Abstract
As stroke results in deficits spanning multiple functional domains, interventions that promote neuroplasticity-like mechanisms across brain and body systems may optimize recovery. In this perspective, we propose a rehabilitation strategy combining operant conditioning with corticomuscular coherence (CMC), a neurophysiological marker of communication between brain and muscle. CMC reflects the synchrony of descending cortical drive with spinal motor output, serving as a real-time index of volitional control. We assert that reinforcing CMC through operant conditioning can strengthen movement-related neural circuits while simultaneously engaging cognitive systems responsible for motor planning, attention, and error correction. Drawing from motor learning, systems neuroscience, and neuroengineering evidence, we outline the conceptual rationale along with the translational potential and implementation challenges of this CMC-based operant conditioning approach. We posit that this framework offers a biologically plausible strategy to strengthen residual motor capacity and restore functional integration across disrupted circuits connecting brain and body systems.
Keywords
Introduction
The central challenge of stroke rehabilitation is not merely the mitigation of motor deficits but the re-establishment of skilled movement through relearning. (Krakauer, 2006, 2015). Despite the advancement of acute stroke interventions, many experience persistent motor impairments that restrict independence and alter daily living (Li et al., 2024; Raghavan, 2015). Traditional physical therapy is vital for rehabilitation, often incorporating task-specific training and goal-oriented movement practice (Belagaje, 2017; Raffin & Hummel, 2018; Winstein et al., 2016). However, its effect may be constrained when such training is not explicitly aligned with mechanisms of neuroplasticity or when cognitive contributors to motor control are insufficiently addressed (Cassidy & Cramer, 2017). This necessitates targeting not only the motor cortex and descending pathways, but also the cognitive processes that support skilled motor output (Cramer et al., 2023). Deficits in attention, executive function, and motor planning can hinder rehabilitation, further compounding motor deficits in motor preparation and execution (Verstraeten et al., 2016). This interplay between motor and cognitive systems suggests that rehabilitation strategies engaging both motor and cognitive processes may optimize recovery outcomes.
One promising approach that combines cognitive and motor processes is operant conditioning, a form of learning that reinforces behavior through targeted feedback, which may include verbal cues, visual signals, and real-time performance metrics (Fetz & Finocchio, 1971; Harrison et al., 2019). Biofeedback, a form of feedback that uses non-invasive sensors/equipment to monitor bodily functions such as muscle activation and recruitment, breathing, and heart rate, may serve as an additional feedback source for operant conditioning (Giggins et al., 2013). In clinical practice, therapists frequently incorporate biofeedback into rehabilitation sessions to enhance learning and reinforce motor control (Giggins et al., 2013). Here, we focus on the utilization of electroencephalography (EEG) and electromyography (EMG) to measure corticomuscular coherence (CMC), a measure of brain and muscle functional connectivity (Liu et al., 2019; Mima & Hallett, 1999). Because operant conditioning has the potential to enhance motor function at both cortical and peripheral levels while also fostering cognitive engagement, CMC is especially enticing as a source of biofeedback. Though we acknowledge that much work is needed, we propose that CMC-based operant conditioning represents a compelling strategy to restore volitional movement by [1] integrating various features of motor learning, [2] addressing neurophysiological mechanisms of biofeedback training, and [3] bridging cognitive and motor domains within a single personalized treatment paradigm. Additionally, we discuss the [4] current applications of CMC biofeedback training, and [5] methodological challenges of CMC operant conditioning that must be addressed.
Motor Learning Following Stroke
Motor learning drives post-stroke recovery by promoting neuroplasticity and functional reorganization throughout the nervous system (Cassidy & Cramer, 2017; Dimyan & Cohen, 2011). It enables individuals to refine movements, restore motor function, and adapt to new task demands. While task-specific training contributes to post-stroke motor learning, research indicates that motor learning also involves use-dependent, instructive, and reinforcement learning and sensorimotor adaptation (reviewed by Leech et al., 2022). Each of these processes engage distinct neural pathways and operate on distinct timescales. Integrating multiple learning processes may offer complementary benefits to stroke rehabilitation, improving both the speed and durability of recovery. Operant conditioning strategies involving biofeedback provide a potential tool to integrate between these learning features (Figure 1).
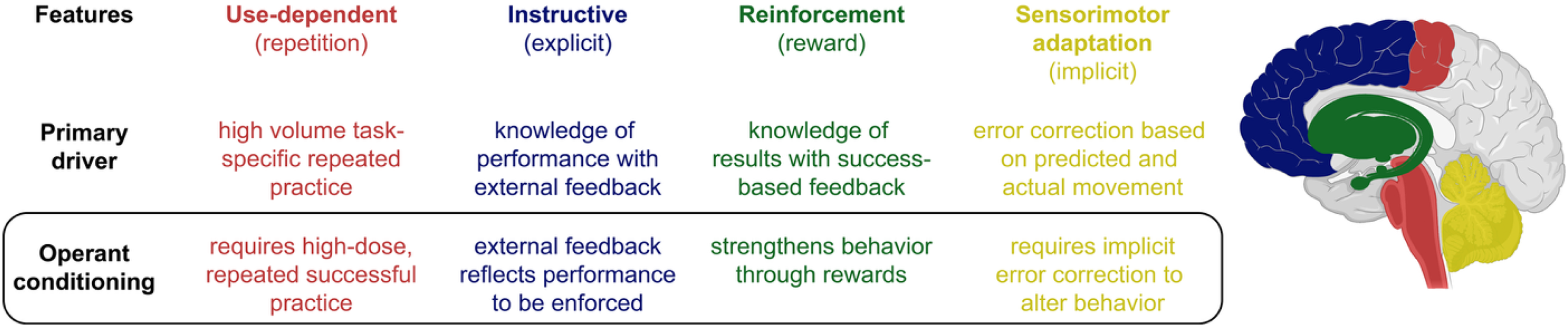
Summary of four core motor learning features relevant to post-stroke recovery. Each feature is linked to a primary driver and operant conditioning component. Brain regions are color-coded by corresponding features, highlighting the distrbuted and interacting systems that support motor learning. Figure adapted from Leech et al., 2022.
Use-dependent learning encompasses repeated practice of a movement, reinforcing motor behaviors through structural and functional plasticity within the corticospinal and sensorimotor networks (Antonioni et al., 2025). This form of learning aligns closely with task-specific training, where extensive repetition strengthens motor representations in the motor cortices and spinal pathways (Rozevink et al., 2023). However, repetition alone is not sufficient for long-term retention, especially when it reinforces maladaptive patterns that do not engage the intended biomechanics or neural circuits (Donnellan-Fernandez et al., 2022; Gregor et al., 2021). Passive movement or excessive physical guidance by the therapist, where the individual is not generating sufficient voluntary movement nor experiencing performance error, hinder learning and do not adequately promote recovery (Abdullahi et al., 2025; Yamaguchi et al., 2020). Active effort and cognitive engagement from the individual are therefore necessary for effective learning during rehabilitation (Huang & Krakauer, 2009; Taylor et al., 2014).
Instructive learning relies on external feedback and cognitive engagement to correct movement errors (French et al., 2021; Leech et al., 2022). This mechanism, primarily mediated by the prefrontal cortex (PFC) and motor planning regions, promotes explicit motor strategy refinement through knowledge of performance and knowledge of results feedback (French et al., 2021; Haith & Krakauer, 2013; Taylor & Ivry, 2014). Unlike use-dependent learning, where repetition strengthens movement, instructive learning requires active cognitive processing where individuals must analyze errors, adjust their movement strategies, and implement corrections (Leech et al., 2022). This process is particularly important in early rehabilitation, as many individuals post stroke exhibit motor planning deficits that impede efficient movement execution (Raghavan, 2007). However, over-reliance on explicit cues negatively impact long-term retention, as individuals may develop an over-reliance on external guidance rather than developing intrinsic error detection strategies (Boyd & Winstein, 2006).
Reinforcement learning relies on reward-based feedback, engaging the basal ganglia and dopaminergic circuits to bias movement selection toward successful outcomes (Leech et al., 2022; Schultz, 2016). This principle underlies operant conditioning, in which reward contingencies shape motor behavior over time (Staddon & Cerutti, 2003). Unlike instructive learning, which depends on explicit feedback, reinforcement learning strengthens implicit movement selection through positive or negative reinforcement (Leech et al., 2022). Recent evidence suggests that reinforcement learning is impaired during the subacute post-stroke period, with individuals demonstrating reduced sensitivity to reward-based adaptation despite preserved motor execution and intact error-based learning (Branscheidt et al., 2025). These findings imply that operant-based rehabilitation strategies may benefit from increased reward salience or delayed implementation early after stroke (Rumbaugh et al., 2007).
