Abstract
Dental home care of pets plays a critical role in preventing periodontal disease. Active methods of dental home care such as tooth brushing are recommended along with oral hygiene measures that can help reduce dental plaque. The study evaluated the effectiveness of a new topical oral gel (TOG) in controlling plaque accumulation in dogs. After scaling and polishing, 32 dogs were randomly assigned to either a control group with no further oral hygiene care or the TOG group that received an application of the oral gel once a day for 30 days. All dogs were fed the same diet during the trial. Five parameters were evaluated in 9 target teeth on days 0 and 30: periodontal disease index (PDI), calculus index (CI), gingival bleeding index (GBI), plaque index, and oral health index (OHI). On day 30, comparison between the groups showed no differences for PDI and CI. However, OHI, PDI, and GBI were significantly lower in the TOG group (P < .05). Furthermore, OHI and GBI decreased over time in the TOG group (P < .05) and not in the control group. These results show that once-daily oral applications of TOG, without mechanical action, reduced the accumulation of dental plaque in dogs.
Keywords
Introduction
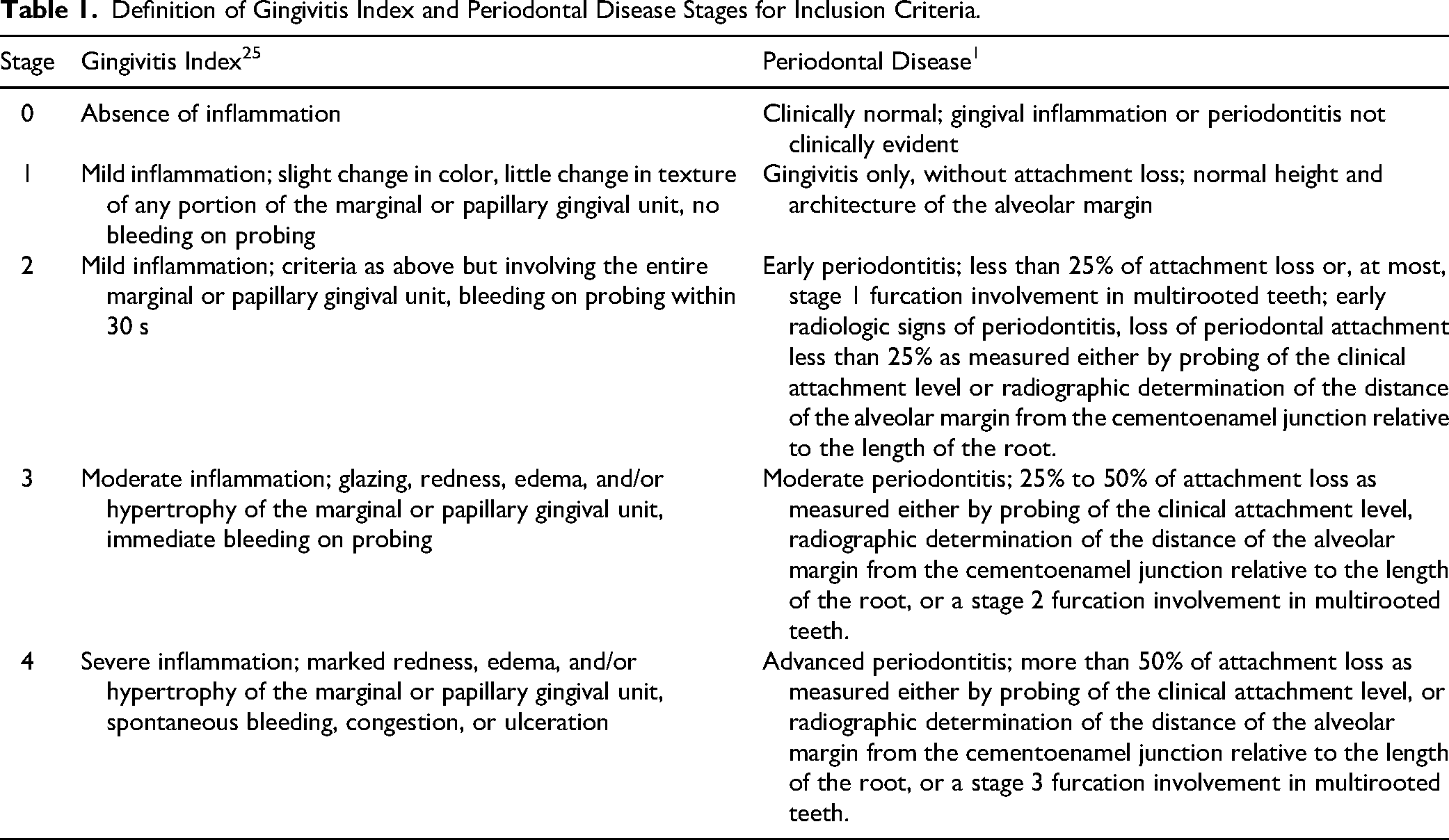
Periodontal disease (PD) is a chronic infection and inflammation of the gingiva and periodontium.1–4 Gingivitis is the first and reversible stage of PD, while early, moderate, and then advanced periodontitis are the next stages of the disease associated with progressive irreversibility.2,5–7 The clinical features of the early stages of PD can include halitosis, red, swollen and bleeding gums, and uncomfortable or painful chewing. Advanced periodontitis is characterized by recessed gums and destruction of supporting alveolar bone leading to tooth mobility and their eventual loss. 8
PD is the most common clinical problem in dogs, with a reported prevalence ranging from 9.3% to 80% in dogs depending on the geographic region and reaching 82% in dogs aged 6 to 8 years, 96% in dogs aged 12 to 14 years, and 100% in poodles older than 4 years.2,3,9,10 PD is also a significant oral health concern in cats. It affects around 50% of cats over 4 years of age and 93% of cats over 8 years of age.9,11
Variables that can place an individual at risk for developing PD include genetic, microbiological, nutritional, and environmental risk factors. 10 The prevalence of PD has also been reported to increase significantly with age, which was also positively correlated with the severity of the disease.2,3,6 Furthermore, breed was shown to play a significant role, with more than 85% of small breed dogs reportedly having PD and small dogs generally developing PD at an earlier age compared to larger dogs.3,6,9 Additional risk factors for the diagnosis of PD also include excess bodyweight and the time between professional dental cleaning procedures. 6
