Abstract
Hyaluronic acid (HA) has been extensively studied and utilized in human and veterinary medicine due to its versatile biological functions, including tissue hydration, anti-inflammatory effects, wound healing, regenerative effects, biocompatibility, biodegradability and antimicrobial properties. Historically, HA was first isolated in 1934 and has since been reviewed and applied across species in numerous clinical and nonclinical settings. In animal studies, canine models have demonstrated the efficacy of HA as an adjunct to periodontal interventions, particularly in improving bone and soft tissue regeneration in controlled experimental settings. Similarly, human studies have shown statistically significant improvements in periodontal outcomes such as probing depth, clinical attachment levels, and decreased periodontal inflammation when HA is used as an adjunct to scaling and root planing or surgical procedures. This review summarizes the current evidence on HA's structure, biological function, and applications in periodontal disease, with a focus on its potential to address unmet needs in veterinary dentistry. Given its demonstrated benign safety profile and proven efficacy in humans, HA holds promise as a valuable therapeutic option for managing periodontal disease in companion animals.
Keywords
Introduction and Background
Periodontal disease is one of the most common diseases diagnosed in veterinary practices, with a prevalence reported between 44% and 86.3%.1,2 While epidemiological surveys vary, it is well established that periodontal disease in companion animals progresses with age, and that smaller breeds are more susceptible. For example, a cross-trial comparison of 3 longitudinal studies showed that 57% of Labrador Retrievers suffered from periodontitis versus 98% for Miniature Schnauzers and Yorkshire Terriers. 3 Periodontal disease can have both local and systemic consequences. Locally, chronic presence of these microbial organisms can drive periodontal destruction through the persistent host inflammatory response to plaque bacteria. Systemically, bacteremia can occur; and periodontal disease has been shown to increase the risk of renal and cardiac disease that may result in negative impacts on an animal's overall health.4–9 Additionally, active periodontal inflammation has been correlated with elevated C-reactive protein, white blood cell counts, and polymorphonuclear cell counts, suggesting a systemic inflammatory response. 10 Given the high prevalence of this disease in companion animals, its progressive nature, and difficulties associated with regular tooth brushing and home care by pet owners, there is a constantly growing need that exists for periodontal interventions and treatments. 11
Hyaluronic acid (HA) was discovered by Karl Meyer and John Palmer in 1934, when it was isolated from the vitreous humors of bovine eyes. 12 HA has since been shown to be a naturally occurring polymer in many organs, and a key component of the extracellular matrix. It has also been found across species, ranging from vertebrates to bacteria, all with an identical chemical structure. 13 Extraction from animal tissues, frequently rooster combs, served as the original source for the production for HA, but fermentation from bacteria has since become the predominant method owing to cost efficiencies in purification and reduced risk of contamination from other animal byproducts. 14 Since its discovery, HA has been shown to play many important roles in biological function and used across a wide range of approved applications. Notably, studies in canine and human periodontal disease have led to multiple approved compounds in human dentistry (e.g., GENGIGELa, hyaDENT BGb).
Structure and Function
HA is a glycosaminoglycan composed of repeating disaccharide units of glucuronic acid and N-acetylglucosamine (chemical formula C14H21NO11). 15 These disaccharide units can form long polymer chains that add structure to the extracellular matrix of tissues. The molecular weight of HA varies depending on the length of these polymers and the source of the HA, ranging from less than 100,000 Daltons to greater than 6,000,000 Daltons. At least one source defines anything greater than 500,000 Daltons as high molecular weight. 16 Notably, high molecular weight HA occupies an extremely large volume because polymer length is inversely correlated with density. 17
