactivity of all removed sections (in some cases, measurements of the residual activity of the ramaining sample); determination of the tracer penetration profile in terms of the dependence of the layer tracer concentration against the panetration depth.
As a result, the information about the lateral (within the sections) distribution of the diffused atoms is lost and a proper microstructure characterization is imperative to provide reliable data.
Analysis of a GB diffusion experiment relies largely on the commonly accepted Fisher model
37
with fast GB diffusion,
The exact solutions of the GB diffusion problem were derived by Whipple 38 and Suzuoka 39 for constant and instantaneous source conditions, respectively, and Le Claire generalized them providing practical numerical approximations feasible for the analysis of the tracer diffusion measurements. 40 For further details, the reader is referred to the original papers38–40 or to the textbooks.4,72
Harrison
41
provided a classification of the kinetic regimes of GB diffusion in a polycrystalline material with one type of short-circuit diffusion paths, e.g., grain boundaries featuring the same kinetics (the GB diffusion coefficient In the A-type regime, the volume diffusion length, In the B-type regime, GB diffusion dominates while the diffusion fluxes from opposite grain boundaries do not overlap, satisfying the condition The C-type regime refers to low temperatures,
The Le Claire parameters 
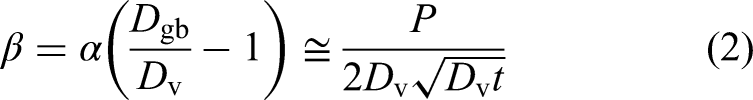
Recently, the original Harrison classification was extended defining transition kinetic regimes, namely the AB and BC ones.44,71,87,88 For further details, the Reader is referred to the textbook. 72
It is important to underline that the GB width,
Using this combination of B- and C-type GB diffusion measurements for selected solutes, the solute segregation factor – typically corresponding to the dilute, Henry-type segregation regime – can be determined as
Typically, GB diffusion measurements are performed in polycrystalline samples with stable and coarse microstructures and the Fisher approximation, i.e. well separated GBs perpendicular to the sample surface, is reliable if the GB diffusion depth is reasonably smaller than the grain size. The analysis of Levine and MacCallum verified the 6/5th power law for the tracer distribution in a GB diffusion experiment for a polycrystalline sample with polygon-shaped grains in the B-type kinetics regime.
42
However, the determined tracer diffusion coefficient would be overestimated by a factor of 1.5 to 2 if the GB diffusion length would be significantly larger than the grain size.
42
Nevertheless, a systematic and consistent analysis of the diffusion data in terms of the same model for the whole dataset will provide correct estimations of the diffusion activation enthalpy. Note that the power 6/5 for the depth dependence of the tracer distribution in the B-type kinetic regime follows from the numerical analysis of Le Claire
40
and this is an approximation only, though a very good approximation, see Kaur et al.
4
for a full analysis. In fact, the corresponding slope of the GB diffusion related branch depends on the value of
Other methods of grain boundary diffusion measurements
Autoradiography is one of the classical radiotracer-based techniques, in which radioactive isotopes are deposited on the surface of a specimen and allowed to diffuse during annealing. After the diffusion annealing, the sample is cross-sectioned and the section is placed in contact with a photographic emulsion or X-ray film, where the emitted radiation generates an image of the tracer distribution. In this way, the diffusion paths can be directly visualized on the X-ray film. The resulting autoradiographic images are often analyzed using a Joyce–Loebl isodensitracer to produce accurate two-dimensional density maps of the grain boundary region, sufficient for the analysis of grain boundary diffusion. Renouf et al. 36 extensively employed this technique to investigate grain boundary diffusion in Cu bicrystals.
While radiotracer methods employing radioactive isotopes are widely used for GB diffusion measurements, several alternative techniques are available that enable such measurements without the use of radioisotopes. These methods are focused almost exclusively on impurity diffusion due to requirement to differentiate the diffusing atoms and the matrix ones. (In this respect, the radiotracer technique is superior, especially for GB self-diffusion.) One such technique is secondary ion mass spectrometry (SIMS), which can effectively detect impurity elements or (highly enriched) stable isotopes with reasonably low natural abundance (below 1%). SIMS provides the depth profiling of diffusion-annealed samples or maps distribution on a prepared cross section, thereby generating penetration profiles of the deposited tracer film. Owing to its high sensitivity and spatial resolution, SIMS is frequently employed to investigate diffusion behavior and interfacial reactions in advanced alloys as well as in semiconductor thin films.
Schwarz et al.
99
investigated GB diffusion in the Cu–Ni system using the SIMS technique. With the high spatial resolution of the SIMS technique, diffusion along GBs of different character was investigated, that is, high-angle, low-angle, and coincidence site lattice boundaries. Further, the Arrhenius parameters of GB diffusion (the pre-factor
Advanced characterization techniques such as analytical transmission electron microscopy (TEM), especially scanning TEM (STEM) have already provided valuable insights into GB diffusion in materials. Schweizer et al.
68
used in-situ STEM to follow jumps of single atoms within a GB and Yang et al.
104
analyzed Ti GB diffusion in Al
Chellali et al.
106
studied the atomic transport along triple junctions in nanocrystalline Cu by the atom probe tomography (APT) and demonstrated that the diffusion of Ni along triple junctions is 100 to 300 times faster than in the high-angle grain boundaries. Similarly, Schwarz et al.
107
used APT to investigate the diffusion behavior of Al in an additively manufactured Mg–Al (AZ111) alloy. With the use of an advanced atom probe tomography technique, the precise detection of the concentration gradient of Al across the grain boundaries was measured. Interestingly, Al was found to diffuse outward during oxidation and incorporated as a surface layer of Mg oxide/hydroxide instead of forming the protective Al
In cases where the concentrations of diffusant are relatively high (several at.%), concentration profiles can also be measured using an electron probe microanalyzer (EPMA), which enables the determination of solute diffusion along individual grain boundaries. However, the spatial resolution of EPMA is limited to approximately 1
Temperature dependence of grain boundary diffusion
As it was stated, a combination of B-type and C-type diffusion measurements for the same solute in the same matrix provides access to the pertinent segregation factor. In Figure 2(a), the results of GB diffusion measurements with different solutes in the same high-purity Cu are presented. By plotting both, the triple products
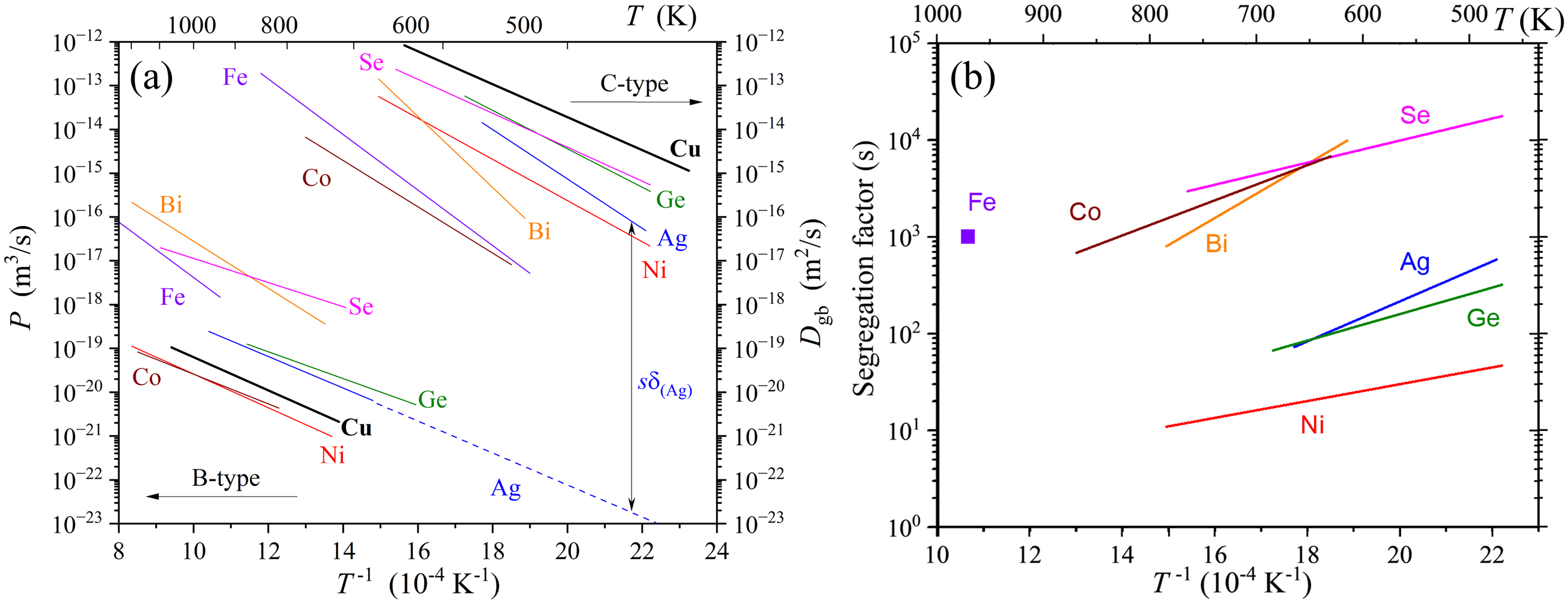
(a) Arrhenius diagram for GB diffusion of different elements in the same pure Cu (99.9998 wt.% purity): Cu (thick black lines
111
), Ni (red lines
112
), Co (wine lines
98
), Ag (blue lines
95
), Bi (orange lines
96
), Se (magenta lines
113
), Ge (green lines
114
), and Fe (violet lines
115
). The results of the B-type (
Assuming that the GB width is the same for self- and solute diffusion, i.e.
One may argue that the GB width
The scatter of experimental estimates of
Dedicated molecular dynamics simulations with empirical potentials showed only minor changes in
The diffusional GB width for impurities (solutes) is not accessible experimentally, since only the product
Independent measurements of the segregation factor
We conclude that
Misorientation and inclination dependencies of grain boundary diffusion
The influence of misorientation on GB diffusion was intensively studied, e.g., by Li and Chou
126
for Cr diffusion in
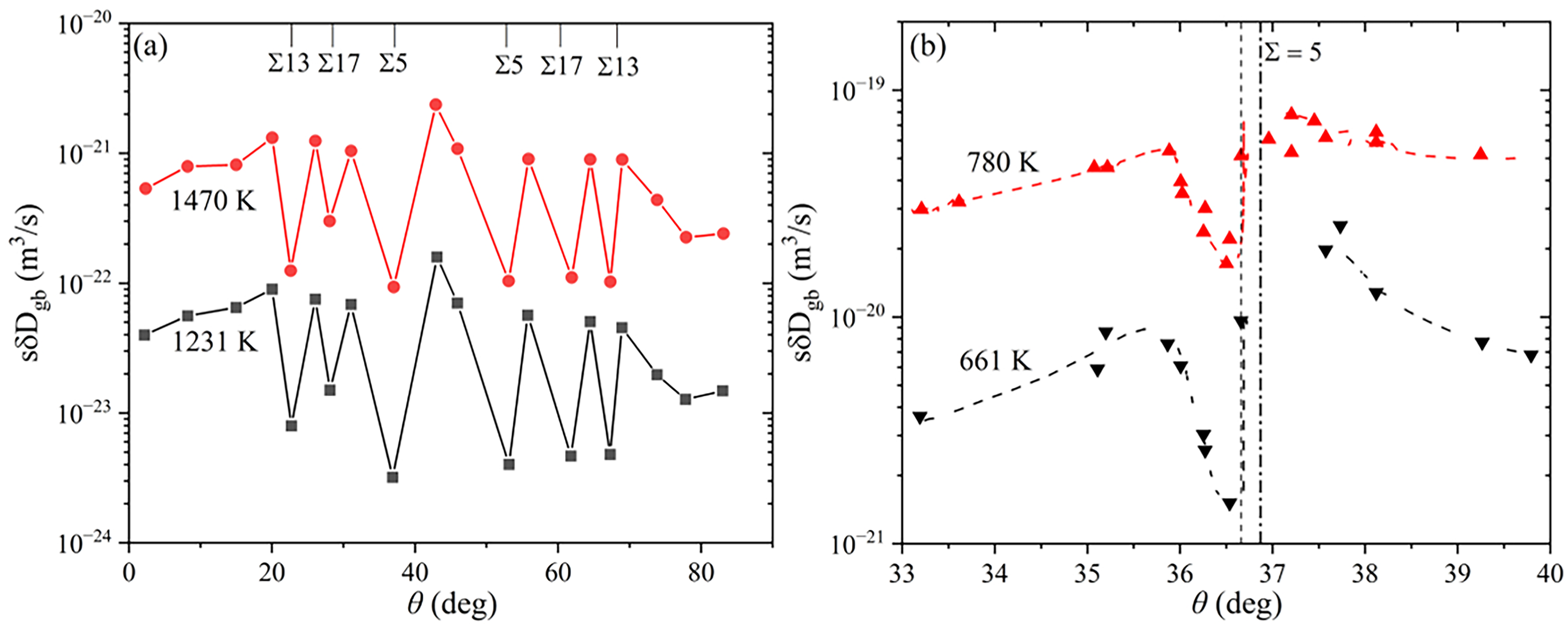
Detailed and still state-of-the-art measurements on the orientation dependence of GB diffusion were performed by Budke et al.
129
for the case of Au diffusion in Cu near
GB inclination,
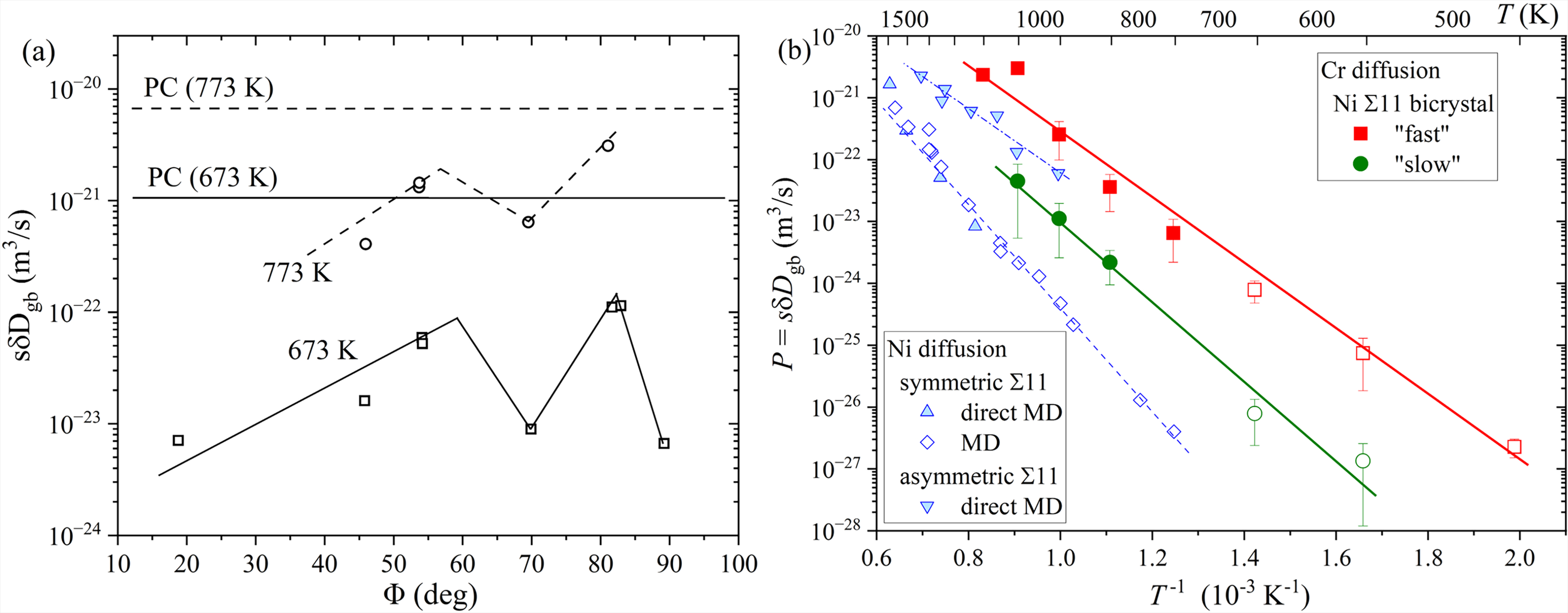
(a) The triple product
The impact of GB inclination of the GB diffusion rates was further investigated for Cr diffusion in Ni
One may further suggest that the GB defects might be responsible for the controversial results of the GB diffusion
131
and GB energy
133
measurements as function of inclination for the
Effect of material’s purity
The influence of material purity on GB self-diffusion was carefully investigated by Surholt et al. 111 for copper. Two, in fact high-purity, copper materials were investigated, namely with the purities 5N and 5N8, Figure 5(a). The authors concluded that the presence of about 1 at.ppm of sulfur in coarse-grained copper significantly retards GB self-diffusion and increases the corresponding activation enthalpy.
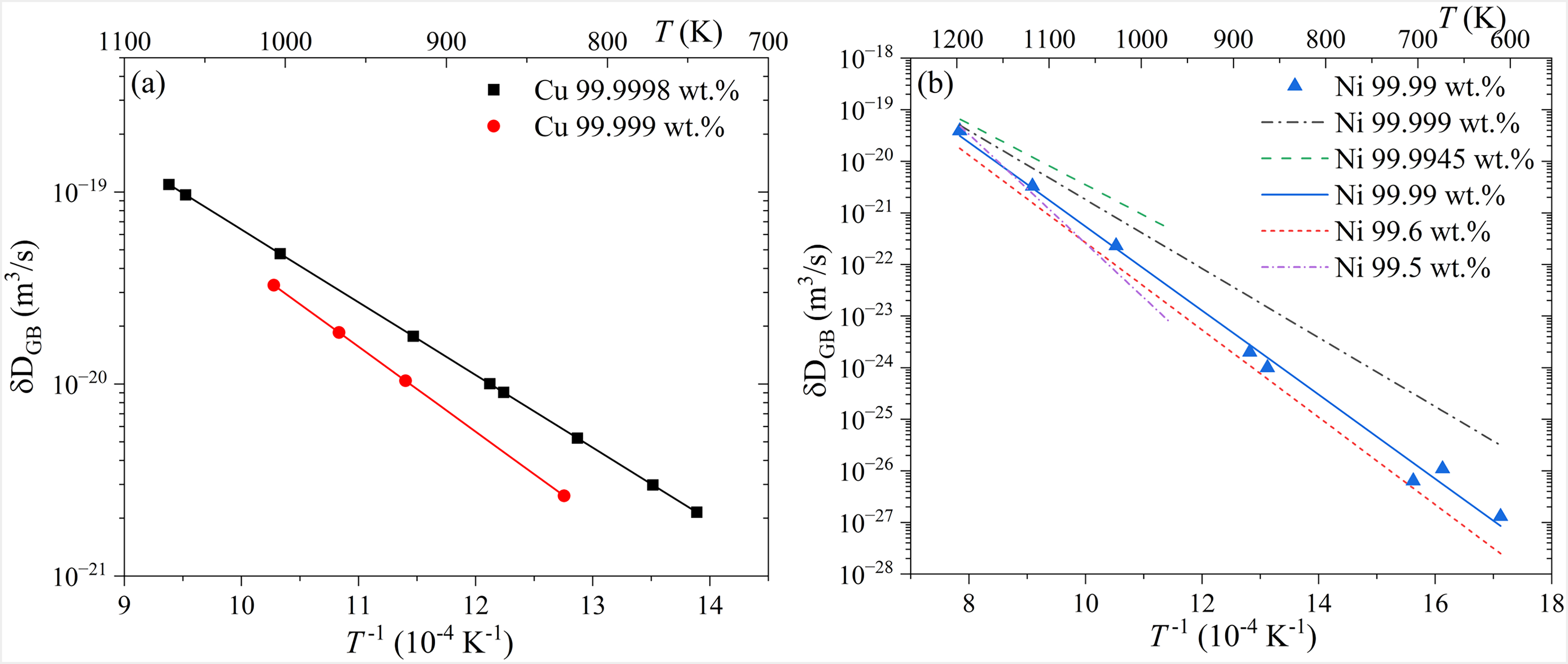
Impact of matrix purity of GB self-diffusion in Cu, after Surholt et al. 111 (a) and in Ni, after Prokoshkina et al. 92 For a reference to the original publications on Ni of different purity levels see Ref. Prokoshkina et al. 92 In (b), the Ni GB self-diffusion data from Refs. Divinski et al., 91 Prokoshkina et al., 92 Rothova and Cermak 139 are collected. (The figures are reproduced from Prokoshkina et al., 92 Surholt and Herzig 111 with permission from Elsevier)
The impact of purity on GB self-diffusion in Ni was studied more extensively,91,92,139 see Figure 5(b). Furthermore, GB width and GB energy as functions of the material purity were investigated by GB self-diffusion measurements in Ni. 92 Thorough analyses of all available results on the GB width in different coarse-grained and nanograined materials suggested that the diffusional GB width is independent of temperature, material purity and material nature and can be taken as 0.5 nm in well-annealed materials, see Figure 6(a). Grain boundary self-diffusion strongly depends on the amount of residual impurities, and the effect is marginal for less pure materials at low temperatures in Ni of the indicated purity levels.