Bacterial plaque is the primary etiologic factor associated with periodontitis. Dental plaque is a result of oral biofilm dysbiosis.4,10,12 These bacteria can infiltrate the subgingival space and produce several metabolites, such as ammonia and volatile sulfur compounds, which lead to halitosis. Moreover, bacterial endotoxins and proteolytic enzymes promote host immune and inflammatory responses.4,10,12,13 The persistent host inflammatory response against dental plaque bacterial aggression leads to progression of PD.8,10,11 Anaerobic Gram-negative bacteria including Treponema denticola, Treponema putidum, Porphyromonas gulae, Porphyromonas gingivalis, and Prevotella nigrescens are suspected to be involved in the development of PD in dogs and cats.14,15 Dental plaque microbiota is, however, highly complex and appears to be distinct according to the PD stage.10,16
PD is a local disease of the oral cavity but can have local and systemic impacts on health and well-being. Local consequences include oronasal fistulas, pulp diseases, pathological fractures, ocular problems, osteomyelitis, and an increased incidence of oral cancer. 17 Associations between PD and a variety of degenerative and inflammatory consequences, such as renal, hepatic, pulmonary or cardiac disease, osteoporosis, adverse pregnancy effects, and diabetes mellitus, have been identified in pets.3,9,11,13,17 The systemic consequences of PD can be explained by the potential spread of bacteria, toxins, and inflammatory mediators from dental plaque to the bloodstream after chewing, tooth brushing, and dental or surgical interventions.9,10,13
Despite its high prevalence, PD is considered to be severely underdiagnosed in dogs and cats.3,6,9,11 Consequently, PD is reported as the most undertreated animal health problem in pets. 17 Prevention strategies of PD rely on the control of dental plaque accumulation 4 by using daily oral hygiene methods including tooth brushing or oral products. 3 However, the evidence for efficacy of the many therapeutic and preventive interventions is highly variable.10,18 Furthermore, a recent survey showed that approximately half of owners never brush their pet's teeth. 18 These findings underline the need for new preventive solutions to increase pet owner compliance with dental home care.19,20
In this context, an easy-to-use topical oral gel (TOG)a has been developed that combines enzyme complexes, antiadhesive polysaccharides, and liquorice extracts. This evidence-based gel formulation was developed to support healthy mouths and to soothe gums in pets by daily applications at home. Bromelain, an enzyme naturally present in pineapples, has been used as a microbial balance agent to manage periodontitis. 21 Peroxidase enzymes have been shown to strengthen natural defences. 22 Polysaccharides are known to be capable of inhibiting biofilm formation 23 and in addition, the inflammatory modulation, antiadhesive, and microbial balance properties of liquorice have been shown to have beneficial effects in oral diseases such as periodontitis. 8
This study aimed to evaluate the efficacy of a new dental care gel to control plaque and calculus accumulation in dogs.
