Abstract
Periostitis ossificans (PO) is a subtype of chronic osteomyelitis characterized by subperiosteal bone formation as a reaction to mild infection or irritation. In the veterinary medicine literature, it has been described in large breed dogs, aged 3 to 5 months, and was postulated to be caused by mild inflammation related to tooth eruption. This report describes 3 Belgian Malinois puppies from the same litter diagnosed with PO at 3 months of age. The puppies presented with non-painful distal mandibular swellings. Diagnostic procedures included intraoral dental radiographs, a computed tomography scan of the head, and bone biopsies submitted for histopathological examination. PO is typically a self-limiting condition and likely caused by inflammation due to tooth eruption. Therefore, treatment focuses on pain management and monitoring until the permanent teeth emerge. Long-term follow up until two and a half years of age showed a significant decrease to total resolution of the swelling, with minor bone changes evident on advanced imaging. Histopathologic findings are not pertinent for the diagnosis of PO and imaging shows pathognomonic features of the condition. Mandibular PO, craniomandibular osteopathy, and calvarial hyperostotic syndrome are all self-limiting proliferative bone diseases of the skull in juvenile dogs with an implied genetic component. The authors’ findings support grouping them as a single disorder called “Idiopathic Canine Juvenile Cranial Hyperostosis” with predilection sites that vary between breeds.
Keywords
Introduction
Periostitis ossificans (PO) is an atypical chronic osteomyelitis characterized by subperiosteal bone formation. Osteomyelitis is an inflammatory reaction of the bone in response to infection. The source of infection can be exogenous or hematogenous. There are 5 phases in the pathophysiology of hematogenous osteomyelitis: inflammation, suppuration, necrosis, new bone formation, and resolution. 1 In PO, chronic low-grade inflammation or irritation results in bone “buildup” rather than the classic appearance of bone loss. 2 PO was first described by Carl Garré in 1893 as 1 of 10 clinical complications of osteomyelitis, thereby referred to as “osteomyelitis of Garré.” Other synonyms of PO include sclerosing osteomyelitis, chronic osteomyelitis with proliferative periostitis, and non-suppurative sclerosing osteomyelitis. 2
PO occurs in children and young adults when the osteoblastic activity of the periosteum is at its peak. 3 The causative factor is usually an odontogenic infection resulting from dental caries 4 but other etiologies such as mild periodontitis, dental eruption, or complication of dental extractions have also been described in the literature. 5
There are 2 published case reports of PO in the veterinary literature. The disease was described in 6 male large breed puppies aged 3 to 5 months, who presented with a non-painful mandibular swelling. Dental radiographs revealed a 2-layered ventral mandibular cortex, which was diagnosed as periosteal new bone formation on histopathologic examination. It was postulated that PO in these puppies was caused by mild inflammation related to tooth eruption.6,7
This report describes the clinical presentation, diagnostic procedures, and short- and long-term follow-up regarding clinical outcomes in 3 Belgian Malinois puppies, from the same litter, diagnosed with PO by the dentistry and oral surgery service at Koret veterinary teaching hospital, Israel.
“Don’t touch” lesions is a concept used in human medicine, describing non-aggressive bone lesions that have very characteristic radiographic features that render a biopsy or additional diagnostic tests unnecessary. 8 The definition “don’t touch” serves to reserve histopathology for aggressive lesions. 9 In human medicine PO is considered a “don’t touch” lesion. 10 To the best of the authors’ knowledge, the concept of “don’t touch” lesions is not used in veterinary medicine. The possibility of introducing this concept to veterinary medicine, as well as acknowledging that PO is a “don’t touch” lesion will be addressed.
The connection between craniomandibular osteopathy (CMO) and calvarial hyperostotic syndrome (CHS), 2 self-limiting proliferative bone diseases of the skull in juvenile dogs, and mandibular PO is also discussed.
Case Description
Signalment
Three intact Belgian Malinois puppies from the same litter were presented at 3 months of age to the dentistry and oral surgery service at Koret veterinary teaching hospital, Israel due to non-painful distal mandibular swellings. The litter consisted of 4 puppies. Puppies #1 and #2 were female and puppy #3 was a male. All attempts to contact the owner of the fourth puppy failed.
History
The chief complaint for all patients was a progressing non-painful distal mandibular swelling. Puppy #1 was presented with a left mandibular swelling, puppy #2 with a right mandibular swelling, and puppy #3 with bilateral mandibular swelling. The time of onset of the swellings was unclear in all puppies. No other clinical signs or developmental abnormalities were noticed by any of the owners.
Puppy #1 had a fine needle aspiration performed by the referring veterinarian 2 weeks prior to presentation. The aspirate contained pink fluid, with few lymphocytes, macrophages, red blood cells, and periosteal cells. No bacteria were detected in the aspirated fluid. Following this procedure, the puppy was treated with amoxicillin/clavulanic acida (12.5 mg/kg) q12h per os for 5 days. The other puppies underwent no diagnostic procedures prior to presentation.
Clinical and Laboratory Findings
A complete physical examination was performed at the time of presentation. No abnormalities other than the distal mandibular swellings were detected in any of the puppies. Complete blood count and serum chemistry results were within normal limits in all of the puppies.
The puppies were anesthetized using an opioid and acepromazine for premedication, propofol and a benzodiazepine for induction, and isoflurane in 100% oxygen for maintenance. An oral examination was performed under general anesthesia. All puppies had normal dentition for their age. All deciduous mandibular premolar teeth were present, and no permanent mandibular premolar or molar teeth had erupted. In all the puppies a swelling was noted on the ventral, buccal and lingual aspects of the affected mandibles, extending from the level of the deciduous mandibular fourth premolar teeth (708/808) distally (Figures 1A-C, 2A and B, 3A and B).
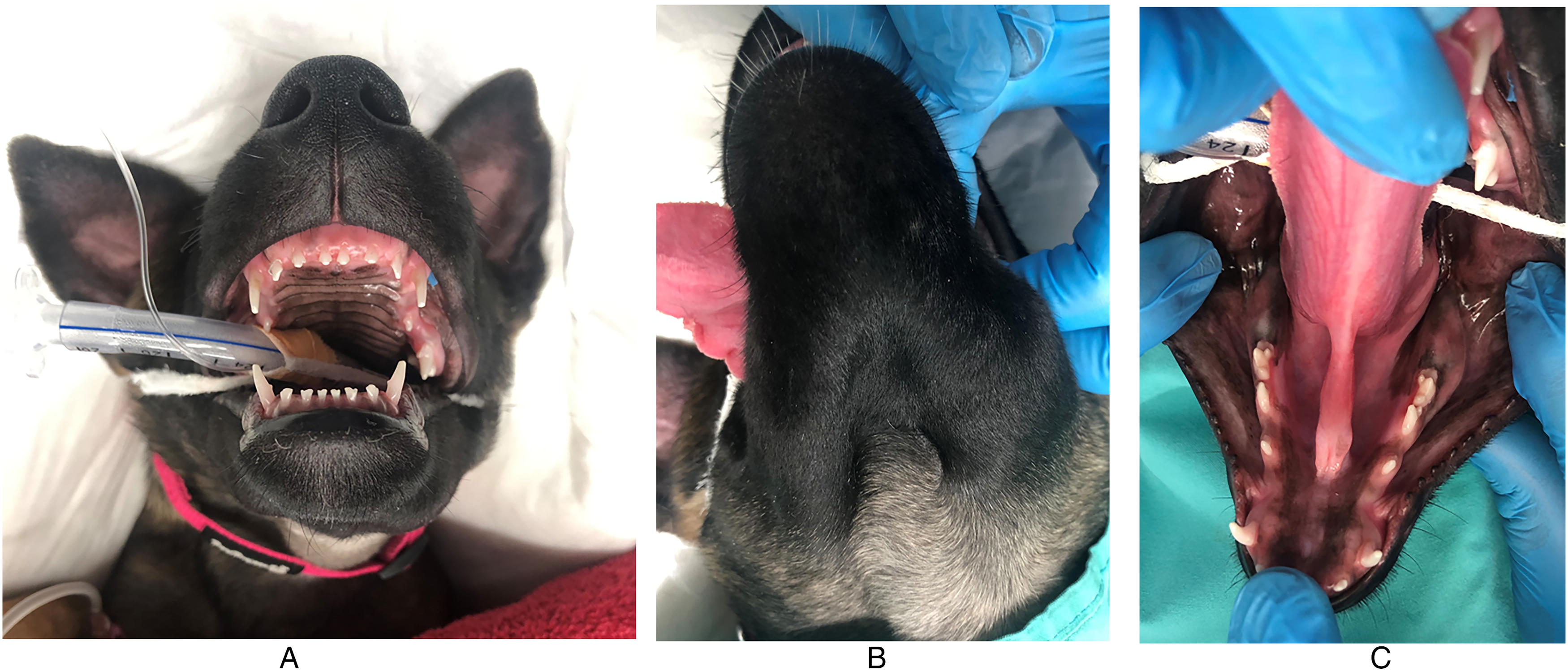
Puppy #1 at presentation. (A) Frontal view, (B) Ventral view and (C) Intraoral view showing swelling on the ventral, buccal and lingual aspects of the left mandible, extending from the deciduous fourth premolar tooth (708) distally.
