Abstract
Biomanufacturing is an exciting scientific and industrial venture involving the use of cells to produce important biological products that positively impact human endeavors including healthcare. This industry offers highly regarded and economically lucrative careers that markedly impact their employees’ and their families’ lives. Unfortunately, entry into these careers is not always clear or obtainable. Barriers include issues as simple as misunderstanding required qualifications as well as more substantial barriers such as the lack of access to the hands-on bioprocessing training needed for participation within this high growth industry. Here we report on our ongoing and developing work in the state of North Carolina (NC) to address workforce inequities at their source, that is, developing a coalition of Historically Black Colleges and Universities (HBCU) and one Historically American Indian University (HAIU) with the sole purpose of deploying regional biomanufacturing training hubs within the most economically depressed counties within NC. Our program has been directly vetted by and has secured funding from member companies that comprise the biomanufacturing industry such that our current venture has the promise of providing a powerful and sustained socioeconomic effect. Here we report on the conception, development and deployment of this unique program.
Keywords
Introduction
HBCU/HAIU coalition logical framework.

Program description
The Biomanufacturing Research Institute and Technology Enterprise (BRITE) at North Carolina Central University (NCCU), an HBCU located in Durham, NC, has led the development of a coalition of Historically Black Colleges and Universities (HBCU) and one Historically American Indian University (HAIU), the first of its kind in NC, in the deployment of regional bioprocessing training hubs within the most economically depressed counties in the state of NC. These regional assets aim to create a pool of diverse workers who can directly participate in NC’s rapidly growing life sciences manufacturing industry and make these regions attractive to investment from the Biomedical and Pharmaceutical industry as the workforce pools grow. We also expect that this newly minted coalition can be leveraged to deploy other programs in these economically distressed areas, and the modular nature of this proposal makes expanding the coalition a relatively easy process.
In response to the 2022 US Economic Development Administration Build Back Better Regional Challenge (EDA-BBB), which sought to transform regional economies through groups of complementary, aligned projects that will help regions recover from the coronavirus pandemic and increase resilience, NCCU and its BRITE Institute proposed a network of bioprocessing training hubs (Figure 1) using an established infrastructure at 10 of the 11 NC HBCUs and the only HAIU. To our knowledge, this is the first coalition in NC comprised of solely minority serving institutions. This program delivers an already established bioprocessing training workshop vetted by the National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) and already in use at BRITE. This modular approach ensures all training sites are equal and functional. At the same time, the modular aspect of these assets permits their future deployment to other regions of the state or country. As described in our logical framework (Table 1), the goal of the coalition is to increase accessibility to biopharmaceutical careers for rural and underrepresented populations by leveraging the BRITE-led coalition of HBCUs and an HAIU. 2022 North Carolina County Distress Tier Designations and Training Hub Locations. NC Federal Information Processing Standard (FIPS) codes, which uniquely identify counties and county equivalents in the United States, are also indicated in “parentheses”.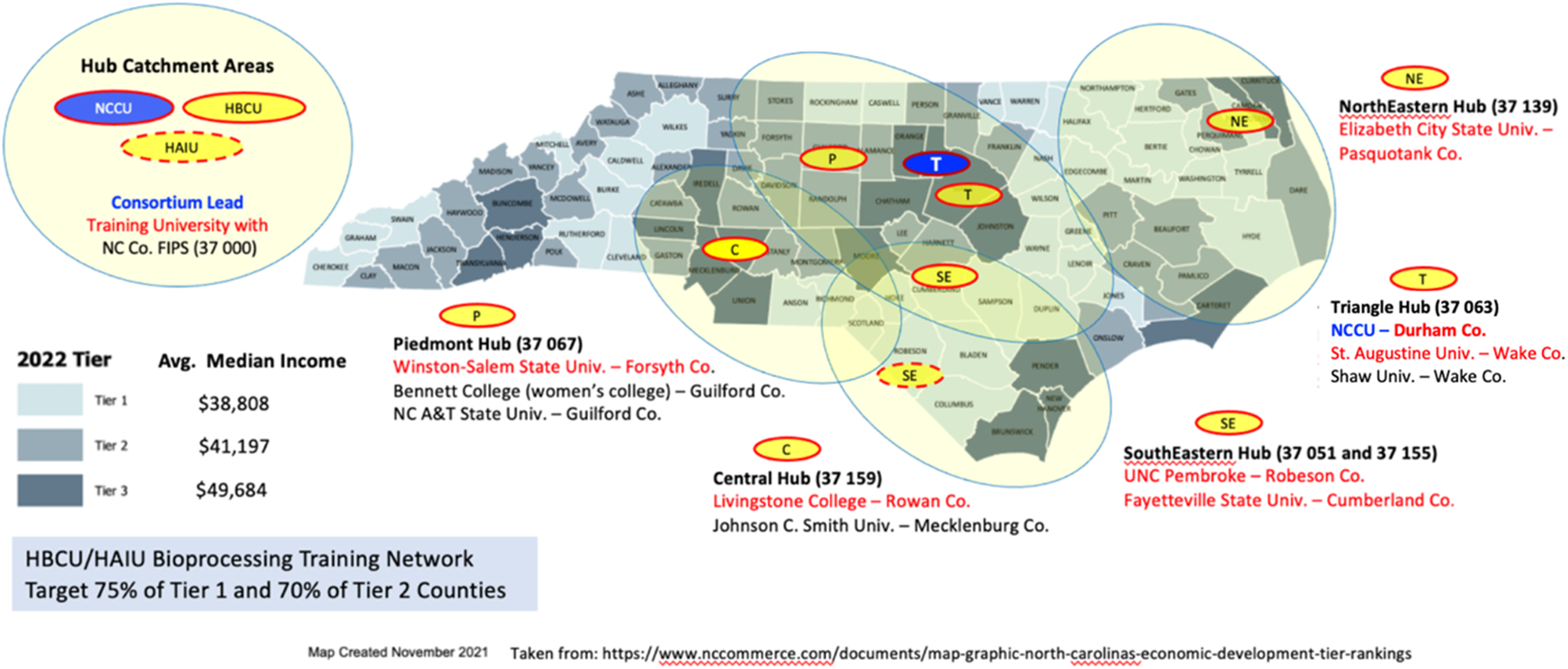
