Abstract
Background
Visual snow is an abnormal visual perception, frequently occurring in the context of visual snow syndrome. Recent literature has suggested a link with serotonin reuptake inhibiting antidepressants, but there is little research on this topic.
Objective
We aimed to identify cases of visual snow and visual snow syndrome linked to serotonin reuptake inhibiting antidepressants to evaluate the possibility of an association.
Methods
Retrospective analysis of patient adverse event reports linking serotonin reuptake inhibiting antidepressants with visual snow or visual snow syndrome using data from RxISK.org, a global database of spontaneous reports of drug-linked adverse events. Each case was subject to a causality assessment: a RxISK score of 0–4 indicates more information is required, 5–8 a likely link and ≥9 a strong possibility of a link between the medication and the symptoms.
Results
24 cases were identified; 16 male and 8 female patients, with a mean age of 30 years. All had visual snow, and 10 patients (42%) had visual snow syndrome. Reports originated from 8 countries and involved 10 different drugs. Symptoms began on the drug in 14 cases (58%), after reducing the dose in 6 cases (25%), and after discontinuation in 4 cases (17%). At the time of reporting, 22 patients (92%) had stopped the suspect drug but without resolution of symptoms. The mean RxISK score was 9.5 (range 2–17).
Conclusion
Serotonin reuptake inhibiting antidepressants may trigger visual snow and visual snow syndrome that fails to resolve or can even worsen or emerge after stopping the drug. With usage of these medications rising worldwide, cases are likely to increase. Further research is vital to understand potential mechanisms and risk factors.
Introduction
Diagnostic criteria for visual snow syndrome.

VSS frequently coexists with migraine and tinnitus which, although recognised as distinct conditions, have shared neurobiological features. 3 The diagnosis is clinical and one of exclusion based on the patient’s history, combined with a normal ophthalmic examination and brain imaging, and not explained by migraine with aura or another disorder, or by psychotropic drug abuse.
The pathophysiology of VSS is not well understood. Measurable neurological changes have been found in patients with VSS compared to controls, indicating a widespread dysfunction in sensory processing and altered cortical excitability, 3 but the aetiology of these changes is uncertain and treatment options remain limited. Our recent study of visual problems associated with serotonin reuptake inhibiting antidepressants suggested a link with VSS, 4 yet this is not a recognised side effect of these commonly prescribed medications, and previous literature on this topic is limited to a single case report. 5 With usage of these medications increasing worldwide, 6 further investigation of this possible connection is of growing importance and may help to further our understanding of the mechanisms that may underlie this debilitating condition.
In the present study, we carried out an analysis of patient adverse event reports to identify suspected cases of VS and VSS linked to the use of serotonin reuptake inhibiting antidepressants, and to describe the characteristics, impact and strength of association.
Methods
RxISK.org is an independent drug safety website where consumers can research and report suspected side effects. 7 It was founded in 2012 by an international group including two of the current authors (DH and DM) with the aim of increasing independent pharmacovigilance data and empowering patients to have better conversations about their medication with their doctor. The adverse event reporting facility consists of a structured questionnaire including age, gender, country of origin, drug consumption, medical history, other relevant health information (e.g. smoking, pregnancy, alcohol use) and clinical details of the suspected adverse drug event. This is followed by a causality assessment based on Koch’s postulates, the Bradford-Hill criteria and the Naranjo algorithm for cause and effect, to help determine whether the reported drug is responsible for the event. A RxISK score of 0–4 indicates that more information is required, 5–8 indicates a likely link, and 9 or higher a strong possibility of a link. The report also includes a set of questions designed to capture the impact on quality of life.
Between 17 June 2012 and 4 April 2024, a total of 7501 patient adverse event reports were submitted to RxISK.org. We searched the database for reports of visual symptoms linked to serotonin reuptake inhibitors using a set of vision-related keywords taken from the Medical Dictionary for Regulatory Activities (MedDRA) terminology for vision disorders. 8 Serotonin reuptake inhibitor (SRI) was defined as any antidepressant drug that strongly inhibits the sodium-dependent serotonin transporter (SERT). This included selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and serotonin reuptake inhibiting tricyclic antidepressants. The RxISK score for each case was evaluated to determine the strength of the reported association with the SRI.
Ethics approval was not required as the study utilised secondary data, and the terms and privacy policy of the adverse event report grants the usage of anonymised data by researchers, regulators and others for the purpose of medication safety. We consulted the UK Research and Innovation Department (UKRI) which agreed that there was no need to seek ethical approval for this work.
Results
Demographics, visual symptoms and RxISK Score.
