Abstract
Oronasal fistula formation, associated with maxillary canine teeth, is a frequent sequela to untreated periodontal disease, and extraction has traditionally been regarded as the primary treatment. A 10-year-old spayed female miniature Dachshund was presented with an oronasal fistula involving the left maxillary canine tooth. After initial non-surgical management with minimally invasive non-surgical technique (MINST) failed to resolve the periodontal pocket and fistula, microscope-assisted periodontal regenerative therapy combined with an Er:YAG laser was performed. The treatment achieved complete closure of the fistula together with reduction of the periodontal pocket. Cone beam computed tomography at 8 months post-surgery confirmed new bone formation. This case demonstrates that the integration of microsurgical visualization and Er:YAG laser therapy can enable preservation of maxillary canine teeth with oronasal fistula, providing a minimally invasive alternative to extraction.
Keywords
Introduction
Periodontal disease is a common condition in dogs that causes progressive destruction of the periodontal tissues and ultimately results in tooth mobility and loss. 1 Maxillary canine teeth are especially predisposed because their roots lie in close proximity to the nasal cavity. As periodontal disease advances, oronasal fistulas may develop, sometimes leading to clinical signs such as nasal discharge, epistaxis, sneezing, or stertor; however, some patients remain asymptomatic. 2 Traditionally, extraction of the affected canine tooth with surgical closure of the fistula has been regarded as the treatment of choice. 3
Nevertheless, canine teeth are of particular functional and esthetic importance. They contribute to proper occlusion and prehension and provide support for the lips and facial structure. Although they are not essential for mastication and some edentulous patients can maintain an excellent quality of life, preserving canine teeth can still provide meaningful structural and functional benefits. Thus, their preservation should be prioritized whenever feasible. Periodontal surgery and regenerative therapies represent valuable alternatives to extraction when preservation is possible.
In human periodontology, minimally invasive techniques under magnification, such as the minimally invasive non-surgical technique (MINST), have gained prominence in recent years. MINST enables debridement of granulation tissue and calculus removal without gingival incision, and has been shown to reduce probing pocket depth (PPD) and improve clinical attachment level, confirming its utility as a regenerative approach.4–6 In practical terms, MINST involves subgingival instrumentation performed under magnification without flap elevation and may include debridement and, when indicated, root planning, depending on lesion characteristics and treatment objectives. The technique is performed under magnification using fine ultrasonic and manual instruments, allowing thorough debridement via the gingival sulcus without flap elevation. These improvements are thought to result from preservation of soft-tissue architecture, minimal surgical trauma, and stabilization of the blood clot, all of which support periodontal healing.7,8 However, when lesions involve deep intrabony defects or other complex periodontal lesions, the efficacy of conventional instruments may be limited, underscoring the need for adjunctive technologies. For this reason, recent studies have reported combining MINST with adjunctive modalities, including regenerative biomaterials and laser therapy to overcome these limitations and enhance clinical outcomes. 9
The Er:YAG laser has been widely used in human periodontal therapy for calculus removal, debridement of inflamed granulation tissue, and root surface modification.10–12 Its high absorption in water allows efficient ablation with minimal thermal damage,12,13 while creating a biologically favorable root surface that supports periodontal healing.13,14 These properties make the Er:YAG laser a valuable adjunct when conventional instrumentation alone is insufficient.10–14
Experimental research in dogs has demonstrated that flap surgery incorporating an Er:YAG laser significantly promotes new bone formation compared with conventional techniques. 15 Likewise, the effectiveness of trafermin, a recombinant human basic fibroblast growth factor (rhbFGF), for periodontal regeneration has been validated in canine models, 16 and enamel matrix derivatives combined with bone grafts have been successfully applied in clinical cases of intrabony defects in maxillary canine teeth. 17 Enamel matrix derivative (EMD) promotes periodontal regeneration by stimulating new cementum and periodontal ligament formation, while collagen matrices function as biocompatible scaffolds that stabilize the blood clot and support defect fill during healing, further broadening the range of regenerative options in veterinary dentistry. 17
Despite these promising advances, reports describing preservation of canine teeth affected by oronasal fistula using regenerative therapy or laser technology remain exceedingly rare in veterinary dentistry. The present case describes successful preservation of a canine tooth with an oronasal fistula through MINST-assisted flapless regenerative therapy combining microsurgical visualization with Er:YAG laser application.
Case Report
A 10-year-old, 4.55 kg, spayed female Miniature Dachshund, body condition score 5/9, was presented with chronic nasal discharge and sneezing. The dog had never received any professional dental treatment. The patient's diet consisted of commercial dry food without the use of dental care products.