The BRITE institute has been a respected leader and partner in workforce development for the NC Pharma industry for over 15 years. The remaining coalition universities are primarily located in the most diverse and economically distressed regions of the state (Tier 1 and 2 counties; Figure 1). The NC Department of Commerce annually ranks the state’s 100 counties based on economic well-being and assigns each a Tier designation. This Tier system is incorporated into various state programs to encourage economic activity in the less prosperous areas of the state. The 40 most distressed counties are designated as Tier 1, the next 40 as Tier 2 and the 20 least distressed as Tier 3. Tier 1 and 2 regions need economic development and careers offering higher pay, advancement, career mobility, health benefits and retirement plans. The fast-growing NC Biopharma industry offers such careers. Unfortunately, the citizens in these communities do not have access to the hands-on bioprocessing training needed for them to participate in this growth and to encourage regional Pharma investment.
Thus, this industry standard bioprocessing short course is being conducted multiple times a year at each hub using NIIMBL-vetted curriculum and instrumentation. To promote this program and grow community awareness, the short course is provided free-of-charge to participants during the funded project period. As follows below, to meet the goal of the coalition, special focus needs to be placed on participant recruitment, training design and implementation, as well as industry relationships.
Intended audience and recruitment
Because the biomanufacturing industry is unknown and/or misunderstood by people outside the industry, an important first step is to educate the community on what bioprocessing is and help them envision themselves in that space. To do this, we took advantage of a parallel program funded by the EDA known as the Ambassador Program (North Carolina Biotechnology Center, 2024). The Ambassador Program initiated biomanufacturing information sessions across the state in an effort to educate the general public on the topic of biomanufacturing. With this support, the regional “hubs” were able to recruit community members and university personnel to become biomanufacturing Ambassadors. This 4-h hands-on educational workshop included information about the industry, the types of jobs that were available, and a hands-on demo of what it would be like to work as part of an assembly line at a biomanufacturing facility. By providing this education to select community members, each region gained several Ambassadors who were tasked with explaining the biomanufacturing industry to those around them and communicating that anyone with a high school diploma or GED could participate in the upcoming bioprocessing short courses. In addition, several steps were taken to publicize the upcoming short courses through local news outlets that included information describing the industry. These trainings were also promoted by the North Carolina Biotechnology Center (NC Biotech Center), which ensured a wide-reaching recruitment effort. The hubs were encouraged to host kick-off events for the inaugural training to involve university and community leadership. For example, at University of North Carolina at Pembroke (UNC-Pembroke), the Chancellor, Provost and Tribal Chairman were all invited to speak.
