Abstract
Background
Haemophilus influenzae type b (HIB) is a gram-negative pathogenic bacterium that mostly impacts the pediatric and geriatric population, sometimes resulting in permanent sequelae or death.
Objective
In this study we set out to discover adverse outcomes from the thimerosal-free HibTITER® vaccine derived from a Florida Medicaid dataset spanning from January 2003 to June 2007 (
Methods
HIB vaccinated children were isolated from the Florida Medicaid dataset. Using other HIB vaccines as the control, we analyzed diagnoses for statistically significant adverse outcomes related to the HibTITER® vaccine (only the strictest
Results
The study revealed 19 individual ICD-9 codes positively associated (
Conclusion
Incidence of 19 different medical conditions was significantly higher compared to the control group who received other HIB vaccine formulations. These results were corroborated using the VAERS database, and have profound medical implications for the estimated 35 million Americans between the ages of 16 and 33 who received the vaccine.
Introduction
Haemophilus influenzae type b (HIB) is a gram-negative pathogenic bacterium that mostly impacts the pediatric and geriatric population, sometimes resulting in permanent sequelae or death. Patients most notably had complications related to respiration. HIB can cause mild disease such as ear infections as well as severe disease including septicemia. 1
The first HIB conjugate vaccine was licensed in December 1987 for individuals 18-month-old and older. In October 1990 it was approved by the FDA for 2-month-old infants and older. Among children 5 years and younger, HIB infection rate dropped from 37 per 100,000 in 1989 to 11 per 100,000 in 1991. 2 Representing a drop of 92%, in 2008 the infection rate had fallen to three per 100,000. 3
The Vaccine Adverse Event Reporting System (VAERS) is mandated by the National Childhood Vaccine Injury Act 4 and is co-administered by the Centers for Disease Control and Prevention (CDC) and the U.S. Food and Drug Administration (FDA). It is a self-described “national early warning...passive reporting system.” The limitations and intentions are clearly laid out by “VAERS is not designed to determine if a vaccine caused a health problem but is especially useful for detecting unusual or unexpected patterns of adverse event reporting that might indicate a possible safety problem with a vaccine.” 5 From its inception through the end of 2007, VAERS received 247,604 reports, 33,955 (or 13.7%) of which were for the HIB type vaccines.
The human immune system response to HIB capsular component polyribosylribitol phosphate (PRP) was leveraged in vaccine development. This component was used in polysaccharide vaccines licensed in the U.S. in 1985. Later specific antigens responsible for T-cell response were covalently linked to the PRP capsule, forming a conjugate vaccine. We investigate the four major types of HIB vaccine: HbOC (HibTITER®); PRP-OMP (PedvaxHIB®); PRP-T (ActHIB® and HIBERIX®); and PRP-D (PROHIBIT®). HibTITER® is a conjugate of diphtheria CRM197 protein and HIB capsular antigen and was discontinued by Wyeth in the U.S. in 2007. PedvaxHIB® is a conjugate of HIB capsular antigen and meningococcal protein and is manufactured by Merck. PRP-T is a conjugate of HIB capsular antigen with tetanus toxoid. ActHIB® is manufactured by Sanofi Pasteur and HIBERIX® is manufactured by GlaxoSmithKline. PRP-D is a conjugate of HIB capsular antigen with the diphtheria toxoid and was manufactured as PROHIBIT® by Connaught Laboratories. PROHIBIT® is no longer available in the U.S. HibTITER®’s original multi-dose formulation contained thimerosal. The multi-dose version was reformulated without thimerosal in the wake of the CDC’s June 2000 Scientific Review of Vaccine Safety Datalink Information 6 at the Simpsonwood Methodist retreat, and leading up to the 2001 publication of Immunization Safety Review concerning thimerosal-containing vaccines and neurodevelopmental disorders. 7 Thimerosal containing HibTITER® expired in 2002. The subject of this research is thimerosal-free (where thimerosal is not listed among ingredients) HibTITER® and began in 2003.