Sensorimotor adaptation, governed primarily by the cerebellum, involves the correction of movement errors based on sensory prediction discrepancies (Krakauer & Mazzoni, 2011; Leech et al., 2022). This allows for real-time motor adjustments when movement outcomes deviate from expected sensory feedback (Krakauer & Mazzoni, 2011). Unlike reinforcement learning, which modifies movement selection, sensorimotor adaptation refines movement execution by continuously updating motor commands (Leech et al., 2022). This mechanism is essential for adjusting to dynamic environments (e.g., navigating uneven terrain, using different movement plans, or adapting to assistive devices).
Importantly, these learning features do not operate in isolation. They instead interact dynamically and contribute in varying extents depending on the stage of learning and/or recovery and task demands (Diedrichsen et al., 2010; Huang et al., 2011; Krakauer et al., 2019; Shmuelof et al., 2012). For example, sensorimotor adaptation and instructive learning tend to dominate early in recovery, when explicit correction and rapid adjustments are needed to re-establish basic motor patterns (Krakauer et al., 2019; Shmuelof & Krakauer, 2011). In later chronic phases of recovery, reinforcement and use-dependent learning become more prominent as behaviors are consolidated and practiced extensively (Diedrichsen et al., 2010; Therrien et al., 2016). Leech et al. (2022) proposed that this shift parallels the transition from the cognitive to the associative and autonomous phases of motor learning, as originally described by Fitts and Posner (Fitts & Posner, 1967; Leech et al., 2022). As illustrated in Figure 1, operant conditioning leverages reinforcement learning (Therrien et al., 2016) and sensorimotor adaptation (Avraham et al., 2022) while also employing use-dependent (Du et al., 2022) and instructive learning (Roemmich & Bastian, 2018). Biofeedback-based operant conditioning may thus provide a multi-dimensional framework for optimizing motor learning in stroke rehabilitation.
Operant Conditioning & Biofeedback in Stroke Rehabilitation
Operant conditioning is a fundamental learning principle where behaviors are strengthened or weakened from reward or punishment (Skinner, 1963; Staddon & Cerutti, 2003; Thompson & Wolpaw, 2014). Originally formalized by Skinner in 1937, operant conditioning has been widely applied in behavioral psychology (Skinner, 1963; Staddon & Cerutti, 2003) and more recently in neurorehabilitation (Nelson, 2007). In the context of stroke recovery, operant conditioning may facilitate motor learning by shaping neural activity and driving neuroplasticity to achieve and reinforce desired functional behaviors (Norman et al., 2022; Wolpaw & Thompson, 2023). As shown in Table 1, recent studies using electrophysiological and neuroimaging tools demonstrate that operant conditioning can induce plasticity across multiple levels of the nervous system, from spinal reflex circuits (Thompson & Wolpaw, 2014) to midbrain dopaminergic structures and cortical motor areas (Cassidy & Cramer, 2017; Dimyan & Cohen, 2011; Sulzer et al., 2013).
Operant Conditioning Approaches Using Neurophysiological and Neuroimaging Feedback.
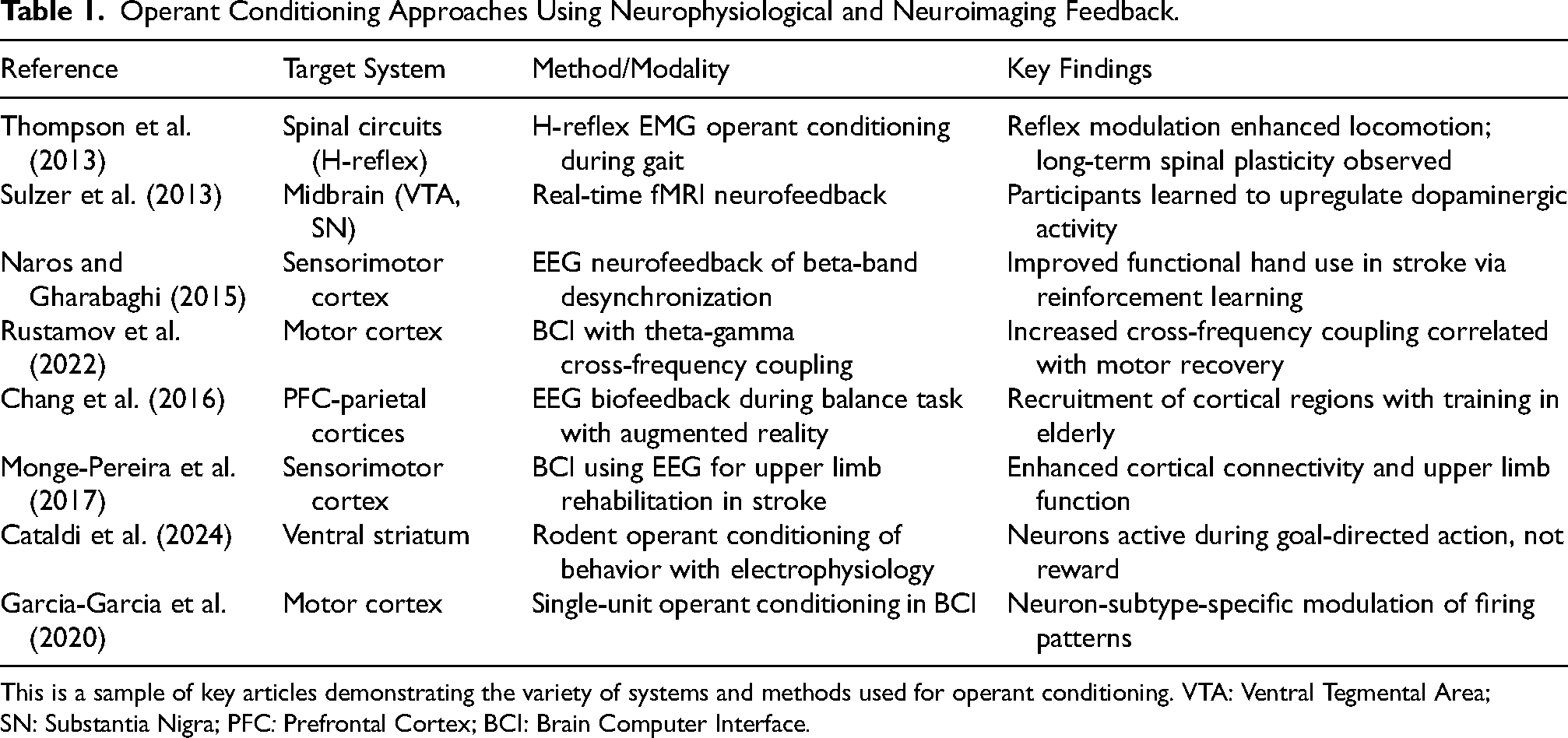
This is a sample of key articles demonstrating the variety of systems and methods used for operant conditioning. VTA: Ventral Tegmental Area; SN: Substantia Nigra; PFC: Prefrontal Cortex; BCI: Brain Computer Interface.
While operant conditioning has been shown to induce plasticity at multiple neural levels (Guerra & Silva, 2010), its applicability and efficacy often vary depending on the nature of the target behavior (Kim et al., 2024; Nelson, 2007). The utility of biofeedback in neurorehabilitation appears to be inversely related to the observability of the target function (Nelson, 2007; Shahani et al., 2024). Biofeedback tends to be most beneficial for physiological signals that do not produce visible movement and therefore lack natural sensory feedback. Examples include reflex modulation, cortical excitability, and patterns of brain-muscle connectivity. Because these processes are not directly observable, individuals must rely on external feedback and trial-and-error learning to gradually shape them (Jueptner et al., 1997). In contrast, overt movements (e.g., reaching or walking) can be adjusted using visual or proprioceptive cues, reducing the need for additional feedback mechanisms (Jueptner et al., 1997). Operant conditioning thus enables individuals to discover agency over these inaccessible systems by reinforcing desirable activity patterns (Weerdmeester et al., 2020).
Recent theoretical models suggest that biofeedback operates not only through physiological learning but also through shifts in cognitive appraisals such as self-efficacy and perceived control (Weerdmeester et al., 2020). These shifts may help reframe internal arousal from a perceived “threat” to a manageable “challenge,” thereby improving emotional regulation and sustaining engagement (Jamieson et al., 2012; Weerdmeester et al., 2020). This parallels educational theories of active learning, where engagement and self-discovery heighten retention (Settles, 2011; Sprenger, 2018).
