Abstract
Background
Health Canada uses risk management plans (RMPs) to identify and monitor risks associated with drugs in the postmarket phase but does not make the contents of these RMPs public.
Objective
To identify information about RMPs that is available in other Health Canada documents and to analyze extent of the information.
Methods
A list of new active substances (NAS) approved by Health Canada between July 1, 2015 and May 5, 2025 was generated. These drugs were searched on the Summary Basis of Decision website and any information about the contents of the RMPs was copied verbatim. The information was read iteratively and grouped into themes.
Results
There were 385 NAS approved and 381 had RMPs. For 301 the only information was that they had been reviewed by Health Canada and found to be acceptable. The information about the other 80 was typically vague and revealed little about how the information that they generated would be used to enhance safety.
Conclusion
Health Canada needs to make RMPs publicly available, should explain how their implementation will be monitored and evaluated, how the information they generate will be used to enhance drug safety and should publish updates to them as necessary.
Introduction
When a new active substance (NAS, a molecule never previously marketed) is approved, relatively little is known about its safety profile. This situation exists for a number of reasons including the population in the preclinical trials being relatively homogeneous 1 and the rule of three in detecting side effects, that is, you need three times as many subjects to observe an event when you assume that the adverse event of interest does not normally occur in the absence of the medication. Therefore, if a side effect occurs one time in a thousand, three thousand patients have to be observed. 2 The median number of patients enrolled in pivotal trials for drugs approved in Canada from 2012 to 2022 was 457 3 meaning that safety problems occurring in fewer than 150 patients may have been missed.
In part, to help fill this knowledge gap about drug safety, since June 2015 Health Canada has adopted the use of risk management plans (RMP). Companies are expected to file RMPs when they submit an application to have a NAS approved if there is a significant degree of uncertainty respecting the risks associated with a particular drug or when a particular drug presents a serious risk of injury to human health that warrants measures to reduce the probability or severity of such an injury. 4 However, the submission of a RMP until April 1, 2027 is voluntary on the part of the manufacturer. 5 An RMP usually involves both pharmacovigilance and risk minimization activities. The former includes signal detection, the submission of regulatory safety summary reports, postmarket safety and utilization studies, clinical trials and patient registries. The latter are activities such as modifying the content of product labeling and the type of packaging, drug administration training, developing educational material for healthcare professionals and patients, restricting access and distribution and risk communication. 6
According to Health Canada, “The main objective of the Risk Management Plan (RMP) is to identify how risks will be minimized and monitored to ensure that the benefit of a particular drug used in the treatment of patients outweigh the risks.” 7 At the same time, the contents of RMPs are not made public, nor is there any information about how they are being used in practice to monitor and evaluate drug safety or when RMPs are updated. However, Health Canada does point to two sources of information about drug safety and RMPs 7 ; the Drug Product Database, 8 which “provides high-level information on additional risk minimization measures included in the product’s RMP” 7 and the Summary Basis of Decision 9 document that “includes information on the approved RMP, if any.” 7
There has not been any previous analysis of Health Canada’s RMP system. The purpose of this study is to investigate the amount of information about RMPs in these two sources. Expanded knowledge about how RMPs work is necessary to understand how they can be used to enhance drug safety.
Methods
List of new active substances approved
A list of all NAS approved between July 1, 2015 and May 5, 2025 was generated from two sources. The first was the annual reports, until March 30, 2022, from the Therapeutic Products Directorate and the Biologics and Radiopharmaceutical Drugs Directorate, previously available from
Retrieval of information in RMPs
Each drug was searched on both the Drug Product Database 8 and Summary Basis of Decision 9 websites and any information about RMPs was recorded verbatim. RMPs requested by Health Canada once drugs were already marketed and for generics and biosimilars were not evaluated.
Data analysis
Counts were made of the number of NAS with and without RMPs. The information collected about the contents of the RMPs was read iteratively and then categorized into themes and the number of times each theme was mentioned was recorded. (A single RMP could mention more than one theme.) No statistical analysis was undertaken.