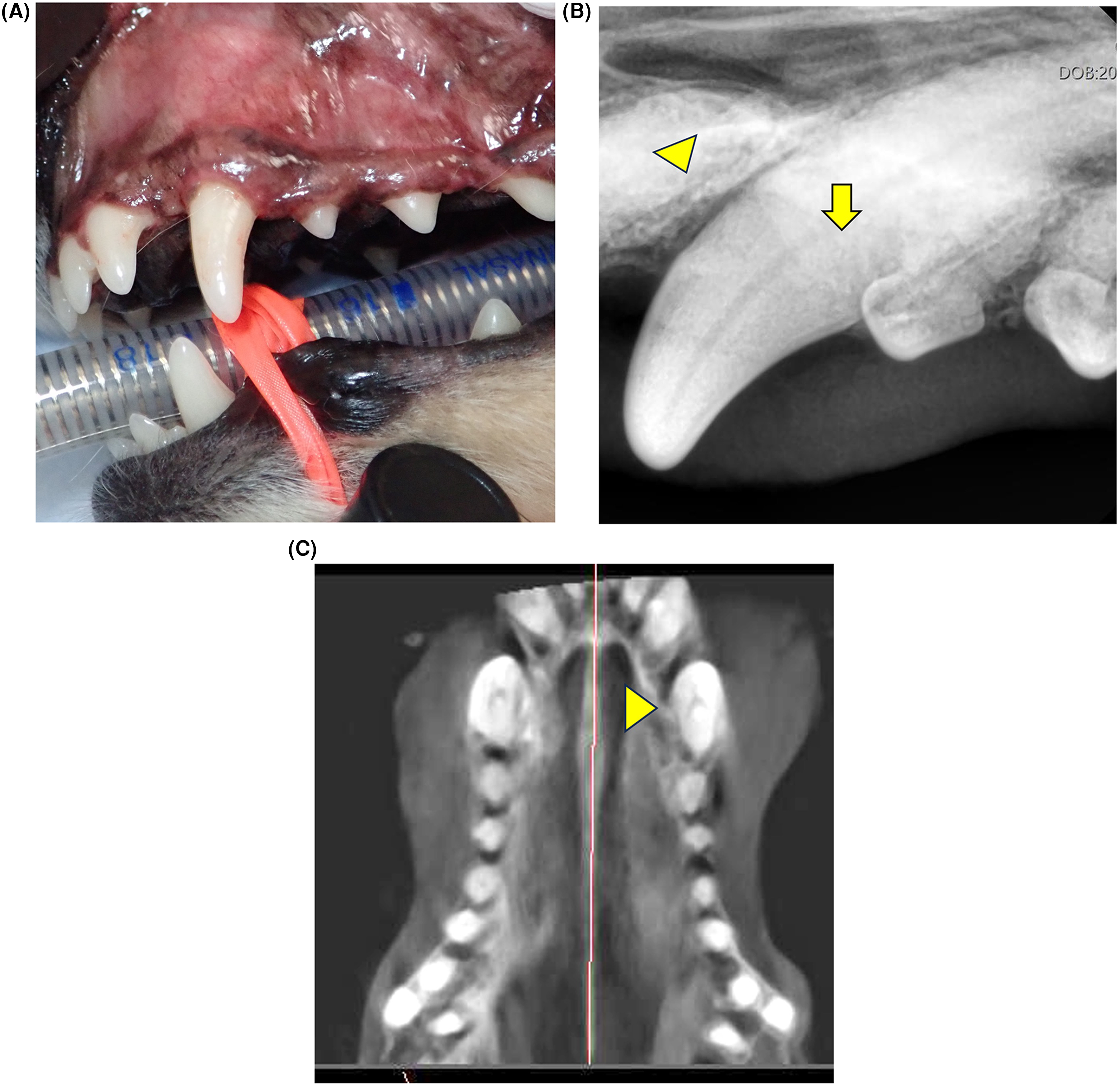
Oral examination in the conscious patient revealed normal occlusion with mild to moderate calculus accumulation (calculus index [CI]1-2) and localized gingival erythema and edema (Figure 1A). 18 The general condition of the patient was unremarkable, with no abnormalities detected on physical examination, blood analysis, or thoracic radiographs.
