Abstract
Alginate has gained extensive attention due to its versatile properties and numerous applications in different fields. Due to the abundant availability of raw alginate in nature and its advantage of being easily recycled, alginate-based materials are conducive to advancing the shift away from an economy dependent on fossil fuels to a more environmentally friendly and sustainable one. In addition, its inherent biocompatibility makes alginate a fascinating biomedical material. This comprehensive review presents a detailed analysis of alginate, focusing on its structural characteristics, physicochemical and biological properties, current processing technologies, and modification strategies. This review explores the diverse applications of alginate-based materials in biomedicine and healthcare, considering drug delivery systems, tissue engineering, and wound dressings. Additionally, this review looks into the environmental applications of alginate in biosensors and waste-water treatment. The challenges associated with alginate utilization and future perspectives for research and development are also presented. This review provides a comprehensive overview of alginate, offering valuable insights into its properties, applications, and potential for future applications across various industries.
Introduction
In the last decade, research in material science has paid much attention to materials which can be produced sustainably. Sustainable materials, also called “green materials”, are substances that are usually derived from natural materials and utilized by humans in an environmentally responsible way. In addition, the term “green materials” can encompass non-toxic materials that are environmentally benign and provide safer options for human life.1,2 Alginate is a naturally occurring polysaccharide, and thus can be extracted from sustainable sources of raw materials. It is a material that is very abundant in nature. This biological resource possesses excellent biocompatibility, minimal toxicity, favorable hygroscopic properties, and is capable of degradation, making it ideal for human use.3,4 It is derived from brown seaweed and can also be extracted from microbial fermentation using bacteria such as
Recently, researchers have been paying increasing attention to the development of alginate fibers, owing to their origin from sustainable raw materials and the fascinating characteristics of their small structures. The generation of fibers greatly broadens the possible applications of alginate materials due to their improved characteristics, including a remarkably high ratio of surface area to volume and tunable porosity. The application of alginate fibers ranges from wound dressings, tissue engineering, drug delivery, wastewater treatment, probiotic bacteria encapsulation, nutrition delivery, sensors, and textiles.7–14 The utilization of alginate-derived fibers across various applications helps to reduce over-reliance on non-renewable resources with environmentally deleterious implications.
Searching in the prevailing literature, numerous review articles on alginate are evident. However, there remains a dearth of comprehensive critiques focusing specifically on alginate in its fiber form. This review article discusses alginate fibers in a comprehensive manner and includes a thorough and objective discussion of their properties, manufacturing methods, properties enhancement, applications, and future research opportunities. It serves as a foundation for future research and promotes the utilization of alginate fibers in diverse industries, which could ultimately lead to advancements in healthcare, biotechnology, and materials science.
Structure and properties of alginate
Structure
Chemically, alginate is composed of random copolymerized blocks of β-1,4-D-mannuronic acid (M) and α-1,4-l-guluronic acid (G), linearly linked to each other by 1–4 glycosidic bonds.
15
The polymer consists of consecutive G-blocks (G-G-G), consecutive M-blocks (M-M-M), and alternating M and G-blocks (M-G-M).
16
Figure 1 depicts the chemical structure of a short alginate chain and the M and G blocks that comprise the alginate structure. These monomers can be arranged in different sequences along the alginate chain, leading to varying properties and functionalities. The ratio of M and G monomers in alginate determines its polymer structure and behavior.
17
Alginates with enriched G units have a rigid molecular structure, whereas alginates with enriched M units show a flexible structure and greater biocompatibility.18,19 In addition, the M/G ratio is crucial in determining the drug release behavior, antibacterial properties, and wound healing efficiency of alginate-based materials in their biomedical applications. According to Liew et al.
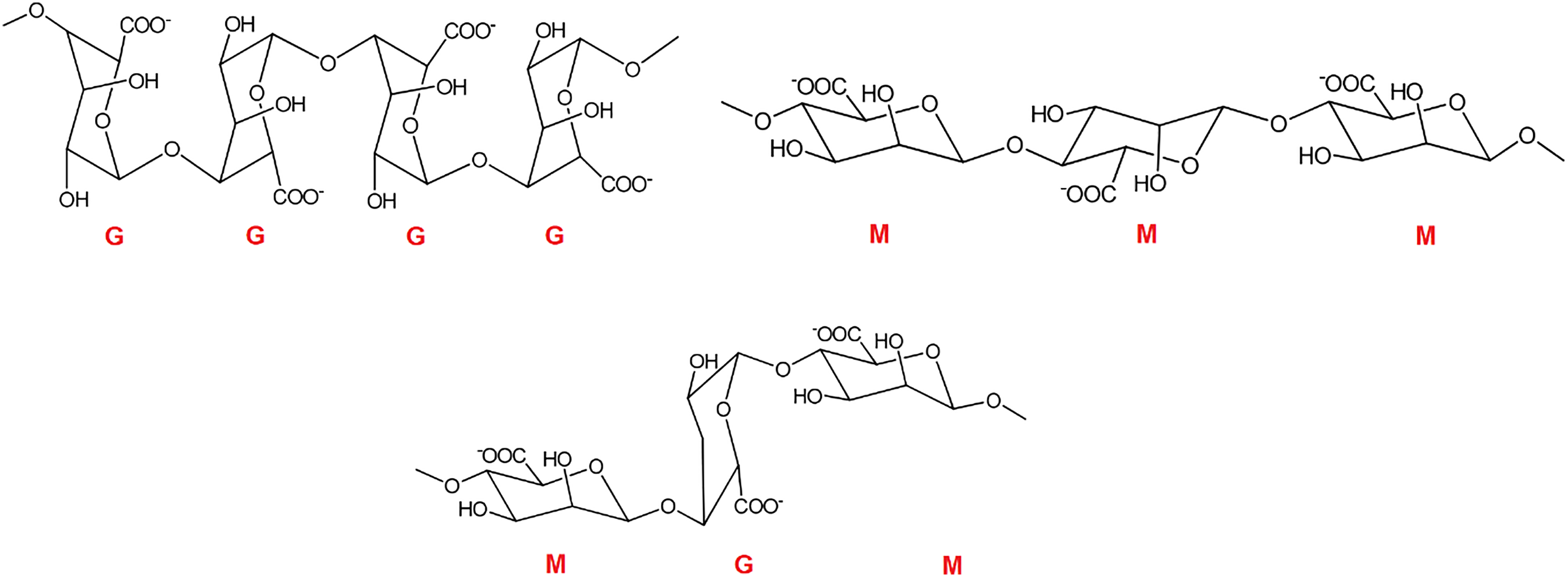
Chemical structures of alginate with different distributions of M and G monomers, GGG (G-block), MMM (M-block), and MGM/GMG (alternating block).
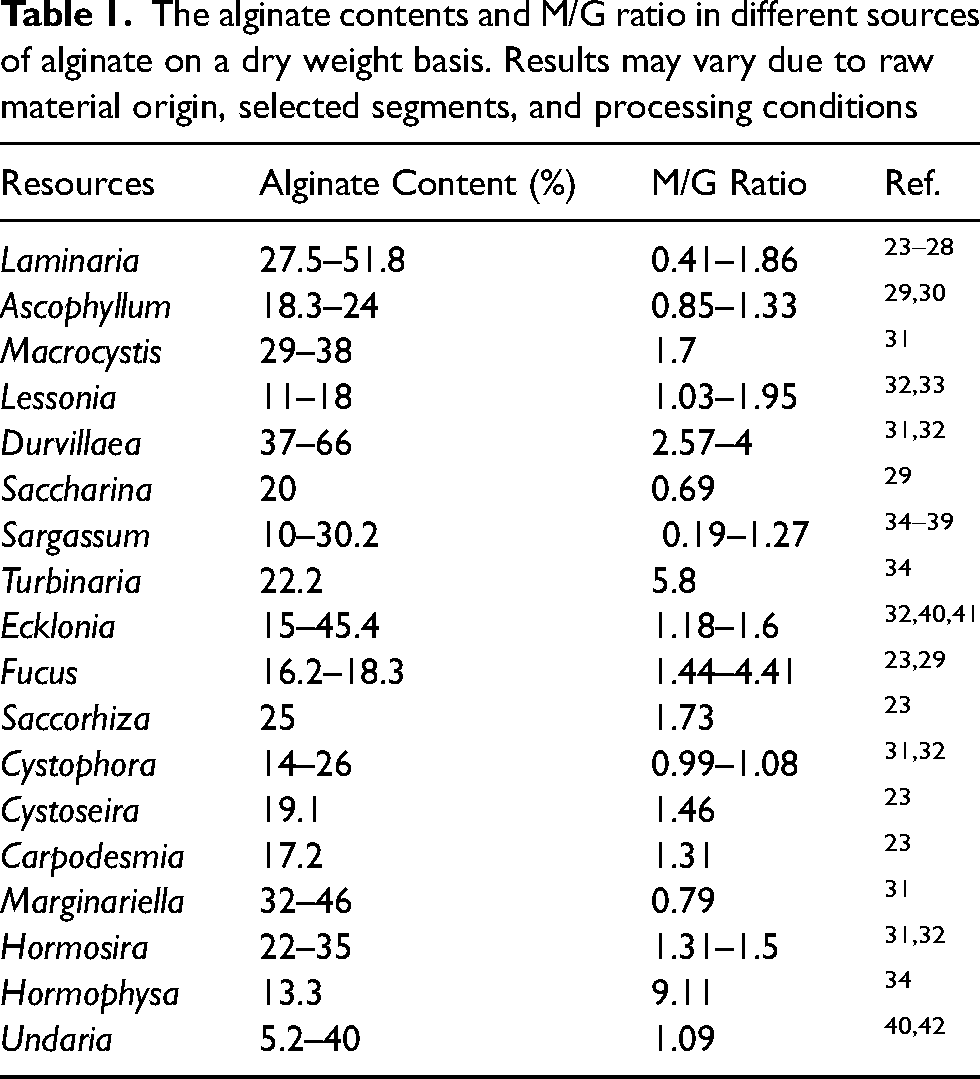
The alginate contents and M/G ratio in different sources of alginate on a dry weight basis. Results may vary due to raw material origin, selected segments, and processing conditions
Physical properties
Alginates can form physical hydrogels via intermolecular chain interactions. When alginate is exposed to solutions that are enriched with divalent cations, particularly Ca2+, the divalent ions establish ionic cross-linkages with the negatively charged carboxylate groups in the alginate chain. 43 This cross-linking reaction leads to gel formation, where the alginate chains intertwine to create a three-dimensional structure.44,45 The egg-box model describes the interaction between alginate chain carboxyl groups and divalent cations (Figure 2). Carboxyl groups are present in the G units of the alginate chain and can chelate with Ca2+ to form an egg-box-like structure. In this structure, the alginate chain serves as the box, and the Ca2+ as the egg. The formation of this structure and the strength of the resulting gel network depends on multiple factors, including pH, temperature, and co-solute. 46 Higher molecular weight or increasing the concentration of gelling ions, for instance, leads to the construction of a denser and more robust gel network. Whereas a low pH of 3.8 can lead to a non-uniform gel structure with increased wrinkles and cavities. 47 Additionally, Puguan et al. 48 reported that excessively high pH levels (pH > 10) result in a significantly increased gel volume. An increase in temperature is reported to promote the formation of a dense network structure in alginate gel, with alginate density increasing from 1.03 g/cm3 to 1.12 g/cm3 as the gelation temperature rises from 5 °C to 85 °C. 49
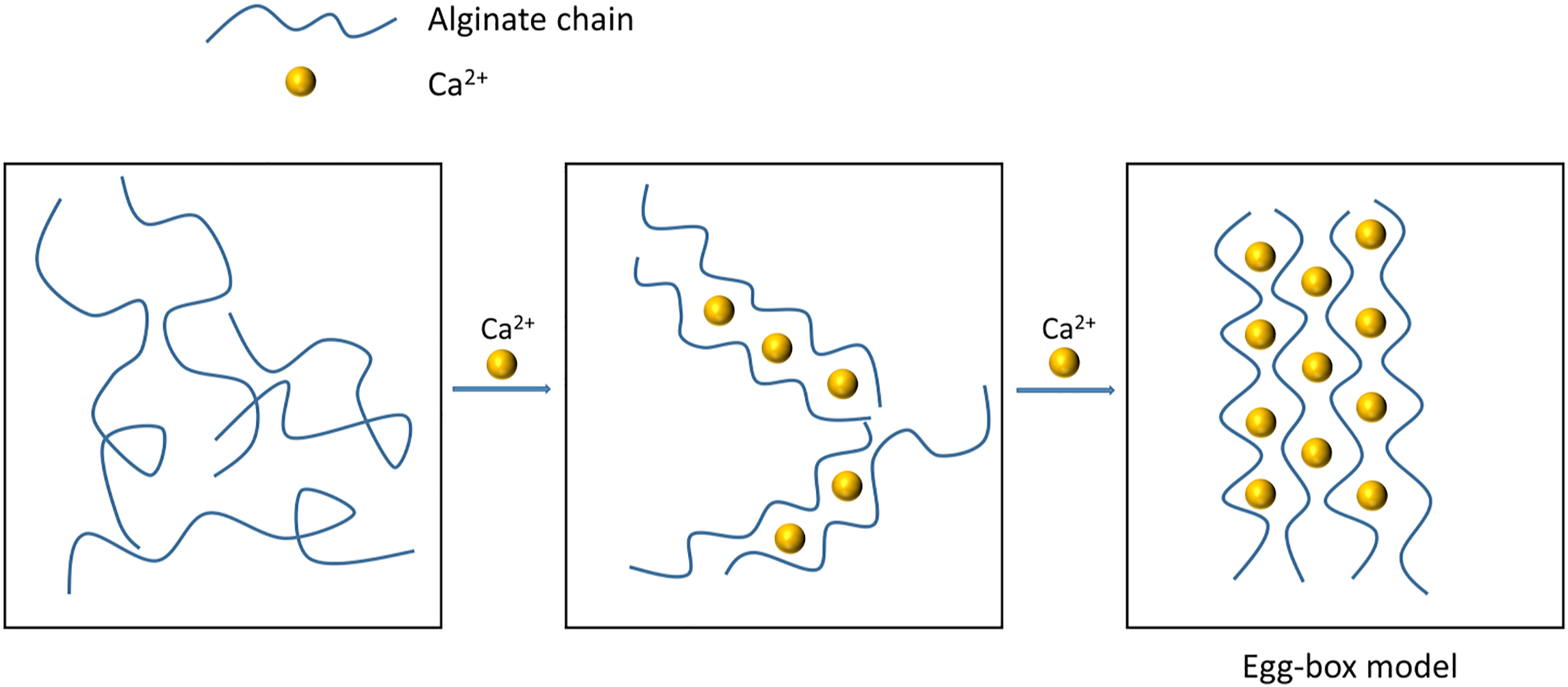
Ionic cross-linking between Ca2+ and alginate chains.