Materials and Methods
Dog Selection
The animals were selected according to the guidelines of the Veterinary Oral Health Council (VOHC) for trials testing compounds for PD control. 24 They were recruited from the patients of Veterinary Clinic Arka, Kraków, Poland a veterinary referral center (dentistry), after obtaining informed consent from the owners. The study was conducted according to standard noninvasive veterinary procedures that did not require Ethical Committee approval.
Healthy client-owned dogs weighing less than 30 kg with mild to moderate gingivitis were enrolled in the study. None of the dogs received any active nor passive homecare other than the TOG. Mild to moderate gingivitis was defined as a gingivitis index ≤3 and a PD stage between 0 and 3 as described in Table 1.1,25 All teeth required as per the VOHC guidelines (maxillary third incisor, canine, third premolar, fourth premolar and first molar and mandibular canine, third premolar, fourth premolar and first molar bilaterally) were present. Advanced periodontal status (PD stage >4), oral mucosal ulcerations, systemic disease, and pregnancy were excluding criteria. 24
Definition of Gingivitis Index and Periodontal Disease Stages for Inclusion Criteria.
Study Design
All procedures were conducted in a registered and approved European Veterinary Dental College training center under the supervision of a Board-Certified veterinary dental specialist.
On day 0 (day of inclusion), all animals received a general physical examination. Blood and urine samples were collected for immediate hematology and urinalysis. Using recommended doses, medetomidineb (10-20 μg/kg) and butorphanolc (0.1 mg/kg) were administered intramuscularly (IM) for sedation, and preoxygenation was administered using an oxygen mask. The animals then received propofold (1-3 mg/kg) intravenously for induction of general anesthesia and endotracheal intubation. General anesthesia was maintained using inhalant isofluranee and oxygenf for the duration of the dental procedure. Intravenous fluid therapy was administered using Ringer's solutiong (4 mL/kg/h). A comprehensive oral health assessment and treatment (COHAT) was performed under supervision of a Board-Certified veterinary dental specialist, and included oral examination and charting, full-mouth radiography, and professional dental cleaning (teeth descaling and polishing). A disclosing solutionh was used to ensure that plaque and calculus were completely removed. If plaque or calculus were still discernible, the scaling and polishing procedures were repeated.
Treatment Groups
The dogs were randomly assigned to 2 treatment groups on D0 after scaling. Randomization was generated with professional software.i The treatment group received the TOG containing an enzymatic complex, consisting of bromelain and peroxidase. The peroxidase system was composed of amyloglucosidase and glucose oxidase. The gel also contained antiadhesive polysaccharides (rhamnose, glucose, and glucuronic acid) and liquorice extracts. Owners applied the gel once daily between the lips and gums with the soft tube applicator, without performing any mechanical cleaning action, and then the cheeks were gently massaged bilaterally. No other means of oral hygiene was provided to these dogs during the study period. The control group received no oral hygiene care. All animals were fed the same small kibble of dry dietj during the study. They received water ad libitum and their lifestyle was unchanged during the study.
Assessment of Oral Health Status
Assessment of the oral health status was performed by the same Board-Certified veterinary dental specialist on D0 and D30 based on the scoring of the entire buccal surface of the 9 VOHC required teeth.