This framework accounts for the growing application of physiologically-derived biofeedback in both clinical and experimental contexts (Kumar et al., 2019; McLeod et al., 2021; Tahayori & Koceja, 2019). Work by Thompson et al. (2013) demonstrate how reflex conditioning can induce plasticity within spinal circuits, enabling retained modulation of reflex activity. Repeated sessions of targeted reinforcement of EMG patterns elicited by H-reflex stimulation in unimpaired individuals was associated with enhanced gait and muscle coordination. These findings underscore the ability of operant conditioning to “shape” neural activity supporting behavioral function (McLeod et al., 2021; Thompson et al., 2013; Thompson et al., 2022).
Operant conditioning has also been applied and assessed at higher levels of the nervous system using functional magnetic resonance imaging (fMRI) (Sulzer et al., 2013; Wang et al., 2018) and EEG (Naros & Gharabaghi, 2015). Functional MRI studies have demonstrated individuals’ capacity to self-modulate midbrain activity in the substantia nigra and ventral tegmental area– areas involved in dopamine regulation and reward processing, respectively (Cataldi et al., 2024; Sulzer et al., 2013). Through operant conditioning-based biofeedback, these regions have been augmented to enhance functional connectivity and neurochemical signaling—processes that may contribute to sustained neuroplastic changes (Cataldi et al., 2024; Sulzer et al., 2013).
Neurofeedback paradigms utilizing EEG-based biofeedback have depicted modifications in beta (13–30 Hz) oscillatory activity, a frequency range closely linked to sensorimotor control within sensorimotor regions including the primary motor cortex (M1) and supplementary motor area (SMA) in individuals stroke (Milani et al., 2022; Quandt et al., 2019). Naros and Gharabaghi (2015) showed that reinforcement learning involving beta-band event-related desynchronization occurring over M1, premotor cortex (PMC) and SMA led to motor improvement and enhanced hand function in chronic stroke.
In parallel, brain-computer interfaces (BCI) using EEG have been used to entrain functional connectivity between cortical regions and across the frequency spectrum, thereby engaging broader motor-cognitive circuits (Mane et al., 2020; Monge-Pereira et al., 2017). One such mechanism, known as cross-frequency coupling, reflects the interaction between distinct frequency bands across the scalp and is associated with a variety of higher-order functions in learning and recovery (Mark et al., 2024; Rustamov et al., 2022; Rustamov et al., 2025). For example, Rustamov et al. (2022) reported increases in theta—gamma cross-frequency coupling in EEG leads overlying bilateral motor cortices following BCI training which were associated with motor learning and the integration of newly formed motor plans into internal models of movement. Subsequent work by this group in chronic stroke showed that a BCI-based intervention resulted in upper limb motor improvement, which supports the therapeutic potential of biofeedback-based operant conditioning using BCI systems (Rustamov et al., 2025).
Neurobiological Mechanisms of Operant Conditioning
Operant conditioning engages the nervous system at the cellular, regional, and network levels to facilitate functional recovery following stroke (Black, 1972; Stein & Belluzzi, 2014; Teitelbaum, 2022). This multiscale engagement reflects the hierarchical organization proposed by Thompson and Wolpaw (2014), in which conditioning applied at any level not only modifies local activity, but also triggers distributed adaptation that supports functional gains (Black, 1972; Stein & Belluzzi, 2014; Teitelbaum, 2022) Thus, the therapeutic utility of operant conditioning derives not just from its target specificity but from its capacity to propagate plasticity across the motor control hierarchy.
Cellular Level: Operant conditioning modulates sensorimotor neurons, particularly those within the corticospinal tract, responsible for transmitting motor commands from the brain to the periphery (Figure 2) (Arduin et al., 2013; Thompson & Wolpaw, 2014). This process is mediated by synaptic plasticity mechanisms, such as long-term potentiation and long-term depression, which selectively strengthen or weaken synaptic connection, respectively, wherein in reinforced motor actions are encoded by strengthening synaptic efficiency in movement-relevant neural pathways (Gruart et al., 2015; Lebel et al., 2001; Munakata & Pfaffly, 2004; Rosenkranz et al., 2007). Cortical pyramidal neurons in M1 undergo synaptic modifications in response to repeated movement attempts (Jones & Schallert, 1994), encoding new motor representations that help mitigate motor control of the affected extremity (Battaglia-Mayer & Caminiti, 2019). Additionally, interneuron circuits in the spinal cord and brainstem contribute to adaptive sensorimotor integration, refining reflexive and volitional movement pathways through reinforcement-driven neuromodulation (Arroyo et al., 2023).
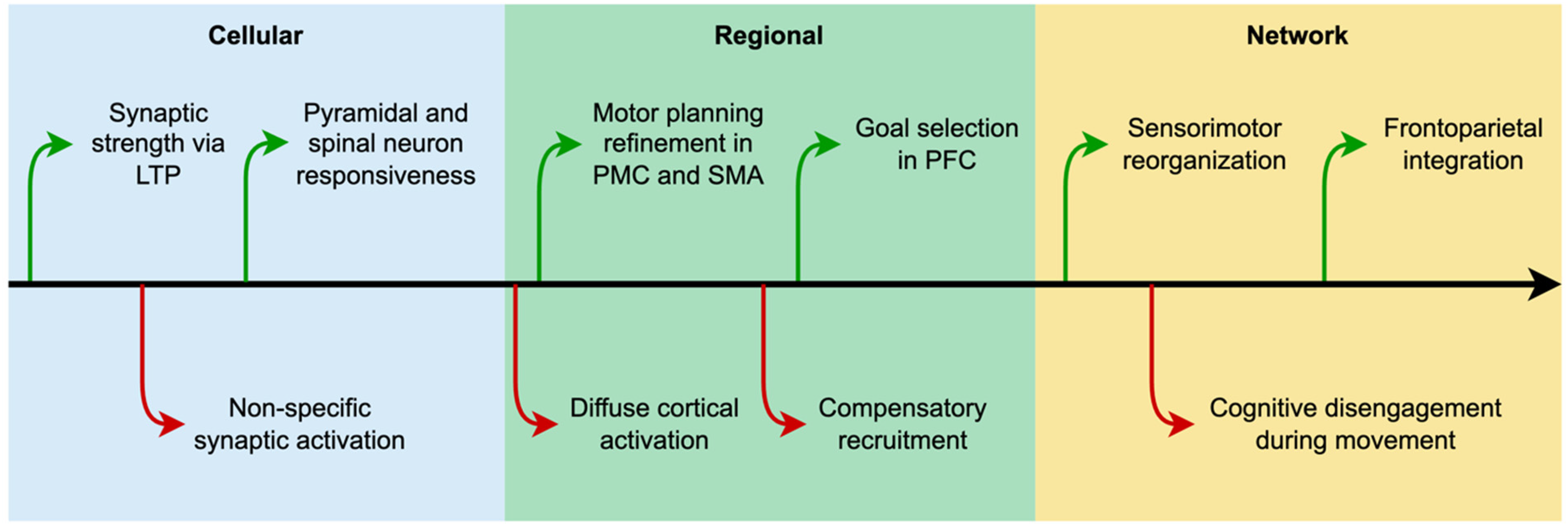
Neurobiological processes influenced by operant conditioning occur across three hierarchical levels encompassing cellular, regional, and network. Green upward arrows represent processes that are enhanced while red downward arrows reflect suppressed functions.
Regional Level: Operant conditioning involves key motor control regions responsible for planning, execution, and adjustment of movement (Figure 2) (Cannon et al., 2014; Ostlund et al., 2009). The M1 serves as the final cortical source for voluntary motor control, refining motor commands based on feedback from reinforcement signals. (Pruszynski et al., 2011; Scott, 2004). PMC and SMA contribute to motor sequencing and preparatory planning, facilitating movement initiation and coordination (Ariani et al., 2025; Halsband et al., 1993; Nakayama et al., 2022). Beyond motor regions, PFC plays a critical role in cognitive control and executive function as they relate to motor control (Fine & Hayden, 2022). The PFC functions as a hierarchical premotor system that integrates sensory inputs, task demands, and motivational cues to shape goal-directed action (Fine & Hayden, 2022). Rather than serving as a passive executive center, PFC actively selects, potentiates, or suppresses action plans across levels of abstraction to guide adaptive motor strategies (Fine & Hayden, 2022). These regions across the brain work in concert, adjusting movement patterns in response to error feedback and reinforcement learning to optimize motor performance (Codol et al., 2020; Mark et al., 2024). By pairing successful movement attempts with positive reinforcement and targeted error feedback, patients can enhance neural engagement in both prefrontal and motor regions, reinforcing effective motor strategies while inhibiting compensatory maladaptive patterns (Codol et al., 2020; Izawa & Shadmehr, 2011).