This is attributed to the enhanced diffusion rate of Ca2+ under a high temperature. Conversely, gelation at lower temperatures promotes the construction of a more regular internal structure, with the resulting alginate gel containing a lower Ca2+ content. These properties make alginate an alternative material for applications requiring gelation, such as drug delivery. In drug delivery applications, an active pharmaceutical ingredient (API) is encapsulated in the alginate gel matrix, which could then be delivered to the patient. The alginate gel will dissolve or degrade after it reaches the target and release the API in a controlled manner.16,50
Alginates containing divalent or trivalent cations do not dissolve in water because these cations bond very strongly to the terminal carboxylic ion (-COO-), creating an insoluble product. Instead, they can absorb up to 300 times their weight in water, forming a hydrogel with a paste-like consistency. In contrast, alginates containing monovalent cations like Na+, K+, and NH4+ dissolve readily in either cold or warm water. The solubility of alginates varies depending on their molecular weight. 51 Alginates exhibit enhanced solubility at higher pH. 52 At lower pH values, the alginate chain undergoes protonation, which reduces the charge density and leads to reduced solubility. It was reported that below pH 2.3, even a degraded product was only partly soluble. 53 Furthermore, the ionic intensity of the dissolving medium has a major impact on the solubility of alginate. This influences solubility through effects on solution characteristics, including polymer conformation, chain extension, and viscosity. 54
Biological properties
Alginate possesses a number of beneficial characteristics, including biocompatibility, non-toxicity, non-immunogenicity, and biodegradability.
55
As a biomaterial, alginate does not induce inflammation, cell death, or tissue damage.56,57 While alginate typically exhibits minimal immunogenicity in its high-purity form, impurities can trigger an immune response. The
Calcium alginate (CA) exhibits several potential healing properties. 60 When in contact with wound exudate (the fluid discharged from wounds), CA forms a gel substance that fosters a favorable environment for wound healing. This gel not only safeguards the wound against damage but also stimulates new tissue growth. Moreover, alginate is considered a biocompatible material due to its inherent biodegradability. Enzymes in the body have the capability to metabolize alginate into sugars, which makes it a safe and compatible material. This property is particularly important in applications where the material needs to be removed or replaced over time. The rate at which alginate degrades depends greatly on factors such as the degree of oxidation, pH, and temperature of the media.16,61 Another important biological behavior of alginate is its capability to sustain cell growth and tissue regeneration. 62
Mechanical properties
The mechanical characteristics of alginate differ due to multiple factors, commonly involving material source, molecular structure, molecular weight, cross-linking degree, and processing method. The egg-box region enriched in G units exhibits a higher affinity for divalent metal ions, particularly Ca2+.63,64 This strong binding capability facilitates the gelation process of alginate. Consequently, alginates rich in G units exhibit higher mechanical strength and stiffness.65–67 Conversely, a higher M unit alginate content leads to a flatter and shallower nest, resulting in weaker binding with Ca2+. 68 Thus, these alginates have increased flexibility and lower mechanical strength. 64
Additionally, a higher molecular weight leads to enhanced chain entanglement and physical cross-linking of alginate, thereby improving its mechanical strength. Cross-linking methods, including ionic gelation, chemical cross-linking, and photo-crosslinking, play a crucial role in modulating the mechanical properties of alginate. Covalent cross-linking, compared to physical cross-linking, leads to the formation of a robust three-dimensional network structure, effectively enhancing the mechanical strength. 69 This transformation is usually irreversible. Nevertheless, alginate typically possesses relatively low mechanical strength due to its amorphous structure. Consequently, identifying suitable strategies to adjust the mechanical properties to meet specific application requirements is vital for alginate processing, as discussed further in subsequent sections of this review.
Overall, the mechanical properties of alginate fibers can vary significantly due to the influence of several factors. According to Zhu et al.
70
and Liu et al.
Common methods to enhance the mechanical properties of alginate-based fibers and their resulting mechanical strength.
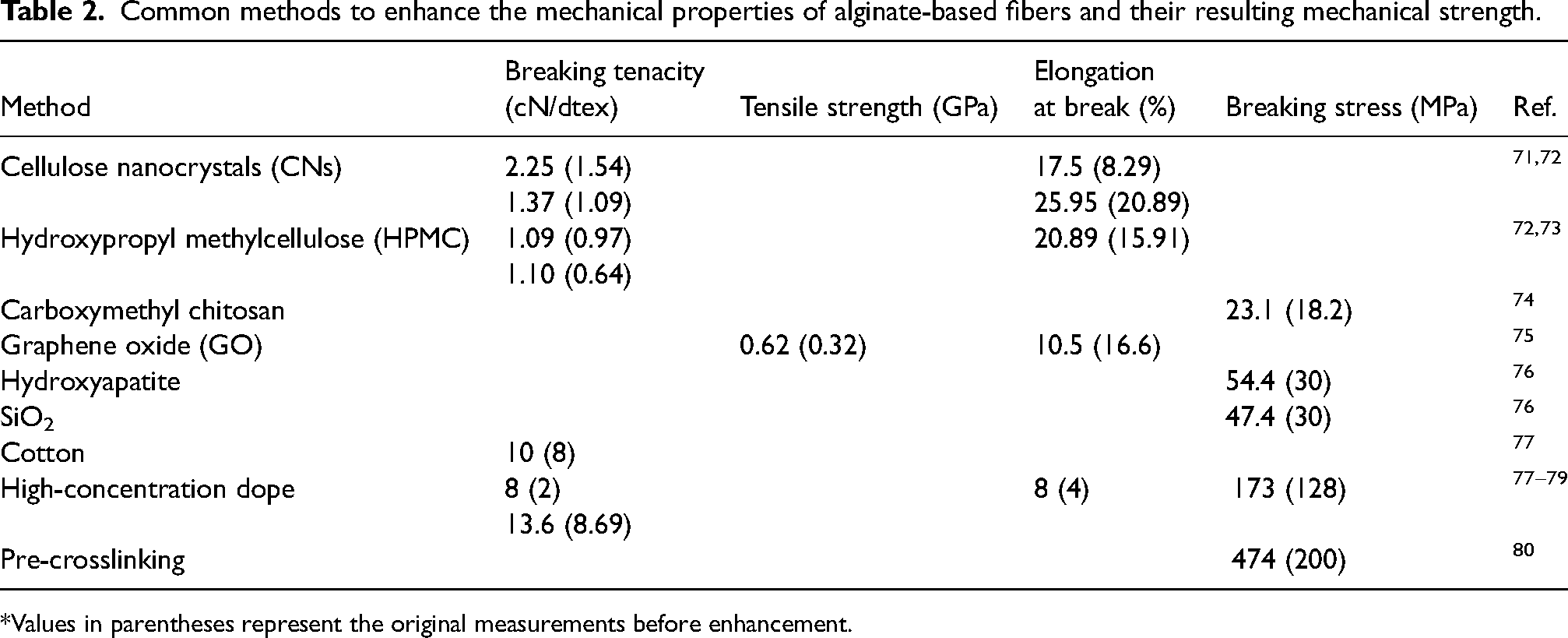
*Values in parentheses represent the original measurements before enhancement.
Degradation of alginate
The degradation of alginate refers to the cleavage of glycosidic bonds in its polymer structure, which is generally associated with a reduction in the average molecular weight and a change in molecular weight distribution. 81 This process is influenced by various factors. Principally, alginate degradation occurs through spontaneous hydrolysis or enzymatic hydrolysis. In aqueous solutions or an environment containing sufficient moisture, alginate hydrolyzes spontaneously. In addition to the inherent characteristics of the alginate material (such as molecular weight, degree of cross-linking, and composition of alginate molecule), the degradation rate is also influenced by extrinsic environmental factors, such as temperature, pH value, presence of ions, etc. Understanding the interplay of environmental factors on degradation rates is critical for the storage duration, shelf life, and disposal process of alginate materials and alginate-based products. Enzymatic degradation of alginate is inherently non-spontaneous in airborne environments or in mammals due to the absence of alginate-degrading enzymes (i.e., alginase, generally isolated from brown algae). 82 However, ionically cross-linked alginate gels can be dissolved by releasing their divalent cations (i.e., Ca2+, Ba2+, Zn2+), subsequently engaging in ion exchange with monovalent cations (i.e., Na+) in the surrounding environment. Overall, avoiding high temperatures and moisture in the air is conducive to prolonging the shelf life of alginate and preserving its material stability. 83 Studies conducted by Hondrum et al. 84 revealed that under controlled storage conditions (room temperature of 20.5° ± 1°C and relative humidity of 40 ± 10%), the tested alginate materials exhibited negligible deterioration over 78 months, indicating their sustained performance.
This review mainly explores the application of alginate in the biomedical field, emphasizing the importance of understanding its
Manufacturing alginate fibers
To maximize the application potential of alginate, several techniques have been explored to process it into fibers, which are renowned for their distinctive large specific surface area. Table 3 summarizes the recent studies which have processed alginate into fiber materials and their prospective application areas.
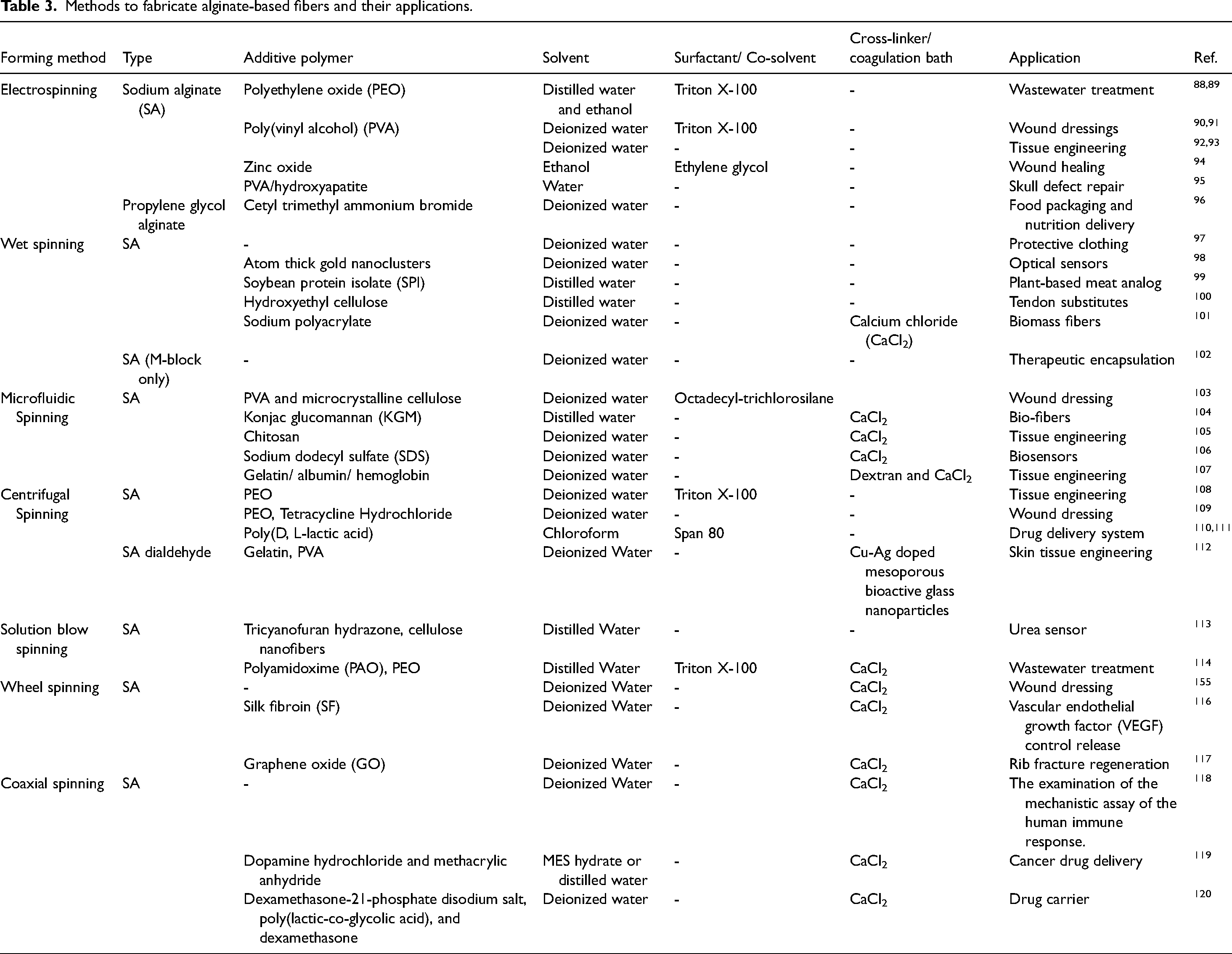
Methods to fabricate alginate-based fibers and their applications.