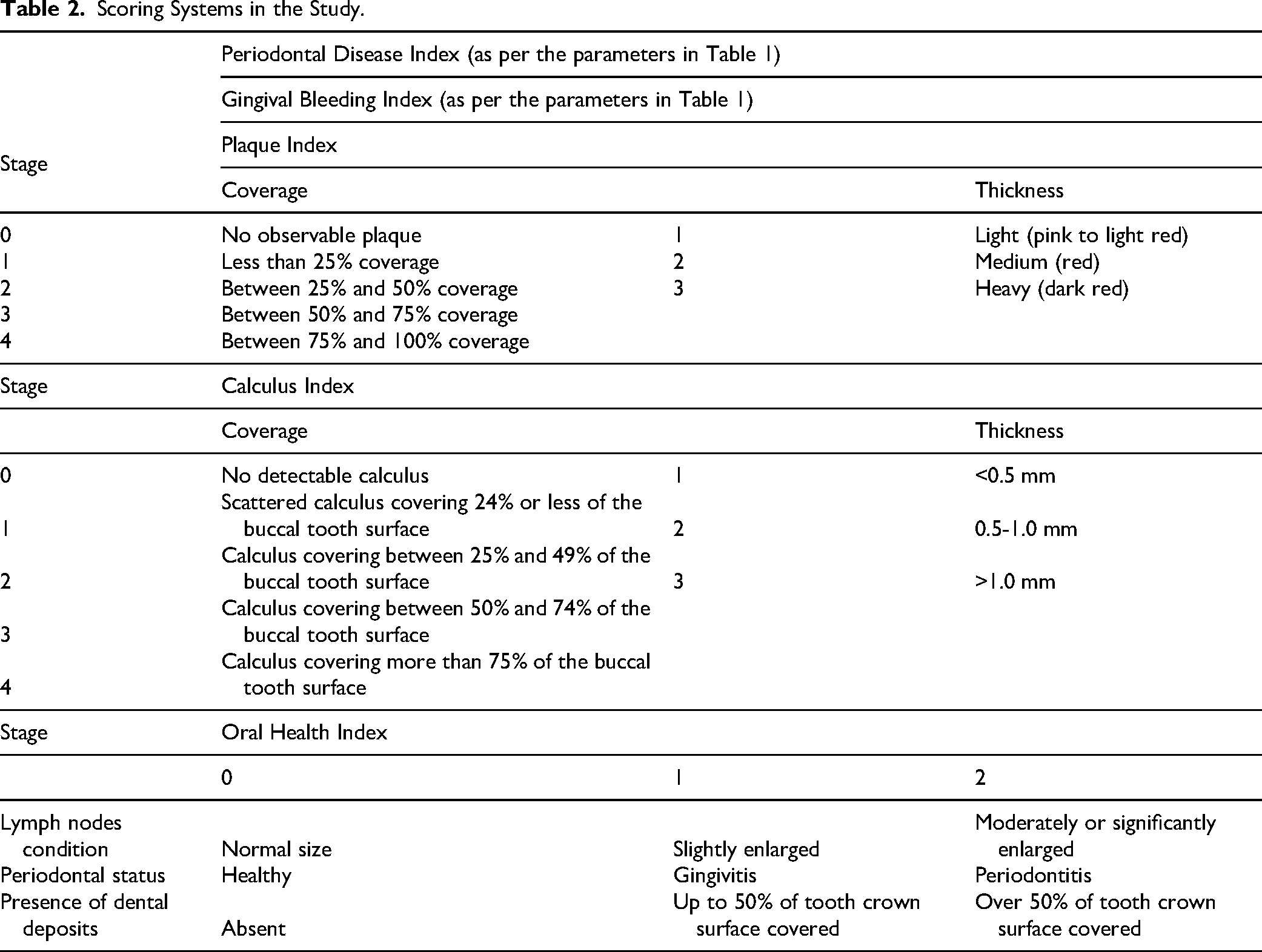
Five parameters were evaluated (Table 2). The PD was evaluated. 1 The gingival bleeding index (GBI), a sensitive marker of local irritation, was scored on a scale of 0 to 3 to assess gel tolerance. 26 The teeth were then stained with the disclosing solution for the assessment of plaque index, which was calculated in accordance with the modified Logan and Boyce method by multiplying the plaque coverage and thickness scores of the plaque. 27 The plaque was then brushed from the surface of the teeth prior to the assessment of the calculus index (CI). A dental explorer was used to determine the edge of the calculus. The coverage of calculus was scored according to a previous study design. 28 Table 2 describes the criteria for evaluating calculus index on a 0 to 4 scale. Finally, the oral health index (OHI) was determined. The OHI is a general oral health assessment index defined as the sum of the scores obtained for the 3 parameters: mandibular lymph nodes status, periodontal tissue status, and the degree of dental deposits present on the tooth surface. Zero points indicated optimal oral health, and 6 points indicated the poorest oral health (Table 2).29,30
Scoring Systems in the Study.
