Abstract
Peripheral facial paralysis results in complete loss of contractibility in facial muscles, atrophy and asymmetry. Patients performed functional electrical stimulation (FES) home training twice daily over 1 year to show its effect and define stimulation parameters. A specific response was obtained in all 10 patients. Since the mean phase duration (PD) could be reduced (155.0 ± 55.0 to 51.0 ± 12.4 msec), this allowed the mean stimulation frequency to be increased (1.3 ± 0.5 to 5.0 Hz ± 1.5 Hz). The PD reduction to generate the same charge increased the voltage (16.2 ± 2.5 to 27.2 V ± 5.9). A suitable combination of PD and amplitude elicited a selective response without activation of other facial muscles using a PD of ≥ 25 ms (> 25 000 µs). FES changes the stimulation parameters for improving the training effect on denervated facial muscles. It prevents denervation atrophy and stops facial asymmetry.
Keywords
Introduction
Complete facial nerve paralysis, the most severe form of facial nerve palsies, is characterized by axonal damage and never recovers completely. This is an important difference compared to most cases of Belĺs Palsy (Urban et al., 2020), which are usually caused by a temporary conduction block and in most cases heal completely within a few weeks. In contrast, in complete facial nerve paralysis all motor axons are damaged and complete denervation of all facial muscles followed by progressive muscle atrophy occurs. If there is no spontaneous regeneration or regeneration by facial nerve reconstruction after this phase, the denervation and muscle atrophy is permanent (Zumbusch et al., 2024).
The complete denervation of the facial muscles results in a progressive facial asymmetry. The loss of the ability to move the face has both social and functional consequences for the patient (Steinhäuser et al. 2022). Functionally, patients present with an asymmetric smile, epiphora or dry eye, absence of blink, problems with speech articulation, drooling, and depending on the location of the lesion, hyperacusis and changes in taste. In social situations patients often have difficulty expressing facial emotions and suffer impairments in eating and speaking. This may lead to stress, anxiety and depression, and a reduction in further social engagement (Dobel et al., 2012; Strobelt et al., 2022).
None of the patients with axonal damage of the facial nerve can recover completely; even in instances where a spontaneous regrowth of the damaged axons and a reinnervation of the denervated facial muscles occurs. In such cases, there will always be some degree of aberrant, misguided reinnervation and therefore some synkinesis, hyperactivity of muscles, mass movements, and discomfort due to the uncoordinated muscle activation (Guntinas-Lichius et al., 2022). If no spontaneous reinnervation occurs, for many patients with cases of irreversible paralysis reinnervation procedures via surgery are the primary solution. In such cases, supportive treatment is nonetheless necessary to assist the patient until reinnervation occurs and to improve surgical outcome (Kurz et al., 2022). Patients affected with facial paralysis therefore require non-invasive approaches to complement surgical treatment options (Dobel et al., 2012). Especially as no single surgical technique can solve all of the patient's problems (Seeberger et al., 2024).
The use of functional electrical stimulation (FES) in the treatment of patients with facial paralysis has been investigated as a non-invasive approach for treatment (Arnold et al., 2023; Fargher & Coulson, 2017; Gittins et al., 1999; Puls et al., 2020; Volk et al., 2020). The first published studies indicate that FES is an appropriate intervention for the treatment of denervation atrophy of the facial muscles and indicates that FES enhances facial movements (Choi, 2016; Mäkelä et al., 2019; Tuncay et al., 2015). However, it is crucial to acknowledge the potential side effects of FES, such as the deterioration of facial nerve regeneration, and the difficulties with adherence to therapy due to resulting depression and a strong aversion towards treatment. These consequences can significantly influence the overall adherence to the therapy (Bradley, 1994; Diels, 2000). In contrast, outcomes indicate that FES has a positive or at least no negative effect on reducing the symptoms of facial nerve paralysis (Arnold et al., 2021; Gittins et al., 1999; Mäkelä et al., 2019; Tuncay et al., 2015) and that it bears no negative effect on the patient's reinnervation or synkinesis (Tuncay et al., 2015; Zealear et al., 2014). Therefore, FES treatment appears to be safe for cases of facial paralysis (Puls et al., 2020).
FES home-training has been used successfully in the treatment of spinal cord injury as it enables effective rebuilding of muscle structure, mass, and strength (Boncompagni et al., 2007; Kern et al., 2005; Kern & Carraro, 2020; Sajer et al., 2018). Furthermore, several studies have initiated the use of FES in patients with facial paralysis (Krauß et al., 2024; Mäkelä et al., 2019; Meincke et al., 2024; Puls et al., 2020). However, none of these studies have adapted the stimulation parameters to create an individualized FES training program adapted to the patient. The stimulation parameters used are dependent on the condition of the denervated muscles and the duration of the denervation. Long-time denervated muscles need electrical impulses with long phase durations (PDs). Several studies have shown that muscles (including facial muscles) show an increase in strength and function over time, such as increasing muscle diameter or improving muscle function (Brüggemann et al., 2017; Choi, 2016; Crameri et al., 2002; Kern & Carraro, 2020; Mäkelä et al., 2019), but also respond to a reduction in phase duration (PD) and increased stimulation frequency to evoke more muscle contractions during the same training time (Krauß et al., 2024; Meincke et al., 2024).
The present study aimed to establish a daily home-based FES protocol with the goal of providing a conservative therapy supplement for patients with complete peripheral facial paralysis. The main objective of this study was to develop a patient set-up by minimizing discomfort while improving facial functionality and symmetry.
Methods
Patients
The data presented were collected from patients presenting at the Department of Otorhinolaryngology, Jena University Hospital, within a longitudinal, open-label, prospective, single-center, proof of principle study. Data were collected from the 12th of June 2018 to the 13th of December 2022.
Ten adult patients diagnosed with unilateral complete paralysis of the ZYG by nEMG were included in this study. Six patients were male (60%) and 4 were female (40%). The median age of the patients at onset of facial paralysis was 60.3 years (range 22.4–76.6). The median age of the patients at baseline was 61.0 years (range 24.0–77.0).