HA is a naturally occurring component of the extracellular matrix and endogenously participates in many cellular processes, including its degradation via native enzymes (hyaluronases). 18 As such, it has multiple biological functions. Due to its hydrophilic properties, HA hydrates tissues. Six liters of water can be absorbed by 1 g of HA. 19 Relatedly, the viscous nature of HA can prevent tissue degeneration through decreased friction. 20 HA has analgesic properties via binding to mechanosensitive stretch-activated ion channels of nociceptive nerve terminals and interaction with free nerve endings. HA with a molecular weight greater than 860,000 Daltons provides high and long-lasting analgesia.21,22 HA can affect biological processes through direct binding to receptors on specific cell types. For example, HA can bind directly to CD44 receptors in immune cells to drive IL-1β-mediated anti-inflammatory effects. 23 As with analgesia, high molecular weight HA has been demonstrated to drive greater anti-inflammatory effects. 24 Additional specific binding functions include the receptor for hyaluronan-mediated motility, which coordinates migration of fibroblasts and keratinocytes to promote regeneration and wound healing. 25 Additionally, HA can provide antimicrobial effects, both via the aforementioned mobilization of immune cells, but also via mechanical prevention of bacterial adhesion. 26
HA plays an important role in healing. Upon traumatic insult, high molecular weight HA is rapidly synthesized by platelets and recruited from the bloodstream. High molecular weight HA works initially by binding fibrinogen to catalyze the extrinsic clotting pathway and stabilizing tissue spaces to facilitate polymorphonuclear leukocyte access. 27 However, within 24 h endogenous enzymes called hyaluronidase degrade high molecular weight HA into low molecular weight HA, which drives angiogenesis to the wound and triggers endothelial cell proliferation. Exogenous administration of high molecular weight HA has been shown to accelerate healing via the endogenous mechanisms described above. 28
Veterinary Applications
HA's first veterinary approval was in 1988 as an intra-articular joint injection for osteoarthritis in horses. Clinical studies leading to its approval showed dose-dependent and statistically significant improvements in lameness scores. 29 A subsequent product replicated this data in both intra-articular and intravenous settings, and it has since been studied in oral formulations in dogs, with a variety of products available.30–33
HA has been studied and approved in ophthalmologic settings in both canines and felines. It has been used in an artificial tear lubricating eye drop as an adjunct to anesthesia, chronically in cases of dry eye, and as a treatment for corneal ulceration.34–37 Subdermal injections of HA have been shown to effectively treat entropion. 38
Human Nondental Applications
In human medicine, HA applications extend beyond those of veterinary medicine to a wide variety of indications. HA has been studied in wounds, both acute and chronic, including those arising from burns, venous insufficiency, diabetes, neuropathies, and surgery.39,40 In 1 meta-analysis, 8 of 9 studies demonstrated significantly improved wound healing using HA versus the control (traditional therapies or placebo), while the 9th study showed that a higher concentration HA formula significantly improved healing versus a lower concentration counterpart. 40 HA has also been used as a bulking agent (i.e., fills space to increase resistance to urine outflow) to treat vesico-ureteral reflux. 41 Similarly, HA has been used as an organ spacer to reduce gastro-intestinal side effects during radiation therapy for prostate cancer. 42 HA in the latter setting is noted to be highly visible on ultrasound and magnetic resonance imaging while retaining consistent shape for 10 to 12 months, while also being easily reversible with hyaluronidase. 43 HA has been used extensively in cosmetics, both as an injection and as a topical cream.44–46 HA fillers have largely superseded earlier bovine- and human-derived collagen products because of its lack of tissue or species specificity, therefore minimizing immunologic risk. 47 Over-the-counter topical formulations have been studied clinically to improve dermatological endpoints. 48 Finally, HA has been used as a drug-delivery excipient due to its biocompatibility and tissue retention qualities. 49
Canine Periodontal Studies
This review identified 5 studies utilizing adult Beagle dogs in a research setting. These studies investigated the effects of HA as a monotherapy adjunct to periodontal procedures, which all demonstrated benefits of HA across multiple endpoints. Similarities across studies include study populations (lab animals, specifically adult beagle dogs), treatment protocols (single application of HA post-procedure), and split-mouth design (i.e., self-control). All animals were euthanized at the conclusion of the study period to obtain tissue samples for histologic evaluation, the gold standard for measuring periodontal disease. 50 Table 1 includes specific details on each study, with notable findings highlighted below.
Summary of Hyaluronic Acid Canine Studies in Research Settings.