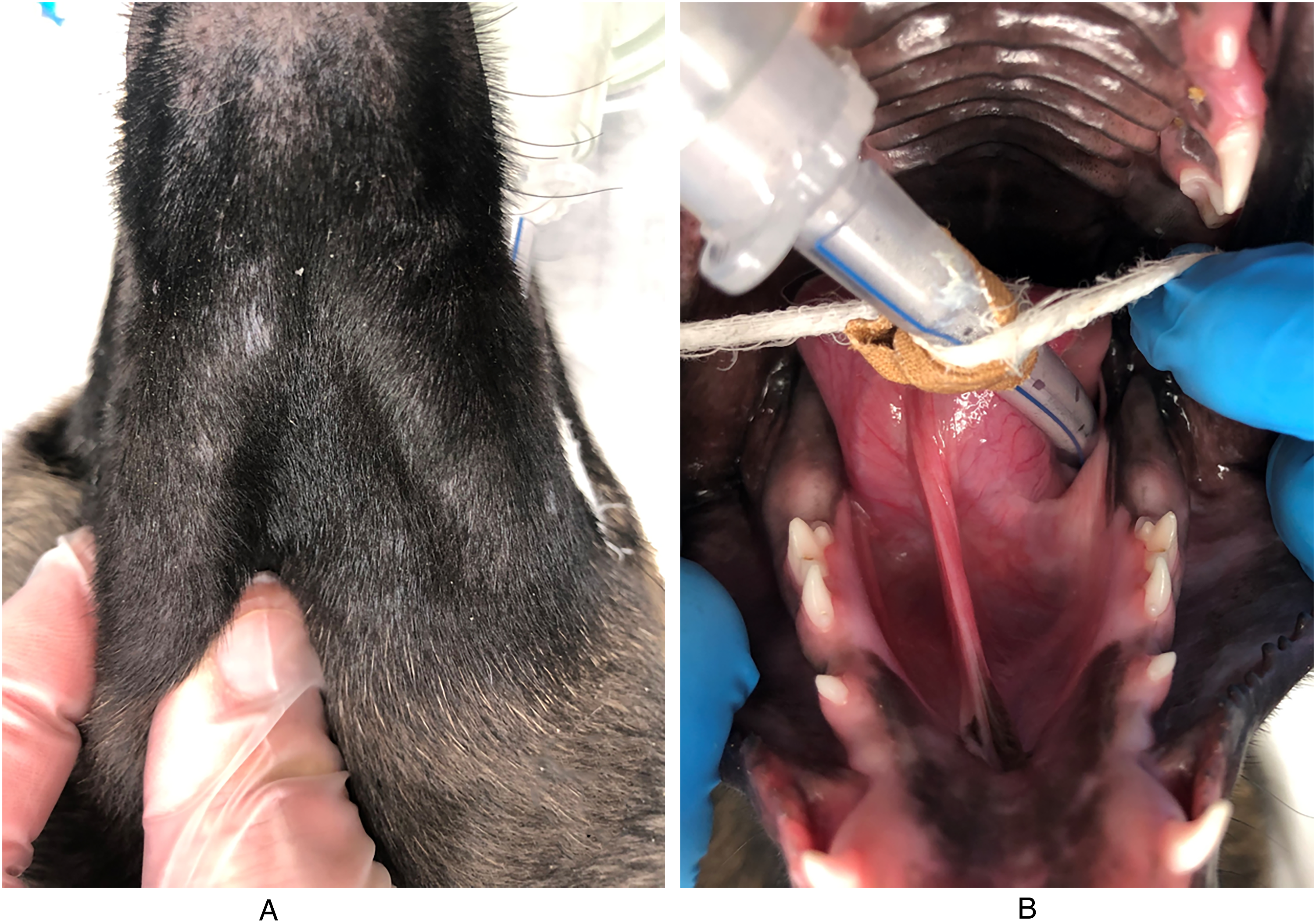
Puppy #2 at presentation. (A) Ventral view, (B) Intraoral view showing swelling on the ventral, buccal, and lingual aspects of the right mandible, extending between the deciduous fourth premolar tooth (808) and the unerupted first molar tooth (409).
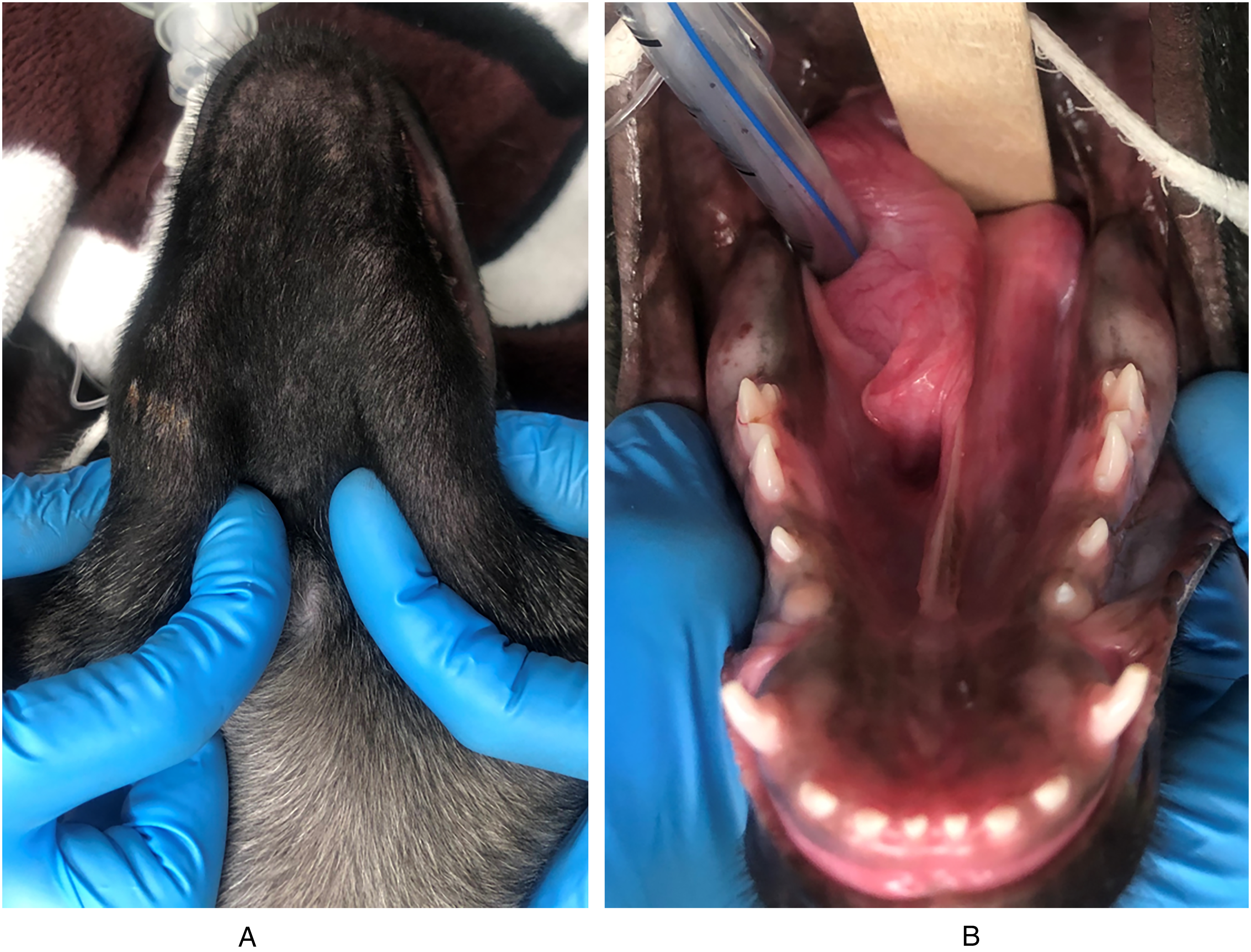
Puppy #3 at presentation. (A) Ventral view, (B) Intraoral view showing swelling on the ventral, buccal, and lingual aspects of both mandibles, more prominent on the right mandible, extending from the deciduous fourth premolar teeth (708 and 808) distally.
Dental Radiographic Findings
Intraoral dental radiographs of the distal mandible were obtained bilaterally. The lateral view of the affected mandibles revealed a double-cortex (“onion skin”) formation along the ventral border in all the puppies, with a radiolucent area separating it from the normal mandibular cortex. This additional cortical layer varied in length between the puppies but was present ventral to the tooth bud of the mandibular first molar tooth (309/409) in all the cases (Figures 4A, 5A, and 6A). The occlusal view showed a double-cortex formation lingually in all 3 puppies (Figures 4B, 5B, and 6B) and buccally in puppies #1 and #3 (Figures 4B and 6B).