Electrospinning
Electrospinning involves utilizing electrostatic force to generate thin polymeric fibers with diameters ranging from several micrometers down to the nanoscale.120,121 A schematic diagram of this method is shown in Figure 3(a). The formation of a Taylor cone is achieved by subjecting the polymer solution, expelled from a needle, to a high voltage. 127 This process involves the extension of the hemispherical surface of the polymer solution. The cone then forms a charged polymer jet that solidifies into fibers upon reaching the collector.120,121,128 Electrospinning has been used to explore a range of polymeric materials, including both synthetic and organic polymers.129,130 However, the process is sensitive to several parameters, such as the solution and processing parameters, and environmental conditions. Therefore, finely controlled alginate fiber fabrication by electrospinning remains challenging. 121
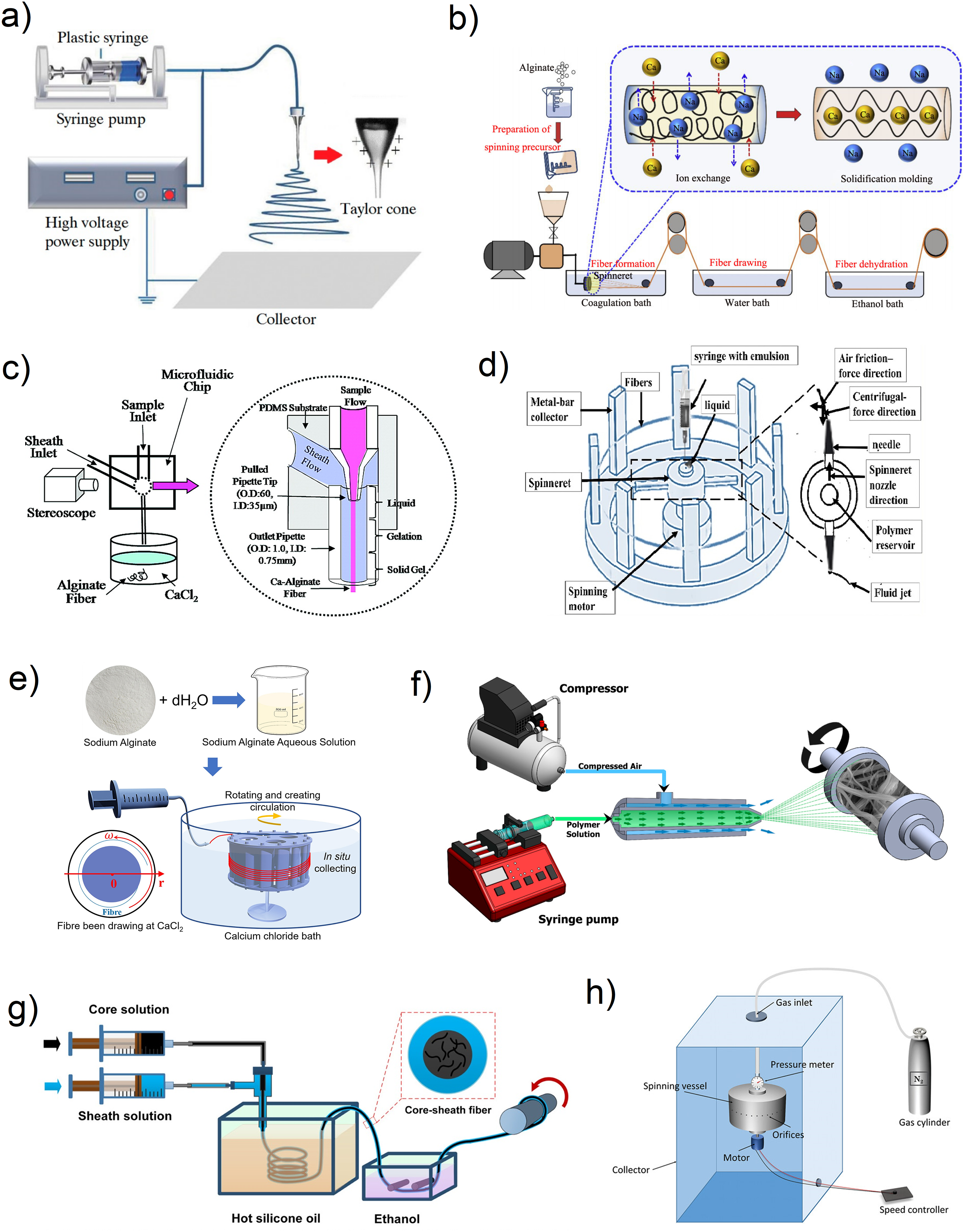
Schematic diagram illustrating different methods of alginate fiber manufacturing; a) electrospinning, 122 with permission from MDPI, licensed under an open access Creative Commons CC BY 4.0 license; b) wet spinning, 123 with permission from Elsevier (copyright 2022); c) microfluidic spinning, adapted with permission from. 8 Copyright 2007 American Chemical Society; d) centrifugal spinning, 110 with permission from Elsevier (copyright 2022); e) wheel spinning, 79 with permission from Elsevier, licensed under an open access Creative Commons CC-BY license; f) solution blow spinning, 124 with permission from MDPI, licensed under an open access Creative Commons CC BY 4.0 license; g) coaxial spinning, 125 with permission from Springer Nature (copyright 2021); and h) pressurized gyration, 126 with permission from Wiley, licensed under an open access Creative Commons CC BY 4.0 license.
However, as shown in Table 3, electrospinning is one of the most widely used techniques for making alginate fibers. The properties of electrospun alginate fibers can be customized by modifying various parameters, including polymer concentration, solvent type, applied voltage, and collection distance. These parameters influence the fiber diameter, morphology, and alignment. 131 Unfortunately, alginate has poor electrospinnability due to its polyelectrolytic nature and solid hydrogen network. 132 The high electrical conductivity causes its tendency to be highly charged and thus presents a challenge in the processing of alginate solutions. In addition, the decrease of chain entanglements, gelation occurring at low concentrations, and high surface tension all play a role in alginate's poor electrospinnability.132–135 Surfactants such as Triton X-100 are commonly used to enhance the spinnability of alginate in electrospinning. 136 Triton X-100 effectively reduces the surface tension of alginate solutions, preventing aggregation of alginate molecules and minimizing issues like clumping or uneven fiber formation. Additionally, it aids in producing finer, more uniform fibers with fewer defects. However, considering the potential toxicity of Triton X-100, exploring non-toxic surfactant alternatives or surfactant-free methods is increasingly appealing.
Wet spinning
Wet spinning is another prevalent technique employed for alginate fiber fabrication. Wet spinning involves extruding a polymer solution through a spinneret into a coagulating bath, as shown in Figure 3(b), where the alginate undergoes gelation or precipitation to form fibers. 137 Alginate fibers are typically generated by extruding the alginate solution into a coagulation bath containing divalent cations, such as CaCl2. 138 The coagulated fibers are then collected, rinsed, and dried. Wet spinning allows the production of continuous alginate fibers with controlled diameter and mechanical properties. The main challenge of producing alginate fibers by wet spinning lies in the gelation process. The gelation of alginate relies on the ionotropic gelation mechanism, where Ca2+ crosslinks the alginate chains to form a network. Due to the brittleness and low tensile strength of alginate, wet-spun alginate fibers often have limited mechanical strength and durability. 139 To address this issue, various techniques have been explored to enhance the mechanical properties of these fibers, as discussed in subsequent sections of this review.
Microfluidic spinning
Microfluidic spinning is a method that uses microfluidic devices to fabricate structures (e.g., such as particles, fibers, and tubes) at the micro- and nano-scale, offering precise control over their dimensions and properties. 140 A schematic of this method is depicted in Figure 3(c). The microfluidic spinning technique provides a wider range of solidification strategies than conventional wet spinning, which is based on the solvent exchange curing process. These strategies include the reactions of photopolymerization, chemical and ionic crosslinking, and solvent evaporation. 141 Microfluidic spinning can produce fibers with desired sizes, structures, and material compositions by controlling factors such as reagent concentration, flow rate, viscosity, and solidification strategy. 141 This technique allows for the continuous production of fibers with a uniform diameter and precise control over their properties. 121 Microfluidic spinning requires reduced amounts of chemicals and simple equipment. Moreover, it can be operated under mild temperature and pressure conditions. 141
Numerous studies have been conducted on fabricating alginate fibers by this method, subsequently advancing to apply the fibers in domains like drug delivery, biomimetic materials, and other areas. Ahn et al. 142 used a microfluidic spinning system to produce isopropyl alcohol-based fibrous alginate drug carriers, which exhibited excellent properties for drug loading and showed promise for wound healing applications. In addition, using a microfluidic spinning device, Zhang et al. 143 and Kang et al. 9 successfully made alginate fibers with a grooved structure capable of mimicking the extracellular matrix and enhancing cell and tissue growth.
Centrifugal spinning
Centrifugal spinning is proposed to be a highly efficient technology capable of producing fibers with a wide range of diameters, from micrometers to nanometers. 108 This cost-effective technique can be used to manufacture fibers at a high production rate. Centrifugal spinning offers the advantage of adjusting porosity, improving mechanical strength, and enhancing the surface-to-volume ratio of the composite nanofiber products. 112 As depicted in Figure 3(d), the process involves forcing a polymer spinning dope through precision nozzles using circular motion. The spinning dope must be in liquid form, which can be achieved through either dissolution or heating. The motor rotates the spinneret around its axis of symmetry. When the angular velocity of the spinneret is above a specific threshold established by the equilibrium between surface tension and centrifugal force, the solution is discharged as a stream through the orifices on the spinneret. 144 As the solvent in the jet evaporates, the material follows a curved path due to the influence of centrifugal force. It continues to move until it reaches the collector's wall. Fiber production is a result of the elongation, distortion, and realignment of the polymer's structure due to high-pressure loss at the nozzle orifice.144,145
Lu et al. 108 utilized this method to produce alginate-rich fibers with up to 97.5 wt% alginate content in an aqueous alginate/PEO system. The key factor enabling centrifugal spinnability of the aqueous alginate/PEO system was the increase of solvent evaporation rate. The primary function of PEO was to establish a hydrogen bonding interaction with alginate, weakening the interplay occurring between alginate and water, and facilitating the formation of fibers.
Ibrahim et al. 111 manufactured variations of polylactic acid (PLA) composites with medium-viscosity alginate for potential medical applications through centrifugal spinning. Composites were prepared with 0.08–0.25 wt% medium-viscosity alginate, while the PLA concentration remained fixed at 6.6 wt%. Composites with medium-viscosity alginate at ≤0.25 wt% and low-viscosity alginate at ≤0.48 wt% exhibited fiber networks interwoven with micro-beads, making them suitable for controlled-drug delivery systems due to the varied surface area. In contrast, using each alginate type at 1.1 wt% in composites with PLA at 6.6 wt% could produce homogeneous fibrous materials more suitable for applications in wound treatment.
Wheel spinning
Wheel spinning is a development of the wet-spinning method where a roller is placed in the coagulation bath to collect the fibers (Figure 3(e)). The working principle involved is similar to that of wet spinning. Firstly, the solution is pumped into a syringe and subsequently ejected into a coagulation bath at a controlled flow rate. The coagulated alginate is then collected and drawn by the wheel system placed in the middle of the fluid container. 116 The wheel spinning directs the fiber emerging from the extruder into the coagulating bath. 79
At the present time, there is limited research that has been reported using this method to produce fibers from alginate. However, Song et al. 115 applied it to fabricate composite fiber for VEGF controllable release. It was shown that the wheel spinning parameters and the morphology of alginate/SF composites have a notable influence on the behavior of VEGF release and the rate of fiber degradation. These effects could be adjusted by changing the concentration of the solution and modifying the morphology of SF.
There is also some work by Chen et al. 79 which revealed that high-strength alginate fibers with a breaking strain of up to 18% and toughness of 16.16 MJ m−3 could be created using the wheel spinning technology. These microfibers exhibited superior overall mechanical properties than others described in the literature.
Solution blow spinning
Solution blow spinning (SBS) is a straightforward and easily scalable technology for producing fibers and has been in use for over a decade. 146 As shown in Figure 3(f), a typical SBS apparatus comprises a gas pressure source, a concentric nozzle, a syringe pump, and a collector. 147 SBS involves two parallel concentric fluid streams, where one stream contains a polymer dissolved in a solvent, and the other is a high-speed air stream. 148 The two streams are directed towards a collector where the solvent evaporates, leaving behind ultrafine fibers that form a non-woven web. The pressurized gas accelerates due to the Bernoulli principle, leading to a reduction in pressure occurring at the interface between the gas and solution. This results in shearing at the interface, stretching the polymer solution towards a collector when the critical air pressure is reached. 146 As the solution droplet is ejected from the inner nozzle, it forms a conical shape due to the high-speed airflow from the outer nozzle. The droplet is then divided into fine streams, which, as they travel towards the collector, are stretched further due to the pressure drop. 149
To date, only a few researchers have reported manufacturing alginate fibers through SBS (Table 3). Xu et al. 114 developed a novel approach for producing PAO/alginate nanofibers using an aqueous SBS on a large scale, using a water-soluble PAO precursor solution. These fibers are suitable for adsorbing uranium after immersion in a CaCl2 solution for crosslinking. Importantly, this approach maintains the 3D porous structure, flexibility, and strength of the nanofibers without a traditional “alkaline-heat activation” treatment, which often causes shrinkage and degeneration.
