Abstract
The aim of this review is to succinctly review recent progress in experimental and theoretical investigation of the viscosity of binary and multicomponent Ni–Al-based industrial alloys, summarising the current state of knowledge of the topic. Viscosity data are important for simulation of microstructural evolution during solidification-related processes such as casting. Therefore, to obtain accurate predictions of defect-free microstructure, only reliable input data are required. Based on the synergy between experiment and theory, a review of the literature provides a critical evaluation of temperature- and composition-dependent viscosity data available from existing studies carried out over the last 60 years. The purpose of this review is threefold: to discuss container-based viscometer methods and alternative containerless processing via electromagnetic levitation during parabolic flights and on board the International Space Station; to analyse the viscosity models including their validation; and to refer to the first two points, to interpret the viscosity data.
Keywords
Introduction
Liquid Ni-based alloys represent examples of Newtonian fluids obeying Newton's law of viscosity. Its story goes back to the experiment performed by Sir Isaac Newton in 1687 and published in ‘Philosophiæ Naturalis Principia Mathematica’. 1 In that work, the concept of Newtonian fluid and Newton's laws of motion of classical mechanics have been introduced. Since the late 19th century, the above-mentioned experiment 1 is known as the Couette flow, in honour of Professor Maurice Couette, due to his contributions to the experiments and theory of fluid flow. 2 From the days of Newton, 1 it took more than 250 years to perform the first viscosity measurements of Ni-based alloys, indicating the complexity of both the experiments and materials.
Ni–Al binary and multicomponent alloys represent a combination of outstanding high-temperature mechanical properties, oxidation and corrosion resistance in harsh environments covering a broad range of applications: from structural, power generation, aerospace and transport to storage technologies, protective coating, catalytic chemistry and biomedical uses. 3 The most common approach to realise the above-mentioned applications achieving high-performance levels is Materials Design. Its methodology involves a series of steps, iterations and periodic feedback between theory, experiment and computations, making it possible to perform investigation based on a quadriade ‘composition-processing-microstructure-properties’. 4 Among the processing routes used for manufacturing Ni-based industrial alloys,3,5–8 an investment casting as one of the oldest known metal forming techniques is still under continuous optimisation 3 aiming to reduce the presence of structural defects on the components made from these alloys. Thermophysical property data analysis has a fundamental role in the theory and experimental determination with the assessment and feedback of more elaborated models and procedures. Accordingly, the development of mathematical models describing the solidification of complex alloy systems in terms of microstructural evolution requires accurate and reliable thermophysical properties data as inputs. On the other side, reliable property data can be obtained by advanced measurement methods, such as those performed under microgravity conditions.9,10
From 2001 to 2020, in the framework of the European Space Agency (ESA)-microgravity application programme (MAP) ThermoLab (MAP-Thermophysical Properties of Liquid Metallic Alloys for Modelling of Industrial Solidification Processes and Development of Advance Components and Products)9,11 and ThermoProp (MAP-Thermophysical Properties of Liquid Metals for Industrial Process) projects, the experimental and theoretical investigations of the thermophysical properties of binary and multicomponent Ni–Al liquid alloys have been performed. In order to study the mass density, surface tension, viscosity, specific heat, electrical resistivity, thermal conductivity and total hemispherical emissivity of liquid Ni-based industrial alloys, container-based, quasi-containerless and containerless experimental methods including the measurements on the International Space Station (ISS) together with different theoretical models based on thermodynamics, statistical mechanics and atomistic approaches have been applied.12–16 Recently, the present authors reported the state of the arts on the surface properties of liquid Ni–Al alloys including a few aspects, such as the experiments and data collection, theoretical modelling, validation of the models and comparison between the experimental data and model predicted values. 15 In the present work, the aforementioned aspects related to the viscosity of Ni-based alloys were analysed. Viscosity is one of the most important thermophysical properties of metallic materials because of its high sensitivity to microstructure evolution and phase transformations. Since the early 1980s, the viscosity reference data of liquid Al and Ni have been measured by many authors and collected.17–24 Within each dataset, the viscosity data differs by up to 20% 24 or more.17,20,21 The situation is even worse for the viscosity datasets of binary and complex Ni–Al-based alloys, indicating a scatter of up to 100% for temperature- and concentration-dependent viscosity.16,24–27 Preliminary analysis of available viscosity datasets of Ni-based alloy systems indicates that, until now, the oscillating cup technique has been mainly used.10,22,24,28,29 Therefore, high chemical reactivity of metallic melts in contact with container materials, melt oxidation and inaccurately defined crucible diameter are the primary sources of experimental errors.24,28 To avoid these inconveniences, over the last two decades, a few measurements using containerless electromagnetic levitation (EML) during parabolic flights (PFs)13,30 or on board the ISS 14 were performed. Concerning the models, the Arrhenius equation is widely used to describe the temperature dependence of viscosity,23,28 while none of the existing models describes fairly well the concentration-dependent viscosity data neither of Ni–Al23,27,31 nor of Ni-based alloy melts. 16 Accordingly, the use of two or more models is useful to construct and interpret confidence intervals for predicted values that, combined with the experimental data obtained by different experimental methods, are necessary for data reliability assessment. On the contrary, speaking about the surface properties, Butler's model 32 is considered the ‘universal one’ describing the surface tension of liquids including metallic melts, either properly 24 or as a first approximation, 15 as it was shown for alloy systems exhibiting one of two limiting types of mixing, that is, phase separation and ordering in alloy systems.12,15 In the case of viscosity, the ‘universal’ model that allows both the predictions and explanations of physical phenomena is still lacking. For the previously mentioned systems, the models describing their surface properties include explicit contribution of short-range order (SRO) in terms of homocoordinated and heterocoordinated pairs of atoms, respectively, 33 while the effects of SRO on the viscosity are implicitly built into the viscosity models based on thermodynamics.23,34
This review represents the state of the art on the viscosity of binary and complex Ni–Al-based industrial alloys and includes the experimental measurement methods, available datasets and analysis of models applied to calculate viscosity isotherms and polytherms of the above-mentioned systems. It is important not only for metallurgists, but also for all people dealing with experimental and theoretical aspects of viscosity of liquid metallic materials. The future prospective of metallic materials design is more oriented to modelling and simulation, a fast development of databases containing properties data and a wide use of a multivariate analysis (MVA) for data elaboration including machine learning, which on the other side, will contribute to developing of new alloys with desired properties.
Experimental methods
Over the last 150 years, viscosity measurements of metallic melts have evolved from conventional container-based methods16,22,24 to containerless processing by means of electromagnetic13,14,35 and electrostatic levitation.36–38 The oscillating cup or similar methods are suitable to measure both dynamic18,27,39 and kinematic26,40,41 viscosity. However, none of the container materials is inert or highly resistant when in contact with metallic melts having high melting temperatures.42,43 Indeed, during conventional processing/measurement by viscometer methods, unavoidable interactions between the melt and the container take place. The formation of reaction products at the metal/container interface occurs and therefore, in many cases, despite the controlled experimental conditions, the obtained viscosity data is often unreliable. In particular, in the case of metallic alloys with elevated melting temperatures and relatively high melt reactivity, such as Ni–Al-based alloys, interfacial interactions can lead to detrimental effects on the precision of the measurement, or even render it plain and simply useless.15,16 In recent years, immense progress has been made in advancing ground-based containerless methods for the measurement of viscosity, such as electrostatic levitation.
44
On the other side, strong electromagnetic forces that keep the liquid sample levitated against Earth's gravity lead to a turbulent fluid flow, making inappropriate operating conditions for viscosity measurements.45,46 This can be overcome using containerless methods in microgravity conditions, that is, the oscillating drop method with an EML (OD–
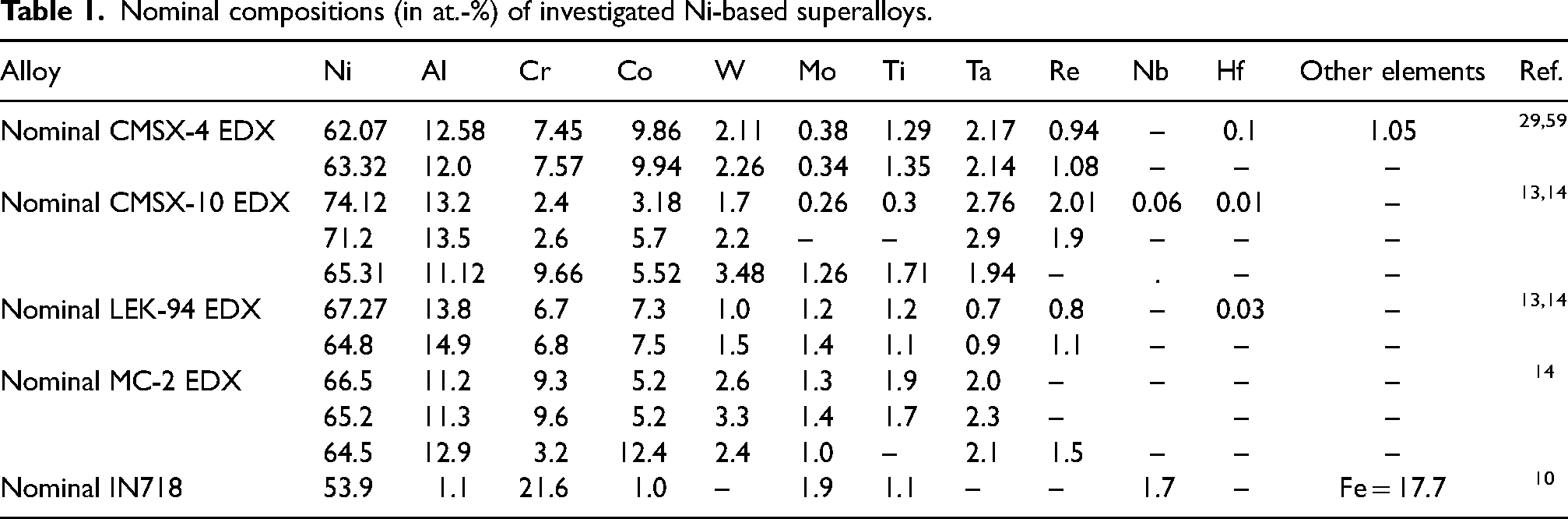
Nominal compositions (in at.-%) of investigated Ni-based superalloys.