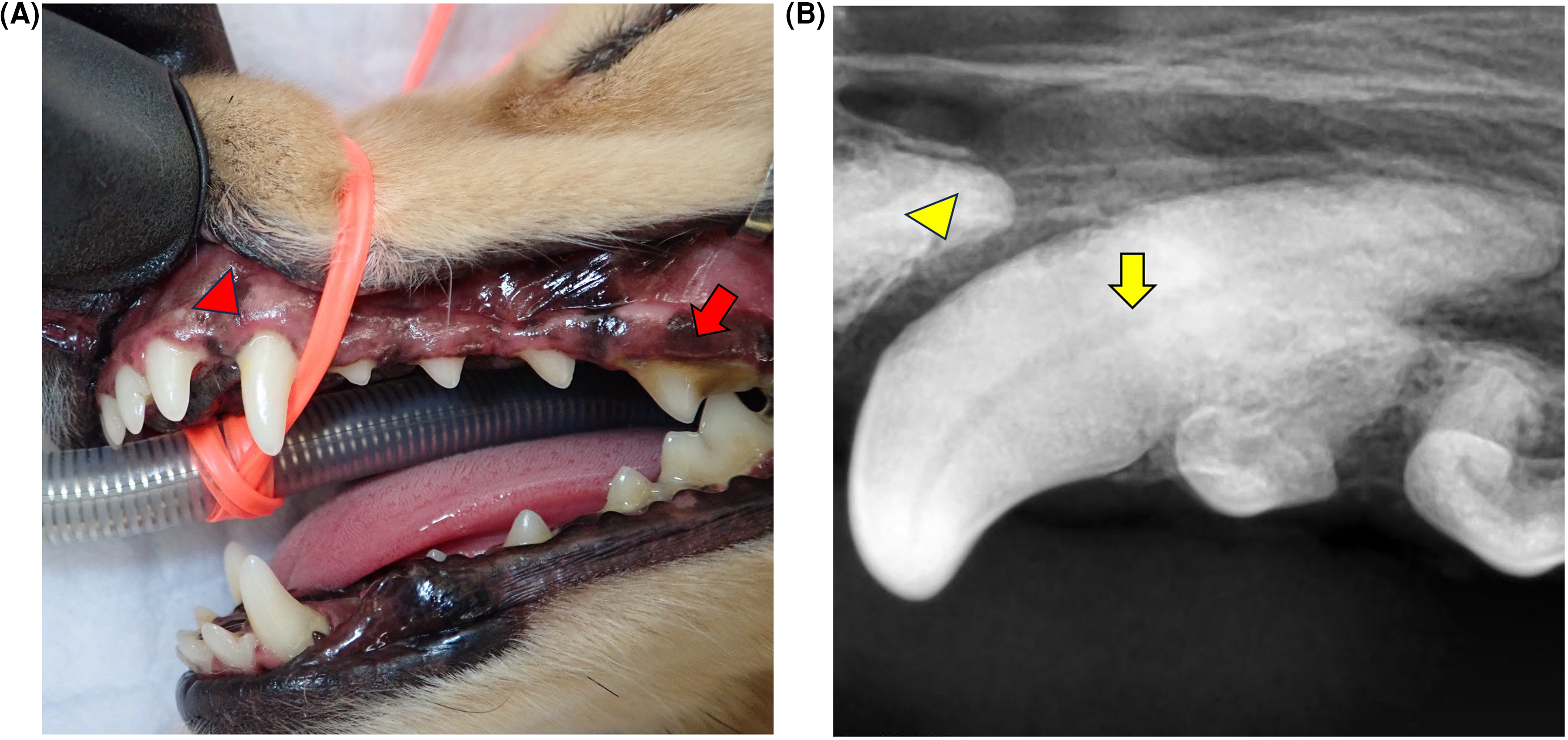
(A) Clinical photograph showing calculus on the left maxillary fourth premolar tooth (208) (arrow), and generalized maxillary gingival edema, including adjacent to the left maxillary canine tooth (204) (arrowhead). (B) Dental radiograph of the left rostral maxillary area showing mesial bone loss adjacent to the third incisor root (203) (arrowhead) and undetermined palatal bone loss adjacent to the canine tooth (204) (arrow).
The diagnostic and treatment plan included preoperative testing, general anesthesia, oral charting, dental radiography and scaling, which consisted of supragingival cleaning and subgingival debridement performed under magnification. A 24 G intravenous (IV) catheter was placed in the right cephalic vein, and lactated Ringer's solution was administered at 3 ml/kg/hr. The patient was pre-oxygenation with 100% oxygen by facemask. General anesthesia was induced with alfaxalonea (2 mg/kg) IV. Following a temporary reduction in respiratory drive sufficient to allow safe endotracheal intubation, along with loss of spontaneous respiration, eyelid reflex, and jaw tone, and sufficient suppression of the laryngeal reflex, a cuffed endotracheal tubeb was placed, and anesthesia was maintained with isofluranec in 100% oxygen. Prior to the dental procedure, cefovecind (8 mg/kg) was administered subcutaneously (SQ), meloxicame (0.2 mg/kg) SQ, tranexamic acidf (10 mg/kg) IV, and buprenorphineg (10 μg/kg) IV. Body temperature was maintained using a veterinary warming systemh and towels. Physiological parameters, including temperature, respiratory rate, heart rate, indirect blood pressure, electrocardiogram, pulse oximetry and capnography, were continuously monitored with a multiparameter monitori.
Periodontal examination was performed using a periodontal probej to measure pocket depth (PPD) and bleeding on probing (BOP) at six sites per tooth (mesio-buccal, mid-buccal, disto-buccal, mesio-palatal, mid-palatal, and disto-palatal). Intraoral radiographs were obtained with a dental portable x-ray unitk and a digital sensorl. Radiographic evaluation revealed mesial bone loss; however, the extent of palatal bone loss was unclear (Figure 1B).
A 10 mm periodontal pocket was detected on the palatal aspect of the left maxillary canine tooth (204), and communication with the nasal cavity was confirmed by a fluid infusion test using sterile saline. This method is consistent with the diagnostic principle of the water-passing test recently reported to improve detection of small oronasal fistulas that are not always identifiable on imaging. 19 All other teeth were stage 1 periodontitis with pocket depths <3 mm.18,20 Based on these findings, 204 was diagnosed with an oronasal fistula secondary to periodontal disease.
The owner was informed of the presence of the oronasal fistula and was offered either extraction or regenerative therapy after 3 months. The owner strongly preferred regenerative therapy. Initially, minimally invasive non-surgical technique (MINST) was performed on all teeth under 6.9× magnification using a surgical microscopem. Root debridement was performed on 204 with a subgingival ultrasonic insertn,o. The owner was advised that if home dental care was insufficient or if nasal discharge or sneezing worsened, regenerative therapy would be contraindicated, and extraction would be required.