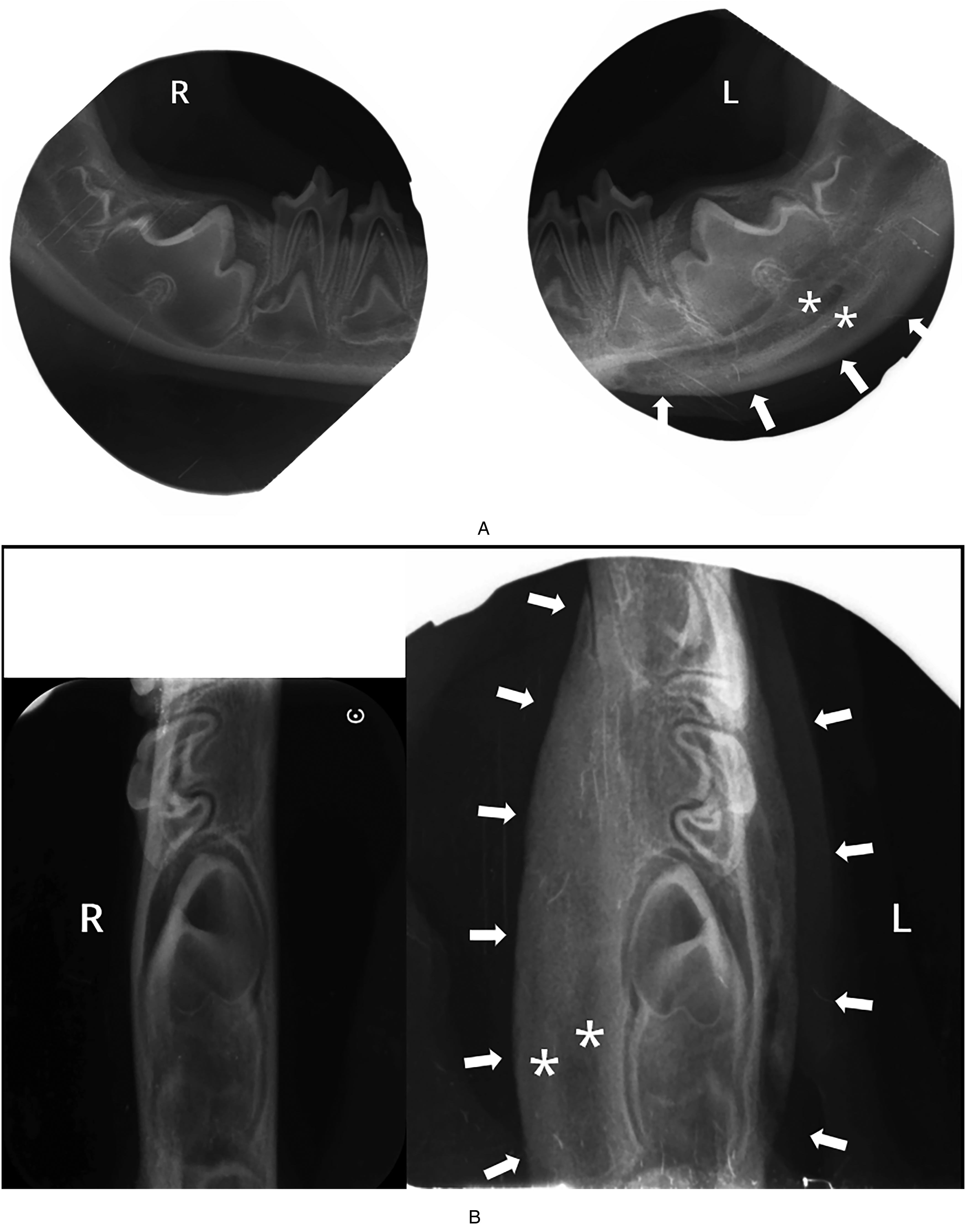
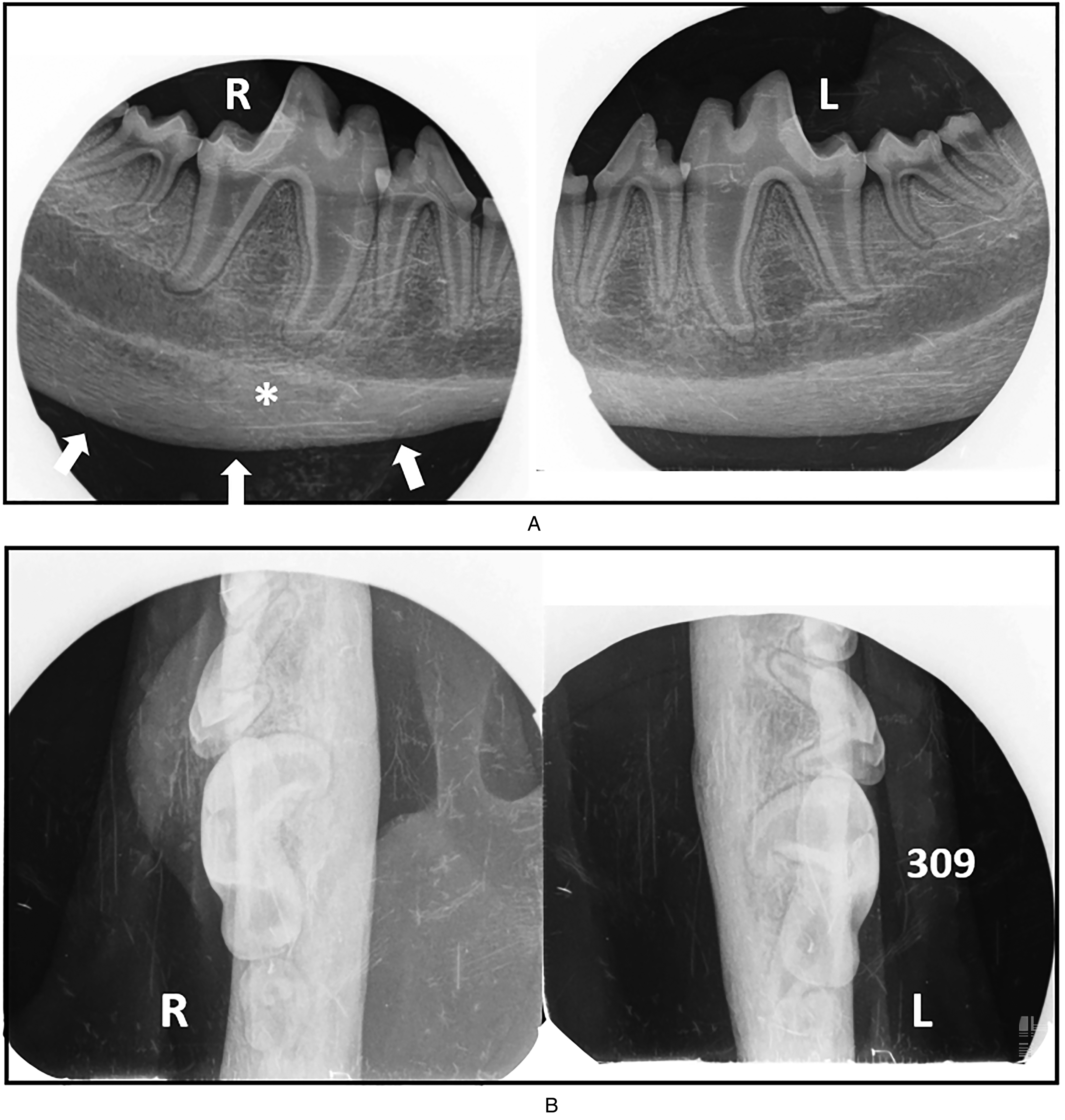
Puppy #1. Intraoral dental radiographs. (A) Lateral view, (B) Occlusal view of the distal mandibles showing a double-cortex formation on the left mandible (arrows). A radiolucent area is visible between the 2 cortices (asterisk).
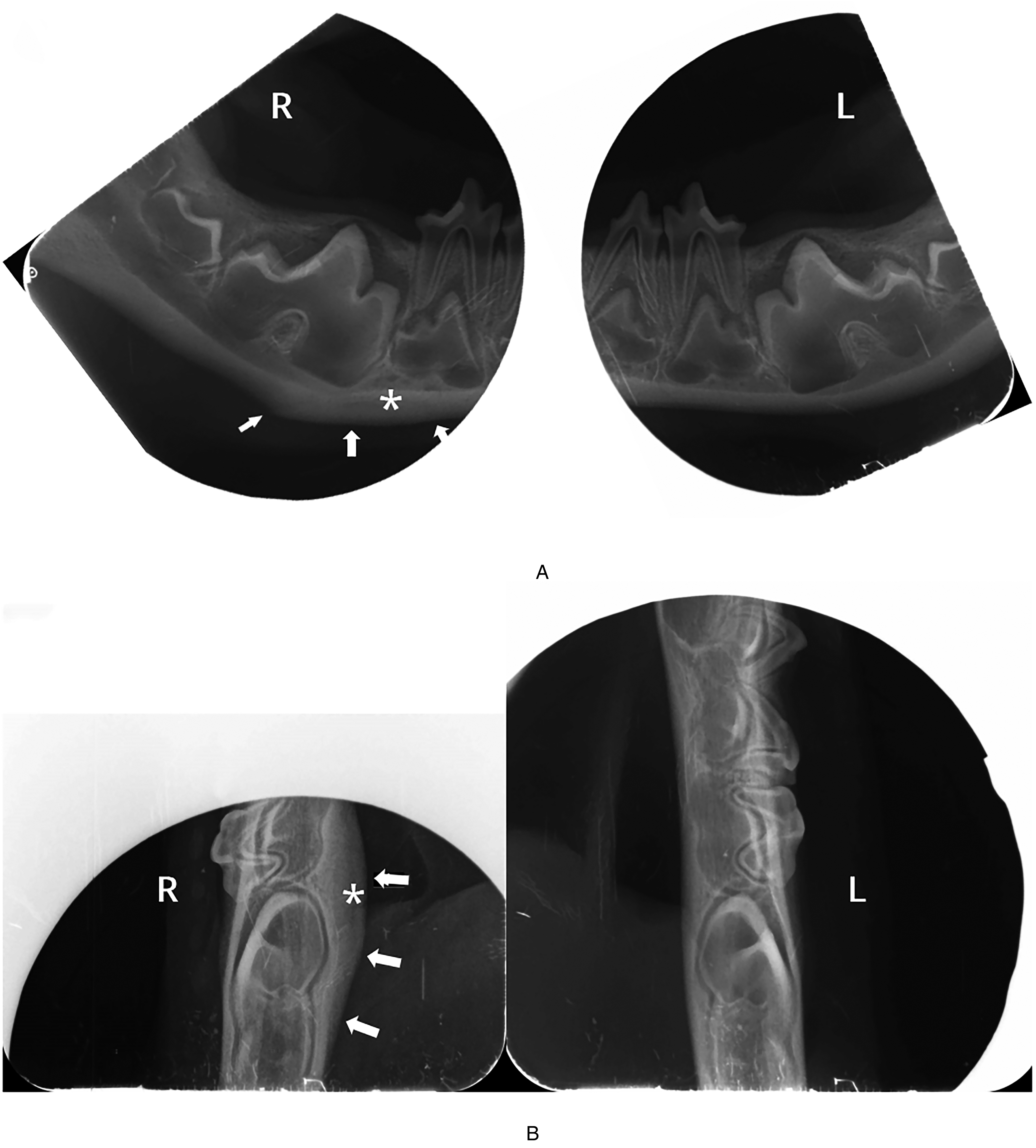
Puppy #2. Intraoral dental radiographs. (A) Lateral view, (B) Occlusal view of the distal mandibles showing a double-cortex formation on the right mandible (arrows). A radiolucent area is visible between the 2 cortices (asterisk).