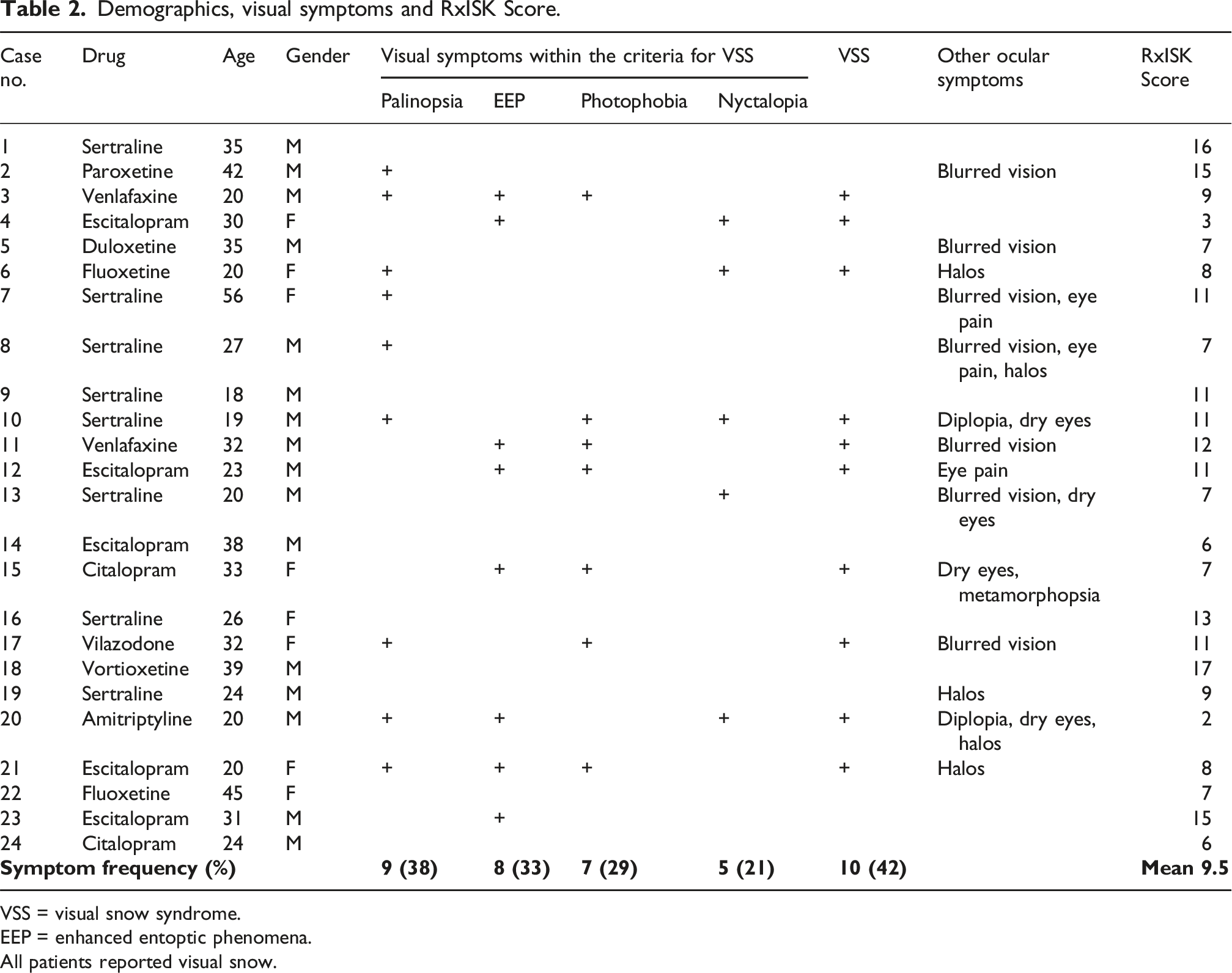
VSS = visual snow syndrome.
EEP = enhanced entoptic phenomena.
All patients reported visual snow.
All patients reported VS, and 10 (42%) reported symptoms consistent with VSS. There were 16 male and 8 female patients, with a mean age of 30 years (range 18–56 years). 10 reports originated from the United States, 7 from the United Kingdom, 2 from India, and one each from Canada, France, Israel, Poland and Türkiye. All reports were completed by the affected individuals.
