Abstract
Many authors recommend abrasive diets to prevent dental overgrowth in pet rabbits. Research, however, indicates that growth adapts to wear. Furthermore, growth in the absence of wear has only been qualitatively but not quantitatively determined. The present study therefore aimed to quantify the growth–wear relation in rabbit incisor teeth by inducing minimal to maximal wear and using the obtained data as well as previously published values. Twelve rabbits were fed a hay-only diet as a control or a gruel as a low-wear diet in a cross-over design for 21 days each. Thereafter, in all animals while on the hay-only diet, the left incisor pair was repetitively trimmed out of occlusion for a maximum of 54 days. Animals received ample gnawing materials except when gruel-fed. Tooth length, growth, and wear were documented throughout. The diet affected proxies (i.e., measurements) only in the maxillary incisors. Growth in response to dental trimming on the other hand was faster in the mandibular incisors. Data indicate a linear adaptation of growth to wear with slopes lower than 1 and a positive intercept in maxillary and mandibular incisors. Although this proposes basal growth and incomplete responsiveness to wear, measurement methods likely cause underestimation and overestimation of the slopes and intercepts, respectively. Importantly, wear always occurs, even when rabbits are fed liquid diets without gnawing opportunities. The present study confirms the adaptive character of growth to wear in rabbit incisors and questions the necessity of dietary abrasiveness to prevent incisor overgrowth.
Introduction
Throughout evolutionary time, an increasing number of mammalian lineages have developed altered ontogenetic patterns, causing different types of hypsodont teeth to evolve from their brachydont basis.1–4 Although differing in extent, all hypsodont types are characterized by greater crown heights and crown-to-root ratios compared to brachydont teeth. This is thought to render dentition more durable, which in the case of herbivores is an adaptation toward processing of more abrasive food.2,4–6 The most extreme form is termed euhypsodonty or hypselodonty, where tooth roots are never formed and are replaced by cervical loops, resulting in “ever-growing” teeth.2,3,7 The best examples are the incisors of lagomorphs and rodents. 2 Many species additionally have hypselodont cheek teeth, among them are popular pets such as rabbits, guinea pigs, degus, and chinchillas.1,8–10
In contrast to their putative durability against abrasion, however, ever-growing teeth appear to make animals more vulnerable to the development of dental problems in a domestic environment. This is evident from the high prevalence of dental disease in hypselodont-companion animals, ranging from 12% to 90% depending on the studied population and examination methods used.11–18 Since malocclusion with concurrent dental overgrowth is the main manifestation,11,18 an evidently assumed factor in its etiology is inadequate abrasion of teeth by the food.19–21 To prevent overgrowth, many authors therefore recommend feeding such pets abrasion-promoting diets.20–24
The concept that abrasive diets are required to prevent dental overgrowth, however, should be questioned. First, on a more logical basis, biological plausibility should be considered. Such a condition implies that animals in their natural habitat would need to constantly select a diet of adequate net abrasiveness to prevent detrimental dental overgrowth. Evidently, this would impose a constraint on diet selection with little energy content or nutritive value for the animal and is therefore unlikely to have evolved. Additionally, it seems unlikely that a system of constant growth would evolve without an adequate feedback control to allow a reaction to actual, temporary circumstances.
Second, the concept does not correspond well to empirical data. Many studies have demonstrated that the magnitude of growth in hypselodont teeth indeed alters in relation to the magnitude of wear. This was already reported for rabbits at the beginning of the 20th century,25–28 and has since been repeated for rabbits and guinea pigs.29–35 Many authors suggest a change in occlusal pressure as being the main regulator. This is particularly supported by the increased growth rates of incisors that have been artificially shortened “out of occlusion,”25–27,33 as well as the reduced growth rate following the application of controlled pressure on them.36,37
Although growth of hypselodont teeth changes according to wear, it could still be argued that very low wear might represent a risk. This concept is, however, difficult to test. Animals with hypselodont teeth would have to be fed a “soft” or “liquid” diet that does not require the use of these teeth. To the authors’ knowledge, only 2 such studies have been conducted in rabbits. The first study kept rabbits for 3 weeks on gruel, in glass cages without any gnawing opportunities. 25 Compared to a conventional diet requiring chewing, tooth growth on the liquid diet was reduced by up to two-thirds. This study, however, only reported tooth growth but not wear. The second study maintained rabbits for a year on a liquid diet, for reasons unrelated to dental biology, without measuring tooth wear or growth. 38 While dental health was not mentioned in the publication, no dental problems were observed (pers. comm., 2017).
The present study aimed to quantify growth rates of rabbit incisor teeth in response to a large range of wear, by feeding diets requiring different incisor action (gruel diet versus hay diet), and by artificially maximizing wear through dental trimming. In addition, the findings from this study were compared to collected data from the scientific literature, to compile the existing evidence on the relation between tooth wear and incisor growth in rabbits.
Materials and Methods
General Study Design
The study consisted of 2 periods, over 82 days, and was conducted under the approval of the Cantonal Veterinary Office in Zurich, Switzerland (No. 35593; ZH041/2023). The first period evaluated the effect of dietary format on incisor wear and growth, whereas the second period investigated the change in growth rate after artificially increasing wear by dental trimming.
Dietary Format — The first period comprised of a cross-over design, in which 12 rabbits were randomly divided into 2 groups of 6 animals. For 21 days, each group received either a hay-only diet (control, C), expected to lead to relevant incisor wear, in enclosures equipped with ample gnawing opportunities, or a gruel-only diet (low wear, L) that did not require incisor action for ingestion in enclosures without gnawing opportunities. Incisor wear and growth rates were recorded throughout.
Dental Trimming — Following the first period, all animals were fed the same hay-only diet during the second period (trimming, T) with ample gnawing opportunities. After 1 week of adaptation and for a maximum of 54 days, dental wear of the left incisor pair was artificially maximized in all rabbits by subsequent trimming to prevent return to occlusion. Incisor “wear” (the length trimmed) and growth rates were recorded throughout.