Network Level: Operant conditioning facilitates large-scale neural interactions between motor, sensory, and cognitive systems, reinforcing functional connectivity essential for stroke recovery (Figure 2) (Teitelbaum, 2022). The sensorimotor network, including M1, somatosensory cortex, thalamus, and basal ganglia, undergoes reorganization as successful movements are reinforced (Darvishi et al., 2017; Kasahara et al., 2024). Reward contingencies first modulate cerebellar circuits, via input from the inferior olive, which in turn influence activity in the sensorimotor cortex (Swain et al., 2011; Velázquez-Pérez et al., 2017). This top-down influence then drives corticospinal and spinal adaptations that directly shape motor output (Thompson & Wolpaw, 2014, 2021).
Beyond sensorimotor loops, frontoparietal networks, including the dorsolateral PFC and posterior parietal cortex, support motor learning by enhancing attention, working memory, and action selection, which are often altered post-stroke (Katsuki & Constantinidis, 2012; Lam et al., 2018). These cognitive-motor interactions are particularly critical in stroke rehabilitation, where lasting functional improvement depends on the reinforcement of goal-directed movement (Verschure et al., 2023). Operant conditioning is thus uniquely poised to engage both motor and cognitive systems through reinforcement-based feedback. This integrative quality may prove valuable in the design and delivery of stroke interventions targeting deficits across multiple systems.
Cognitive-Motor System Integration in Operant Conditioning
Post-stroke recovery is often complicated due to the disruption of both cognitive and motor systems (Cramer et al., 2023). The predominant focus has involved motor re-training, often neglecting the cognitive processes essential for effective movement control (Cramer et al., 2023). However, a growing body of research supports an integrated approach that engages cognitive and motor systems across the brain (Table 2) (Eschweiler et al., 2021; VanGilder et al., 2020; Rice et al., 2022).
Key Brain Regions and Circuitry Utilized in Operant Conditioning.
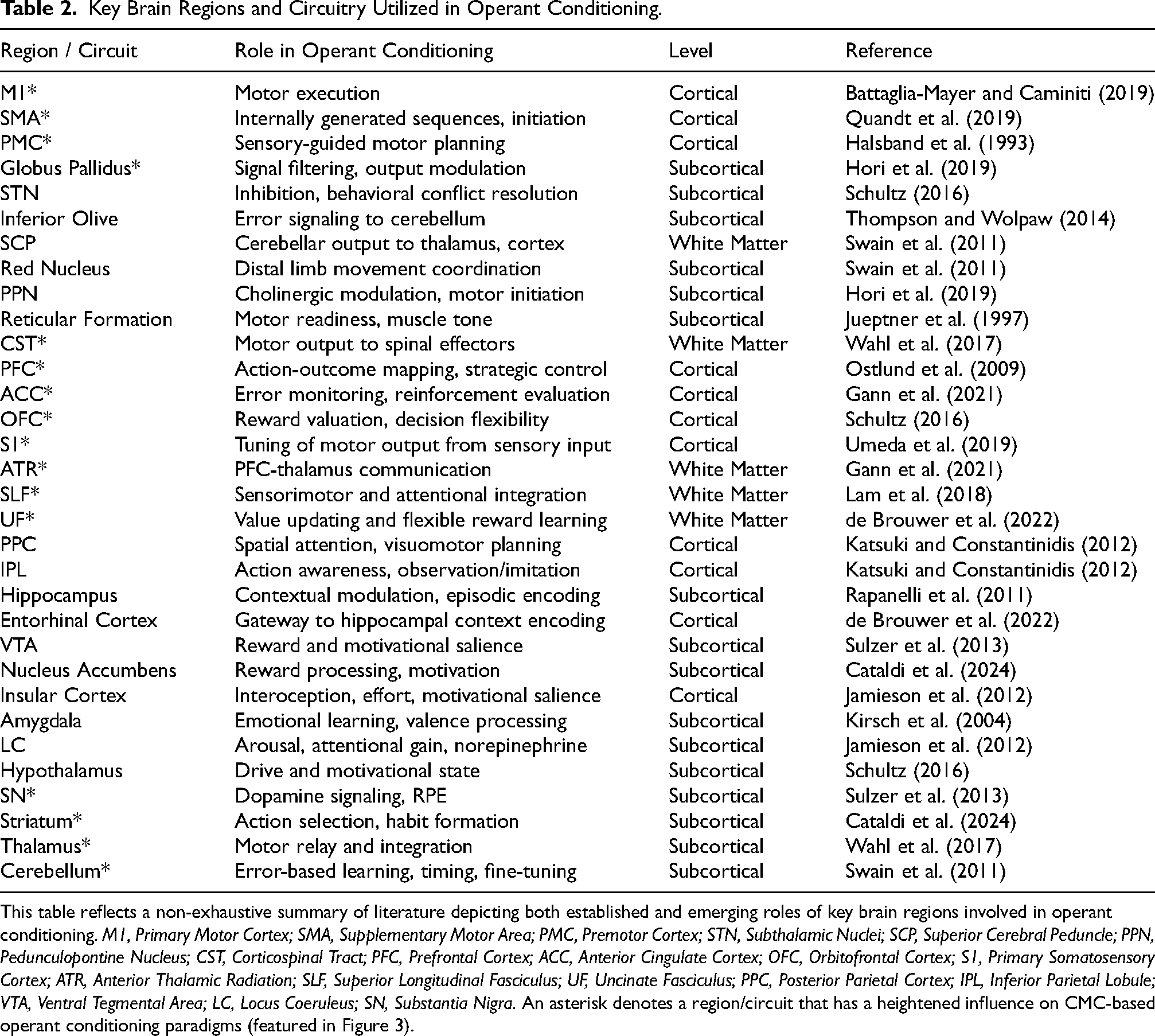
This table reflects a non-exhaustive summary of literature depicting both established and emerging roles of key brain regions involved in operant conditioning. M1, Primary Motor Cortex; SMA, Supplementary Motor Area; PMC, Premotor Cortex; STN, Subthalamic Nuclei; SCP, Superior Cerebral Peduncle; PPN, Pedunculopontine Nucleus; CST, Corticospinal Tract; PFC, Prefrontal Cortex; ACC, Anterior Cingulate Cortex; OFC, Orbitofrontal Cortex; S1, Primary Somatosensory Cortex; ATR, Anterior Thalamic Radiation; SLF, Superior Longitudinal Fasciculus; UF, Uncinate Fasciculus; PPC, Posterior Parietal Cortex; IPL, Inferior Parietal Lobule; VTA, Ventral Tegmental Area; LC, Locus Coeruleus; SN, Substantia Nigra. An asterisk denotes a region/circuit that has a heightened influence on CMC-based operant conditioning paradigms (featured in Figure 3).
Higher-order cognitive functions contribute to motor learning, including executive control, attention, working memory, and motivation (Krakauer et al., 2019). Deficits in these processes following stroke often impede recovery efforts (D’Imperio et al., 2021). Rehabilitation techniques that actively engage cognitive circuits, such as dual-task training (He et al., 2018) and neurofeedback-based reinforcement (Nan et al., 2019), may significantly improve motor retention and re-learning. Several studies have explored the utility of dual-task training, integrating both cognitive and motor systems, in neurorehabilitation (He et al., 2018). These works have identified a break-down in cognitive-motor systems integration in stroke. Gait studies with the addition of mental arithmetic, for example, have shown disparate effects on either task performed together as opposed to isolation (He et al., 2018; Patel & Bhatt, 2014). Operant conditioning provides an internally directed framework for this dual engagement, leveraging intrinsic learning pathways to drive neurophysiological adaptations that enhance both motor execution and cognitive-motor coordination.
Motor recovery is fundamentally driven by reinforcement learning mechanisms, where successful movements are repeated and refined through feedback-based reinforcement (Nikooyan & Ahmed, 2015). This process parallels operant conditioning where desirable motor behaviors are strengthened through reward-driven learning while maladaptive behaviors are suppressed. The two primary reinforcement mechanisms in motor learning relevant to stroke rehabilitation are (1) intrinsic reinforcement, where the individual's awareness of improved movement fosters self-motivation and engagement (Gangwani et al., 2022; Vaughn et al., 2025; Yoshida et al., 2021), and (2) extrinsic reinforcement, where biofeedback, neurofeedback, and reward-based training systems provide external reinforcement cues to reinforce correct movement patterns (Oyake et al., 2020; Palidis et al., 2025; Quattrocchi et al., 2017). Error is another important component in learning as movement-related errors prompt adaptive neural modifications such as long-term potentiation induced by dopamine release (Waelti et al., 2001) and changes in functional connectivity between motor and cognitive areas (Seidler et al., 2013) that refine motor planning and execution (Diedrichsen et al., 2010). These reinforcement-driven changes occur at both cortical and subcortical levels, supporting the restoration of disrupted neural pathways (de Brouwer et al., 2022; Palidis et al., 2019; Seidler et al., 2013).