On D30, the animals were sedated as previously described for D0, including additional preoxygenation. A blinded assessment of oral health was performed after sedation by the same investigator in all dogs (treated and control groups). In the treated group, this assessment was performed within 24 ± 3 h after the last application of TOG.
After completion of the oral health evaluation, the animals were allocated to an intensive care unit cage with oxygen supply and received atipamezolek (using recommended dose) IM for sedation reversal. Body weight, body condition score, daily food intake, and side effects were also monitored throughout the study period.
Statistical Analysis
The Gaussian distribution of the registered dental health parameters was evaluated using the Shapiro-1814k normality test. Intergroup comparisons at D30 were performed using an unpaired t-test when normal distribution was confirmed or if not, then a Mann–Whitney Wilcoxon test was used. A sign test was used for intragroup comparisons between D0 and D30.l The dog was the experimental unit and the statistical significance was established at P < .05.
Results
Thirty-three dogs were selected for the study. One dog was excluded due to having a PD stage greater than 2. The remaining 32 dogs were randomly divided into 2 groups of 16 dogs. Thirty purebred dogs—20 Jack Russell Terriers, 6 Miniature Schnauzers, 3 Chihuahuas, and 1 Miniature Pinscher—were included in the study. The 2 remaining dogs were crossbred. Twenty-nine dogs were female (91%). The mean age was 4.3 ± 2.5 years (range 1.0-10.0 years) and the mean body weight was 6.3 ± 2.5 kg (range 2.5-15.0 kg). The body condition score was 5 out of 9 in 31 dogs and 6 out of 9 in 1 dog and these remained stable during the study. The characteristics of the study population and the dental health parameters were not significantly different in the 2 treatment groups at D0.
The mean values for the 5 dental health parameters at D0 and D30 are presented in Table 3. Dogs receiving TOG once daily had a significantly reduced mean OHI (−53%, P = 9.11 × 10−7), PI (−23.9%, P = .0268), and GBI (−61%, P = .0241) compared to the control group at D30. However, CI and PD were similar in both treatment groups at D30. Intragroup comparisons between D0 and D30 showed no significant differences in the control group, whereas the mean OHI and GBI significantly decreased over time in the TOG group (P = .0003 and P = .0162, respectively). Furthermore, the GBI values indicated that TOG use did not worsen the oral health status measured by gingivitis. The mean bodyweight of the dogs was not significantly different on D30 between treatment groups.
Mean Scores of Dental Health Parameters in Each Treatment Group on Day 0 and Day 30.
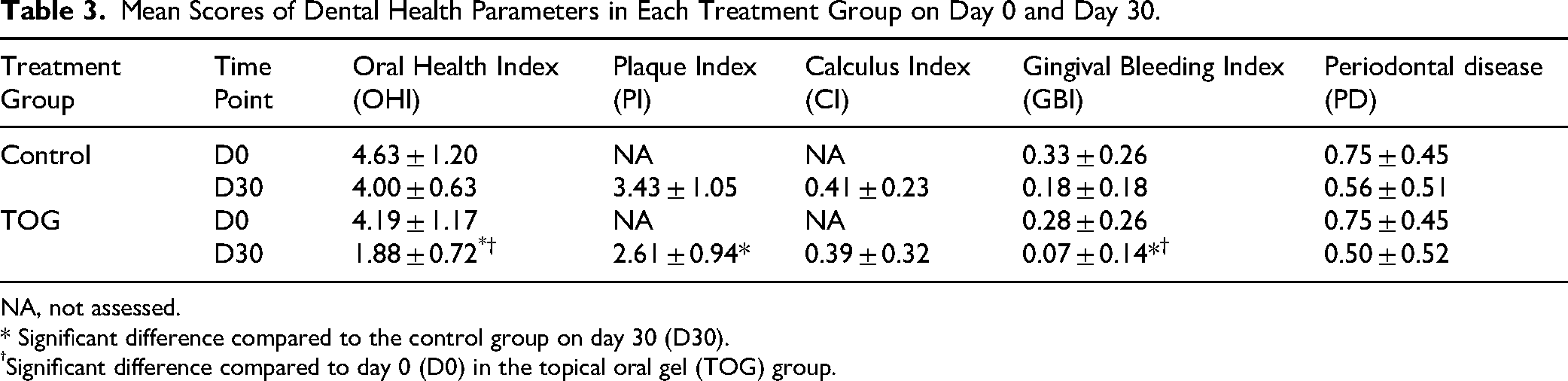
NA, not assessed.