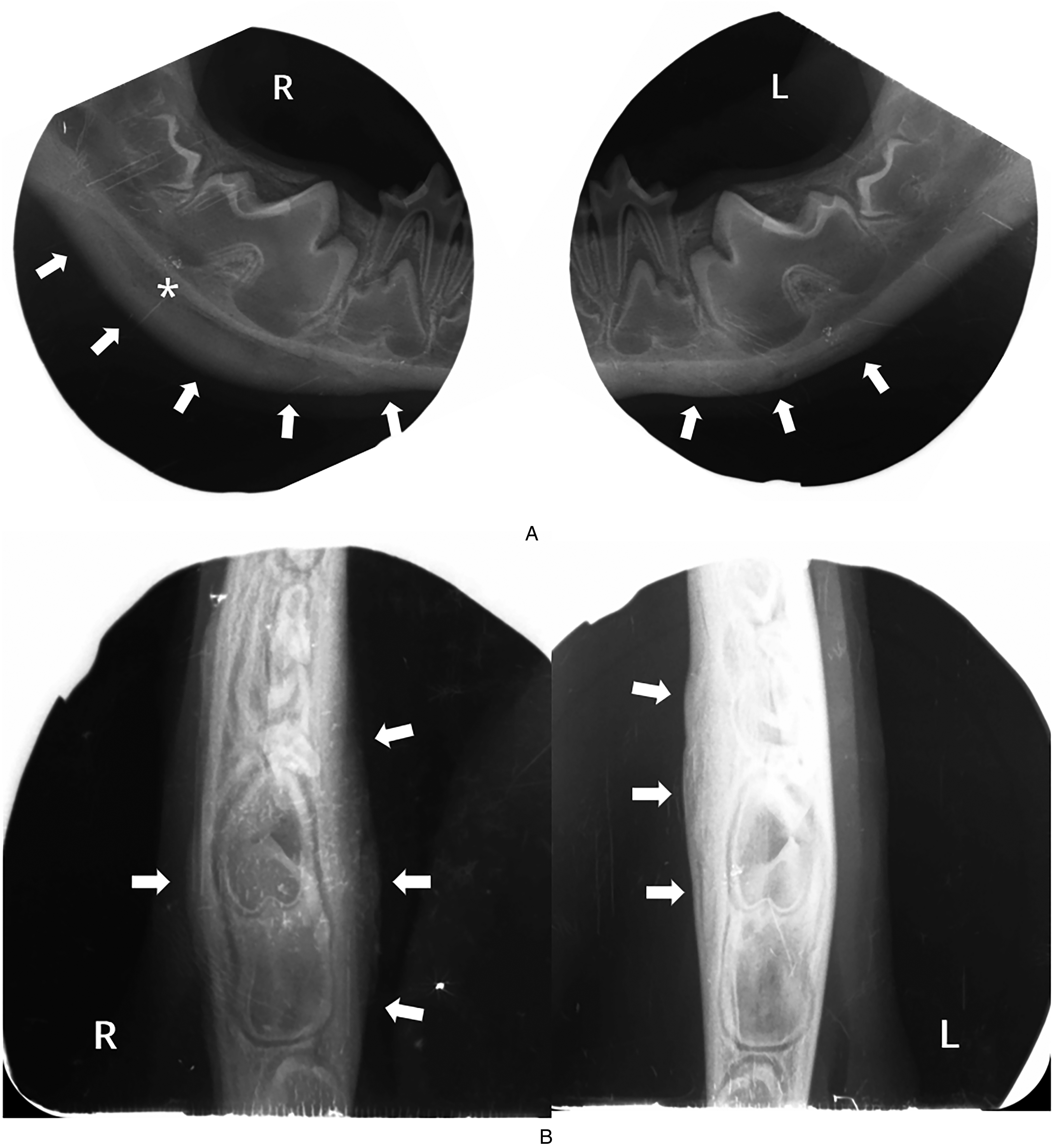
Puppy #3. Intraoral dental radiographs. (A) Lateral view, (B) Occlusal view of the distal mandibles showing a double-cortex formation bilaterally (arrows). A radiolucent area is visible between the 2 cortices (asterisk).
Computed Tomography Findings
Puppy #3 underwent computed tomography (CT) scans of the head, both pre- and post-contrast administrationb (700 mg I/kg). The scans were performed using a multi-slice scannerc (MSCT).
The CT images showed a smoothly marginated, solid periosteal reaction with bony proliferation along the ventrolingual aspects of both mandibles. A mild, similar periosteal reaction was observed along the buccal aspect of the right mandible. The bony changes were more pronounced on the right side, extending from the level of the tooth bud of the permanent mandibular right fourth premolar tooth (408) to the angle of the mandible. On the left side, the changes extended from the tooth bud of the permanent mandibular left canine tooth (304) to the angle of the mandible. There was asymmetry of the mandibular canals on each side, with the left mandibular canal having a slightly irregular contour compared to the right. No cortical lysis was observed (Figures 7, 8A and B, and 9).
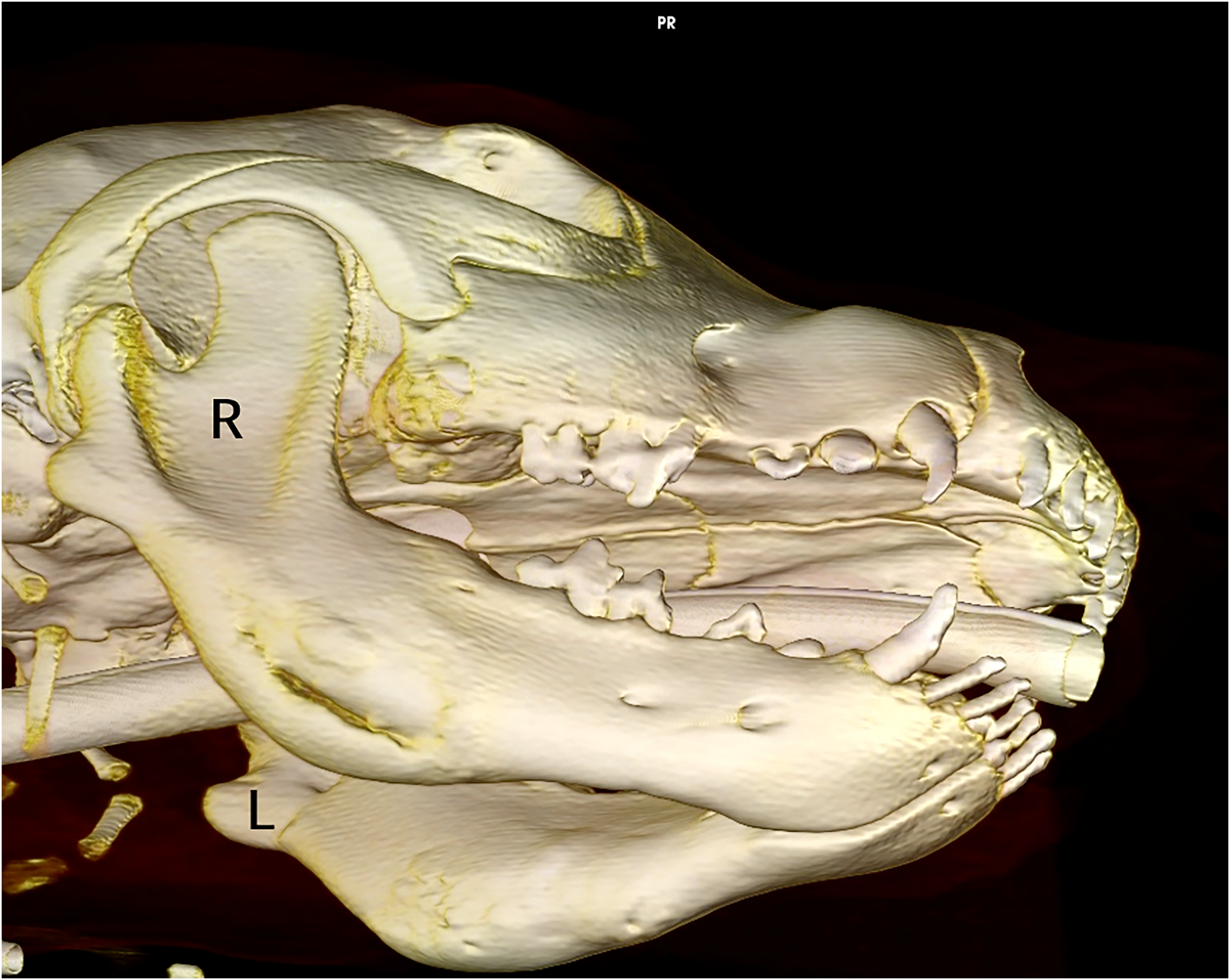
Puppy #3. A 3-dimensional computed tomography (CT) reconstruction showing bony proliferation on the cortices of both mandibles.
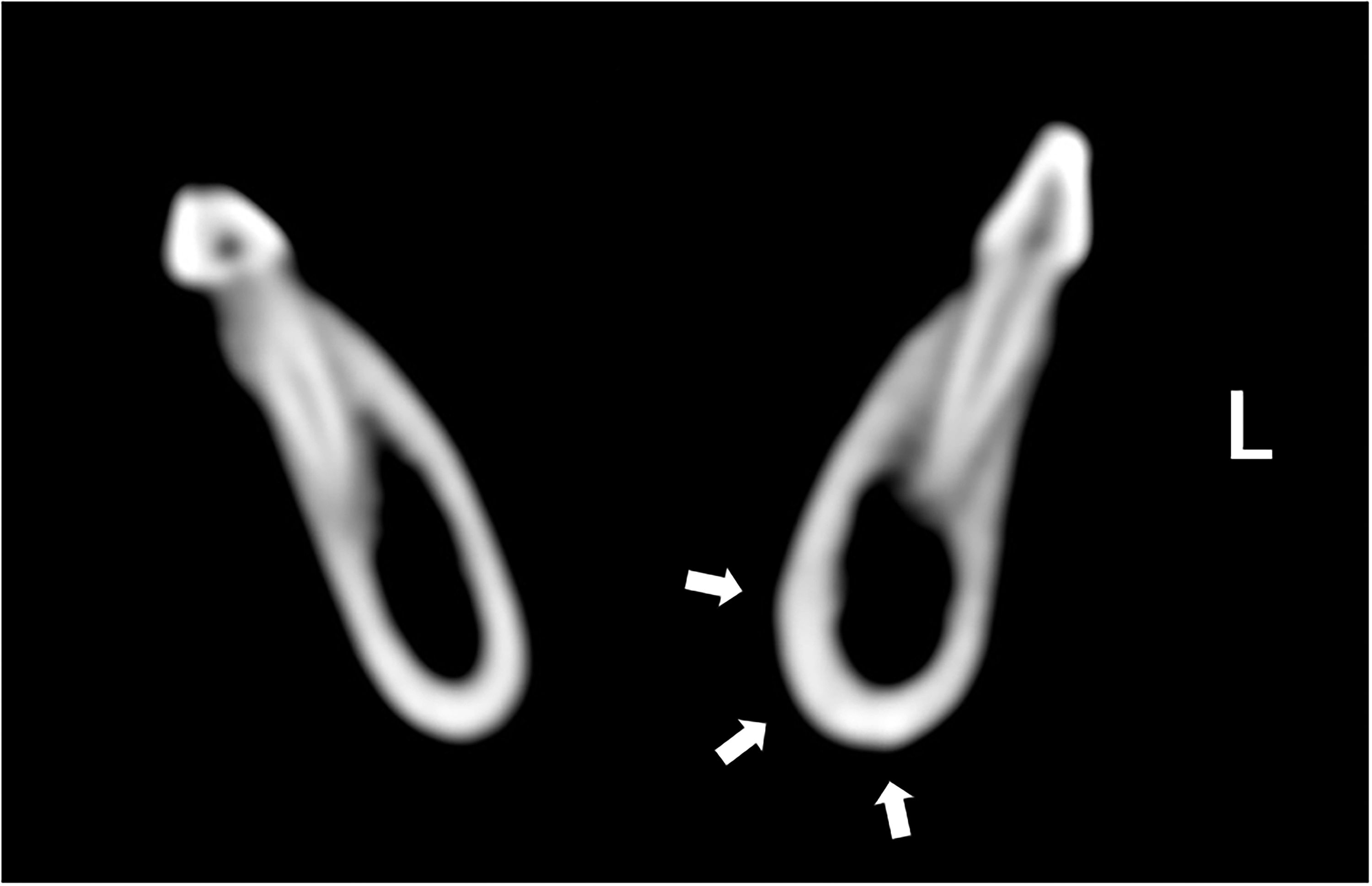
A thin hypoattenuating separation line was identified along most of the ventrolingual aspect of the right mandibular margin with the adjacent bony proliferation resembling the double-cortex appearance detected on the radiographs (Figure 8C). Two bony fragments were observed, one along the ventral margin of the left mandible at the level of the tooth bud of the permanent left third premolar tooth (307) (Figure 10A and B) and a second, larger fragment along the lingual margin of the right mandible at the level of the tooth bud of the right first molar tooth (409) (Figure 10C and D).