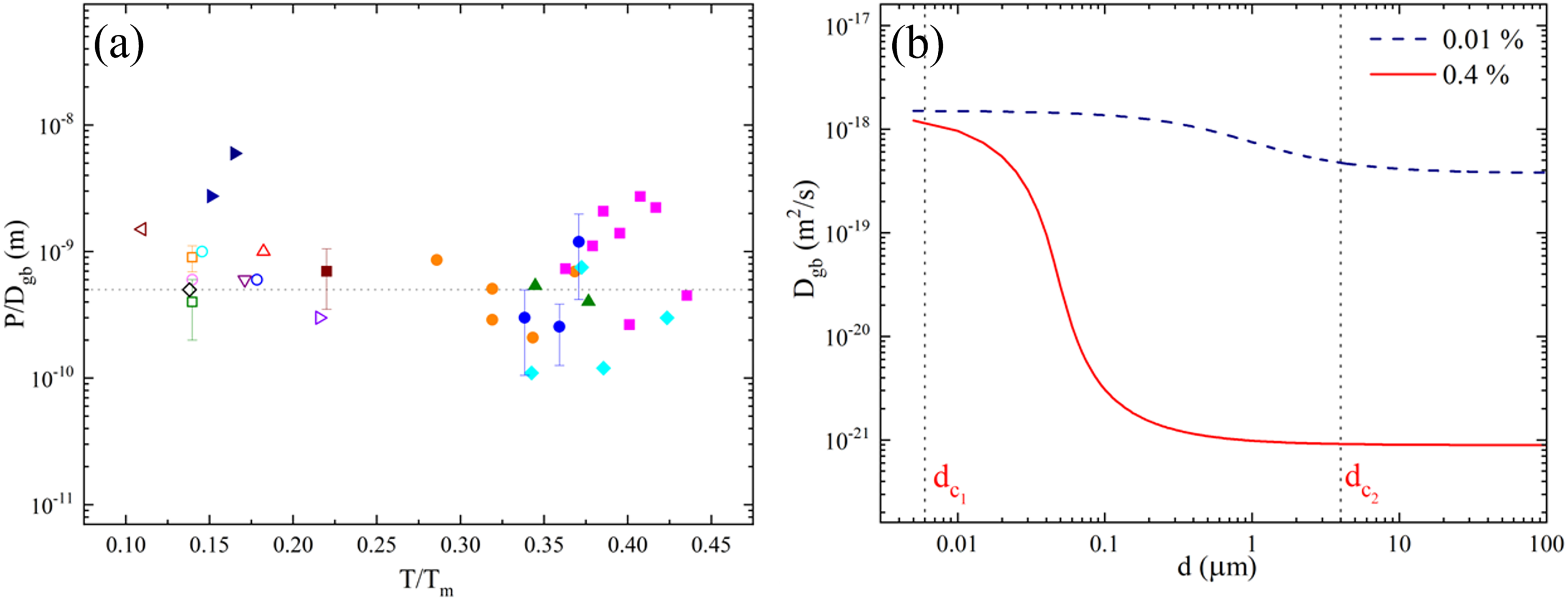
(a) GB width as determined by GB self-diffusion measurements (filled symbols),
The effective dependence of the GB energy and the self-diffusion coefficient on grain size in a material of a given purity level can be evaluated using a model proposed by Prokoshkina et al.,
92
see Figure 6(b). The dependence is monotonic, but highly non-linear. In fact, two critical grain sizes,
Grain boundary structure transitions
GB diffusion measurements across an extended temperature interval were shown to provide a unique access to the solute segregation factor in the matrix, Section “Temperature dependence of grain boundary diffusion”. Though, the main pre-requisite of the corresponding analysis is that the atomistic structure of the grain boundary remains largely unchanged at different temperatures. Linear temperature dependencies in the Arrhenius coordinates were thought to support such an analysis. 72 Deviations of the Arrhenius-type temperature dependencies provide hints towards the evolution of the GB structure.
Recently, GB diffusion of Ag in a Cu
Being motivated by those findings, atomistic computer simulations discovered phase transformations in high-angle GBs in metals.
141
Subsequent atomistic simulations of Ag diffusion and segregation in two different structural phases of the Cu
Those studies initiated intensive research revealing a large spectrum of GB phase transitions.9–11 Choi and Brink 145 motivated that well-known faceting transitions can be considered as GB phase transitions. Grain boundary phase diagrams were proposed to be constructed.146–148 Moreover, machine learning algorithms were combined with atomistic simulations to predict GB properties as functions of the five macroscopic degrees of freedom of GBs plus temperature and composition for a binary alloy in a 7-D space. 148 The concept is further extended to account for defect phase diagrams 135 which describe the coexistence and transitions of general defect phases (dislocations, stacking faults, GBs, triple junctions, etc).
General aspects of grain boundary diffusion in binary alloys
Addition of a second element, either wanted (as a solute) or unwanted (as a residual impurity), modifies GB diffusion. The related aspects were already spotlighted in Section “Effect of material’s purity” with a dedicated discussion on the impact of material’s purity, Figure 5. Here the discussion will be extended towards binary dilute and afterwards concentrated alloys and intermetallics. Selected studies on high-purity copper are presented in Table 2, while binary, ternary and multi-component alloys are listed in Table 3, along with comments on the most important findings.
Arrhenius parameters for GB diffusion of different elements in high-purity 5N8 copper measured in the B- and C-type kinetic regimes and the estimated segregation behavior in the dilute limit assuming
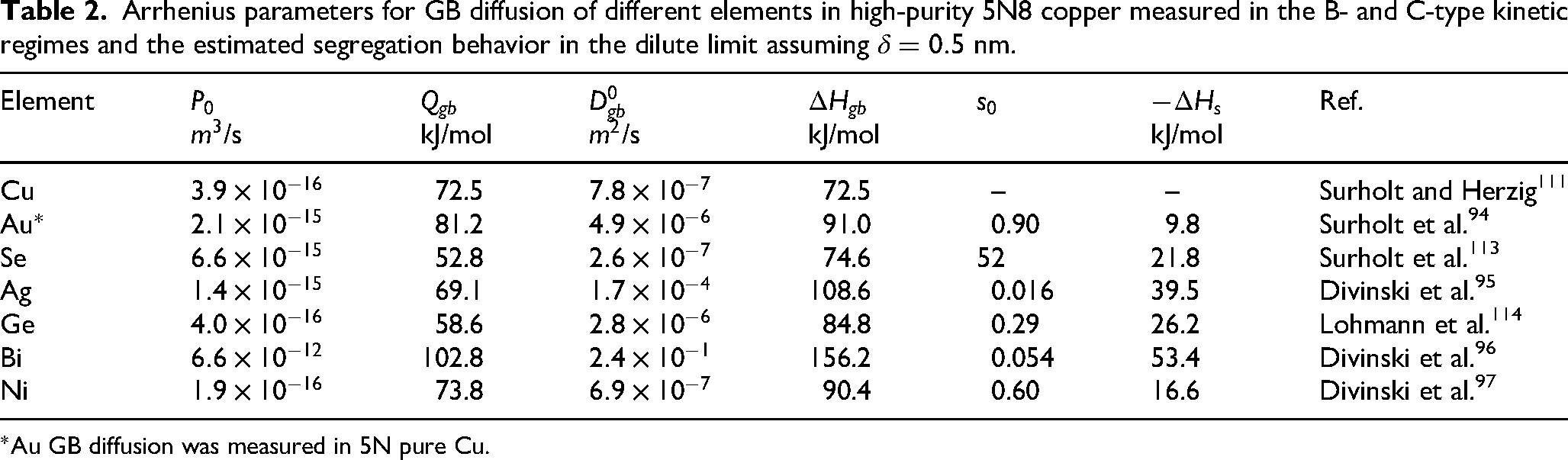
Experimental investigations of GB diffusion using radiotracer method in selected binary, ternary and multi-component alloys available so far in literature. ECAP

Dilute Alloys
In the past, GB diffusion in binary alloys has been studied extensively, e.g. by the group of Gas and Bernardini for GB diffusion in the Ag-Sn, Fe–Sn, Fe–Sb alloys.149–151 Selected studies are listed in Table 3. Alloying of Ag by Sn up to a concentration of 0.2at.% was found to increase the triple products of Ag and Sn GB diffusion by a factor of three.
149
Further alloying with Sn does not practically change the triple product of Ag, but that of Sn decreases considerably, approaching a value of about 50% of that of pure Ag.
149
These tendencies were established for the triple product,
Cu alloys also received a particular attention due to their widespread industrial applications and well-understood crystal structure.152,153 Moreover, GB diffusion of different solutes in the same high-purity copper was measured in the C-type kinetic regime over the years.94,95,97,98,111,113,114,140,154 The Arrhenius parameters of GB diffusion in both B- and C-type kinetic regimes are summarized in Table 2 approximating the measured temperature dependencies as
In fact, independent measurements of the segregation factor
Commonly, the atomic size effect (elastic mismatch) was identified as the main driving force for solute segregation, e.g., for Bi in Cu. 160 However, electronic (chemical) and configurational entropy effects cannot be excluded, too, as it was found to explain the segregation anisotropy in Pt–Au alloys. 161 Gibson and Schuh 162 proposed simple bond-breaking arguments to clarify the variations in the solute-induced changes in boundary cohesion. Although simple Miedema-like models 163 are attractive,164,165 a whole spectrum of computational modeling techniques (density-functional theory, MD simulation, mesoscale phase-field, continuum defect theory, etc.) has to be used as complementary tools for analysis of GB segregation. 166
GB diffusion was found to be influenced by various factors such as the nature of the alloying element, their segregation behavior, induced phase transitions and microstructural features.
As Figure 5(a) suggests, the addition of already 1 ppm of sulfur retards Cu GB self-diffusion dramatically. Similarly, segregation of phosphorus or sulphur hinders/reduces the GB self-diffusion in Fe 49 or Ni.91,92,183 Moreover, in high-purity copper the segregation of sulfur resulted in reduced grain boundary diffusion rates of Ni. 184 Bernardini and Gas 185 introduced the so-called “poisoning effect” to explain the strong impact of the segregated impurity. According to their model, the segregated atoms ”poison” neighboring sites and consequently reduce GB diffusivity. 185
In the Cu-Ag system,
167
Divinski et al. reported that the addition of 0.2 at% Ag shows almost no change of the Ag GB diffusion coefficient,
Prokoshkina et al.
154
investigated the impact of alloying on GB diffusion in Cu–Fe alloys. In Figure 7, the determined
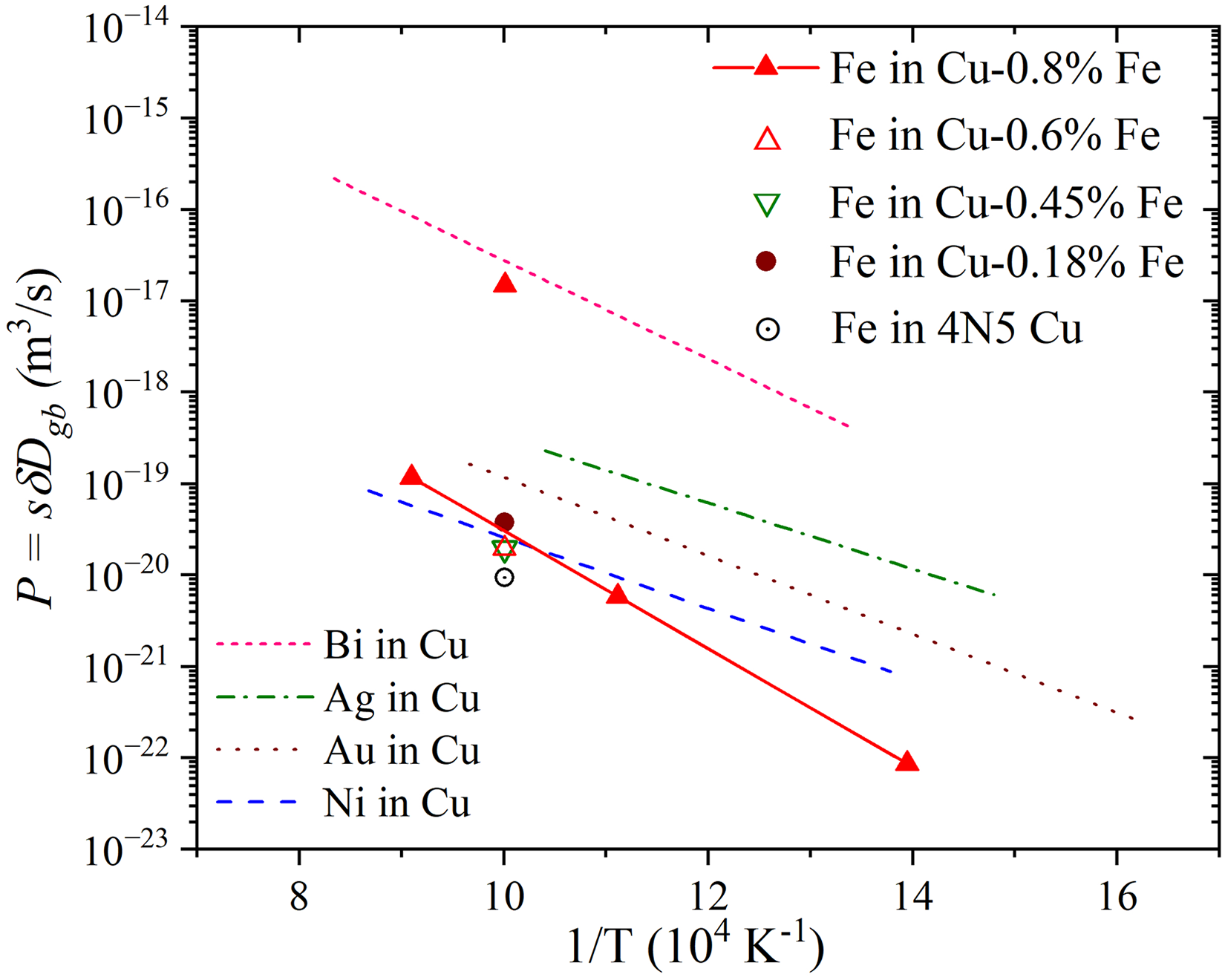
Phase transitions critically influence GB diffusion in binary dilute alloys by modifying GB structure, segregation patterns and the diffusion rates. In Cu-Bi alloys, 170 the GB pre-melting (pre-wetting) transition causes formation of liquid-like films at GBs, dramatically increasing both Cu (host) and Bi (solute) diffusivities. The critical Bi concentration, corresponding to an abrupt jump in the GB diffusivities, depends on temperature and signifies the onset of pre-wetting. Experiments at 1093 K and 1116 K show complete GB wetting by a liquid phase, as these temperatures exceed the GB wetting transition temperature of 820 K. The presence of liquid-like films enhances atomic mobility, though a strong heterogeneity of Bi coverage was recorded. In fact, the coexistence of both high-angle GBs with a high Bi coverage (high values of the triple products) and high-angle GBs with low Bi segregation and lower mobilities was reported. These findings were supported by recent measurements of Ag GB diffusion in the same Cu-Bi alloys. 187
A strong interplay between the segregation patterns, evolving as grain boundary phase transitions (or complexions), and the diffusion behavior was found for the the Ni–Bi system.
116
The determined grain boundary diffusion map is shown in Figure 8, which illustrates how the changes in Bi concentration influence the diffusion rates through a sequence of distinct GB phase transitions. Three characteristic regions are observed, depending on the Bi concentration and temperature:
116
Region I with a slight enhancement in Ni diffusion due to Bi segregation at the grain boundary; Region II with a significant increase in Ni diffusivity associated with multi-layer Bi segregation; and Region III revealing the highest Ni diffusivity values corresponding to the formation of liquid-like Bi layers at the grain boundaries.
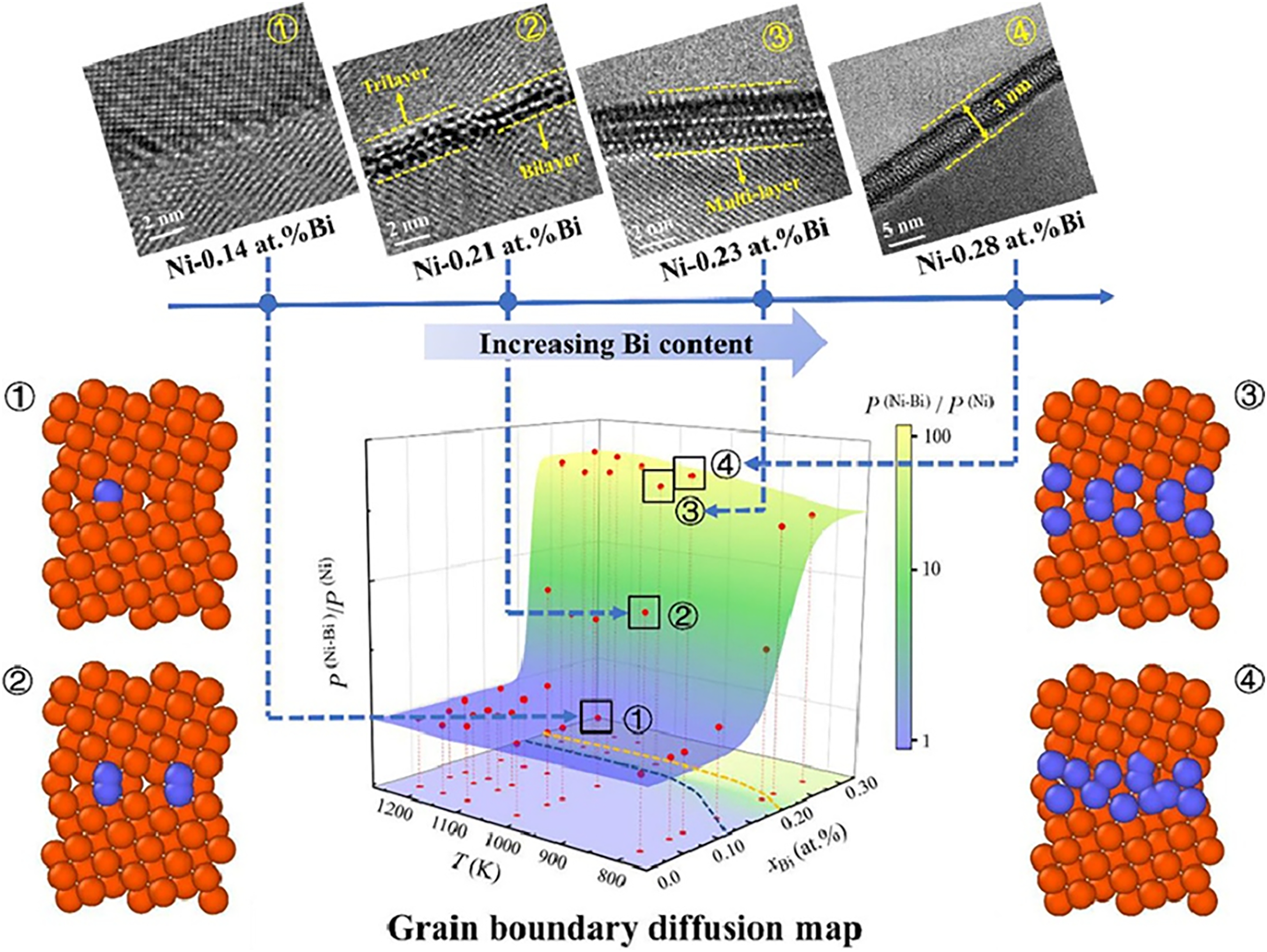
Schematic of a grain boundary diffusion map of the Ni–Bi system.
116
The normalized triple product of Ni GB diffusion in Ni–Bi alloys,
The Ni-Bi system shows also a considerable heterogeneity in Bi segregation between general high-angle grain boundaries. 116 This heterogeneity implies that not all GBs behave uniformly, with local variations in structure and composition leading to significant differences in diffusion behavior. Moreover, the measured grain boundary energies correlate well with the observed diffusion trends, with abrupt energy increases aligning with structural transitions from segregated layers to liquid-like GB films. 116
In the Fe–Sn system, 150 a contrasting behavior in comparison to that depicted for Ni–Bi was established via detailed diffusion measurements. Sn acts as a fast diffuser in bulk Fe but it significantly slows down at GBs due to segregation effects. 150 Sn GB segregation reduces the diffusivities of both Fe and Sn, sometimes by more than an order of magnitude. These results support Borisov’s theory (1964), 188 which connects the GB energy reductions caused by segregation to changes in the diffusion behavior. The study highlights the necessity for incorporating segregation factors when analyzing GB diffusion in such systems, especially if the measurements are performed at higher temperatures under the B-type kinetic regime conditions.
Concentrated alloys – disordered solid solutions
GB diffusion of the constituent elements (Fe, Ni) and of a solute (Ag) was systematically studied in a nanocrystalline
In fact, the hierarchy of internal short-circuits in the nano-crystalline Fe-40at.%Ni alloy was theoretically discovered by analyzing the whole amount of the diffusion data, both as penetration profiles and as Arrhenius plots.44,46 The key point was that the B-type profiles (
Despite the different mathematical treatments required for each combination of the kinetic regimes, the derived diffusion characteristics remained consistent and reliable.46,189,190 A key finding was that the diffusion rates along the inter-agglomerate boundaries were significantly enhanced, by several orders of magnitude, with respect to those along the nanocrystalline grain boundaries. Additionally, the activation enthalpy for inter-agglomerate boundary diffusion was notably lower than that for nanocrystalline GB diffusion. Specifically, for nanocrystalline GBs, the activation enthalpies were 189 kJ/mol for Fe, 177 kJ/mol for Ni, and 173 kJ/mol for Ag. In contrast, for inter-agglomerate boundaries, the activation enthalpies were significantly lower: 148 kJ/mol for Fe, 134 kJ/mol for Ni, and 91 kJ/mol for Ag. These values clearly highlight the more facile diffusion along inter-agglomerate boundaries.