The patients’ demographics and history of unilateral facial paralysis are shown in Table 1. Three patients dropped out: Patient 02 after 40 ± 2 weeks (due to personal reasons, Patient 03 after 12 ± 2 weeks due to a stroke, and Patient 06 after 12 ± 2 weeks to participate in rehabilitation after total hip arthroplasty. One patient was withdrawn during the course of the study after 8 ± 2 weeks when a lymph node metastasis was detected. The treatment plan was changed and instead of the planned hypoglossus-facial nerve jump nerve suture, the metastasis and the left facial nerve were surgically removed. The remaining six patients performed home training according to the protocol until strong signs of clinical and electrophysiological reinnervation occurred.
Patient Demographics.
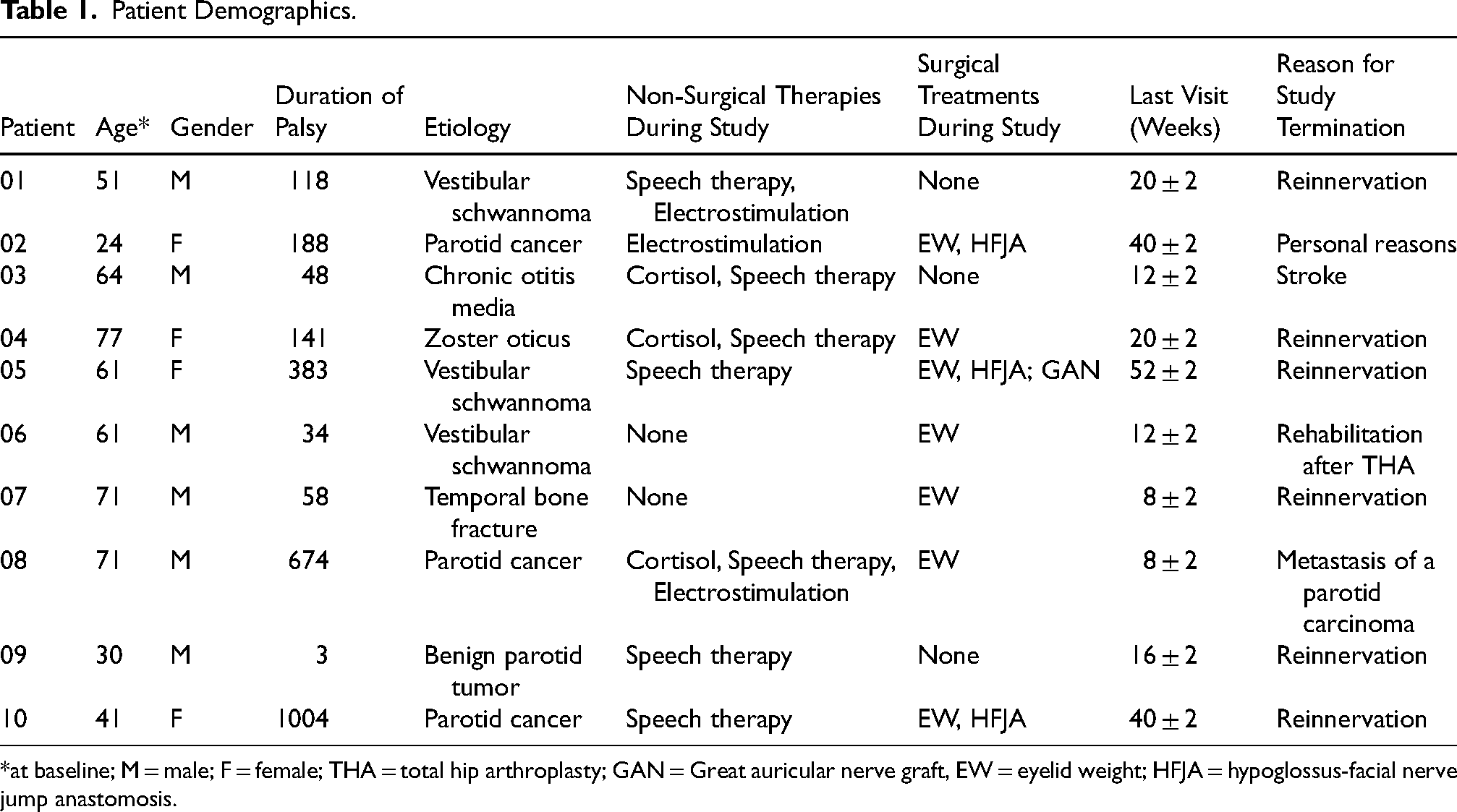
*at baseline; M = male; F = female; THA = total hip arthroplasty; GAN = Great auricular nerve graft, EW = eyelid weight; HFJA = hypoglossus-facial nerve jump anastomosis.
The etiology of paralysis for the patients included was vestibular schwannoma in three, parotid cancer in three, benign parotid tumor in one, chronic otitis media in one, temporal bone fracture in one and zoster oticus (Ramsey-Hunt syndrome) in one. Five patients were affected on the right side and 5 patients on the left side. A summary of the patients’ surgical and non-surgical treatments are shown in Table 1.
All patients gave written informed consent for the inclusion of their data in this study. Ethics committee approval was obtained (no. 5505-03/18) and the study was conducted in compliance with the Declaration of Helsinki and registered in the German Clinical Trials Registry (Deutsches Register Klinischer Studien; DRKS00015015).
Inclusion/Exclusion Criteria
Adult patients (≥18 years) were included in the study if they had unilateral complete peripheral facial muscle paralysis as determined via needle electromyography (nEMG) of the facial nerve. The frontalis muscle, the orbicularis oculi, the orbicularis oris, and zygomaticus muscle (ZYG) were tested. All muscles were tested to increase the sensitivity of needle EMG to detect any remaining innervation or any form of reinnervation. Patients were observed at monthly on-site follow-up visits for signs of reinnervation and were treated and observed continuously until strong signs of clinical and electrophysiological reinnervation occurred to evaluate the optimal stimulation parameters. The two previous published studies, based on the same patient collective, had stronger criteria to exclude later follow up time points with first signs of reinnervation to avoid any bias on facial muscle diameter or optical parameters of the faces (Krauß et al., 2024; Meincke et al., 2024). The maximum study period per patient was 52 ± 2 weeks. Patients were also excluded if they had undergone any conservative treatment, e.g., injection with botulinum toxin or physiotherapeutic treatment during the last 3 months; or had undergone any prior surgical intervention.