Short courses were advertised through regional and statewide news outlets that included an interest link with a survey. Although many people across the region, state and nation wanted to take advantage of a grant-funded biomanufacturing short course, priority was given to residents of NC residing in Tier 1 and 2 counties. Selection and registration for each short course was managed through the university hub offering the course. Up to 12 participants could be selected for each short course. This number was determined by the capacity of regional hubs to administer hands-on biomanufacturing training in a lab setting.
Once selected, participants worked with the hub site coordinator to obtain information about registering for the five online biomanufacturing courses that must be completed before the 2-week short course began, that is, a prerequisite for entry to the hands-on short course training. Participants had approximately 1 month to complete the online courses, with each course taking approximately 2-3 h to complete, and participants were expected to complete the online courses 1 week in advance of the beginning of the short course. After completion, participants received a “Certificate of Achievement” from the hub site for completing the required online courses, which was in addition to the short course certificate itself, that is, the “Advanced Certificate in Biopharmaceutical Manufacturing.”
Learning time
The training consists of five online biomanufacturing modules taken prior to attending the 2-week (M-F), hands-on, lab-based, bioprocessing short course. Two instructors lead each hands-on training session.
Prerequisite student knowledge
Five online biomanufacturing modules, as described below: (1) Introduction to pharmaceutical manufacturing – What is pharmaceutical manufacturing? A beginning step to demystifying this industry. (2) Understanding the science of the biopharmaceutical industry – An overview of the overall science(s) employed within the majority of biopharma companies. (3) cGMP (current Good Manufacturing Practice) procedures and documentation – Biopharma requires a highly regulated environment for production purposes throughout the process which is combined with multiple levels of documentation. (4) Pharmaceutical facility operations – Large complex operations require sophisticated and nimble oversight. (5) Safety in the biopharmaceutical industry – Safety is first and foremost for this industry and is further discussed here.
Learning objectives
Our overall goal is to both train individual participants and to promote community awareness of this industry and opportunities available. (1) Participants will be introduced to the field of biomanufacturing. (2) Participants will understand and be able to explain the overall processes involved in biomanufacturing from start to finish. (3) Participants will understand and have a working knowledge of upstream and downstream bioprocessing. (4) Through increased exposure and hands-on learning, participants will understand what it would be like to work within this field.
Procedures
Subject matter covered and the overall program
Week 1 – Upstream bioprocessing (hands-on labs and lectures)
The first week’s module begins
On
On
Week 2 – Downstream bioprocessing (hands-on labs and lectures)
The second week
On
The workshop concludes
Instructors
Instructors apply to a “train-the-trainer” event where they participate in the 2-week in-person training from start to finish. Minimum qualifications to be an instructor included a bachelor’s degree and previous documented prior experience as an instructor.
Based on availability, two instructors are selected to lead each 2-week training course. Participation is limited to 12 students per course so the teacher to student ratio is one to 6. Daily instruction is a mixture of lecture and lab. Lecture spaces are equipped with white boards and presentation screens. All students have physical binders of all course materials prepared for them in advance.
Industry partnerships
The key assumption, as outlined in the logical framework (Table 1), revolves around our life science manufacturing partners. Most importantly, growth of the biomanufacturing industry in NC is essential for our program to be effective. With a $9.1 billion investment and over 12,500 new jobs, life science companies have already made a significant investment in NC (Haskins and Parilla, 2024). Industry partners such as Novo Nordisk and Amgen made significant financial commitments to this project, thus registering their dedication to a “growing our own” mentality and specifically targeting our minority communities. The planned growth of biomanufacturing companies such as FUJIFILM Diosynth Biotechnologies promises additional job creation, suggesting strong demand for talent in the coming years. Ultimately, the coalition and industry partners need a close integrated job placement plan to reach our goal of successfully placing 50% of graduates in jobs and internships in biopharmaceutical positions.
Current progress
At present (mid-Oct. 2024), we are beginning year three of our 4-year project performance period. We have now fully equipped and outfitted all six hub site facilities so that all sites are now capable of deploying the short course, which was the major goal for year 1 (Oct. 2022- Sept. 2023) of this project. During year 2 (Oct. 2023- Sept. 2024) of this project, instruction began, and instructors have now been deployed to deliver the short course offering at 4 (Fayetteville State University, Livingstone College, UNC-Pembroke and Winston Salem State University) of the six hub sites. In total, we have enrolled 5 separate cohorts of students, with two cohorts having participated at short course sections at UNC-Pembroke. The total for the enrolled participants across these five cohorts was 56 with 49 of these 56 individuals (an 87.5% completion rate) fully completing the short course, that is, earning the Advanced Certificate in Biopharmaceutical Manufacturing.