Methods
VAERS
VAERS 8 is not designed to be externally valid, as it is a voluntary reporting system with strong and complicated biases. VAERS was internally analyzed for this study. In this observational study we assessed the four major types of HIB vaccine for an intra-group comparison. Intra-group comparisons are useful in a retrospective study because, although the types of vaccines are different, the distribution and administration guidelines do not differ significantly. Though subtle factors such as location availability or institutional price negotiation may affect the decision on which HIB vaccine to administer, when distributed through the whole of the U.S. there is unlikely to be a significant selection bias. Based on this assumption we may also infer that the biases that exist surrounding VAERS report submissions do not differ significantly between the different vaccines within the HIB group. VAERS data was downloaded 9 and its elements were extracted from reports that allow for the discernment of adverse event symptoms.
Medicaid
A subset of the Medicaid database for the state of Florida utilized for this study contains a record of infants who received a HIB vaccination identified by Current Procedural Terminology (CPT) code. The HIB vaccine delineated in CPT codes are type: HbOC; PRP-OMP; PRP-T; PRP-D. The subject of this study (HibTITER®) is the only vaccine of type HbOC.
Basic demographic information for the cohort receiving the Haemophilus influenzae B vaccine (from the Florida Medicaid Database).
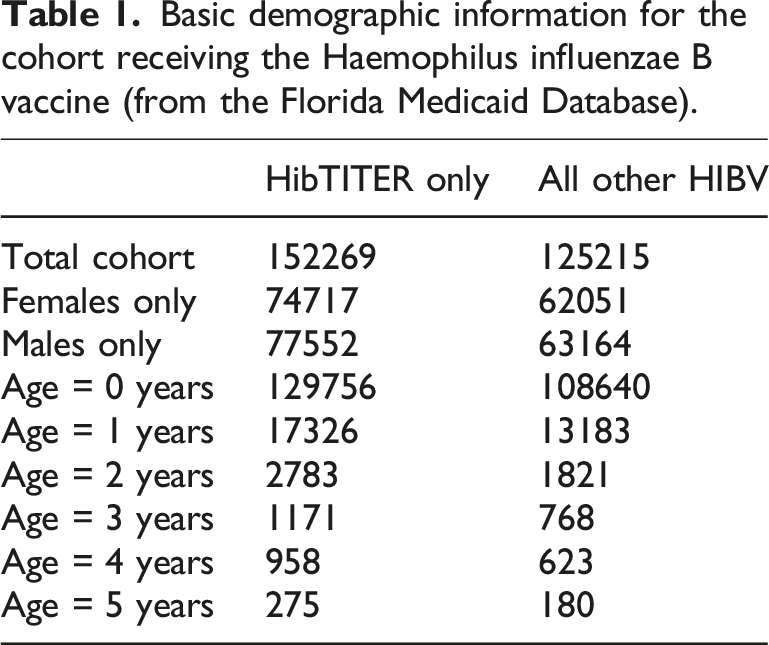
Estimated exposure
HibTITER® was the dominant HIB vaccine between 1991 and 1994, and it later shared roughly half of the market-share with other FDA approved vaccines. If we assume 3.5 million U.S. children born every year adhere to the U.S. government approved, institutionally endorsed, and pediatrician recommended schedule, we may assume the vaccine has been administered to roughly 35 million infants who are now between the ages of 16 and 33.