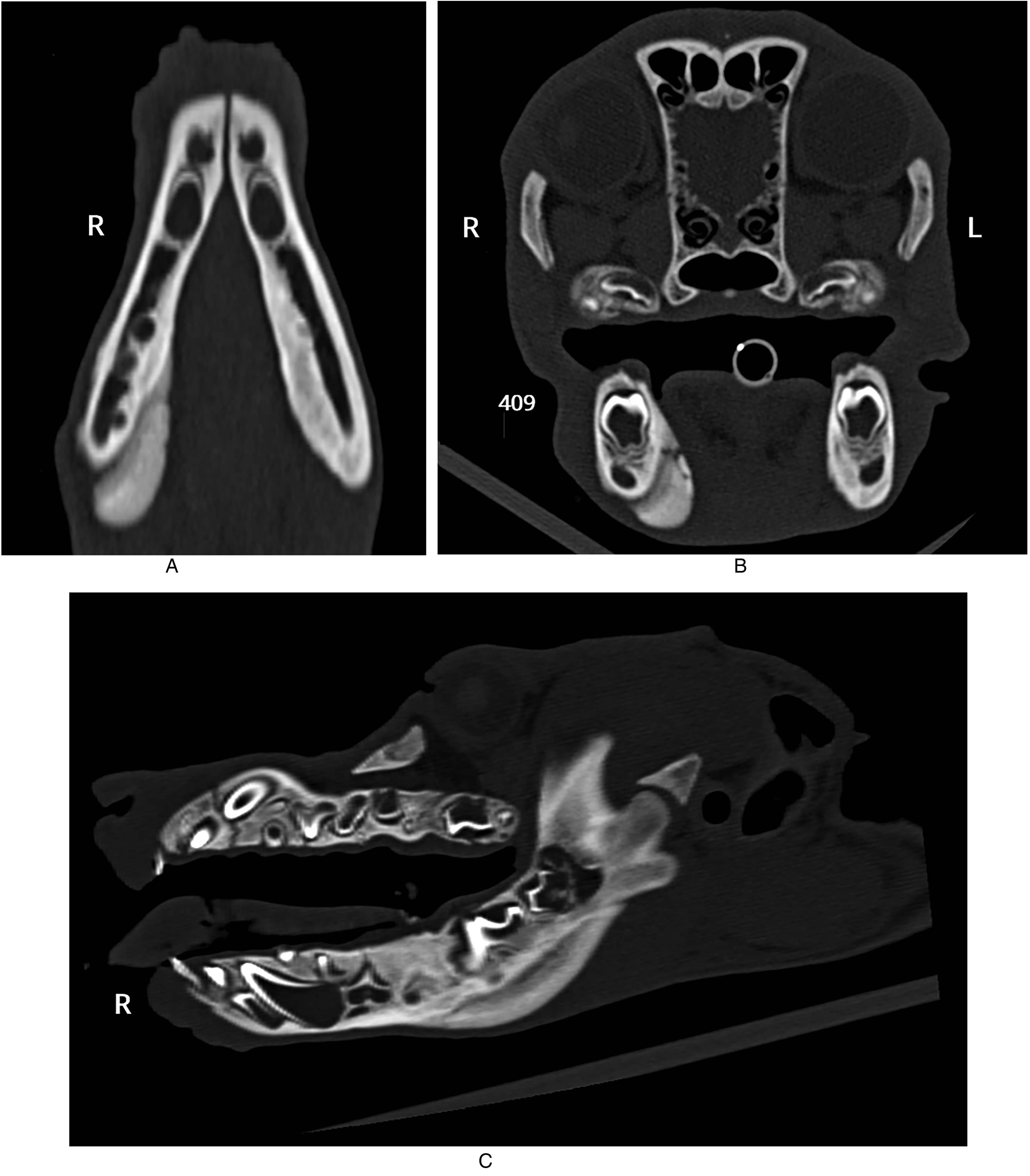
Puppy #3. Computed tomography (CT) images of both mandibles. (A) Dorsal view, (B) Transverse view at the level of the tooth bud of the mandibular first molar tooth (309/409) and (C) Sagittal view demonstrating a smoothly marginated, solid periosteal reaction with bony proliferation along the ventrolingual aspects. Notably, the right mandible exhibits a distinct periosteal reaction, with bony proliferation separate from the mandibular cortex, resembling a “double-cortex” formation.
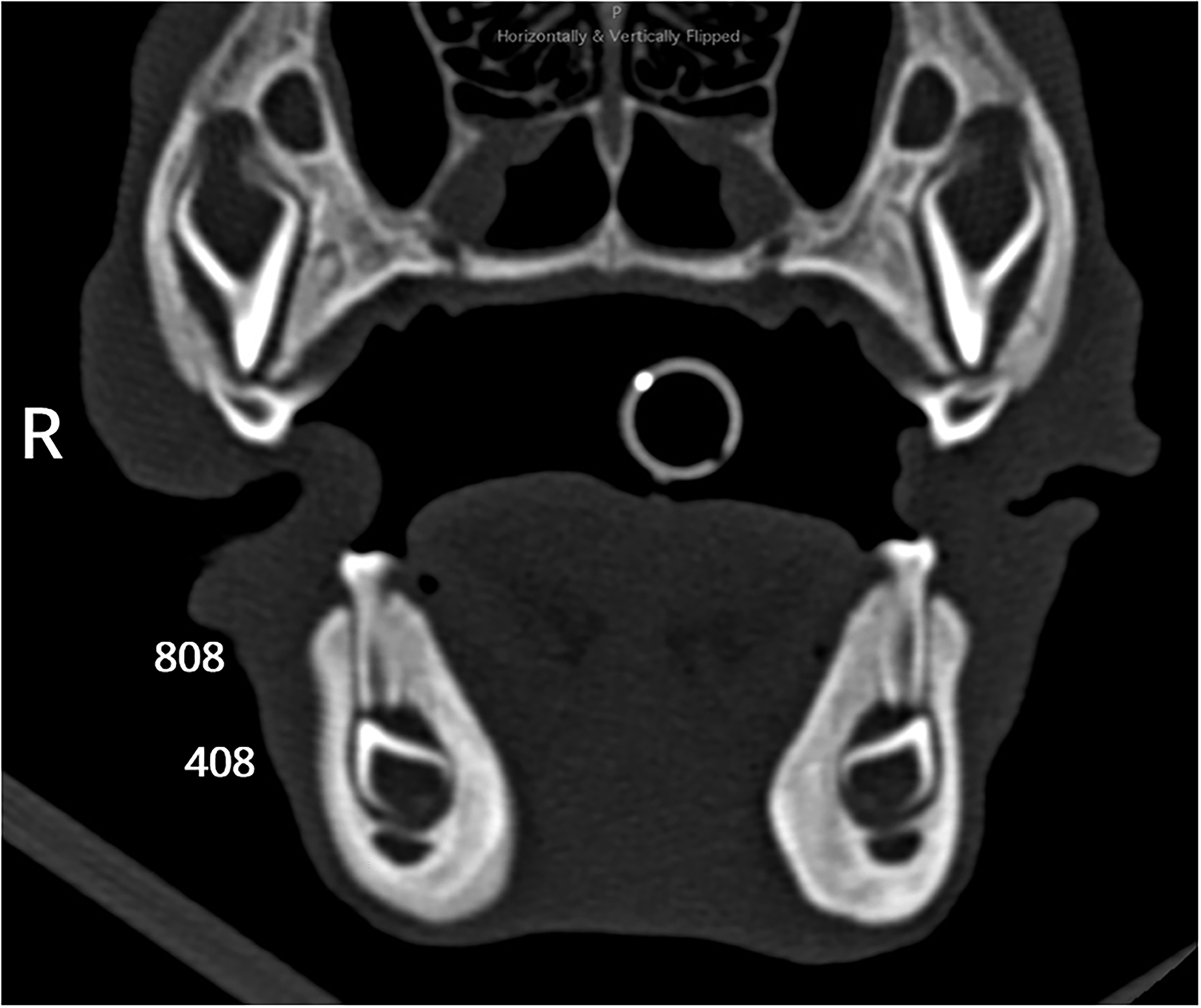
Puppy #3. Computed tomography images showing transverse view at the level of the tooth bud of the mandibular fourth premolar tooth (308/408), demonstrating a subtle focal smooth and solid bony proliferation on the buccal aspect of the right mandible.