* Significant difference compared to the control group on day 30 (D30).
Significant difference compared to day 0 (D0) in the topical oral gel (TOG) group.
Discussion
The results of this study showed a significant impact on OHI and PI on D30 compared to the control group and on OHI and GBI over time (D0 versus D30) after daily administration of TOG. These observations can be explained by the combined effects of the TOG components.
The absence of chlorhexidine in the formulation is an important feature, addressing potential concerns of side effects such as staining and altered taste reported with chlorhexidine use in oral products. 31 The combination of polysaccharides, peroxidase enzymes, and liquorice extracts contributes to maintaining oral health.
In humans, polysaccharides have been demonstrated to have inhibiting effects on biofilm formation and therefore to help maintain the balance of the oral microbiota.23,32 Their mode of action is not clearly understood since it has been shown that they have no bacteriostatic or bactericidal activity. 23 It has been proposed that their antibiofilm activity involves surfactants that interfere with and break down cell membrane components. 23 A second hypothesis suggests that polysaccharides can act as signaling molecules that modulate the gene expression of bacteria. 23 A third possible mode of action is the inhibition of lectins or sugar-binding proteins, fimbriae and pili tip adhesins on bacterial surfaces. 23 Peroxidase enzymes are innate defense factors in human and canine saliva.22,33,34 These proteins contribute to control of the formation of dental plaque and regulation of the oral bacterial microflora. 34 They interfere with bacterial glucose uptake or its metabolism, thus limiting bacterial growth 22 and they promote the production of hypothiocyanite, a potent antimicrobial oxidant.10,34 Bromelain, a group of proteolytic enzymes naturally present in the fruit and stem of the pineapple plant, has been shown to have in vitro antibacterial efficacy against aerobic and anaerobic human periodontal microorganisms. 21 In another in vitro study, bromelain showed a stain removal effect by removing the protein portion of the dental pellicle that forms on the surface of human teeth over time. 35 Bromelain has also been widely used as an anti-inflammatory agent. 21 The antiadhesive, antimicrobial, anti-inflammatory, and immunoregulatory properties of liquorice have shown beneficial effects in the prevention and treatment of human oral diseases such as periodontitis.4,8 The antibacterial effect is explained by the ability of its root polysaccharides to reduce bacterial adhesion. 8 Liquorice also inhibits the inflammatory cytokines and matrix metalloproteinases present in high concentrations in macrophages from inflamed gingival tissues. 8 Furthermore, liquorice has been proven to prevent inflammatory bone loss in mice by attenuating osteoclast activity. 8 Other studies show that liquorice extracts and bioactive ingredients in liquorice have effects on the host immune response involved in oral–dental diseases. 4 In conclusion, the effects of TOG observed in the present study on various parameters of oral health can be explained by the complementary properties of its components that inhibit both the formation of plaque and the host immune response, the 2 main etiological factors of periodontitis.