Visits
The monthly on-site visits for the patients were as follows: Baseline (week 0); 4 ± 1 week; 8 ± 2 weeks; 12 ± 2 weeks; 16 ± 2 weeks; 20 ± 2 weeks; 28 ± 2 weeks; 40 ± 2 weeks; and 52 ± 2 weeks.
Electromyography
Standardized nEMG was performed on the frontalis muscle, ZYG, orbicularis oculi and oris muscle using the VIASYSY Synergy (version 15.0. VIASYS Healthcare UK Ltd. Warwick, United Kingdom) as described by Geißler et al. and Guntinas-Lichius et al. (Geißler et al., 2016; Guntinas-Lichius et al., 2020). The nEMG was used to determine the denervation, synkinesis and reinnervation status of the muscles.
Innervation Status
The paralysis of the frontalis muscle, the orbicularis oculi, the orbicularis oris, and ZYG for each patient was assessed via nEMG from baseline to the end of the study period at each follow-up to determine if synkinetic reinnervation occurred. nEMG measurements were performed according to previous protocols (Grosheva et al., 2008; Guntinas-Lichius et al., 2020). A complete denervation of the facial muscles was declared when no volitional activity could be detected. To also detect aberrant reinnervation, for each muscle's measurement the patient was asked to perform volitional activations of all regions of the face. If not the agonistic, but any other facial expression triggered a clear electrophysiological response in nEMG it was classified as synkinetic reinnervaion (Guntinas-Lichius et al., 2020). nEMG measurements were saved electronically in their original format accompanied by a human-readable report.
Surface Electrode Placement for Testing and Home-Training
The electrodes were placed with the patient in an upright sitting position, two 6.4 × 4.0 cm oval surface electrodes (PALS® Neurostimulation electrodes, Axelgaard Manufacturing Co., Ltd., Lystrup, Denmark CE-certified, REF 896230)) were placed in correspondence with the paralyzed ZYG as close as possible to the corner of the mouth in order to prevent unspecific stimulation of other facial muscles and the cables of the electrodes were connected to the external stimulator (Figure 1).
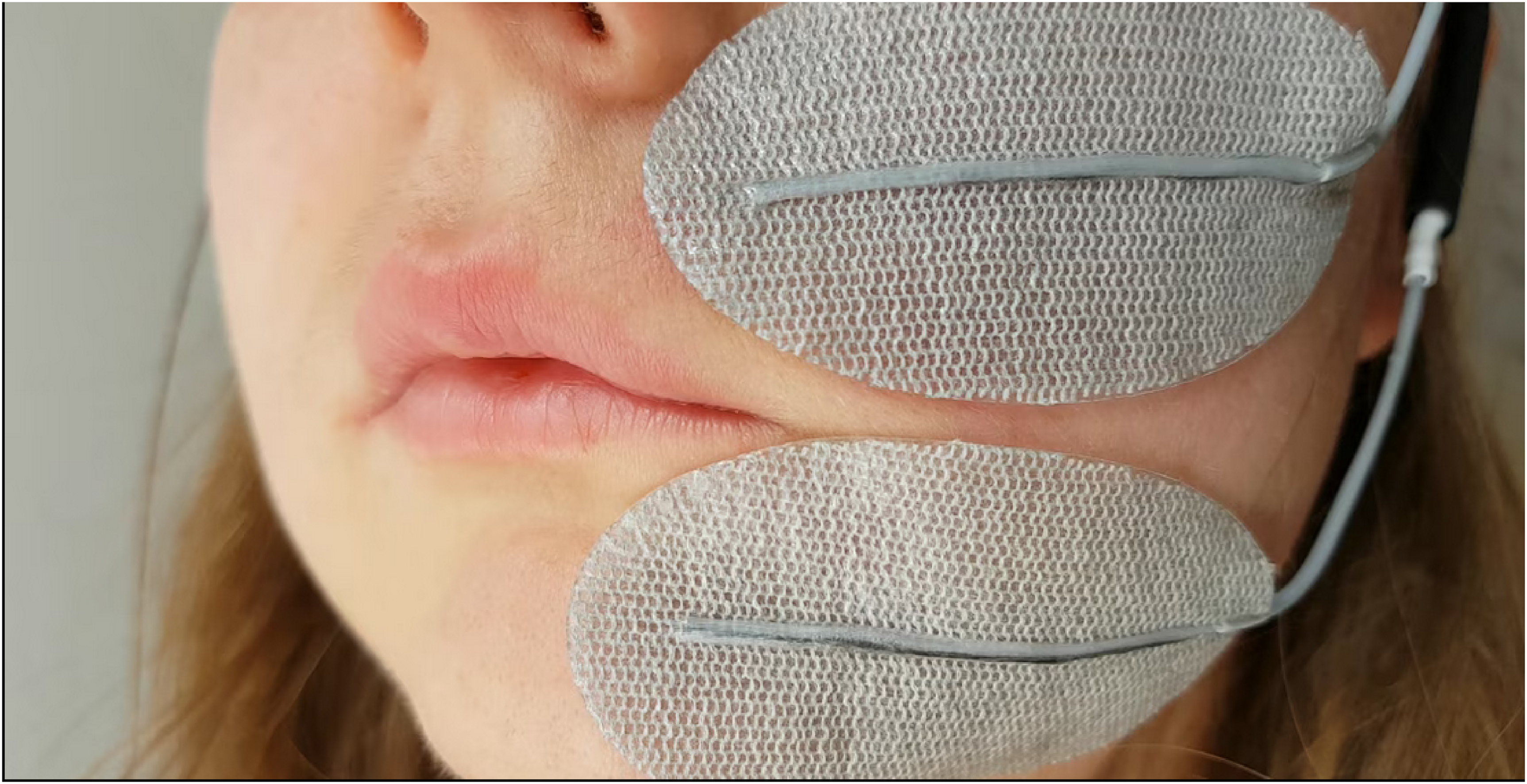
Female Patient with Complete Peripheral Facial Palsy on the Left Side, Denervation Since 7 Months, Performing FES Since 1 Month. FES with 50 ms Phase Duration, 14–18 V, 5 Hz, 2 s oN, 1 s OFF, Triangular Impulses – FES-Video Can be Seen at: https://vimeo.com/1070284595 [Volk, Gerd Fabian; FES on Female Patient; https://vimeo.com; Publication Date: 28th of March 2025; Last Call: 29th of March 2025].