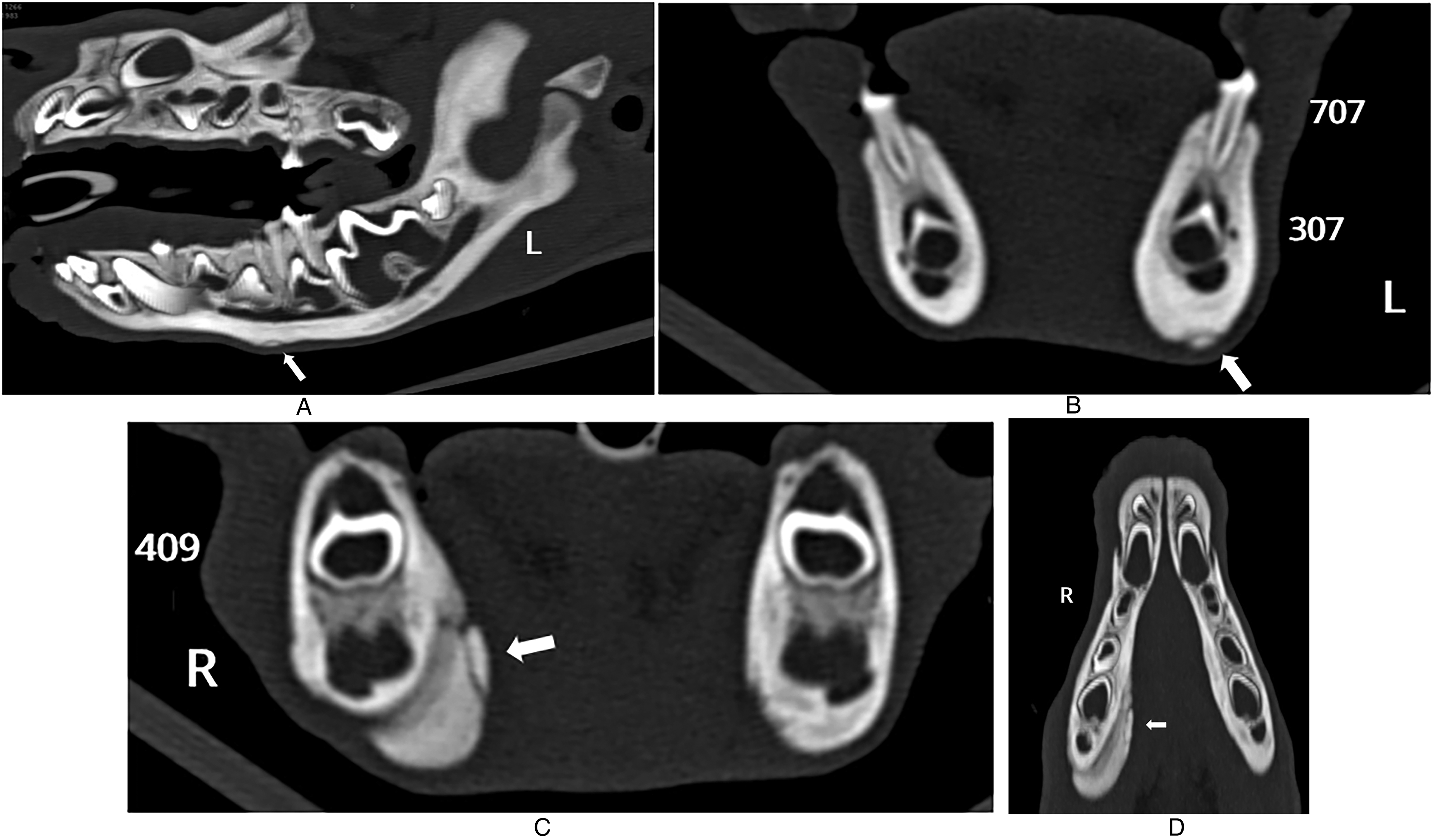
Puppy #3. Computed tomography images of small focal bony fragments in the periphery of the bony proliferation in both mandibles (arrow). (A) Sagittal view, (B) Transverse view at the level of the tooth bud of the permanent third premolar tooth (307/407) demonstrating a fragment in the left mandible. (C) Transverse view, (D) Dorsal view at the level of the tooth bud of the first molar tooth (309/409) demonstrating a fragment in the right mandible.
There was no evidence of soft tissue swelling, edema, or significant enhancement in the post-contrast images.
The described CT findings, including the smoothly marginated periosteal reaction, lack of cortical lysis, and absence of significant contrast enhancement, were most consistent with a benign, non-aggressive bone process.
Histopathology Findings
Biopsies were obtained from the left mandible of puppy #1 and the right mandible of puppy #3. The histopathologic evaluation of both samples was identical. The sample consisted of woven bone forming a lattice and fibrous connective tissue with atrophic skeletal muscle. Part of the bone was fragmented. The histopathologic diagnosis was subperiosteal proliferation. Therefore, the histopathologic findings were compatible with, but on their own not diagnostic of PO (Figure 11A and B).
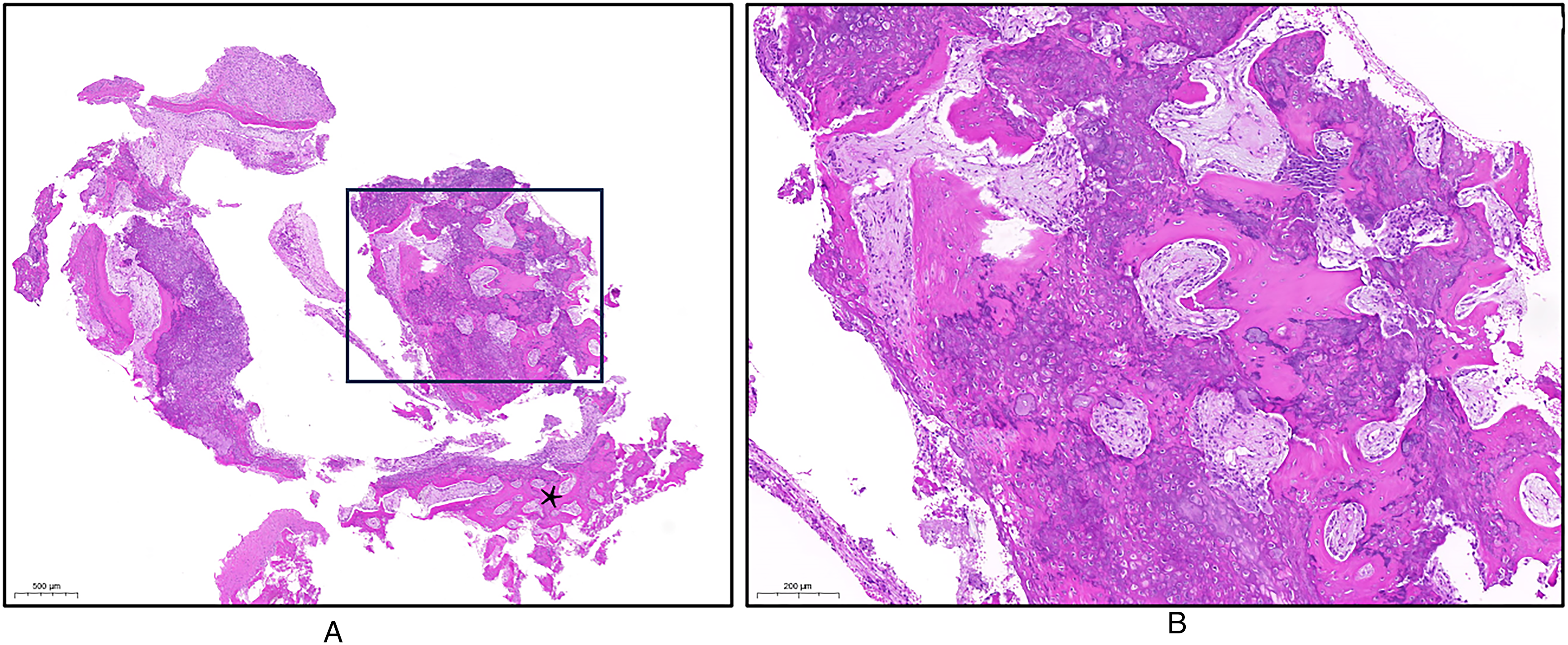
Puppy #1. Photomicrograph of the mandibular lesion [H&E]. (A) The tissue consists of woven bone. Normally cartilage is found as a cap on top of the woven bone (asterisk). (B) Higher magnification of the area within the rectangle showing woven bone (pink spicules) admixed with a large amount of cartilage (purple-blue material), characteristic of rapidly produced woven bone. In this area the cartilage is being converted into bone. Courtesy of Dr. Ori Brenner.
Treatment
Puppy #1 and puppy #3 were treated with ampicillin sodiumd (25 mg/kg) intravenously (IV) and an inferior alveolar nerve block with bupivacaine hydrochloride 0.5%e (0.3 mg/kg) for each mandible prior to collecting the biopsy. The puppies were discharged with buprenorphinef (0.02 mg/kg) q8h per os for 3 days and soft food for 2 weeks following the procedure.
Follow-up Examination
It was recommended that all puppies undergo a follow-up examination at 8 months of age. Puppy #3 was presented for follow-up at 8 months of age, puppy #1 at 11 months of age. Puppy #2 was lost to follow-up.
The puppies were anesthetized as previously described and an oral examination was performed. Puppy #1 and puppy #3 had normal dentition for their age, with all 42 permanent teeth fully erupted, normal occlusion and symmetry (Figure 12A and B). On palpation of both mandibles, no abnormalities or differences between the left and right mandibles were detected in puppy #1. Puppy #3 presented with a soft tissue swelling surrounding the biopsy site in the right mandible but no bone abnormalities were palpated (Figure 12C).
