Abstract
Introduction
Post-acute COVID-19 vaccination syndrome (PACVS) emerges as a syndrome of persistent symptoms of a multisystemic nature, even in previously healthy people. Its pathophysiology involves a neural phase characterized by the neuroimmune reflex and persistent secondary neurogenic inflammation. Frequent manifestations such as dysautonomia, neurological alterations, and musculoskeletal symptoms have been described.
Objective
To review the current evidence on PACVS and explore the therapeutic potential of Neuraltherapeutic Medicine (NTM), illustrated with a clinical case.
Material and methods
A targeted literature review search was conducted in MEDLINE (up to June 2024) using the terms “post-COVID-19 vaccination syndrome”, “covid vaccine adverse effects”, “inflammatory reflex” and “neural therapy”. In addition, a clinical case of a 75-year-old patient with persistent musculoskeletal pain post-vaccination, treated with NTM, was documented.
Results
The pathophysiology of PACVS includes neuroimmune mechanisms such as response, neurogenic inflammation, and autonomic dysfunction. NTM has shown the ability to modulate the nervous system by desensitizing sources of irritation. In the case presented, the application of 0.5% procaine to the vaccination site and contralateral reflex zone resulted in complete resolution of pain within 48 h and sustained functional improvement during the three months of follow-up.
Conclusions
NTM could represent a therapeutic option in the management of PACVS and other syndromes involving neurogenic inflammation. Controlled studies are required to validate its efficacy and establish standardized protocols.
Keywords
Introduction
The COVID-19 pandemic, caused by SARS-CoV-2, triggered a global health emergency, with more than 777 million cases and 7.1 million deaths reported as of March 2025 (World Health Organization, 2025). Vaccines, especially messenger RNA and viral vector vaccines, demonstrated remarkable efficacy, preventing approximately 14.4 million deaths in 2021 and reducing hospitalizations (89.1%), ICU admissions (97.4%), and mortality (99%) (Scholkmann & May, 2023; Zheng et al., 2022).
Serious adverse effects, according to WHO, are rare, with an incidence of 0.0045 per 10,000 doses, while transient effects (local pain, myalgia, low-grade fever, and headache) subside in more than 95% of cases within 48 h (Centers for Disease Control and Prevention, 2024). Although most musculoskeletal reactions following COVID-19 vaccination are mild and self-limiting, higher-intensity events, such as bursitis, capsulitis, and tendonitis, have been reported, with odds ratios between 1.4 and 3.7 compared to unvaccinated people (Park et al., 2023).
In addition, post-acute COVID-19 vaccination syndrome (PACVS) has been described as a chronic clinical entity associated with immunization against SARS-CoV-2 (Mundorf et al., 2024), characterized by persistent multisystemic symptoms, including chronic fatigue, autonomic dysfunction, neurological manifestations, and cardiovascular alterations (Mundorf et al., 2024; Platschek & Boege, 2024). Although its estimated prevalence is low (∼0.02%), the increasing documentation of cases and the absence of official recognition represent an emerging problem that requires greater clinical attention and specific research (Platschek & Boege, 2024).
Although clinical trials are essential to evaluate the safety and efficacy of vaccines, their controlled design may limit the generalizability of results to diverse epidemiological contexts. Growing evidence has emerged on rare adverse events associated primarily with messenger RNA-based vaccines.
From the perspective of Neuraltherapeutic Medicine (NTM), Bustamante et al. explore the pathophysiological bases that support the integration of the human being in a unified response to injury, called a non-specific response. This response comprises the coordinated interaction of cellular and humoral components of inflammation and highlights the inflammatory reflex as a phenomenon regulated by the nervous system (NS). This reflex is triggered when the NS detects the injury and modulates, through a reflex arc, the inflammatory expression. In this context, local anesthetics emerge as neuromodulatory tools of the neurogenic inflammatory process (Bustamante et al., 2024).
PACVS shares similar pathophysiological mechanisms and clinical manifestations with other functional syndromes, such as chronic fatigue/myalgic encephalomyelitis syndrome (ME/CFS) and post-acute COVID-19 syndrome (PACS). These entities are characterized by a nonlinear clinical evolution, mediated by dysfunction of the central and peripheral nervous system, peripheral and central sensitization (corticalization), dysautonomia, metabolic dyshomeostasis, neurogenic and systemic inflammation, and alterations in the extended autonomic nervous system (psychoneuroimmunoendocrine). These alterations generate a complex adaptive response to the initial injury, perpetuating a state of multisystemic hyperreactivity that explains the persistent and fluctuating symptoms in these patients (Bustamante et al., 2024).
The remarkable overlapping of these syndromes suggests the existence of common pathways, which may lead to therapeutic strategies based on the modulation of the extended autonomic nervous system, the control of neurogenic inflammation, and the restoration of homeostasis.