I/t Curve Determination
Determination of the I/t curves was performed accordingly to Arnold et al. (2021). FES of the ZYG was regarded as effective only when it lifted and pulled the corner of the mouth on the same side, without causing activation of any other facial muscles on either side, such as the mentalis, depressor labii inferioris, depressor anguli oris, platysma, or muscles involved in chewing like the masseter. The I/t curve was created with the X-axis representing the PD assessed and the Y-axis indicating the corresponding threshold amplitudes. PD of 1, 2, 5, 10, 15, 25, 50, 100, 250, 500, 1000 ms were tested with increasing amplitudes between 0.1 and 20 mA. Biphasic triangular and rectangular waveforms presented as single pulses were used. The impact of facial paralysis duration, prior treatments, and its underlying cause were analyzed in relation to parameter selection and stimulation efficacy (Arnold et al., 2021).
For all test in the hospital we used electrical stimulators with mA as output. But for the hometraining, the STIWELL med4 devices were used, working with V instead of mA.
Unspecific Effects of Stimulation
In order to minimize unspecific motoric stimulation, the electrode placement and stimulation parameters were optimized to activate mainly the ZYG (Arnold et al., 2021).
Using two 6 × 4 cm electrodes in the face affects more than one muscle at a time. When we increased the amplitude, more and more muscles got stimulated and start contracting. But by carefully choosing the optimal location and just the right amount of electrical energy, mainly specific movements could be triggered while avoiding relevant co-contraction of muscles close by.
To allow the patients to find the same spots and settings at home, we took pictures and videos from the electrical stimulation while being in the hospital for the patients to take home and check when placing their electrodes themselves. Also a mirror was used at home to find the right position, but also the right amplitude for a specific “electrical triggered smile contraction.
Patients were withdrawn from the clinical investigation if such unspecific muscle activation compatible with clear signs of reinnervation, either normal or synkinetic, was observed.
A metallic taste on the tongue caused by unspecific stimulation of the lingual nerve and/or flash sensations by unspecific stimulation of the retina and/or the optical nerve are typical for long triangular impulses and are not an unexpected side effect or any reason for the withdrawal of a patient (Puls et al., 2020).
The parameters and the electrode position chosen during the first session at the hospital were confirmed or adjusted as need be at each of the subsequent sessions, in order to maintain an effective and selective ZYG response to the stimulation.
For home-training, biphasic stimulation was delivered in triangular waveform, with a burst duration of 5 s and a burst pause of 1 s The patient was instructed and trained to place the electrodes in front of a mirror in the same position depicted in the manual, to switch on the STIWELL med4 (STIWELL® med4 device (CE 0297; P/N 9001015; MED−EL Medical Electronics, Innsbruck, Austria), and to start the saved program. For all patients monthly study visits were planned, plus a baseline visit, and two remote study visits (after 2 and 6 weeks of home training) conducted via telephone. The stimulation was performed twice a day (morning and evening with a break in between of at least 6 h) for 20 min. The parameters chosen at baseline were adjusted at the subsequent follow-up visits, as need be in order to maintain a safe, specific, and effective ZYG stimulation.
Program for Home-Training
FES was performed with an approved electrostimulation device, the STIWELL med4. The individual shortest PD without causing a selective activation of the ZYG and without uncomfortable sensation was used to program the device. The amplitude at which a visible contraction of the ZYG could be painless elicited without the simultaneous unspecific reaction of other facial muscles was determined for each evaluated PD, unless the discomfort threshold or an amplitude causing unspecific reactions of other facial muscles was reached before. Because the STIWELL device is voltage controlled when using long pulse stimulation programs, the amplitude is determined in V instead of mA as used when the I/t-curve was determined. At this stage, a 20-min stimulation session was conducted on the patient using the device to verify the efficacy of the selected combination. The chosen PD was saved on a close map on the STIWELL med4. The map could not be changed by the patient, who could only increase or decrease the voltage of about 2–4 V. The program became available to the user when the device was switched on.
For home training we only used triangular waveforms. But for all test in the hospital like I/t-curves, we used rectangular and triangular waveforms to be able to compare the differences for the planed scientific evaluation. While the 4 weeks of twice daily home training with triangular impulses had a training effect on the muscles while being well tolerated, the I/t-cuves tests once every 4 month used rectangular and triangular waveformes and phase durations of a wide spectrum with the intention to quantify the electrophysiological changes.
Data Analysis
Data were analyzed using IBM SPSS statistics software (Version 28; IBM, New York) for medical statistics. Descriptive statistics were used to report demographic data (e.g., age, gender). Distribution of continuous data was described using mean values with standard deviation. Qualitative data are presented as absolute and relative frequencies. The Friedman test was used to determine differences in PD and frequency over time.
Results
Patients
All patients performed home training according to the protocol until drop-out or exclusion (Table 1). The patients did not report any severe side effects of the FES on the facial muscles over the treatment period, nor were any observed by the clinicians. One patient experienced minor skin irritation localized to the skin surface in contact with the adhesive of the electrodes, and one patient reported an “unpleasant feeling”.