Animals and Husbandry
Intact female rabbits were purchased from Charles River Laboratories, Châtillon, France, a specific pathogen-free breeding facility. From their arrival (age 5-6 weeks) until the end of the study, the animals remained within their study environment. During a 26-week adaptation period, training for habituation to the study procedures (weighing, restraint, and dental examination) was conducted at least twice weekly. The study started when animals exceeded the age of 31 weeks. Two rabbits on the gruel diet shortened their lower incisors by excessive rubbing (gnawing was not possible) against the enclosure. As this would have exerted a large effect on the recorded measurements, the study was repeated with 2 replacement animals.
During the adaptation period, as well as the C and T periods, rabbits were group-housed in the same room within an enclosure of 7 to 8 m2. Each group was provided with 5 hay racks, 14 to 16 nipple drinkers, 2 toy types (various balls and chains exchanged weekly), 6 wooden shelters, half of which also functioned as elevated platforms, and varying additional gnawing opportunities. When rabbits were fed diet L, they were kept in pairs in the same room in 2 m2 enclosures, with visual contact to other group members. Each pair was provided with 4 nipple drinkers, 2 ceramic feeding bowls, 1 interchanging (nongnawable) toy type and a nongnawable shelter. All other gnawing opportunities were eliminated during L. The bedding of all enclosures consisted of wood shavings. Natural lighting was provided through a window with additional artificial lighting ensuring a constant 12 h cycle from 06:00 to 18:00. The temperature was maintained between 18 and 20°C.
Diets and Sampling
Rabbits received hay for ad libitum consumption complemented with a pelleted dieta until they were 16 weeks of age. The latter was partly fed as a gruel to habituate the animals to this format. For the rest of the adaptation period, only a mixture of different hay types was provided for ad libitum consumption. The hay used during the study (C and T) was also fed for ad libitum consumption and comprised of an artificially dried third cut (type GR, stage 5) 39 from a natural pasture (Table 1). The gruel diet, on the other hand, was fed with restriction. Per pair, the allotted quantity was based on the calculated average digestible energy (DE) requirement per kg metabolic weight of the group when fed a hay-only diet during the adaptation phase. The change from a complete hay diet to the gruel occurred gradually over 5 days at 20% increments on DE basis to avoid gastrointestinal disturbances. The nutrient content of the hay and gruel diets was determined by an accredited laboratoryb or by the manufacturerc respectively. The DE content of all diets was calculated according to Villamide et al. 40 Prior to feeding, diet L pellets were pulverized, mixed with water (1:3 w:w) and fed as a gruel (Table 1). Food intake was monitored daily during the first period of the study. For measurement of intake of the gruel diet, left-over feed was corrected for moisture loss by exposing a portion of gruel to the same environment but out of the rabbits’ reach and using the weight difference as a correction factor.
Experimental Diets’ Content of Selected Nutrients.
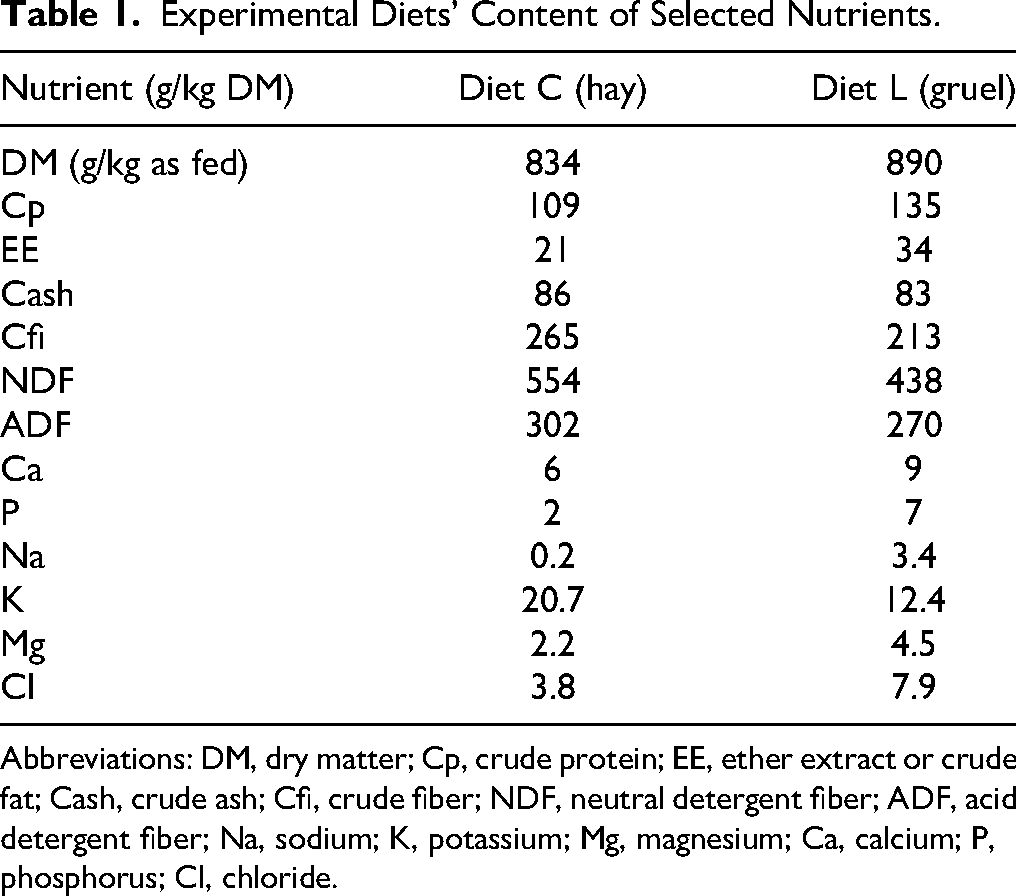
Abbreviations: DM, dry matter; Cp, crude protein; EE, ether extract or crude fat; Cash, crude ash; Cfi, crude fiber; NDF, neutral detergent fiber; ADF, acid detergent fiber; Na, sodium; K, potassium; Mg, magnesium; Ca, calcium; P, phosphorus; Cl, chloride.