Coaxial spinning
A schematic diagram of coaxial spinning is given in Figure 3(g). This setup could be regarded as an unconventional wet-spinning method, where two or more solutions are simultaneously extruded through concentric spinnerets, forming core-shell fibers. The core/shell material can be selected based on the desired properties of the final fiber, such as strength, flexibility, or biocompatibility. Coaxial spinning enables the fabrication of alginate fibers with tailored release kinetics and multifunctionality. The core-shell structure provides versatility in controlling the fiber properties and encapsulating bioactive substances within the core. 150 This structure has the potential to address the typical challenges associated with monolithic fibers, such as the initial burst release phenomenon.131,151
Miranda et al. 152 developed a co-axial drug delivery system using wet-spun microfibers for potential applications in chronic wound care. The core of the microfibers consisted of polycaprolactone (PCL) and alanine-alanine-proline-valine (AAPV), while the shell is made of SA and N-carboxymethyl chitosan (NCMC). The fibers demonstrated ideal flexibility, mechanical resilience, and structural stability. Cytocompatibility evaluations demonstrated the modified fiber system was safe when in direct contact with L929 and HaCaT cell lines. The coaxial fibers loaded with AAPV and NCMC hold significant promise to take effect in chronic wound care applications. Coaxial fibers of alginate and polyion complexes loaded with chitosan and chondroitin sulfate were also fabricated using a coaxial wet spinning process by Sagawa et al. 153 The polyion complex-coated fibers exhibited improved strength, durability, and sustained drug release when compared to uncoated alginate fibers, making them suitable for drug carriers.
Pressurized gyration
Pressurized gyration (PG) is a technique that integrates centrifugal spinning with solution blowing to create polymer fibers. 154 When comparing centrifugal spinning to the high-pressure system in PG, the latter enables the generation and stretching of spinning jets more effectively. This leads to a higher production rate, and thinner fibers. The specific advantages of pressurized gyration for fiber customization include highly adjustable processing parameters, easily replaceable spinnerets, and a wider range of material options. 126 In this method, the polymer solution's instability is influenced by the Rayleigh-Taylor interaction caused by the PG equipment (Figure 3 (h)). The polymer solution is displaced due to the amplified centrifugal force resulting from the rotation of the vessel. The gas pressure provided causes the liquid to be expelled from the container, resulting in a difference in pressure. 155 The force exerted on the polymer jet is amplified as it is pushed through the openings, leading to a reduction in jet thickness and, subsequently, the production of finer fibers. 156
To the best of our knowledge, there is only one study that tried to utilize PG to produce alginate-based fibers. Brako et al. 157 blended 5 wt% of medium viscosity SA into 15 wt% of PEO to prepare SA/PEO nanofibers that offer substantial mucoadhesive capabilities. Scanning electron microscopy verified that the fibers are clearly defined, uniformly cylindrical throughout their lengths, and possess high structural integrity. While there is currently no existing literature on the production of pure alginate fibers utilizing PG, Dai et al.158,159 pioneered a novel approach by introducing a coagulation bath into a modified nozzle-PG device, successfully producing pure cellulose fibers through dry-jet wet spinning technique. Given the analogous principles underlying the processing of cellulose fibers and alginate fibers, we hypothesize the feasibility and significant potential of extending this method to the production of alginate fibers. Leveraging the simplicity and high efficiency inherent to nozzle-PG, 158 this innovative method holds promise as a transformative advancement in scaling up alginate fiber production capacity.
Strategies to improve alginate properties for fiber forming
The inherent properties of alginate may not always meet specific requirements. In such cases, strategies can be employed to modulate its properties and expand its potential applications. We set out below the key approaches which have been used to this end.
Molecular weight
One of the primary factors influencing alginate properties is its molecular weight. The molecular weight of alginate is a critical parameter that controls the physical and mechanical properties of the resulting fibers. 160 Alginate with a low molecular weight is required for creating nanoscale micelles. 161 It was discovered by Boontheekul et al. 162 that the degradation rate is governed by both oxidation extent and the ratio of high to low molecular weight alginates used to form gels. Rostami et al. 163 investigated the relationship between the molecular weight and biological properties of alginates extracted by various techniques. Enzyme-assisted extractions yielded alginates with the lowest protein and polyphenol contaminations, which had a molecular weight of 386.4 × 103 g mol−1 after acid treatment.
There are several ways in which the molecular weight of alginate can be modified. The most common method is to reduce the molecular weight through chemical degradation. This deterioration can be accomplished by using an oxidation reaction. Alginate with lower molecular weight obtained by oxidation is reported to show better biodegradability than the parent SA. 164 Phosphorylation is an alternative approach to decrease the molecular weight of alginate. This process produced alginate derivatives with a lower molecular weight than their source material and a diminished gelation capacity. 165 The observed breakdown of the alginates (a 2–4-fold decrease in the molecular weight of the phosphorylated alginate compared to the original polysaccharide) is caused by phosphoric acid. 164 Feng et al. 166 explored the impact of ultrasonic treatment on alginate at different frequencies. The treatment led to degradation and marked changes in the properties of the alginate, such as the viscosity and molecular weight. This indicates a decrease in the M/G ratio after ultrasound treatment. The untreated alginate had molecular weight of 1.927 × 105 g mol−1. Ultrasound treatment decreased molecular weight ranging from 3.50 × 104 g mol−1 to 7.34 × 104 g mol−1. 166
Crosslinking
The crosslinking process involves connecting chains through ionic and/or chemical methods to create hydrophilic materials in diverse forms, including fibers, nanoparticles, films, and beads.
167
Crosslinking is vital to enhance alginate stability,168,169 control drug release,
170
protect embedded ingredients,
171
enhance absorption performance,
172
and improve mechanical properties.173,174 These are all crucial in applications for drug delivery, tissue engineering, energy storage, food technology,
The solubility of alginate in water can be modulated through processes like covalent crosslinking, thermal gelation, and cell crosslinking. Among these methods, ionic crosslinking is the most practical method, with Ca2+ commonly used as the preferred ion. 175 Recent studies have shown that crosslinking alginate-based films with calcium leads to improved tensile strength and cohesion, resulting in stronger films that are less soluble in water. 64 The process of crosslinking alginate occurs during the polymerization of its constituent monomers when exposed to divalent or trivalent cations. 176
Mokhena and Luyt 177 reported notable stability improvement of electrospun alginate nanofibers in water when utilizing dual crosslinking (calcium and glutaraldehyde). Shen and Hsieh 178 conducted an aqueous electrospinning and physical crosslinking approach. The stability of the fibers in water was achieved by employing a dual approach involving the ionic crosslinking of SA and the crystallization of PVA. Stone et al. 179 cross-linked citric acid of electrospun alginate/PVA fibers. As depicted in Figure 4, the thermogravimetric analysis (TGA) curves illustrate that non-crosslinked fibers exhibited a major weight loss peak due to thermal degradation. Crosslinked fibers showed a distinct profile with steeper curves, indicating a slower degradation rate. This suggests that citric acid crosslinking enhances the thermal stability of the nanofibers compared to their non-crosslinked counterparts.
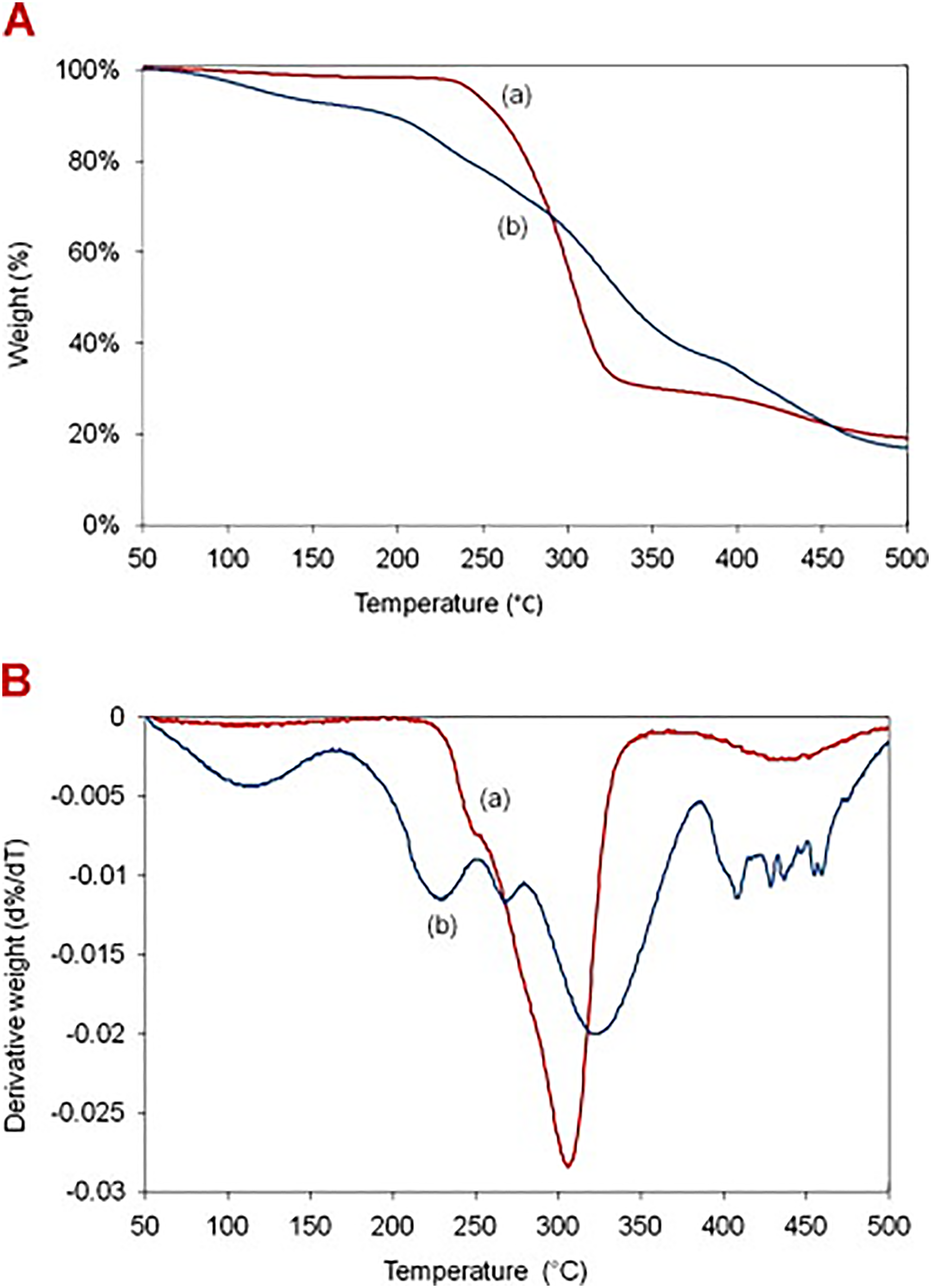
TGA (A) and first derivative weight loss (B) curves of (a) electrospun alginate/PVA nanofibers before crosslinking and (b) after crosslinking, with permission from Elsevier (copyright 2013).
Blend formation
Blending is a useful and efficient way to enhance the properties of polymer materials. In order to create high-performance materials, blending has been employed to combine the benefits of individual materials. 180 As shown in Table 3, alginate can be blended with a variety of different materials to generate fibers and create new applications. The electrospinning process is hindered by the high electrical conductivity of alginate due to its polyelectrolytic nature, which poses challenges in achieving electrospinnability. The addition of other materials to alginate solutions can improve their viscosity and electrospinnability. Safi et al. 181 studied the electrospinnability of SA and its blended systems. The alginate/PVA and alginate/PEO blended systems demonstrated the ability to be electrospun into nanofibers with exceptional fineness and uniformity. Blend formulations containing alginate can improve the mechanical properties of alginate fibers, such as tensile strength and modulus. Dey et al. 182 explored the impact of CA fiber reinforcement in PVA matrix on its mechanical strength. The mechanical properties of the composites were substantially superior to those of the pure PVA matrix.