Patient involvement, ethics, and reporting
No patients were involved in this study, all data were publicly available and ethics approval was not required. Data were gathered by a single individual between May 10 and 15, 2025. The 2007 STROBE guidelines for cross-sectional studies were followed. 11
Results
Three hundred and eight-five NAS were approved. One drug did not have an entry in the Summary Basis of Decision website and three drugs did not have a RMP leaving 381 NAS with a RMP for analysis (Figure 1). The only information on the Drug Product Database about RMPs was the statement that “A Risk Management Plan (RMP) for this product was submitted.” The only information in the Summary Basis of Decision about the RMP for 301 (79.0%) drugs was that the RMP was reviewed by Health Canada and found to be acceptable (Supplemental File 1). PRISMA flow diagram.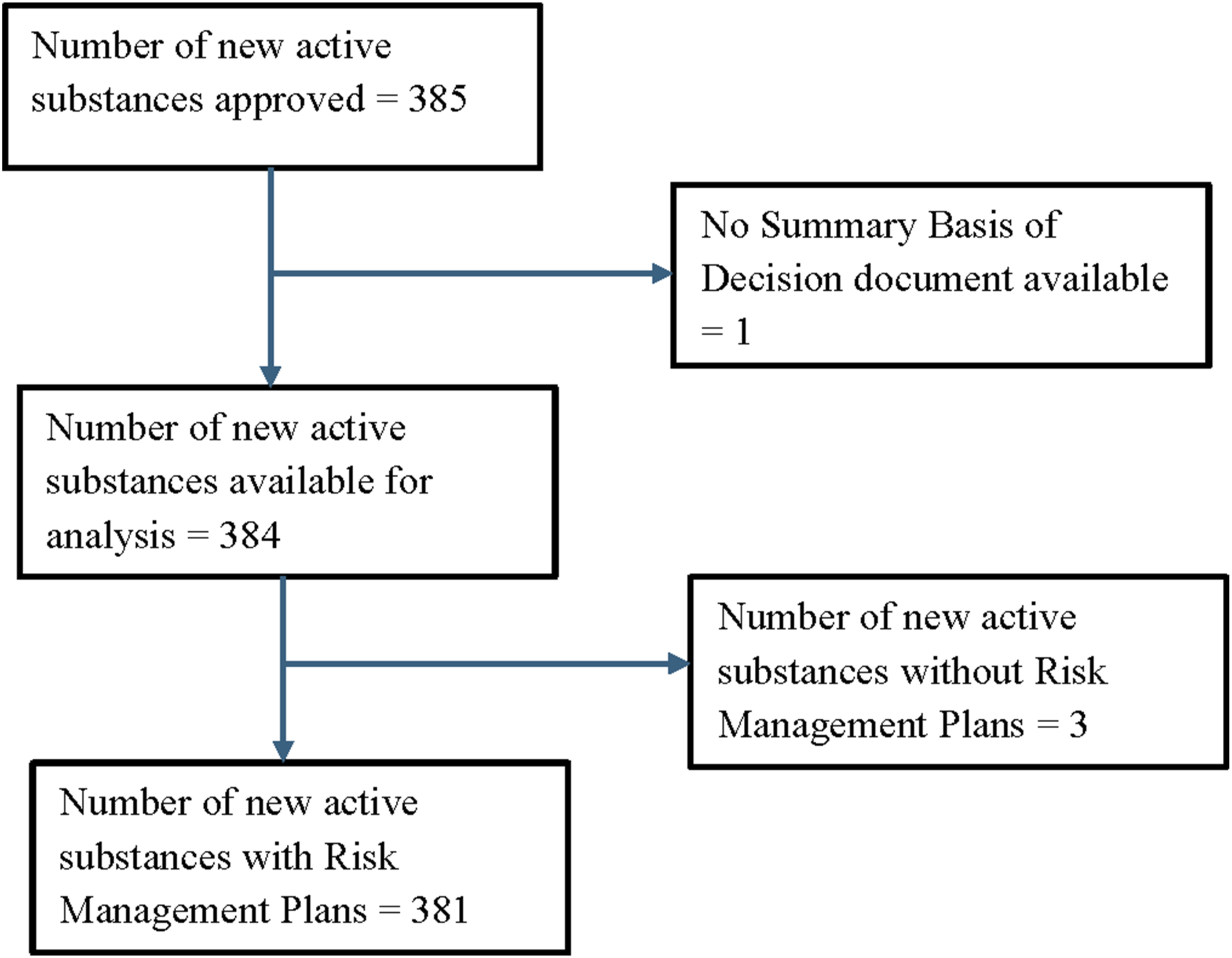
Themes in information about Risk Management Plans.