Re-evaluation was intentionally scheduled 3 months later for several reasons: (1) initial MINST-based debridement can reduce local inflammation and improve soft-tissue conditions; (2) regenerative therapy is ideally performed after inflammation has been minimized through initial therapy; and (3) this interval allowed assessment of the owner's ability to maintain adequate plaque control at home. Although some clinical improvement was anticipated, the initial MINST session was not performed with the expectation of resolving the entire 10 mm defect. Rather, it was intended as phase-1 therapy to reduce inflammation, improve tissue quality, and allow more accurate reassessment of the defect prior to considering regenerative surgery. The 3-month interval also allowed the authors to evaluate whether the owner was able to maintain appropriate plaque control at home, which is essential for the success of periodontal regenerative therapy. In addition, the ultrasonic insert used during MINST provides effective debridement to depths of approximately 6–7 mm. While the tip can physically reach 8–9 mm pockets, anatomical constraints limit controlled instrumentation at these depths, and complete debridement of a 10 mm defect is not feasible. This mechanical limitation further supported the decision to perform subsequent flapless regenerative therapy.
Three months after the initial treatment, re-evaluation was performed under general anesthesia, including probing, intraoral radiography, and cone beam computed tomographyp (CBCT). The owner reported improvement in nasal discharge and sneezing. Mild calculus and plaque accumulation were observed on all teeth (Figure 2A). PPD on the palatal aspect of 204 remained at 10 mm (Figure 2B) and the oronasal fistula persisted. Radiographs still revealed mesial bone loss, while palatal bone loss remained indistinct, and overall improvement was insufficient (Figure 2C). CBCT images confirmed the loss of palatal bone and the palatal bone defect extended into the nasal cavity (Figure 2D), therefore the lesion represented a non-contained open defect and could not be classified using the conventional periodontal bone-wall system.
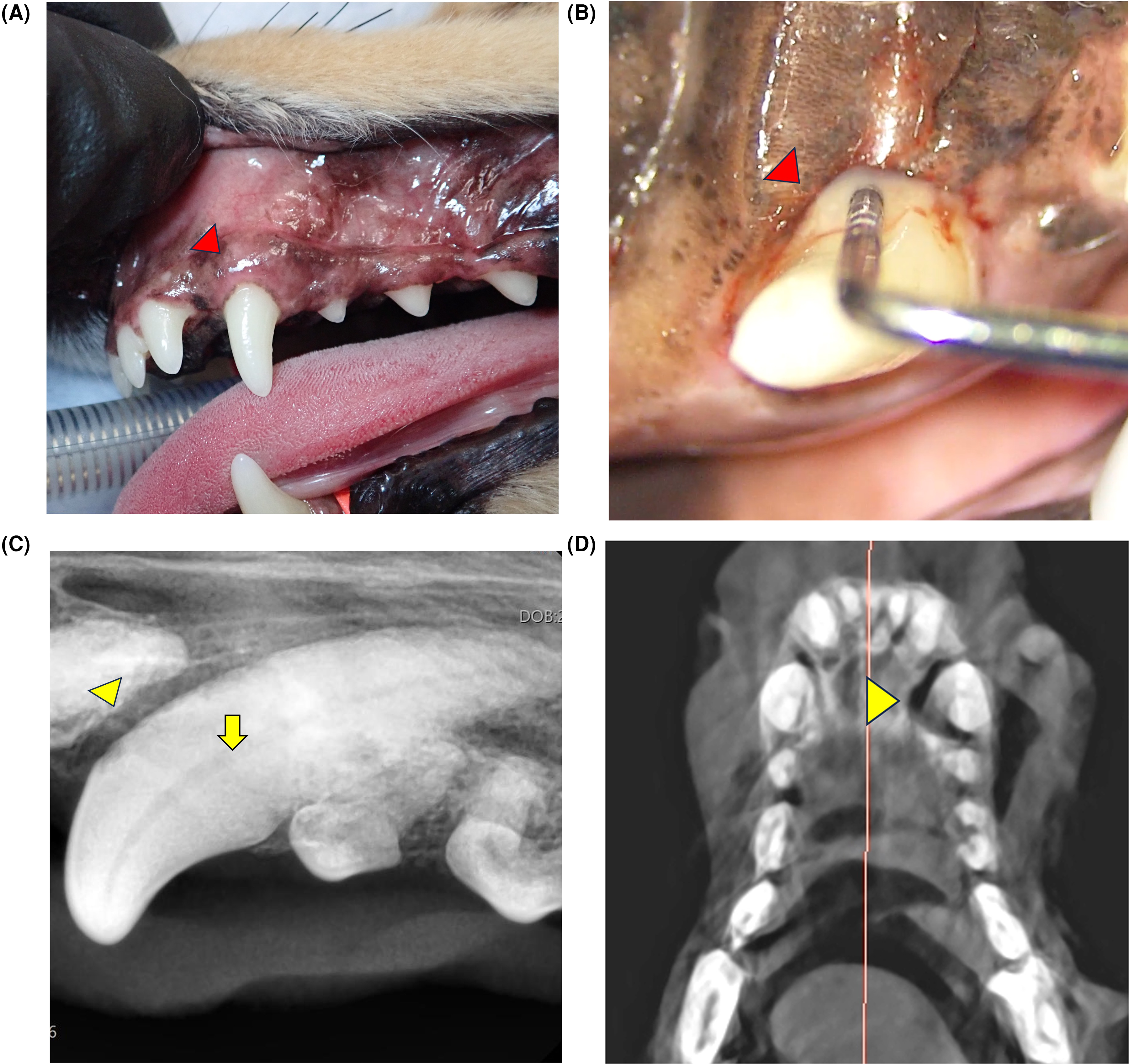
3-month visit post-initial treatment. (A) Clinical photograph showing generalized maxillary gingival edema, including adjacent to 204 (arrowhead). (B) Probing of the palatal aspect of 204 showing a 10 mm periodontal pocket with suppuration. (C) Dental radiograph the left rostral maxillary area showing continued mesial bone loss adjacent to 203 (arrowhead), while the extent of palatal bone loss was still indistinct adjacent to 204, showing insufficient improvement (arrow). (D) Dorsal view CBCT image showing the palatal bone defect of tooth 204 (arrowhead).