Blending alginates with natural polymers such as chitosan or gelatin can enhance the biodegradability, biocompatibility, thermal stability, and antimicrobial properties of the resultant fibers. Owing to its relatively rapid degradation and weak bioadhesion, pure alginate is unsuitable for use alone in drug-delivery systems. By adding chitosan to alginate-based nanoparticulate formulations, the duration of contact with the epithelium can be prolonged, and absorption can be improved by opening the tight junctions between cells. Chitosan-alginate nanostructures have been extensively studied for biomedical applications, and they have demonstrated promising results in enhancing
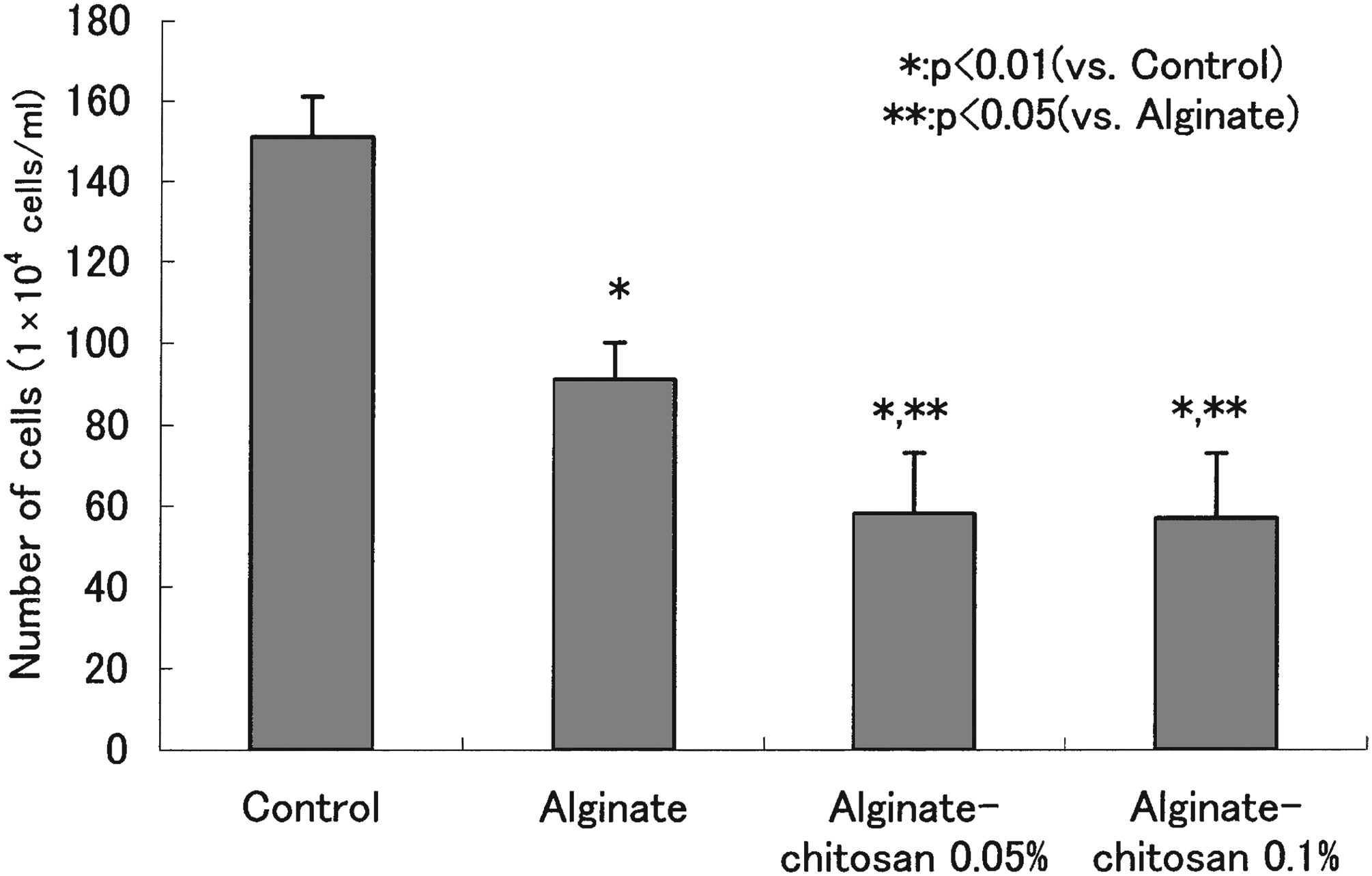
Number of fibroblasts washed out from both the control group and all samples, 184 with permission from Elsevier (copyright 2005).
Alginate–chitosan fibers were also prepared by wet spinning by Sibaja et al. 185 The fibers could deliver a biocidal substance in a controlled fashion that may prevent the growth of bacteria. These fibers are proven to have the desired tensile characteristics and biocompatibility. They are appropriate for creating and manufacturing biodegradable bandages for particular sorts of wounds, such as chronic wound. 186 The fibers create a barrier against bacteria, aid in preventing body fluid movement, and provide a moist environment, which is beneficial for wound healing. Additionally, the high hydrophilicity of these fibers may aid in preventing infections that could make healing more difficult.
Surface modification
Alginate fibers can be treated with various surface modifications to improve their properties. The treatments include a pretreatment process, a nano-finishing process, and a post-treatment process. Antibacterial fibers can be achieved by treatment with an aqueous solution of silver nitrate. Xu et al.
187
demonstrated that silver nanoparticle-coated CA fibers exhibited exceptional antibacterial properties even at the lowest silver concentration (1 mg g−1). An increase in silver content completely inhibits the growth of
CA fibers can be converted into a flame-retardant material by surface modification. Superhydrophobic surface treatment can improve the deformation resistance of alginate fabrics in water. Zheng et al. 188 conducted work on the preparation of superhydrophobic and flame-retardant alginate fabrics using a hexadecyltrimethoxysilane-based surface treatment. The treated alginate fabrics demonstrated exceptional water repellency, salinity resistance, and fire resistance. The superhydrophobic surface treatment significantly enhanced the resistance of the fabrics to deformation in water. Moreover, with a limiting oxygen index value of 28.0, the treated alginate fabrics exhibited exceptional fire resistance.
Alginate fibers can also be coated with chitosan for wound dressing applications. Using the wet-spinning method, Dumont et al. 189 fabricated alginate fibers coated by chitosan that possess antibacterial activity against Gram-negative and Gram-positive bacteria, as well as antibiotic-resistant strains. The antibacterial activity is ascribed to surface effects, and β-irradiation does not alter the antibacterial properties of the processed fibers. In textile and wound dressing applications, chitosan-coated alginate fibers are favorable material candidates due to the combination of the wound-healing properties of CA with the antibacterial activity of chitosan.
The above surface modification strategies can be combined or tailored to meet specific requirements and desired functionalities of alginate-based materials. The choice of surface modification technique depends on the intended application, compatibility with the target material, and the properties need to be enhanced.
Potential applications of alginate fibers
Drug delivery systems
Alginate has great potential in drug formulation due to its non-toxicity, and is prevalently used in food. 190 Alginate is a favored option for ionic gelation-based encapsulation, forming cross-linked structures through ionic bonds with divalent cations. 191 Alginate is also adaptable for pharmaceutical and biomedical purposes, serving as an excipient and tool for controlled drug delivery. Alginate-based systems ensure drug release in a consistent and predictable manner. SA and alginic acid have traditional roles as tablet-binding agents and disintegrants, respectively. 190
Liu et al. 192 investigated a potential of alginate fibers as drug delivery system. They report an innovative technique for producing methylene blue-loaded alginate fibers by spinning an aqueous mixture containing alginate and drug-containing alginate nanocapsules. The resulting fibers demonstrate exceptionally high-water absorbency and follow the pseudo-second-order kinetics model. The release of drugs from the fibers is influenced by both the nanocapsule content and the conditions of the surrounding medium. Fibers containing drug-loaded nanocapsules demonstrate improved controlled release characteristics (Figure 6).
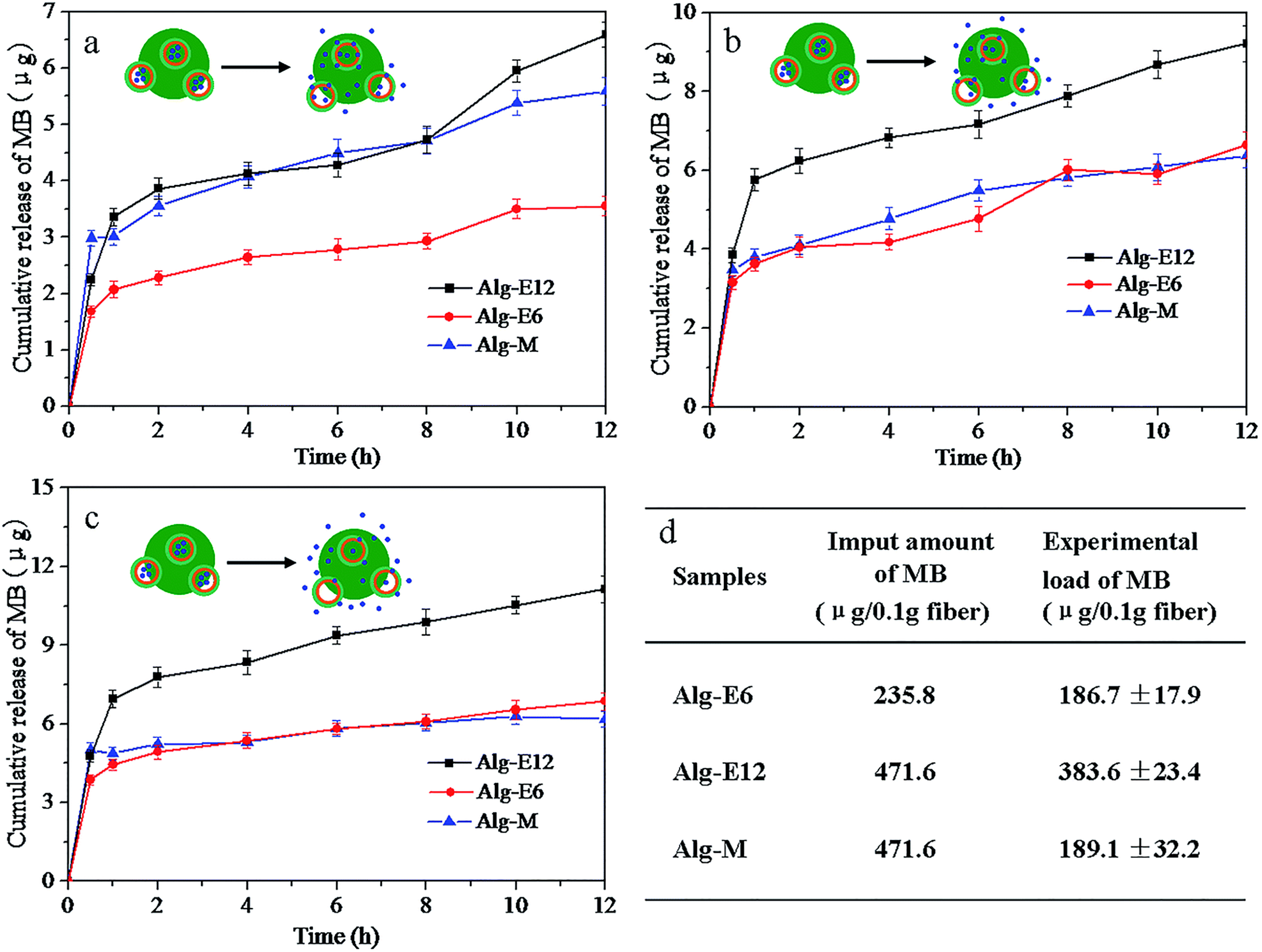
Cumulative release of methylene blue from different alginate fibers in different media: (a) distilled water, (b) solution A, and (c) PBS buffer solution (c). Different amounts of drug-loaded nanocapsules incorporated in alginate fibers (d), 192 with permission from Royal Society of Chemistry (copyright 2014).
A drug-loaded fiber made from alginate and starch was created by Wang et al.
193
by extruding their solution into a coagulating bath containing CaCl2 and ethanol. The findings from the controlled release experiments revealed that higher proportions of starch in the fiber led to greater amounts of model drug (salicylic acid) being released (Figure 7). Additionally, as shown in Figure 8, while the release rate of the drug decreased with higher drug loads in the fiber, the total cumulative release amount increased. The fiber holds promise for localized drug delivery applications
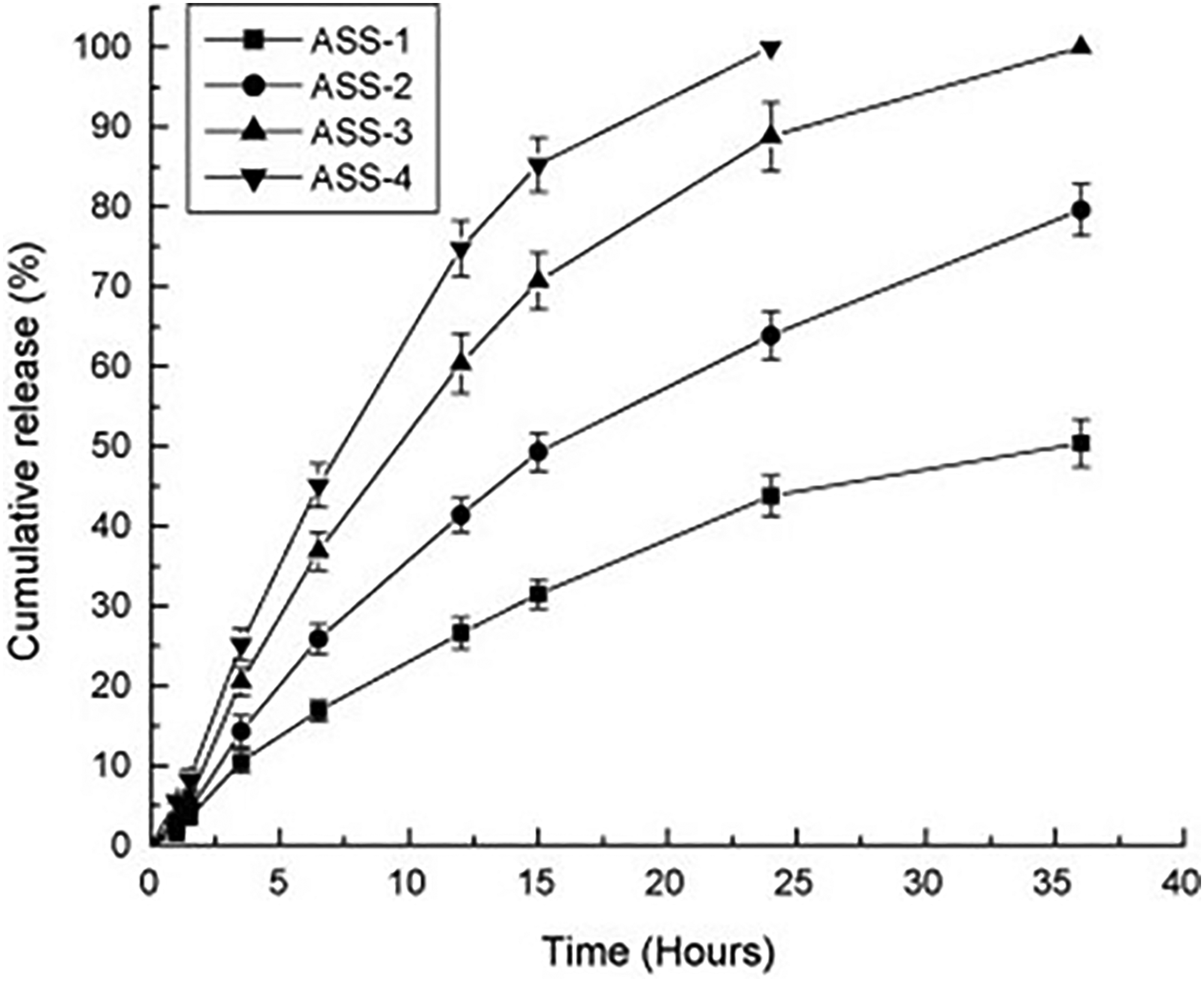
The influence of the drug composition incorporated in alginate/starch fibers on the controlled drug release process. ASS-1, ASS-2, ASS-3 and ASS-4 (A—alginate; S—starch; weight ratios of alginate: starch were 90:10, 70:30, 50:50 and 30:70, respectively), 193 with permission from Elsevier (copyright 2010).