The animals’ weight and body condition were monitored daily and weekly during pair- and group-housing, respectively. When feeding gruel, the allowed quantity of feed per pair was adapted with 10% changes per week, depending on weight changes. Hay was fed as 2 equal portions in the morning and the afternoon. To ensure liquid consistency, gruel was provided in 3 meals per day of which the latter comprised of 50% of the daily allowance.
Dental Measurements and Manipulations
Inspections of the incisors were conducted at least twice weekly, and procedures were only carried out when necessary.
Dietary Format — To study the growth and wear rates of all 4 incisors during C and L periods, each incisor was marked on its labial side (0.2 mm deep) with a dental diamond-tipped burd. The distance from the marking's edges to the gingiva and to the crown's occlusal surface, as well as the total tooth length were first determined with a Castroviejo-type calipere (accuracy 0.1 mm) and subsequently measured with a regular or digital caliperf (accuracy 0.01 mm). Tooth growth and wear were calculated as the difference in distance between 2 measurements, from the gingiva to the marking and from the occlusal surface to the marking, respectively (Figure 1A). Four measuring points were considered for analysis. From 0 to 3, they comprise 0, 7, 14 and 21 days after initiation of the study or the diet switch. Values obtained represent a summation of the separate measurements within the respective week.
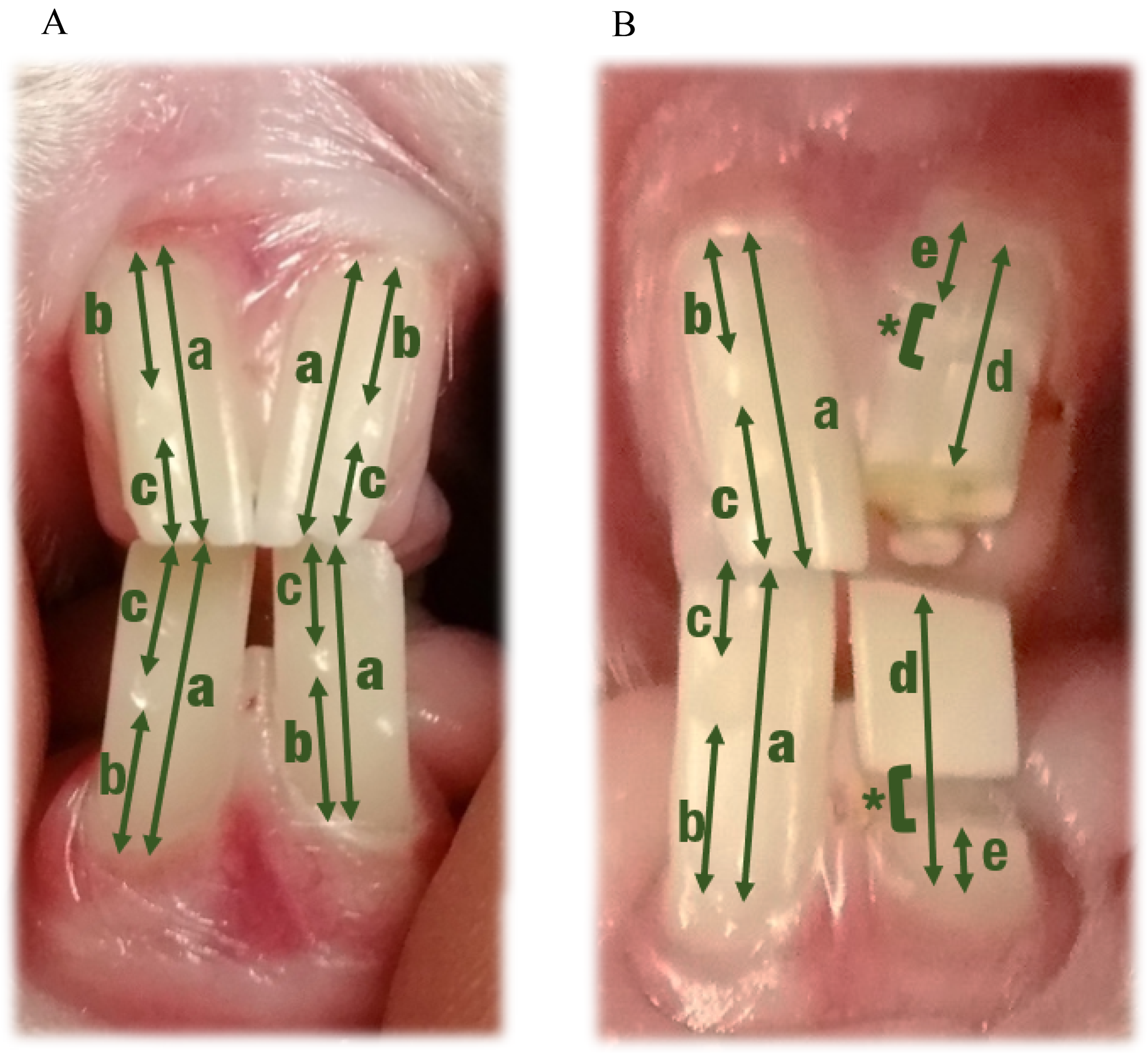
Measurements taken to determine dental proxies during the dietary (A) and the trimming periods (B). In uncut teeth (all teeth during the dietary period and the right incisor pair during the trimming period), length was directly measured (a), whereas growth and wear rate were calculated as the difference in (b) and (c) respectively, between measurements. In cut teeth (left incisor pair during the trimming period), growth rate was calculated as the difference in tooth length between cuttings (from (e) in previous measurement to (d) in present measurement), whereas wear rate was calculated as the difference in tooth length due to cutting (from (d) in present measurement to (e) in present measurement). Wear was not taken as the length of the cut off piece due to the variable loss of dental tissue by the cutting procedure (*).