Another important conclusion was that the GB diffusivities in nanocrystalline (grain size of about 100 nm) and coarse-grained (grain size of about 0.5 mm)
The diffusivities of Fe, Ni, and Ag along the nanocrystalline GBs in the
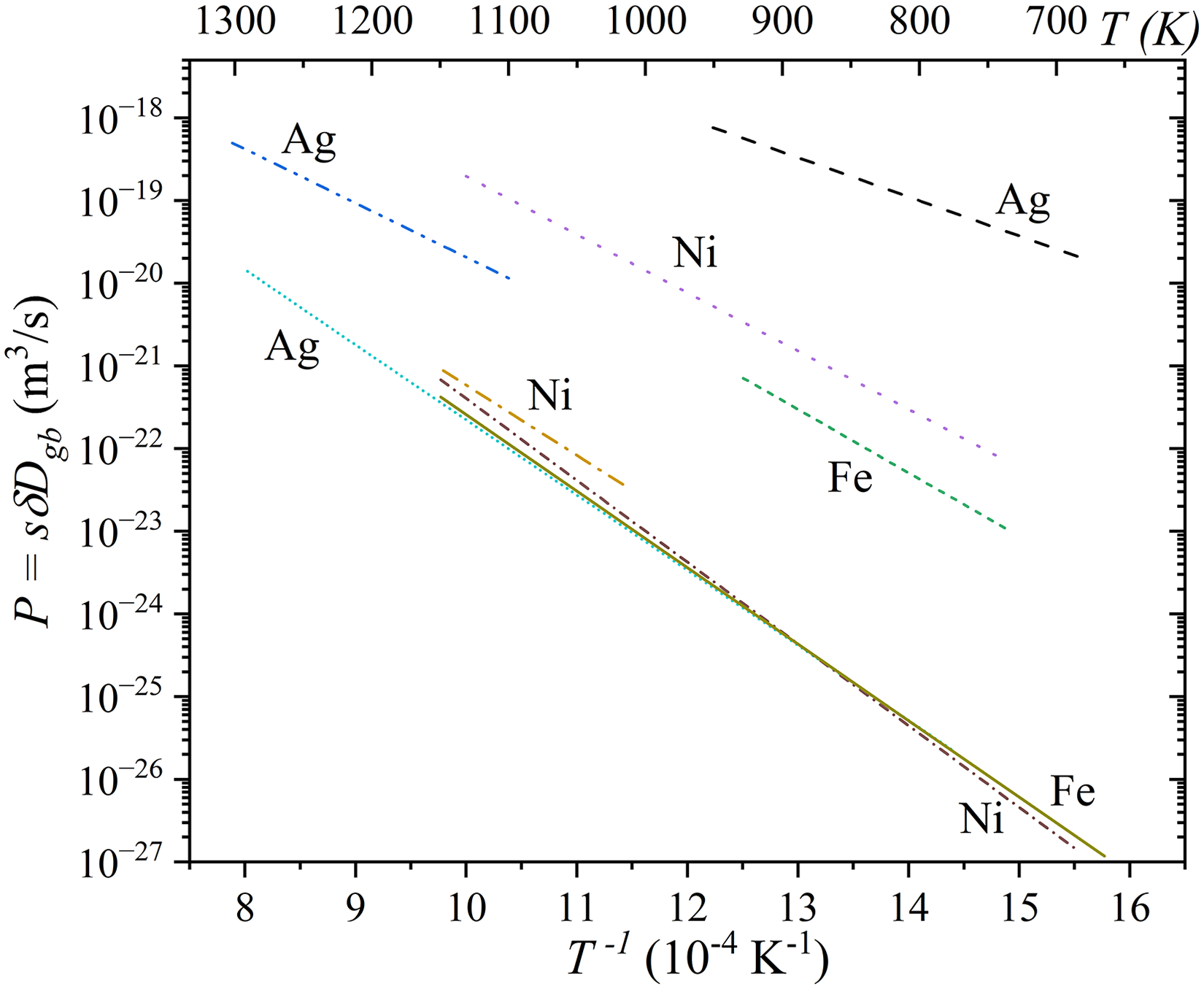
Arrhenius dependencies reported for Ag, Fe, and Ni diffusion in nanocrystalline GBs (solid lines) and inter-agglomerate boundaries (dashed lines) of nano
In the Ni–Cr alloy system, Moulin et al.
171
extensively investigated the effect of the carbon content on GB diffusion. Their study revealed that while volume diffusion remains practically unaffected, the GB diffusion coefficients increase with the carbon precipitation at grain boundaries, which also raises the activation energy for both Ni and Cr diffusion. The interaction between carbon and chromium atoms within the grain boundaries was advocated to explain the effect rather than segregation.
171
Moreover, dissolved carbon does not alter GB diffusion in Ni, but significantly affects the frequency pre-factor,
Ordered intermetallic compounds and the impact of the order-disorder transitions
Significant advancements have been made in understanding GB diffusion in intermetallic compounds; however, its kinetics remains less explored compared to bulk diffusion. Experimental investigations conducted over the years were primarily focused on Ni, Ti, and Fe-based aluminides, revealing distinct diffusion behaviour influenced by composition and atomic ordering. For example, Frank et al.
193
reported that in L1
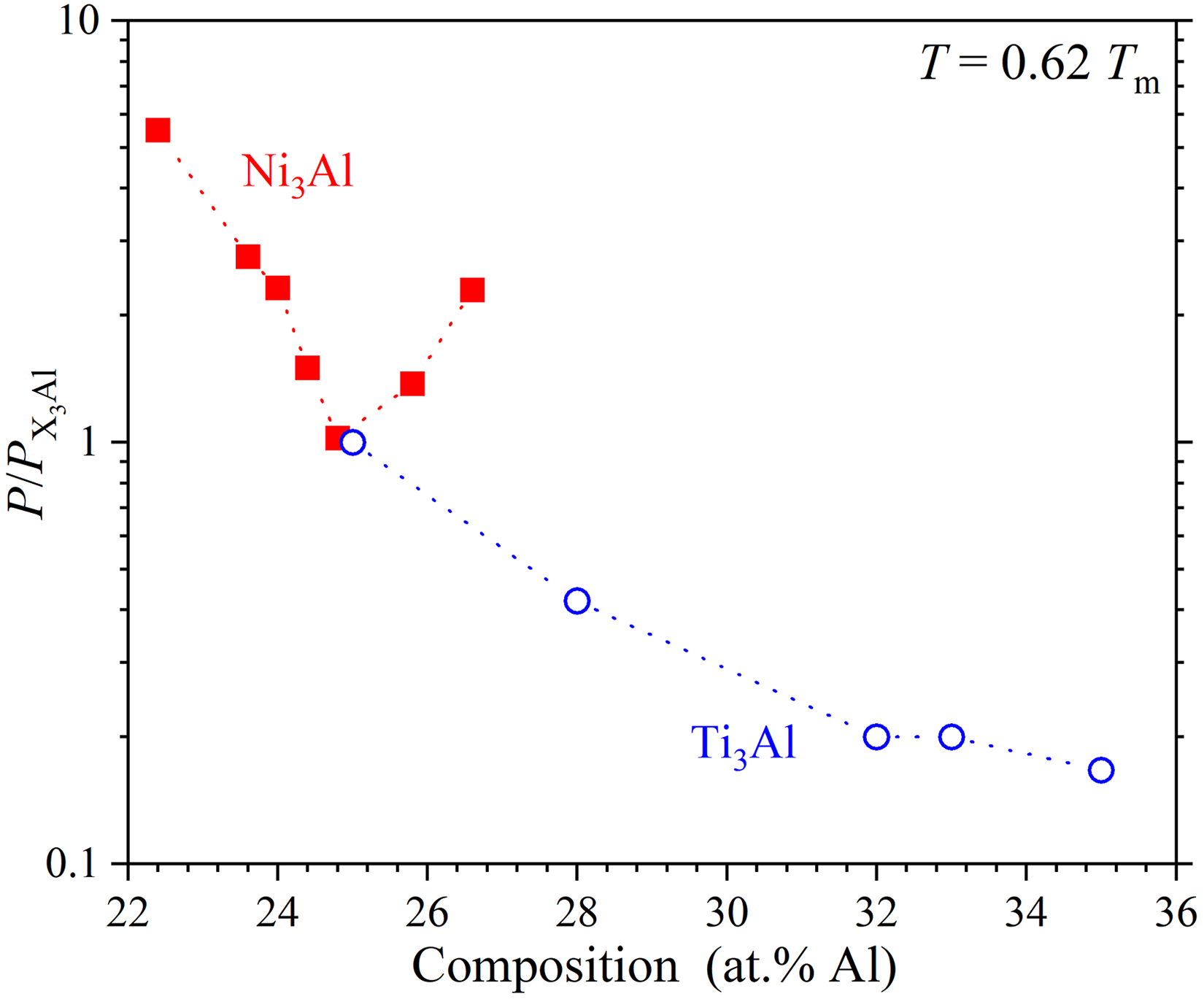
Composition dependence of the normalized GB diffusion rate of the majority component,
Rothova and Cermak
139
used Ga tracer to mimick Al diffusion in Ni
Ti GB diffusion was extensively measured for Ti aluminides. For example, Ti GB diffusion in L1
The impact of atomic ordering on GB diffusion has been highlighted in several studies. Tokei and coworkers
172
discussed that order-disorder transitions, such as B2–DO
Typically, atomic ordering results in a reduction of the triple products below the order-disorder transition temperature. However, the studies emphasized that segregation can induce significant shifts of the transition temperatures between GBs and the bulk.
172
Generally, intermetallic compounds features higher ratios of GB-to-bulk diffusion activation energies,
In the case of B2-ordered NiTi alloys, 173 GB diffusion of both Ti and Ni reveal strong deviations from linear Arrhenius-type temperature dependencies, Figure 11. Such deviations were suggested to be induced by structural modifications at the grain boundaries, potentially driven by precipitates’ formation and their evolution along the GBs. 173 In particular, transitions from coherent (GB diffusion contribution marked as NiTi-I in Figure 11) to incoherent (GB diffusion contribution marked as NiTi-II in Figure 11) precipitation with increasing temperature turned out to influence the diffusion rates by altering the GB structures and atomic segregation. 173 Nonlinearities in GB diffusion induced by coherency loss during precipitate growth were reported for Al-based alloys. 182
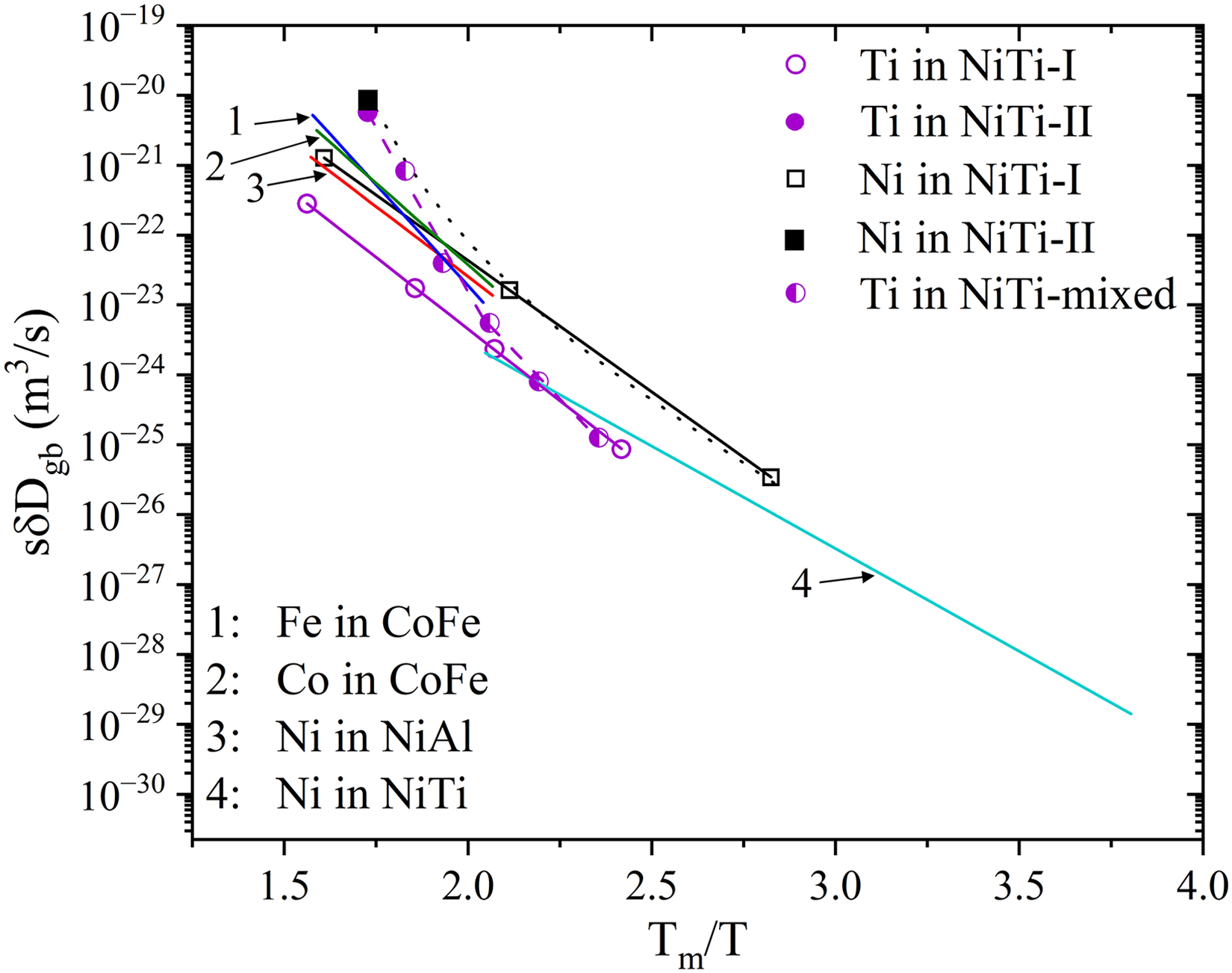
Arrhenius plots for GB triple products
Thus, GB diffusion in binary alloys is significantly affected by different factors, including solute segregation, phase transitions, and microstructure transformations. The presence of a second element, whether as an alloying addition or an impurity, alters the diffusion behavior by modifying the atomic mobilities. In intermetallic compounds, the ordering influences further the diffusion kinetics. Due to segregation, the order-disorder transition at GBs differs from that in the crystalline bulk. These effects become even more complex in multi-component systems due to additional interactions between multiple elements.
Grain boundary diffusion in multi-component alloys
Grain boundary structures in multi-component alloys: interplay of segregation, precipitation and phase decomposition
The thermal and chemical stability of grain boundaries, particularly in terms of their affinity to segregation and possible precipitation and phase decomposition phenomena is crucial for GB diffusion in multi-component alloys. The concomitant development of elastic stresses might result in the creation of networks of GB dislocations which further affect the GB diffusion rates, with requirements to account for complex mechano-chemical coupling phenomena at GBs. Even in the case of nominally pure Ni, the appearance of networks of secondary GB dislocations due to the variation of GB inclination revealed a dramatic impact on the GB diffusion for a single grain boundary in a Ni bicrystal.
66
Similar effects were previously reported for GB diffusion in Cu near
In the case of pure metals, due to the absence of chemical complexity, the character of the grain boundaries, i.e. the GB bi-crystallography (Section “Grain boundary bicrystallography”), alone will influence the properties of the respective pure metal. In multicomponent alloys/compositionally complex alloys, the chemistry of grain boundaries, the segregation of elements along the grain boundaries, and the precipitation along grain boundaries play crucial roles in the material’s performance, including technological applications. Casales et al.
198
have reported that a continuous network of carbides along grain boundaries increases the resistance to stress corrosion cracking. Thus, GB segregation can have both beneficial or detrimental effects on the material’s performance, and it is important to understand such segregation behavior in technologically relevant materials199,200 and its impact on diffusion kinetics.
56
Solute segregation may change the grain boundary character/structure as well, and it may directly influence the GB diffusion behavior. The segregation of Ag along the
Recently, GB diffusion-related mechano-chemical coupling phenomena were thoroughly investigated in two alloys of formally similar compositions, namely in the Ni–Cr–Mo-based Hastelloy C-22
20
and Ni–Cr–Fe-based 602 CA19,21 alloys, see Table 4. These alloys are well-known for their exceptional resistance to corrosion and oxidation at high temperatures. According to equilibrium thermodynamics, a single-phase FCC (
In Figure 12, the evolution of grain boundary structure with precipitation and growth of GB carbides is shown. The impact of the annealing treatment at 873 K is comparatively examined for two states: a pristine one directly after homogenization at 1423 K (group A) and one produced by an intermediate heat treatment at 1173 K (group B). Direct heat treatment at 873 K has resulted in the formation of Cr-rich M23C

Mechano-chemical coupling in the Ni-Cr-Fe 602CA alloy: evolution of GB phases and GB structures for random high-angle GBs influenced by formation and growth of Cr-rich M23C
In the case of group B, which was intermittently annealed at 1173 K, the carbide precipitation occurred uniformly along the high angle grain boundaries (HAGs) and minimal growth was observed after prolonged annealing, Figure 12. The carbides grew continuously along the grain boundaries with minimal intrusion into adjacent grains while maintaining a nearly equiaxed morphology. The intermittent annealing has modified the growth behavior of the GB carbides. Moreover the intermittent annealing has formed stable carbides which grew in equilibrium fashion along the grain boundaries without altering the structure of the grain boundaries. The M23C
Carbide precipitation resulted in Cr loss and Ni enrichment within the FCC matrix. In addition, a certain Al loss in the FCC matrix was due to the formation of Ni
The disturbance of local chemical equilibria at grain boundaries due to carbide precipitation can not only influence the diffusion kinetics but can also alter the phase stability. Atomic scale decomposition resembling a spinodal–like decomposition along the grain boundary has been observed,19,21 with enrichments towards Ni+Al and Cr+Fe alternative layers, Figure 13. Furthermore, two families of high-angle GBs can be distinguished in the Ni-based alloy, namely with respect to the evolution of the precipitate types and the GB appearance. The existence of the two types of high-angle GBs correlated with the appearance of two distinct short-circuit diffusion paths measured by Bian et al. 21 A comprehensive analysis substantiated that the straight GBs with small globular precipitates corresponded to the slow GB diffusion branch and the modified, highly distorted and hackly boundaries exhibited significantly enhanced diffusivities, 21 Figure 14. The observation of various complexions in these alloys indicate a significant influence of the heat treatment parameters (temperature and time) which predominantly affected the kinetics along the GBs.
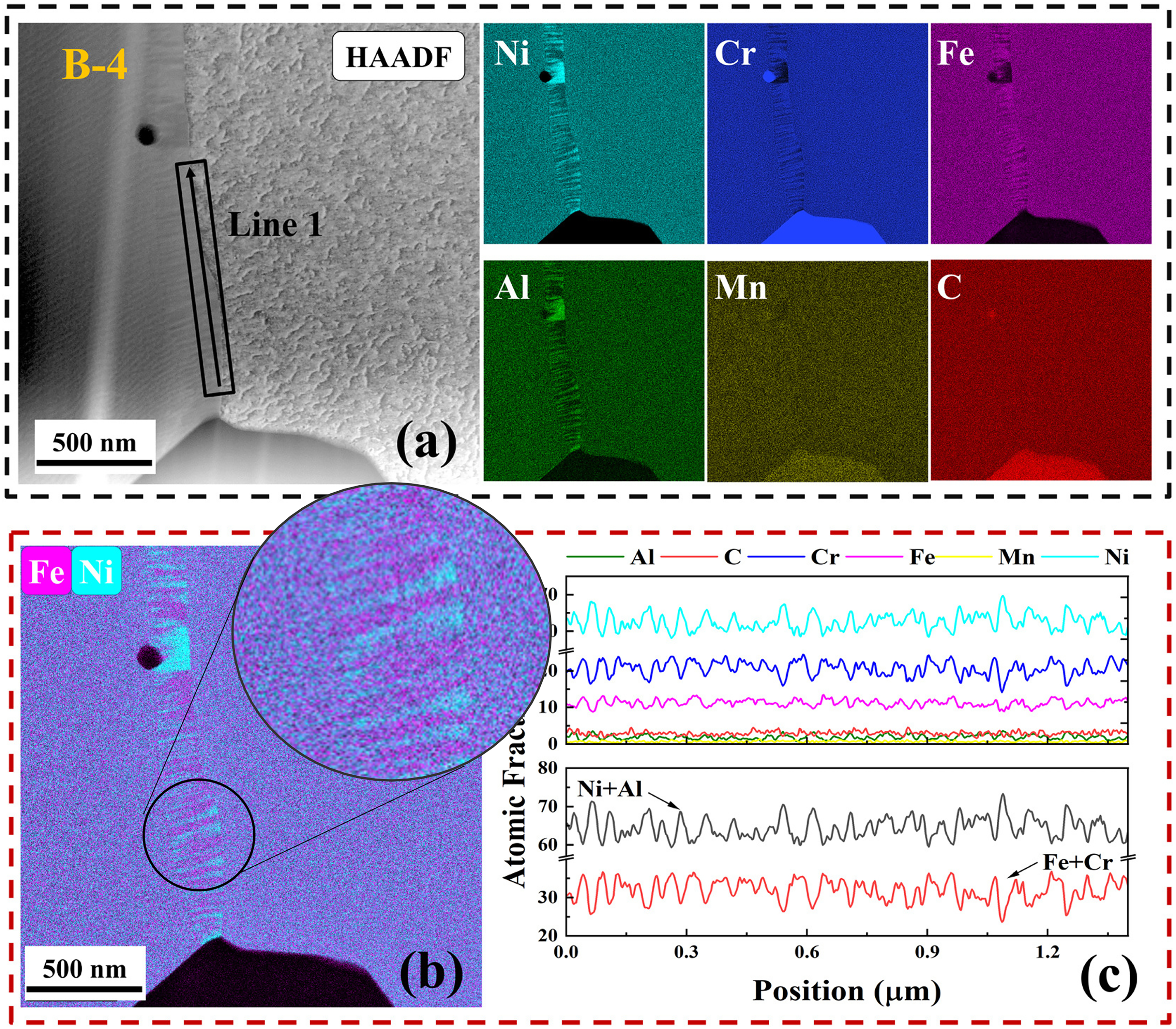
Grain boundary structures in 602CA alloy pre-annealed at 1173 K for 16 h and aged at 873 K for 744 h as seen by HAADF-STEM image and the elemental maps (a). A spinodal-like decomposition along the given GB showing alternative layers with Ni+Al vs. Fe+Cr enrichments, highlighted by insert (b), (c). Note that the given GB is inclined with respect to the electron beam and the GB decomposition is seen in projection. (The figures are reproduced from Bian et al. 21 ) and modified to highlight the spinodal-like decomposition seen for the inclined grain boundary.