At the owner's request, regenerative therapy was performed on tooth 204. The lips were retracted with a clipq and the oral cavity was disinfected with chlorhexidine solutionr. Under 10.4× magnification with a surgical microscope, granulation tissue removal and root debridement were performed using an Er:YAG lasers and hand curettest (Figure 3A and B). The Er:YAG laser was used with a PSM600 T tip at 20 Hz, 50 mJ, with water irrigation. Enamel matrix derivativeu (EMD) was applied to the root surface, and a collagen matrixv was placed within the bony defect, positioned adjacent to the nasal mucosa associated with the oronasal fistula; however, direct contact between the collagen matrix and the nasal mucosa could not be visually confirmed. The site was closed by suturing the gingival tissues over the defect using interrupted sutures with 6-0 absorbable monofilament suture materialw without fixation of the collagen matrix itself. (Figure 3C–G). Recovery from anesthesia was uneventful, and the dog was monitored for one-hour post-extubation. The owner was instructed to avoid brushing the surgical site for 14 days, performing only gentle wiping with gauze, while brushing other teeth could resume after 24 h. A soft diet was recommended for 14 days.
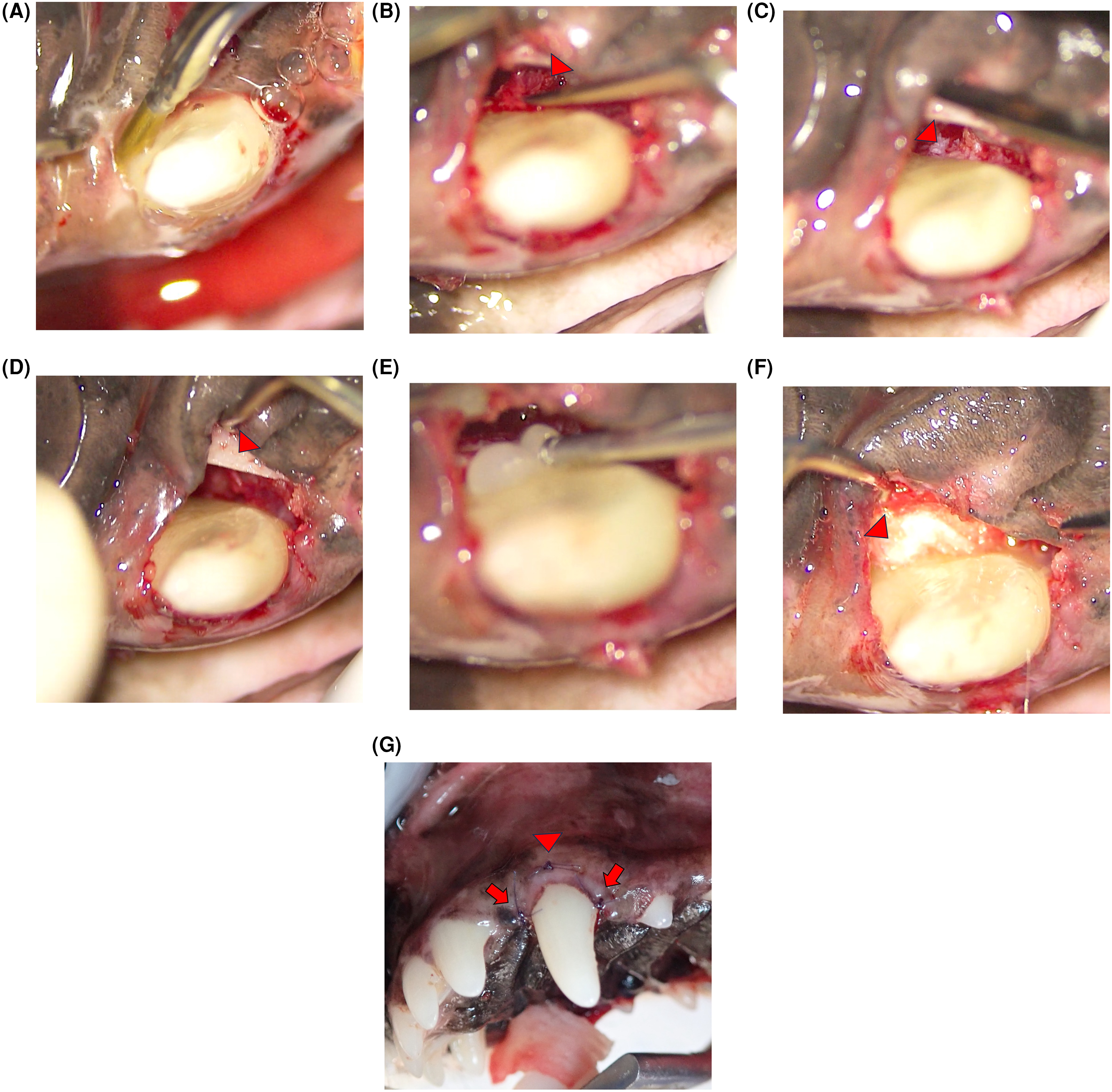
Clinical procedure using periodontal regenerative therapy to treat the 204 periodontal pocket. (A) Er:YAG laser irradiation is being used for debridement of granulation tissue and root surface cleaning. (B) Residual granulation tissue (arrowhead) is being removed with a hand curette. (C) The base of the bony defect (arrowhead) where communication with the nasal cavity was observed. (D) Bony defect after granulation tissue removal and root surface cleaning, showing visible palatal bone (arrowhead). (E) Application of enamel matrix derivative (EMD) to the root surface. (F) Placement of collagen-based socket protection material (arrowhead) into the bony defect. (G) Closure of the palatal flap using palatal and buccal horizontal mattress sutures (arrowheads) and simple interrupted sutures at the mesial and distal sites (arrows).
At 14 days postoperatively, the owner reported that epistaxis, nasal discharge, and sneezing had resolved by day 2. Clinical examination revealed no swelling, calculus, or plaque, and sutures had resorbed. Brushing and dry food were resumed after this visit.