Dental Trimming — The same procedure as described for the dietary format period was used for the occlusal right incisor pair. The nonocclusal left incisor pair was cut at 2.1 to 5.4 mm from the occlusal surface with a diamond disk burg at the beginning of T, and for 7 subsequent times when approaching occlusal length. Growth for the nonocclusal incisor pair was calculated as the increase in tooth length between 2 cuttings. The difference in tooth length due to cutting was taken as a value for wear (Figure 1B). The end point of the study was set at 8 tooth length reductions per animal. Nine measuring points (the initial value and after each cutting event) were used for analysis. At each measuring point, average daily growth and wear were calculated and transformed to a nominal value in mm per week for analysis. Measuring points 1 to 9 correspond with 0, 6, 12 to 13, 18 to 19, 25 to 26, 32 to 33, 38 to 39, 43 to 45, and 47 to 51 days after the first cut.
Literature Data
To contextualize the relationship between tooth growth and wear, data obtained from the present study was plotted alongside previously published values. Only studies that measured both growth and wear of rabbit incisors (maxillary, mandibular, or both) or from which these values could be deduced were included in the comparison (Figures 2 and 3).19,29–33,35,41
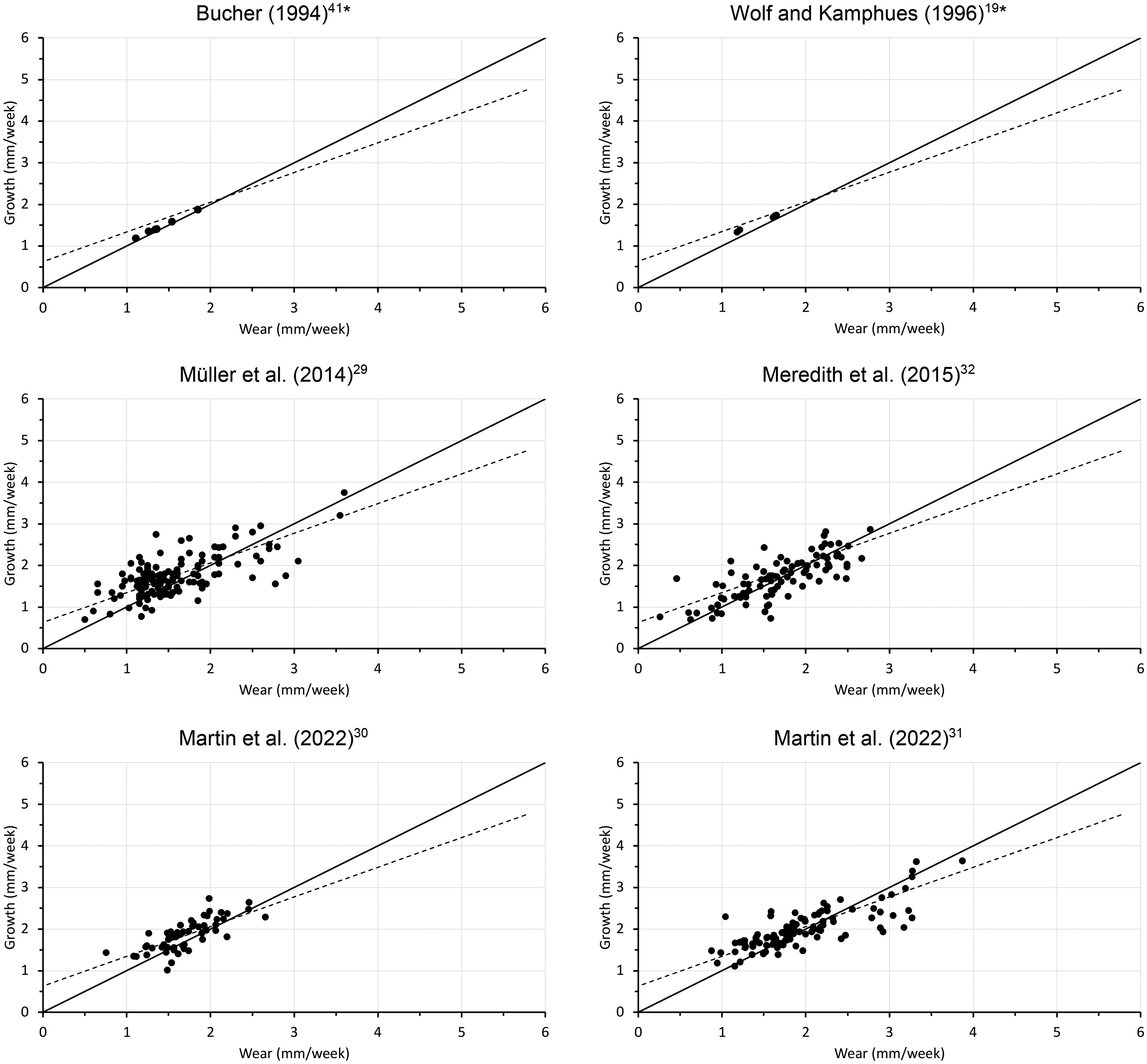
Relationship of maxillary incisor wear and growth from previous published studies. The solid black line represents y = x, and the dotted black line is the regression line for the whole dataset (Figure 11). * Denotes average values for different diets (individual data not available).