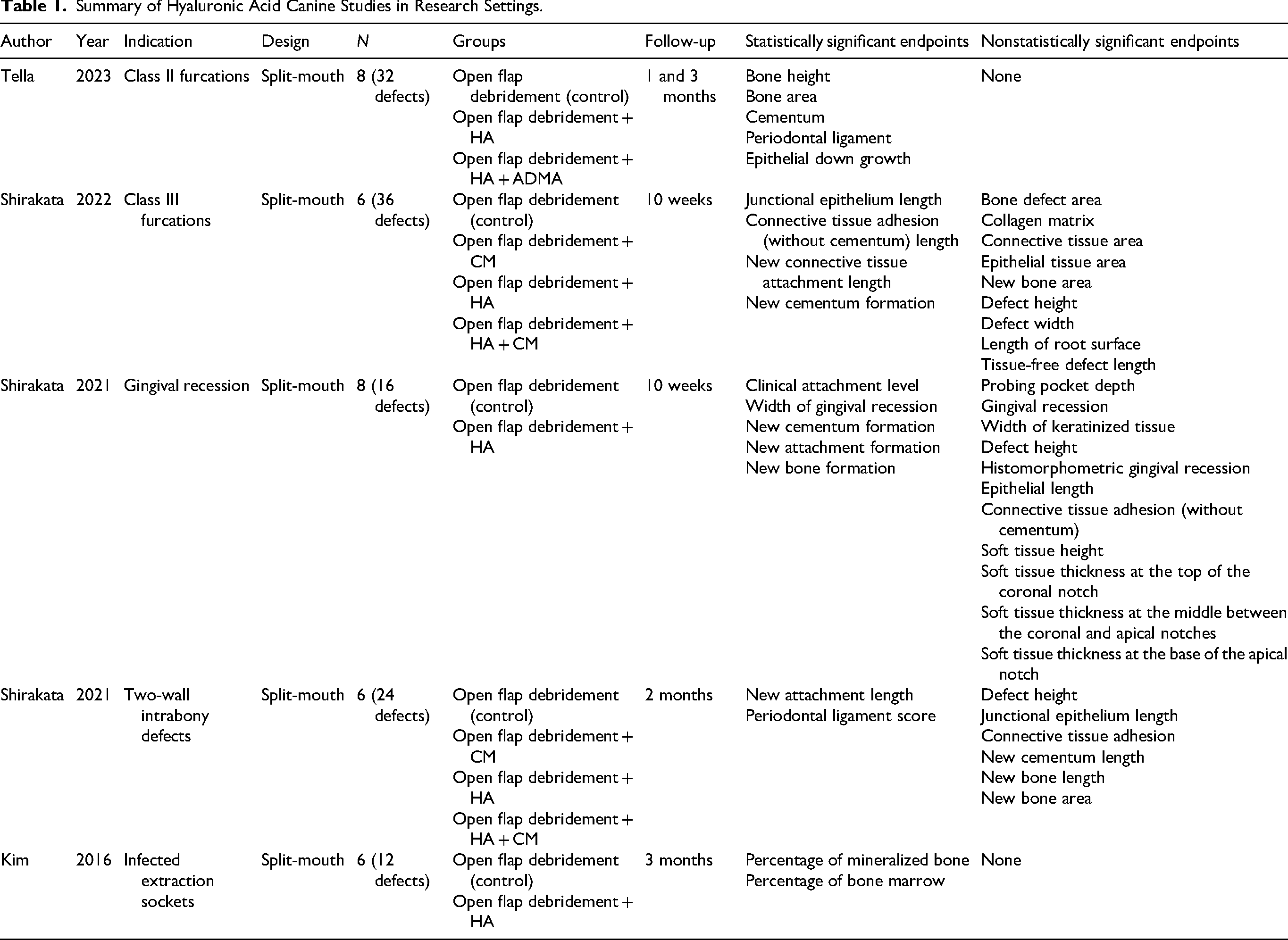
The effects of HA with and without acellular dermal matrix allograft (ADMA) in Class II furcation defects have been examined. 51 Bilateral defects were surgically created in the buccal aspect of mandibular premolars for a total of 32 defects across 8 dogs. To prevent spontaneous repair of the defects, a rubber base impression material was placed within the furcation of these teeth, to allow plaque and gingival inflammation to accumulate. Two weeks later, open flap debridement was performed on all defects, with 8 receiving adjunctive HA, 8 receiving both adjunctive HA and ADMA, and 16 receiving open flap debridement only (control). Histomorphometric measurements of bone height, bone area, cementum, periodontal ligament, and epithelial down growth were measured at 1 and 3 months. Improvements in all categories reached statistical significance at all time-points for HA versus control, including highly significant P-values of <.000 for bone height and bone area at 3 months. Numerically, HA demonstrated 88.95% regeneration of newly formed bone height relative to the size of the created defect, reaching 8.72 mm of regeneration in absolute terms (vs 38.55% and 5.11 mm, respectively, for control). Addition of ADMA versus HA alone reached statistical significance across all categories at 3 months, though the absolute difference in effect size was clinically minimal (e.g., 90.34% and 9.34 mm in newly formed bone height). Unlike the control group, neither test groups (HA alone, and HA with ADMA) showed histological evidence of epithelial downgrowth into the periodontal pocket. Clinically important, this study showed favorable periodontal regeneration and clinical improvements following HA application.