Container-based methods
In the vast majority of liquid metals and alloys, the most reliable data are still measured using a conventional method, namely the oscillating cup viscometer.24,60 Such a viscometer typically consists of two parts: the furnace and the oscillation system. The device at the German Aerospace Center (DLR – Deutsches Zentrum für Luft- und Raumfahrt)
61
uses a vertical torsional steel wire (
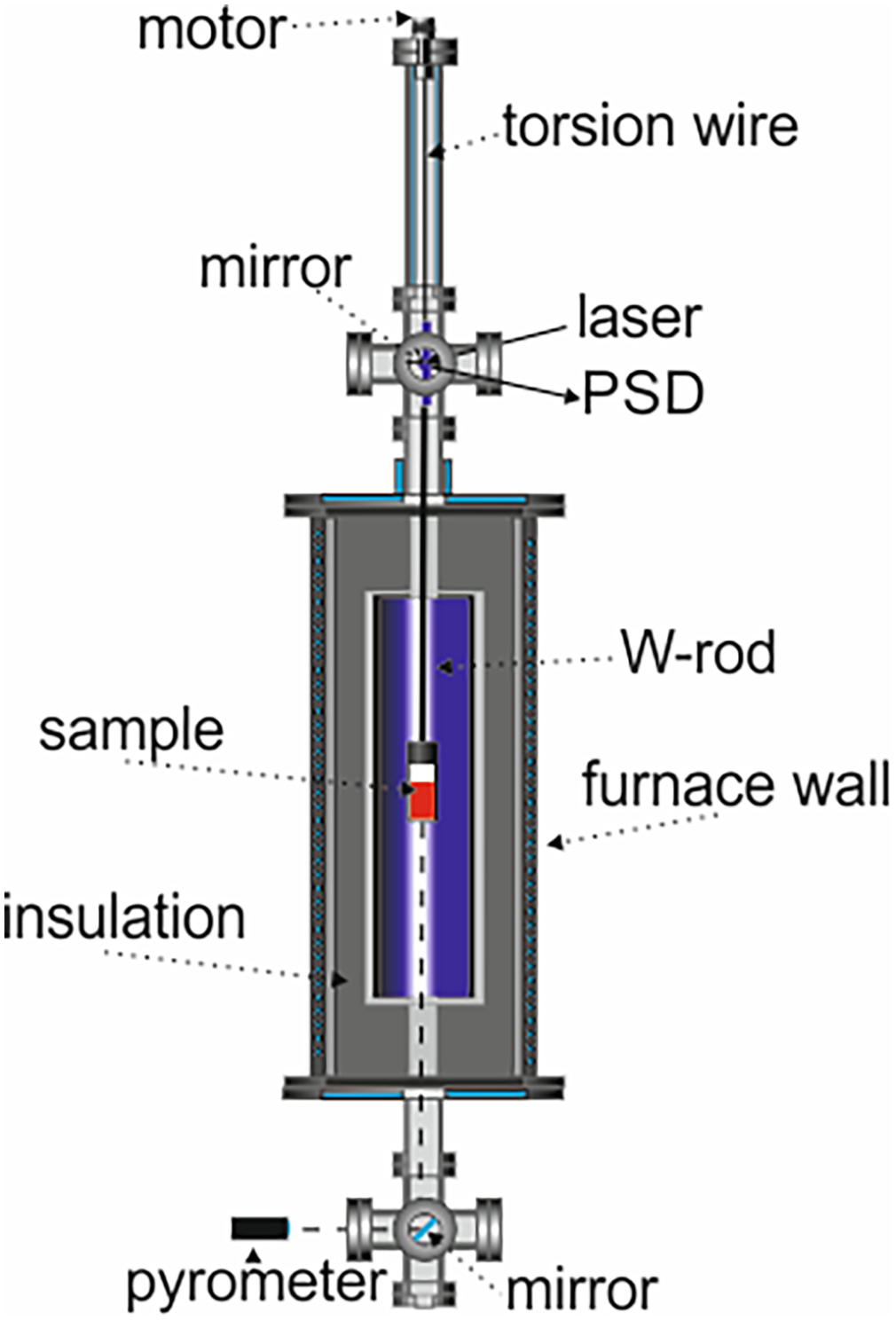
Schematic depiction of the oscillating cup viscometer used at the German Aerospace Center (DLR). 24 The crucible, placed inside the furnace, performs torsional oscillations excited by the motor. The pyrometer is directed at the crucible via an adjusted mirror.
The setup used at DLR
61
forms a vacuum system. Before each experiment, it is evacuated to 10−4 Pa in order to remove gaseous impurities. Experiments are done under pressure of 4×104 Pa in Ar atmosphere (99.999%), whereas a proportional, integral and derivative) loop assured constant pressure. The furnace, which is part of the vacuum system, can reach temperatures of up to 2300 K. The pyrometer is directed via a mirror to the bottom face of the crucible. A laser is shone at the mirror. The spot of the reflected beam is received by a position-sensitive detector that outputs a voltage from which the torsion angle 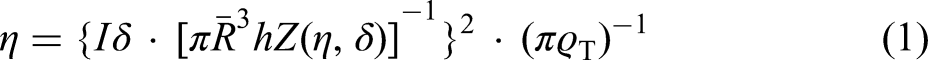
Containerless methods
The logic step to avoid the systematic source of measurement errors is to remove the container and to use containerless processing methods in order to prevent any contaminations with foreign material. An alternative to container-based methods for viscosity measurements is the oscillating drop (OD) method in an EML on board a PF aeroplane or the ISS.13,16,49 EML appears to be more versatile regarding the large variety of metallic and semiconducting alloys to which it was successfully applied as well as its applicability under different gas atmospheres and in vacuum. 64 High-precision experiments can be performed by levitating the sample using a levitation setup, such as an electromagnetic levitator. 45 Under microgravity, the reduction of levitation/positioning forces, quantified by a factor of 1000 with respect to those on the ground, leads from a turbulent to laminar flow transition and finally to laminar flow conditions, that make measurements of viscosity possible and reliable.46,65 In this way, heating and positioning by the electromagnetic fields get decoupled. As a consequence, a larger temperature range, from the stable liquid to the undercooled liquid phase, becomes experimentally accessible. In addition, the fluid flow, driven by the positioner field can be sufficiently low, to prevent turbulent flows.46,66
Currently, an EML (ISS-EML) is installed on board the European science module ‘Columbus’ on the ISS. A similar device, called TEMPUS (‘Tiegelfreies ElektroMagnetisches Prozessieren unter Schwerelosigkeit’, Engl. ‘Containerless electromagnetic processing under weightlessness’), is regularly installed on board an A310 parabolic flight aeroplane. With these devices, the sample can be positioned, heated, molten, overheated and cooled down in either a gas atmosphere (Ar or He) or in a vacuum. During the free cooling, well-established measurement methods are applied, in order to obtain several thermophysical properties, such as the viscosity and surface tension.
49
The measurements of viscosity in EML facilities under microgravity by the OD method
67
can be performed in PF experiments and on board the ISS. PF offer 20 s of microgravity
68
and serves as an important precursor for long-time duration ISS experiments, of about 20 min.