Case Presentation
Patient's Life Medical History
The patient, a 75-year-old female of low socioeconomic status, has a history of numerous chronic symptoms such as insomnia, headache, vertigo, otalgia, odynophagia, dysphonia, generalized musculoskeletal pain, and genitourinary symptoms. These conditions were successfully treated by NTM between 2016 and February 2020, through 17 interventions with 0.5% procaine from a segmental and meta-segmental therapeutic perspective, achieving complete remission of symptoms.
Consultation
In January 2021, the patient consulted the Neuraltherapeutic Medicine service again, reporting that on May 6, 2020, the day after receiving the second dose of the Sinovac vaccine, she began to present the following musculoskeletal symptoms, characterized by:
Pain in the right shoulder with radiation to the cervical region (visual analogue scale [VAS] 7/10) Severe low back pain with radiation to the lower limbs (VAS 6/10)
In addition, physical examination evidenced:
Skin hypersensitivity in C4-C5 dermatomes Presence of subcutaneous nodules on the right arm Palpable fascial retraction in the deltoid region (vaccination site) Limitation of active mobility in the right shoulder (60° abduction)
Beyond these localized findings, a comprehensive musculoskeletal and neurological examination was conducted. Additional investigations such as inflammatory markers (CRP, ESR, ferritin), autoantibody screening (ANA), coagulation profile (D-dimer, fibrinogen), and a metabolic/hepatic panel (transaminases, LDH), were ordered but not completed by the patient due to limited access. Imaging studies, including MRI and CT scans, were not requested as they were not deemed essential for the initial clinical assessment.
Diagnosis, Treatment and Follow-Up
Given the clinical findings and the temporal relationship with vaccination, the case was interpreted as a probable vaccine-related neural irritation. The therapeutic intervention, initiated at the first consultation in January 2021, infiltration was performed at the application site (right shoulder) with 0.5% procaine, complemented with intervention in the contralateral reflex zone (left shoulder). The procedure was well tolerated and no adverse effects were observed, and no additional pharmacological treatment was prescribed.
In the follow-up carried out on February 1, 2022, the patient reported complete resolution of pain in both the shoulder and lumbar region (VAS 0/10), with restoration of full shoulder mobility and normal functional activity. On examination, segmental alterations previously observed (hypersensitivity in C4–C5 dermatomes, fascial retraction, and palpable subcutaneous nodules) were no longer present.
At the three-month control, the patient remained asymptomatic, with no recurrence of musculoskeletal or neurological findings. She also reported improvement in sleep quality and daily activities, consistent with a systemic recovery beyond the musculoskeletal domain.
Discussion
Temporal Relationship and Neuroimmune Mechanisms in Post-Vaccine Exacerbation
This paper illustrates a clinical case of chronic musculoskeletal symptoms, with persistence of six months, characterized by musculoskeletal pain, with a temporal association of 24 h after the application of the COVID-19 vaccine (Sinovac). The patient, with a history of multiple chronic symptoms linked to sensitization of the NS, had achieved complete remission through interventions with NTM.
The reactivation of musculoskeletal symptoms in temporally associated with the vaccine raises the hypothesis that the stimulus generated acted as a focus of neuroimmune activation of the extended autonomic system, triggering persistent neurogenic sensitization mechanisms for 6 months.
The complete resolution of symptoms after the application of 0.5% procaine at the vaccination site and in contralateral reflex areas supports the hypothesis that the application of the COVID-19 vaccine generated a pathological neuroimmune trigger point. This explanatory hypothesis would be in accordance with the principles of A.D. Speransky's “second blow” theory, according to which successive stimuli can induce amplified neuroimmune responses (Pinilla-Bonilla et al., 2024). This irritation behaved as a center or focus of pathological dominance (Konradi, 1960; Zueva & Zuev, 2015), whose desensitization through a single neuraltherapeutic stimulus was sufficient to completely reverse the symptoms, maintaining the therapeutic effect over time.
Although a direct causal association between the components of vaccination and certain adverse effects has not been established, the appearance of post-vaccination neuroinflammatory disorders has been observed. These are characterized by the presence of symptoms like musculoskeletal pain, cutaneous hypersensitivity, among others, without structural findings identifiable by conventional studies, which raises the hypothesis of a possible involvement of nonspecific neuroimmunological mechanisms in their pathophysiology (Li et al., 2022).
Reported Adverse Events Associated with COVID-19 Vaccination
In an observational cohort study using data from the Global COVID Vaccine Safety (GCoVS Project), 13 adverse events were assessed within 42 days after vaccination. The study evaluated neurological, hematological, and cardiovascular conditions after vaccination, such as transverse myelitis, Guillain-Barré, cerebral venous thrombosis, and myocarditis. Although it did not reach the threshold for priority safety signals, continuous monitoring is recommended (Mundorf et al., 2024).
In a series of four cases with neurological manifestations—such as weakness, paresthesias, hyperreflexia, and gait disturbances— anti-angiotensin II antibodies were detected in the absence of other immunological markers (Bellucci et al., 2024). Likewise, a study in Vienna reported significant cognitive deficits in 29 patients with PACS or PACVS, especially in attentional functions, and a possible increase in the incidence of Alzheimer's disease in this population has been suggested (Roh et al., 2024).