The effects of HA, collagen matrix (CM), and HA + CM as an adjunct to open flap debridement in Class III furcations in beagle dogs (N = 6) have also been examined. 52 Six defects per dog were surgically created at bilateral mandibular 2nd, 3rd, and 4th premolars. At 10 weeks, 4 of 13 histomorphometric endpoints met statistical significance in the intervention arms versus control: junctional epithelium length, connective tissue adhesion (without cementum) length, new connective tissue attachment length, and new cementum formation length. The improvement in new connective tissue attachment length was the most dramatic in the HA treatment group proportionally, reaching 6.25 ± 1.45 mm for HA alone versus 1.47 ± 0.85 for control (and 6.4 ± 1.35 mm for HA + CM). The HA and HA + CM groups achieved statistical significance across all 4 endpoints, while the CM alone group did not for any endpoints. Additionally, complete closure of Class III furcation defects was not observed in any of the treatment groups, further reinforcing that this is a challenging pathology that likely requires extraction.
In a different study, the same lead author's group investigated the effects of open flap debridement with or without HA in gingival recession. 53 Bilateral defects were surgically created on the buccal aspect of both maxillary canines in 8 dogs, for a total of 16 defects. After 8 weeks, the defects were treated with debridement, with or without HA. At 10 weeks, 5 out of 16 endpoints showed a statistically significant improvement in favor of adjunctive use of HA versus open flap debridement alone: clinical attachment level, width of gingival recession, new cementum formation, new attachment formation, and new bone formation. Improvement in new cementum formation reached 4.31 ± 1.78 mm versus 2.40 ± 1.35 mm and improvement in new bone formation reached 1.84 ± 1.16 mm versus 0.72 ± 0.62 mm for treatment and control arms, respectively. This was clinically relevant, demonstrating periodontal regeneration of defects with gingival recession is improved when using HA in addition to open flap debridement.
In a 3rd study, the group investigated the effects of HA, CM, and HA + CM as an adjunct to open flap debridement in two-wall intrabony defects. 54 Two-wall intrabony defects were surgically created at the distal and mesial aspects of mandibular premolars in 6 dogs, resulting in 24 defects for randomization. At 2 months, histomorphometric endpoints met statistical significance in HA and HA + CM arms for 2 of the 8 endpoints measured versus open flap debridement alone: new attachment length and periodontal ligament score. HA demonstrated a 2.43 ± 1.25 mm versus 0.55 ± 0.99 mm improvement in new attachment length for HA alone versus control arms. CM alone did not meet statistical significance in any of the 8 endpoints.
In a separate study the effects of HA in infected sockets was examined. 55 Combined periodontal and endodontic periapical lesions were surgically induced and iatrogenically infected with Porphyromonas gingivalis for a period of 4 months to generate infected sockets in 6 beagle dogs at the mandibular third premolars for a total of 12 lesions. Subsequently, the mesial roots were removed bilaterally and treated with either HA or left untreated (control). At 3 months, histomorphometric endpoints met statistical significance for 2 of 2 endpoints demonstrating improvements in the treatment arm: percentage of mineralized bone and percentage of red bone marrow.
Beyond these studies, 2 other trials tested the effects of HA without a monotherapy HA arm. The first showed that HA in combination with sodium hypochlorite/amino acid gel exhibited statistically significant improvements in pocket probing depth, bleeding on probing, new bone formation, new attachment length, and new cementum when compared to control at 8 weeks for patients with two-wall intrabony defects. 56 Separately, the second showed that HA used in conjunction with an absorbable collagen sponge demonstrated statistically significant improvements in bone formation (measured via micro-computed tomography) and the number of bone growth mediating-cells (as determined by osteocalcin staining). Qualitative review of histology suggested regrowth of tissue was visually favorable for the treatment arm versus control (e.g., continuous cortication with adjacent cortical bone). The authors of this study conclude that “HA can promote bone formation and improve the wound healing rate comparable to recombinant human bone morphogenetic protein-2 in infected extraction sockets,” which has been FDA approved for maxillary sinus augmentation and alveolar ridge augmentation.57,58
Human Periodontal Studies
HA has been studied extensively in human periodontal settings. Variations in studies include HA concentration, treatment frequency, endpoints, delivery mechanism (e.g., topical, injectable), study design, and molecular weight.