Response to Triangular vs. Rectangular Wave Form Stimulation
Triangular Wave Stimulation
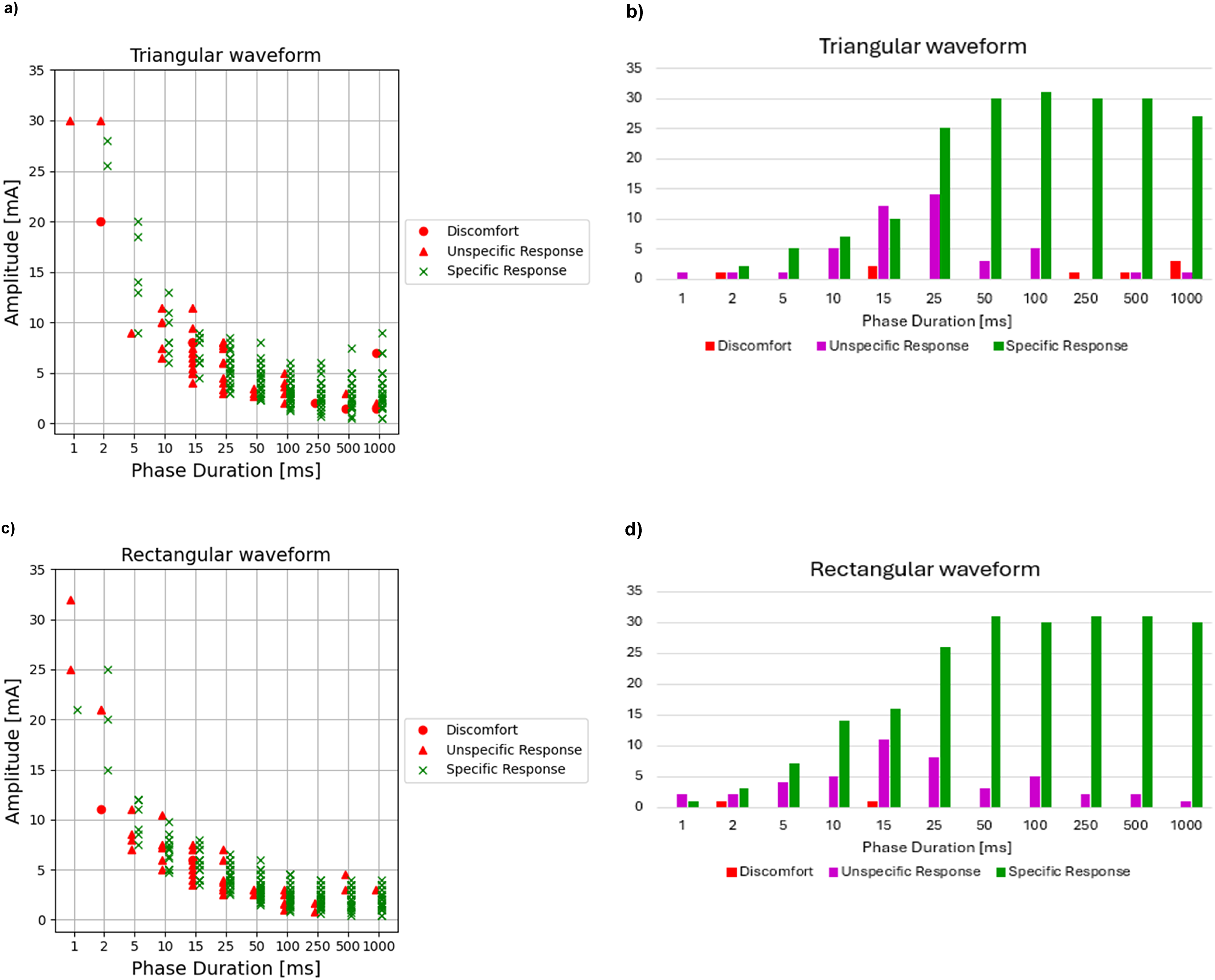
A specific response to the stimulation without unspecific activation of other facial muscles and/or pain/discomfort for the patients was observed in all the patients with a PD ≥ 25 ms. With shorter PD unspecific response of the lips, chin, masseter and sometimes eyes could be observed. In these cases, it was impossible to select a parameter combination eliciting a selective ZYG stimulation only. Figure 2(a) shows the amplitude per patient for the PDs using triangular wave stimulation. The occurrence of discomfort, unspecific responses, and specific response using triangular wave stimulation according to PD is shown in Figure 2(b).
Rectangular Wave Stimulation
The response observed with rectangular wave stimulation showed a pattern very similar to that observed for triangular wave stimulation (Figure 2(c)). In general, a lower amplitude was required to reach the same results with rectangular wave form versus the triangular wave stimulation.
The amplitude required to elicit an effective in home-training over time showed a trend towards increasing over time. The occurrence of discomfort, unspecific responses, and specific response using rectangular wave stimulation according to PD is shown in Figure 2(d). Interestingly an appreciable increase was observed at different checkpoints depending upon the individual patients, although it appeared that the amplitude required by freshly diagnosed patients was always ≤ 10 V, while that required by patients suffering from unilateral facial paralysis for several years was almost always above 10 V.
Triangular vs Rectangular
The amplitude required to selectively stimulate the ZYG was always lower when rectangular rather than triangular wave form was applied, because charge of the rectangular pulse is double that of a triangular pulse - since the area of a rectangle is twice that of a triangle. Given that the electrostimulation device used is voltage-controlled, no charge assessment (PD *amplitude) could be calculated.
Stimulation Parameters
Data is shown up to 11th follow-up interval at 52 ± 2 weeks, the end of the follow-up period. The mean PDs required to elicit the specific response of the targeted ZYG during home training over the follow-up period are shown in Figure 3(a), Table 2.