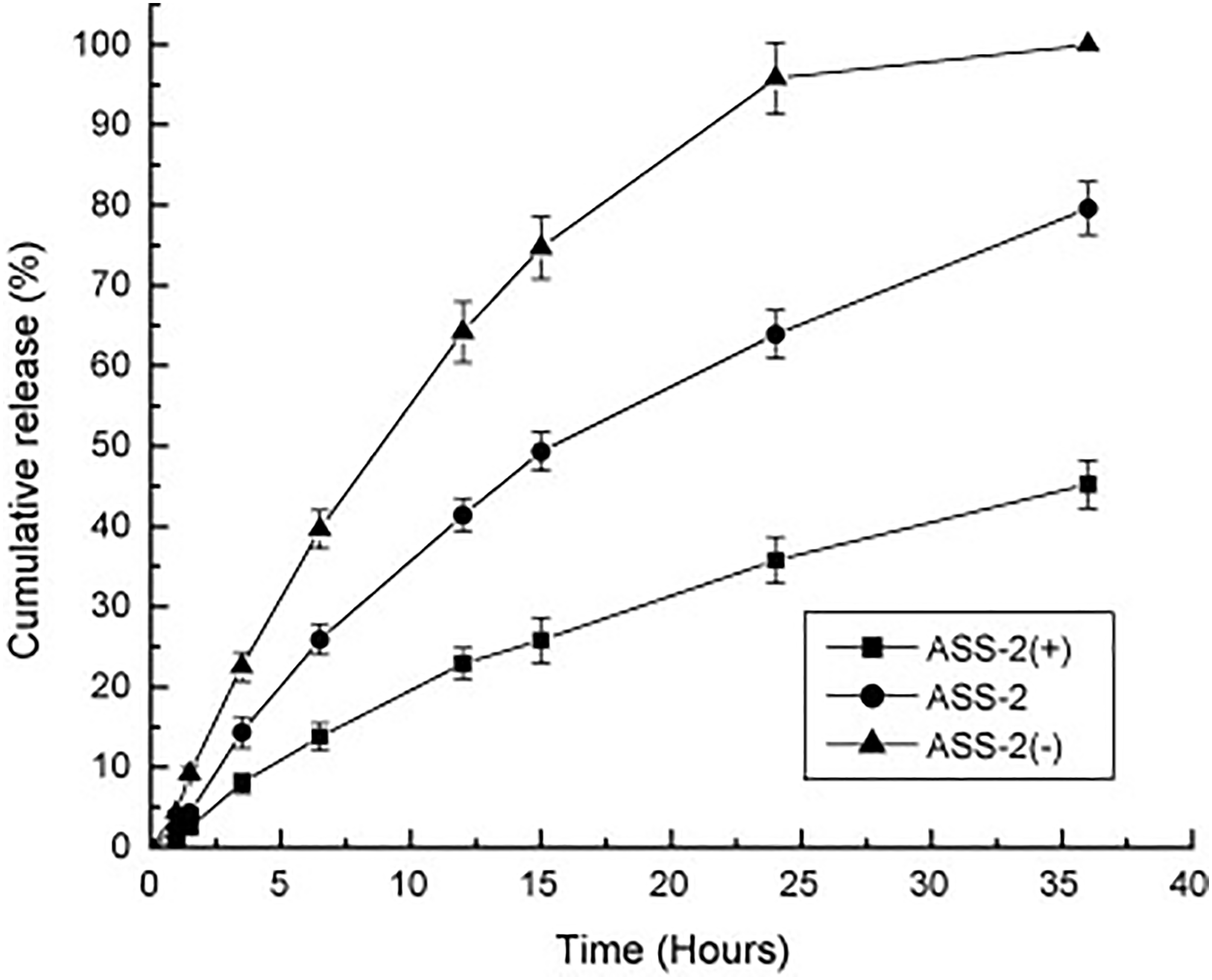
The influence of the amount of drug incorporated in ASS-2 fibers on the controlled drug release process. ASS-2(+), ASS-2, and ASS-2(-) (1.0, 2.0, and 3.0 g of salicylic acid, respectively), 193 with permission from Elsevier (copyright 2010).
Dabiri et al. 194 developed a pH-sensitive alginate-brushite hydrogel as a targeted drug delivery system. In this system, the -COO- groups in alginate are converted to -COOH at pH levels below 3. These resulting -COOH groups form hydrogen bonds with -OH groups in the alginate, which decreases the swelling rate in acidic environments, such as gastric fluids, and minimizes drug release efficiency under such conditions. Conversely, in intestinal fluid (pH = 7.4), these alginate composites exhibit a burst release of ibuprofen due to significant swelling and electrostatic repulsion between the COO- groups.
Scaffolding in tissue engineering
The ideal scaffolding material should possess biocompatibility, appropriate microstructure, desired mechanical strength and degradation rate, and, most importantly, the capacity to sustain cell residence and maintain metabolic functions. 195 Alginate fibers have several advantages as a scaffold material for tissue engineering. They have a high surface area-to-volume ratio, which allows for efficient nutrient and oxygen transport to cells. Their high porosity allows for cell infiltration and proliferation within the scaffold. Alginate-based scaffolds can be prepared by cross-linking the alginate molecules to form a three-dimensional network structure. The porosity and mechanical properties of the scaffold can be tuned by adjusting the cross-linking conditions and the concentration of alginate used. These scaffolds are then seeded with cells, which can grow and differentiate within the scaffold to form functional tissues.
Luo et al.
196
produced fibrous scaffolds based on highly concentrated alginate/gelatin-methacryloyl bioinks via coaxial 3D printing. Nanoapatites were uniformly coated on the inner and exterior surfaces of the hollow fibers. Direct protein loading into the bio-inks allowed for sustained release from the scaffolds over a 28-day period.
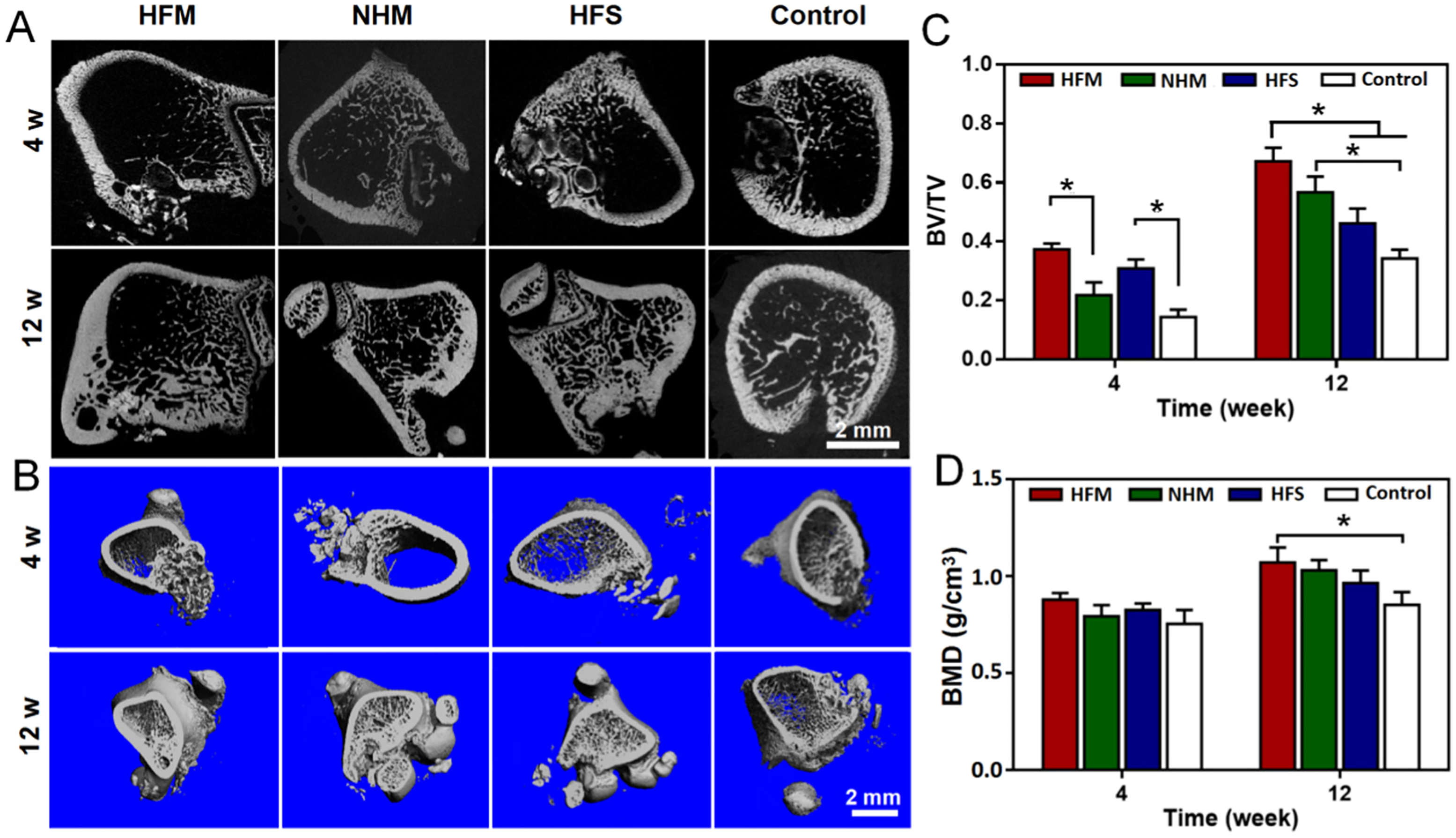
Micro-CT analysis performed on the bone repairing regions using different scaffolds in 2D slices (A) and 3D images (B). The bone volume/total volume (BV/TV) (C) and bone mineral density (BMD) (D) of the samples assessed after 4 and 12 weeks of treatment (*p < 0.05), 196 with permission from Elsevier (copyright 2022).
Alginate-based scaffolds were also successfully made by electrospinning by Rezaie et al. 197 They created CA and sodium carboxyl methyl cellulose (SCMC)-based scaffolds for skin tissue engineering applications. The outcomes demonstrated the production of bead-free fibers with a porosity of more than 80%. The presence of CA improved the materials’ flexibility, water absorption, wettability, and rate of biodegradation. According to biological analysis, the SCMC/CA scaffold had greater fibroblast cell adhesion and proliferation than a pure SCMC scaffold. Statistical analysis indicated that the incorporation of CA led to a notable enhancement in cell viability. The overall conclusions pointed to the SCMC/CA nanofiber scaffold's chemical, mechanical, hydrophilicity, and biological compatibility, making them a suitable candidate for further research into skin tissue engineering applications.
Wound dressings
Hydrogel-based dressings are favored for their potential to promote efficient wound healing due to their ability to facilitate rapid healing, alleviate pain, and minimize scar formation. 198 CA's widespread use as a wound dressing is attributed to its effective exudate removal capacity. 199 Upon contact with wound exudates, water-insoluble CA triggers an exchange of Ca2+ with sodium ions in the body fluid, potentially serving as a hemostatic agent. Gradually transitioning into SA, it absorbs a significant exudate volume, forming a gel that maintains a moist wound interface. 200 This moist environment is advantageous for healing, as it facilitates faster migration of epithelial cells from moist wounds to the affected area, contrasting with slower migration in dry wounds. 201
Hu et al.