Demographic data for the 2nd project year.
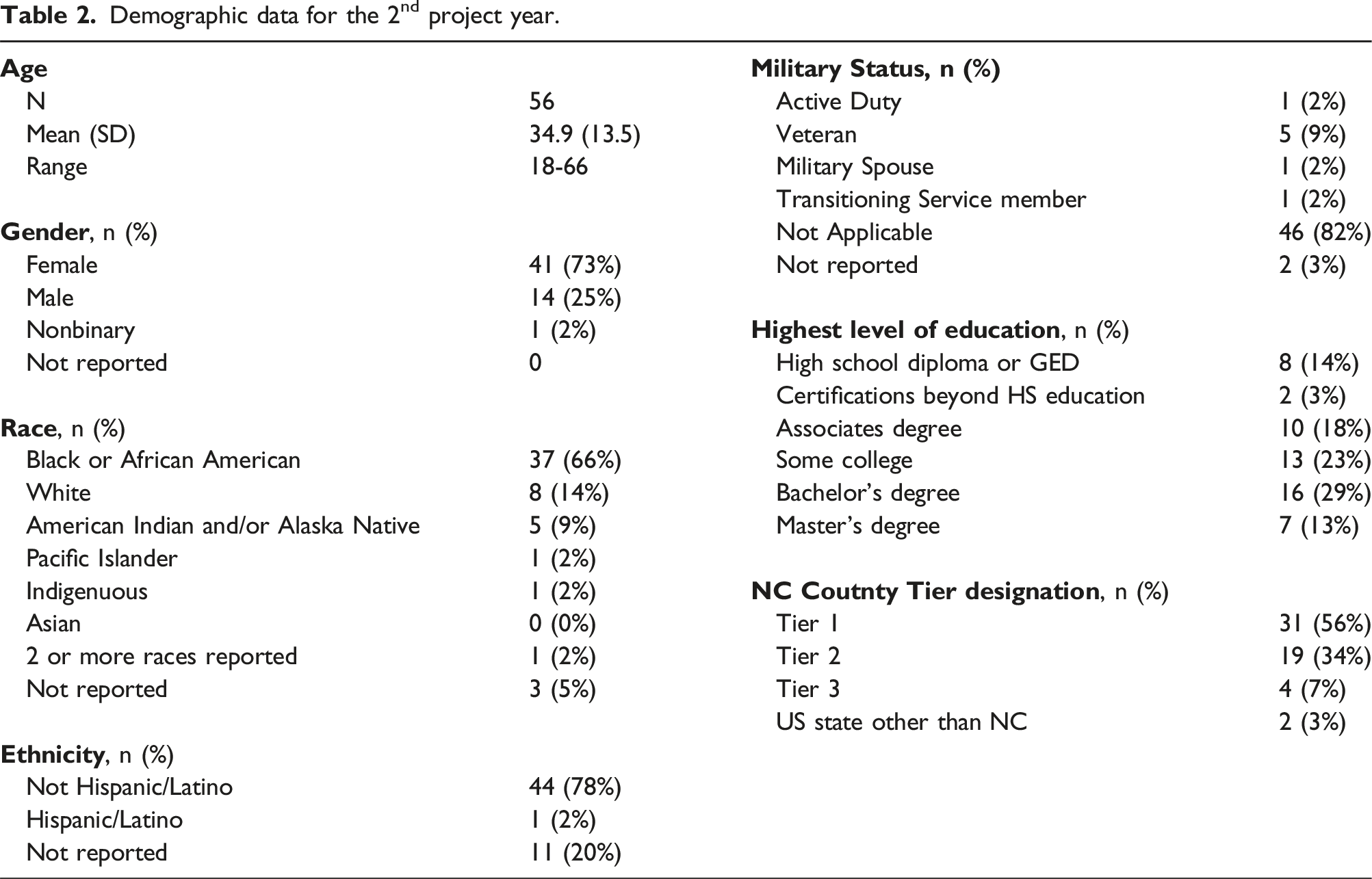
Of our success indicators outlined in Table 1, we have successfully established regional training hubs at all 6 locations. We have now successfully administered the two-week in person training program at 4 hubs with our target of 50% of program participants coming from underrepresented groups being exceeded. Although it is still too early to determine successful job placement, we recently held an information session for all of our program graduates to meet with industry partners to learn more about the companies’ missions, open positions, benefits, etc. Close partnership with our industry partners will be a key to success. In addition, after the first cohort struggled with their resume development, we have now added resume workshops to our training program.