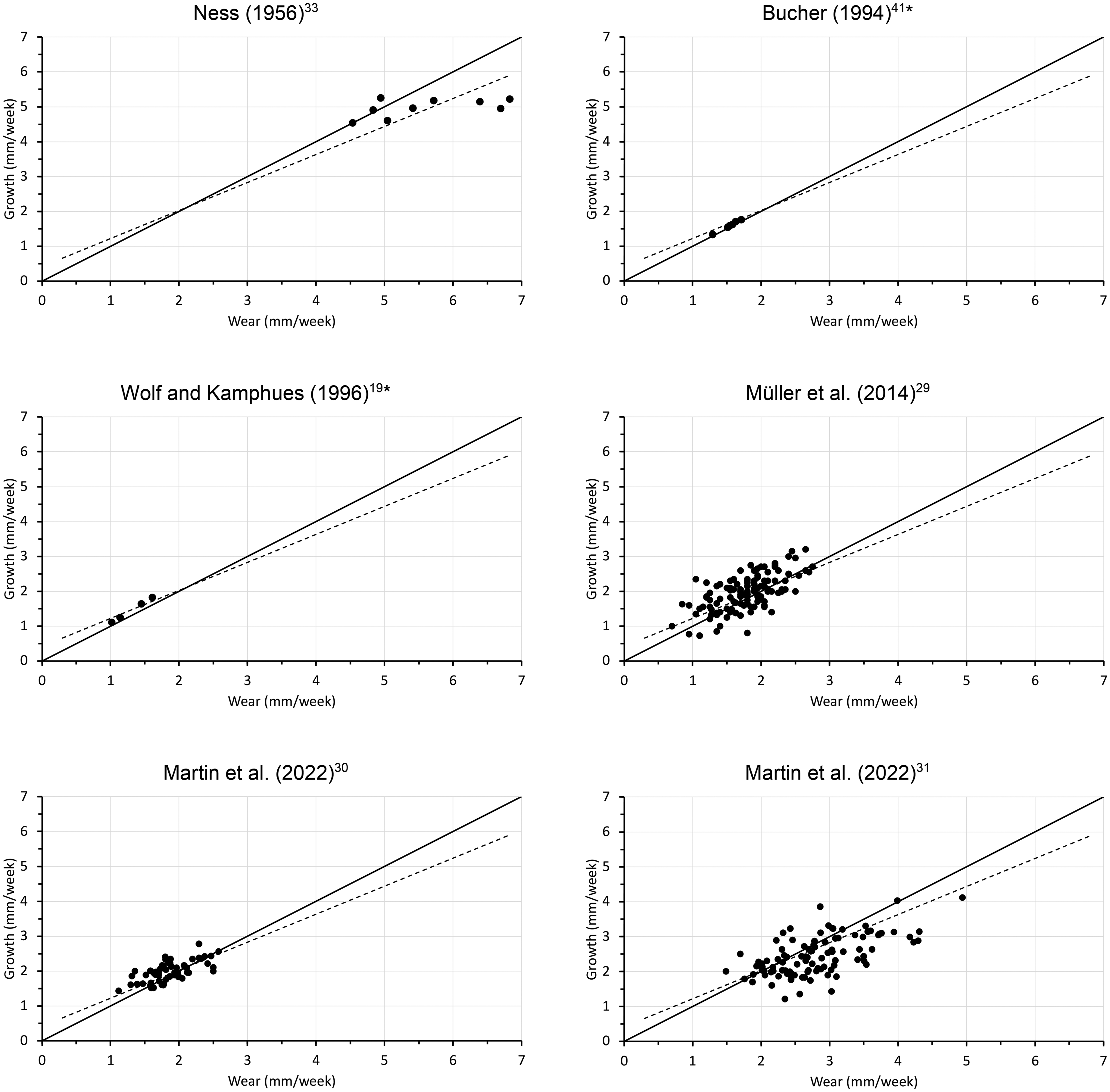
Relationship of mandibular incisor wear and growth from previous published studies. The solid black line represents y = x, and the dotted black line is the regression line for the whole dataset (Figure 12). * Denotes average values for different diets (individual data not available).
Statistical Analysis
Data for specific groups are summarized as mean ± standard deviation. For the diet switch study, the authors used linear mixed models to test whether and how body mass (BM), as well as tooth length, wear, and growth varied across diets (gruel or hay), phases of the crossover study (period 1 or 2), and over time (weeks 0, 1, 2, 3; treated as a categorical predictor variable, and nested within period). For the tooth morphometric response variables, the authors also evaluated the effect of jaw (maxillary or mandibular) and side (left or right). For each response variable, the full models included all 2-way and higher-order interaction terms among fixed (and the nested term) effects, with individual included as a random effect to accommodate repeated measures in the study design, that is, ensuring all individuals were compared from similar “zeroed” baselines. Models were fit using a software package Imer Test,
42
and goodness-of-fit of the full subsets of alternative predictor variable combinations was compared based on the second-order Akaike's information criterion (AICc package MuMIn).
43
For each model i the authors calculated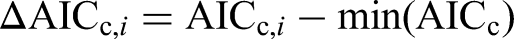
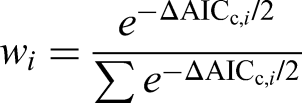
Models with low ΔAICc are best supported by the data: in general ΔAICc,i < 2 indicates strong support, 2 < ΔAICc,i < 10 indicates moderate fit, and wi is a proportion (
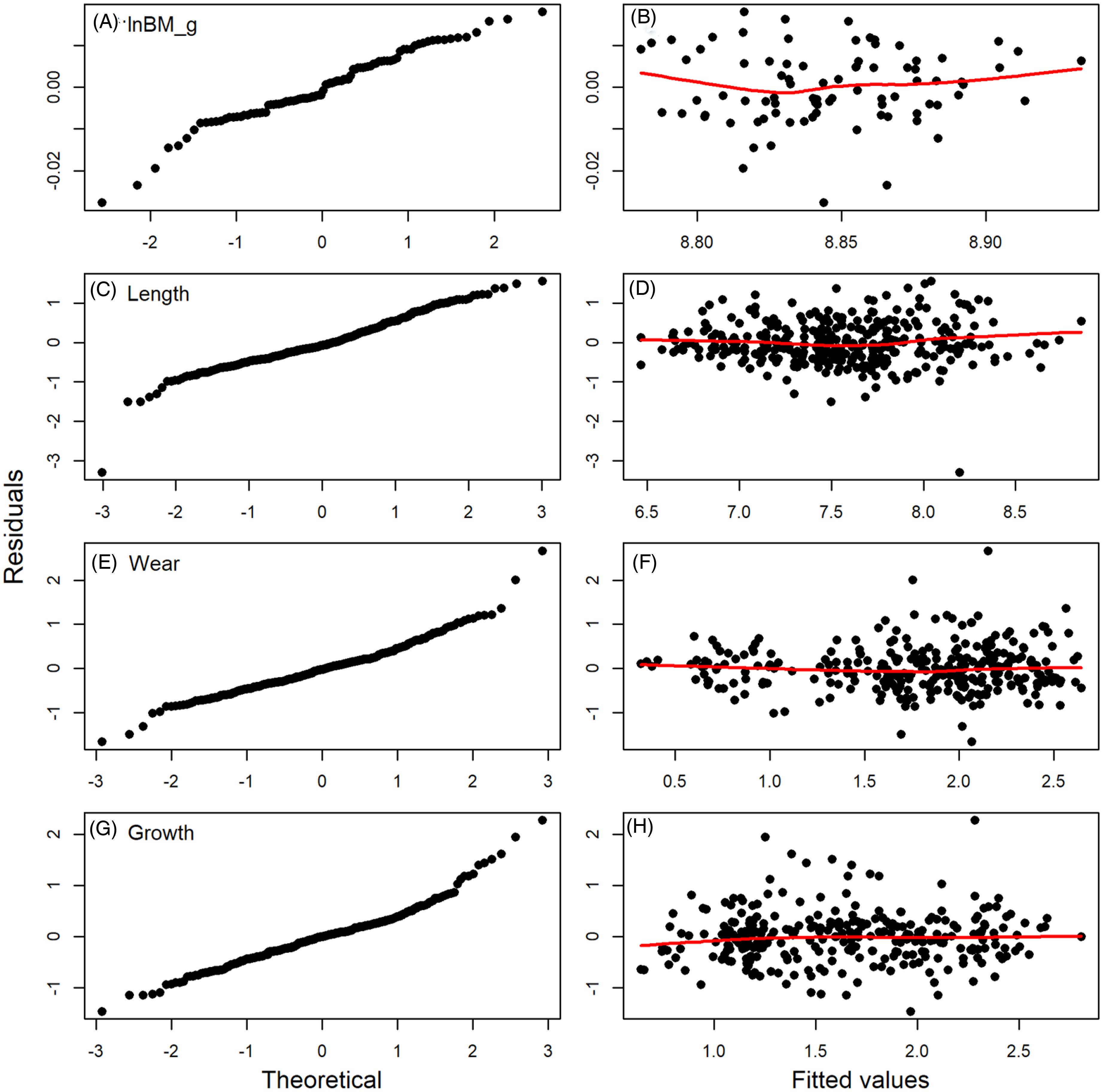