Autoimmune phenomena have been described after vaccination, such as immune thrombocytopenic purpura, autoimmune liver diseases, Guillain-Barré syndrome, IgA nephropathy, rheumatoid arthritis, and systemic lupus erythematosus (Chen et al., 2022). Other authors have reported thrombocytopenia, myocarditis, allergic reactions, autoimmune hepatitis, facial paralysis, functional neurological disorders, and lymphadenopathy (Gopalaswamy et al., 2024; Li et al., 2022; Padilla-Flores et al., 2024). Surveillance of these events is key to supporting informed clinical decisions.
Although a definitive causal relationship has not been demonstrated, several reports suggest a possible association between COVID-19 vaccination and the appearance of autoimmune syndromes. In this context, the adjuvant-induced autoimmune/inflammatory syndrome (ASIA), proposed by, (Shoenfeld & Agmon-Levin, 2011) has been considered a useful theoretical framework to understand certain post-vaccination immunological phenomena. Recent studies (Jara et al., 2022) have documented cases compatible with this entity after the administration of vaccines against SARS-CoV-2, which underscores the need to continue investigating the possible immunopathogenic mechanisms involved.
Assessing causality in adverse events following vaccination requires epidemiological studies and robust statistical analyses that minimize bias and chance. Individual reports are insufficient to establish causality, which reinforces the need for systematic reviews with a higher level of evidence.
Post-Acute Vaccination Syndrome COVID-19 (PACVS)
Although most side effects following COVID-19 vaccination are transient, a minority of cases symptoms may persist, making it difficult to distinguish between vaccine reactions and reactivation of underlying diseases. An emergent syndrome, known as PACVS, with an estimated prevalence of approximately 0.02%, characterized by persistent symptoms for weeks or months, is recognized (Mundorf et al., 2024; Scholkmann & May, 2023).
General symptoms of PACVS (Table 1) were initially identified through patient reports of persistent, unexplained complaints following COVID-19 vaccination—symptoms that resembled but remained clinically distinct from Long COVID. Subsequent clinical case series supported these observations. Schieffer and Schieffer described more than 350 SARS-CoV-2–negative individuals presenting with chest pain, dyspnoea, fatigue, insomnia, paraesthesia, neuropathy, dermatological manifestations, and gastrointestinal complaints (Yong et al., 2025).
Comparative Table of Symptoms by Study, Taken and Modified from (Yong et al., 2025).

Population-based surveys indicate that prolonged post-vaccination symptoms are relatively uncommon. A Czech cohort reported symptoms lasting longer than one month in 1.3% of healthcare workers, and German surveys in children and adolescents showed similarly low prevalence rates.
Observational cohorts—including the Yale LISTEN study and the dataset described by Mundorf et al.—consistently document multisystem involvement. The most frequently reported symptoms include fatigue, exercise intolerance, paraesthesia, neuropathy, cognitive impairment, tachycardia, palpitations, dyspnoea, anxiety, sleep disturbances, and musculoskeletal pain. Identified symptom clusters comprise chronic fatigue, peripheral nerve dysfunction, cardiovascular involvement, cognitive impairment, psychomotor dysfunction, and sleep dysregulation (Yong et al., 2025).
Although most available studies rely on self-selected populations and lack control groups, the current evidence supports a heterogeneous yet recognizable pattern of manifestations that warrants further scientific investigation.
Long-term sequelae of severe adverse events (SAEs) following COVID-19 vaccination—such as myocarditis, vaccine-induced immune thrombotic thrombocytopenia (VITT), and immune thrombocytopaenia—are rare. However, follow-up studies demonstrate that a subset of affected individuals develop persistent symptoms consistent with PACVS (Yong et al., 2025).
In post-vaccine myocarditis, 10–50% of patients continue to report chest pain, dyspnoea, palpitations, fatigue, or persistent abnormalities on cardiac imaging for several months. In children, symptoms may persist for weeks or months but generally improve over time.
Recovery from VITT is frequently incomplete. Only about one-third of patients achieve good functional recovery within 3–6 months; others experience ongoing disability, recurrent thrombosis or thrombocytopaenia, or long-term neurological sequelae. Qualitative reports describe significant impacts on daily functioning, mental health, and socioeconomic stability (Yong et al., 2025).
Although infrequent, these prolonged outcomes demonstrate that certain SAEs can lead to chronic, PACVS-like manifestations, underscoring the importance of long-term clinical monitoring and structured follow-up.