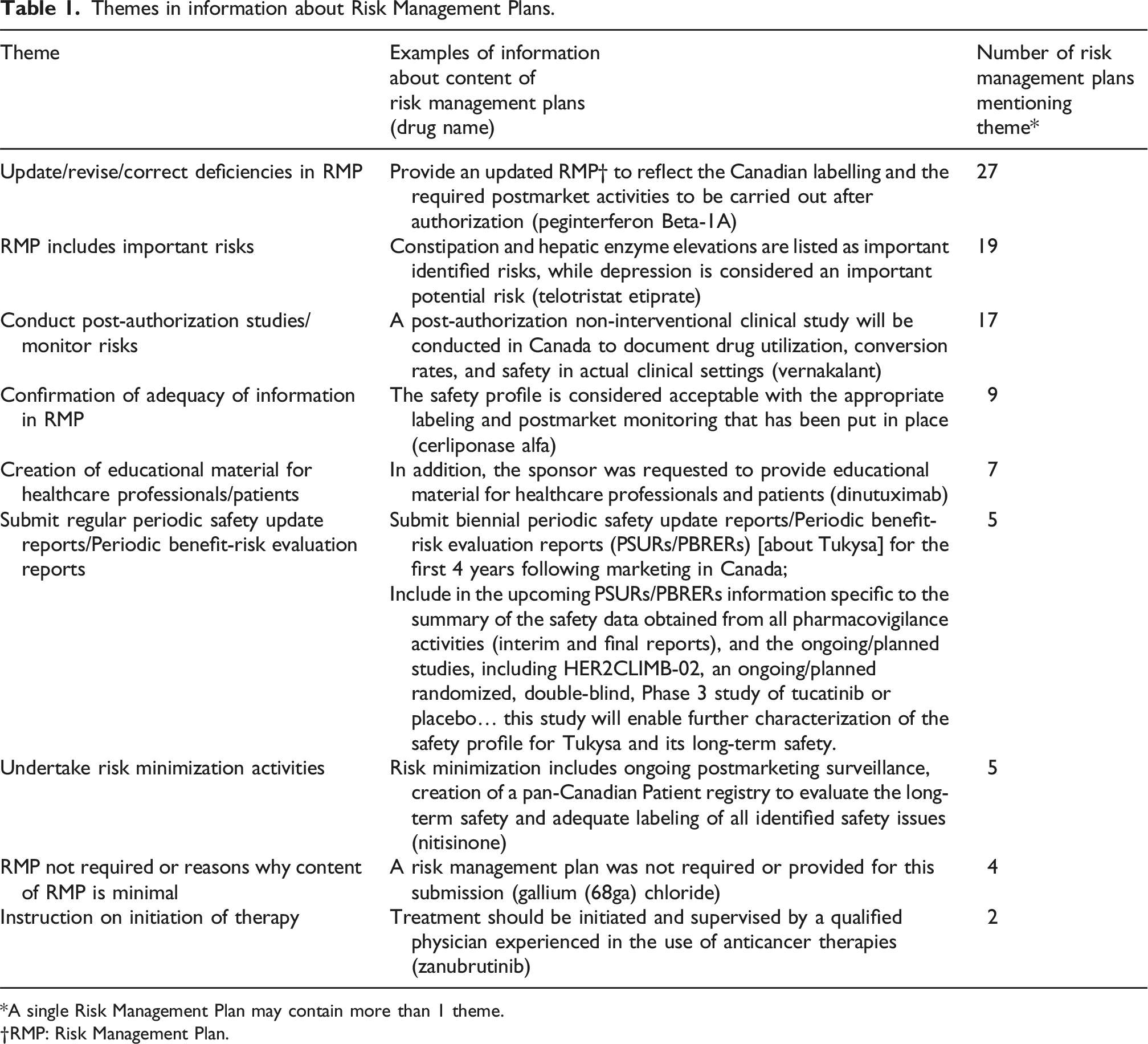
*A single Risk Management Plan may contain more than 1 theme.