Discussion
Expected impact
Based on data from the NC Biotech Center, BioPharma in NC expects to create ∼8000 jobs over the next 5 years (North Carolina Biotechnology Center, 2023). These jobs offer high salaries, excellent working conditions, health benefits and career mobility (North Carolina Biotechnology Center, 2023). Importantly, by prioritizing participation from Tier 1 and 2 counties and establishing training hubs at HBCUs and HAIUs, this program will increase the diversity of the industry in NC. Based upon data from a small sample of NC biotechnology companies (North Carolina Biotechnology Center, 2023), only 26% of employees identified as African American, 5% Hispanic/Latino and 0.4% American Indian or Alaska Native. These numbers are likely skewed higher due to reporting biases, yet they still indicate the need for more diversity within this industry. The success of these training hubs will create a diverse workforce pool that supports future Biopharma investment and expansion in these regions, ultimately transforming these economies. This network can also be leveraged to deploy other economic initiatives in these distressed areas.
Best practices
Based on our first five cohorts, several best practices have emerged regarding recruitment and instruction. First, in terms of recruitment and participant communication, it is preferable to have a site administrator who handles participant recruitment and correspondence. This individual is responsible for overseeing recruitment efforts, collecting potential participant survey responses, sending out course invitations, answering questions regarding the online courses and communicating with participants on what they can expect for the 2-week in-person training. In addition, a site lab manager is essential to prepare the site lab for the training. Given that the trainers are not always affiliated with the institution, the instructors need a support staff member who can assist in organizing/preparing/ordering supplies. Communication between the site administrator, site lab manager and two training instructors should be strong and is typically coordinated by the site administrator. Two to 3 weeks in advance of the training, the site administrator shares contact information for the participants with the instructors so they can reach out directly to answer more technical questions regarding the training. This level of communication has been successful in terms of keeping student interest and increasing retention from the online course portion to the in-person training portion of the program. Second, every site hosts a “kickoff” for the cohort. The “kickoff” event involves the site administrator, site lab manager, two instructors and other hub administrators meeting with the participants at the beginning of their training to provide encouragement, emphasize the importance of the program and offer support. Similarly, sites host a certificate ceremony upon completion of the program to celebrate the success of the cohort, award certificates and provide an opportunity for participants to share what they have learned. Third, many of our instructors are educators; therefore, they understand best practices for student learning particularly with adult students. For example, the “lecture” portions of the curriculum involve significant discussion and application as well as ample opportunities for participants to ask questions. In addition, instructors frequently review previously learned material to improve retention and the integration of new material. The one to six teacher-to-student ratio also impacts learning as participants receive significant individualized attention during lectures and labs.
Sustainability plan
Over the final 3 years of the grant funding period, each hub will train up to 72 participants who will be eligible for entry level positions in the field. Across six hubs, this impact will multiply to 432 participants. Beyond this training, each hub is now fully equipped to provide biomanufacturing trainings and courses for students and community participants. The bioprocessing labs and associated short course will become a self-sustaining asset of each hub’s university by project end. Our model permits hubs to deliver content using NCCU’s contract instructor model or by hiring dedicated staff. Regardless, these assets can be maintained through course fees covering short course operating costs. However, high course fees could be a barrier to entry in the Tier 1 and 2 counties. Therefore, we are working with private and public partners to provide operating support, reduced short course cost, or both. We are also leveraging the extensive and well-established system of NC Minority Serving Institutions (MSIs), which has sped acceptance of the programs because of the deep and long-standing relationships these institutions have within their regional communities. Thus, this consortium of MSIs is of significant importance to the success of the training hubs, which is essential to attracting future economic investment. By building sustained workforce capacity in these regions, industry partners will be more likely to provide additional investments in these communities, contributing to their economic growth and vitality. In serving as a liaison between industry and community, the MSI consortium is well-established to change the biomanufacturing workforce landscape of NC.
Footnotes
Acknowledgments
The support and collaboration received by the following academic institutions, companies and their representatives are gratefully acknowledged (in alphabetical order): Amgen, Elizabeth City State University, Fayetteville State University, Livingstone College, Novo Nordisk, the North Carolina Biotechnology Center, Saint Augustine’s University, the University of North Carolina at Pembroke and Winston Salem State University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported in part by a grant from the US Economic Development Administration (EDA; Award# 04-79-07800) as well as funding from Amgen, Novo Nordisk and the North Carolina Biotechnology Center.