Diagnostic plots of residuals for the full models fitted to data for body mass (A, B) (BM, in g, ln-transformed, tooth length (C, D), wear (E, F), and growth (G, H) from period 1. The QQ plots (right-hand column) show approximate normality of distribution of residuals, but deviations are notable; more importantly, the lack of patterns on the right-hand column indicate little to no evidence for heteroscedasticity. Abbreviation: BM, body mass.
The authors adopted a similar approach to evaluate data for the tooth cutting period (Figure 5). Here, however, sampling intervals occurred over 9 measurements, and so “time point” was treated as a quantitative predictor. For BM, only the random effect individual and time points were included as predictors, for tooth length data the maxillary and mandibular jaw were also compared, and for tooth wear and growth the cut status (i.e., cut or uncut tooth) was included as a third fixed effect. Finally, the effect of wear on growth was evaluated, including jaw as well as whether the neighboring tooth was cut or uncut as cofactors, and individual as a random effect. For both sets of analyses, the full models for each response variable included all 2-way and higher-order interactions terms, and full subsets were compared using AICc and MMI. All analyses were carried out in Rh v 4.4.1. 46
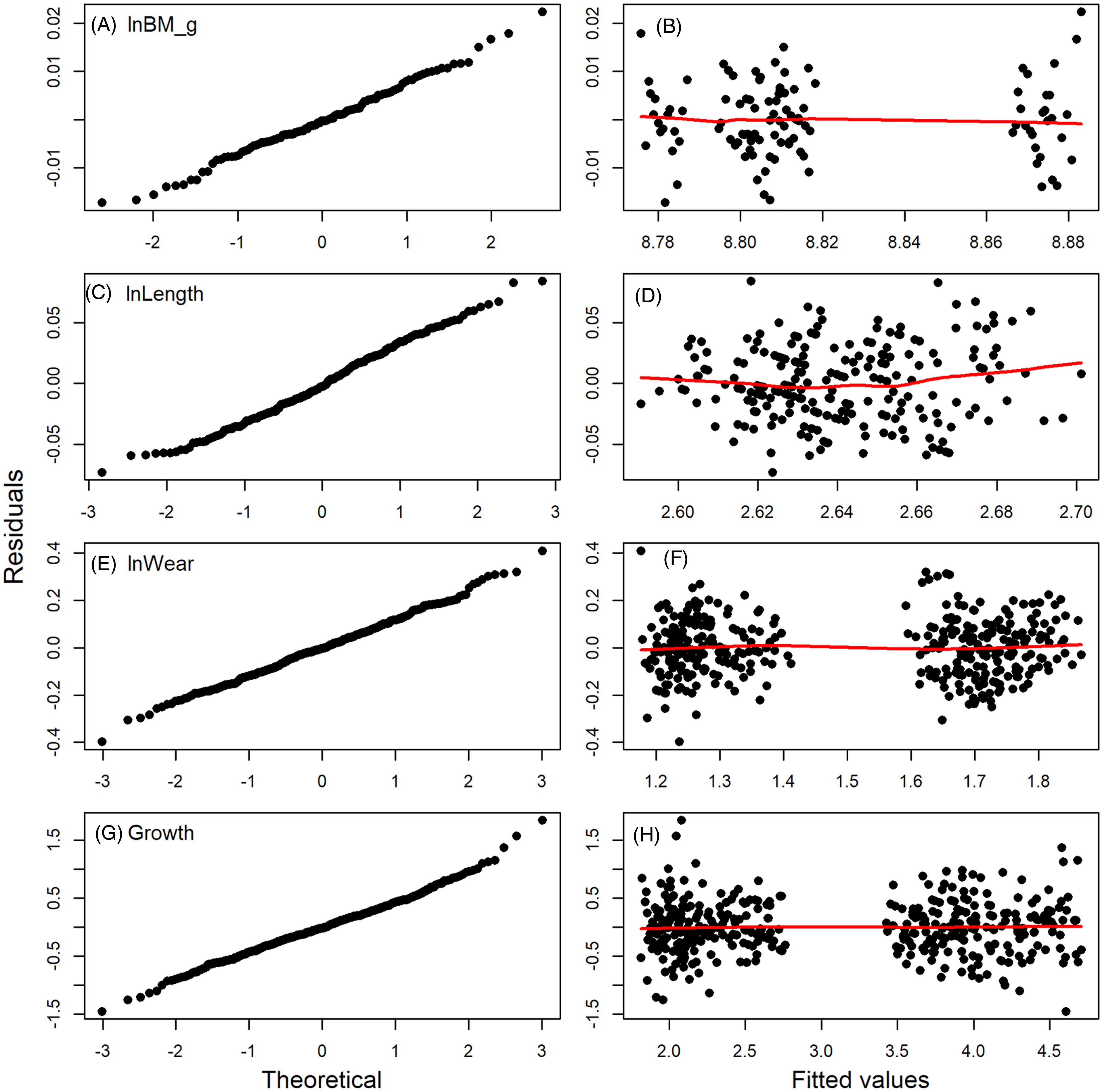
Diagnostic plots of residuals for the full models fitted to data for body mass (A, B) (BM, in g, ln-transformed), tooth length (C, D), wear (E, F), and growth (G, H) from period 2. The QQ plots (right-hand column) show approximate normality of distribution of residuals, but deviations are notable; more importantly, the lack of patterns on the right-hand column indicate little to no evidence for heteroscedasticity. Abbreviation: BM, body mass.