Three months after regenerative therapy, the dog underwent general anesthesia for an unrelated condition, and follow-up evaluation was performed. Probing revealed a palatal PPD of 4 mm with no BOP (Figure 4A and B). CBCT confirmed increased radiopacity at the site of the previous bony defect (Figure 4C). The owner was instructed to continue brushing and feeding dry food.
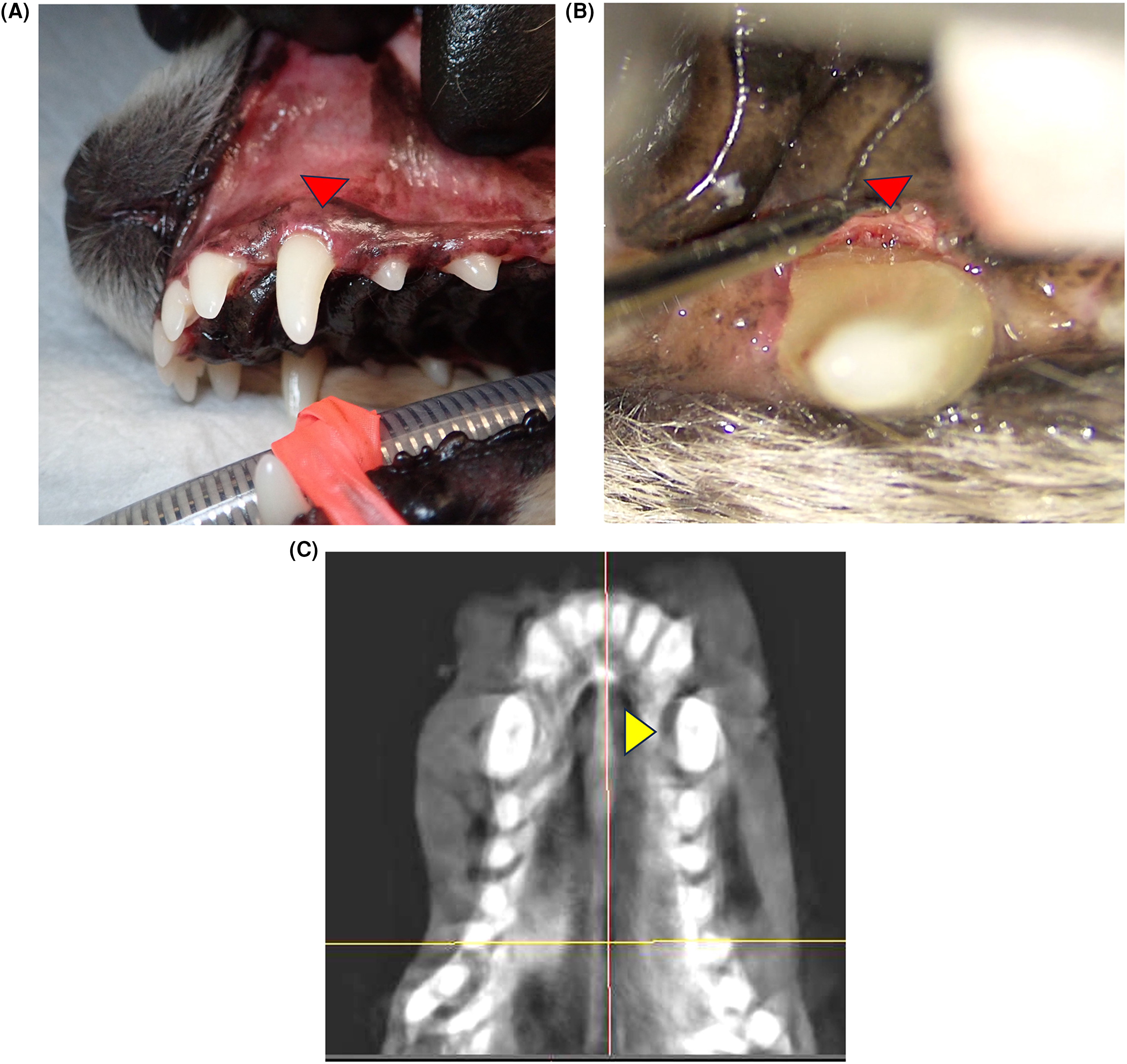
3-month visit post-regenerative therapy. (A) Clinical appearance showing mild gingival edema adjacent to the 204 (arrowhead). (B) Clinical appearance showing bone regeneration on the palatal aspect of 204 (arrowhead). (C) Dorsal view CBCT image showing increased radiopacity of the palatal bone adjacent to 204 (arrowhead).
At 8 months postoperatively, re-evaluation was again performed under general anesthesia. Probing revealed a palatal PPD of 2 mm with no BOP (Figure 5A and B). CBCT confirmed further increased radiopacity consistent with bone regeneration (Figure 5C). Recovery was uneventful, and the owner was advised to maintain annual periodontal re-evaluations with professional cleaning.
8-month visit post-regenerative therapy. (A) Clinical appearance showing minimal calculus deposition and gingival edema of 204. (B) Dental radiograph showing bone regeneration on the mesial aspect of 203 (arrowhead), while the palatal bone defect remained indistinct on 204 (arrow). (C) Dorsal view CBCT image showing further increase in radiopacity of the palatal bone adjacent to 204 (arrowhead).
Discussion
This case describes a rare instance in which a maxillary canine tooth with an oronasal fistula secondary to periodontal disease was successfully preserved through regenerative therapy, rather than extraction, which has traditionally been considered the first-line treatment. 3 Maxillary canines play a critical role in prehension, occlusal stability, and maintenance of facial support.2 Their loss may lead to a significant reduction in quality of life. Thus, this case is clinically meaningful as it demonstrates the potential for canine preservation as an alternative therapeutic option.