Dental home care is a crucial aspect of PD prevention because, in the absence of effective methods to maintain oral hygiene and periodontal health, dental plaque accumulates on the teeth crown surface within 24 h after dental cleaning and microbial counts return to pretreatment levels within a week. 36 In pets, as in humans, daily tooth brushing is regarded as the gold standard for the prevention of PD progression,3,20 however, survey results show that fewer than 5% of the owners effectively brush their pet's teeth daily. 19 Therefore chemical plaque controls, including agents to prevent plaque biofilm production, especially in areas of the mouth that are less accessible to tooth brushing, remain a necessary complement to mechanical plaque control. 4 Additionally, the generally good persistence of oral products in the oral cavity enables maintenance of oral hygiene between brushings. 4 In order to improve dental home-care compliance, the TOG tested in this study has been developed with an easy-to-use tube with a premounted applicator that can be inserted directly into the mouth.
Several factors might have affected or influenced the findings of the present study. First, fewer than 20 animals were enrolled in each treatment group. The small number of dogs was due to the single centrally located design of the present study. As all cases were managed at a referral center by an experienced investigator specializing in veterinary dentistry this ensured that the inclusion criteria and approach were consistent. A larger number of animals could have maximized the chances of avoiding type II error and allowed identification of more significant relationships or connections between TOG administration and oral health parameters in this study. Another limitation of this trial might have been the variations, however small, in the characteristics of the study population that are inherently associated with a prospective clinical study using client-owned pets. Inclusion criteria included a bodyweight of less than 30 kg, and no statistical significance was found on D0 between the groups regarding the breeds and mean age and bodyweight. Nevertheless, a stratified randomization by age, breed and breed size might have limited the imbalance between treatment groups regarding known risk factors for periodontitis in dogs. Finally, a longer follow-up period might have been useful to investigate the long-term effectiveness of TOG. A longer study duration would also have enabled the time to relapse to be evaluated, i.e. the time interval between the first COHAT and the next professional dental cleaning procedure being required. Despite these limitations, this study showed significant improvement in oral health after daily application with the TOG over a 30-day period.
Conclusion
The daily oral application of a topical gel containing an enzymatic complex, antiadhesive polysaccharides, and liquorice extracts was safe and effective in reducing plaque accumulation and improving general oral health in dogs, even without tooth brushing. These findings highlight the potential of the gel as a valuable addition to routine active oral care for dogs. Further investigations are needed to better characterize the long-term benefits of this product, and to assess whether more frequent applications could enhance the positive outcomes observed. Finally, its usefulness in cats should be confirmed to broaden the scope of this innovative approach to companion animal dentistry.
Materials
Buccaclean gel, MP Labo, Grasse, France
Sedator, Eurovet Animal Health B.V., Bladel, Netherlands
Torbugesic, Zoetis, Parsippany, NJ, USA
Propomitor, Orion Pharma Poland sp. Zoo, Warsaw, Poland
Isoflurin, VetPharma Animal Health SL, Barcelona, Spain
Tlen medyczny, AirLiquid Polska sp. Zoo, Kraków, Poland
Plyn Ringera, Fresenius Kabi Polska sp. Zoo, Stryków, Poland
IC Plaque, IM3, Duleek, Ireland
Research Randomiser, https://www.randomizer.org
Science Diet Canine Maintenance food for adult dogs, Hill's, Topeka, KS, USA
Antisedan, Orion, Finland
Statgraphics Centurion software, version XVI.II, Statgraphics Technologies, Inc., The Plains, VA, USA
Footnotes
Acknowledgments
The authors thank all pet owners involved in the study. The authors would like to thank Dr. Karin de Lange for her valuable help in proofreading the manuscript and providing insightful comments.
Ethical Approval and Informed Consent Statements
Dogs were recruited from the Veterinary Clinic Arka, Kraków, Poland, after signature of an informed consent form by the owners. The study was conducted according to standard veterinary dental care procedures that did not require Ethical Committee approval.
Author Contributions
JG: coordination, study protocol writing, clinical evaluation and data analysis. DZ: clinical evaluations. MJ: statistical analysis. CG: coordination, study protocol validation, data analysis, and manuscript validation. CN and BJ: study protocol validation, data analysis, and manuscript preparation.
Funding
The study was funded by MP Labo, the manufacturer of the tested product.
Declaration of Conflicting Interests
C. Gard, C. Navarro, and B. Jahier are employees of MP Labo, the manufacturer of the tested product. The other authors have nothing to disclose.
Data Availability Statement
Data is available upon reasonable request from the corresponding author at christelle.demongeot@mplabo.eu.