A review of the literature identified 4 meta-analyses with statistical analysis calculating the effect size of HA across multiple studies and showing the benefits of periodontal HA application. While research protocols and inclusion/exclusion criteria varied, and each meta-analysis came to its own quantitative conclusion, some overlap existed in terms of included studies for analysis. The findings are summarized in Table 2.
Summary of Hyaluronic Acid Meta-Analyses in Humans.
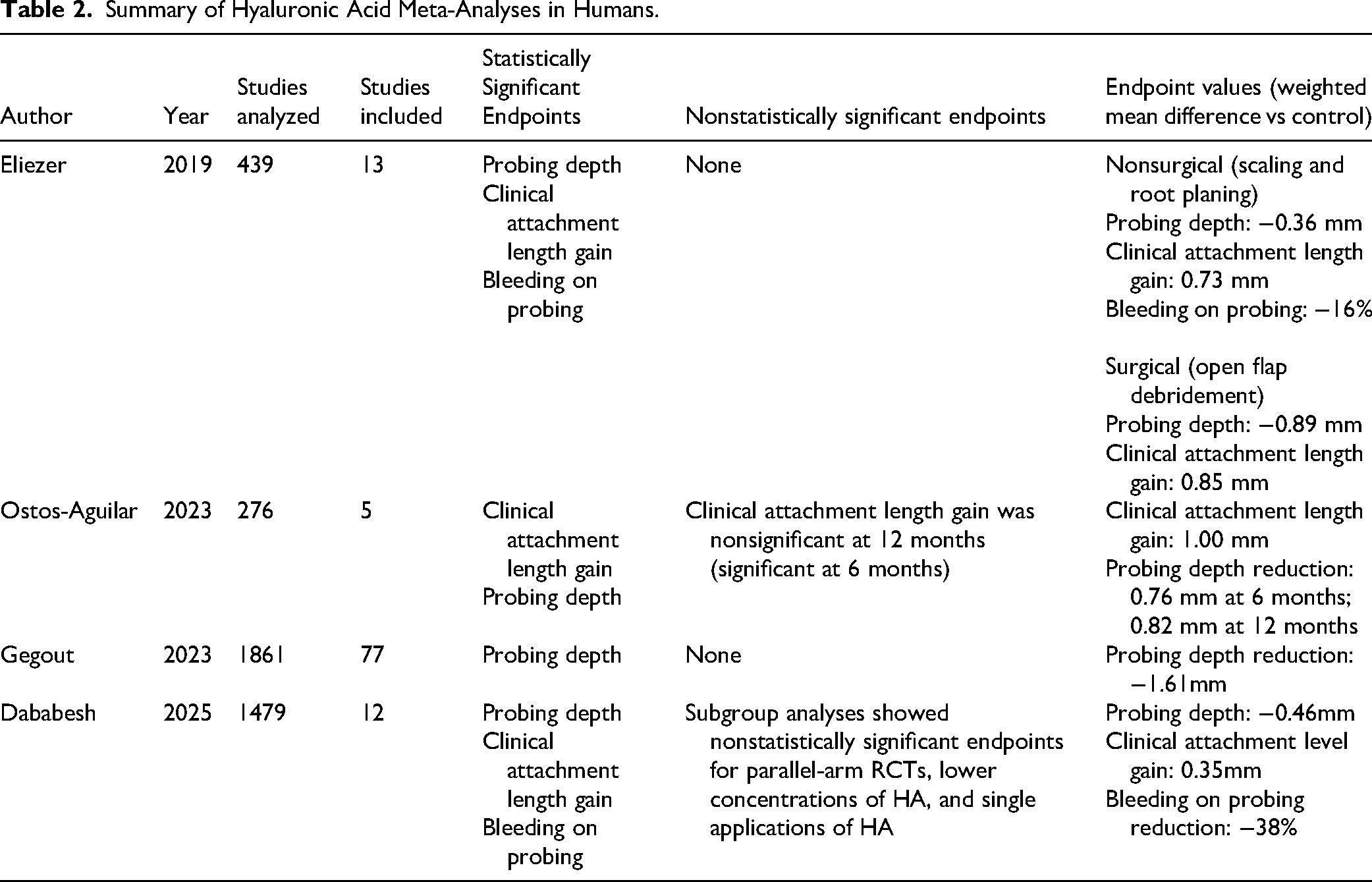
The first study performed a meta-analysis of 13 randomized controlled trials classified into nonsurgical versus surgical interventions for chronic periodontitis. Their analysis showed statistically significant improvements in probing depth, clinical attachment level gain, and bleeding on probing in both settings when HA was used compared to controls. 59
The second study performed a meta-analysis of 5 randomized controlled trials and found statistically significant improvements in clinical attachment level gain and probing depth at 6 months when HA was used as an adjunct to open flap debridement for the treatment of intrabony defects. Probing depth improvements remained statistically significant out to 12 months for 2 out of 2 studies examined to that duration (3 of the included studies only had 6-month durations). 60
The third study performed a meta-analysis of 77 randomized controlled trials where eight topical adjuncts were compared versus scaling and root planing for the treatment of periodontitis. HA showed a statistically significant weighted mean difference in probing pocket depth of 1.61 mm, which was also numerically the highest out of all 8 interventions included in the analysis (other interventions in order of effect size: metformin, statins, macrolides, tetracyclines, metronidazole, chlorhexidine, and bisphosphonates). 61
The final study performed a meta-analysis of 12 studies examining the effects of HA used as an adjunct to nonsurgical periodontal therapy in adults with periodontitis. Statistically significant improvements were noted in probing depth, clinical attachment level gain, and bleeding on probing. Subgroup analyses revealed that higher HA concentrations (0.8%) showed more pronounced probing depth reduction. 62
Beyond these 4 meta-analyses, 7 additional systematic reviews were identified, each analyzing the impact of HA as an adjunct to periodontal procedures. For all the systematic reviews, the conclusions by the authors were similar, noting the statistically significant benefits of HA observed across multiple studies and endpoints. However, the authors also remark on the wide heterogeneity in study design, HA formulation, and time frame, suggesting that further studies must be conducted before recommending an optimal regimen.
While some of the examined trials included application frequencies as high as twice daily for 3 weeks, 3 identified studies examined the impact of a single application immediately postdebridement—similar in structure to the aforementioned canine studies. Findings are summarized below and in Table 3.
Summary of Hyaluronic Acid Human Studies With Single-Dose Treatment Protocols.

One study involved 60 patients randomized equally into 3 groups. Group I received scaling and root planing only, Group II received adjunctive subgingival application of HA gel, and Group III received adjunctive subgingival application of chlorhexidine gel. Inclusion criteria required each patient to have a minimum of 8 sites with probing pocket depths between 4 and 8 mm for examination. At 3 months, Group II showed the highest improvements in probing pocket depth and clinical attachment level, including a statistically significant improvement versus scaling and root planing alone (Group I). Absolute improvements in probing pocket depth were 1.6 mm and 2.5 mm from baseline for the control and treatment groups, respectively. When normalized for baseline pocket depth (4.9 mm and 5.93 mm for control and HA treatment groups, respectively), HA provided a 1.3× improvement versus scaling and root planing alone. Likewise, absolute improvements in clinical attachment level were 0.55 mm and 1.25 mm from baseline for the control and treatment groups, respectively. When normalized for baseline clinical attachment level (6.1 mm and 6.13 mm for control and HA treatment groups, respectively), HA provided a 2.25× improvement versus scaling and root planing alone. 63