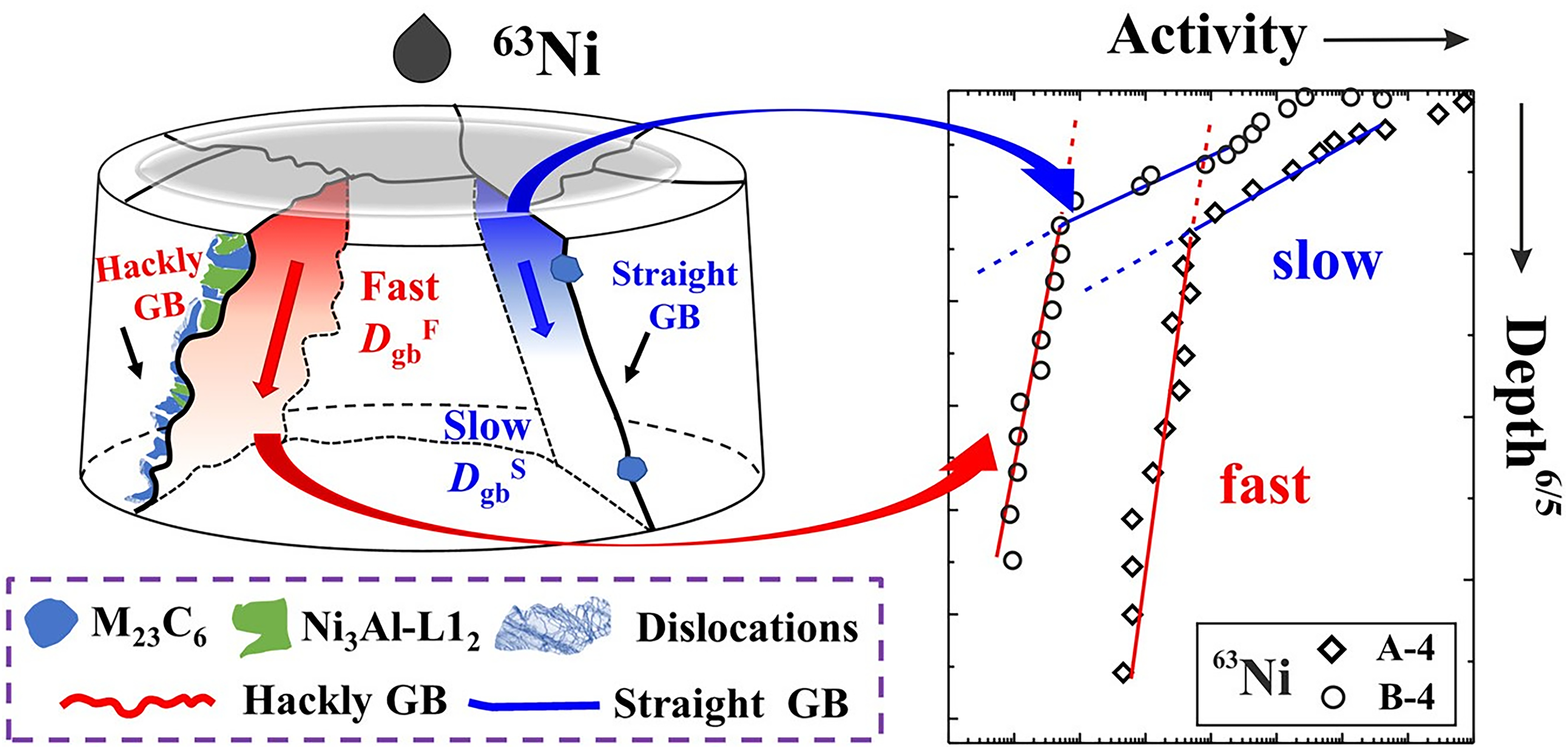
Correlation of the appearance of high-angle GBs and the types of short-circuit diffusion paths due to mechano-chemical coupling in a Ni–Cr–Fe 602 CA alloy. (Adopted from Bian et al. 21 )
In contrast, carbide precipitation accompanied by a strong solute (Mo and W) segregation in the Ni–Cr–Mo Hastelloy C22 hinders GB diffusion. 20 Figure 15 shows the evolution of carbides along grain boundaries in the Ni–Cr–Mo C22 alloy. Unlike in the Ni–Cr–Fe alloys, carbides are formed continuously along the grain boundary, occupying almost the entire grain boundary area, Figure 15.

Evolution of GB precipitates in the Ni–Cr–Mo Hastelloy C22 subjected to solutionizing (B-0) at 1123 K, followed by further heat treatments at 1123 K for 1 h (B-1) up to 744 h (B-6). The observed GB carbides are found to be of M6C type. Upon heat treatment for about 168 h (B-4), a new M23C
On first sight, the C-22 20 and 602CA 21 Ni-based alloys show quite similar compositions, though with an important difference in the presence of strongly segregating elements like Mo and W in C-22, Table 4.
It is exciting that opposite trends in the tracer GB diffusion coefficients of Ni were measured for both, as-homogenized states (Groups A) and initially annealed at 1123 K (C-22) or 1173 K (602CA) states (Groups B), see Figure 16.
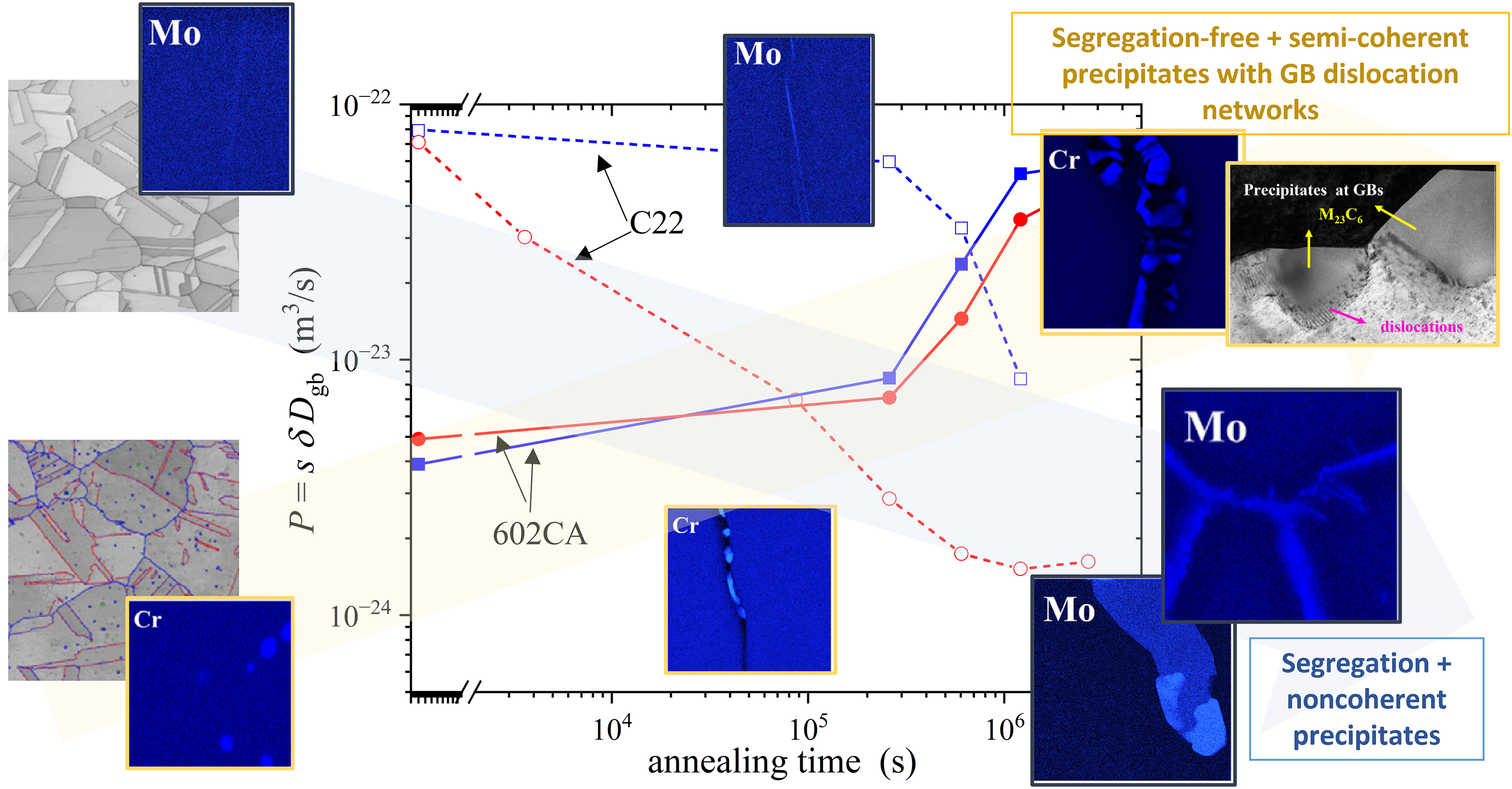
Triple products of Ni GB diffusion in C-22 (dashed lines and open symbols 20 ) and 602CA (solid lines and filled symbols 21 ) Ni-based alloys as function of annealing time at 873 K. The data were measured for two states, namely the homogenized one (blue) and after an intermittent annealing treatment (red), in the two alloys. Developing GB dislocation networks due to precipitation of semi-coherent carbides in 602CA enhances GB diffusion, while strong segregation and precipitation of incoherent carbides in C22 results in GB diffusion retardation.20,21
In addition to the data presented in Figure 16, the relative contributions from GB diffusion along the straight GBs were measured and those did not evolve with time.20,21 The corresponding high-angle GBs were sporadically covered (or immobilized 21 ) by nano-scaled, globular carbides. In the segregation-free case, an enhancement of GB diffusion along hackly GBs with plate-like precipitates is obvious and it is induced by mechano-chemical coupling phenomena during nucleation and growth of GB precipitates. As the dominating effect for the GB diffusion enhancement, the developing network of GB dislocations was proposed, 21 see Figure 16. In contrast, if a strong Mo (and partially W) segregation is present and the carbides grow in non-coherent fashion, a strong retardation of Ni GB diffusion was observed. 20
The above discussion elaborates the competing effects of segregation, precipitation, and dislocation network formation reveal that grain boundaries are not passive interfaces but dynamic entities that govern diffusion, phase stability, and mechanical response. From a GB engineering standpoint, these findings suggest a tuneable route for GB diffusivity. By regulating GB character distributions, segregation behavior, and complexion states, it becomes possible to design boundary networks that mitigate detrimental diffusion paths while enhancing beneficial ones. Heat treatment protocols and compositional design, when integrated with GB engineering approaches, provide a pathway for creating alloys with predictable, superior performance in aggressive environments, transforming GBs into assets in advanced materials engineering.
Grain boundary phase transitions in multi-component alloys
In Section “Grain boundary structure transitions”, it was shown that due to multiplicity of GB structures,144,206,207 i.e. the availability of different GB structures at a fixed GB crystallography, temperature-induced GB structure transitions can be present even in pure metals.95,142 Alloying, i.e. addition of one or more elements, might result in segregation-induced GB transitions, called as GB phase transitions 124 or complexion transitions. 208 These transitions, influencing the GB structures,9,10 might affect GB kinetic properties, including GB diffusion. The latter was unambiguously demonstrated for GB pre-wetting and wetting transitions, e.g., for Cu–Bi 170 or Ni–Bi 116 binary alloys.
High-resolution TEM (HR-TEM) imaging became a powerful tool for investigating the GB structures and the wide spectrum of GB phase transitions. Devulapalli et al.
10
used HR-TEM to investigate the impact of the Fe alloying on GB structures in pure Ti thin films. Pristine (Fe-free) symmetric
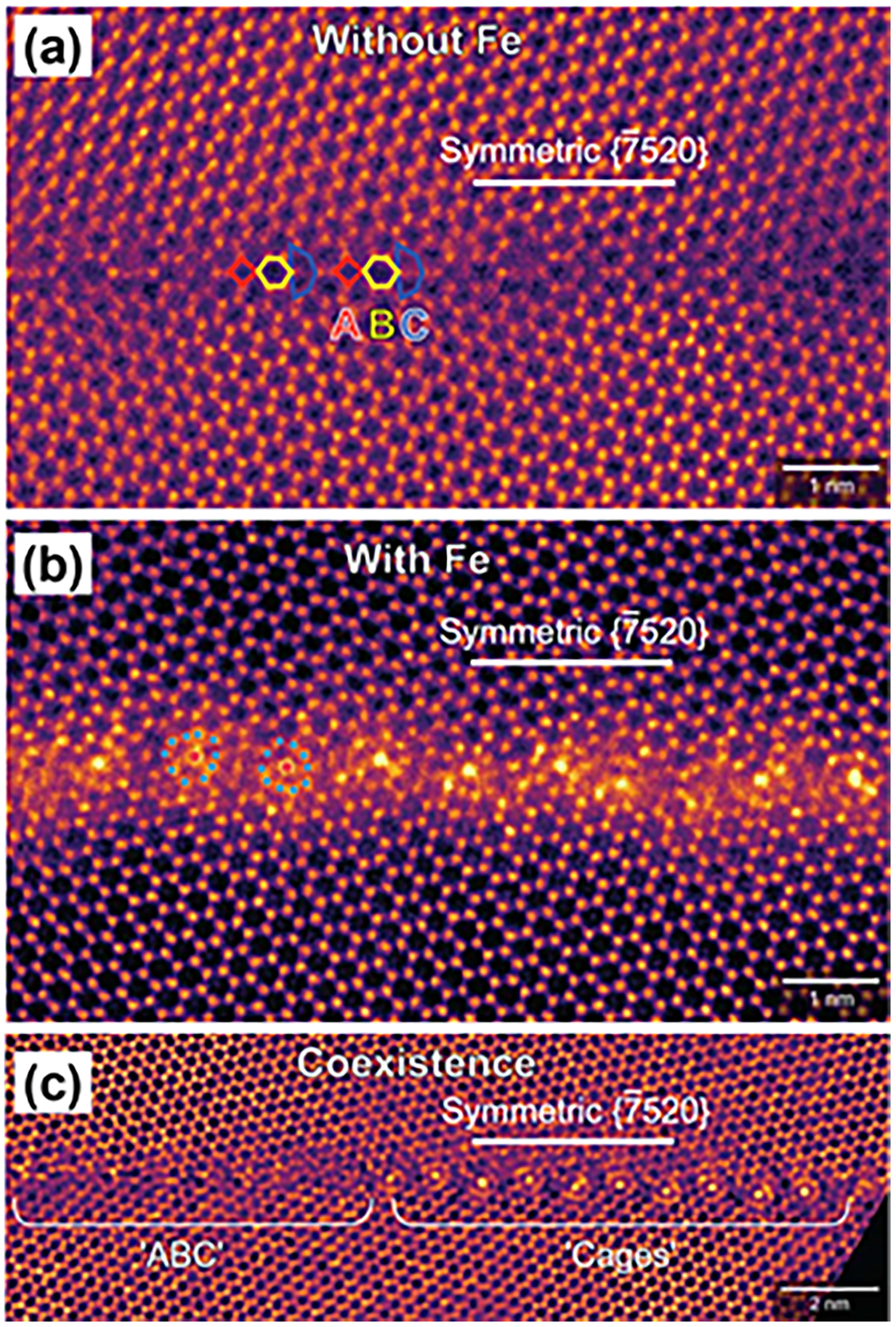
Devulapalli et al.
10
argued that the accommodation of Fe atoms in the Ti GB by formation of cages with a fivefold symmetry, Figure 17(b), prevents the formation of ordered phases and might be reminiscent of quasi-crystals or a glassy state in the GBs. It is reasonable to suggest that Fe GB diffusion in
In HEAs (or even more generally in MPEAs) such experiments with atomic resolution of GB structures are almost absent. GB segregation in a representative equiatomic CoCrFeMnNi high-entropy alloy (HEA) aged at
These results correlate generally with the findings of Glienke et al. 118 where phase decomposition and precipitation were observed at high-angle GBs in nominally the same equiatomic CoCrFeMnNi HEA. The GB segregation and phase decomposition at lower temperatures were correlated with the appearance of a specific, ”ultra-fast” GB diffusion path, revealed by GB diffusion measurements. 118 Such GB diffusion path is absent at higher temperatures corresponding to stability of the random solid solution and absence of any segregation.
The element segregation and GB phase decomposition might be affected or triggered by impurities, which demonstrate a strongly GB segregation. For example, Yang et al.
213
correlated the decomposition of the Al11La
Due to the comprehensive GB structure–impurity atom interactions, GB diffusion of those impurities might reveal characteristic dependencies on the impurity segregation at the GBs. Such GB diffusion measurements would be highly desirable. The evidence of segregation-induced GB phase transitions and complexion formation in HEAs offer unique opportunities for alloy design. Unlike conventional alloys, HEAs exhibit strong temperature and chemistry dependent segregation behavior, leading to GB phase decomposition, nano-precipitate formation, and the emergence of “ultra-fast” diffusion paths. These features directly impact creep resistance, oxidation, and corrosion performance at elevated temperatures, making the control of GB states critical for several applications such as energy and aerospace systems. The processing strategies involving combination of thermo-mechanical treatment can be used to stabilize favorable GB complexions while suppressing harmful segregation.
Is grain boundary diffusion in high-entropy alloys “sluggish”?
One of the key attributes of HEAs is their high configurational entropy, which, in general tendency, stabilizes single-phase solid solutions. 214 This entropy arises from the vast number of atomic configurations in the alloy, and was speculated to lead to unique properties such as enhanced mechanical strength, corrosion resistance, and thermal stability.215,216 However, recent studies emphasize the well-known importance of factors such as mixing enthalpy, atomic size differences, and processing conditions for single phase formation in HEAs. 24
One of the earliest claims about HEAs was the concept of “sluggish diffusion,” which suggests significantly slower atomic mobility compared to conventional alloys. 217 This concept has been widely debated. Tsai et al. 218 were among the first to quantitatively investigate interdiffusion in HEAs, using a quasi-binary diffusion couple approach. Their findings supported the sluggish diffusion hypothesis, citing estimated tracer diffusivities and activation energies. However, their methodology has been criticized, particularly for assuming ideal solution behavior and neglecting vacancy wind effects, as highlighted by Paul. 219 Subsequent studies also challenged the early report on sluggish diffusion. Kulkarni and Chauhan 220 observed strong diffusional interactions in the Fe-Ni-Co-Cr system, contradicting Tsai et al.’s assumptions. Similarly, Sohn’s group 221 reported that interdiffusion in Al-Co-Cr-Fe-Ni HEAs was not inherently slower than in compositionally less complex systems, further questioning the sluggish diffusion hypothesis.
Tracer diffusion measurements further contradicted the sluggish diffusion hypothesis by showing that diffusion coefficients in HEAs were comparable to those in conventional FCC alloys. Vaidya et al.
222
and Gaertner et al.
223
demonstrated that increasing the number of components does not necessarily lead to slower diffusion. Similarly, Nadutov et al.
224
systematically measured Co tracer diffusion in Al
More recent studies reveal that the diffusion behavior in HEAs is complex and depends on factors such as element interactions, vacancy formation enthalpies, lattice type, and lattice distortions. Nayak et al. 225 showed that vacancy formation enthalpies increase with alloy complexity, affecting the diffusion kinetics. Muralikrishna et al. 226 demonstrated the importance of the interactions between constituent elements and the corresponding defects generated by the addition of these elements in understanding the diffusion behavior in multicomponent B2 aluminides. Zhang et al. 227 investigated diffusion in sigma-phase HEAs and found that bulk diffusion was not sluggish when analyzed on an absolute temperature scale. Similarly, Zhang et al. 228 examined BCC refractory HEAs and reported that diffusion in HfTiZrNbTa was not significantly slower than in conventional BCC alloys. These findings challenge the idea of sluggish diffusion as a universal property of HEAs. For HCP HEAs, even “anti-sluggish” diffusion has been reported,229,230 when one refers the measured diffusivities in the multi-principal alloy with respect to “predictions” by a simple geometric mean of the respective diffusion coefficients in the unaries. In fact, Ti diffusion in AlScHfTiZr HEA was by orders of magnitude enhanced with respect to this simple rule of mixture. 229
While bulk diffusion has been extensively studied, GB diffusion presents a more nuanced picture. Vaidya et al.
117
conducted the first radiotracer studies on GB diffusion of Ni in CoCrFeNi and CoCrFeMnNi HEAs, showing that GB diffusion is not universally sluggish. At around 800 K, GB diffusion was faster in the quinary alloy than in the quaternary, but at lower temperatures, the trend reversed. Compared to pure Ni, CoCrFeNi exhibited an order-of-magnitude slower GB diffusion, whereas CoCrFeMnNi displayed a more complex temperature-dependent behavior. GB diffusion in HEAs shows a mild retardation compared to pure metals, similar to the effects of impurities or ordered phases like Ni
Additional studies indicate that GB diffusion in HEAs is governed by composition, temperature, and defect structures rather than configurational entropy alone. Li et al. 232 found no sluggish diffusion effect in FeCrMnCoNi HEA across both, absolute and homologous temperature scales. Gaertner et al. 233 demonstrated strong diffusional interactions in Co-Cr-Fe-Mn-Ni HEAs, reinforcing the idea that GB diffusion cannot be solely attributed to high entropy effects.