The pathophysiology of PACVS continues to be investigated, with recent findings revealing biomarker patterns. In a key study with 191 participants with chronic fatigue and post-vaccination dysautonomia (Mundorf et al., 2024), antibodies against angiotensin II type 1 receptors and alpha-2 adrenergic receptors, as well as elevated levels of interleukin-6 (Mundorf et al., 2024), were identified, suggesting an autoimmune mechanism. Subsequent research by these authors described elevation of interleukins 6 and 8, a decrease in free triiodothyronine, alteration in immunoglobulin G subclasses, iron storage dysfunction, and an increase in soluble neurofilament light chains, (Mundorf et al., 2024) suggesting an inflammatory and autoimmune compromise with neuroendocrine dysfunction in this syndrome.
Spikeopathy Hypothesis
The hypothesis proposes that the SARS-CoV-2 Spike protein — whether derived from natural infection or expressed following COVID-19 vaccination — may itself be pathogenic, with effects extending beyond the acute phase. Interference with the cholinergic anti-inflammatory pathway has been suggested as a key mechanism by which Spike-related signals precipitate neuroimmune dysregulation. The vagus-mediated cholinergic circuit, acting through acetylcholine stimulation of α7-containing nicotinic receptors on macrophages and microglia, normally suppresses proinflammatory cytokine release. Disruption of this pathway may therefore result in uncontrolled systemic and central inflammation, microglial activation, blood–brain barrier dysfunction, and subsequent neuronal injury. Spike protein-related effects — including direct receptor interaction, functional antagonism, receptor downregulation, or impairment of vagal signaling — could plausibly drive this cascade, contributing to the persistent neuroinflammatory phenotype observed in Post-Spike Syndrome (PSS) (Parry et al., 2023).
Several additional mechanisms may underlie this proposed pathology: widespread biodistribution of Spike protein or mRNA through lipid nanoparticles or viral vectors; endothelial injury secondary to Angiotensin-Converting Enzyme 2 (ACE2) binding and downregulation; activation of inflammatory and thrombotic cascades; oxidative stress with mitochondrial dysfunction; translocation of the S1 subunit across the blood–brain barrier; prion-like aggregation of neuronal proteins; and autoimmunity triggered by ectopic antigen expression (Parry et al., 2023).
Clinically, these mechanisms have been associated with a broad spectrum of multisystem manifestations collectively described as PSS, which shares several features with PACVS and other dysautonomic conditions. From the perspective of nonspecific neuroimmunological mechanisms, the stimuli mediated by the Spike protein could be directly implicated in the disruption of this pathway.
Overlap with Other Dysautonomic Symptoms
PACVS has a remarkable similarity in clinical characteristics with other post-immunization syndromes, such as those described after the human papillomavirus vaccine. In a similar way, it happens with other dysautonomic syndromes such as PACS, ME/CFS, postural orthostatic tachycardia syndrome, mast cell activation syndrome, and small fiber sympathetic neuropathy (Mundorf et al., 2024; Ndeupen et al., 2021; World Health Organization, 2025).
An overlap between the complications of PACS and PACVS has been observed, with a possible increase in the incidence of neurological diseases, particularly in the peripheral nervous system. These include Guillain-Barré syndrome (8.1 cases per million vaccinated people), Parsonage-Turner syndrome—characterized by pain and weakness in the shoulders—small fiber neuropathy and Bell's facial palsy (Khatami et al., 2025; Zheng et al., 2022).
ME/CFS, PACS, and PACVS share proposed pathophysiological mechanisms and clinical symptoms. These could be related to phenomena such as viral persistence, immune dysregulation, autoimmune diseases, and hormonal imbalances (Calvacanti et al., 2024; Scholkmann & May, 2023).
Neuroimmune Reflex
Irritative phenomena of the NS, which can be generated by stimuli such as vaccination SARS-CoV-2, trigger complex reflex responses that involve the interaction between the nervous system and the immune system. This mechanism, called the inflammatory reflex by Tracey, is characterized by the involvement of the NS, which reacts within milliseconds or seconds in response to peripheral nociceptive or inflammatory stimuli (Centers for Disease Control and Prevention, 2024; Tracey, 2002).
This activation can trigger an exacerbated inflammatory response, known as a hyperinflammatory reflex, that triggers neurogenic inflammation, mediated by the release of proinflammatory neuropeptides and other neurotransmitters from peripheral nerve endings (Bustamante et al., 2024; Fischer et al., 2022; Park et al., 2023).
Neurogenic inflammation not only contributes to peripheral and central sensitization but also compromises the integrity of surrounding tissues, disrupting homeostasis. This phenomenon can evolve into neurogenic dystrophy, an entity characterized by trophic dysfunction of the affected tissue, decreasing the capacity for regeneration, alterations in cellular metabolism, and an inadequate response to new physiological or pathological stimuli (Bustamante et al., 2024).
In contrast to systemic and integrative approaches to the pathophysiology of PACS, Leitzke et al. have proposed a hypothesis focused on disruption of the cholinergic system, specifically at the level of nicotinic cholinergic neurotransmission, as one of the key mechanisms underlying this condition. This proposal has served as the basis for a clinical case report in which significant neurological improvement was documented after the administration of transdermal nicotine in low doses (Leitzke, 2023; Mundorf et al., 2024).