14
In both cases, in order to excite surface oscillations, during the sample cooling, short heater pulses are applied. These oscillations are usually damped exponentially, due to internal friction. The damping time constant can be related to the liquid viscosity
69
using
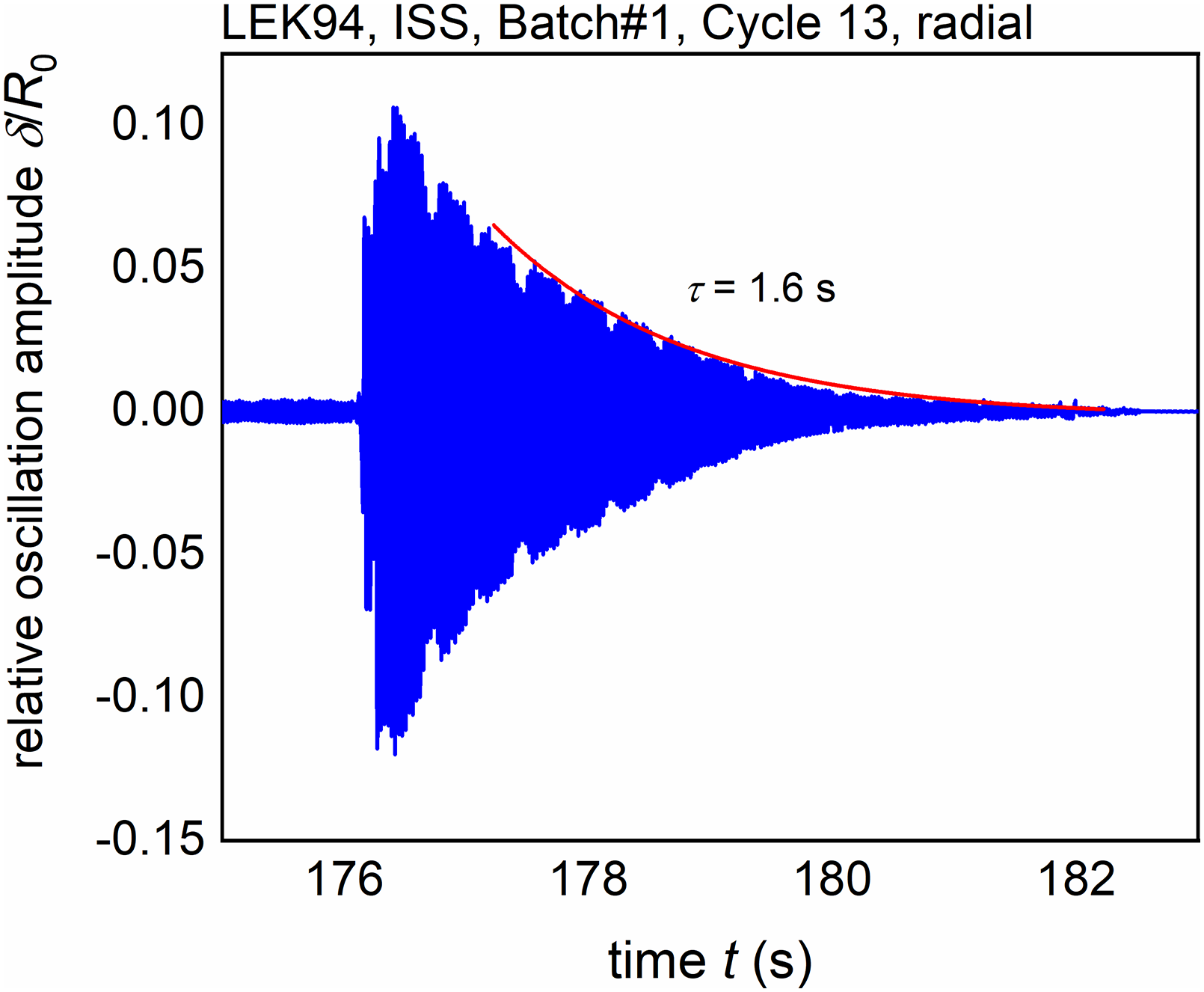
Exponential decay of the surface oscillation excited by the second heater pulse. 49
Modelling
Atomic-scale motion in the liquid phase can be explained in terms of viscosity, as one of the main transport properties. The viscosity of metallic melts can be experimentally determined as a function of temperature and as a function of composition. The viscosity datasets have been used to study the functional dependence of the fitting parameters in the models proposed to calculate the polytherms and the isotherms, respectively. Among the models used to describe the temperature-dependent viscosity of liquid metals and alloys, an empirical model known as the Arrhenius equation22,23 is given by
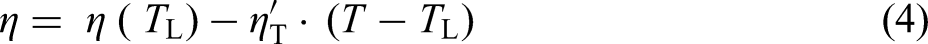
To describe the viscosity of alloy melts over a large range of temperatures, other models such as the Mauro–Yue–Ellison–Gupta–Allan (MYEGA), Vogel–Fulcher–Tammann (VFT) or Avramov–Milchev (AM) should be used. 70 The viscosity calculated by each of these three-parameter models can be expressed in terms of a common set of parameters that have a physical foundation. Among them, the MYEGA model is more accurate. 70
The model-predicted viscosity in terms of polytherms and isotherms exhibits discrepancies with respect to those obtained by the measurements and this reflects the difficulty of obtaining accurate experimental data.22,23,28 There are many empirical and semi-empirical viscosity models, but until now, there is no universal mathematical model that allows both predictions and interpretations of physicochemical phenomena involved. These models contain several arbitrary constants that deprive them of some practical interest except for comparison with experimental data.23,34 Accordingly, depending on the shape of the experimental isotherm, to assess the reliability of viscosity datasets of an alloy system, the most appropriate models have to be applied. Otherwise, the lack of viscosity experimental data of an alloy system leads to the use of different theoretical models resulting in arbitrary predicted isotherms.
Concerning the viscosity isotherm of liquid Ni–Al alloys, there are few investigations related to the applications of various models and their validation using available datasets.27,31,71 Indeed, the values calculated by the Kozlov, Kaptay, Hirai and Shi27,31 models as well as the predictions of Ni–Al viscosity by means of embedded-atom simulations 71 have been used and compared with the corresponding literature data obtained at the same temperature.26,27,30,31,55 In order to establish an agreement between the model predicted values and the experimental data of liquid Ni–Al alloys, the preliminary test of the viscosity models reported in the literature23,34 and their validation by means of the available datasets have been done, indicating the Terzieff model,72,73 as the most appropriate. 16 This model represents an extension of Iida's model23,74 and it was developed in the framework of thermodynamics and hard-sphere-like theory, describing the energetics and the structure of the liquid system and their contributions to its viscosity in terms of the enthalpy of mixing and the packing fraction, respectively. The hard-sphere contribution to the viscosity of liquid Ni–Al alloys was investigated by the molecular dynamic (MD) simulations analysing the Al–Al and Ni–Ni atomic distances in the Al- and Ni-rich parts of the Ni–Al system. 75 The Ni–Ni distance remains almost constant over the whole composition range, while for Al-rich alloys, the Al–Al distance increases with respect to the corresponding values observed for other alloy compositions, resulting in non-linear behaviour of the atomic volume and therefore, the change in packing fraction with composition.22,23 These theoretical findings were substantiated by inelastic neutron scattering measurements and experimental determination of Ni self-diffusion coefficient indicating the predominant packing effects on the transport properties with respect to those of the SRO phenomena or the composition changes. 75
The Terzieff model72,73 is expressed as the ratio of the excess viscosity and additive viscosity, giving the relative viscosity of an alloy melt, as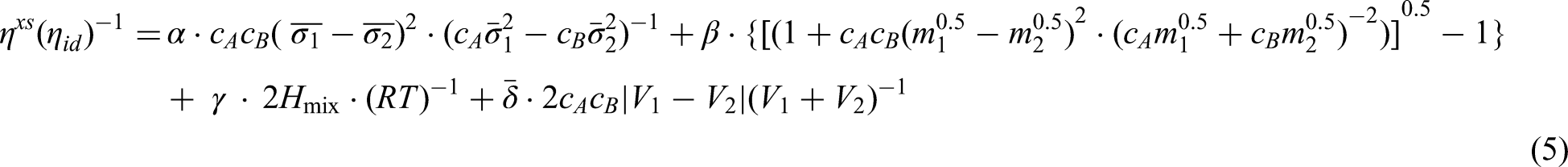
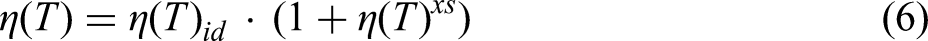
Based on the Stokes–Einstein relation between diffusivity and viscosity for pure liquid metals and its extension to binary alloys,
76
the assessment of an empirical viscosity model containing four constants has been developed in the framework of thermodynamics and statistical mechanics.
77
The viscosity model is represented by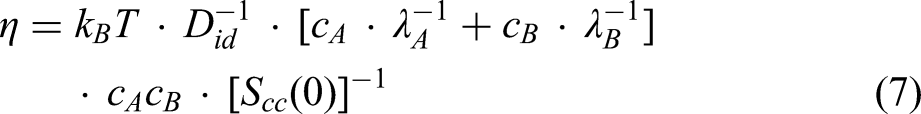
Results
Until 2000, there was a limited amount of viscosity datasets related to liquid Ni–Al alloys and all of them have been obtained by container-based viscometric methods.26,40,56,79,80 Regarding Ni–Al-based alloys, the lack of data was the biggest challenge, that in the last 20 years, was partially compensated by the viscosity measurements performed in the frameworks of the EU FP6-IMPRESS, 81 ESA-MAP ThermoLab82–84 and ThermoProp Projects. 85 The aforementioned projects included the experimental determination of the viscosity by container-based methods and advanced containerless processing techniques under microgravity conditions as well as the application of viscosity models reported in the literature.23,28,34,72,73,77 An important issue of the viscosity data analysis is the sources of errors that can explain why the experimental results differ among them and deviate from the model-predicted values. Roughly, one can distinguish two types of errors, that is, systematic errors and random errors. The first type is inherent in the measurement process and affects the accuracy of a measurement. Repeating the experiments, no matter how carefully, systematic errors cannot be eliminated. Random errors are related to the precision of instruments, purity of materials and surrounding atmospheres that can be reduced but never with absolute certainty. In the laboratories of project participants, round-robin measurements of the viscosity by using different experimental methods and different equipment improve the reliability of data and accuracy of measurements, reducing potential sources of systematic errors.9,16,24,81–85
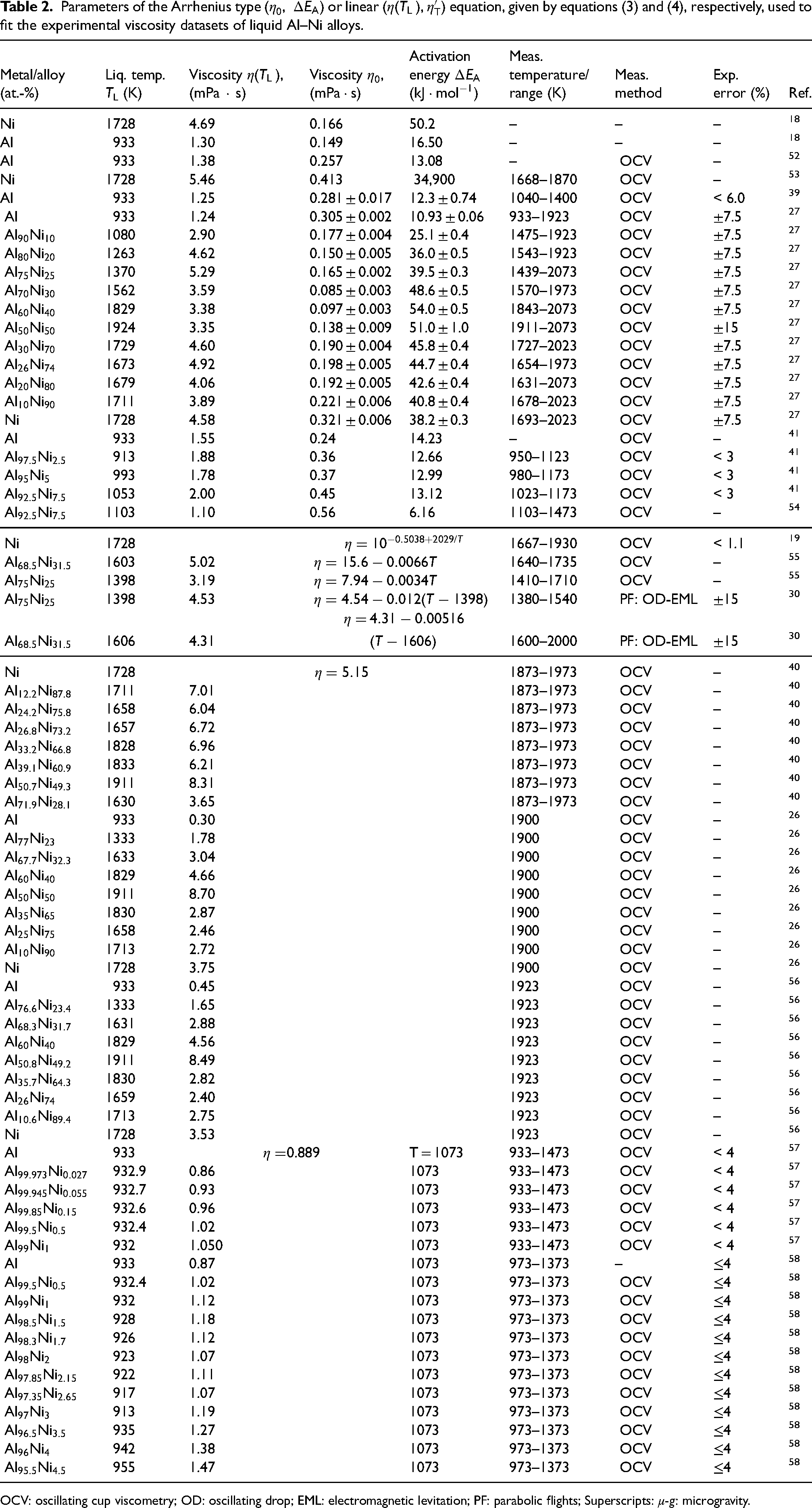
The available viscosity datasets of liquid Ni–Al and multicomponent Ni–Al-based alloys were collected and given in Tables 2 and 3, respectively. The analysis of viscosity data was carried out taking into account the two aspects of the investigation, that is, the experimental methods and theoretical models applied.