Statistical methods
In the medicaid analysis we limit our analysis to a post-Bonferroni correction
Results
Comparison of HibTITER® and “All other HIBV” groups with the cohort. VAERS corroboration
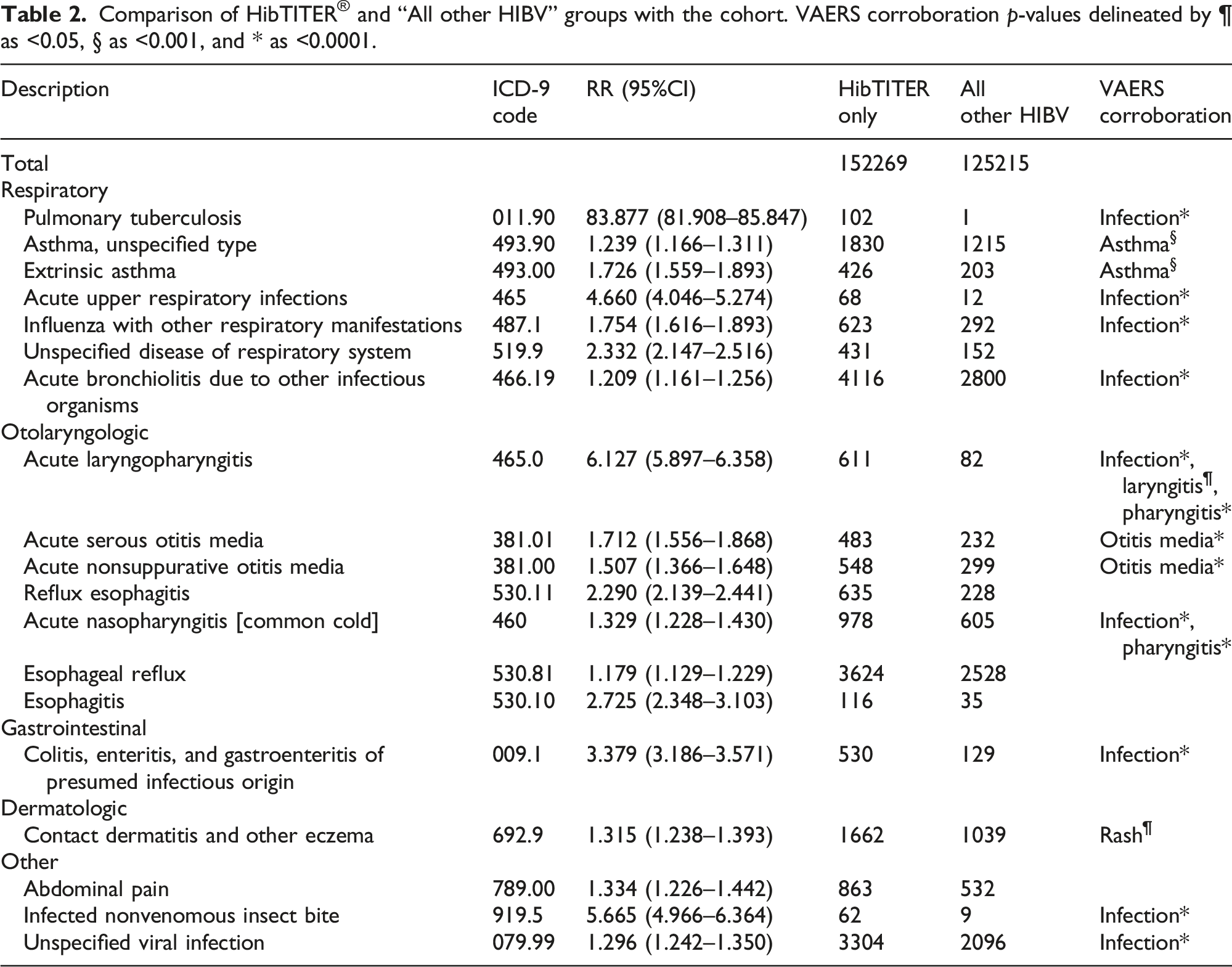
There are six other respiratory diseases that this study identifies as adverse outcomes within 30-days of HibTITER® vaccination: asthma, unspecified type (ICD-9: 493.90) with a relative risk of 1.239 (95% CI: 1.166–1.311); extrinsic asthma (ICD-9: 493.00) with a relative risk of 1.726 (95% CI: 1.559–1.893); acute upper respiratory infections (ICD-9: 465) with a relative risk of 4.660 (95% CI: 4.046–5.274); influenza with other respiratory manifestations (ICD-9: 487.1) with a relative risk of 1.754 (95% CI: 1.616–1.893); unspecified disease of respiratory system (ICD-9: 519.9) with a relative risk of 2.332 (95% CI: 2.147–2.516); and acute bronchiolitis due to other infectious organisms (ICD-9: 466.19) with a relative risk of 1.209 (95% CI: 1.161–1.256). All respiratory diseases can be corroborated in VAERS as infection (
There are seven otolaryngologic diseases classified as adverse outcomes, with the most prominent being acute laryngopharyngitis (ICD-9: 465.0) where 88.17% of all patients diagnosed within 30-days of their first HIB vaccine were HibTITER® recipients yielding a relative risk of 6.127 (95% CI: 5.897–6.358). The remaining otolaryngologic diseases include: acute serous otitis media (ICD-9: 381.01) with a relative risk of 1.712 (95% CI: 1.556–1.868); acute nonsuppurative otitis media (ICD-9: 381.00) with a relative risk of 1.507 (95% CI: 1.366–1.648); reflux esophagitis (ICD-9: 530.11) with a relative risk of 2.290 (95% CI: 2.139–2.441); acute nasopharyngitis [common cold] (ICD-9: 460) with a relative risk of 1.329 (95% CI: 1.228–1.430); esophageal reflux (ICD-9: 530.81) with a relative risk of 1.179 (95% CI: 1.129–1.229); and esophagitis (ICD-9: 530.10) with a relative risk of 2.725 (95% CI: 2.348–3.103). VAERS corroboration occurs with infection (
Additional discovered adverse outcomes include: the gastrointestinal disease of colitis, enteritis, and gastroenteritis of presumed infectious origin (ICD-9: 009.1) with a relative risk of 3.379 (95% CI: 3.186–3.571); and the dermatologic disease of contact dermatitis and other eczema (ICD-9: 692.9) with a relative risk of 1.315 (95% CI: 1.238–1.393). Other diseases include: abdominal pain (ICD-9: 789.00) with a relative risk of 1.334 (95% CI: 1.226–1.442); infected nonvenomous insect bite (ICD-9: 919.5) with a relative risk of 5.665 (95% CI: 4.966–6.364); and unspecified viral infection (ICD-9: 079.99) with a relative risk of 1.296 (95% CI: 1.242–1.350). VAERS corroboration occurs with infection (