Intrinsic reinforcement, where individuals derive motivation and learning from their internal awareness of performance improvements, is central to both operant conditioning and post-stroke recovery, particularly given the link between cognitive and motor systems (Kirsch et al., 2004; Nargeot & Simmers, 2011). PFC is hypothesized to contribute to goal-directed motor activity by integrating sensory feedback, task demands, and reinforcement signals (Kobayashi et al., 2010; Riddle et al., 2022; Yamagata et al., 2012). As motor learning progresses, PFC activity becomes more refined, enabling enhanced executive oversight over movement selection, performance monitoring, and error correction (Gann et al., 2021; Mark et al., 2024; Ono et al., 2015). This top-down regulation is important in stroke, where compensatory strategies often compete with re-learning. Interactions between PFC and other brain regions (Table 2 & Figure 3), including SMA, basal ganglia, and parietal cortex, ensures that reinforcement signals are contextually grounded and strategically applied, promoting both behavioral flexibility and skill retention.
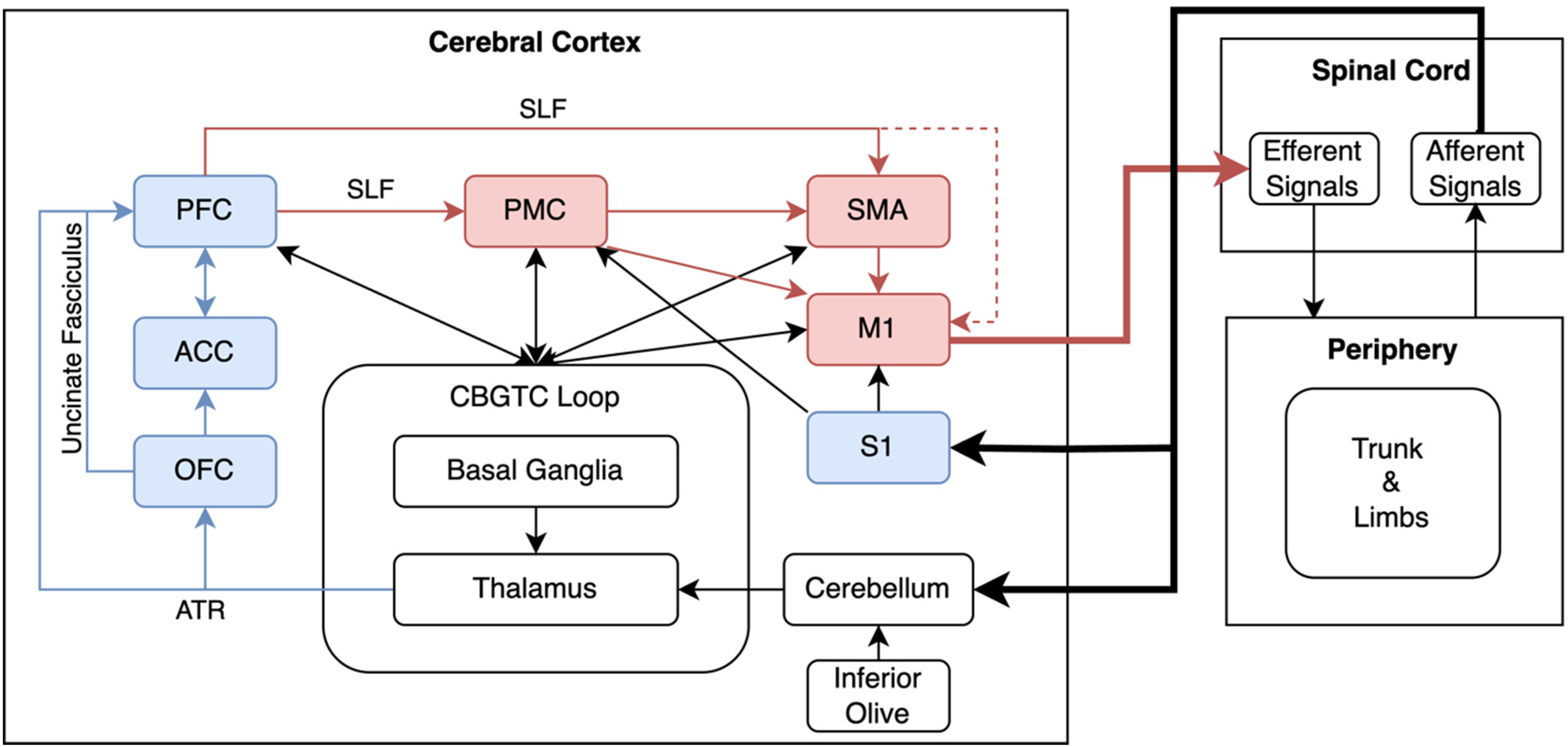
Multisystem Architecture Supporting CMC-based Operant Conditioning. This schematic illustrates the flow of neural information between cortical, subcortical, cerebellar, and spinal systems that support top-down conditioning motor control. Cognitive (blue) and motor (red) cortical regions project to the cortico-basal ganglia-thalamo-cortical (CBGTC) loop, which governs reinforcement-based action selection. In parallel, the cerebellum, receiving error signals from the inferior olive, projects to overlapping thalamic territories that relay refined motor signals back to cortex. These systems converge on the primary motor cortex (M1), which issues descending motor commands via the corticospinal tract (CST). Real-time afferent and efferent signals from the periphery provide the substrate for learning-based modulation of corticomuscular communication. Long-range integration among cognitive and motor regions is supported by white matter tracts, including the CST, superior longitudinal fasciculus (SLF), anterior thalamic radiation (ATR), and uncincate fasciculus. Together, this architecture enables operant conditioning of corticomuscular coherence. PFC, Prefrontal Cortex; ACC, Anterior Cingulate Cortex; OFC, Orbitofrontal Cortex; PMC, Premotor Cortex; S1, Primary Somatosensory Cortex.
Most evidence supporting PFC contributions encompasses explicit motor learning paradigms. The role of PFC in operant conditioning, specifically involving covert physiological signals and limited perceptual feedback, remains less understood. However, emerging work suggests the PFC may still contribute meaningfully during implicit learning. For instance, Taylor et al. (2014) highlighted cerebellar-prefrontal coactivation during cognitive-motor tasks, indicating that the PFC may participate in early error monitoring and strategy formation even when task goals are not consciously articulated. Rapanelli et al. (2011) further demonstrated that operant conditioning promotes neurogenesis in the hippocampus and medial PFC, suggesting the idea that implicit reinforcement learning can reshape prefrontal circuits. Additionally, Chang et al. (2016) showed that EEG-based motor biofeedback in older adults recruits prefrontal and parietal activity during balance tasks, implying cortical involvement in feedback-based modulation of covert motor functions. These findings align with the broader framework proposed by Dayan and Cohen (2011), who assert that motor learning, even during implicit conditions, engages distributed networks that include top-down prefrontal regions to support error-based adjustment, attention, and cognitive control.
Operant conditioning further strengthens motor learning through interactions between both top-down executive control and bottom-up sensory feedback. Repeated reinforcement and error correction promote connectivity between prefrontal and motor regions, while also engaging somatosensory pathways critical for adaptive control (Garcia-Garcia et al., 2020; Hori et al., 2019). PFC is proposed to play a central role in top-down modulation of operant conditioning, guiding goal-directed actions through attention, motivation, and decision-making processes (Fine & Hayden, 2022). Its strategic oversight may enable flexible motor adjustments based on evolving task demands. In contrast, the primary somatosensory cortex (S1) provides essential bottom-up input for refining motor output in real time, especially when external cues are limited (Gale et al., 2021). These systems are hypothesized to operate in a reciprocal loop, where PFC directs attention toward relevant sensory inputs, while S1 conveys performance-related feedback that shapes executive adjustments (Umeda et al., 2019). By co-activating both regions, operant conditioning may facilitate the integration of cognitive and sensorimotor processes, supporting a more adaptive motor control system. While the PFC's role as a convergence hub for cognitive, motor, and reward circuits is theoretically plausible, direct evidence linking PFC activity to CMC-based operant learning remains limited and future studies are needed to clarify its specific contributions in this context.
Operant Conditioning with Corticomuscular Coherence
As previously discussed, biofeedback is most effective when directed toward covert physiological processes (Nelson, 2007). This is particularly relevant in stroke rehabilitation, where motor impairments may arise from disrupted corticospinal descending signal propagation rather than just peripheral muscle weakness alone (Madhavan et al., 2011; Wahl et al., 2017; Ward et al., 2006). Rehabilitation should thus entail retraining of these neural pathways responsible for motor execution (Pekna et al., 2012).