Although these preliminary findings open up a promising therapeutic pathway, the model based solely on nicotinic receptor function has limitations. This view oversimplifies the multifactorial complexity of the PACS and omits the dynamic interaction between the nervous, immune, and endocrine systems.
Mechanisms of Nonspecific Response to Injury: Integrative Role of the Extended Autonomic Nervous System
The NS irritation observed in patients with PACVS triggers a non-specific response to injury, comparable to secondary irritation induced by viral infection in the context of PACS. This model reflects the body's ability to react systemically through a neuroimmunoendocrine communication network, hierarchically coordinated by the nervous system.
In this framework, stimuli perceived as harmful activate rapid reflex pathways, on the order of milliseconds, along with slower endocrine responses that develop over the course of minutes to hours, integrated within the extended autonomic nervous system (Bustamante et al., 2024).
The endocrine response, which is activated in an approximate range of 10 to 30 min after exposure to the stressing stimulus, is part of the “general stress adaptation syndrome”, described by Hans Selye. This response is mainly mediated by the hypothalamic-pituitary-adrenal axis, with cortisol as the main humoral effector (Bustamante et al., 2024).
Bustamante et al. describe this phenomenon as a “neurological symphony”, alluding to a systemic, non-linear, and non-localized response, which involves different levels of the nervous system. This complex functional network acts in integration with other systems, operating as a unit in the face of disturbing stimuli. In this context, nociceptor activation can induce local irritation phenomena that, in addition to being expressed at the primary site, can propagate at a distance following patterns of metameric and/or metasegmental segmental irritation, evidencing dysfunctions in neuroanatomically linked terrains (Bustamante et al., 2024; Pinilla-Bonilla et al., 2024).
Clinically, this non-specific response generates manifestations depending on individual susceptibility and previous history of NS irritation. These alterations could explain the appearance of symptoms accompanied by tissue changes, manifested as sensorimotor and autonomic dysfunctions, even in the absence of objectifiable structural damage, attributable to the phenomenon of neurogenic inflammation (Bustamante et al., 2024; Engel et al., 2022).
Neuraltherapeutic Medicine
Neuraltherapeutic Medicine (NTM) is a medical system based on a systemic and complex conception of biological organisms. Its central focus is the modulation of the nervous system (NS) and its interaction with other regulatory systems, especially the neuroimmune axis. This model integrates historical contributions from the Russian Nervism school, as well as the clinical and therapeutic developments of the neural therapy school of the Huneke brothers.
With the incorporation of the principles of complexity sciences, MNT has undergone a significant evolution in Colombia, where it has generated a notable clinical and academic impact as a complex medical system.
The foundation of the Institute of Experimental Medicine and the Department of Physiology gave rise to the school of Nervism, headed by Ivan Pavlov. This trend was deepened for more than a century by several generations of scientists, including authors such as Speransky, Bykov, Orbeli and Vischnevsky. These authors explored the regulatory role of NS in the pathophysiology of pathological processes (Klimenko & Golikov, 2003) and introduced the therapeutic use of infiltrations with local anesthetics, known as “novocaine blocks”, with the purpose of modulating neurovegetative and neuroimmune responses. These techniques were applied in the management of various acute and chronic medical conditions, such as septic and hypovolemic shock, as well as in the treatment of inflammatory and infectious processes (Bustamante et al., 2024).
NTM uses local anesthetics, such as procaine, applied to irritated NS structures to modulate the nonspecific response to injury. This medical system focuses on modulating pathological processes mediated by neurogenic inflammation, consequent reduction of tissue oxidative stress, and finally, promoting the recovery of organ function. This phenomenon, currently recognized as a “neuraltherapeutic effect”, transcends the transient pharmacological action of local anesthetics by inducing a functional regulation of the extended autonomic nervous system (Bustamante et al., 2024).
The therapeutic use of local anesthetics has been well documented in the treatment of various pathologies. In a case report, Bustamante et al. described a neural stimulation approach using local anesthetics to modulate neurogenic inflammation manifested as persistent musculoskeletal pain following post–SARS-CoV-2 viral infection (Bustamante & Pinilla-Bonilla, 2025). Additionally, in a systematic scoping review, Vinyes et al. analyzed 129 studies, of which 37.98% corresponded to clinical trials, 55.03% to observational studies, and 6.97% to systematic reviews. The main criterion of indication was pain. The majority of the cases were chronic pain (72.86%), followed by acute pain (13.17%), with other non-pain-related conditions accounting for the smallest portion (13.95%). These findings highlight the safety profile and usefulness of local anesthetics in the management of acute and chronic pain, as well as in inflammatory conditions unrelated to pain (Vinyes et al., 2023).
This relevant review, unprecedented in this area of knowledge due to quantitative statistical analysis, highlights the usefulness of local anesthetics as a therapeutic tool in various diseases. However, its scope is limited, as it does not address the complexity of the neuromodulation mechanisms involved in the responses of living systems, according to the MNT's proposal.