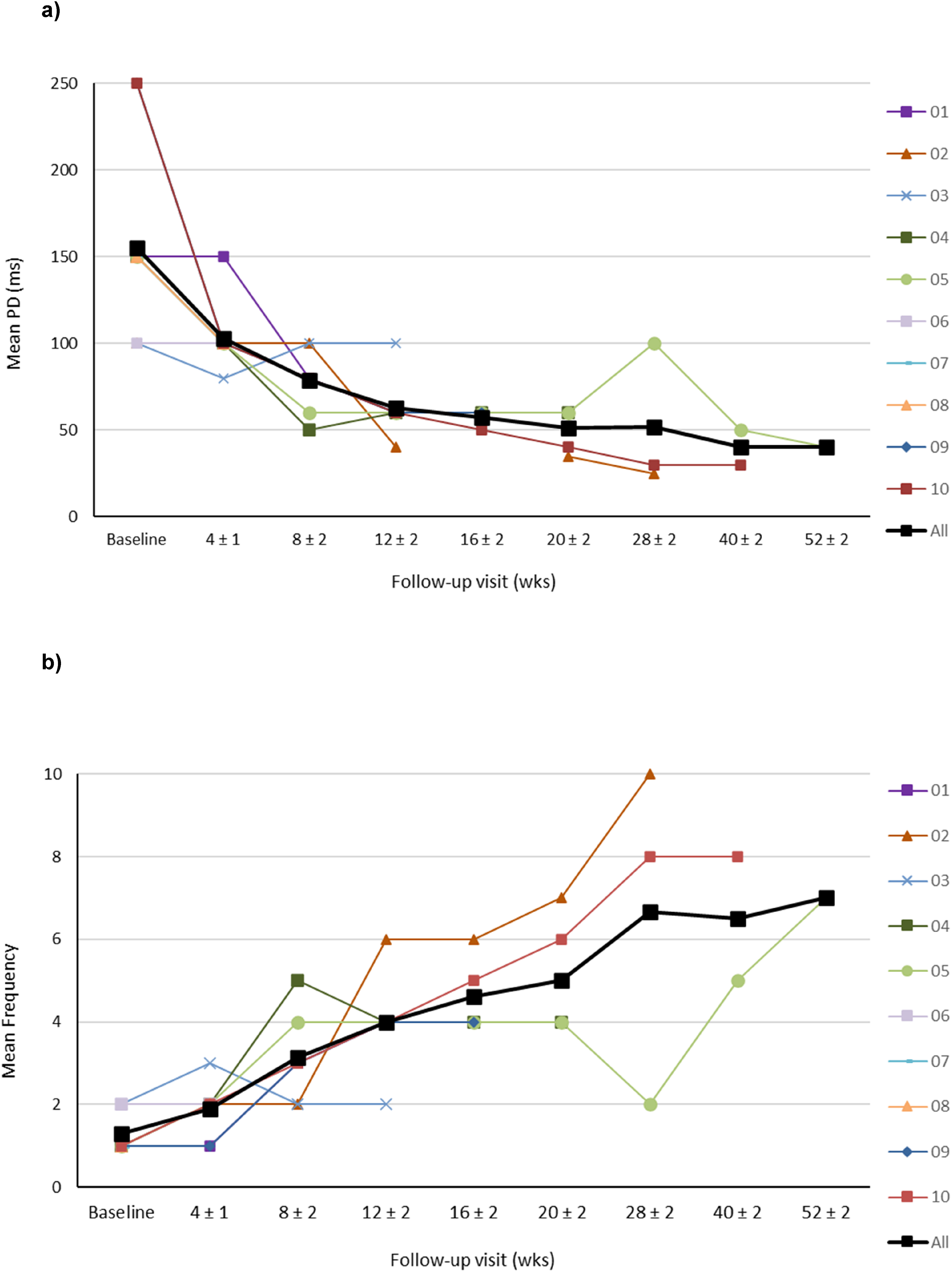
Phase Duration Over the Treatment Period.
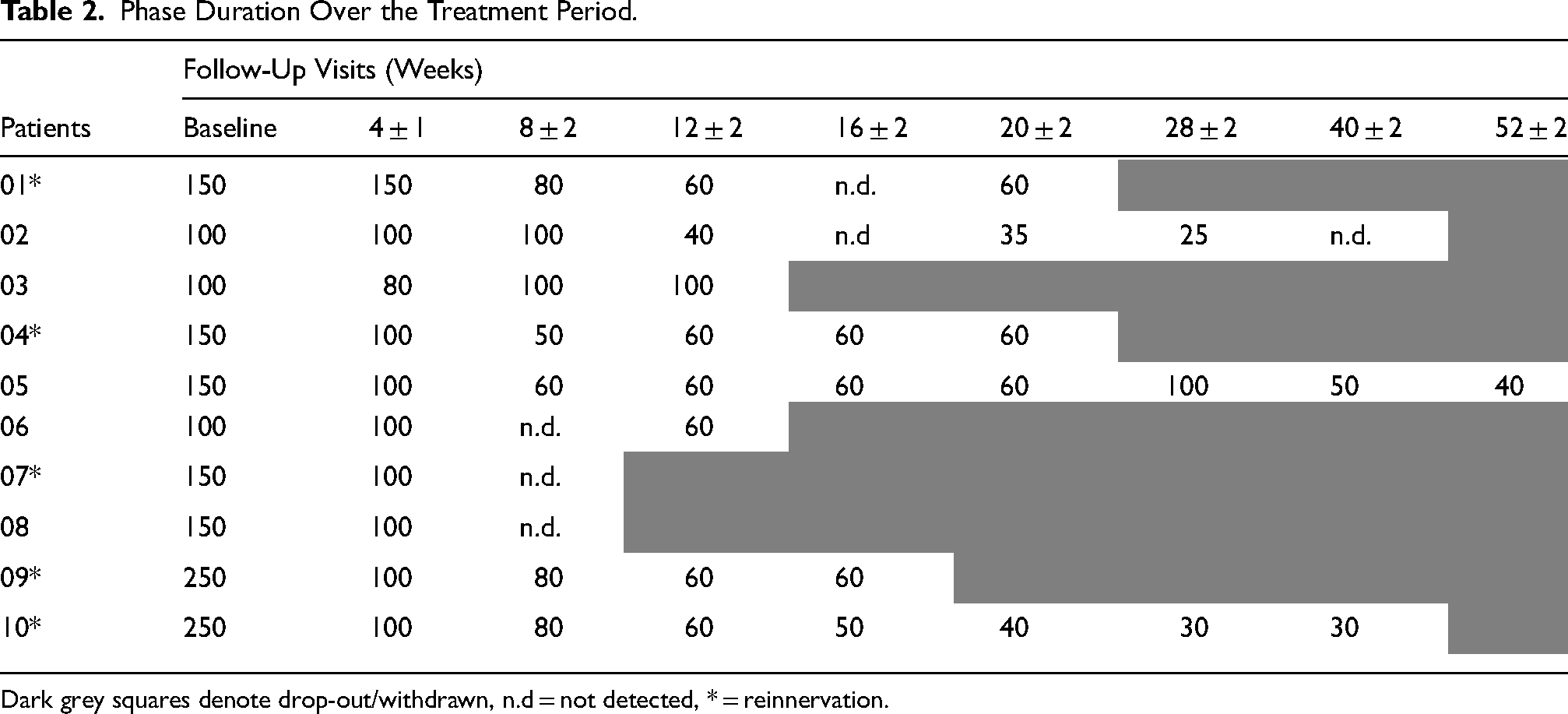
Dark grey squares denote drop-out/withdrawn, n.d = not detected, * = reinnervation.