Of the 19 disease diagnoses, spanning from mild to life threatening, identified as statistically significant to the utmost scientific standard (
Discussion
Literature review
The published studies on the safety of HibTITER® are numerous, but largely underpowered to detect the associations reported in this study. These include safety comparisons: of HibTITER® in combination with other vaccines (
Disease
The scope of the nine infectious diseases and diseases of infection from the 19 identified adverse outcomes suggests significant and rapid immunological defense impairment. The impairment spans into the respiratory system (with pulmonary tuberculosis, upper respiratory infection, influenza, and bronchiolitis), into the otolaryngologic system (with laryngopharyngitis and nasopharyngitis), into the gastrointestinal system (with colitis, enteritis, and gastroenteritis), and into the dermatological insult (with infected insect bite). Other exhibits of the immune system dysregulated are signals for asthma and dermatological reactions. Extrinsic asthma implicates an overreaction by the immune system to harmless environmental substances. Dermatitis and eczema implicates the immune system misidentifying harmless environmental substances and mounting a response.
Interpretation
The interpretation of the results in the context of cause and effect is best viewed as indirect. The HibTITER® vaccine does not directly cause a condition to manifest (e.g., infection), but may create or co-create a condition (e.g., suppressed immune system) for which the condition manifests. Additionally, receiving a diagnosis is inherently biased as sicker persons: are more likely to receive medical care; will receive medical care at a higher frequency; will have greater health documentation; and will have more diagnosis codes. Two different interpretations are entertained by the authors. One interpretation is that HibTITER® causes the conditions for a suppressed immune response (consistent with Adabor’s findings, 19 “the immune response may be compromised in repeated vaccination … compared with those with lesser vaccination” where the HIB vaccine is most often first given at 2 months of age along with the other CDC recommended vaccinations of hepatitis B, rotavirus, diphtheria, tetanus, acellular pertussis, pneumococcal conjugate, and inactivated poliovirus. Additionally, Cowling et al. 20 noted “Receipt of [a vaccine] could increase [a virus] immunity at the expense of reduced immunity to [other] viruses”) leading to a greater probability or severity of disease. Another interpretation, not mutually exclusive, is that HibTITER® causes the conditions for more interactions with medical care providers, and thus the greater documentation of disease.