Contralateral Reflex Irradiation
The phenomenon of the “mirror site” (Figure 1), that is, the appearance of clinical manifestations contralateral to a unilateral lesion, has been documented in various clinical and experimental contexts. Within the framework of NTM, this finding refers to the pioneering observations of Alexander Speransky, who described in his work “Bases for a New Theory in Medicine (1936)” how unilateral irritative stimuli in paired organs could induce symmetrical morphophysiological alterations on the opposite side. In a specific study, Speransky demonstrated that a unilateral injury to the sciatic nerve, induced by irritating or infectious substances, generated an inflammatory response both in the ipsilateral structures (paravertebral ganglia and nerve roots) and on the contralateral side, a finding confirmed by histopathological analysis (Speransky, 1954)
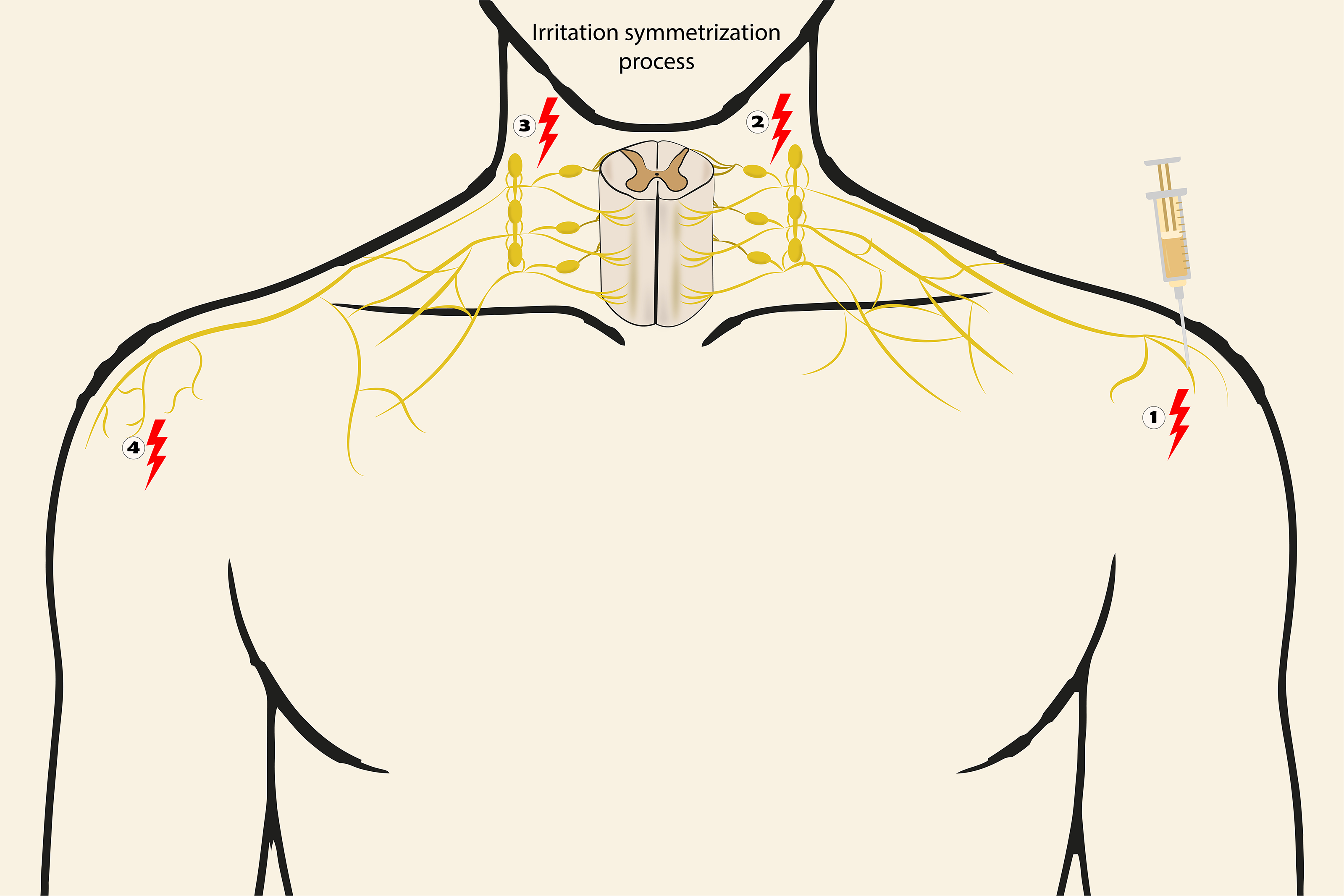
Irritation symmetrization process. As part of the sensitization processes following an initial irritation, a phenomenon may occur in which the original irritation becomes mirrored or symmetrized, leading to the appearance of a secondary focus of irritation in the contralateral anatomical site. This process is part of the segmental irradiation of irritation and follows four non-linear phases:
In the current scientific literature, several studies have shown that a nerve injury can trigger a cascade of functional, morphological, and molecular alterations in apparently undamaged contralateral structures of the nervous system. This phenomenon can affect different levels of the NS, both at the central and peripheral levels. In a cerebral and cerebellar context, this phenomenon has been recognized as crossed or transhemispheric diaschisis.