At the initial treatment, root debridement with MINST failed to reduce the 10 mm PPD. This was likely due to incomplete removal of granulation tissue, calculus, and plaque. Subsequent therapy incorporating an Er:YAG laser resulted in a marked reduction in PPD. The laser provided access to areas that are difficult to reach with conventional instruments, enabling more thorough removal of residual granulation tissue. In this case, MINST alone was insufficient, whereas the adjunctive use of Er:YAG laser achieved clinical improvement. While MINST offers the advantages of minimal invasiveness and wound protection, it can be limited in cases with fistulas or deep bony defects. The present case highlights both these limitations and the potential benefits of a MINST-assisted flapless regenerative therapy, in which the subsequent laser-assisted procedure complemented the initial MINST session. In particular, the ablative effect of the Er:YAG laser facilitated treatment of deep granulation tissue and constricted sites, which likely contributed to successful healing.
The surgical microscope also played a pivotal role in achieving treatment success. The magnified view allowed precise removal of calculus and granulation tissue around the fistula without incision, while promoting blood clot stability. Although flap surgery can also be used to manage fistulas, it carries the risks of greater tissue trauma, flap dehiscence, and impaired wound stability. By contrast, MINST provides a narrower surgical field but permits adequate debridement while maintaining the benefits of minimal invasiveness and wound protection. In the present case, these advantages contributed to improving tissue conditions during the initial session, which subsequently facilitated the flapless regenerative procedure.
Dental radiographs and CBCT are valuable diagnostic imaging tools for assessing bony defects and evaluating treatment outcomes. However, they cannot definitively diagnose an oronasal fistula. 21 Confirmation requires fluid leakage testing or direct visualization. A recent study reported the diagnostic utility of a fluorescent water-passing test for detecting small fistulas, which may improve diagnostic accuracy. 19 In this case, the size and morphology of the fistula could not be fully determined preoperatively by imaging, and intraoperative findings guided case selection. This limitation suggests that large fistulas may not be suitable for a flapless regenerative approach, as the defect size may exceed what can be adequately managed without flap elevation.