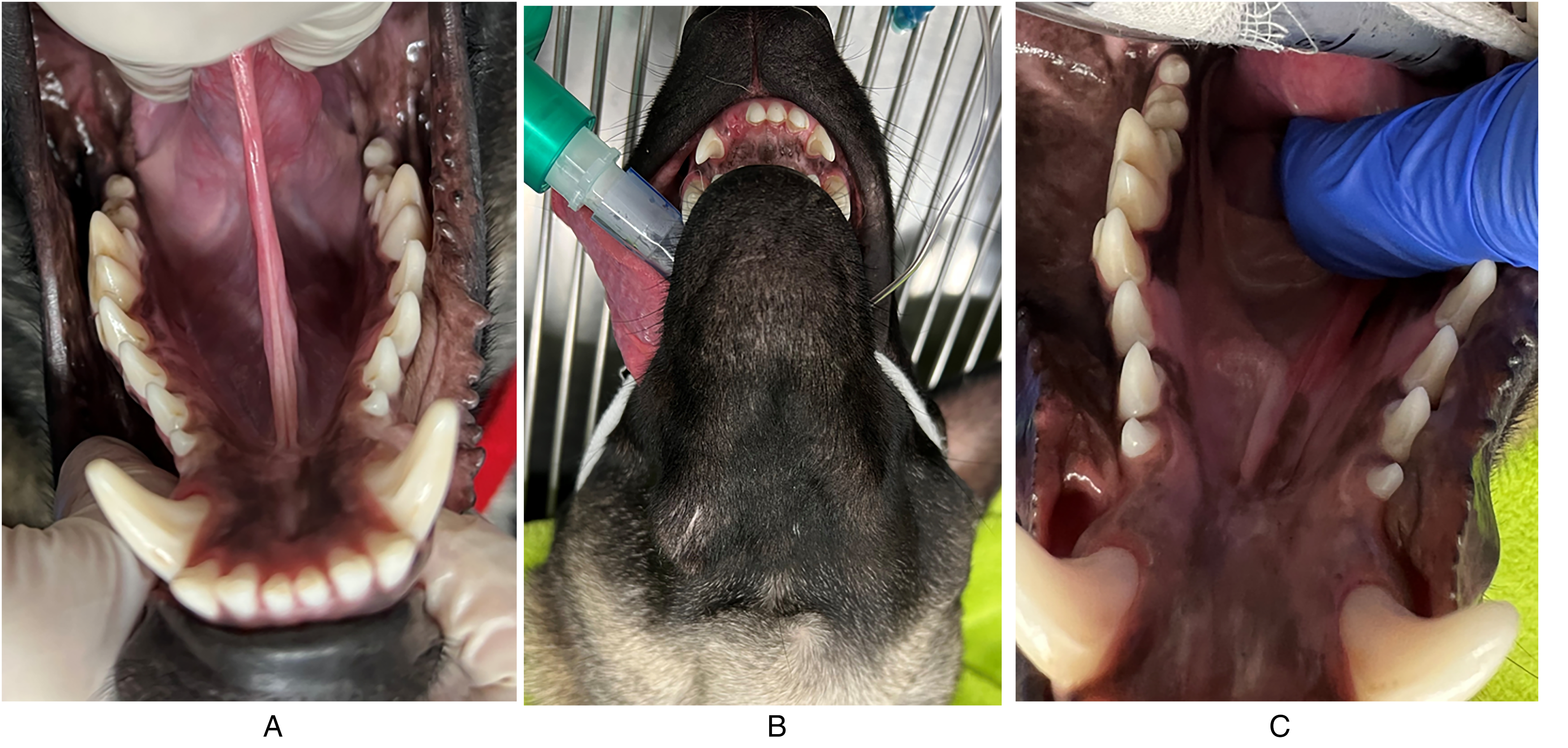
(A) Puppy #1 at follow-up examination at 11 months of age. Intraoral view showing the mandibular swelling has resolved and all permanent teeth have erupted. (B) Puppy #3 at follow-up examination at 8 months of age, ventral view showing a soft tissue swelling in the right mandible. (C) Puppy #3 at the same follow-up examination, intraoral view showing all permanent teeth have erupted.
Intraoral dental radiographs of the distal mandible revealed a significant reduction in the size of the double-cortex in both puppies, but did not have a completely normal radiographic appearance, with a double-cortex separated from the normal mandibular cortex by a radiolucent area still evident (Figures 13A and B, 14A and B).
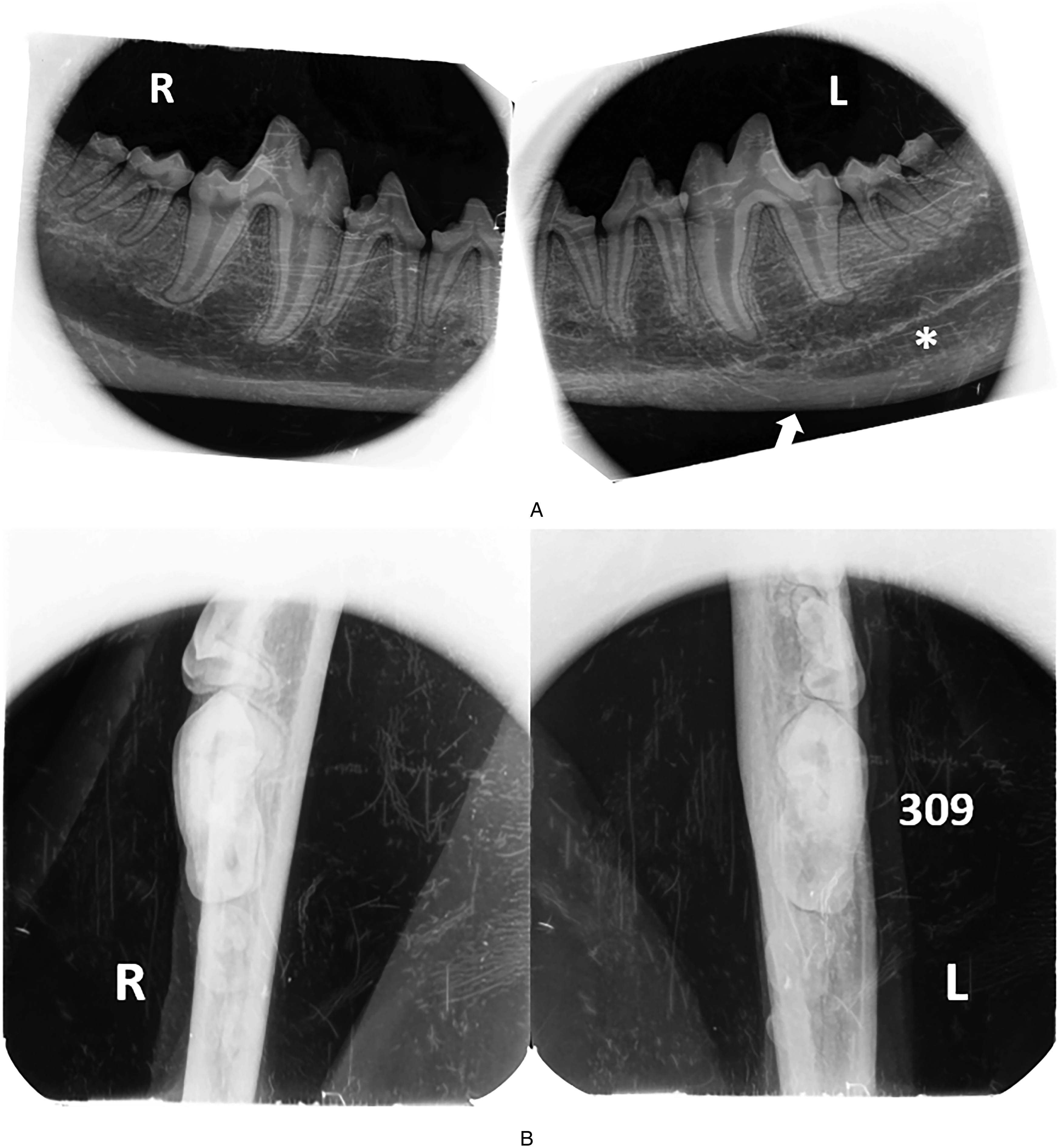
Puppy #1. Intraoral dental radiographs at follow-up examination at 11 months of age. (A) Lateral view, (B) Occlusal view of the distal mandibles. On the lateral view of the left mandible, a double-cortex is evident from the level of the first molar tooth (309) distally (arrow). Note the radiolucent area between the 2 cortices (asterisk).
Puppy #1 underwent a head and neck CT scan approximately 2 years after the initial presentation due to an unrelated condition. The scan revealed mild asymmetry in mandibular shape and size, with the left mandible being more prominent than the right. A subtle, smooth, and asymmetric cortical thickening was present along the ventrolingual aspect of the left mandible. Additionally, there was contour irregularity of the mandibular body, extending from the region caudal to the left third premolar tooth (307) to the angle of the mandible, and expansion of the left mandibular canal (Figure 15). No imaging evidence of active inflammation, osteomyelitis, or other aggressive lesions was identified.
Puppy #3. Intraoral dental radiographs at follow-up examination at 8 months of age. (A) Lateral view, (B) Occlusal view of the distal mandibles. On the lateral view of the right mandible, a double-cortex is evident from the level of the first molar tooth (409) distally (arrows). Note the radiolucent area between the 2 cortices (asterisk).
Puppy #1. Computed tomography image at follow-up at approximately 2.5 years of age. Transverse view at the level of the fourth premolar tooth (308/408) demonstrating subtle, smooth cortical thickening of the ventrolingual aspect of the left mandible along with mild widening of the left mandibular canal.
Discussion
PO refers to the diagnosis of periostitis with subperiosteal bone formation. 2 The pathogenesis is one of sequential periosteal stripping. The periosteum, lifted from the bone sequentially by an inflammatory exudate, reacts by producing woven bone.4,11 In human patients, it is recognized that PO is a condition affecting young people, most commonly occurring before the age of 25, when osteoblastic activity of the periosteum is at its peak. 3 It has been postulated that this may be related to the ease by which the young periosteum may be stripped from bone. 4 All puppies in this report were presented at 3 months of age. This is in agreement with the previous reports of PO in dogs, in which patients were presented at the age of 3 to 5 months,6,7 with a median age of 4.1 months. 6 All affected dogs, both in previous studies and in the present study, had no permanent premolar and molar dentition at the time of presentation.
In a recent human study of 38 patients diagnosed with PO affecting the long bones of the limbs, muscle attachment at the site of PO was noted in 89% of cases. 10 The authors of that study concluded that this muscle attachment leads to stripping of the periosteum resulting in soft tissue and osseous edema and centripetal ossification following traumatic injuries, including minor ones which the patients could either not recall or which they perceived as being insignificant. 10 All reported cases of PO in dogs affected the distal portion of the mandibular body.6,7 This area on the medial aspect of the mandible is the origin of the genioglossus muscle, one of the tongue muscles. The rostral belly of the digastricus muscle, the muscle which opens the mouth, inserts on the ventromedial border of the mandible in this area. 12 An interesting finding in the CT scan of puppy #3 was the 2 bony fragments along the cortical margin of the mandibles. These fragments were separated from the cortex by a hypoattenuating line, leading the authors to believe that the fragments represent an initial stage in the formation of another cortical layer as part of the pathogenesis of sequential periosteal stripping. The follow-up CT imaging of puppy #1 identified a wider mandibular canal on the affected left side when compared to the right. This finding raises the possibility that mechanical forces of muscles pulling the periosteum during mandibular growth results in mandibular canal expansion. Further studies are needed to determine if muscle attachment plays a role in the pathogenesis of mandibular PO in dogs and to what extent.
In human literature there is no gender predilection for PO, although a slight male preponderance has been reported. 4 In the veterinary literature, the 6 puppies previously reported were male.6,7 In the present study, the male:female ratio was found to be 1:2. When combining the previously reported cases with the present study, this ratio becomes 3.5:1. However, the low number of dogs diagnosed with PO so far does not permit significant statistical conclusion. More data is required to establish the statistical significance of this ratio, to determine whether gender plays a role in PO in dogs.