Fifteen patients (63%) reported at least one additional symptom within the diagnostic criteria for VSS: palinopsia (n = 9), enhanced entoptic phenomena (n = 8), photophobia (n = 7) and nyctalopia (n = 5). Ocular symptoms outside of the criteria were reported by 14 patients (58%): blurred vision (n = 7), diplopia (n = 2), dry eyes (n = 4), eye pain (n = 3), halos (n = 5) and metamorphopsia (n = 1). Two patients reported an ocular comorbidity: cataract (n = 1) and retinopathy (n = 1).
Ten different SRIs were reported as the suspect drug. The most common was sertraline in 8 cases, followed by 5 cases on escitalopram, 2 each on citalopram, fluoxetine and venlafaxine, and one each on amitriptyline, duloxetine, paroxetine, vilazodone and vortioxetine. The duration of treatment was reported in all cases and ranged from a single dose to more than 18 years.
Symptoms began while taking the drug in 14 cases (58%), after reducing the dose in 6 cases (25%), and after discontinuation of treatment in the remaining 4 cases (17%). No patients reported experiencing symptoms of VSS prior to SRI usage.
At the time of reporting, 22 patients (92%) had already discontinued the suspect drug and were experiencing an enduring vision problem, although one had switched to another SRI which may account for the ongoing issue. Only one patient who stopped treatment reported some improvement in symptoms, but not resolution. The remainder reported a persistence, and in 4 cases, a worsening of symptoms. Eleven patients reported having an enduring vision problem for at least a year since stopping the suspect drug, with the longest duration being over 4 years since stopping sertraline.
The mean RxISK score was 9.5 (range 2–17), with 9 cases (38%) scoring between 5 and 8 indicating a likely link, and 13 cases (54%) scoring 9 or above indicating a strong possibility of a link between the medication and the side effect. It is worth noting that our causality algorithm will typically produce a lower score for events happening on discontinuation of treatment.
Twelve patients (50%) indicated that symptoms were bad enough to affect everyday activities, and 11 (46%) indicated a change in usual social activities, friendships, or relationships as a result of the problem, including descriptions of becoming socially withdrawn and isolated. 19 patients (79%) sought healthcare, and 15 (63%) reported that their work had been affected, with some noting a period of absence. Reported investigations had included standard eye examination, retinal imaging, blood and urine analysis, neurological examination, electroretinography, magnetic resonance imaging (MRI), computed tomography (CT), and magnetic resonance angiography (MRA). Five patients had received a formal diagnosis of either VSS (n = 4) or VS (n = 1).
The RxISK reporting template asked users to rate their experience on the suspect drug considering the treatment of their original condition and the side effects they experienced. From a set of 5 options (very unsatisfied, unsatisfied, neutral, satisfied and very satisfied), the results were unsatisfied (n = 3), very unsatisfied (n = 20), and one report did not have a rating.
Discussion
VS is a neuro-ophthalmological condition that frequently occurs in the context of VSS, a recently recognised syndrome comprising of VS for at least 3 months, not explained by typical migraine aura or another condition, and accompanied by at least 2 additional visual symptoms: palinopsia, enhanced entoptic phenomena, photophobia and nyctalopia.
On imaging and electrophysiology studies in patients with VSS, there is a widespread dysfunction in neurological processing. 3 On MRI, measurable changes in grey matter volume in both visual and extra-visual cortices have been found, 9 as well as white matter abnormalities in areas involving visual processing and conceptualisation 10 and changes in cortical blood flow and perfusion. 11 Electrophysiological tests indicate occipital hyperexcitability and differences in visual evoked potentials, with increased N145 latencies suggesting altered processing in the extra-striate visual cortex. 12 Changes in oculomotor control have also been found: VSS patients have unique saccadic behavioural profiles, with faster eye movements towards novel stimuli, differences in saccadic latency, and higher rates of erroneous saccades compared to controls.3,13
Most recently, researchers investigated the activity of neurotransmitters in VSS using a new technique known as receptor-enriched analysis of functional connectivity by targets (REACT), combining functional magnetic resonance imaging (fMRI) with positron emission tomography (PET) to study synchronized activity within neural networks in the brain. 14 Interestingly, they found that compared to controls, patients with VSS had significantly less-synchronised activity in networks specifically involving SERT, 5-HT2A and NMDA receptors. SRIs act not only on SERT and 5-HT2A receptors but have also been shown to modulate NMDA receptors. 15
However, central changes on imaging and other tests do not mean that the disorder arises in the brain rather than the eyes. SRI medicines are known to influence intraocular pressure through a number of actions on serotonin receptors in the iris and ciliary body.16,17 They are also carbonic anhydrase activators, and this action may lead to increased aqueous production and raised intraocular pressure resulting in some cases of glaucoma, which may respond to carbonic anhydrase inhibitors (CAI) like acetazolamide.18,19 Raised intraocular pressure can cause blurred vision, and some reporters to RxISK with closely related visual problems, complaining of visual ‘blur’ rather than visual snow, also reported benefits with CAI agents. Acetazolamide has also been used to treat visual vertigo and vestibular migraine, both of which are related to VSS.20,21
Our previous study looked at all visual side effects associated with SRIs but did not focus specifically on VSS.4,22 Aside from that study, only a single case report exists, describing a 31-year-old female who developed VSS 2 weeks after starting citalopram. 5 Despite this, 34% (53/158) of reports of VSS in the FDA Adverse Event Reporting System (FAERS) as of 31 March 2025 were linked to SRIs, 23 pointing to a possible association. FAERS reports began in 2017, possibly because the code was only added at that time.