There did not appear to be a difference in the mean PD needed in the study period before any signs of reinnervation appeared between the five patients that later showed some reinnervation (Patient 01, 04, 07, 09, 10) versus the five patients without signs of reinnervation during the study period (Patient 02, 03, 05, 06, 08), although the data did not allow for statistical evaluation (Table 3). The decrease in PD from baseline to 12 ± 2 weeks (n = 7, p = 0.003), and the decrease from baseline to 20 ± 2 weeks (n = 4, p = 0.001) were both significant. Since the mean PD could be reduced from 155.0 msec ± 55.0 to 51.0 msec ± 12.4 over time, the mean stimulation frequency could be increased during the observation period of 20 ± 2weeks, from 1.3 Hz ± 0.5 at baseline to 5.0 Hz ± 1.5 after 20 ± 2 weeks (Figure 3(a)). As a consequence of the reduction of the PD to generate the same charge of stimulation (following the strength/duration-curve), the mean voltage was increased from 16.2 V ± 2.5 to 27.2 V ± 5.9 (data not shown). The mean frequency from baseline to 20 ± 2 weeks is shown in Figure 3(b). Despite the general trend of reducing the PD and increasing the frequency as a result of the home training, in 13 of 41 follow-ups the parameters were not changed because the motor response was weaker. At 3 follow-ups, patient 3 at 8 ± 2 weeks, patient 4 at 12 ± 2 weeks, and patient 5 at 28 ± 2 weeks, the PD had to be increased again because the patients’ denervated muscles did not yet tolerate the higher frequency applied.
Mean phase duration in all patients and in patients with/without reinnervation.

NA = not available.
Discussion
The results of our study showed that it is possible to find a suitable combination of PD and amplitude capable of eliciting a selective response of the targeted ZYG without the patient´s discomfort or the simultaneous unspecific activation of other ipsi- or contralateral facial muscles. We did not interpret, nor did patients report, any harmful side effects of the home-based FES on the facial muscles over the treatment period.
Using triangular wave stimulation this specific response was observed in all patients with a PD ≥ 25 ms. Lowering the PD usually resulted in the onset of unspecific responses. The response observed with rectangular wave stimulation showed a pattern similar to triangular wave stimulation. Similarly, our 2021 study (Arnold et al., 2021), demonstrated that a PD of ≥ 50 ms, regardless of the waveform being triangular or rectangular, resulted in a distinct ZYG response following surface stimulation. This response was not influenced by the stimulation duration and/or the duration of pre-existing facial paralysis before treatment. Unspecific responses usually occurred with a PD of ≤ 25 ms and occasionally with a PD of 50 ms. In general, our experience showed that at the same PD, the unspecific response was observed with lower amplitudes if the rectangular rather than the triangular wave stimulation was used.
During home training it was observed that the PD required to induce a constant visible contraction of the targeted muscles could be reduced over time. Consequently, both stimulation amplitude and frequency could be increased throughout the majority of the treatment period without causing fatigue in the patients during the 20 min home training. At three follow-up intervals, the denervated muscles showed signs of fatigue. Thus, in Patient 3 at 8 ± 2 weeks, Patient 4 at 12 ± 2 weeks, and Patient 5 at 28 ± 2 weeks a better response was reported with lower home training frequencies. This resulted in a reduction in the frequency and an increase in the PD to target more not yet well-trained muscle fibers at these follow-up intervals. There was a general trend toward decreasing PD and increasing frequency that led to an increase in training intensity on the ZYG. Nevertheless individual adjustment is necessary. In general the maximum training capacity of the patients improved over the complete follow-up period (Krauß et al., 2024). Increasing the strength of the stimulation recruits more muscle fibers (Doucet et al., 2012; Meincke et al., 2024). This has also been shown in several other studies investigating FES in both facial muscles (Choi, 2016; Mäkelä et al., 2019) and muscles excluding the face (Brüggemann et al., 2017; Crameri et al., 2002; Kern & Carraro, 2020), which showed an increase in strength and function after training.
The main limitation of our study was that the volitional and unintended facial muscle movement of the participants was assessed subjectively by the examiners. An objective measurement of the muscle force or acceleration is not established for facial muscles, but could improve the inter-investigator variability. The evaluation of the degree of synkinesis was performed by facial paralysis experts supported by nEMG to objectify the assessment. The use of an automated system to further objectify the assessment would also be of advantage.
Furthermore, the limited population size and the absence of a control group in this study were also limitations. The low initial number of participants was confounded by their discontinuation from the study; either for reasons of reinnervation or drop-out. Bias due to missing data resulting from their exclusion or drop-out cannot be excluded. However, the study was designed as a pilot study and going forward we would increase the number of participants and consider adding a control group for comparative purposes. Besides it must be emphasized that complete peripheral facial paralysis is a rare disease. The precise treatment protocol, the study procedures, the well-established diagnostic procedures in the Facial-Nerve-Center, and high-level of patient compliance, nonetheless allowed us to conclude that surface electrical stimulation is both safe and effective in treating facial muscle atrophy in complete peripheral facial paralysis. The resulting absence of voluntary residual nerve activity demonstrated clearly that FES is capable of halting and even reversing the atrophy of denervated mimic muscles and there were no adverse incidents related to the treatment.