199
coelectrospun alginate and PCL as composite fibers using a dual-jet system. A moist environment for wounds was created by highly absorbent alginate, and the presence of PCL improved cell adherence. Microorganism growth was reduced by silver nanoparticles, incorporated in the PCL fibers for long-term release. The platelet-derived growth factor-B (PDGF-B) gene was also included and allowed cells to be
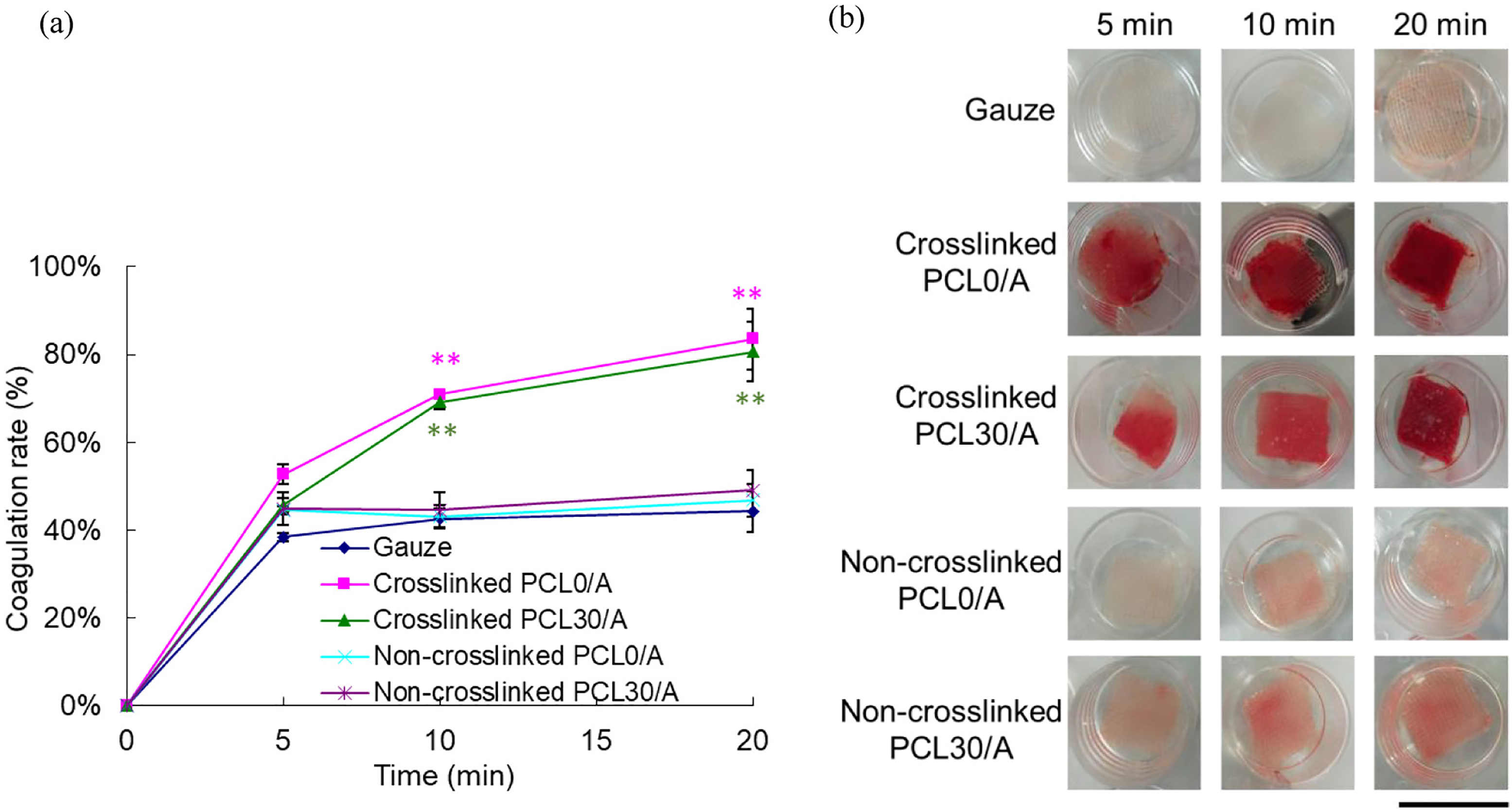
a) The coagulation rate of PCL/alginate composite fibers. (**: p < 0.01 compared to the gauze group); b) The photos of blood clots on PCL/alginate composite fibers. (scale bar = 2 cm), 199 with permission from Elsevier (copyright 2022).
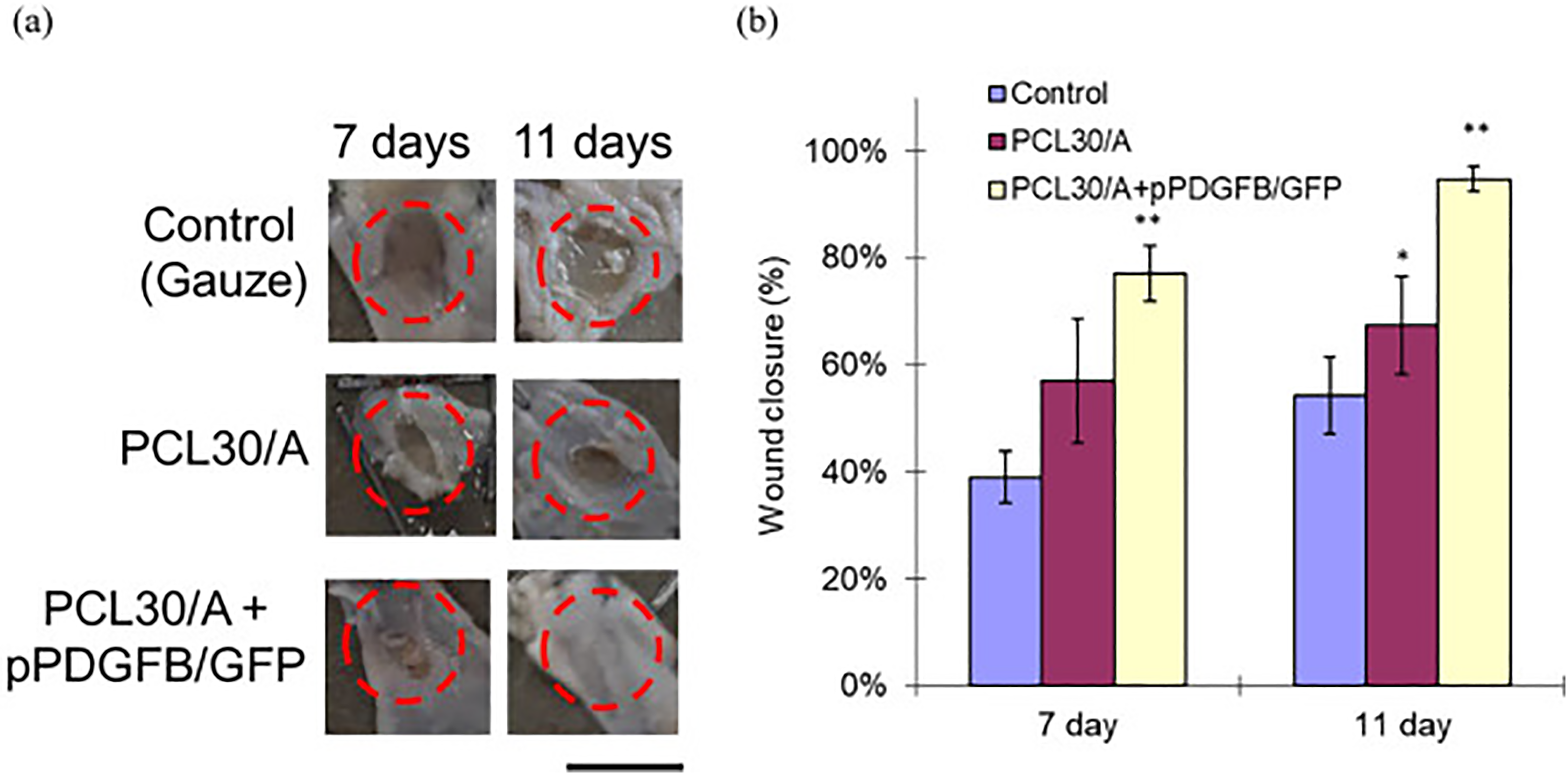
Ma et al. 202 used microfluidic spinning to form alginate into helical fibers, which were then mixed with polyacrylamide (PAM) to create a composite polymer film. The composite film exhibits enhanced flexibility, enabling it to undergo stretching up to 14 times its initial length without significant deformation under 300% strain for 8 cycles. It also possessed features of transparency and adhesion to the skin, making it ideal for wound dressings. The CA-containing fibers exhibit a compact structure, which enables enhanced loading efficiency and improved control over drug release compared to pure PAM. This study offers a multifunctional solution to facilitate wound healing, particularly when incorporating antibacterial agents or healing-promoting drugs.
Recently, Mavrokefalou et al.
203
produced nanofiber films through the electrospinning process using SA and gelatin as the matrix and betamethasone as an anti-inflammatory component. These films were discovered to effectively speed up blood clotting and wound healing. The scratch assay was utilized to evaluate the

Time-lapse sequence of the
Wastewater treatment
Alginate fibers exhibit favorable characteristics such as cost-effectiveness, ease of regeneration, and minimal susceptibility to temperature variations, making them a very promising material for the purpose of wastewater treatment. 204 Alginate beads have also been used for decontamination of aquatic pollutants such as heavy metals, dyes, and organic/inorganic pollutants through various pathways such as adsorption, precipitation, ion exchange, reverse osmosis, and evaporation.205,206 Alginate beads have been used in the remediation of actual meat processing effluent. A specially built annular photobioreactor was created to enhance the transmission of light in situations where there is a high ratio of beads to wastewater. 207
With a breakdown rate that was quicker than that of non-porous analogs and bulk P25 titanium dioxide (TiO2) powder, alginate/TiO2 fibers created by Papageorgiou et al. 10 demonstrated excellent efficiency in the removal of methyl orange from contaminated water. The study used a hybrid photocatalytic/ultrafiltration water treatment technology, wherein composite alginate/photocatalyst porous fibers and TiO2 membranes were utilized. As a result, there was a threefold increase in the efficacy of methyl orange elimination, and the membrane retentate underwent dilution instead of condensation. In the presence of the fibers, the permeability of the photocatalytic membrane increased by about 20%. The technologies used to develop the relevant materials are easily scaled up, and it is not anticipated that they will significantly increase the cost of the suggested water treatment procedure.
In another study, Pan et al. 204 synthesized alginate composite fibers reinforced with GO to remove heavy metal ions from wastewater. The interlaced fibers provided favorable transport channels and an enlarged surface area, resulting in a higher adsorption rate for heavy metal ions. As shown in Figure 13, the maximum adsorption capacity for Pb2+ and Cu2+ was 386.5 and 102.4 mg g−1, respectively, higher than other reported sorbents.
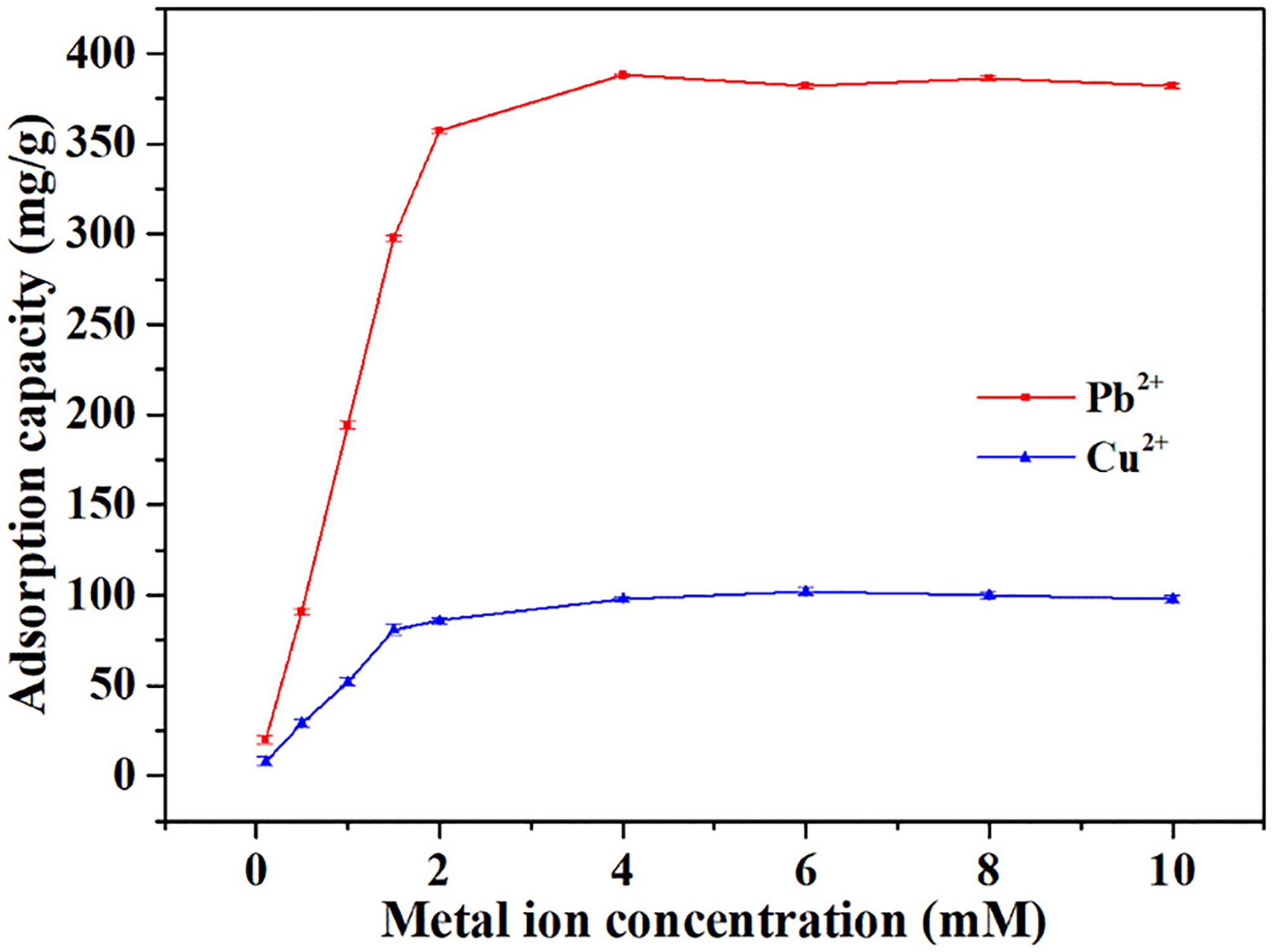
Maximum adsorption capacity of alginate composite fibers for Pb2+ and Cu2+. determined by mixing 50 mg of the fibers with 50 mL of solutions containing varying concentrations of the metal ions, 204 with permission from Elsevier (copyright 2019).