Combining the data from this study with that in the literature, the effect of wear on growth rate was assessed by using growth as a dependent variable and wear as the independent variable in a linear regression analysis. The assumption was that if there was a baseline tooth growth regardless of wear, the intercept of the linear regression equation would be significant (i.e., its 95% CI would exclude zero). To assess whether the inclusion of trimmed teeth affected the relationship, the same regression analysis was performed after exclusion of trimmed teeth data.
Results
General Health
No signs of discomfort or problematic food intake were exhibited by any of the animals during this study. Aside from the abovementioned 2 rabbits that were excluded, no other issues of any kind occurred. None of the animals required clinical intervention throughout the study.
Effect of Dietary Format
Rabbit BM variations through time appeared to differ between the 2 diet treatments (Figure 6). Although the null (intercept and random effect only) model was the best fit, models including diet as well as the interaction between diet, period, and week were moderately supported by the data (wi = 0.193 and 0.178, respectively) (Table 2). On the gruel diet, BM increased within the first week of the study (parameter 95% CI [0.035; 0.056] and [0.024; 0.048] for periods 1 and 2, respectively), and moderate (but not statistically significant) increases were observed in subsequent weeks. The situation was reversed for the hay diet: BM dropped markedly in the first week, but only significantly in period 2 [−0.045; −0.024] (during period 1, parameter 95% CI excluding zero was only observed by week 3).
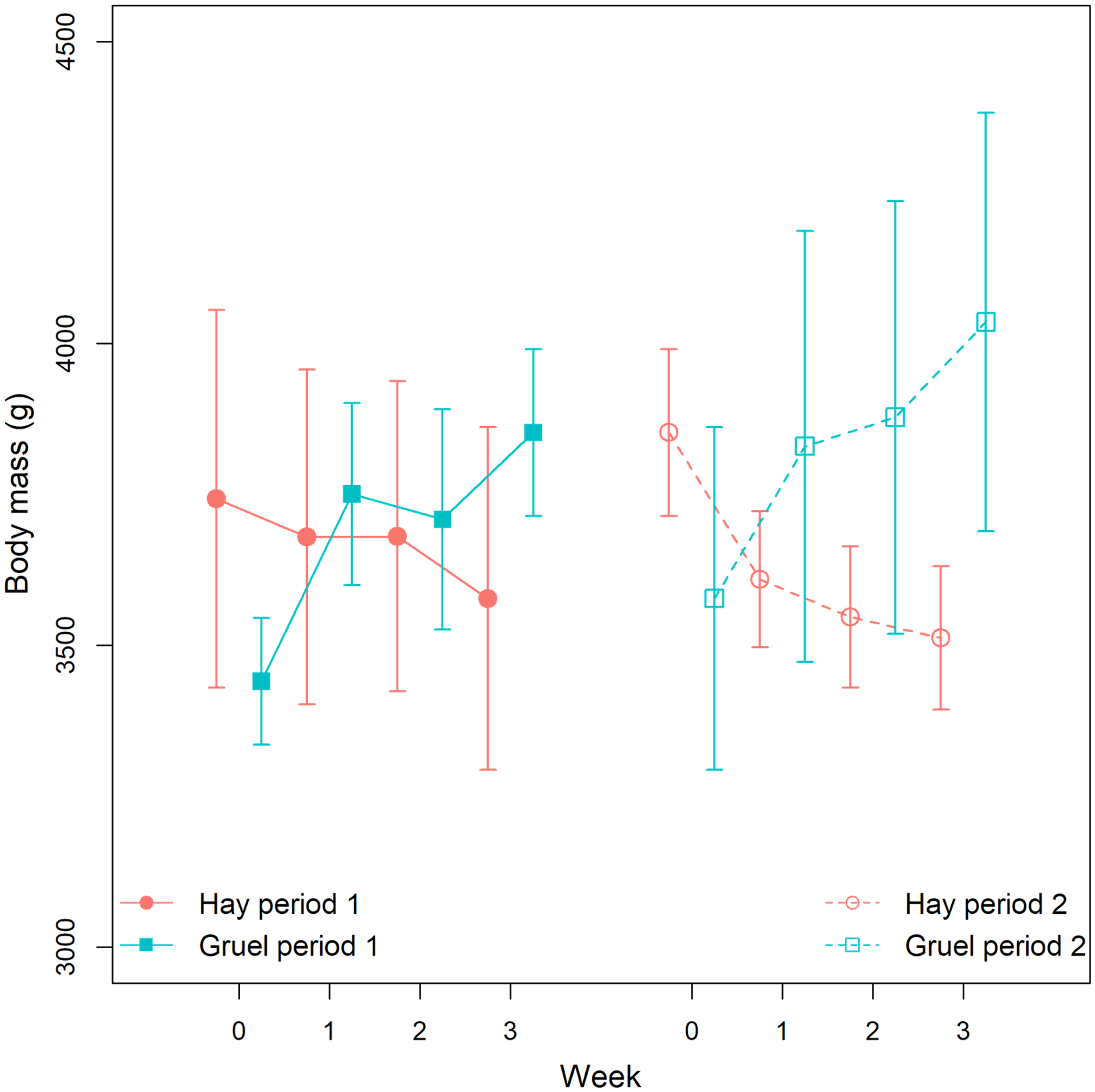
Comparison of mean (symbols) body mass of rabbits between the gruel and hay diets over time, and across period 1 and 2 of the crossover design. Error bars are 95% confidence intervals of the mean.