Multisystem biofeedback addresses this challenge by integrating cortical, spinal, and muscular activity to reinforce top-down motor control. This approach facilitates cognitive-motor engagement, as higher-order brain regions involved in attention, motor planning, and reinforcement learning actively shape movement strategies. Enhanced sensorimotor synchronization ensures that cortical motor commands are more precisely aligned with peripheral muscle activation, supporting more efficient motor learning and execution (Wu et al., 2020). This model mirrors real-world motor control, which depends on coordinated activity across the brain, spinal cord, and musculature.
A promising neurophysiological marker of top-down motor control is CMC, a measure of the synchronization between cortical motor areas and muscle activity (von Carlowitz-Ghori et al., 2014a). CMC reflects the functional communication along the corticospinal tract, which is highly relevant for understanding and modulating motor function (Gao et al., 2024; Ibáñez et al., 2021). Most prominent in the beta-band (13–30 Hz) frequency range, CMC plays a critical role in motor precision and stability, with higher coherence indicating more effective motor-cortical signaling (Ibáñez et al., 2021; Parmar et al., 2024; Rossiter et al., 2013). Increased CMC is associated with improved corticospinal efficiency, and functional movement control (Kristeva et al., 2007). Critically, CMC evolves dynamically with motor practice and training, establishing its value as both a biomarker of recovery and a target for intervention (Fauvet et al., 2021; Lattari et al., 2010; Rossiter et al., 2013).
While these features position CMC as both a biomarker of recovery and a potential target for operant conditioning, most supporting evidence stems from studies in unimpaired individuals. Its application in stroke rehabilitation remains exploratory, and its clinical relevance requires further investigation. Early findings suggest that CMC-based neurofeedback may enable reinforcement of brain-muscle connectivity in real time, potentially enhancing volitional motor control post-stroke (Aikio et al., 2021; Velázquez-Pérez et al., 2017). Unlike traditional biofeedback methods that focus on either cortical or muscular activity alone, CMC-based conditioning enables real-time reinforcement of brain-muscle connectivity. Because this measure reflects how well motor intent is translated into action, CMC conveys a more holistic view of motor system function. By conditioning CMC, individuals post-stroke may learn to reinforce this brain-muscle synchrony in real-time, strengthening the pathways responsible for voluntary motor control. Further clinical validation to confirm its therapeutic potential is needed.
Several recent studies support the efficacy of CMC-based biofeedback in enhancing motor control across various populations (Chang et al., 2016; Handiru et al., 2024; Khademi et al., 2022; Naros & Gharabaghi, 2015). Mendez-Balbuena et al. (2012) have shown that reinforcing beta-band CMC through real-time feedback enhanced volitional motor control and strengthened sensorimotor connectivity in unimpaired individuals. Similarly, Khademi et al. (2022) showed that proprioceptive feedback, delivered via a robotic orthosis, increased CMC between the sensorimotor cortex and target muscles in individuals post-stroke, further demonstrating their capacity to voluntarily control their brain-muscle synchronization. Together, these findings suggest that CMC is not just a marker of motor system integrity but also a modifiable target for reinforcement-based training.
The relationship between CMC and motor performance appears to be strongly correlated, with improvements in one often paralleling change in the other. While this bidirectional association supports the rationale for using CMC in neurorehabilitation, causal links remain unproven, particularly in stroke. As such, CMC may serve as a promising marker or mediator of adaptive motor change, rather than a direct driver. Notably, von Carlowitz-Ghori et al. (2015) demonstrated that individuals can volitionally modulate CMC strength using real-time neurofeedback, independent of overt changes in muscle activity (von Carlowitz-Ghori et al., 2014b). In this study, participants successfully increased or decreased beta-band coherence between EEG and EMG signals through mental strategies alone, confirming that CMC is not only responsive to peripheral feedback but can also be shaped by top-down, intentional modulation (von Carlowitz-Ghori et al., 2014b). This capacity for volitional control supports the feasibility of applying operant conditioning frameworks to enhance CMC as a target of stroke rehabilitation. Furthermore, prior work in spinal cord injury has shown that operant conditioning of neural activity can yield meaningful improvements in functional movement (Manella et al., 2013). CMC-guided biofeedback protocols, though still exploratory, may eventually form a core component of modern neurorehabilitation by targeting both central and peripheral contributors to motor impairment.
Design Elements for Corticomuscular Coherence-Based Operant Conditioning Tasks
Developing operant conditioning paradigms based on CMC measurement requires thoughtful design and consideration of feedback and volitional control strategies to effectively promote neural and behavioral adaptation.
Feedback Paradigm Design
A core component of operant conditioning is the reinforcement of desirable physiologic and neural states. In CMC-based paradigms, feedback must be closely tied to the coherence signal, providing participants with a clear and timely indicator of performance. Two primary feedback strategies have emerged: (1) Real-time CMC biofeedback is provided when beta-band coherence crosses a dynamic threshold during volitional movement attempts (von Carlowitz-Ghori et al., 2015), and (2) Post-trial Performance Feedback is given in blocks, summarizing coherence trends and reinforcing adaptive changes across trials (Figure 4) (Thompson et al., 2013; Thompson & Wolpaw, 2014, 2021). This dual approach may help balance immediate neuroplastic reinforcement with higher-order cognitive engagement. Comparison of these feedback timing strategies to determine potential differences in learning and retention is a necessary next step.
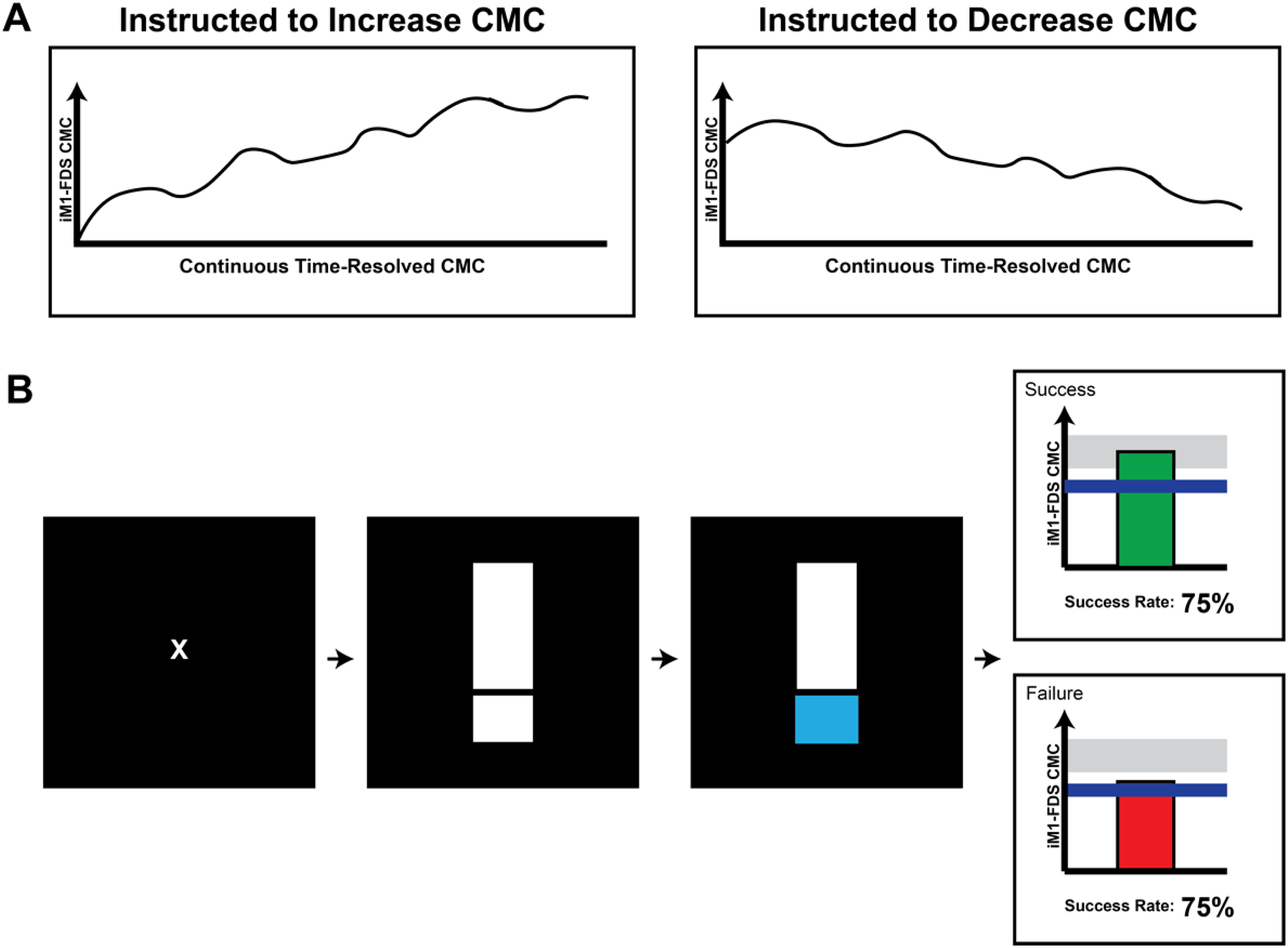
Two proposed operant conditioning feedback mechanisms for corticomuscular coherence (CMC) measurement between ipsilesional primary motor cortex and flexor digitorum superficialis (iM1-FDS). Both approaches rely on internal, volitionally driven operant conditioning mechanisms to shape CMC but differ in the timing of reinforcement (continuous vs post-trial). (A) An exemplar design wherein instantaneous CMC is continuously displayed to participants during a grip task where they receive instruction to voluntary “increase/decrease their brain-muscle communication.” (B) Schematic of complementary feedback where participants receive visual feedback at the end of each grip trial indicating success or failure based on whether the mean CMC value met a predefined target. In this case, participants focused on a fixation cross and then squeezed the grip device at the stimulus onset (thermometer, middle black box) to raise the thermometer to a desired force output target (right black box).