†RMP: Risk Management Plan.
Even when the RMP contained a greater amount of information, there were still significant deficiencies. For example, according to the RMP for tucatinib upcoming safety reports were to include safety data obtained from all pharmacovigilance activities and the ongoing/planned studies, including HER2CLIMB-02, an ongoing/planned randomized, double-blind, Phase 3 study of tucatinib or placebo. But there was no timeline attached to this commitment and no indication how this information would be used beyond that it will further enable the characterization of the drug and its long-term safety.
Discussion
Virtually all the NAS approved by Health Canada have a RMP and since the submission of RMPs at the time of drug approval is still voluntary, it indicates that manufacturers seem to believe that there are significant unresolved safety issues associated with their drugs when they are approved. However, Health Canada provides almost no transparency about contents of those RMPs nor how they are used. Seventy-nine percent of the time, Health Canada merely says that the RMP was reviewed and found to be acceptable. The other 21% often just say that the RMP needs revising or updating, describes the risks included in the RMP and says that the manufacturer has committed to ill-defined postmarket studies or trials, although occasionally there is some concrete information.
In addition to problems with transparency there are also concerns about the design and administration of the RMPs. Both functions are carried out by manufacturers although Health Canada has to approve the RMPs. However, the commercial interests of companies may introduce biases in them, exemplified by the Suboxone Education Programme. 12 The program, with a brand name in its title, was developed by Indivior, the corporate rights-holder for Suboxone, as part of a Health Canada regulated RMP. It is mandatory in some provinces to complete the program before healthcare professionals are able to prescribe buprenorphine-naloxone despite there being good, accredited, non-industry educational programs already available to teach appropriate opioid addiction care and prescribing. The inclusion of the brand name in the title flies in the face of the recommended practices and accreditation standards for Canadian medical education. 13
Equivalents to the RMP system exist in other jurisdictions. The Food and Drug Administration uses its Risk Evaluation and Mitigation Strategies (REMS) program to identify and minimize risks in the United States in the postmarket period but is much more transparent about how REMS work. There is a public website that lists all the drugs with REMS and contains the goals of the individual REMS, a summary of the measures included in them, the materials that have been developed as part of the REMS and how the effectiveness of the REMS plan is going to be assessed. 14 The European Medicines Agency also publishes the risk management plans that it has approved along with RMP updates. 15
Limitations
All the material was gathered by a single individual and that may have resulted in transcription errors although this problem was unlikely since information about the content of the RMPs was copied verbatim. The identification of themes also may have had a subjective element.
Conclusion
Health Canada should make RMPs publicly available, should explain how their implementation will be monitored and evaluated, how the information they generate will be used to enhance drug safety and should publish updates to them. In addition, Health Canada should design the RMPs, with input from the drug companies, and have them administered by a neutral third party to avoid any commercial biases in them.
Supplemental Material
Supplemental Material - Risk management plans and drug safety in Canada: A cross-sectional study
Supplemental Material for Risk management plans and drug safety in Canada: A cross-sectional study by Joel Lexchin in International Journal of Risk & Safety in Medicine
Supplemental Material
Supplemental Material - Risk management plans and drug safety in Canada: A cross-sectional study
Supplemental Material for Risk management plans and drug safety in Canada: A cross-sectional study by Joel Lexchin in International Journal of Risk & Safety in Medicine
Footnotes
Ethical considerations
All the information was publicly available and ethics approval was not necessary.
Consent to participate
No patients were involved in this study.
Consent for publication
There were no participants in this study, and therefore, no consent for publication was necessary.
Author contributions
Joel Lexchin developed the idea for this study, gathered and analyzed the data, wrote and revised the manuscript and approved its submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Between 2022 and 2025, Joel Lexchin received payments for writing a brief for a legal firm on the role of promotion in generating prescriptions for opioids, for being on a panel about pharmacare and for co-writing an article for a peer-reviewed medical journal on semaglutide. He is a member of the Boards of Canadian Doctors for Medicare and the Canadian Health Coalition. He receives royalties from University of Toronto Press and James Lorimer & Co. Ltd for books he has written. He has received funding from the Canadian Institutes of Health Research in the past.
Data Availability Statement
All the data collected for this study is in the two Supplementary Files.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