Another limiting factor derived from the targeted muscle size and position. Surface electrodes cover a relatively large area. To target a specific muscle the stimulation parameters could only be adjusted in a small range. In the future, in a real-live clinical setting, higher amplitudes could be chosen to maximize the recruitment of all muscle fibers and also surrounding muscles. It is also possible that by stimulating agonistic and antagonistic muscles at the same time the training effect might increase because the muscles being trained would work against each other causing resistance. Also, training unspecific muscles, i.e., not only the ZYG, but additional affected facial muscles, might increase the positive effect on the whole side of the face by improving the general functionality of the muscles and the perceived training effect to an even greater degree. Even so, the results of our study showed that selective stimulation of a single muscle was possible using surface stimulation, not only during supervised study visits, but also in its every-day application during the long-term home-based training. Moving forward the potential to selectively stimulate a single muscle can be leveraged for the development of implantable solutions that could address the challenges associated with surface stimulation.
Previous work has indicated that the effect of FES on the muscle could be seen as early as 4–8 weeks after starting FES (Krauß et al., 2024). This finding is consistent with previous studies that have quantified muscle condition during chronic phase. FES has the potential to restore facial muscle function in peripheral facial paralysis patients, even if the patient has had persistent facial paralysis for several years (Mäkelä et al., 2019).
Tuncay et al. have also demonstrated that the use of FES in addition to conventional therapeutic approaches, such as physiotherapy, improves patients’ outcomes in the acute phase of facial paralysis. This benefit seems to extend to both complete and incomplete denervated facial muscles (Tuncay et al., 2015).
Di Pietro et al. have even shown a shortened recovery time in Belĺs palsy cases receiving FES in addition to physical therapy, in contrast to a control group receiving only physical therapy (Pietro et al., 2023). Participants in the electrical stimulation group achieved maximal recovery twice as fast as the control group (2.5 weeks versus 5.2 weeks) with no significant differences in facial function or synkinesis between groups at any time point. The Di Petro study was the first human trial of electrical stimulation in Bell's palsy to follow patients 6 months from recovery and also supports that selective electrical muscle stimulation accelerates recovery and does not increase synkinesis. Together with the preliminary data presented herein this suggests that FES has a future potential therapeutic benefit outside of facial paralysis in the targeted rehabilitation of spinal cord injuries or plexus disorders, to stimulate specific muscles in the legs, arms, hands or other areas; potentially improving muscle mass, range of motion, and supporting nerve regeneration in the targeted areas. In more distal areas of the body, the time until motor axons regenerate after e.g., plexus lesion, is much longer than in facial paralysis. Therefore, the positive effect of preventing denervation atrophy is much more relevant for denervated limb muscles.
Likewise, within the framework of this study, Krauß et al. demonstrated, through the use of photographic analysis and other methodologies that functional-objective changes in the facial nerve mimics occur in facial paralysis patients undergoing FES (Krauß et al., 2024). The authors would recommend a regular adjustment of the stimulation settings at least every 4 weeks or even earlier to maximize the therapeutical effect of FES according to the individual training progress. Therefore, telemedicine could be used as an alternative approach to facilitate regular contact between the patient and therapist in regular context, avoiding long travel distances to the individual clinics.
Conclusion
A suitable combination of PD and amplitude is capable of eliciting a selective zygomaticus response without discomfort or simultaneous non-specific activation of other ipsi- or contralateral facial muscles using a PD of ≥ 25 ms only (25 ms = 25 000 µs), which is 100 times longer than what can be applied by other stimulation devices on the market. Electrostimulation devices other than the device used in the present study and only generating shorter PDs do not allow specific stimulation of denervated zygomatic muscle without concomitant stimulation of surrounding muscles or nerves while creating discomfort to the patient. The right choice of stimulation device is therefore very important.
It has been shown that FES has significant potential to halt and even reverse the atrophy of denervated mimic muscles in peripheral facial nerve paralysis patients. FES may therefore be used as a “bridge technology” or a “baby-sitter-procedure” to restore facial muscle capacity before undergoing reinnervating surgery treatment.
Within the scope of this proof-of-principle study, we could show that the stimulation parameters should be individualized to each patient and continually readjusted throughout the FES to enhance training intensity. FES serves to complement existing invasive facial nerve paralysis treatments. Future research requires further investigations with a larger patient cohort and adapted study design with a control group. Surface electrical stimulation has the potential to become an interesting addition to current facial paralysis treatments, particularly in cases for which no suitable therapy has been found.
Footnotes
Acknowledgements
This paper received statistical and medical writing support on a version of this manuscript from MED-EL Elektromedizinische Geräte GmbH, Innsbruck, Austria.
Author Contributions
Conceptualization: M.G, G.F.V; Data curation: W.M, C.K; Formal analysis: D.A, T.L; Funding acquisition: M.G, O.G.-L, G.F.V; Investigation: M.G, J.B,J.K,G.M, A-M.K, D.A, V.M, G.F.V; Methodology: D.A, W.M, C.K, G.F.V; Project administration: M.G, G.F.V; Resources: M.G, O.G.-L, G.F.V; Supervision: D.A, W.M, G.F.V; Validation: D.A, W.M; Visualization: D.A, W.M; Writing of original draft: M.G, J.B, G.F.V; O.G.-L.: Writing of review and editing: J.B; M.G.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by MED-EL Elektromedizinische Geräte GmbH, Innsbruck, Austria.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors M. Geitner, J. Krauß, G. Meincke, AM. Kuttenreich, D. Arnold, J. Ballmaier, O. Guntinas-Lichius and G.F. Volk have received stimulation devices and financial support for travel costs from MED-EL, Innsbruck, Austria.
M. Geitner declares financial support was received for the research, authorship and publication of the article as being a member of IZKF Jena. Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)–(KL 2926/2-1), Clinician Scientist-Program OrganAge, funding number WI 830/12-1.
The remaining authors have no conflict of interest to report.
Data Availability Statement
The data supporting the findings of this study are available upon reasonable request from the corresponding author.