The Er:YAG laser served as an essential adjunct in this case. Beyond removal of granulation tissue and calculus, it offers multiple benefits, including root surface smoothing, bactericidal effects, stabilization of the blood clot, and stimulation of regenerative factor expression.10–14 One study demonstrated in a canine model that Er:YAG laser use promotes new bone formation, 15 which is consistent with the favorable outcome observed in this case. Although the absence of a control group precludes definitive conclusions, the clinical improvement observed during the laser-assisted flapless regenerative procedure, in contrast to the lack of improvement after the initial MINST session, strongly suggests the effectiveness of this adjunctive approach. EMD and a collagen scaffold were also used in this case. EMD has been shown to promote cementum and periodontal ligament regeneration and is widely applied in human dentistry. In veterinary dentistry, experimental studies with trafermin 16 and clinical reports of EMD combined with bone grafts in canine teeth 17 have also demonstrated efficacy. In this case, PPD reduction and resolution of BOP were observed by 3 months postoperatively, and CBCT at 8 months revealed bone-like radiopacity at the defect site. These findings indicate that the multimodal flapless regenerative approach, including laser and EMD, contributed to the favorable outcome. In addition, the collagen matrix used in this case functions as a scaffold consistent with principles of guided tissue regeneration (GTR), supporting space maintenance and selective cell repopulation. Although our procedure was performed in a flapless manner rather than as a traditional GTR technique, the biological concept underlying the collagen scaffold reflects a GTR-related regenerative approach commonly recognized in veterinary dentistry. 22
This report is limited by its nature as a single case with only 8 months of follow-up. In addition, the absence of histologic evaluation prevents definitive confirmation of the nature and extent of true bone regeneration at the defect site, which is a common limitation in reports involving guided tissue regeneration techniques. Long-term prognosis and recurrence risk remain uncertain. Furthermore, the applicability of this flapless regenerative approach may be restricted by the size and morphology of a fistula, which cannot always be predicted preoperatively. Future studies involving case accumulation, long-term monitoring, and comparative trials with conventional therapies are needed to define indications and limitations of this approach.
In conclusion, this case demonstrates the novelty of preserving a canine tooth affected by oronasal fistula as an alternative to extraction. The combination of microscope-assisted minimally invasive debridement and a laser-assisted flapless regenerative procedure enabled precise, incision-free management of the fistula and periodontal regeneration. This MINST-assisted flapless regenerative approach represents a promising new therapeutic strategy in veterinary dentistry, with potential for broader clinical application in the future.
Conclusion
In this case, periodontal regenerative therapy conducted as a MINST-assisted flapless regenerative therapy enabled preservation of a maxillary canine tooth affected by an oronasal fistula, thereby avoiding extraction. While the initial MINST session alone did not provide sufficient improvement, the subsequent flapless regenerative procedure using an Er:YAG laser facilitated the removal of residual granulation tissue and calculus, which contributed to periodontal healing. This case suggests that such a combined approach may serve as a novel therapeutic alternative to extraction, which has traditionally been regarded as the first-line treatment. Further case accumulation and long-term follow-up studies are warranted to clarify the indications and clinical effectiveness of this technique.
Materials
Alfaxan Multidose, Meiji Animal Health Co., Ltd, Kumamoto, Japan
Wire Reinforced Endotracheal Tube, Fuji Systems Corporation, Tokyo, Japan
Isoflurane for animal, MSD Animal Health Co., Ltd, Tokyo, Japan
Convenia, Zoetis Japan Inc., Tokyo, Japan
Meloxirin® Injection 0.5%, Fujita Pharmaceutical Co., Ltd, Tokyo, Japan
Transamin® Injection 10%, Daiichi Sankyo Co., Ltd, Tokyo, Japan
Lepetan® Injection 0.2 mg/mL, Otsuka Pharmaceutical Co., Ltd, Tokyo, Japan
Cocoon™ Warming System, Nihon Kohden Corporation, Tokyo, Japan
Bio-Scope AM140, Fukuda M-E Co., Ltd, Tokyo, Japan
CPUNC15, Hu-Friedy Group, Tokyo, Japan
EzRay Air, RayVision, Saitama, Japan
Intraoral CMOS sensor, RayVision, Saitama, Japan
BrightVision® LED 5103 Surgical Microscope, Pentron Japan Inc, Tokyo, Japan
Cavitron® Select™ SPS Ultrasonic Scale, DENTSPLY Sirona K.K, Tokyo, Japan
. Cavitron® THINsert® Ultrasonic Insert FITGRIP, DENTSPLY Sirona K.K, Tokyo, Japan
GREEN18, Vatechjapan, Tokyo, Japan
Raney Scalp Clip, Mizuho Co., Ltd, Tokyo, Japan
0.5% Glucodine W Water, Nichi-Iko Pharmaceutical Co., Ltd, Toyama, Japan
Erwin AdvErL EVO Er:YAG Laser, J. Morita Mfg. Corp, Kyoto, Japan
NEW O・K Micro Exca, Seto Seisakusho K.K, Ibasaki, Japan
Emdogain® Straumann Japan K.K, Tokyo, Japan
TERUPLUG® GC Corporation, Tokyo, Japan
6-0Monosyn, B.Braun Aesculap Japan Co., Ltd, Tokyo, Japan
Footnotes
Ethics Statement
This case report describes a clinical patient treated as part of routine veterinary care. No experimental procedures were performed. Informed consent was obtained from the owner for all diagnostic and therapeutic procedures, as well as for the publication of anonymized clinical data and images.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