Volitional Control and Task Design
The success of CMC-based operant conditioning depends on participants’ ability to volitionally modulate brain-muscle synchrony. This modulation can take several forms depending on the design of the task and the motor or cognitive capacity of the participant. Most commonly, individuals engage in overt motor actions such as repetitive gripping or sustained isometric contractions (illustrated in Figure 4B), which provide measurable motor output synchronized with underlying neural activity often captured with EEG.
Alternatively, motor imagery, the mental rehearsal of movement without physical execution, may be employed, particularly in populations with severe impairment and limited mobility. Action observation may serve as a complementary form of motor imagery (Li et al., 2018). By viewing others perform goal-directed actions, both clinical cohorts and unimpaired young adults have demonstrated engagement of similar neural pathways involved in active movement (Mark et al., 2023), including the mirror neuron system (Li et al., 2018; Mulder, 2007).
In addition to motor-based approaches, cognitive strategies such as attentional focus, reinforcement framing, or visualization techniques can support volitional control of CMC. These strategies are useful when the neural target is abstract or when participants require support in conceptualizing how to engage relevant brain-muscle networks. Instructional framing is therefore critical as participants must understand that they are not simply performing a motor task but actively training a neurophysiological signal. To maximize the effectiveness of operant learning, CMC protocols often incorporate structured guidance, dynamic thresholding, and performance-based feedback. These elements help sustain motivation and ensure that reinforcement aligns with genuine changes in brain-muscle synchrony. Over time, such scaffolding may facilitate the internalization of control strategies to promote lasting change.
Reward Type and Reinforcement Framing
The type and framing of reinforcement in CMC-based operant conditioning tasks may influence motivation, neural engagement, and learning outcomes. Research has shown that different reward modalities, such as monetary or social-, token-, and sensory-based, activate reward-related neural substrates (i.e., striatum, insula, and anterior cingulate cortex) to varying extents likely driven by context and salience factors (Bieniek & Bąbel, 2023; Delgado et al., 2000; Metereau & Dreher, 2013). Social- and token-based rewards, for instance, have been shown to outperform verbal feedback in shaping conditioned responses, suggesting that tangible, goal-directed reinforcers may be more effective in modulating learning-relevant physiology (reviewed by Weinstein, 2023). In biofeedback paradigms, symbolic rewards such as points, visual progress bars, or success indicators may serve as accessible substitutes for more complex reinforcement structures, which may further support their clinical application. Furthermore, framing the task in terms of “training brain-muscle communication” or “enhancing internal control” may optimize cognitive engagement and operant responsiveness (Wolpaw, 2018). Reward type and feedback framing are essential design elements for tailoring operant protocols to individual learners.
Methodological Challenges and Considerations
There are several methodological and translational challenges with CMC-based biofeedback application in a rehabilitation setting (summarized in Table 3). Reliability and specificity of CMC measurement is a primary concern. Signal quality is highly sensitive to confounding factors such as movement artifacts, volume conduction, and non-motor cortical activity (Gangwani et al., 2023; Liang et al., 2020; Sun et al., 2023). Robust estimation of true corticospinal interactions requires advanced signal processing techniques, and, critically, the necessary steps for real-time CMC biofeedback preprocessing are currently under debate (Delorme, 2023; Robbins et al., 2020).
Methodological Challenges and Solutions in Implementing CMC-Based Feedback Paradigms.
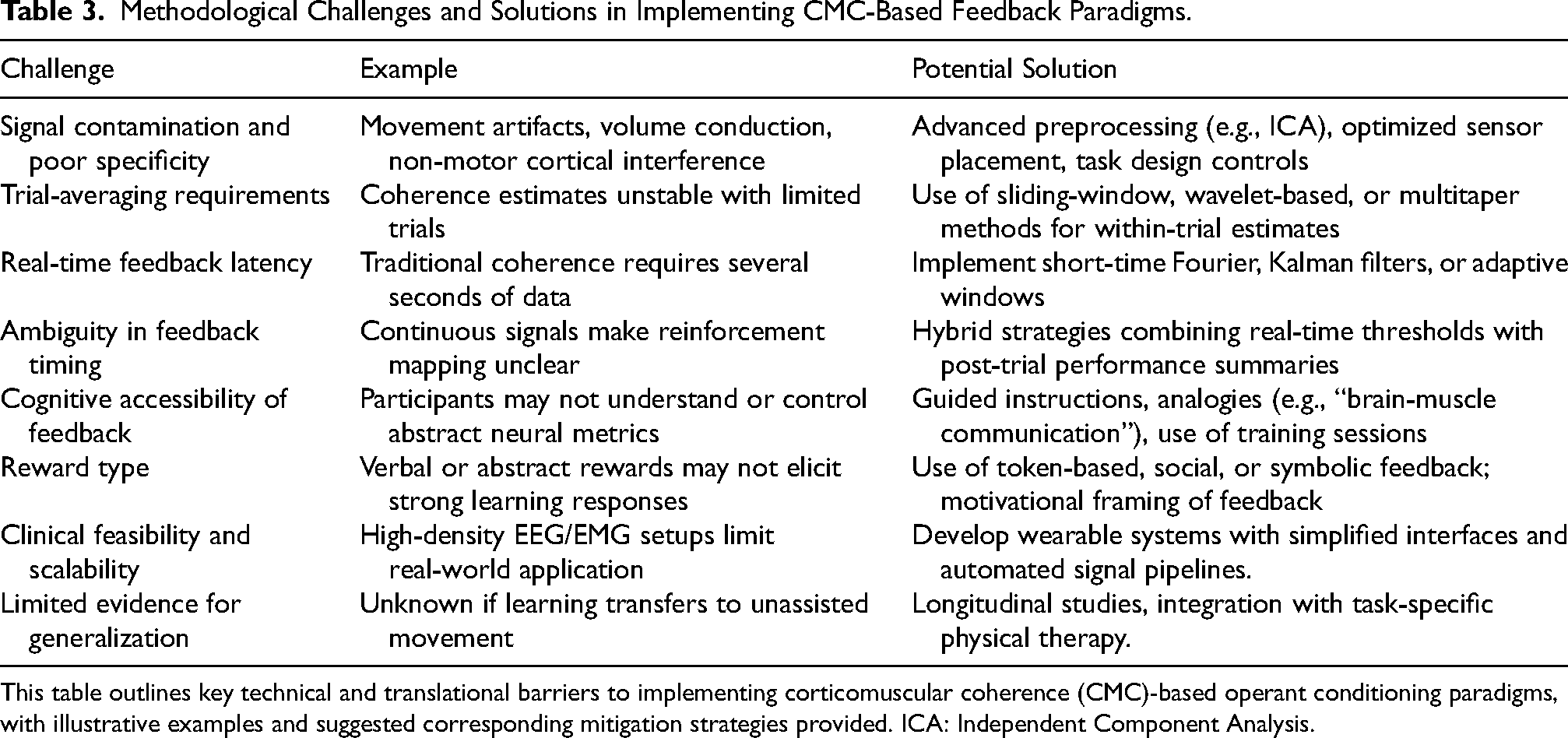
This table outlines key technical and translational barriers to implementing corticomuscular coherence (CMC)-based operant conditioning paradigms, with illustrative examples and suggested corresponding mitigation strategies provided. ICA: Independent Component Analysis.