Impact of chemical complexity on grain boundary diffusion in FCC solid solutions
The change in grain boundary diffusivity (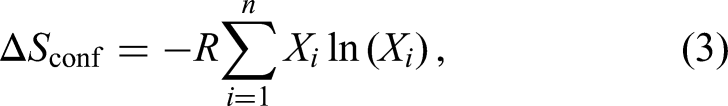
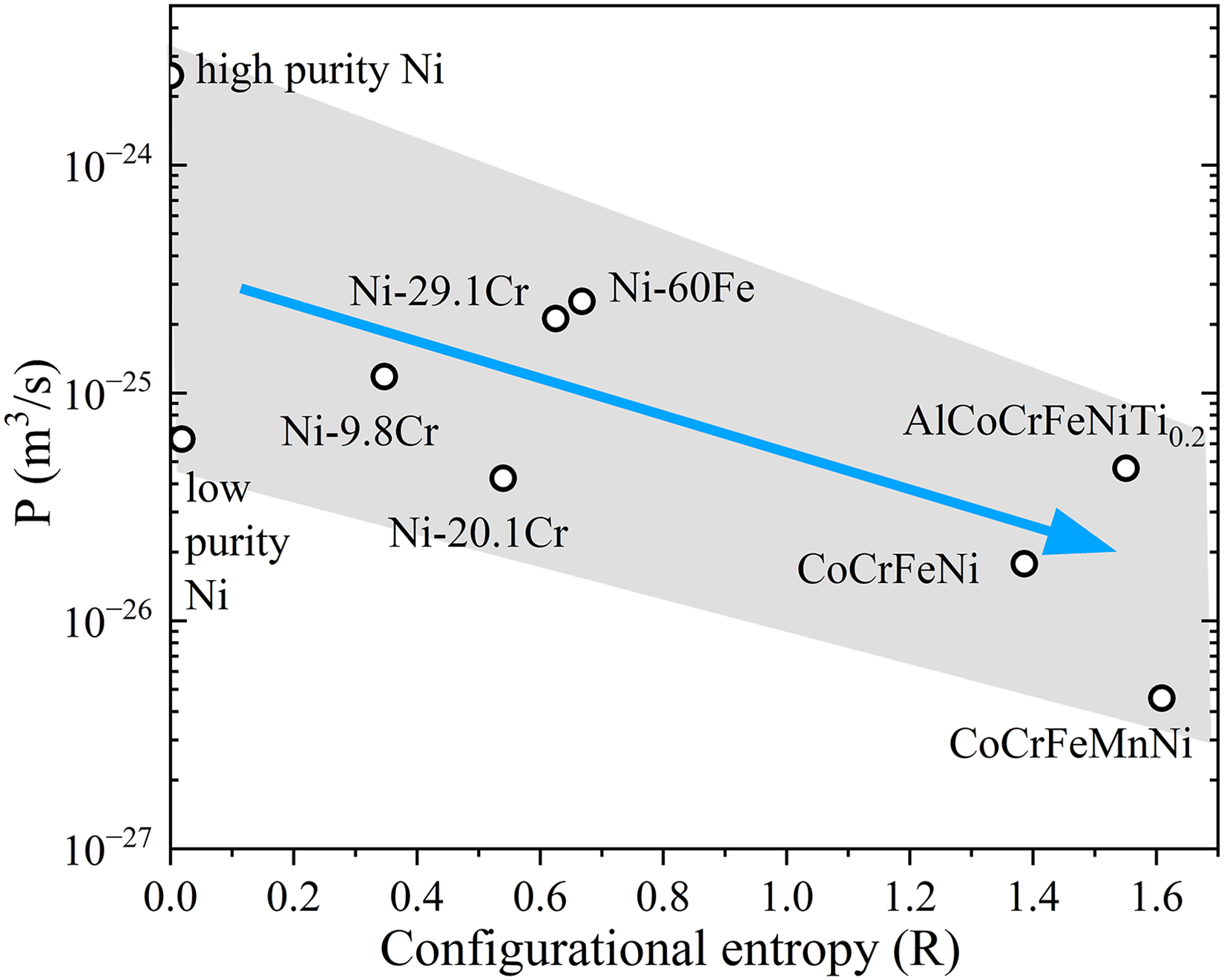
The change in grain boundary diffusivity (
The retardation tendency shown in Figure 18 is very pronounced for the FCC solid solutions at lower temperatures of about
Figure 18 presents a generic tendency, not a dependency of GB diffusion on the chemical complexity at the given temperature. Matrix purity has a strong impact on GB diffusion, as it is highlighted by the scatter of the data for nominally pure Ni. A presence of others, often not documented, interstitial impurities, like S, C, O, P, and others, even at ppm level in coarse-grained polycrystals influences GB diffusion and is partially responsible for the scatter, seen in Figure 18. Other factors, like GB character distribution, influence the GB diffusion results as well. We conclude that the correlation seen in Figure 18 (with all scatter of the data) is the best which can be drawn using the experimental studies on polycrystalline materials. In principle, one might produce bicrystalline samples from different alloys with almost the same misorientation parameters and measure GB diffusion of suitable elements. In view of expected huge hurdles for growth of reasonably large bicrystals though, this issue can be tackled by atomistic modelling more consistently.
Other crystalline structures
The experimental data are primarily available for FCC HEAs, while the data for other crystalline lattices are almost absent. GB diffusion in BCC or B2-ordered multi-component alloys was measured in a very limited number of studies, while GB diffusion data for HCP HEAs are simply absent. Though, Heng et al.
120
observed a retardation of GB diffusion of Ni for the B2-ordered alloys with increasing chemical complexity (quantified as the configurational entropy), which is still less pronounced as that for FCC alloys, within an order of magnitude at
In Figure 19, the triple products
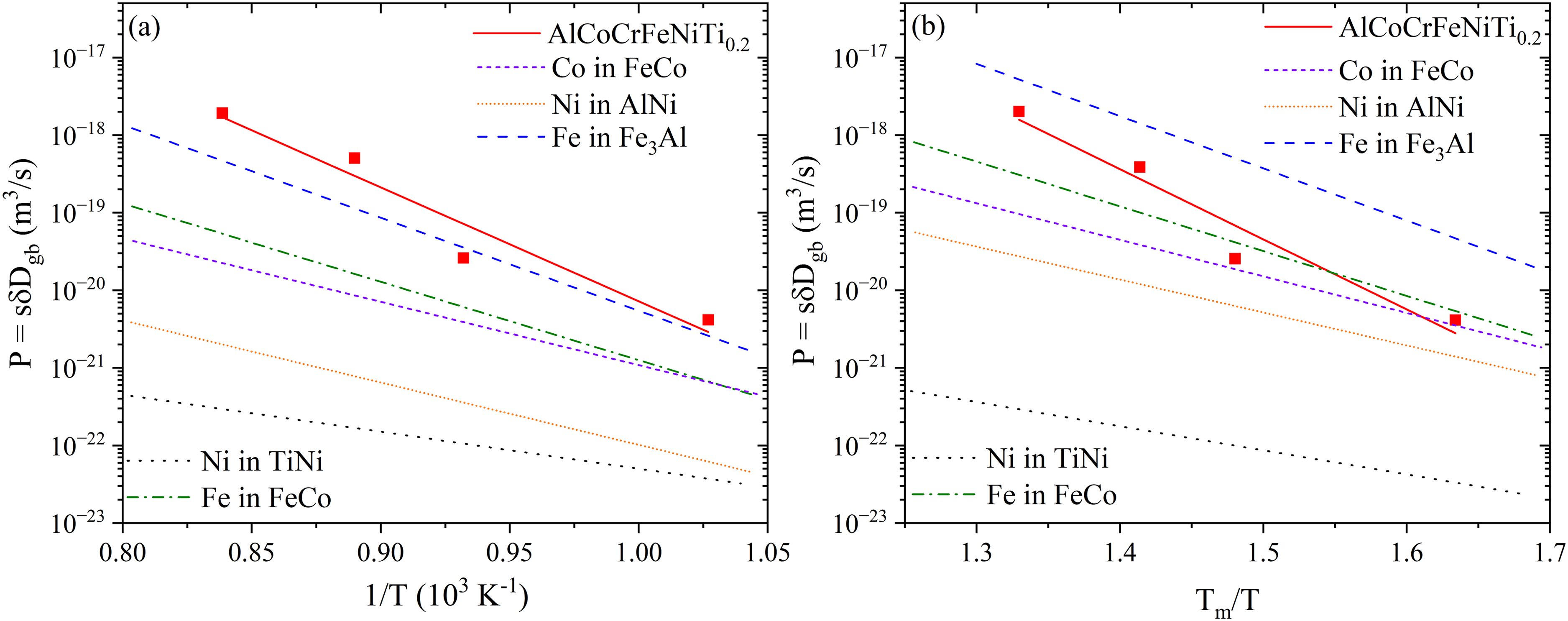
Ultimately, the hypothesis of sluggish diffusion in HEAs remains far from being accepted or proven. While bulk diffusion frequently contradicts the hypothesis, GB diffusion exhibits no universal trend. Instead, its behavior is intricately linked to composition and microstructure. Some HEAs show slower GB diffusion, while others defy expectations with enhanced diffusivity. Experimentally, diffusion properties of general (random) high-angle GBs, as present in cast, homogenized and equilibrated (via a certain mechanical deformation plus recrystallization annealing) polycrystals, were addressed. Bicrystalline samples were almost never investigated for HEAs. Further insights are provided by atomistic simulations, see Section “Computational assessment of grain boundary and interphase boundary diffusion”. The evidence shows that neither bulk nor grain boundary diffusion is universally suppressed; rather, diffusion is strongly governed by alloy composition, lattice type, defect structures, and impurities. This complexity means that diffusion in HEAs can be strategically tuned rather than assumed to be inherently sluggish. Grain boundary engineering therefore becomes essential, enabling control over boundary character distributions and segregation tendencies to manage diffusion pathways. By integrating experimental diffusion data with atomistic modeling, HEA compositions and processing routes can be tailored to balance strength with diffusion-controlled phenomena for technological applications.
Metastable grain boundaries in multi-component alloys
Processing methods, particularly severe plastic deformation techniques like equal channel angular pressing (ECAP) or high-pressure torsion (HPT), influence GB diffusion by modifying microstructural characteristics.55,235
In ultrafine-grained Cu–Zr alloys (0.17 wt.%Zr), 168 ECAP processing results in a bimodal distribution of GB diffusivities. According to Divinski et al., ”fast” GBs are likely remnants of original high-angle grain boundaries that absorbed dislocations and thus exhibit diffusivities over two orders of magnitude higher than ”slow” GBs. Similarly, in Cu–Pb alloys, ECAP introduces distinct GB diffusion pathways. Ribbe et al. 236 reported that the relatively slow paths are related to relaxed HAGBs, while fast paths correspond to non-equilibrium GBs. The activation enthalpy for fast paths constitutes about 50% of the value corresponding to high-angle GBs in coarse-grained polycrystalline Ni.
In a simulation study by Lappas et al. 237 , a triple diffusion model was applied to ultra-fine grained Cu–0.17 wt.% Zr, categorizing GB diffusion into fast, mixed, and slow paths. This model uncovered three distinct GB types, potentially resulting from SPD, and demonstrated that cross-diffusion between these paths adheres to Arrhenius behavior. These simulation insights underscore the critical role of GB segregation, structural disordering, and microstructural complexity in governing GB diffusion in binary Cu alloys.
Additive manufacturing, which is a rapidly developing area, modifies characteristically the GB structures and properties, too.179,238
Additively manufactured (AM) metallic materials is known to contain numerous grown-in defects which limit the durability and mechanical properties of a workpiece. However, the existence and impact of non-equilibrium vacancies and dislocations and especially the state of grain boundaries in AM materials remained largely unexplored until very recently. Grain boundary diffusion of Ni in the equiatomic CoCrFeMnNi high-entropy alloy, produced by additive manufacturing, was found to be strongly enhanced with respect to the coarse-grained cast-homogenized state. 179 A low-temperature annealing treatment was found to relax the ‘non-equilibrium’ grain boundary state without invoking grain growth, representing an ultimate proof of the metastable, ‘non-equilibrium state’ of general high-angle grain boundaries in an as-produced AM high-entropy CoCrFeMnNi alloy.179,180 Furthermore, a non-monotonous temperature dependence of the Ni GB diffusion coefficients was seen and explained by relaxation of the non-equilibrium state induced by rapid solidification during fabrication. 238 The intermittent retardation of the diffusion rates with increasing temperature is spectacular and highlights a discontinuity in the governing GB diffusion mechanisms. In fact, the energy barriers for atomic transport become larger at higher temperatures due to GB re-structuring. The temperature-induced evolution of the grain boundary state was analyzed in terms of the concomitant structure evolution, segregation, phase stability and precipitation in the multi-component alloy. 238
The metastable states created by additive manufacturing were reviewed by Hu et al. 239 As it was experimentally shown, 179 rapid heating/cooling cycles drive GBs to a non-equilibrium state with distinct energy and kinetic properties compared to conventional equilibrium GBs. Atomistic simulation has shown 240 that metastable GBs exhibit a spectrum of energies due to the existence of microstructural and chemical disorder.
Grain boundary energy
Grain boundary energy is the excess energy per unit area associated with the presence of a grain boundary in a polycrystalline material. It arises due to the discontinuity in the crystal structure at the boundary between two grains, leading to a mismatch in atomic positions and bonding.
Grain boundary energy plays a pivotal role in determining the microstructural evolution and properties of polycrystalline materials. 241 It drives processes such as grain growth and recrystallization, as materials naturally seek to minimize their total Gibbs free energy by reducing the area of high-energy grain boundaries. This has significant implications for the thermal stability and mechanical behavior of materials. 242 High-energy grain boundaries can also act as weak points, making materials more susceptible to creep, intergranular corrosion, and cracking under certain conditions.243,244 Furthermore, GBs serving as preferential sites for short-circuit diffusion are critical for processes like sintering, 245 phase transformations, 246 and oxidation. In ultrafine-grained and nanocrystalline materials, where the GB volume fraction is significant, grain boundary energy plays a dominant role in governing properties such as diffusion rates, phase stability, and mechanical strength.235,247 Additionally, grain boundary engineering modifying the boundary character to lower energy states offers a powerful tool for improving material properties, including creep resistance, corrosion resistance, and fatigue life. 5 Thus, understanding and controlling grain boundary energy is essential for tailoring the performance and reliability of materials across a wide range of applications, especially in technologically-relevant multi-component materials.
The magnitude of GB energy is influenced by several critical factors, which are schematically depicted in Figure 20. It includes temperature, chemical complexity, purity, segregation, crystal structure, microstructure, and deformation. In Figure 20, the Read-Shockley equation,
248
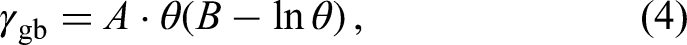
Some factors influencing the GB energy in a fictitious metal. Dependencies on the temperature, misorientation angle, and segregation are explicitly shown using the Read-Shockley equation, 248 equation (4), as a case example. The black arrow indicates a decrease of the GB energy due to an impurity segregation. Further parameters, such as GB inclination, GB defects, chemical complexity, purity, precipitation, crystal structure, external action (plastic deformation or non-equilibrium processing), influence the GB energy, too.
Measurements of grain boundary energy
Primarily, two methods of measurements of GB energy are widely employed: (i) the thermal grooving approach and (ii) a semi-empirical approach utilizing bulk and GB diffusivities.
Following the thermal grooving method, Figure 21, the equilibrium angle at the root of thermal grooves 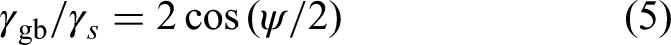
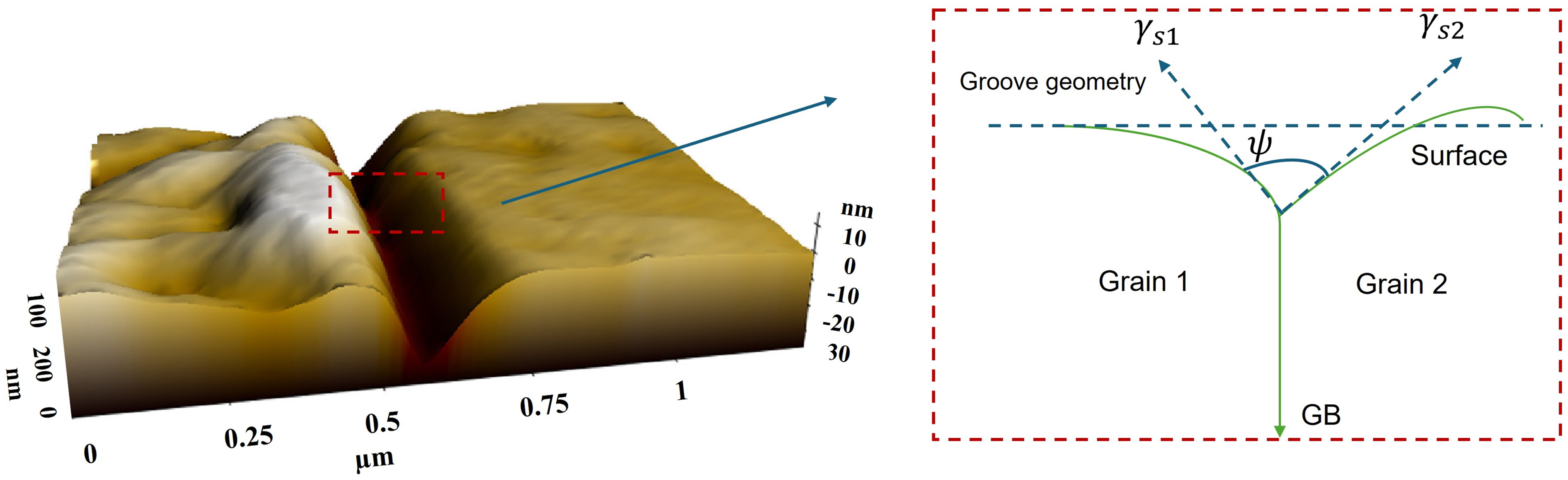
The thermal grooving method has been effectively applied to estimate the GB energies in various metals,including fcc254,256–259 and bcc260–262 metals and alloys.
Borisov et al.
188
proposed a semi-empirical approach, which was further developed by Guiraldenq
263
and Gupta.
264
In the case of vacancy-mediated diffusion within both, the crystalline bulk and the GB slabs, the excess GB energy is proposed to account for the enhancement of the GB diffusivity,
188
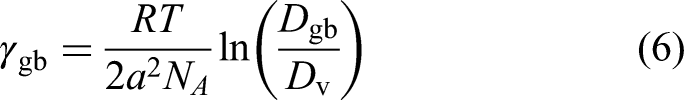
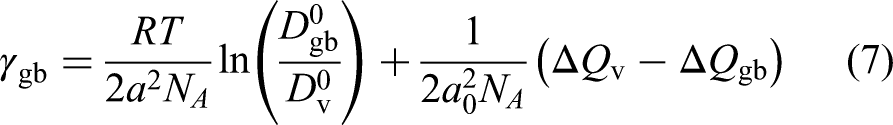
Pelleg
265
reported a consistent agreement between the values of GB energies determined using the thermal grooving and diffusion methods for fcc metals like silver, nickel, and
In addition to experimental approaches, Density Functional Theory (DFT) has become a powerful computational method to estimate GB energies, providing fundamental atomic-level insight into grain boundary structures and energetics at 0 K. Several key studies have systematically calculated GB energies in pure metals using DFT. For BCC metals, Li et al. 267 calculated GB energies for various tilt boundaries in metals such as Fe, Mo, and W, showing excellent agreement with available experimental data and establishing reliable scaling relationships between materials. For FCC metals, similar studies by Li at al. 268 focused on high-throughput DFT calculations of GB energies across ten FCC metals, further showing strong correlations between GB energies and low-index surface energies. Table 5 summarizes GB energy values for selected BCC and FCC metals obtained from thermal grooving, diffusion, and DFT methods. It highlights the good agreement between DFT and experimental data, especially for the FCC metals.
GB energies in BCC and FCC metals at 0 K. All values in J/m2. The experimental values are extrapolated to 0 K and averaged values of the ab initio (DFT) calculations are listed. For further details see Li et al.267,268 TG is thermal grooving method and DGB corresponds to the borisov semi-empirical formalism applied to the GB and volume diffusion data.
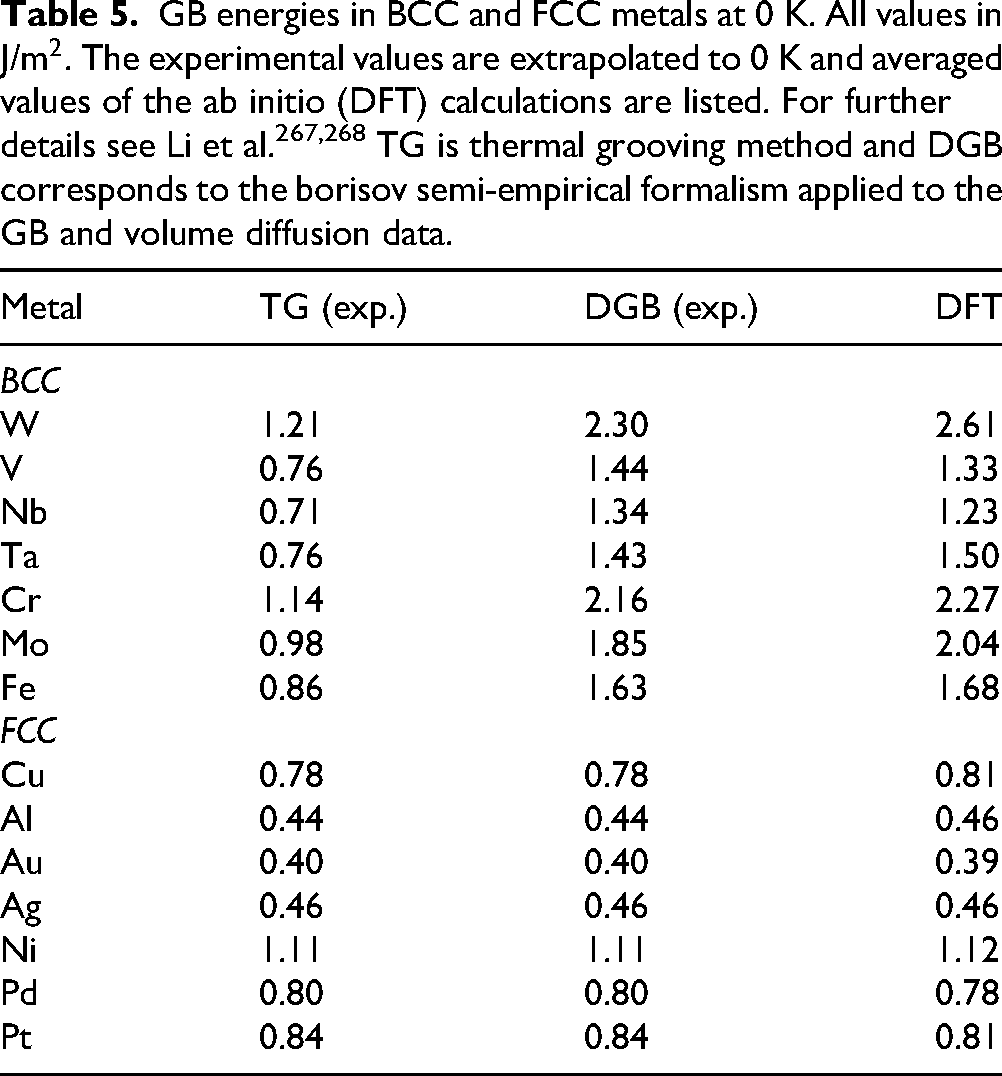
As a general statement, one can conclude that the thermal grooving experiments (TG) yield the values which are about 50% of the values estimated following the Borisov formalism (DGB),
The DFT values and experimental data presented in this table are sourced from Li et al. 267 for BCC metals and Li and Lu 268 for FCC metals. Detailed references for the experimental data can be found within these publications.