A similar study was conducted which included 30 patients with chronic periodontitis with baseline pocket depth >4 mm and the added comorbidity of diabetes mellitus type 2. Subjects were randomly allocated into equal treatment and control groups, where the treatment group received HA as an adjunct following scaling and root planing and the control group received scaling and root planing alone. Clinical parameters including plaque index, gingival index, probing depth, and clinical attachment level were measured at baseline, 6 weeks, and 12 weeks. Statistically significant improvements for the treatment group were observed for all clinical parameters except for plaque index at both 6 and 12 weeks. For 12-week values, probing depths were improved 0.88 mm and 1.1 mm in the control and treatment groups, respectively. When normalized for baseline pocket depth (3.56 mm and 3.49 mm for control and HA treatment groups, respectively), HA provided a 1.28× improvement versus scaling and root planing alone. Clinical attachment levels were improved 0.83 mm and 1.32 mm in control and treatment groups, respectively. When normalized for baseline clinical attachment level (4.29 mm and 4.35 mm for control and HA treatment groups, respectively), HA provided a 1.57× improvement versus scaling and root planing alone. Finally, gingival index scores were improved to 1.03 and 0.85 in control and treatment groups, respectively. When normalized for baseline gingival index scores (1.91 and 1.88 for control and HA treatment groups, respectively), HA provided a 19% reduction in gingival scores. 64
Another study selected 33 patients with chronic periodontal disease with probing depth >4 mm for inclusion. This trial was a split-mouth design where each patient received full mouth scaling and root planing. Half of each maxilla and mandible was randomly allocated to the treatment group (HA adjunct) or control group (scaling and root planing only). At baseline and after 6 weeks, patients were assessed for periodontal parameters: gingival index, plaque index, probing depth, and clinical attachment level. Statistically significant improvements in probing depth and clinical attachment level were observed. Nonsignificant improvements were noted for plaque and gingival indices. At 6 weeks, absolute improvements in probing pocket depth were 0.12 mm and 1.49 mm for the control and treatment arms, respectively. When normalized for baseline probing depths (5.45 mm and 5.64 mm for control and HA groups, respectively), HA provided a 12× improvement versus scaling and root planing alone. Likewise, for clinical attachment level absolute improvements were 0.31 mm and 1.03 mm for control and treatment arms, respectively. When normalized for baseline clinical attachment levels (5.73 mm and 5.91 mm for control and HA groups, respectively), HA provided a 3.22× improvement versus scaling and root planing alone. 65
Optimal regimens for veterinary medicine would likely benefit from minimal follow-ups/reapplications, hence the importance of single applications driving statistically significant benefit across multiple endpoints. Also relevant to veterinary medicine, the healing effect of HA has been shown to occur as early as within the first 24 h postsurgery. Another study was conducted using a randomized, split-mouth, double-blind clinical trial to assess healing at 24 h and at 1 week. Not only did the HA group experience a statistically significant improvement in healing at 24 h and 1 week versus control, but the HA group's healing score at 24 h was numerically higher than that of the control group at 1 week. 66 Given the challenges of managing animals postsurgery, rapid healing can potentially reduce short-term complications.