Another concern regards the feasibility of computing CMC at the single-trial level as coherence is typically defined across repeated trials, with estimates requiring multiple observations to generate a stable cross-spectrum (Figure 5) (Halliday et al., 1995; Schoffelen & Gross, 2009). While emerging techniques such as sliding window coherence and time-frequency decompositions (e.g., wavelet, multitaper) offer promising alternatives for time-resolved feedback (Figure 5) (Bayraktaroglu et al., 2011; Bigot et al., 2011; Cohen, 2014; Fauvet et al., 2021), several of these approaches are still under refinement and not yet widely adopted.
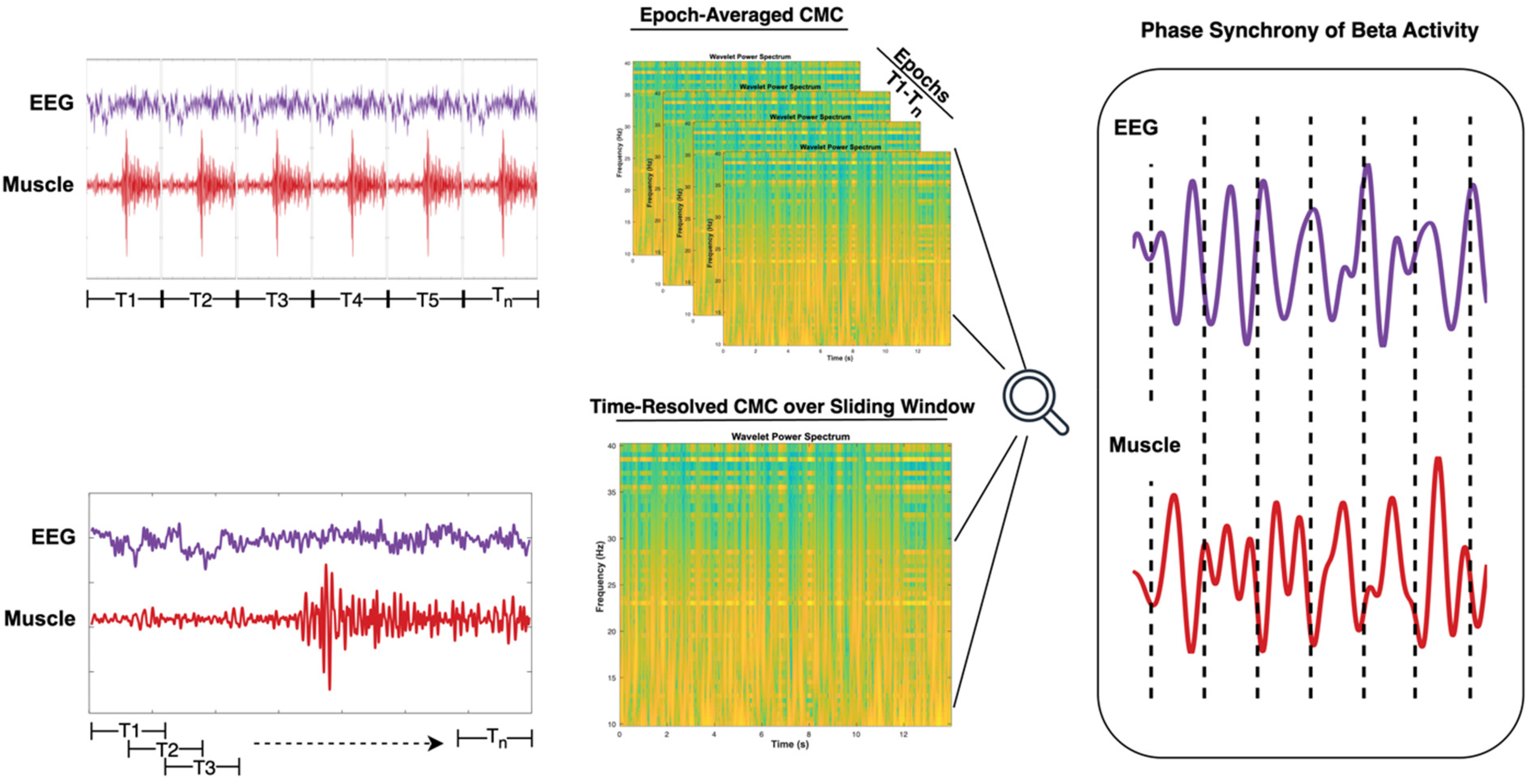
Two approaches to CMC estimation between EEG (purple) and EMG (red) signals. The top illustration exemplifies a traditional method using trial-averaged time-frequency decomposition (e.g., wavelet power spectrum across repeated epochs, T1–Tn), suitable for detecting stable coherence but requiring many trials. The bottom illustration presents a dynamic approach using moving-window analysis to resolve time-varying changes in CMC during single trials. The output of both approaches is beta-band phase alignment, or coherence, between both EEG and EMG signals. Together, these methods support both retrospective analysis and real-time feedback applications for motor learning and neurorehabilitation.
Importantly, Bigot et al. (2011) introduced a statistical framework that enhances the interpretability of wavelet-based coherence estimates, even with a limited number of trials. By applying a thresholding method to wavelet cross-spectrum estimates, their approach permits robust detection of time-frequency dependencies without relying on large sample sizes. Building on this framework, Fauvet et al. (2021) applied a wavelet-based coherence framework to assess time-resolved CMC during voluntary movement, showing that dynamic CMC metrics can meaningfully reflect motor control impairments in individuals post-stroke. Together, these advances challenge the notion that CMC must be computed over many trials, and they support the feasibility of continuous CMC estimation for real-time biofeedback applications (Figure 5). These developments are particularly relevant as the field moves toward within-trial operant conditioning paradigms that require temporally aligned neural feedback. Future research efforts focusing on validation and their robustness and scalability for clinical research use are needed.
Real-time computation of CMC introduces additional obstacles. Traditional spectral coherence calculations rely on windowed analyses that require several seconds of data, limiting the immediacy of feedback (Cohen, 2014). While millisecond-scale updates may not be essential, as seen in H-reflex and motor-evoked potential paradigms (Thompson et al., 2009; Thompson et al., 2013; Thompson et al., 2022; Thompson & Wolpaw, 2021), timely yet stable feedback remains necessary for linking CMC change to movement (Pruszynski et al., 2011; Scott, 2004). Advanced approaches using short-time Fourier transforms, wavelet-based coherence, sliding windows, and Kalman filtering can improve responsiveness while maintaining signal fidelity (Fauvet et al., 2021; von Carlowitz-Ghori et al., 2015). These techniques and approaches require additional study before more widespread implementation.
From a translational perspective, current EEG-EMG systems used in CMC studies often utilize stationary equipment/set-up schemes (Stoyell et al., 2021). Moving toward clinically viable and portable solutions will require the development of user-friendly, wearable EMG interfaces, and real-time signal integration platforms (Armand Larsen et al., 2024; Chang et al., 2023). While promising prototypes exist, these systems remain in development and must further contend with several challenges prior to real-world use: signal quality optimization in mobile conditions, minimizing setup time, ensuring data security, and designing intuitive user interfaces that support both patients and clinicians.
Lastly, long-term retention and functional generalization of CMC-based learning remain uncertain (reviewed by Liu et al., 2019). The timeframe of learning-related motor improvement persisting beyond training or the extent to which learning-related gains transfer to other movements are important questions to elucidate. Continued evaluation of how CMC training interacts with broader learning mechanisms, including reinforcement and use-dependent plasticity, are therefore necessary to determine its utility in stroke rehabilitation.
Conclusion
CMC-based operant conditioning offers a promising multisystem strategy for stroke rehabilitation, targeting both motor execution and the cognitive processes that drive learning. By reinforcing brain-muscle connectivity through dynamic feedback, this approach engages distributed cognitive-motor neural networks, from prefrontal and sensorimotor cortices to spinal effectors, to promote adaptive plasticity. The integration of top-down control and bottom-up sensory feedback positions CMC as a biologically grounded, functionally relevant biomarker of recovery. Future efforts should be focused on developing clinically viable, user-friendly EEG-EMG systems; validating optimal feedback timing and structure; and assessing long-term retention and real-world transfer. Continued research should examine how CMC-based conditioning can complement existing therapies, personalize rehabilitation, and ultimately enhance independence and quality of life for individuals post-stroke.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Foundation for Neurofeedback and Neuromodulation (Mini-Grant #602-2024).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JMC serves as a consultant for Astellas Pharma, Inc. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