The following subsections briefly discuss some of the key factors that influence the GB energy.
Impact of alloying on grain boundary energy
Figure 22 shows the influence of alloying on the GB energy for nominally pure metals (Cu, Au, Fe) and their alloys. In general, alloying elements can either increase or decrease
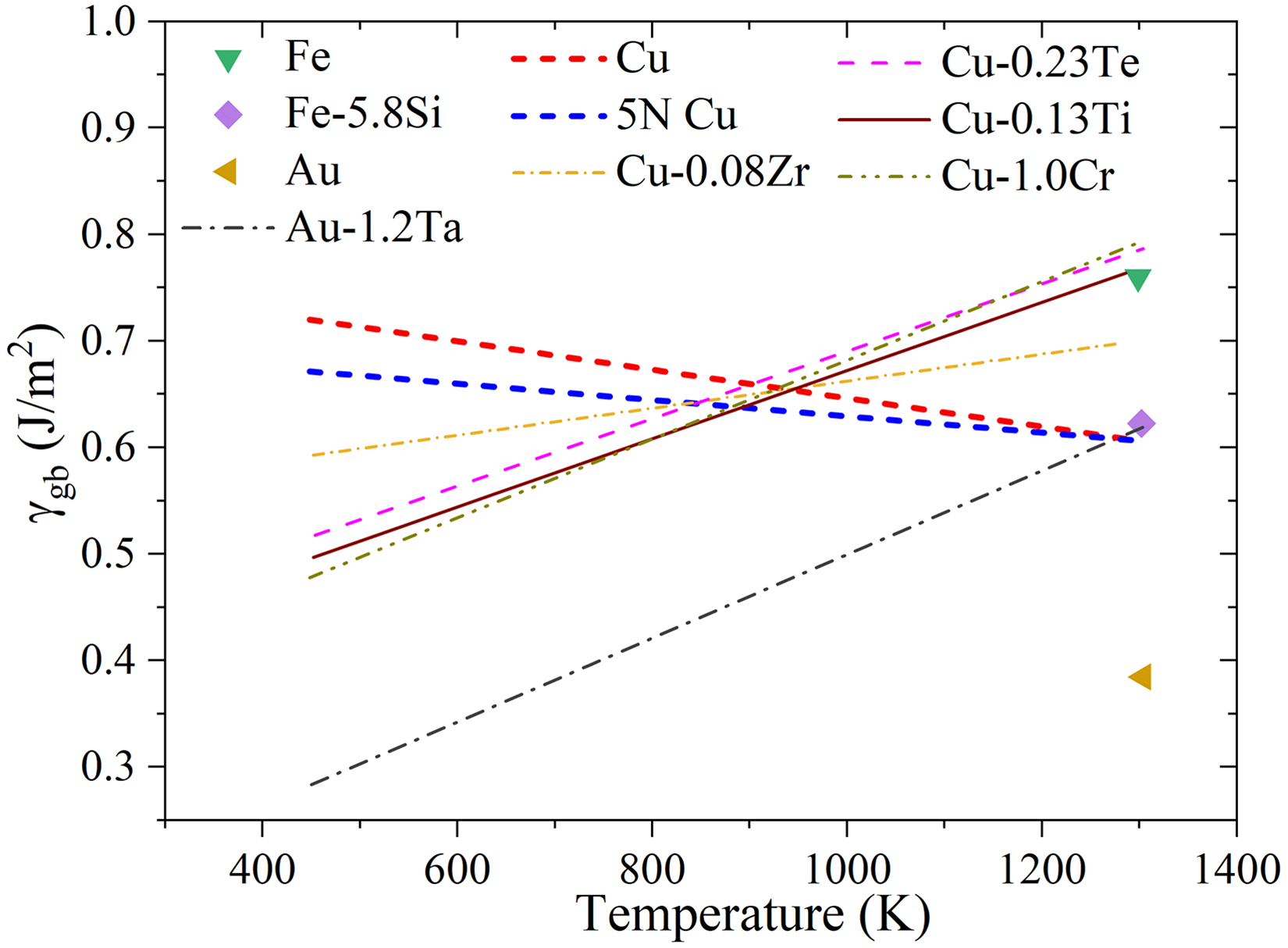
Similarly Gupta,
269
in the Au system, the Au-1.2 at% Ta alloy also shows a higher GB energy than pure Au, since Ta has a higher melting point. Such elements do not segregate to the GB core, but occupy preferentially GB sites which are more distant from the GB core.
270
Contrarily, segregation of lower melting point elements decreases GB energy, compare the
Effect of processing route
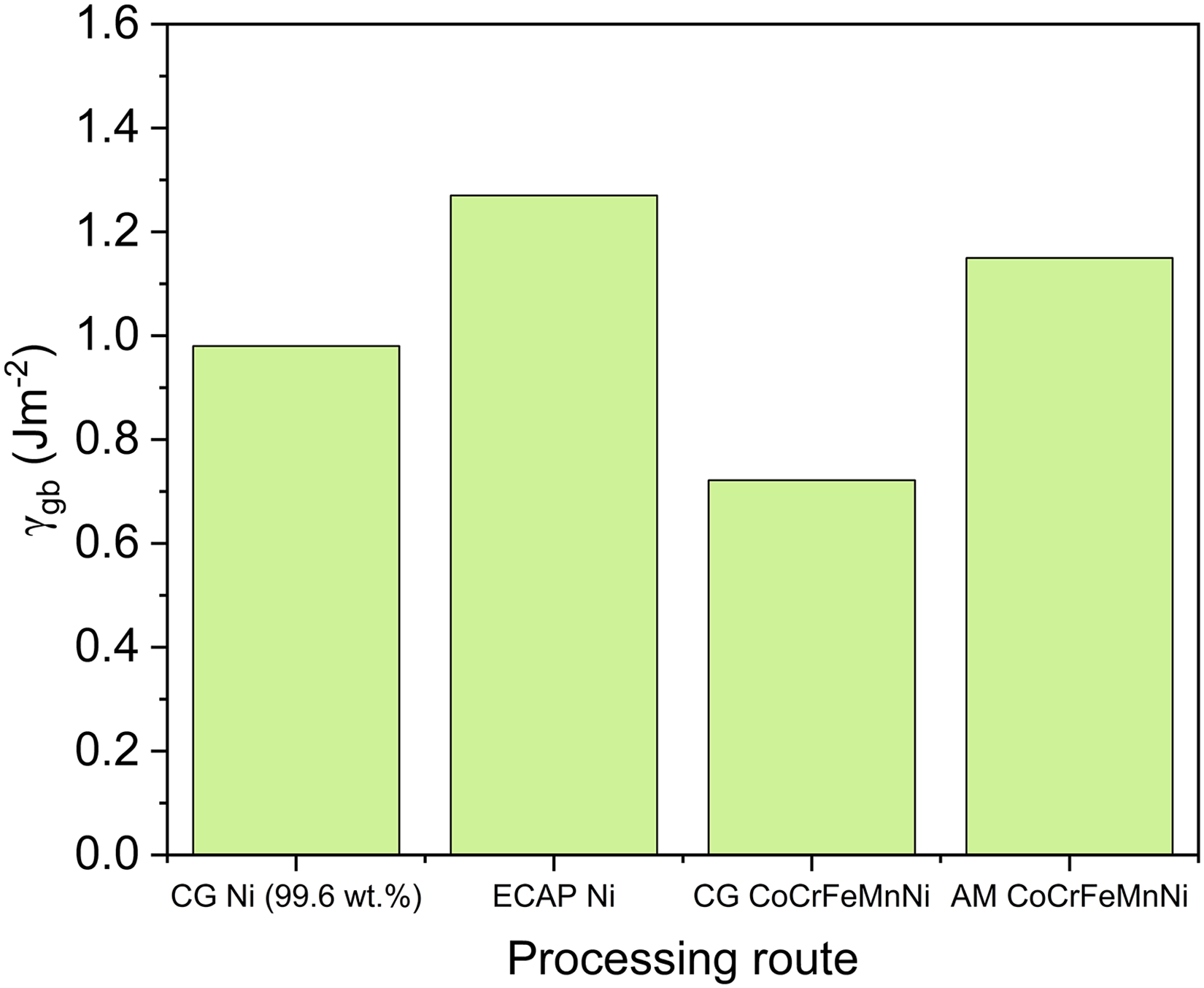
The grain boundary energy is significantly influenced by the processing method. Severe plastic deformation can significantly elevate the GB energy by introducing so-called ”non-equilibrium” (or ”deformation-modified”) grain boundaries.235,272 As shown in Figure 23, the GB energy of pure Ni after severe plastic deformation via equal channel-angular pressing (ECAP) is increased to about 1.27 J/m
Effect of chemical complexity
The chemical complexity of an alloy, determined by the number and type of elements present, significantly affects GB energy through solute segregation, atomic size mismatch, electronic interactions, and thermodynamic stability. In pure metals, GB energy is relatively high due to the absence of solute-induced stabilization. As additional elements are introduced, solute segregation to GBs reduces interfacial energy by altering local atomic bonding and minimizing excess free energy. In multi-component alloys, further reductions in GB energy arise due to a combination of chemical interactions and high configurational entropy.
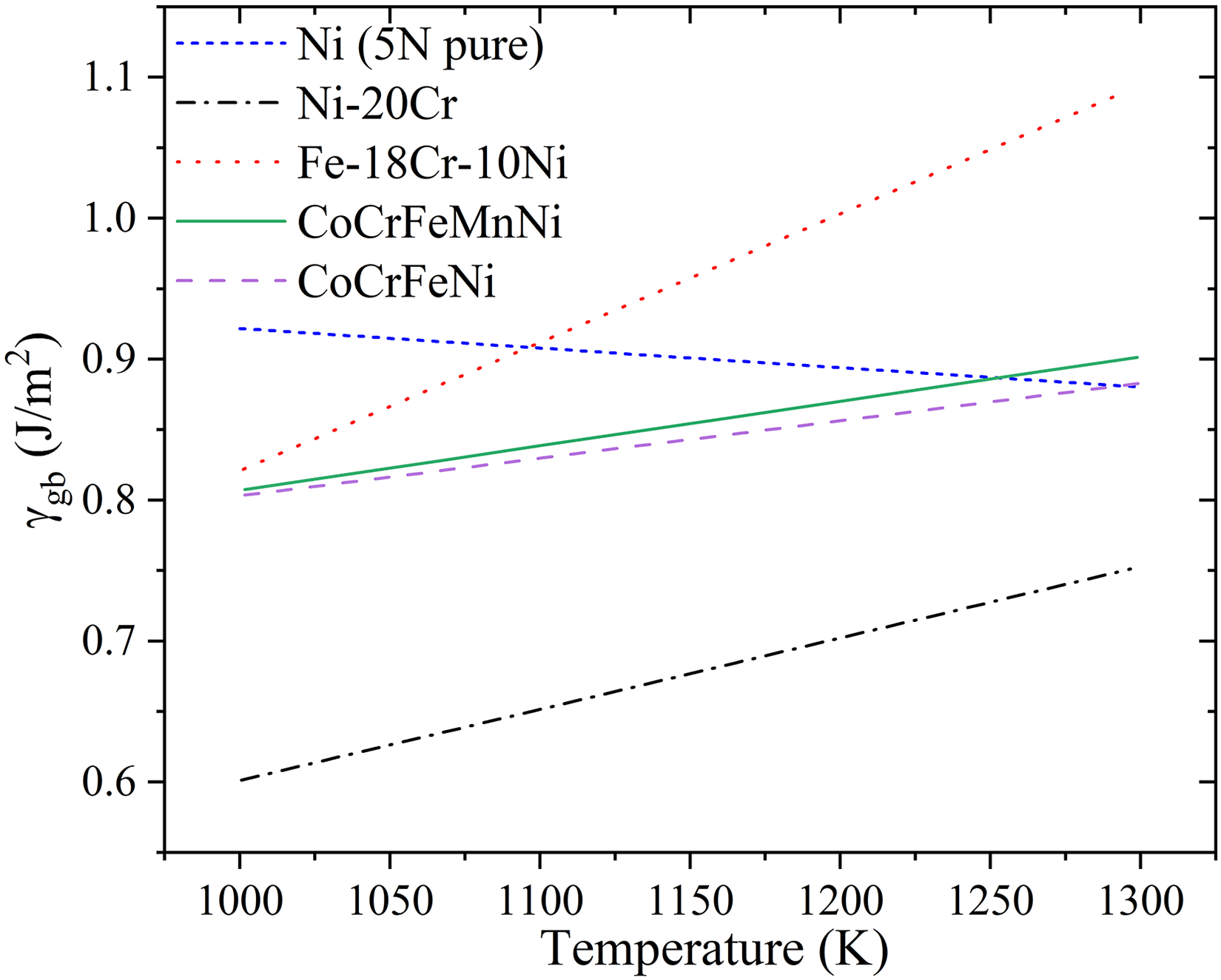
In Figure 18, the impact of configurational entropy on GB diffusion in single-phase random solid solutions is shown. However, the specific influence of chemical complexity on GB energy is not always straightforward and depends on the nature of solute interactions at the boundaries. Figure 24 illustrates the variation in GB energy with temperature for unary (pure Ni),
91
binary (Ni-20Cr),
171
ternary (Fe-18Cr-10Ni),
274
quaternary (CoCrFeNi),
178
and quinary (CoCrFeMnNi)
118
alloys. A general trend of decreasing GB energy with increasing chemical complexity is observed, but an interesting anomaly is seen for the binary Ni-20Cr system, which exhibits the lowest GB energy among all compositions. This suggests that Cr segregation in Ni-20Cr plays a dominant role in stabilizing GBs, leading to a significant reduction in
Effect of crystal structure
No clear trend has been established regarding the effect of crystal structure on GB energy, Figure 25(a). Limited studies conducted on non-cubic materials suggest that GB energy tends to have lower values compared to FCC materials.
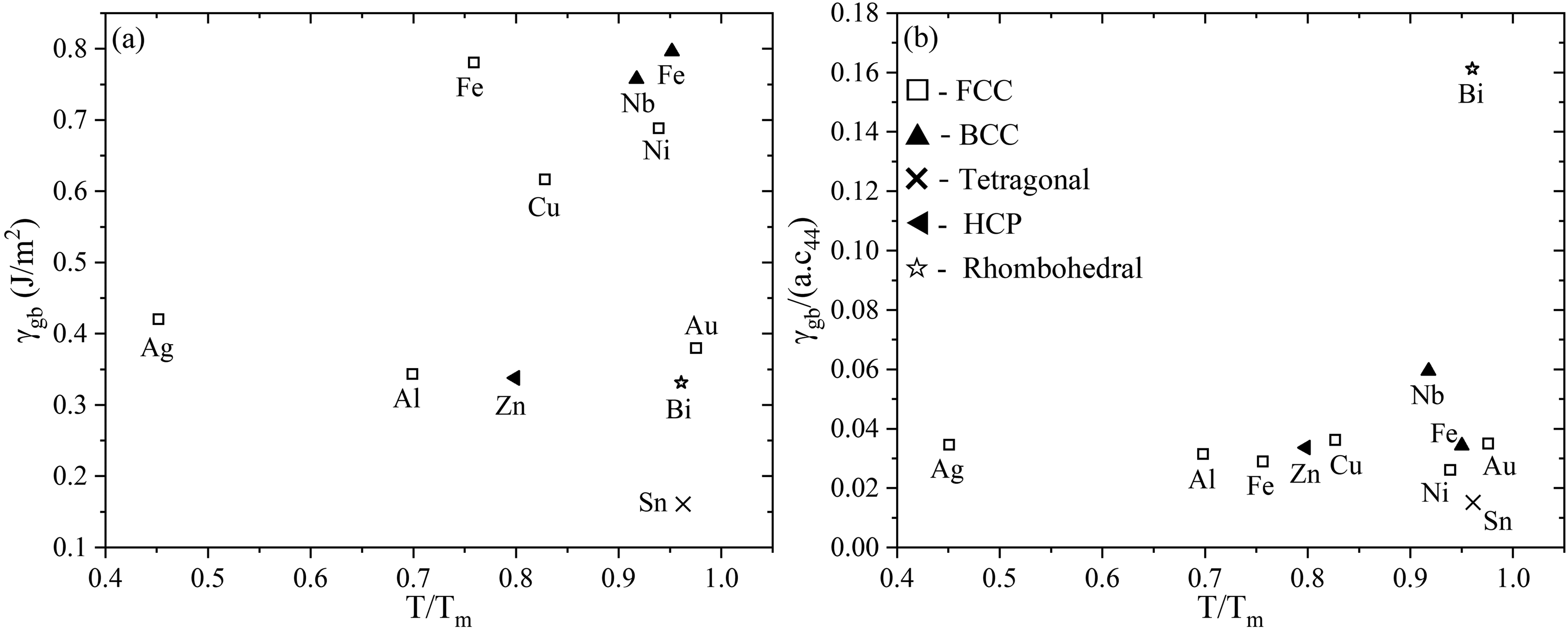
Effect of crystal structure on grain boundary energy for various metals. The absolute value of the GB energy,
In Figure 25(b), the values of the normalized GB energy,
On the other hand, the
Interphase boundary diffusion in binary and compositionally complex alloys
Experimental measurements on eutectic binary systems have demonstrated that diffusion along interphase boundaries is influenced by a number of factors, such as coherency, misorientation, and phase composition. 4 Aaron and Weinberg 275 interpreted that the preferential diffusion pathways in a Cu-14.3 at.%Al alloy were related to the incoherent interphase boundaries, where Ag diffusion was significantly enhanced compared to the bulk.
Chadwick 276 measured extremely fast interphase boundary diffusion in eutectic Pb-Sn alloys and explained those rates by a dominating interstitialcy mechanism. These diffusion rates exceeded those for volume diffusion of noble metals (Cu, Ag, and Au) in single crystalline Pb or Sn277,278 by at least three orders of magnitude. 276 Turnbull with co-workers277,278 interpreted the exceptionally high diffusion coefficients of the noble metals in Pb or Sn via an interstitial mechanism. A similar mechanism was suggested for the interphase boundary diffusion in Pb-Sn alloys, too. 276 In contrast, the diffusion rate of Ag along the Ag-Cu eutectic interphase boundaries 276 was found to be about two orders of magnitude lower than the value obtained by Turnbull and Hoffman 279 for Ag GB self-diffusion along low-angle GBs.
The mechanisms of interphase boundary diffusion depend on the coherency relationships and the available misfit dislocations play a crucial role, as has been shown by Sommer et al. 280 in their study of Ag diffusion in the Ag/Cu interphase boundaries. In the latter case, diffusion was anisotropic, strongly depending on the misorientation, with the highest values observed along the [011] directions, see Figure 26(a).
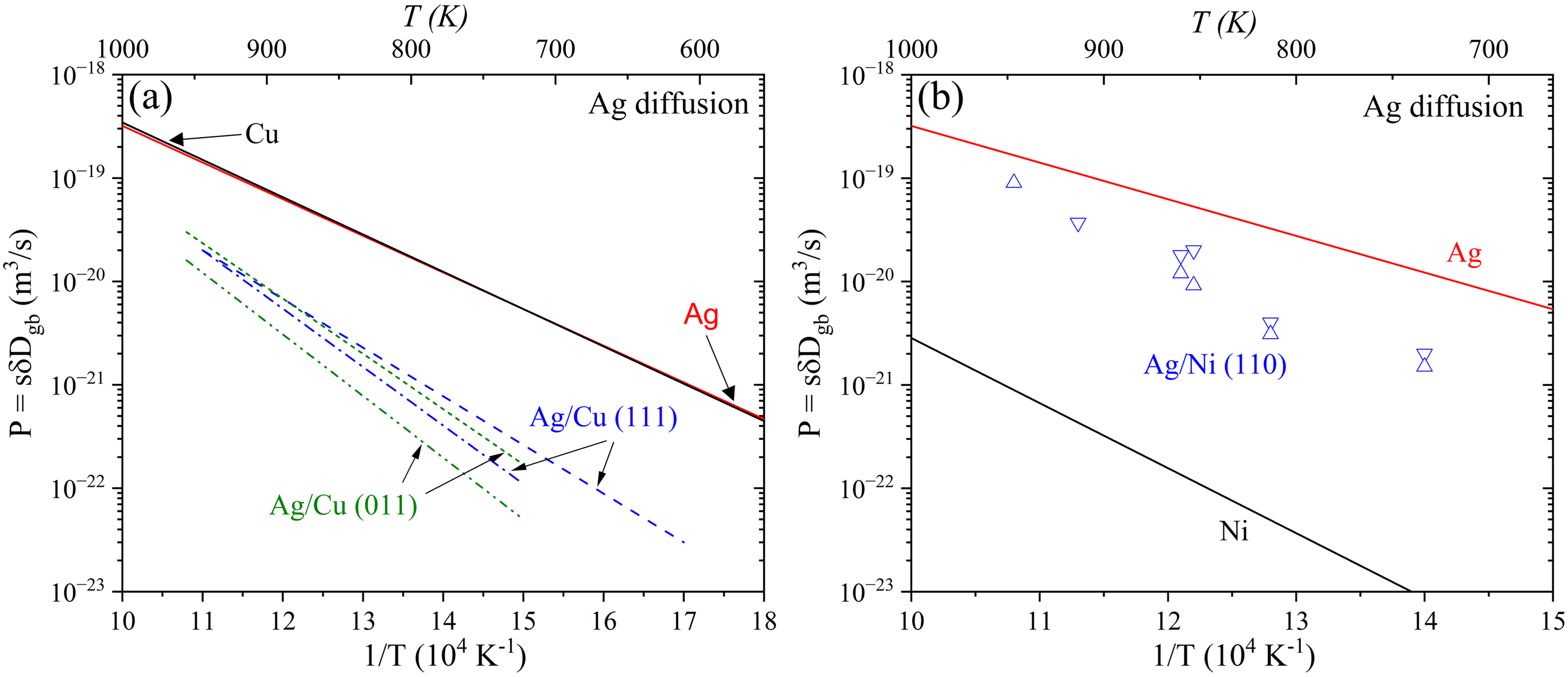
Ag interphase boundary diffusion in comparison to Ag GB diffusion in pure elements: (a) Ag diffusion along Ag/Cu (111)- and (011)-oriented interphase boundaries measured parallel to
Minkwitz et al.