Population perspective
The constellation of adverse outcomes associated with the HibTITER® vaccine in the Medicaid database assemble a population profile with: a compromised immune system allowing bacterial and viral parasites to infect the host (tuberculosis, influenza, bronchiolitis, enteritis, gastroenteritis, infected insect bites, and other unspecified infections); a poorly adapted overreacting immune system causing extrinsic asthma via the inappropriate release of antibody immunoglobulin E; dispersed inflammation likely exacerbated by an overtaxed lymphatic system (colitis, dermatitis, esophagitis, nasopharyngitis, laryngopharyngitis); respiratory complications; otitis media; other otolaryngologic complications; dermatologic complications; ophthalmologic complications; and other diseases of wide-spread organ systems. The mild nature of adverse reactions listed in the package insert of HibTITER® (apnea; appetite loss; convulsions; prolonged crying; diarrhea; erythema; fever; hives; irritability; rash; local reactions; redness; restless sleep; sleepiness; swelling; tenderness; vomiting; warmth; 4 cases of GBS; and 1 seizure) 21 stand in stark contrast to the severity of conditions described in this study.
VAERS corroborates many of the conditions highlighted by this study. VAERS succeeds in its singular purpose of “detecting unusual or unexpected patterns of adverse event reporting that might indicate a possible safety problem with a vaccine.” HibTITER® was not removed from the market based on safety concerns, but “discontinued...following a reassessment of [the manufacturer’s] portfolio and priorities.” 22
Conclusion
This study set out to discover adverse outcomes of thimerosal-free HibTITER® within 30-days of vaccination. Serious or life-threatening conditions were associated with HibTITER®. The HibTITER® published side-effects do not include the severe or life-threatening conditions, nor most of the significant findings of this study. We have corroborated many of our findings with VAERS data, and demonstrate successful detection of patterns with implications in vaccine safety.
In developing the framework necessary to conduct this study, the authors created a template for a passive surveillance protocol utilizing secondary-use health information data. The addition of a health information exchange layer to this template would constitute a low-budget, low-maintenance, population-wide, concurrent vaccine safety vigilance system.
This study is not without limitations. VAERS is considered a passive surveillance system with voluntary reporting only. VAERS data accounts for less than 1% of all vaccine adverse events. 23 In addition, the Florida Medicaid Database accounts for only vaccinations given within the Medicaid system. This does not include free vaccinations distributed by the CDC’s Vaccines for Children program. There may exist a selection bias on who receives which HIB vaccine. The authors went to great lengths to seek out selection bias based on racial/ethnic distributions and wealth disparity, and found none. Still, it remains an assumption that the clinician’s selection of which HIB vaccine to administer is independent of the particular patient as administration guidelines do not differ significantly among HIB vaccines.
Supplemental Material
Supplemental Material - Adverse outcome detection of childhood administered thimerosal-free HibTITER®
Supplemental Material for Adverse outcome detection of childhood administered thimerosal-free HibTITER® by Karl Jablonowski and Brian Hooker in International Journal of Risk & Safety in Medicine.
Footnotes
Acknowledgements
The authors would like to express their gratitude to Lauren Mulvey for sharing her expertise in lymphology, inflammation, and detoxification in the context of this study’s findings. We would also like to recognize Dr. J. Jay Couey for his conversations and contributions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was made possible by the Katherine G. Weisman fellowship at Children’s Health Defense.
Data availability statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