SRIs are also linked to post-SSRI sexual dysfunction (PSSD) and persistent genital arousal disorder (PGAD). These two conditions can begin on treatment but most clearly manifest on or after discontinuing treatment and can endure for decades afterwards.24,25 The symptoms of VS and VSS described by the patients in our study had similar characteristics in that they began whilst on the SRI, did not resolve when the SRI was stopped, or in some cases emerged or worsened upon stopping. The published case report also describes symptoms that arose during treatment with citalopram and continued despite stopping the medication. 5 It is conceivable that in patients taking SRIs, the symptoms of VSS reflect an analogous process to PSSD and PGAD occurring within the visual system. We also have reports of patients with persistent postural perceptual dizziness (PPPD), another syndrome closely related to VSS which in these cases began on discontinuing SRIs.26–28
As well as the primary symptom of VS, there was a high prevalence of additional ocular symptoms reported by the patients in our study, with 63% reporting at least one of the symptoms included in the diagnostic criteria for VSS and 58% reporting a symptom outside the criteria. Serotonin is found in the retina, where evidence suggests it has a neuro-modulatory role, 29 and even in the tear film. 30 SRIs have been linked to disruption of the outer retina and retinal pigment epithelium, 31 thinning of ganglion cell and retinal nerve fibre layers 32 and ocular surface inflammation. 30 It is therefore possible that the symptoms reported by the patients in our study could reflect direct SRI effects on the eye. Nyctalopia, for example, is known to occur in retinal disease, while photophobia is commonly attributed to dry eyes, which SRIs have been shown to exacerbate. 30 A recently completed study involving two of the current authors (JL and DH) found consistently reduced thickness across the macular ganglion cell complex (perifoveal and parafoveal regions) linked to SSRIs. This area includes communication with bipolar cells which have a serotonergic input. These unpublished data are currently under review. 33
SRIs are amongst the most commonly used medications worldwide. Current usage in many countries, such as the UK, is at more than 12% of the population and seems likely to rise further, with prescriptions almost doubling over the last 10 years. 34 It is thus likely that the number of patients presenting with SRI-associated visual adverse effects will also increase. At present, many of the symptoms consistent with VSS are possibly dismissed in clinical practice because they are uncommon, poorly understood, treatment options are limited, and the condition is not considered sight threatening. The impact on the patient, however, should not be underestimated. The fact that all but two of the patients in our study stopped taking their SRI and the majority indicated that their symptoms were bad enough to affect their work, supports the distressing and potentially disabling nature of VSS, both on a personal and socioeconomic level. Raising awareness of a potential trigger is thus vital.
Limitations of this study include the retrospective design, potential for recall bias, and modest sample size. However, it is the largest published analysis of adverse event reports to date involving VS and VSS linked to one drug group. The RxISK scores generated by the patients in our study support the possibility of the medication being responsible for the adverse effect, with a ‘likely’ possibility indicated in 38% and a ‘strong’ possibility in 54%, and possibly higher bearing in mind the limitations of causality algorithms in cases like these.
In summary, our data suggest that SRIs may trigger VS as well as a range of visual disturbances consistent with VSS that generally fail to resolve upon discontinuation of treatment and can even worsen or in some cases emerge upon stopping the drug. Further research is called for to better understand mechanisms and identify risk factors.
Supplemental Material
Supplemental Material - Serotonin reuptake inhibiting antidepressants: A trigger for visual snow syndrome?
Supplement Material for Serotonin reuptake inhibiting antidepressants: A trigger for visual snow syndrome? by Hannaa Bobat, David Healy, Dee Mangin, Jonathan Lochhead in International Journal of Risk & Safety in Medicine
Footnotes
Author contributions
DH and DM collected and supplied the data. HB analysed the data. All authors contributed to writing and editing the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