OCV: oscillating cup viscometry; OD: oscillating drop; EML: electromagnetic levitation; PF: parabolic flights; Superscripts:
Parameters of the Arrhenius type (
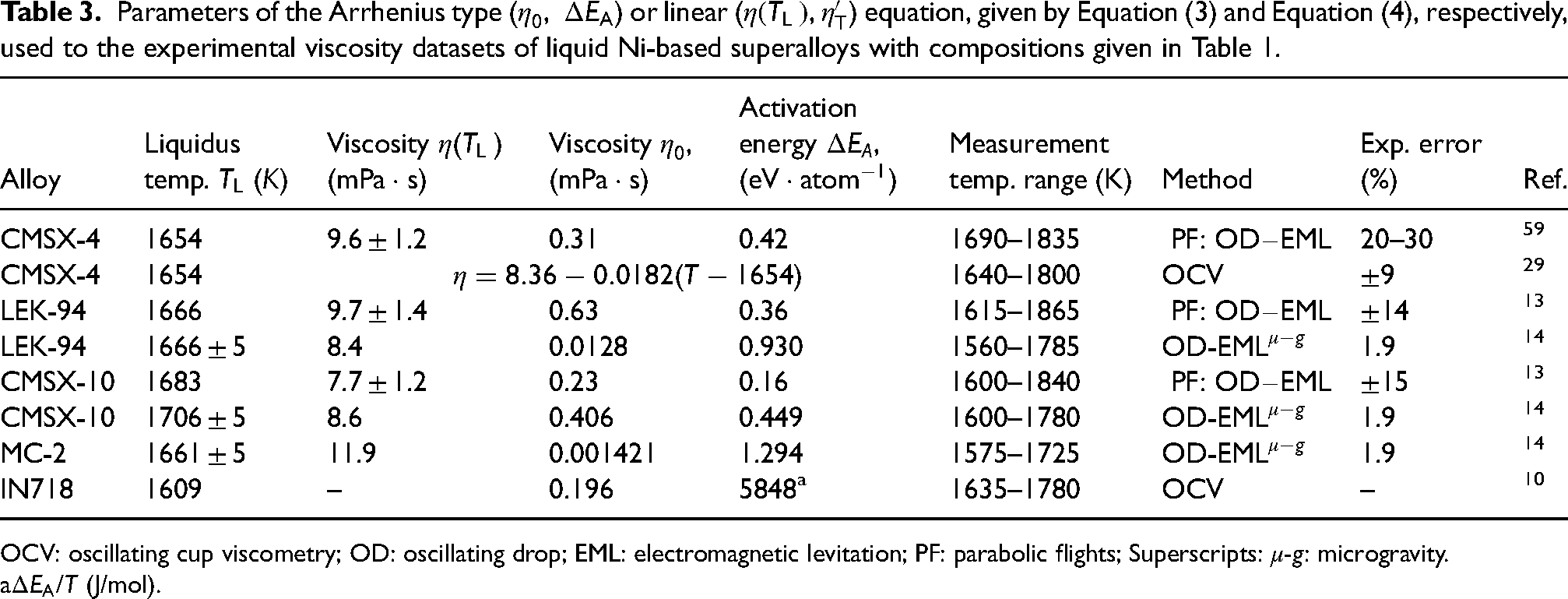
OCV: oscillating cup viscometry; OD: oscillating drop; EML: electromagnetic levitation; PF: parabolic flights; Superscripts:
Viscosity data of liquid Ni–Al alloys
Container-based methods
Over the last 60 years, the reference data for the viscosity of pure liquid Al18,26,27,39,41,52,56–58 and Ni 10,18,19,26,27,40,53,56 have been determined by different viscometric methods10,22,24 and collected in the literature.17,18,22–24 The same measurement methods have been mostly used to obtain the viscosity datasets of liquid Ni–Al binary alloys,26,27,40,41,54,55–58,79 either obtained over the entire composition range (Figure 3) or for Al-rich alloys,41,54,57,58,79 containing up to 7.5 at.-% Ni (Figure 4).
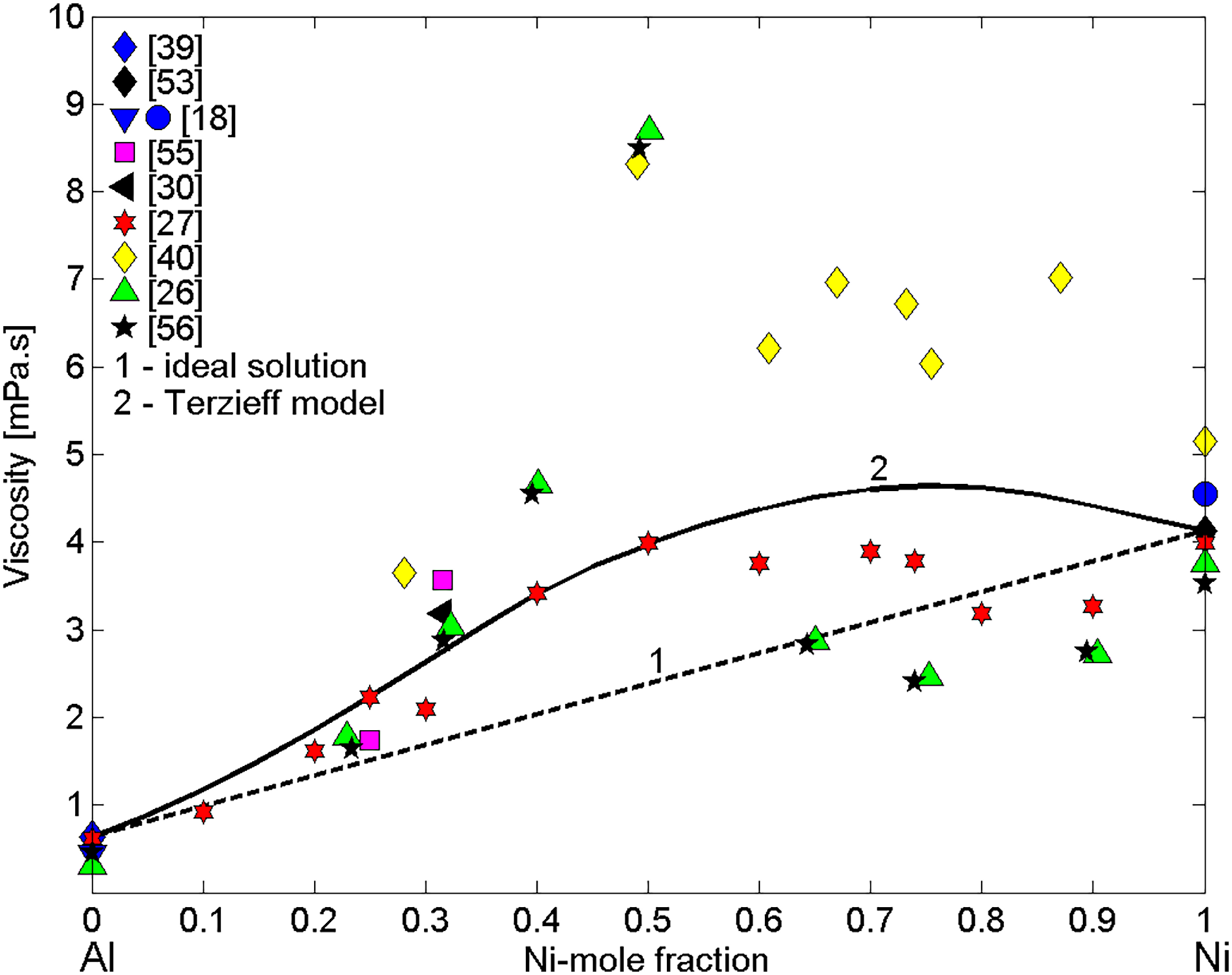
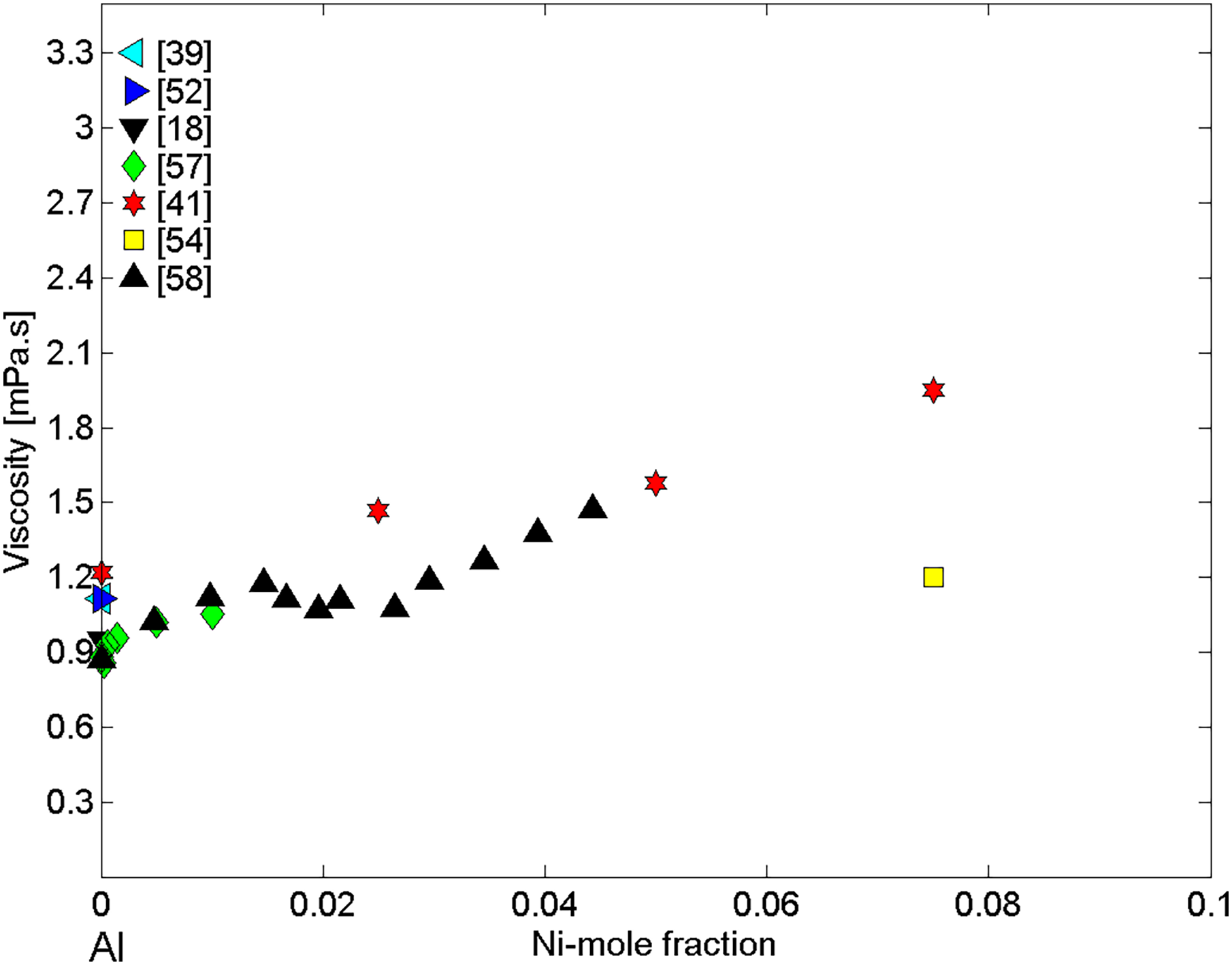
The viscosity measurements have been performed using mainly Al2O3 crucibles, but under different atmospheres. Indeed, the experiments were carried out using Al2O327,55,57,58 and BN 27 as container materials under protective purified helium41,57,58 or flowing argon27,55 atmosphere as well as in high vacuum. 54 In order to obtain reliable viscosity data, a preliminary investigation into the suitability of ceramic materials for the containers enabling accurate viscosity measurements as well as suitable surrounding atmosphere is necessary. Therefore, to avoid chemical reactions at the melt/ceramic interface, the material selection has to be based on the wetting experiments under a controlled atmosphere indicating the non-reactive wetting behaviour of a melt in contact with candidate materials.15,42,43,86 The viscosity polytherms either represent dynamic27,41,54,55,79 or kinematic26,40,57,58,80 temperature-dependent viscosity.
In order to compare available viscosity datasets, kinematic viscosity was transformed into dynamic using the density datasets.24,87–89 The experimental viscosity isotherms were determined using the methods proposed in the literature.26,27,40,56,80 Except for the datasets,40,79 the operating conditions for the aforementioned experiments, have been described in detail. In addition, the viscosity dataset of Al-rich alloys containing up to 3.3 at.-% Ni, 79 differ up to 300% from the datasets41,57,58 and these data were not taken into consideration.
Containerless methods