281
further highlighted the influence of specific atomic transport mechanisms, showing that the triple product for Ag diffusion along the Ag/Ni interphase boundaries (e.g., about
Figure 26 suggests a strong dependency of the interphase boundary diffusion on the chemical nature of the elements. Whereas it is systematically slower than Ag GB diffusion in both, pure Cu and Ag (the latter are almost identical on the absolute temperature scale), Ag diffusion along the Ag/Ni (110) interphase boundary is in between the Ag GB diffusion rates in pure Ag and Ni.
Straumal et al. 282 demonstrated that boundary structures could either enhance or decrease diffusion rates depending on the interface misorientation and the energy states, with interphase boundaries at special misorientation angles exhibiting lower excess Gibbs free energies and higher activation energies for diffusion.
In the past, autoradiography has been used for quantifying diffusion kinetics and for visualizing the diffusion pathways in polycrystals.34–36,283 Time-of-flight secondary ion mass spectroscopy (ToF-SIMS) with 3D resolution can also be used to distinguish the predominant diffusion pathways. 120 In CCAs, interphase boundary diffusion is strongly affected by chemical complexity, phase stability, and temperature, with the activation energy being significantly influenced by alloy composition and boundary structure. While interphase boundary diffusion shares similarities with grain boundary diffusion, unique factors such as misfit dislocations and phase coherency create distinct diffusion behaviors.
Recently, Heng et al.
120
measured short-circuit diffusion of Ni in the multi-principal element eutectic AlCoCrFeNi
Interestingly, at elevated temperatures above 800 K, Ni diffusion along the coherent or semi-coherent FCC/B2 interphase boundaries in eutectic AlCoCrFeNi
It is important to note that the distinct diffusion behavior along interphase boundaries not only offers key fundamental insights but is also highly relevant from an engineering standpoint for alloy design. The strong sensitivity of diffusion rates to coherency, misorientation, and chemical interactions suggests that interphase boundaries can be strategically tailored to control mass transport in complex alloys. In eutectic HEAs, such as AlCoCrFeNi2.1, interphase boundary diffusion dominates over grain boundary diffusion, underscoring its importance in governing creep resistance, phase stability, and high-temperature performance. Coherent or semi-coherent boundaries provide pathways for thermal stability and corrosion resistance, whereas incoherent boundaries enhance diffusivity and may accelerate degradation. Grain boundary engineering concepts can thus be extended to interphase boundaries, where controlling orientation relationships, defect densities, and segregation tendencies becomes a design tool. By coupling experimental diffusion studies with predictive modeling, interphase boundary diffusion can be harnessed to develop next-generation structural alloys with tuneable stability, improved service reliability, and application-specific performance.
Computational assessment of grain boundary and interphase boundary diffusion
Thermodynamics of grain boundary phases and grain boundary phase transitions
Presently, there exist numerous experimental examples of characteristic structural transformations at GBs in materials from pure metals to binary and even multi-component alloys, Sections “Grain boundary structure transitions” and “Grain boundary phase transitions in multi-component alloys”. With critical advances in atomically-resolved characterization methods of GB structures,9,10 our understanding of GB phase equilibria and GB phase transitions relays on the computer simulations. Originally, the compelling evidences for the structural transformations in high-angle GBs were provided by atomistic modelling.141,142 Frolov et al. 141 elaborated adequate simulation methodology which account for variations in the GB atomic density and unambiguously revealed multiple GB phases with different atomic structures. Frolov and Mishin presented a thermodynamic theory of plane coherent solid-solid interfaces in multicomponent systems and provided expressions for the GB free energy as the reversible work of GB formation under stress284,285 and analyzed phases, phase equilibria, and phase rules in low-dimensional systems. 206
Following a phase-field approach, Kamachali 286 proposed a model to assess the GB thermodynamics through modifying the relative atomic density field and its spatial gradients within the GB regions with a reference to the homogeneous bulk. In this way, the GB Gibbs free energy functional was consistently developed that allowed calculating the GB segregation isotherms and the GB phase diagrams.147,286 It was shown that the GB structural variations can amplify segregation transitions and can also stabilize co-existing spinodal interfacial phases, 287 as they were observed, e.g. for Ni-based, 21 see Figure 12, or Fe–Mn-based alloys. 288
Thermodynamic (phase-filed) modelling was used by Ikeda et al. 289 elaborating on how temperature, GB type and the chemo-structurally coupled phase decomposition at the GB impact the segregation transition in the case of a Fe–Zn system. Segregation transitions were found to be related to spinodal decomposition into Zn-rich phases within GBs. GB phase diagrams were constructed and a dependence of the miscibility gap on the type of the interfaces (in terms of the relative GB density) was shown. 289
Zhou, Hu and Luo 143 proposed the concept of “high-entropy grain boundaries” as the GB counterparts to high-entropy materials. The “high-entropy grain boundaries” are considered to be distinct from GBs in high-entropy materials and should not be considered as “compositionally complex GBs”. 148 Luo and co-workers related specific thermodynamic characters to the high-entropy GBs.11,148,290 The high-entropy GBs were suggested to be characterized by both, structural and chemical disorder and might stabilize, e.g., nanocrystalline alloys at high temperatures via thermodynamic and kinetic effects. 148 An interplay of kinetic and thermodynamic stabilization mechanisms in binary alloys was recently analyzed by several groups of authors291,292 and HEAs seem to offer ”intrinsic” mechanisms combining the two types of phenomena.
Zhou et al. 11 developed a systematics of GB segregation transitions and critical phenomena. In this way, the classical GB segregation theories12,293,294 were elaborated. The GB layering and prewetting transitions are sketched in Figure 27. In so-called ”strong” segregation systems, 11 first-order layering transitions occur at low temperatures and become continuous above GB roughing temperatures, Figure 27(b, c). ”Weak” segregation reduces the layering transitions, forcing them to gradually merge, causing a prewetting transition without quantized layer numbers to appear, as in the Kikuchi–Cahn 295 critical-point wetting model. Zhou and Luo proposed GB complexion diagrams with universal character based on the regular solution description of alloy thermodynamics.11,148
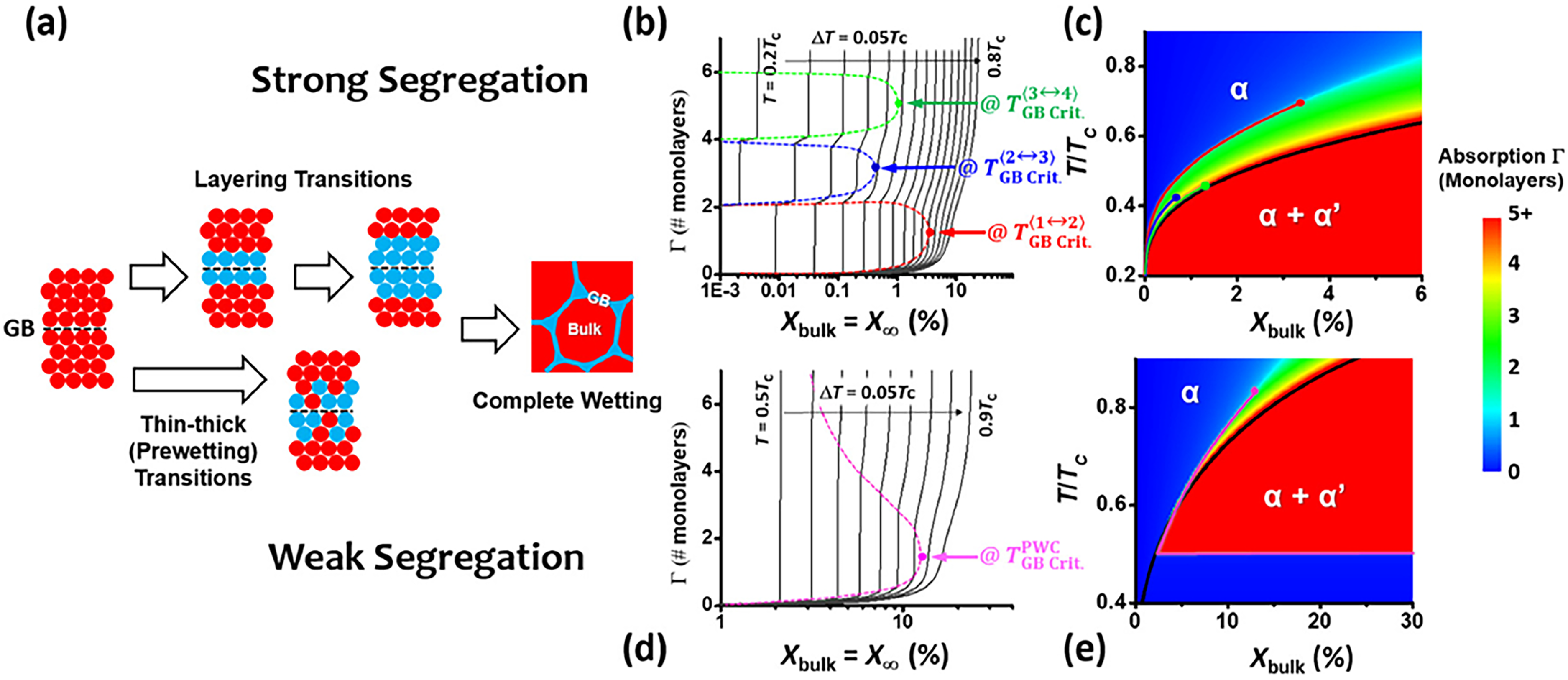
(a) Schematic illustration of GB layering vs. prewetting transitions.
11
Comparison of calculated GB segregation transitions for (b,c) strong and weak segregation systems. The calculated
Using the density-based thermodynamic model, 286 Li et al. 210 reveals that the highly negative energy of mixing Ni and Mn is the main driving force for the experimentally observed nanoscale cosegregation to the GBs. This is further assisted by the opposite segregation of Ni and Cr atoms with a positive enthalpy of mixing. It is also found that GBs of higher interfacial energy, possessing lower atomic densities (higher disorder and free volume), show higher segregation levels. By clarifying the origins of GB segregations in the FeMnNiCoCr HEA, the current work provides fundamental ideas on nanoscale segregation at crystal defects in multicomponent alloys.
Grain boundary segregation models for high-entropy alloys were extensively analyzed by Luo. 290 Differing from classical models where one component is taken as a solvent and others are considered solutes, these models are referenced to the bulk composition to enable improved treatments of HEAs with no principal components. Ideal and regular solution models were presented. 290 This analysis is a convenient framework for addressing GB segregation in multi-component alloys.
While the volumetric properties of high-entropy alloys have been widely studied and discussed with respect to the configurational entropy of mixing, there is a lack of general information regarding the configurational entropy of the grain boundaries. Lejcek with co-workers derived the basic relationships of this thermodynamic quantity related to the solute segregation at grain boundaries. 249 It was stated that the role of grain boundary configurational entropy in interfacial properties is not completely clear and represents a challenge for future research.
Atomistic modeling of grain boundary structures
Atomistic simulations are directly related to the availability and correctness of the interatomic potentials. Empirical interatomic potentials are typically used for large-scale simulations of atomic GB structures in high-entropy alloys296–299 due to obvious limitations of more advanced (and accurate) schemes with respect to the computational speed.
Wynblatt and Chatain 296 modelled GB segregation in equiatomic CoCrFeMnNi and found strong a Cr segregation at 1200 K which is accompanied by a weak Mn segregation. It was suggested that a significant depletion of bulk composition can occur if the grain size drops below about 100 nm. 296 Though, GB diffusion experiments 118 have not confirmed any measurable GB segregation in the alloy. An absence of a significant segregation in the equiatomic CoCrFeMnNi HEA at elevated temperatures was also concluded by atomistic simulation of Farkas. 298 However, the GB structures are significantly affected by the chemical complexity, see Figure 28. Furthermore, Barnett at al. 300 showed that chemical complexity enhances GB absorption efficiency for point defects widening the available microstate range of accessible GB structures.
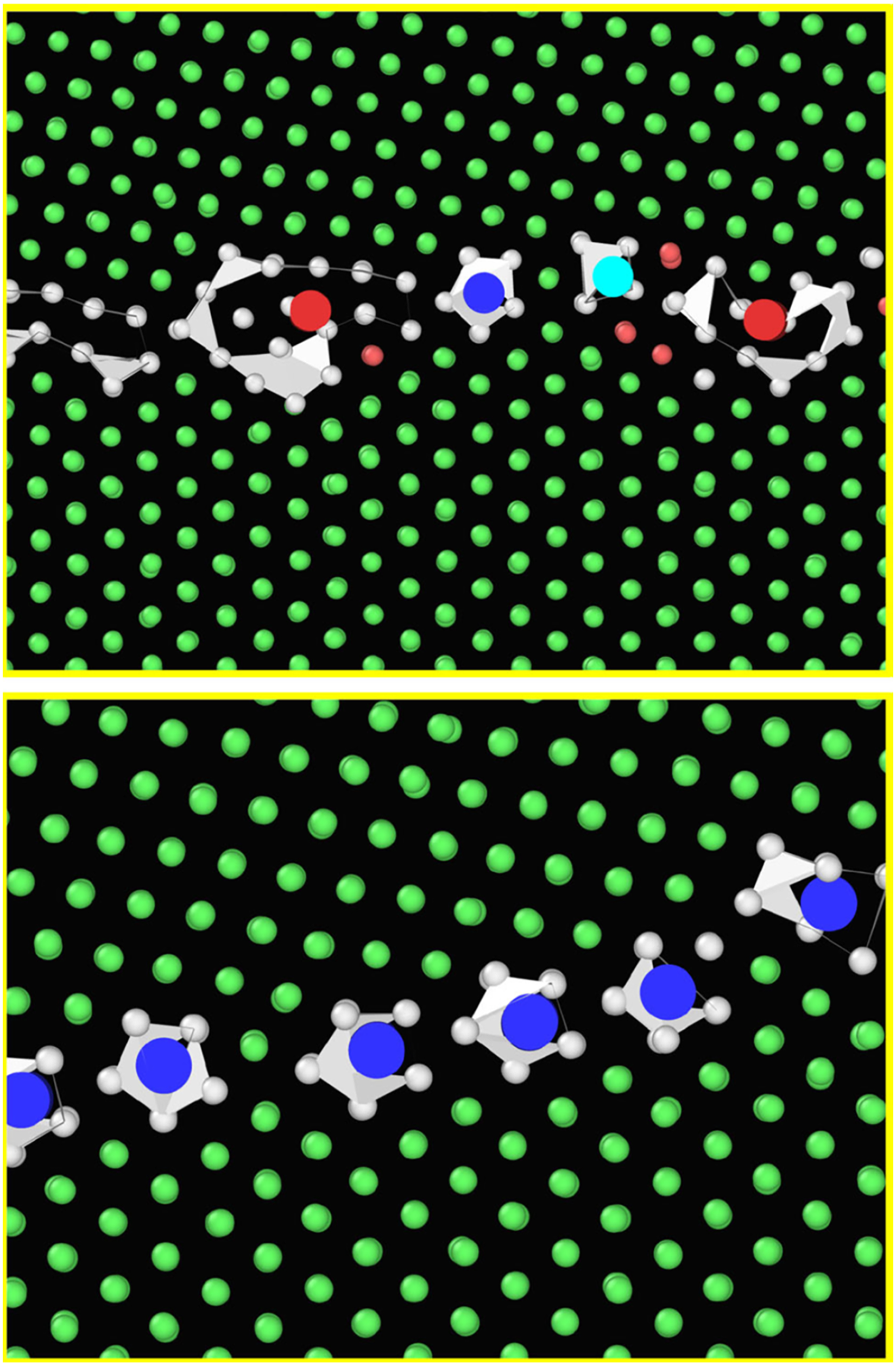
Using empirical potentials, Wang et al.
301
quantified solute interactions and segregation to
The atomistic simulation studies296–299,301 underlined the importance to understand and quantify the chemical interactions within GBs, including GB–defects interactions, especially for designing novel MPEAs.
The classical molecular dynamics and molecular statics simulations are limited in predictive accuracy for specific alloy systems. Though, the performed comparative studies of GB structures, 298 as well as of GB diffusion 302 and migration, 297 in terms of a direct comparison of the predictions for empirical potentials and for an ”average atom” potential are general and very valuable. However, the predictions for any specific system have to be treated with caution.
These issues, at least for structural and thermodynamic properties of GBs, are largely bypassed by recent advances in machine-learning interatomic potentials (MLIPs), which improved substantially the quality of atomistic simulations leveling them to almost DFT accuracy.303,304
MLIPs overcome the challenges of high computational costs in DFT and the relatively low accuracy in classical (largely empirical) interatomic potentials. 305 For example, the Graph Atomic Cluster Expansion (GRACE) potential 304 is parameterized across the periodic table as a GRACE foundation model. 306 In this way, thousands or millions of atoms with complex chemistries are simulated for extended time scales that opens completely new routes for materials discovery and design. The GRACE foundation model 304 allows accurate and predictive analysis of co-segregation effects in real materials accounting for a spectrum of available impurities, the task hardly accessible previously. Generally, machine learning is becoming a method of choice for modelling complex materials and processes, see e.g. the recent reviews with respect to formulations, frameworks and existing challenges.307,308
Still, these developments with respect to GBs in solids are presently limited to unary, binary and several ternary systems. Ito et al.
303
constructed MLIP and computed energy, atomic structure, and dynamics of general GBs in
Xie et al. 305 combined effective two- and three-body potentials in a cubic B-spline basis with regularized linear regression to obtain MLIPs that are sufficiently accurate for applications and as fast as the fastest traditional empirical potentials that directly allows large-scale simulations. Explicitly accounting for symmetry relations, a MLIP surrogate model has been suggested 309 to address the long-standing issues of GB structure–segregation property relationships. With this, the segregation isotherms were computed for a spectrum real GBs. 309
Tuchida and Schuh 310 used MLIPs to assess solute segregation at finite concentrations and temperatures accounting for solute-solute interactions and excess vibrational entropy of segregation. As a result, a self-consistent spectral database for 16 elements comprising 240 binary alloy polycrystals were generated. 310
Ou et al.
67
developed MLIPs and performed large-scale, long-time, and high-accuracy simulations of complex atomic structures and diffusion mechanisms in Li
Atomistic modeling of grain boundary diffusion
Although MLPIs demonstrated immense progress for addressing structural and segregation properties of random high-angle GBs in complex materials, they application to GB diffusion is presently impeded by the challenging computational costs. The application of direct MD
67
or advanced thermodynamic integration schemes for transition states
311
are largely limited by pure metals or highly symmetric low-
Seoane et al.
302
performed extensive molecular dynamics simulations of grain boundary diffusion in a model equiatomic FeNiCrCoCu HEA. Particularly, diffusion along
It was found that GB diffusion in the model HEA is slower than that in the ”average atom” metal as well as with respect to the average of the pure components, Figure 29, left panel. These findings support the “sluggish” diffusion concept, at least, for the special
Junhong and Weiqiang
315
performed molecular dynamics simulations of grain boundary diffusion in CoCrFeMnNi HEA. The vacancy formation energy and atomic migration energies were determined for both, the crystalline bulk and for
High-entropy alloys are known to reveal an increased stability against grain growth, 316 which can be related to a decreased GB mobility or decreased driving force for GB motion. Baruffi and Curtin 317 have shown that an initially flat GB can become spontaneously rough, driven by natural local compositional fluctuations. The roughening decreases the GB energy and the driving force for curvature-driven grain growth. GB segregation and/or second phase precipitation decreases further the GB energy and thus GB mobility.
The recent computational developments with respect to GB-related phenomena in HEAs were reviewed by Ferrari et al. 318 The key achievements concerning the usage of MLIPs for very accurate thermodynamics, impact of short-range order on the macroscopic properties, and developments of interstitial alloying for emerging technological applications are presented in detail, 318 though the analysis was limited to bulk and surface properties.
Atomistic mechanisms of GB diffusion can effectively be studied using a combination of ab initio calculations and MD simulations, as it was recently reported for GB self-diffusion in
For example, Simonnin et al. 63 used large-scale molecular dynamics simulations and examined vacancy-mediated diffusion in model Ni–5at.%Cr alloy with low and high-energy GBs. Diffusion at elevated temperatures of 1300–1600 K was studied. It was found that Cr diffusion is faster than Ni in the bulk but slower in GBs. 63 The phenomenon was attributed to formation of Cr clusters at the grain boundaries. 63
Analysis of the GB diffusion mechanisms is largely limited to pure metals and binary alloys. Li et al. 320 analyzed in detail the mechanisms of GB, dislocation and triple junction diffusion in the Al/Si layers. Hussein et al. 321 elaborated a densiton concept for atomic diffusion in disordered systems or general GBs. Densitons migrate by either single-atom jumps or concerted rearrangements of 2–3 atoms 321 and a remarkable similarity between the strings in disordered and crystalline structures was revealed.
Jha et al.
322
performed intensive simulations of GB diffusion in the Ni–Nb alloys and found that the GB diffusivities of solute and solvent atoms exhibit distinct composition and temperature dependencies. The GB diffusion rate of Ni atom decreases first with Nb alloying due to preferential segregation of Nb atoms at the most favorable sites (as ”trapping” or originally ”poisoning” effect of Bernardini and Gas,
185
see Chapter 33.1). However, with an increase of Nb concentration (and of temperature), the GB structures become disordered, modifying the GB diffusion mechanism.