At the brain level, different findings have been reported at the electroencephalographic level and in blood flow. Juhász et al. (1997) conducted a study in patients recovering from a hemispheric stroke in whom they analyzed the EEG frequency and found a reduction in the alpha peak frequency in the contralateral hemisphere in the same territory of the infarction. Van Kaam et al. (2018) found similar results in patients with a unilateral ischemic lesion, finding an alteration in the delta/alpha EEG frequency and a decrease in functional connectivity in both hemispheres (Kamouchi et al., 2004; Sommer et al., 2016). demonstrated contralateral cerebellar hypoperfusion (crossed cerebellar diaschisis) in acute stages of cerebral infarction, associated with frontal, temporal, and thalamic hypoperfusion. These findings confirm what Andrews (1991) had already reviewed in 1991 about how the process of transhemispheric diaschisis could affect both blood flow and metabolism in the contralateral hemisphere after cerebral ischemia.
At lower levels of the nervous system, phenomena involving a contralateral alteration after nerve injury have also been described. Hasmatali et al. (2020) documented a contralateral increase in the expression of the transcription factor FOXO3 at the dorsal root ganglia (DRG) level. Likewise, Jancálek et al. (2010) have found contralateral increases in TNF-α and IL-10 in DRG after unilateral peripheral nerve injury. Hatashita et al. (2008) reported an increase in TNF-α in DRG-activated glial cells and astrocytes following injury to the contralateral dorsal horn of the spinal cord in an experimental rat model.
Similarly, in experimental animal models of unilateral sciatic nerve section, Wall and Devor (1981) observed a decrease in contralateral dorsal root evoked potentials. And also, Wells & Vaidya (Wells & Vaidya, 1989) reported bilateral morphological alterations, such as changes in somatic size and nuclear position.
In conclusion, after a peripheral nerve injury, a series of phenomena can occur that generate a systemic plastic response that includes changes not only at the unilateral and local level of the injury but also corresponding neurochemical, neurophysiological, synaptic, and morphological alterations that occur at multiple levels, such as the medulla, brain stem, thalamus, and even cerebral cortex (Navarro et al., 2007).
Ultimately, the available evidence underscores the neural system as the core of an extended communication network encompassing the nervous, immune, endocrine, and interstitial tissue systems and cells within the living organism, which sustains its functional unity. Experimental neurophysiology and clinical observations demonstrate that nervous system activity operates through systemic, nonlinear, and integrative mechanisms, which can explain non-local phenomena such as contralateral reflex irradiation and neurogenic inflammation. (Figure 2) NMT provides a practical framework to modulate these complex interactions, offering a coherent rationale for the observed therapeutic effects and highlighting the central regulatory role of the nervous system on the pathogenetic processes of organism.
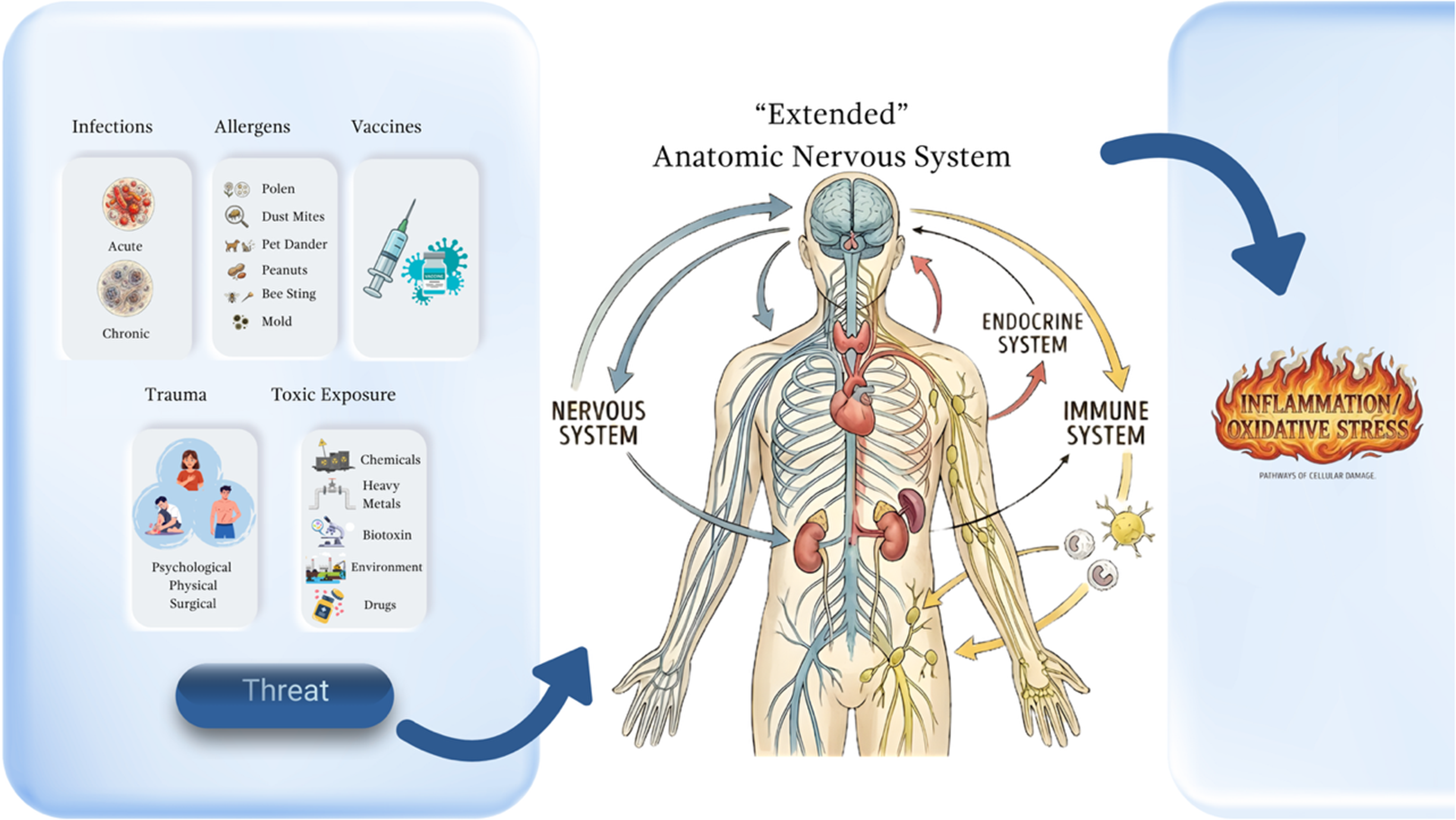