The key advantages of containerless viscosity experiments under reduced gravity conditions are twofold: the absence of turbulence makes it possible to perform measurements in laminar flow and the only contaminant of alloy samples may be trace amounts of oxygen.9,30 Concerning binary Ni–Al alloys, there are two viscosity datasets related to Al-rich compositions, that is, the Ni–68.5Al and Ni–75Al (in at.-%), obtained using the OD method in an EML device under reduced gravity conditions. 30 The experiments were performed on board PF and a TEXUS sounding rocket flight, in an Ar–He gas mixture and under flowing argon, respectively. The oxygen content was estimated to be in the range of 50–80 ppm. PF provides 20 s of reduced gravity, while the total duration of the experiments on TEXUS was 320 s, of which 150 s were available for the measurements under microgravity conditions. The viscosity of liquid Ni–68.5Al and Ni–75Al (in at.-%) alloys was measured over temperature ranges of about 400 K (Table 2) and in the investigated ranges, the two viscosity polytherms show a linear temperature dependence. A relatively large experimental error of ±15%, observed for the dataset 30 obtained by the OD–EML on board PF (Table 2), may occur due to concomitant effects of oxygen in the surrounding atmosphere and possible Al evaporation from the molten alloy samples. Indeed, SEM–EDS analyses of solidified samples indicated that the phase composition did not resemble the phase composition expected from the equilibrium phase diagram. 30
Viscosity data of liquid Ni–Al-based alloys
Container-based methods
Temperature-dependent viscosity of pure liquid metals, components of Ni–Al-based alloys (Table 1), has been analysed by many authors and their polytherms mainly obey the Arrhenius equation.17,18,22–24,87 Viscosity measurements on the IN718 10 and CMSX-4 29 liquid alloys have been performed using an oscillating cup viscometer with an alumina crucible in the flowing Ar atmosphere, detailed in the literature.19,25 Experimental viscosity polytherm of liquid IN718 alloy 10 was described by the Arrhenius equation, while that of the CMSX-4 29 obeys the linear law (Table 3).
Containerless methods
Contrary to the container-based methods used to obtain the Ni–Al viscosity datasets, those of liquid Ni–Al-based alloys (Table 3) have been mainly obtained by containerless methods. Indeed, the viscosity of four Ni–Al-based alloys, that is, CMSX-4, CMSX-10, LEK-94 and MC-2 was measured under microgravity conditions.
16
All experiments were carried out by the OD–
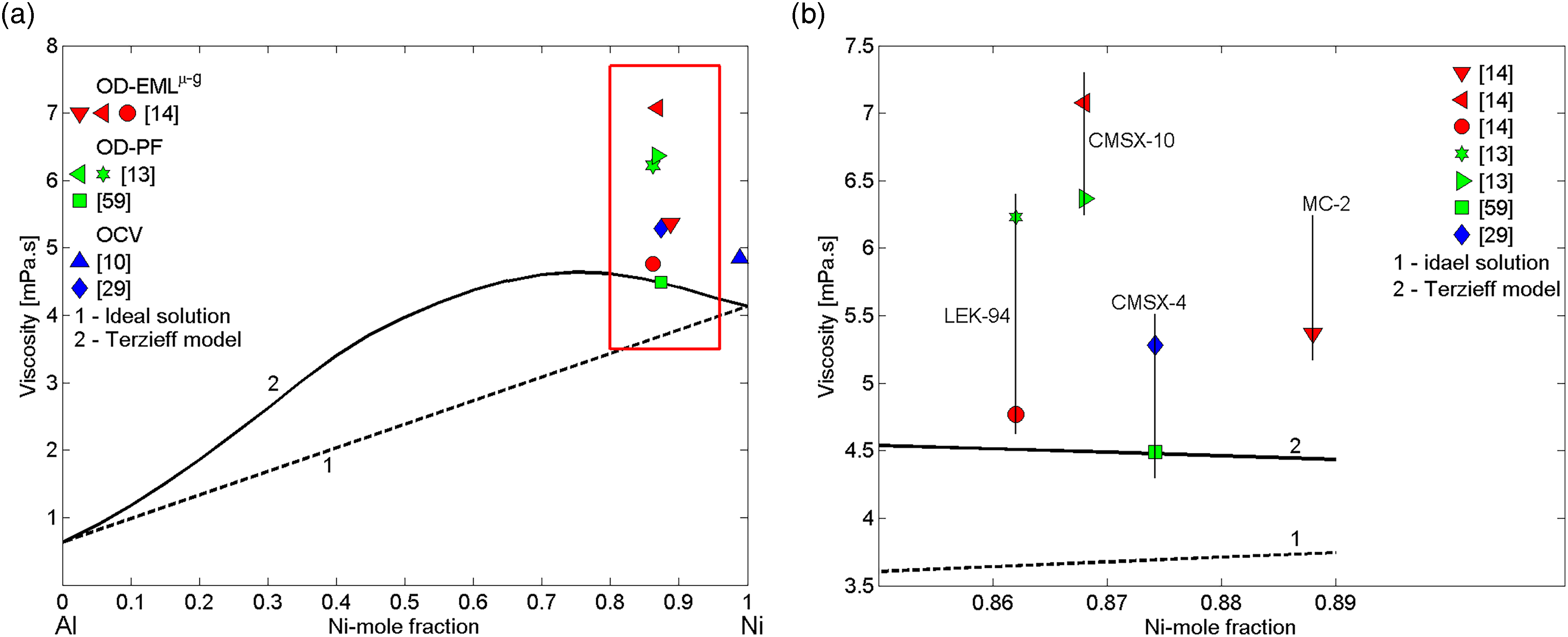
(a) Viscosity experimental datasets of liquid Ni–Al-based alloys (Table 1) obtained at T = 1823 K,10,13,14,29,59 together with the viscosity isotherms of liquid Ni–Al alloys calculated for the same temperature are shown: 1, the ideal solution; 2, Terzieff's model. (b) An enlarged area of (a) with respect to Al content in Ni–Al-based superalloys.