The histopathologic findings were compatible with PO, but on their own not diagnostic of the disease. This can be explained by the pathogenesis of PO. The histopathologic diagnosis is subperiosteal proliferation, which is nonspecific. The role of histopathology in PO is to rule out other conditions but it is not pertinent for the diagnosis. Interestingly, in spite of the role of the inflammatory exudate in the proposed pathogenesis of sequential periosteal stripping attributed to PO, inflammation was not detected in this study sections or in the sections in the previous cases reported in dogs.6,7
“Don’t touch” lesions is a concept first introduced by the human radiologist Clyde Helmes regarding bone lesions, characterizing them as those processes that are radiographically so characteristic that a biopsy or additional diagnostic tests are unnecessary to make a final diagnosis. 8 This definition was later expanded to include all imaging methods. 13 The goal of “don’t touch” lesions is to characterize lesions based on imaging and reduce unnecessary biopsies, reserving histopathology for aggressive lesions and using various imaging modalities to characterize the non-aggressive ones. 9 Obtaining a biopsy results in additional morbidity and cost 8 and, as a general rule, is indicated when histopathology will alter patient management. Any musculoskeletal lesion that can be confidently diagnosed as “benign” or “non-aggressive” based on radiological and clinical information should not be biopsied. Follow-up is indicated for cases where the lesion does not satisfy all of the classical imaging criteria but lacks aggressive features. 9 In human medicine, PO is considered a “don’t touch” lesion. This is in spite of the fact that the imaging features of PO may overlap with other bone lesions. Careful radiographic evaluation and diagnosis is required to ensure appropriate management is initiated. Traditionally, the first-line of investigation for PO in humans is radiography. CT imaging can further delineate the lesions that affect the external cortex, with preservation of the internal cortex. Magnetic resonance imaging is important to assess soft tissue involvement. 10 Contrary to a previous report in dogs which emphasized the importance of a biopsy for the diagnosis, 6 the authors believe that a biopsy is not necessary for diagnosing PO. In the authors’ opinion, signalment and the pathognomonic appearance of PO on dental radiographs and CT scans are sufficient for diagnosis of PO. Furthermore, the authors propose adopting the concept of “don’t touch” lesions in veterinary medicine. Adopting this concept presents the clinician with a diagnostic challenge. The first step is classifying the musculoskeletal lesions as “non-aggressive” or “aggressive.” Invasive diagnostic tests are considered inappropriate in the “non-aggressive” cases. This implies accepting the fact that reaching a definitive diagnosis will not always be possible in those cases which are “non-aggressive” but at the same time don’t have a pathognomonic appearance. 9 Similar to human medicine, the authors claim PO is a “don’t touch” lesion as it is non-aggressive. This does not mean that any patient presenting with a distal mandibular mass should be diagnosed with PO. A diagnosis of PO should be made only in those cases which have the pathognomonic combination of the appropriate signalment, presenting clinical signs, and imaging findings. Cases that do not fulfil all these criteria and lack aggressive characteristics should be followed up. Cases that have aggressive characteristics should be biopsied. Histopathology is the gold standard for various disease processes and is an invaluable diagnostic tool when indicated.
Several studies in humans and animals have shown that cone beam CT (CBCT) has better image quality contrast and spatial resolution of dentoalveolar and maxillofacial structures when compared to MSCT.14,15 CBCT would prove to be superior to MSCT for diagnosis of PO, unfortunately the authors did not have access to this modality at the time.
In a review of 34 human cases of mandibular PO, the most common causes of PO were an infection from dental caries (32%) and tooth extraction (26%). No definitive causative factors other than a developing unerupted tooth or tooth follicle in the vicinity of the PO lesion were found in 20% of cases. In the remaining cases (approximately 20%), the causative factors could not be determined because of insufficient clinical records or poor-quality dental radiographs that did not exhibit clear pictures of tooth structures. 16 A previous veterinary study suggested that mandibular PO in large breed puppies resulted from an inflamed or infected dental follicle, developing unerupted tooth (most likely the permanent mandibular first molar tooth [309/409]), and/or is secondary to pericoronitis. 6 In the authors’ opinion, all the cases described in dogs to date were a reaction to mild inflammation related to tooth eruption. This inflammation is self-limiting and will spontaneously resolve following completion of the eruption process. Therefore, no treatment is required other than pain management and monitoring until the teeth emerge. It is expected that the swelling due to the bony reaction will resolve only after tooth eruption is completed.
Although the left mandible of puppy #1 was normal on palpation at a follow-up examination 8 months post diagnosis, dental radiographs obtained at the same visit revealed that the affected mandible did not have a completely normal radiographic appearance. The double-cortex, although smaller, was still present. Similar changes were found in puppy #3 at the 5 months post diagnosis follow-up. Changes both in bone and mandibular canal width were also detected on a head CT scan of puppy #1 performed 2 years post diagnosis. These findings are consistent with the previous report, where a reduction in the mandibular swelling was noted over time, and dental radiographs obtained 1 year post diagnosis showed changes similar to the present study. 6
All the puppies in this report were from the same litter, with at least 3 puppies affected out of 4. The prevalence of PO in this litter was therefore 75% if the fourth puppy did not suffer from PO, or 100% if it did. The authors found no reference in the human literature regarding a genetic basis for this disease. A genetic basis can be more easily suspected in animals when dealing with litters. When considering environmental versus genetic causes for a disease it can be argued that the calculated prevalence of 75% in this litter is highly suggestive of environmental causes. However, it must be considered that there is a high level of inbreeding in dogs in the authors’ country, which can lead to an increase in the expression of genetic diseases. If a genetic component is established in dogs, it may provide the initial impetus to commence research in human patients.
CMO17,18 and CHS 19 are self-limiting proliferative bone diseases of the skull in juvenile dogs that should be considered when forming differential diagnoses for mandibular PO.
CMO is a self-limiting, idiopathic, hyperostotic lesion commonly diagnosed in, but not limited to, West Highland White and Scottish Terriers aged 4 to 7 months. 17 Clinical signs include swelling and pain around the cranium, mandibles, and temporal regions, difficulty opening the mouth, excessive salivation, lethargy, and anorexia. 18 Multiple skull bones are affected concurrently, including the mandibles, temporal, occipital, and less often, the maxillary bones. 17 Lesions are usually bilateral, but can also be unilateral. The proliferative new bone formation typically ceases at around 1 year of age, when skeletal maturation is complete.17,18 Histologically, CMO is characterized by proliferative bone extending from both the periosteal and endosteal surfaces, a combination of both woven and lamellar bone. Some degree of mixed inflammation may also be present within the remodeling bone. 17
CHS is a self-limiting proliferative bone disease of the skull that was described in 5 male bullmastiffs, aged 5 to 10 months. Cases were presented with painful unilateral or bilateral masses on the frontal, temporal, and occipital bones. Radiographic findings revealed periosteal proliferation. Histologically, remodeled subperiosteal trabeculae of woven and lamellar bone were seen, and a few sections showed neutrophilic infiltrate. It was suggested that the similarities between CMO and CHS may imply a genetic component in CHS, similar to CMO. 19
A more recent case report described an 8-month-old Pit Bull Terrier that was presented with painful swellings in the region of the frontal sinuses. 20 A CT scan of the head revealed periosteal bone formation and hyperostosis on the frontal, parietal, and mandibles bilaterally. Histologically, thick trabeculae of immature woven and mature lamellar bone were seen. The diagnosis was CMO with similarities to CHS. Since Pit Bull Terriers are not predisposed to either of these diseases; and given the clinical, radiographic, and histological similarities between CMO and CHS; the authors suggested that CMO and CHS may represent a single disorder, with predilection sites that vary between breeds of dogs. They proposed to include both CMO and CHS under a new term, “idiopathic canine juvenile cranial hyperostosis.” 20 The recently published case report of mandibular PO in a Great Dane suggested that given the similarities between CMO, CHS, and mandibular PO, mandibular PO may also fall into this category. 7 The authors’ suspicion of a genetic basis for mandibular PO strengthen this hypothesis. The similarities between these 3 self-limiting proliferative bone diseases of the skull in juvenile dogs combined with the fact that a genetic component is implied in all of them cannot be ignored. The authors support grouping them as the single disorder, “idiopathic canine juvenile cranial hyperostosis,” with predilection sites that vary between breeds of dogs. Furthermore, the authors propose acknowledging that all 3, separately or as a group, are skeletal “don’t touch” lesions.
Conclusions
PO is a chronic subtype of osteomyelitis, affecting young large-breed dogs. The insinuating factor is mild inflammation related to tooth eruption.6,7 All the cases described to date in dogs were presented with a non-painful distal mandibular swelling, prior to the eruption of the permanent mandibular fourth premolar (308/408) and first molar (309/409) teeth. Since this inflammation is both sterile and self-limiting, no treatment is required. Over time, a significant reduction in the swelling will be detected, although advanced imaging may reveal that the bone and mandibular canal do not return to a completely normal structure. Histopathology serves to rule out other conditions since the findings in PO are nonspecific. When weighing the contribution of histopathology to the diagnosis versus the morbidity caused when obtaining a bone biopsy, the authors find biopsy to be unnecessary in these cases. Diagnosis should therefore be based on imaging, such as dental radiographs, CBCT, and/or MSCT, as the changes detected by these modalities are pathognomonic. Furthermore, in the authors’ opinion, PO should be considered a “don’t touch” lesion due to its non-aggressive features.
Based on the findings of the present study, and on those of the previous cases reported in the veterinary literature, mandibular PO should be suspected in any large breed puppy presenting with a non-painful distal mandibular swelling.
Furthermore, the authors support grouping mandibular PO, CMO, and CHS as a single entity, “idiopathic canine juvenile cranial hyperostosis,” and acknowledge that all 3 are skeletal “don’t touch” lesions.
Materials
(a) Synulox, Haupt Pharma Latina S.R.L., Italy
(b) Iohexol (Omnipaque 350), GE Healthcare Ireland, Cork, Ireland
(c) Philips Brilliance 40-slice Multi-Slice Computed Tomography, Koninklijke Philips N.V., Amsterdam, Netherlands
(d) Penibrin, Sandoz GmbH, Kundl, Austria
(e) Bupivacaine-Baxter, Baxter Pharmaceuticals India Private Ltd, Vasana-Chacharwadi, Ahmedabad-382 213, India
(f) Bupaq, Richter Pharma AG, Durisolstrasse 14, 4600 Wels, Austria
Footnotes
Acknowledgments
The authors would like to thank Dr Ori Brenner, BVSc DACVP for the histopathology reports and photomicrographs; and Dr Natalia Vapniarski-Arzi, DVM, DACVP for her help and valuable input.
Ethical Considerations
All participants provided a verbal informed consent for the publication of any potentially identifiable images or data included in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