There were no reports of adverse events or safety issues across cited human reviews and individual studies.
HA Versus Other Adjunctive Therapies
Given the increased awareness of appropriate antibiotic stewardship to reduce the development of antibiotic resistance, utilization of unnecessary systemic antibiotics is discouraged, particularly for prophylactic indications. Nevertheless, up to 35% of veterinary patients treated at dental specialist referral clinics received systemic antibiotics, with prophylaxis accounting for nearly half of prescriptions despite little evidence for its efficacy. 67 Practitioners commonly utilize antibiotic-based topical perioceutics as adjuncts to mechanical debridement, which likely reduces the risk of resistant strains when compared to systemic antibiotics. Still, multiple studies have shown at least transient increases of resistant bacterial strains from local administration of both doxycycline68–70 and chlorhexidine. 71 When compared to antibiotic-based perioceutics, HA does not risk the development of antibiotic-resistance strains of bacteria given that its ability to reduce antimicrobial colonization is related to mechanical (biofilm preventing bacterial adhesion) and endogenous (mobilization of local immune system) mechanisms. Additionally, cross trial comparison of canine studies suggests the effect size for HA in probing pocket depth is twice that of clindamycin and doxycycline perioceutics when normalized for baseline (35% vs 14-18% improvement over control, for HA and antibiotic perioceutics, respectively).53,72–74 This finding is corroborated by a meta-analysis of human studies, where the weighted mean difference in millimeters versus scaling and root planing was 1.61 mm, 0.71 mm, and 0.51 mm for HA, macrolides, and tetracyclines, respectively. 61
Platelet-rich fibrin has also been used as an adjunct in periodontal settings, given its natural healing properties, including stimulation of soft and osseous tissue healing. 75 One human study comparing HA to platelet-rich fibrin in alveolar ridge preservation showed a statistically significant improvement in tissue healing: 9.78 mm (22.4% increase vs control), 8.6 mm (7.6% increase vs control), and 7.99 mm for HA, platelet-rich fibrin, and control, respectively. 76 Another study examined the impact of HA and platelet-rich fibrin on bone formation in dental implant sockets in sheep. This study had a combination HA and platelet-rich fibrin arm that demonstrated statistically significant improvements for HA alone, platelet-rich fibrin alone, and the combination arm versus control. Additionally, the combination demonstrated statistically significant improvements in all tested clinical endpoints versus monotherapy arms, suggesting synergistic effects. 77
One other combination that has been investigated in humans is HA with bone graft substitutes, specifically hydroxyapatite. One randomized, double-blinded, split-mouth study showed the combination of HA with hydroxyapatite drove statistically significant improvements in both clinical and radiographic endpoints at 12 months: probing pocket depth (5.06 mm vs 3.21 mm), bone probing depth (4.22 mm vs 3.21 mm), clinical attachment level gain (4.00 mm vs 2.86 mm), defect depth reduction (1.92 mm vs 1.14 mm), and percentage of defect depth reduction (48% vs 20.14%) were all shown to have greater improvements for HA in combination with hydroxyapatite versus hydroxyapatite monotherapy. 78
HA in Human Stomatitis