Model-predicted viscosity isotherms of liquid Ni–Al binary and multicomponent alloys
None of the models such as the Kaptay, Kozlov, Hirai, Moelwyn-Hughes, Iida-Ueda-Morita, Terzieff as well as Calphad type modelling, MD simulations and ab initio calculations16,30,31,71,72,90,91 compared to the Ni–Al viscosity experimental data26,27,30,55,56,61 describe accurately the behaviour of the viscosity. Around the equiatomic composition, the largest differences between the experimental data26,27,40 and the model-predicted values are observed (Figure 3). Among tested models, the viscosity isotherm calculated by the Terzieff model 72 exhibits a good agreement with those obtained by the experiments.16,30 Indeed, this model has been first used by Wunderlich and Fecht 30 to compare the viscosity data of the two binary Ni–Al alloys (Table 2) calculating the temperature dependences over the temperature ranges within 1550–2023 K. Subsequently, it was applied to calculate the viscosity isotherm of liquid Ni–Al alloys for T = 1800 K. 16 The model combines thermodynamic functions of mixing, basic thermophysical properties of pure liquid alloy components,22–24 the classical hard-sphere model parameters92,93 and four empirical constants. 71 In the present work, to calculate the viscosity isotherm of liquid Ni–Al alloys by the Terzieff model, the enthalpy of mixing data obtained at temperatures of T = 1823 ± 50 K, that is, T = 1773 K, 94 1800 K 95 and 1823 K, 96 the viscosity reference data of liquid Al 39 and Ni 53 pure metals, the molar volume and atomic mass of Al and Ni 22 together with their hard-sphere diameters,97,98 respectively, were used (Figure 3). To increase the accuracy of predicted values reducing the cumulative effect of experimental errors, the reference data39,53 taken as input data have been measured in the same apparatus and under the same operating conditions. Both experimental datasets obey Arrhenius's equation. Comparing the viscosity data for elements from different sources,17,18,22–25,28,90 the selected reference data agree well with those reported in Assael et al., 20 , 21 respectively. At high temperatures (T > 1800 K), theoretical and experimental viscosity isotherms of liquid Ni–Al alloys deviate positively from the ideal mixture (Figure 3), in agreement with the opposite trend of their thermodynamic mixing properties. Indeed, the thermodynamic properties of compound forming and segregating/phase separating alloys show negative and positive deviations, respectively, from the ideal behaviour, while their thermophysical properties exhibit an opposite trend.22,23
The viscosity datasets of Al-rich alloys41,57,58 measured at T = 1073 K exhibit a good agreement among them (Figure 4). An exception represents the viscosity data 54 that differs significantly from the experimental isotherm.41,57,58 In the case of Al-rich alloys, it was not possible to calculate the viscosity isotherms due to a large difference between the melting temperatures of Al and Ni pure metals. Indeed, the isotherm calculations for T = 1073 K imply unreliable undercooling of 700 K for Ni, in comparison with the maximum value of 300 K experimentally determined.99,100
Generally, a thermophysical property of liquid multicomponent alloys can be estimated taking into account the corresponding property of a principal binary and/or ternary subsystem, formed by major alloying elements, and the effects of the minority components on that property. 15 Bearing in mind that the viscosity values of liquid or undercooled Cr, Co, W, Ta, Mo, Ti, and Hf17–22 minority components are higher than that of Ni, these elements contribute to an increase in the viscosity of Ni–Al-based multicomponent alloys. Accordingly, the viscosity datasets of Ni–Al-based alloys (Table 1) were analysed with respect to the Terzieff isotherm for liquid Ni–Al alloys (Figure 5). The effects of the minority components on the viscosity are estimated quantifying their cumulative impact on it. To this aim, the viscosities of Ni–Al-based industrial alloys with respect to the Al content in each alloy are shown in Figure 5. For the alloys investigated (Table 1), their Al content ranges between 11 and 15 at.-%, and, therefore, in order to have a clear picture of their viscosity, the data are shown in an enlarged area for 86 < Ni (at.-%) < 89 (Figure 5(b)). An exception represents IN718 with 1.1 at.-% Al (Figure 5(a)). Taking into account the experimental errors up to 30% of the measured viscosity values of Ni–Al-based alloys, the corresponding values of binary alloys can be useful to provide lower bounds for the viscosity of these complex alloy systems.
Thermodynamic input data