322
The analysis of Jha et al.
322
was performed for a wide spectrum of
A detailed analysis of the diffusion mechanisms in terms of propensities of the specific GB structures to trap or facilitate diffusion of defects and atoms in HEA GBs is still pending, despite of a huge progress in evaluating the bulk diffusion properties.323–326
Atomistic GB simulations provide a required knowledge of the diffusion properties of internal interfaces in HEAs which is used as input parameters for mesoscale phase-field models enabling prediction of long-term evolution of GB-related properties.
Interphase boundary diffusion
Recently, Mishin with co-workers performed detailed modeling of diffusion along Al–Si interphase boundaries.64,65,320 The atomistic computer simulations allowed to trace characteristic diffusion mechanisms for such metal-nonmetal interfaces. Diffusion of both Al and Si atoms along the Al–Si interfaces was found to be slow compared to the GB diffusion rates of the two elements in Al, 64 in line with an early experimental report of Chadwick 276 for Ag–Cu eutectic boundaries. However, interface disconnections were found to accelerate the interphase boundary diffusion. Atomic mechanisms of interphase boundary diffusion were reported 276 to be similar to those in metallic grain boundaries. Correlated atomic rearrangements in the form of strings and rings of collectively moving atoms were found to dominate the atomic transport on the time and length scales of the MD simulations. 64
To the best of our knowledge, atomistic simulations of interphase boundary diffusion in multi-component alloys are simply absent, though, in view of the immense progress with MLIPs one might expect further advances in a near future, see Section “New horizons for grain boundary engineering and elaboration of predictive mesoscopic models” below.
The convergence of machine learning interatomic potentials (MLIPs), tomographic GB mapping, and advanced simulations opens a transformative pathway for interface engineering in HEAs. By linking experimentally accessible GB character distributions with predictive models of segregation, energy, and diffusion, it becomes possible to design GB networks that actively enhance performance rather than passively limit it. In HEAs, where configurational entropy, lattice distortion, and sluggish kinetics intersect, such predictive tools could identify optimal boundary chemistries and orientations for targeted application such as creep-resistant GBs in refractory HEAs, corrosion-resistant networks in marine alloys, or crack-resistant boundaries in TRIP-HEAs. Additive manufacturing adds another dimension, where metastable non-equilibrium boundaries can be stabilized or relaxed through data-guided post-processing. Ultimately, extending GB engineering into the ML-accelerated design space paves way for rational GB network design in HEAs for several engineering applications.
Grain boundary structure–property relations for high-entropy alloys
New horizons for grain boundary engineering and elaboration of predictive mesoscopic models
The chemical complexity of HEAs and generally of MPEAs opens new perspectives for GB engineering. A wide spectrum of GB-inherent characteristics – GB character distribution, GB segregation, phase decomposition and second phase precipitation – can be engineered to improve a broad range of mechanical and corrosion properties of advanced alloys. Atomic-level distortions characteristic for the HEAs influence the dislocation–GB interactions and propencity of GBs to act as dislocation sources. Thus, next-generation GB engineering can be achieved in complex concentrated alloys, including single-phase HEAs.
You et al.
327
applied GB engineering concepts to optimize the GB character distribution and achive improved tensile properties of a transformation-induced plasticity-assisted high-entropy alloy. The authors discussed evolution and mechanisms of GB character distribution in detail
327
and found optimized processing parameters to increase the fraction of
GB engineering in HEAs can be achieved not only via optimization of the fraction of the
In additively manufactured alloys, in addition to a common spectrum of interfaces, melt pool boundaries are present and often act as weakening spots due to presence of brittle phases, residual stresses, and coarsened microstructures. As a result, these melt pool boundaries limit significantly the tensile ductility in additively manufactured alloys. Li et al. 330 proposed engineering of melt pool boundaries to mitigate the severe limitations of additively manufactured alloys by introducing nano twins at those boundaries. Tailoring the W segregation and its solubility in the FCC matrix a promising route for improving mechanical properties in HEAs has been elaborated. 330
The propensity of multi-principal element alloys for phase decomposition, the tendency which is enhanced at interfaces, can be used as a further GB engineering approach to stabilize grain growth by intermittent annealing treatments in suitable temperature–time windows provoking GB phase separation and/or secondary phase precipitation. For example, in Cantor-based systems, precipitation of the Cr-rich sigma phase might be helpful as a source of Cr atoms for oxidation protection and precipitation of L1
A specific GB engineering method via GB wetting was proposed and discussed by Straumal et al.
331
Formation of chains or continuous layers of second-phase particles along GBs was observed in many HEA systems. The GB wetting by a second solid phase was proposed to analyze constructing GB tie-linesin the two- or multiphase areas in the multicomponent phase diagrams for HEAs.
331
Chen et al.
332
used one-step recrystallization to engineer the GB character distribution in CoCrFeMnNi, CoCrFeNi, and CoFeNi. It was shown that CoFeNi is promising material for GB engineering with the highest fraction of special GBs and twin density.
332
Wang et al.
29
have shown that GB segregation engineering can enhance the strength–ductility synergy in CoCrNi medium-entropy alloys by boron alloying. GB engineering can be used to improve functional properties of HEAs, as e.g. both mechanical properties and corrosion resistance of Al
Finally, GB engineering via dedicated alloying and concomitant manipulation of the GB energy, see Section “Impact of alloying on grain boundary energy”, in multi-principal element alloys might be a further strategy which is hardly explored so far. Metalloid substitutions (Si or Ge) were found to elevate simultaneously the strength and ductility of FCC HEAs. 334 Interstitial atoms, like C or N, enable joint twinning and transformation induced plasticity in strong and ductile HEAs.335,336 Addition of strongly segregating elements in HEAs might be followed for optimal fine-tuning their functional properties.
The internal (grain and/or interphase) boundaries affect a wide range of thermal, electrical, optical, and mechanical properties of solids. 31 Moreover, interface structures/defects/phases, thermodynamics, mobility, and diffusion play a significant role in solidification, microstructure evolution, and phase transformations in solids. Phase-field modelling became a powerful tool for computational assessment of these complex processes.337–339 Nevertheless, the interphase properties are typically modelled without a full accounting for the interface bicrystallography, interface defects (steps, kinks, facets, GB dislocations, disconnections 134 ), temperature, impurities, alloying, and the full mechano-chemical coupling, which represent in fact 7D+ configurational space. Already accounting for the anisotropies of GB energy and diffusivity in terms of 5D configurational space improves the phase field modelling significantly. 340 The current state-of-the-art corresponds to the representation of the GB energy via Read-Shockley equation 248 and the atomic mobilities by Humpherey equation 341 overlooking the inclination dependence of these parameters, Section “Misorientation and inclination dependencies of grain boundary diffusion”. These are valuable approximations for symmetric GBs indeed, but they they not only oversimplify the details of the GB diffusion anisotropy (characteristic already for pure metals) presented in this review. More importantly, as the present review documented, the interfaces in multicomponent alloys, especially in multi-principal element alloys, reveal further unique features related to GB segregation, phase decomposition, precipitation and full mechano-chemical coupling related to formation and evolution of GB defects, which are temperature and time dependent in real materials, see, e.g., Figure 16. A proper assessment of these properties is challenging but possible, especially with proper computational efforts.
Grain boundary diffusion-related creep in high-entropy alloys
At elevated temperatures and low-to-moderate stresses, GB-mediated processes often control creep where major mechanisms include diffusional (Coble) creep, grain-boundary sliding and GB-facilitated nucleation/absorption of dislocations and cavitation that accelerate tertiary creep and rupture. 342 These mechanisms depend on temperature, grain size, GB chemistry, structure, morphology, segregation, and/or GB precipitation. 343 A critical influence of GBs on creep in HEAs has also been observed particularly under intermediate to high temperatures. 344
In nanocrystalline or fine-grained HEAs, creep is frequently controlled by GB diffusion or GB sliding, analogous to Coble creep in conventional alloys. Shen et al.
345
demonstrated that nanocrystalline VNbMoTaW exhibits GB diffusion dominated creep, with the slowest diffusing species (Mo, Nb) controlling the creep rate at 973–1073 K. Nanoindentation creep studies in Ref. Lee et al.
346
showed that nanocrystalline HEAs deform faster than their coarse-grained counterparts. The creep stress exponent,
Atomistic simulations by Zafar et al. 27 revealed a size dependence of creep, predicting that sub-15 nm grains favor GB sliding and diffusional flow, while larger grains facilitate a transition towards dislocation-mediated creep. It was also revealed that the local chemical order at GBs especially in HEAs can suppress sliding and improve creep resistance.
Beyond size, GB chemistry and segregation are central to the creep behavior. Atom probe studies on the Cantor alloy revealed strong Cr segregation at GBs, with Mn enrichment to a lesser extent, altering GB energy and potentially enhancing or degrading diffusional creep depending on the segregating element.
211
Such findings challenge the oversimplified “sluggish diffusion” paradigm and instead highlight element-specific GB diffusion as a governing factor – in a full agreement with direct radiotracer diffusion measurements.222,347 Lee et al.
26
demonstrated the role of GB morphology on enhancing the creep resistance. An Al
Molecular dynamics simulations have depicted the effects of short-range order on the high-temperature creep performance of TiVTaNb refractory high-entropy alloy. 348 The analysis considered the short-range order, steady-state creep rate, and creep mechanisms across different samples and showed that in nanocrystals, having a high fraction of grain boundaries, the GB energy of constituent elements influences the short-range order formation. Since short-range order affects creep behavior, tuning the GB energy could provide an indirect strategy to enhance creep resistance. The short-range order markedly suppresses atomic diffusion, especially at grain boundaries, and causes notable differences in the diffusivity of different elements. This leads to the formation of enrichment zones containing fast-diffusing species, which undergo pronounced deformation and result in heterogeneous strain distribution.
The above studies underscore the role of GBs in regulating the creep behavior of HEAs. While fine grains and active GB diffusion accelerate creep, the strategic GB engineering via chemistry (segregation control), morphology (serration, twin networks), and topology (stable GB networks) can significantly enhance high-temperature performance. Future alloy design should therefore prioritize controlling GB chemistry, structure and transport, integrating advanced characterization (APT, high-resolution TEM) with computational modeling to optimize creep resistance in HEAs.
Summary and outlook
The present overview documents the complexity of GB diffusion in multi-principal element alloys. In fact, an intricate interplay of chemical complexity, segregation, secondary phase precipitation and phase decomposition in addition to macroscopic and microscopic misorientation parameters and temperature- and/or segregation-induced phase transitions (complexions) complicates the creation of generic ”GB diffusion maps” as it was done, e.g., for the binary Ni–Bi alloys. 116 Even simple visualizations of such maps would be intricate in view of the multi-dimensionality, at least 7D+, of the problem. In Figure 30, potential impacts of chemical complexity, precipitation of coherent/incoherent particles, and of segregation are sketched for a Ni-based alloy as a projection from a five-dimensional space for a GB with fixed macroscopic degrees of freedom (misorientation/inclination).
Schematic impact of the configurational entropy in single phase (random solid solution) FCC alloys and potential influence of GB segregation (blue arrow), coherent precipitation (red arrow), and incoherent precipitation (green arrow). These trends are sketched for an arbitrary selected general high-angle GB corresponding to a single point within the 5D configurational space determined by the GB bi-crystallography. Direct accounting for the GB bi-crystallography will effectively increase the dimensionality of the problem by further 5 coordinates corresponding to the GB misorientation and GB plane inclination.
This scheme is sketched for a general, symmetric tilt high-angle GB in a Ni-based alloy, in which an increased chemical complexity of the random solid solution (the ”configurational entropy” axis) retards diffusion, as well as potential segregation of an alloying element, like W or Mo (the ”segregation” axis). However, segregation of a low-melting point element like Bi is expected to induce layering/premelting phase transitions and would give rise to an enhancement of the GB diffusion rates. The impact of precipitation might be two-fold. Nano-scale coherent particles at GBs induce local strains and generate networks of GB dislocations which would enhance GB diffusivity (the ”coherent precipitates” axis). On the other hand, incoherent particles, typically with higher melting point, would provide diffusion barriers and a retardation of GB diffusion is expected (the ”incoherent precipitates” axis).
In a real material, GB diffusivity depends further on the macroscopic misorientation parameters, as GB misorientation and inclination, GB defects (faceting, dislocations, disconnections, etc.), purity with respect to the critical (typically interstitially solvable) elements like C, O, S, or P, see Section “Misorientation and inclination dependencies of grain boundary diffusion”. In view of co-segregation and segregation-induced structure transitions, including different scenarios of stress/strain relaxation, the problem is not only multi-dimensional, but intrinsically sophisticated in view of governing mechano-chemical coupling phenomena. GB precipitations (semi-coherent/incoherent) provide further facets to the complexity of the governing mechano-chemical coupling phenomena and expected modifications of the GB diffusion rates, see Section “Impact of chemical complexity on grain boundary diffusion in FCC solid solutions”. Although ultimate mapping of the GB diffusion properties in terms of all hypothetically possible degrees of freedom is still illusive, a critical mass of fundamental understanding with respect to working structure–property relations seems to be accommodated or, at least, clear roadmaps for their assessment are formulated. Some coarse-graining with respect to the whole spectrum of interfaces and their atomistic structures in real materials is inevitably required, allowing acceptable level of the uncertainties of the GB diffusion properties in a given polycrystal.
GB diffusion measurements in bicrystalline samples for multi-principal elements alloys, largely absent so far, are required to provide deeper insights from experimental perspectives. Influence of the microscopic parameters, giving rise to multiplicity of GB structures, and GB defects, which are further degrees of freedom, can be investigated as well. Impurity elements, deliberately or unintentionally added to the material, can influence the GB structures affecting GB diffusion (of both, matrix and alloying elements, probably in different ways). Such studies would be highly valuable and expected to provide further aspects of GB structure–property relations in multi-component systems.
Though, to the best of our knowledge, GB diffusion in bicrystals of CoCrFeNi and CoCrFeMnNi was measured only in a single study.
349
Those bicrystals were occasionally produced during the fabrication of corresponding single crystalline alloys.223,350 Unfortunately, the grain boundaries, produced by a chance, were largely curved, featuring varying inclinations at different locations, and numerous small-angle GBs were present as well. The GB diffusion measurements have to be extended to carefully prepared and characterized bicrystals with designed interfaces. The CoCrFeNi and CoCrFeMnNi HEAs are stable at elevated temperatures and a progressive phase decomposition at GBs was seen after annealing for prolonged times only.
24
In this case, GB diffusion properties of pristine GBs can carefully be evaluated. GB diffusion measurements at lower temperatures (about 700 K) in CoCrFeNi and CoCrFeMnNi revealed a strong impact of phase decomposition (formation of the
Having performed a critical set of experiments and/or computational work, we believe that a diagram like Figure 30 can be constructed for other HEA families, e.g. refractory HEAs which are extremely important for advanced applications. Such a generic view present a roadmap for GB engineering of technological alloys depending on the spectrum of properties which are required, see discussion in Section “New horizons for grain boundary engineering and elaboration of predictive mesoscopic models”.
Diffusion along the interphase boundaries is a further important subject for future studies, both experimentally and theoretically. Recently, two investigations reported important insights into the corresponding diffusion mechanisms in multi-principal element alloys.25,120 It was found that deviations from exact orientation relationships between FCC and BCC phases influence the interphase diffusion considerably. 120 Systematic investigations with respect to the influence of bi-crystallography of the interfaces, misfit strains, and chemical complexity are required.
In a diffusion couple, an enhanced interdiffusion can occur along grain boundaries.351,352 The contribution of GBs during formation and growth of intermetallic phases during interdiffusion was recently highlighted.353,354 GB interdiffusion can lead to lateral GB motion as diffusion-induced GB migration (DIGM) or give rise to GB diffusion-induced solid state reactions. 355 Typically, the former are observed in systems forming a wide range of solid solutions and the latter were reported for systems containing intermetallic phases. 355 Interdiffusion of elements with different atomic radii gives rise to the appearance of elastic strains which in turn influence the GB interdiffusion rates (in addition to Kirkendall stresses356,357).358–360 Typically, DIGM takes place on both sides of a binary thin film, although the solute content in the DIGM zone is higher on the side of the component with a higher melting point. 355
In addition to DIGM and resulting alloying, nucleation and growth of entirely new grains via diffusion-induced recrystallization (DIR) can occur. 361 Interestingly, the new grains correspond to homogeneous random solute solutions which are characteristic for the diffusion couples. 361 In thin films, the coherency stresses could be very high and DIR occurs as a break of the coherency by spontaneous relaxation. 361 Similar phenomena are expected in multi-component materials, too, but they are still to be investigated.
Dislocation diffusion in multi-principal element alloys presents a further missing block in the present knowledge of the diffusion phenomena in such materials. The GB diffusion measurements have relied heavily on the radiotracer measurements, which is arguably the most sensitive tool to probe the complexities associated with atomic transport along the boundaries. Given the limited availability of tracer facilities, and the increasing focus on multicomponent alloys, it is also important to develop alternative methods to estimate the GB diffusivities. Techniques such as secondary ion mass spectrometry (SIMS) have been used to measure grain boundary diffusivities in Ni. 362 However, due to its limited penetration depth, SIMS primarily captures diffusion along relaxed, slow grain boundaries and does not resolve the faster, deformation-modified boundaries. As a result, it has inherent limitations in analyzing the full spectrum of grain boundary diffusivities. The development of computational tools to assess the GB energy, structures and diffusivites in multicomponent alloys will also be crucial.
Recent advances in atomic modelling with the use of MLIPs open perspectives of a predictive simulation of the diffusion properties of (almost?) arbitrary interfaces with respect to the basic (experimentally accessible) 5D microscopic parameters and even for complex chemical environments. 67 One may imaging tomographic measurements of GB character distribution in a real material (done e.g. for pure metals or binary alloys77–79,363) with subsequent calculations of the interfaces’ ground states at the given temperature(s) and simultaneous assessment of the corresponding GB (including those of interphase boundaries, if present) diffusion properties. This vision is inseparable with a progress on fast, robust and predictive interatomic potentials, presently associated with the machine learning methods.
The chemical complexity of grain boundaries in HEAs has provided a new dimension and opportunity to expand the realm of grain boundary engineering. GB segregation, phase decomposition, precipitation, and GB character distribution can be tailored to achieve desirable structural and functional properties. Experimental advances, such as the optimization of
Predictive modeling of these complex boundaries remains limited by oversimplified assumptions, such as isotropic GB energy or diffusion behavior. State-of-the-art approaches, including phase-field simulations, already highlight the benefits of accounting for GB energy and mobility anisotropy, yet fully capturing the configurational space of GB defects, chemistry, and mechano-chemical coupling remains an open challenge. Looking ahead, advancing GB engineering strategies will require a paradigm shift from conventional thermodynamic equilibrium approaches toward more nuanced, application-specific methodologies. One promising direction is the development of “complexion engineering” in HEAs. The deliberate stabilization of specific GB complexions, chemically and structurally distinct interfacial states can provide avenues to tailor interfacial energies, mobilities, and segregation behaviors, thereby unlocking new levels of control over microstructural evolution and performance in extreme environments. Simultaneously, the surge of additive manufacturing introduces unique non-equilibrium thermal profiles and microstructural features that challenge traditional GB engineering frameworks. Here, ”non-equilibrium GB engineering”, focused on exploiting transient thermal gradients, rapid solidification pathways, and kinetic stabilization of metastable boundary configurations can enable the design of tailored boundary networks post-build or even in situ during printing. This might involve directed energy deposition techniques combined with real-time thermal management or post-processing heat treatments informed by in-situ diagnostics and modeling. Furthermore, integration of non-equilibrium behaviour in computational tools coupled with advanced characterization tools can lead to modern predictive approach that combines material complexity and processing realities.
Understanding the GB structure and transport is critical to deciphering the creep behavior in HEAs. The current literature demonstrates that when grain sizes are fine (micrometer-large) or nanocrystalline, creep is dominated by GB diffusion and GB sliding, with element-specific diffusion kinetics rather than a universal “sluggish diffusion” effect dictating deformation rates. 27 Segregation phenomena, as observed in the Cantor alloy 211 and various refractory HEAs, 345 highlight how GB chemistry alters the GB energy and mobility, thereby accelerating or suppressing creep. At the same time, morphology-driven strategies such as serrated boundaries, 267 twin networks and stabilized GB structures 28 have been experimentally shown to suppress GB sliding and extend creep rupture life significantly.
The future work to establish a stronger correlation between the GB characteristics and creep properties can have multiple facets. A systematic quantification of GB diffusion coefficients across different HEA families is needed, ideally using tracer diffusion or isotope experiments coupled with high-resolution methods (atom probe tomography or analytical TEM) to resolve the segregation effects. Predictive modeling must integrate atomistic simulations with CALPHAD and mesoscale frameworks to link the GB chemistry and local order to macroscopic creep response. Such multiscale approaches will be crucial for bridging the gap between nanoscale mechanisms and component-level performance. It is also imperative to look ahead integrated design strategies that combine the GB chemistry control, morphological engineering, and microstructural stabilization. Such approaches have the potential to unlock alloys with superior creep strength, ductility, and oxidation resistance, enabling their deployment in aerospace turbines, nuclear reactors, and next-generation energy systems.
In conclusion, the research roadmap ahead should not only focus in optimizing the intrinsic diffusion properties of HEAs, but also involve strategically engineered GBs with respect to chemical, structural, and topological parameters to tune the materials’ structural and functional properties for advanced technological applications.
Nomenclature of the symbols used thoughout the paper.

Footnotes
Acknowledgments
A partial financial support from the German Research Foundation (DFG) via the research projects DI 1419/19-1 (number 467491887), DI 1419/24-1 (number 509804947), and WI 1899/44-1 is acknowledged.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from the German Science Foundation (DFG) via research grants DI 1419/19-1 (number 467491887), DI 1419/24-1 (project number 509804947), and WI 1899/44-1 is acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.