Additionally, there are some efforts to increase the performance of alginate fibers wastewater treatment. Incorporating GO into alginate fibers is one the most effective methods investigated. Wu et al. 208 utilized wet spinning for producing GO and CA biocomposite fibers. The fibers were utilized as an adsorbent to remove ciprofloxacin (CPX) from an aqueous solution. As shown in Figure 14, at a 2 g L−1 adsorbent dose and 6% GO loading, the removal percentage of GO/CA fibers reached 78.9%. At an equilibrium concentration of 60 mg/L, the adsorption capacity increased from 18.5 mg g−1 to 39.1 mg g−1 as the GO content in the fibers increased from 0 to 6 wt%.
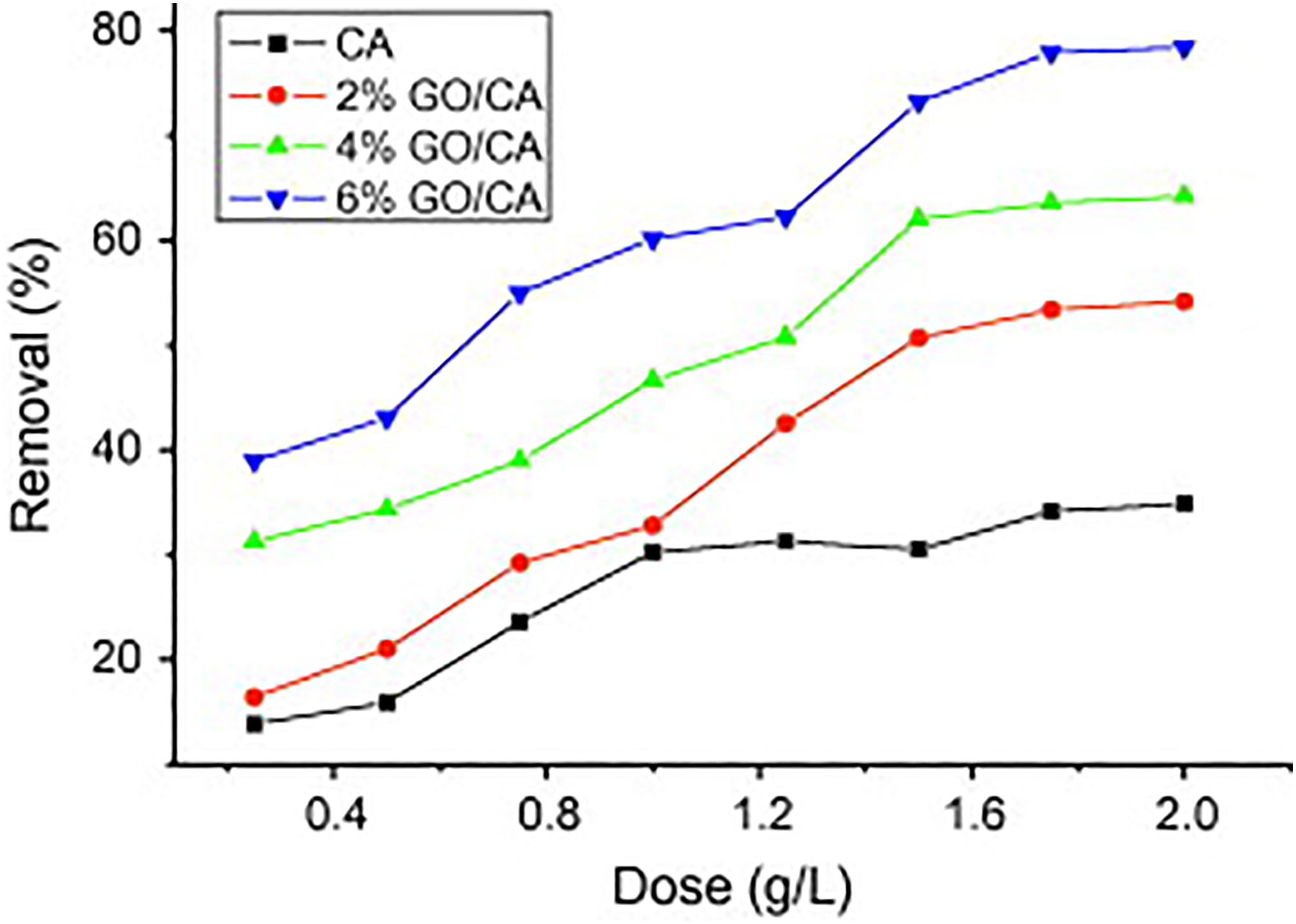
The effect of dosage on the adsorption of CPX onto CA and GO/CA fibers investigated under specific conditions: concentration of CPX = 20 mg/L, temperature = 293 K, and pH = 6.1, 208 with permission from Elsevier (copyright 2013).
Biomaterial-based sensors
Alginate fibers have applications as sensing components in different types of sensors such as for pH, glucose, and gas. They function by allowing the target substance to interact with the alginate material, resulting in variations in fiber properties. These alterations are then identifiable through methods like optics, electricity, or mechanics.
A smart pH sensor based on CA fibers was created by Cui et al. 209 to track the healing process of wounds. Two samples of CA fibers modified with hydroxypropyl trimethyl ammonium chloride (HACC) were prepared and underwent dyeing processes, one with alizarin and the other with anthocyanin. When the HACC concentration was 10 g L−1, the modified CA fiber exhibited the best dyeing performance. Over the pH range of 2–11, the smart CA fibers showed a rapid and noticeable color change as well as a reversible response that was visible to the naked eye.
Chen et al. 13 explored the development of conductive gels toughened by SA fibers in an oil-water system for the preparation of skin-like ionic sensors. PVA and SA fibers were used to create a dynamic network, which offered a wide range of mechanical properties, including high toughness, remodelability, and anti-fatigue fracture. These gels had a sensitive capacity for stress and strain; therefore, they were used as sensors to track different body motions. As shown in Figure 15, biosensor developed was able to monitor the real-time signals of human body motions, including bending of fingers, arms, knees and walking, as well as speaking, coughing and burping. For instance, when varying degrees of bending were applied by the fingers, the gel-based sensor underwent deformation. Subsequently, this movement signal was translated into electrical signals, which were then documented. These biosensors were affixed to the joints of the arm and knee, demonstrating consistent and distinct electrical signals mirroring the physical deformation. This indicates the reliable stability of the biosensors in producing repeatable results. 13
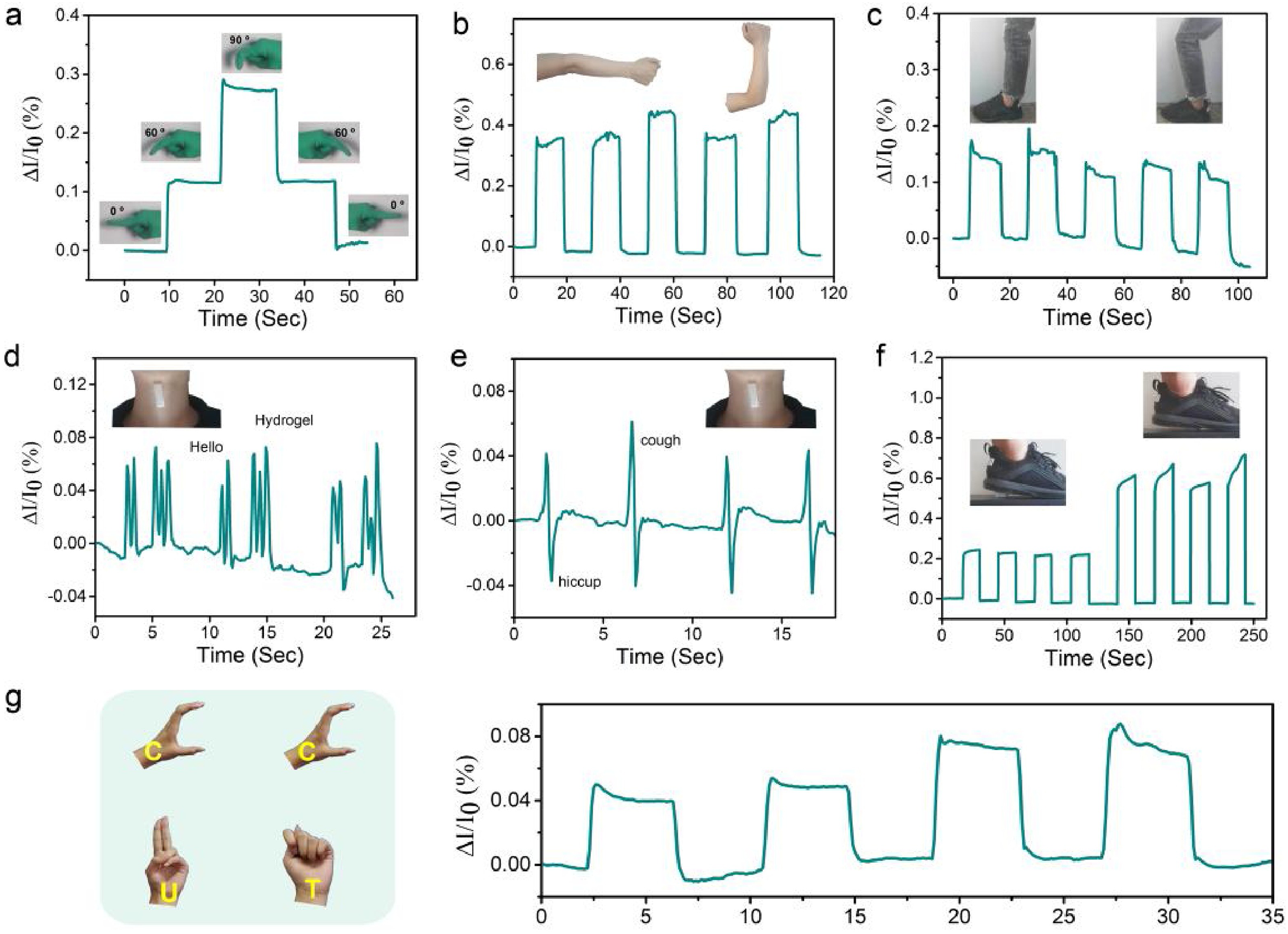
Tracking human body movements using SA gel-based sensors conducted across various body parts: a) Finger; b) arm; c) knee joint; d) throat; e) throat); f) walk; g) hand, 13 with permission from Elsevier (copyright 2020).
Hu et al. 98 investigated the creation of smart wearable and flexible optical sensors. Wet spinning was used to create fluorescent and durable fibers based on alginate loaded with gold nanoclusters. Under ultraviolet irradiation, these fibers displayed impressive red emission and good mechanical strength. With a detection limit as low as 188.0 nM for Cu2+ and 82.1 nM for Hg2+, these fibers could be employed as an easy-to-use, inexpensive, and highly selective fluorescence sensor for detecting Cu2+ and Hg2+ among other metal ions in aqueous solution. By being knitted into textile materials, the luminous fibers were also proposed to have potential as an anti-counterfeiting label.
Future prospectives
In light of the diverse applications of alginate fibers, there is no doubt that these fibers will stand at the forefront of material science advancements. However, the current methodologies for alginate fiber production are fraught with challenges. Wet spinning and modified variations of this technique are used as the primary approaches to produce alginate fibers. A pervasive limitation in wet spinning is the low extrusion rate, which consequently leads to suboptimal yields in alginate fiber manufacturing. Additionally, fabricating fine fibers with uniform morphology via wet spinning remains a considerable challenge, owing largely to the multifaceted parameters associated with the coagulation bath. The fibers resulting from wet spinning usually have dimeters in the micrometer scale.
Due to the marked hydrophilic nature of SA and its low solubility in common organic solvents, there has been to date minimal success where alginate fibers have been produced through prevalent polymer fiber processing techniques like electrospinning, centrifugal spinning, gas blowing, etc. The advancement of processing methodologies is a fundamental requirement for enhancing the properties of alginate fibers. There are some novel methods that have been proven to realize in high-productivity fibers, such as PG 154 and nozzle-PG. 158 As previously mentioned, the significant success achieved with the modified nozzle-PG in producing cellulose fibers has aroused great interest in using this method to expand the production of other natural polymer fibers. Efforts are currently underway to use this innovative process to produce alginate fibers for antimicrobial wound dressings, with exciting progress achieved. Given the distinctive biochemical properties of alginate and the exceptional fiber processing capabilities offered by nozzle-PG, this endeavor holds immense importance in transforming alginate into high-value products tailored to meet the demands of specific biomedical applications. Further elaboration of these findings will be presented in our forthcoming publications. 210
Additionally, several challenges need to be addressed to advance alginate-based materials into clinical practice, particularly concerning material safety and regulatory compliance. While alginate itself is recognized as non-toxic and highly biocompatible, the safety of its composites and potential risks associated with their use remain to be evaluated. Variability in alginate derived from natural sources can significantly impact consistency between batches, necessitating adherence to Good Manufacturing Practices (GMP) to ensure uniformity and reliability in the final product. Establishing rigorous quality control measures and comprehensive regulatory frameworks is essential to ensure they meet the standards required for clinical use. Therefore, addressing these issues is crucial for the successful translation of alginate materials from research settings to real-world clinical applications.
In conclusion, alginate fibers have attracted considerable attention due to their biocompatibility, biodegradability, and versatile applications in various fields. The fabrication of alginate fibers has been extensively studied, and several techniques have been developed to enhance their properties, such as mechanical strength and water resistance. Moreover, the future of alginate fibers looks promising as researchers continue to explore novel strategies to improve their properties and develop new applications. With the increasing demand for eco-friendly and sustainable materials, alginate fibers have the potential to play a significant role in addressing these challenges and contributing to a more sustainable future.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