The thermodynamic94–96,101,102 and phase diagram103,104 data of the Ni–Al system indicate the presence of intermetallic compounds in the solid state that imply strong effects of SRO on the thermophysical properties of liquid Ni–Al15,75,105 and Ni–Al-based alloys. 16 Among five intermetallic/intermediate phases103,104 of the Ni–Al system, the NiAl results in an energetically favoured,101,102 and therefore, in the models describing the Gibbs-free energy of the liquid phase, the prevalence of NiAl dimers in the liquid phase 12 has been postulated.12,15 The concentration dependence of the equilibrium values of NiAl chemical complexes for T = 1923 K, calculated by the compound formation model (CFM), 12 exhibits the symmetry around the equiatomic composition, with the maximum value of about 0.375. 15 Decreasing the temperature to 1823 K, the number of NiAl associates in Ni–Al melts including a broad range of undercooled alloys (37 < Ni, at.-% < 67) increases reaching its maximum of 0.392 (Figure 6). A well-pronounced chemical SRO in alloy melts that are characterised by very strong interactions between the constituent atoms, such as the Al–Ni, 15 Al–Co 77 and Ag–Hf 106 affects significantly the shape of the curves describing thermophysical properties. Indeed, the evidence for irregularities on their isotherms is at or close to the composition of an energetically favoured intermetallic compound. In strongly compound forming systems, the presence of few intermetallics may cause concomitant effects of SRO elements with different stoichiometries, 15 resulting in slightly displaced irregularities on the isotherms with respect to the composition of an energetically favoured intermetallic phase.77,106
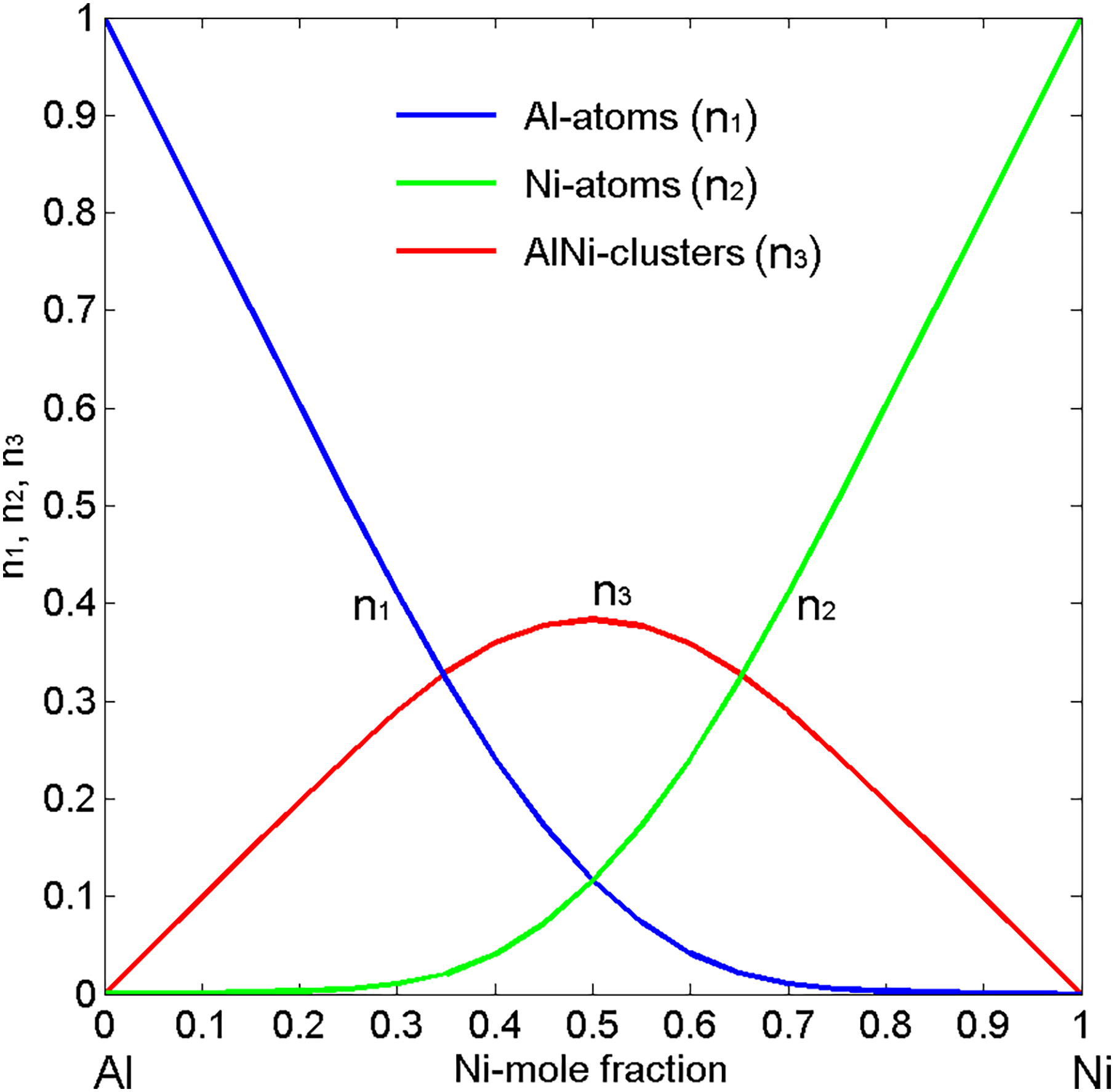
Further information on the ordering phenomena in the Ni–Al system can be deduced from the two microscopic functions, that is, the concentration fluctuations in the long-wavelength limit
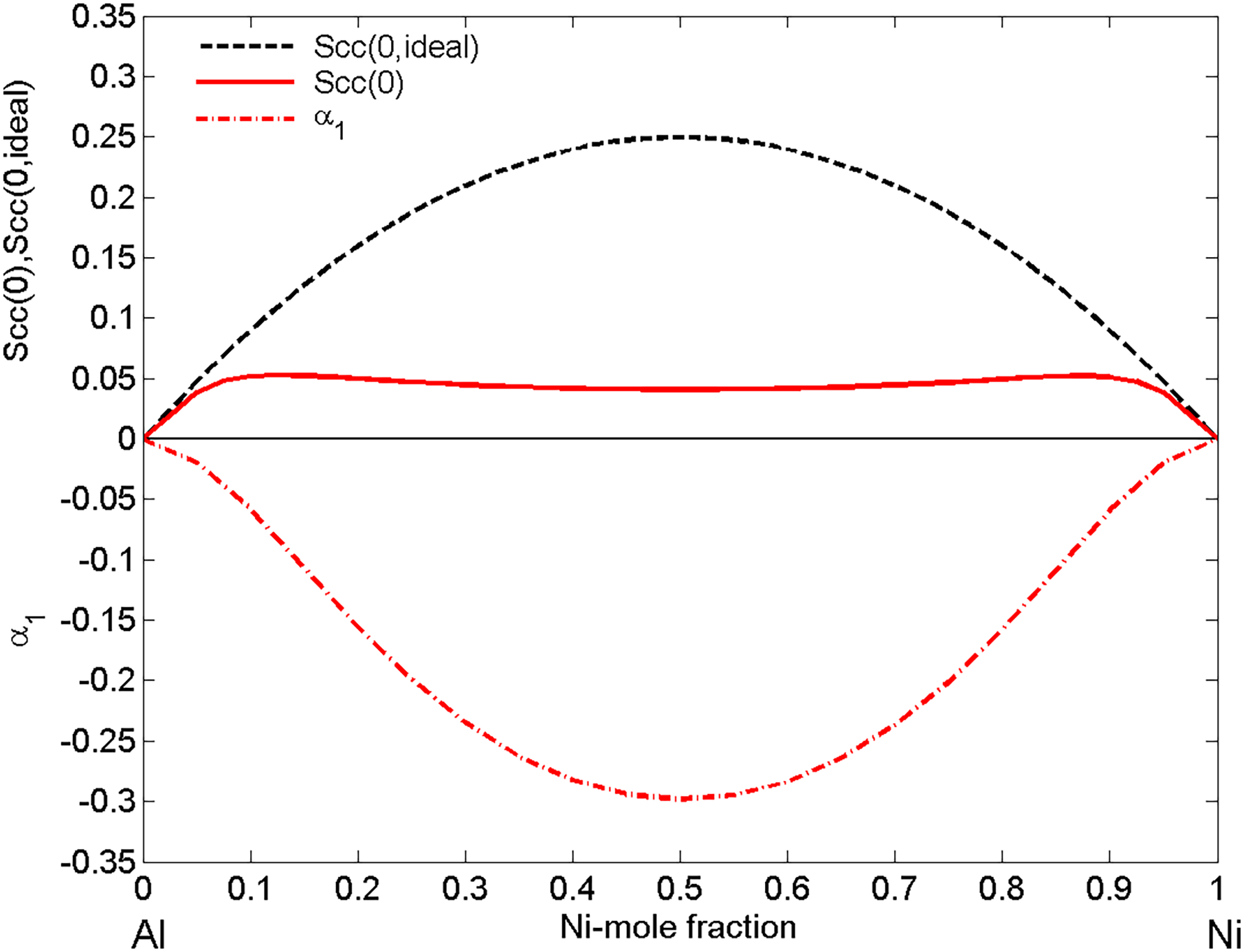
Concentration dependence of the microscopic functions of liquid Ni–Al alloys: the concentration fluctuations in the long-wavelength limit (
The microscopic functions
Discussion
Strong exothermic reactions in the Ni–Al and Ni–Al complex compound forming systems, as well as a close structural similarity of the solid and liquid phases by means of the arrangement of atoms in the two phases, are evident at or near the melting temperatures of these alloys. These characteristics of binary and multicomponent Ni-based alloys are supported by their thermodynamic, thermophysical and structural properties.15,16,22,24,101–104,111 In the following, the viscosity datasets of liquid Ni–Al26,27,30,40,55,56 and Ni–Al-based multicomponent alloys10,13,14,29,59 were analysed separately with respect to the measurement methods applied. The alloys of each group were compared among themselves and with the corresponding predicted values calculated by Terzieff's model.72,73
Viscosity and SRO in Ni–Al melts
Short-range ordering phenomena cause anomalies in thermophysical properties that exhibit large deviations from ideal behaviour and are visible on more or less irregular isotherms.15,77,78,106 Among the theoretical descriptions of thermodynamic and surface properties there are a few models that take explicitly into account the effects of SRO on these properties.12,15,22–23,33,112 Contrarily to the above-mentioned, until now SRO has not yet understood how SRO affects the viscosity of metallic melts and these effects are not explicitly included in the viscosity models.22,23,34,72 An exception represents the viscosity model based on the Stokes–Einstein relation76–78 that is indirectly related to the chemical SRO in terms of
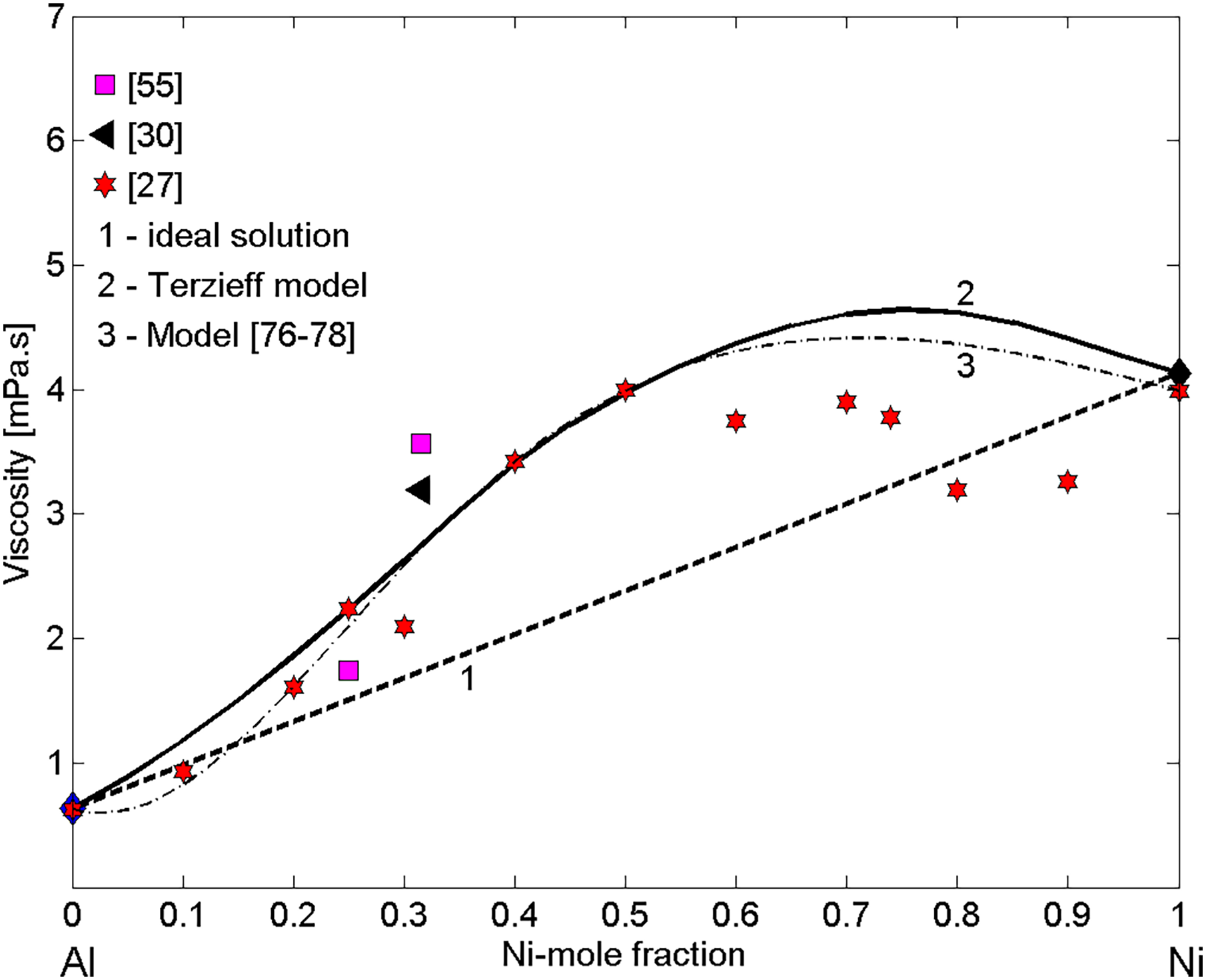
Viscosity experimental datasets of liquid Ni–Al alloys (Table 1) obtained by container-based methods27,55 and containerless method 30 together with the viscosity isotherms calculated for T = 1823 K. 1, the ideal solution model; 2, Terzieff's model; 3, the viscosity model based on the Stokes–Einstein relation.76–78
The two viscosity isotherms are similar and none of them indicates the pronounced effects of SRO at the equiatomic composition, as has been shown by the experimental data reported in the literature.27,30,55 According to the thermodynamic properties including the phase diagram of the Ni–Al system,101–104 among five intermetallics, the NiAl is energetically favoured and thus, as it was already mentioned, the prevalence of NiAl dimers in alloy melts occurs. The viscosity isotherms (curve 2) and (curve 3) calculated by Terzieff's model and by the model based on the Stokes–Einstein relation exhibit the maximum values of 4.65 and 4.36 for 76 and 71 at.-% Ni (Figure 8), respectively, that can be attributed to concomitant effects of NiAl, Ni5Al3 and NiAl3 SRO elements on that property.