HA has been shown to be effective in other oral pathologies, including human analogs to canine stomatitis: aphthous ulcers,79,80 Behçet's disease, 81 and oral lichen planus. 82 While the exact etiology of canine chronic ulcerative stomatitis (CCUS) is unknown, hypotheses include an autoimmune origin as well as a microbial one. 83 Treatments in veterinary and human medicine include both immunosuppressive and antimicrobial mechanisms. 84 Topical HA use in humans has demonstrated statistically significant improvements in ulcer size by day 7 accompanied by reductions in pain. 79 A systematic review of 9 studies in humans also showed statistically significant improvements in healing time and pain alleviation, while no side effects were reported. HA showed superior results compared to triamcinolone in one study, and comparable results in 2 others. 80 Behçet's disease, known to have an autoimmune etiology, often manifests in oral stomatitis. One study showed reductions in number of ulcers, reduction in total ulcer area, and improvements in pain, swelling, and heat in patients with both Behçet's disease and recurrent aphthous ulcers. 81 Finally, a systematic review analyzed 7 studies for HA in oral lichen planus, which has been noted to be the closest human analog to CCUS. 82 Though not identical, oral lichen planus and feline chronic gingivostomatitis also share many similarities as they are both chronic inflammatory oromucosal diseases. 85 One review noted individual trials demonstrating statistically significant findings in favor of HA for oral lichen planus, however meta-analysis was inconclusive, likely due to the high degree of heterogeneity among the included studies. 82 It can be inferred from this body of evidence in humans that HA has potential as a new therapeutic option for challenging stomatitis cases in veterinary medicine, though confirmatory studies still need to be performed.
Possible Application in Veterinary Patients With Periodontal Disease
As previously cited, animal (canine) models in research settings have been used to evaluate the clinical and histological outcomes of use of HA in patients with periodontal disease. Although there are no clinical studies in veterinary medicine analyzing the use of HA in periodontal disease, inferring from the available literature, the authors propose that any dog with periodontal disease that would benefit from closed or open root planing and pocket treatment with a perioceutic would be an appropriate candidate for the use of HA. Furthermore, use of HA alongside a collagen membrane in guided tissue regeneration (GTR) may be an option to result in improved outcomes over the same therapy without HA. Additionally, cases with teeth that are clear candidates for extraction could benefit from HA application postremoval. Importantly, the reviewed literature suggests that HA should be used strictly as an adjunct to—and not a replacement for—thorough debridement.
Conclusions
Based on the reviewed literature, HA has potential to improve periodontal outcomes in veterinary dentistry and oral surgery. Given its minimal risk and demonstrated efficacy in multiple canine and human settings, ranging from clinical improvement in mild gingival recession to more severe periodontitis, HA shows promise as an important adjunct to routine dental care. Specifically, HA could be used to accelerate healing and improve outcomes as an adjunct to scaling and root planing, open flap debridement, GTR, and postextraction. HA could also potentially be beneficial in combination with other adjuncts, including collagen matrices, bone grafts, and platelet-rich fibrin. Additionally, HA may have efficacy in oral pathologies such as stomatitis. Finally, given its identical chemical structure and ubiquitous presence across many species, the potential therapeutic benefit of HA could be extended to cats and other species receiving veterinary dental care.
Materials
a. Oral Science, Brossard, QC, Canada
b. regedent, Zurich, Switzerland
Footnotes
Acknowledgements
The author thanks Dr Suzanna Hatunen, DVM, DAVDC, Dr Erin Ribka, DVM, DAVDC, Jennifer Redmon, DVM, Dr Kerry Mead, DVM, Dr Jamie Anderson, DVM, DAVDC, DACVIM, Dr Bonnie Shope, DVM, DAVDC, Dr Jan Bellows, DVM, DAVDC, and Dr Janet Hong, MD for their editorial reviews of earlier drafts of this article.
Ethical Considerations
This article does not involve any human or animal participants.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by PerioVive.
Declaration of Conflicting Interest
Bryan Song, MD, is the Founder and CEO of PerioVive, which manufactures and markets a hyaluronic acid adjunct for veterinary dental applications.