Nonspecific responses to threatening stimuli—such as trauma, microorganisms, toxins, vaccines, or allergens—activate the extended autonomic nervous system or the neuroimmunoendocrine system. This integrated network responds as a unit through multiple cellular pathways and chemical communication mechanisms associated with stress and inflammation. (Source: Own elaboration).
Conclusion
COVID-19 vaccines have demonstrated a favorable safety and efficacy profile. However, in a minority of patients, persistent symptoms of an inflammatory and dysautonomic nature, called PACVS, have been identified. This syndrome has been poorly documented, and its therapeutic approach is not yet clearly defined, which represents a considerable clinical challenge.
The findings are described from a single case of a patient who achieved resolution of the symptoms associated with CNS irritation by NTM and who subsequently presented a reactivation of musculoskeletal pain for six months, establishing a temporal relationship with vaccination. An immediate and sustained therapeutic response was observed after a new application of NTM, with complete remission documented at a three-month follow-up.
The neuraltherapeutic approach modulates irritation at different levels of the NS, modulating neuroinflammatory circuits. This approach aligns with the concept of the extended autonomic nervous system, or the nervous system unit, which is a psychoneuroimmunoendocrine network that regulates all global physiological dynamics. It highlights the capacity for peripheral interventions to have systemic effects. These findings support the exploration of NTM in post-vaccination syndromes and other neuroinflammatory-based clinical entities.
However, current evidence remains limited, and the present findings are preliminary, highlighting the need for validation through broader research models incorporating complexity science, along with long-term follow-up, before any clinical recommendations can be made.
Footnotes
Acknowledgements
To the living God, source of all light and wisdom, for allowing us to glimpse, though dimly, the sublime perfection of the human being as His creation. To my master, María Luisa Piraquive, whose life and example embody the noblest ideal of what we “ought to be” a constant reminder that truth, compassion, and excellence are inseparable.
Ethics Approval and Consent to Participate
This research was reviewed and approved by the institutional review board of the National University of Colombia and the Integrated Subnetwork of Southern Health Services, UHMES Tunal.
The participant's informed consent was obtained.
This study was approved by the research ethics committee of the Faculty of Medicine of the National University of Colombia and the ethics committee of the “El Tunal” Hospital. Colombian ethical principles were followed (Article 11 of Resolution 008430 of 1993 of the Ministry of Social Protection).
Consent to Publish
The participant's informed consent for publication was obtained.
Authors Contributions
CB: conceptualization (coworking); writing – original draft (lead); formal analysis (lead); writing – review and editing (lead)
LP: Conceptualization (coworking); formal analysis (supporting); Writing – review and editing (supporting).
MP : conceptualization (coworking).
ME: Writing – review and editing (supporting).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability and Materials
Data supporting the findings of this study are available from the corresponding author upon request.