Viscosity of liquid Ni–Al and Ni–Al-based alloys: experiments versus theory
Excluding the viscosity dataset
40
which differs significantly from the other data of liquid Ni–Al alloys,26,27,55,56 obtained by container-based methods, an agreement within experimental errors among these datasets can be observed (Figure 3). On the other side, very high viscosity values26,40,56 measured at the equiatomic composition over the temperature interval of 1873 ≤ T/K ≤ 1973, exhibit significant scatter with respect to the most recent data reported in Kehr et al.
27
This may be caused by oxidation and/or evaporation phenomena, because each phenomenon separately leads to an increase in the viscosity.
30
Unfortunately, the microstructural analysis of the solidified samples26,40,56 was not reported. A lack of experimental details in the literature26,55,56 excludes the possibility of repeating the experiments and may be a potential reason for inconsistent experimental results. The viscosity datasets of liquid Ni–Al alloys26,27,55,56 generally agree among them over the whole composition range, while those of the alloys with Ni-content ≤ 50 at.-% are also comparable to the corresponding model predicted values, in particular with regard to Kehr et al.
27
On the contrary, the experimental viscosity data of Ni-rich alloys containing more than 60 at.-% Ni26,27,56 indicates the values close to that of pure liquid Ni, differing up to 100% from the values calculated by the Terzieff model (Figure 3). One of the main reasons may be related to the high evaporation losses of Al. A good agreement between the viscosity datasets of Al-rich alloys41,54,57,58 measured at T = 1073 K by container-based methods can be observed (Figure 4). Concerning the theoretical predictions, neither the ideal solution isotherm nor the Terzieff isotherm cannot be calculated correctly because the extrapolation of Ni viscosity reference data
53
from its melting temperature to 1073 K (
Regarding the containerless measurements, there are two viscosity data of liquid Ni–75Al and Ni–68.5Al (in at.-%) alloys 30 measured using the OD–EML in microgravity experiments on PF, while the second one was also processed on a TEXUS. The viscosity data of Ni–68.5Al (in at.-%) alloys 30 is close to that reported in the literature26,55,56 and agrees with the corresponding calculated value within the limits of experimental error (Figure 3). The measurements of the Ni–68.5Al (in at.-%) have been performed for temperatures ranging between 1380 and 1540 K, which is about 300 K lower than T = 1823 K, and therefore viscosity data are not comparable to the other datasets.
The main problem of developing and approving viscosity models for complex liquid alloys, that is, mathematical and statistical models that involve multiple variables, is related to the lack of experimental data or divergent trends of experimentally determined datasets used as input.22–24,34 Regarding multicomponent alloys such as Ni–Al-based superalloys, the discrepancies in the reported viscosity data may occur due to slightly different alloy compositions, declared for the same alloy by producers, different measurement methods and measuring equipment as well as different operating conditions. 16 On the other side, depending on the composition of Ni–Al-based alloys, the viscosity measurements were performed within the temperature range of 1560–1865 K (Table 3) and therefore, the validation of the experimental datasets includes the viscosity isotherms calculated for the above-mentioned temperatures or those representing reasonable extrapolation of the model calculations into the metastable undercooled region. Otherwise, the theoretical predictions become unreliable. For example, in the case of liquid Ni–Al-based alloys (Table 1), the calculations of the viscosity isotherm for T = 1823 K using the simplest model such as an ideal mixture, require the viscosity reference data of undercooled transition and refractory metals, that is, Hf, Nb, Mo, Ta, Re and W 44 involving unreliable undercooling between 700 and 1800 K. Accordingly, assuming ideal solution behaviour, the evaluation of viscosity of liquid Ni–Al-based alloys taking into account all alloy components, is not possible because a theoretical statement needs to be validated through experimental observations. 16 This implies that for liquid Ni–Al-based industrial alloys, reliable theoretical predictions of their viscosities can be obtained by taking into account only the viscosities of the majority components and estimating the cumulative effects of minor components (see the ‘Results’ section). Indeed, the viscosity experimental datasets of liquid Ni–Al-based alloys (Table 3) were analysed with respect to the measurement methods applied and subsequently compared to the Terzieff isotherm72,73 calculated for Ni–Al alloys.
The viscosity of IN718 10 and CMSX-4 29 were measured over temperatures ranging between 1635 and 1800 K by container-based methods using an oscillating cup viscometer.16,22,89 Low Al content, absence of Ta and Re refractory metals together with high Fe content in IN718 alloy (Table 1) contribute that its viscosity is about 20% higher than that of the corresponding Ni–1.1Al (everywhere metal content is in at.-%) alloy (Figure 5(a)). The two viscosity datasets of liquid CMSX-429,59 were compared with the calculated value for the Ni–12.58Al, indicating the data 29 as more reliable, while the lower viscosity value 59 overlaps with that of Ni–Al binary alloy (Figure 5(b)). Possible causes of low viscosity 59 may be attributed to short time measurements during PF. The slightly higher viscosity of liquid MC-2 with respect to that of CMSX-4 29 (Figure 5(b)) can be justified by higher cumulative contents of Ni + Co + Cr (81 at.-%) and refractory metals (about 7.8 at.-%) in comparison with the corresponding values of CMSX-4, equal to 80 and 6.9 at.-%, respectively. Among Ni–Al-based alloys (Table 1), LEK-9413,14 has the highest Al content combined with the lowest cumulative content of transition and refractory metals (about 4.9 at.-%), and thus, one may expect the lowest viscosity value of LEK-94. This implies that the viscosity value of LEK-94 measured in long-duration microgravity experiments on board the ISS 14 seems to be more reliable than that obtained during PF. 13 On the contrary, high cumulative contents of Ni + Co + Cr (80 at.-%) and refractory metals (7.1 at.-%), in particular Re + Ta (about 4.8 at.-%) that significantly increase the viscosity of CMSX-10,13,14 indicating the value obtained on board ISS 14 as more reliable (Figure 5(b)).
Taking into account Terzieff's isotherm for the Ni–Al system, Al and Ni contents, as well as cumulative contents of transition and refractory metals in each multicomponent alloy (Table 1), the recommended viscosity values for liquid Ni–Al-based alloys are suggested. To make effective data analysis further measurements are needed.
Summary
Thermophysical properties data including viscosity data are used as input for numerical simulations of solidification, which is the key step of all liquid-assisted industrial processes. Viscosity measurements of Ni–Al alloys have been mainly performed by container-based methods, while for Ni–Al-based alloys, alternative, containerless methods were used. The most advanced containerless methods under microgravity conditions, performed with EML on board the ISS or during PF, make possible accurate measurements at high temperatures over large temperature intervals with significant ranges of undercooling. The viscosity of liquid Ni–Al and Ni–Al-based industrial alloys has been measured by container-based and containerless methods and, within each group of alloys, the results obtained were compared among them as well as with Terzieff's model predicted viscosity isotherm of Ni–Al alloys. Regarding the viscosity datasets of liquid Ni–Al alloys, the validation of Terzieff's model confirms a good agreement for the alloys containing up to 50 at.-% Ni, while the viscosity data of Ni-rich alloys differs significantly from the corresponding calculated values. Indeed, the viscosity of Ni-rich alloys is close to that of pure nickel, suggesting Al evaporation loss during the experiments. The viscosity datasets of liquid CMSX-4, CMSX-10, LEK-94, MC-2 and IN718 agree among them within experimental error and after careful data analysis, the recommended viscosity values of these Ni–Al-based alloys were assessed.
From the scientific point of view, microgravity experiments using the OD method in EML on board PFs and the ISS have very great importance, but due to the extremely high costs of these experiments, terrestrial techniques, capable of dealing with high-temperature, highly reactive materials should be developed for viscosity measurements. In the context of material design, this review gives guidelines for future research that has to include: development of predictive viscosity models, in particular those for multicomponent alloy systems; a wide use of databases containing properties data and built-in predictive models making possible comparisons between measured and calculated property values; applications of MVA for data elaboration including machine learning and more advanced deep learning. A further step may be to explore the possibilities of artificial intelligence in materials science.
Footnotes
Acknowledgements
The authors acknowledge the access to the ISS-EML, which is a joint undertaking of the ESA and the German Space Agency DLR. Parts of the reported work were conducted in the framework of the ESA projects ThermoProp and ThermoLab (AO-2009-1020 and AO-099-022).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.M. and H.-J.F. received funding from the German Space Agency DLR with funds provided by the Federal Ministry for Economic Affairs and Climate Action (BMWK) under Grant No. 50WM2143.