Effect of Dietary Format. Comparative Goodness-of-Fit of the 5 Best Models’ Fit to Data for Body Mass, Tooth Length, Wear, and Growth of Rabbits from the Dietary Format Experiment.
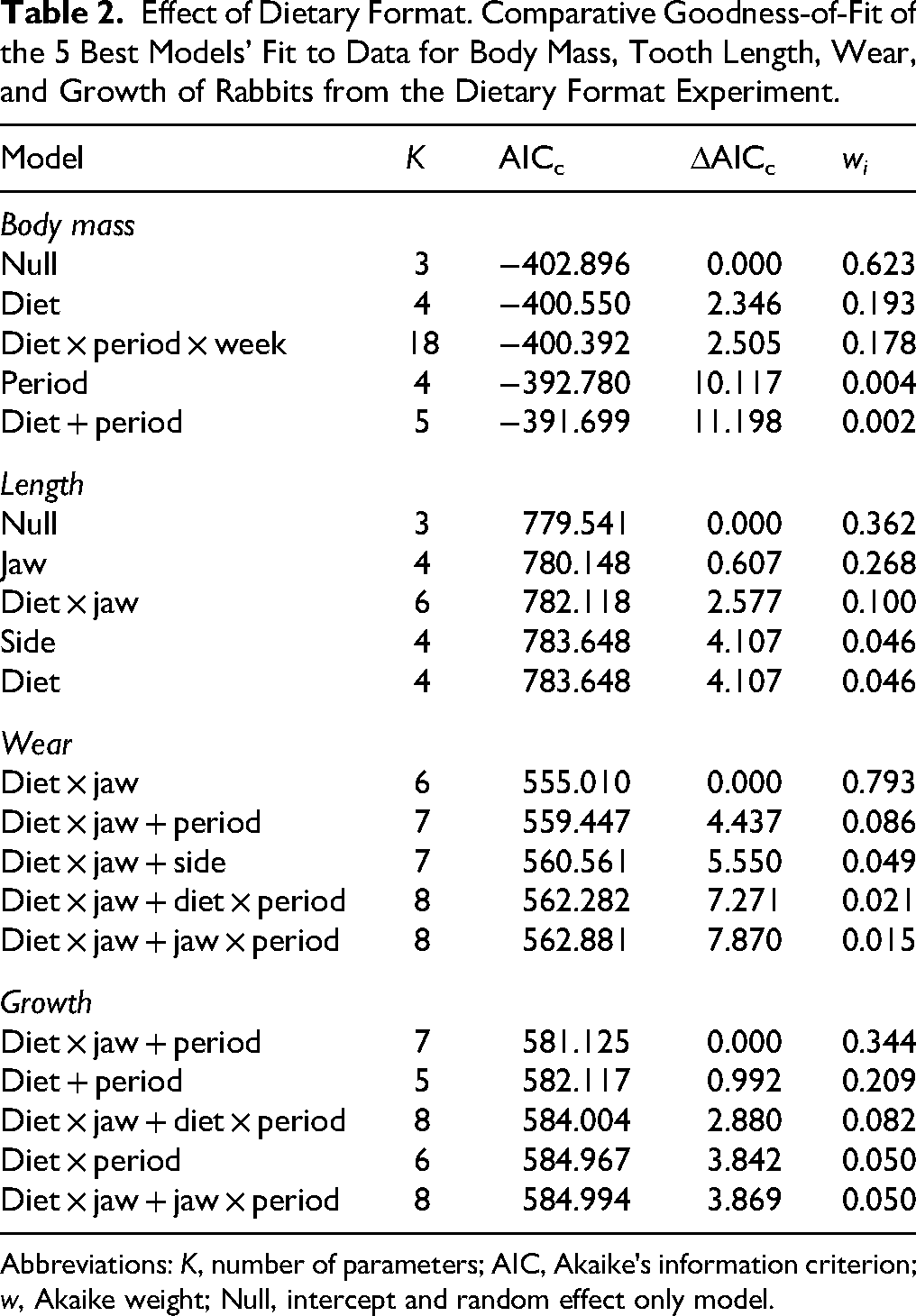
Abbreviations: K, number of parameters; AIC, Akaike's information criterion; w, Akaike weight; Null, intercept and random effect only model.
Tooth length variations were also best explained by individual-level stochasticity, and by differences between the maxilla and mandible, but the model including the diet × jaw interaction was moderately supported (wi = 0.10; Table 2) implying some degree of diet-based difference. Model-weighted parameter estimates indicated that maxillary incisor length of rabbits on the hay diet was less than for the gruel diet [−0.580; −0.075], although this pattern is only visually evident in period 2 of the study. By contrast, mandibular incisor length did not vary across diets (Figure 7A and B). The authors’ analyses revealed no evidence for an effect of time (period or week), nor sidedness, on tooth length. Tooth wear and growth showed similar patterns, with best-supported models fitted to these variables including the diet × jaw interaction term (Table 2). As with tooth length, wear and growth did not differ between diets in the mandible, but in the maxilla both wear [0.780; 1.336] and growth [0.1432; 0.7256] were higher on hay than on gruel (Figure 7C-F; individual data displayed in Figure 8). In the case of growth, the effect of period was also significant, with greater tooth growth occurring in period 2 of the study [0.053; 0.463]. For wear and growth, no sidedness was detected, either.
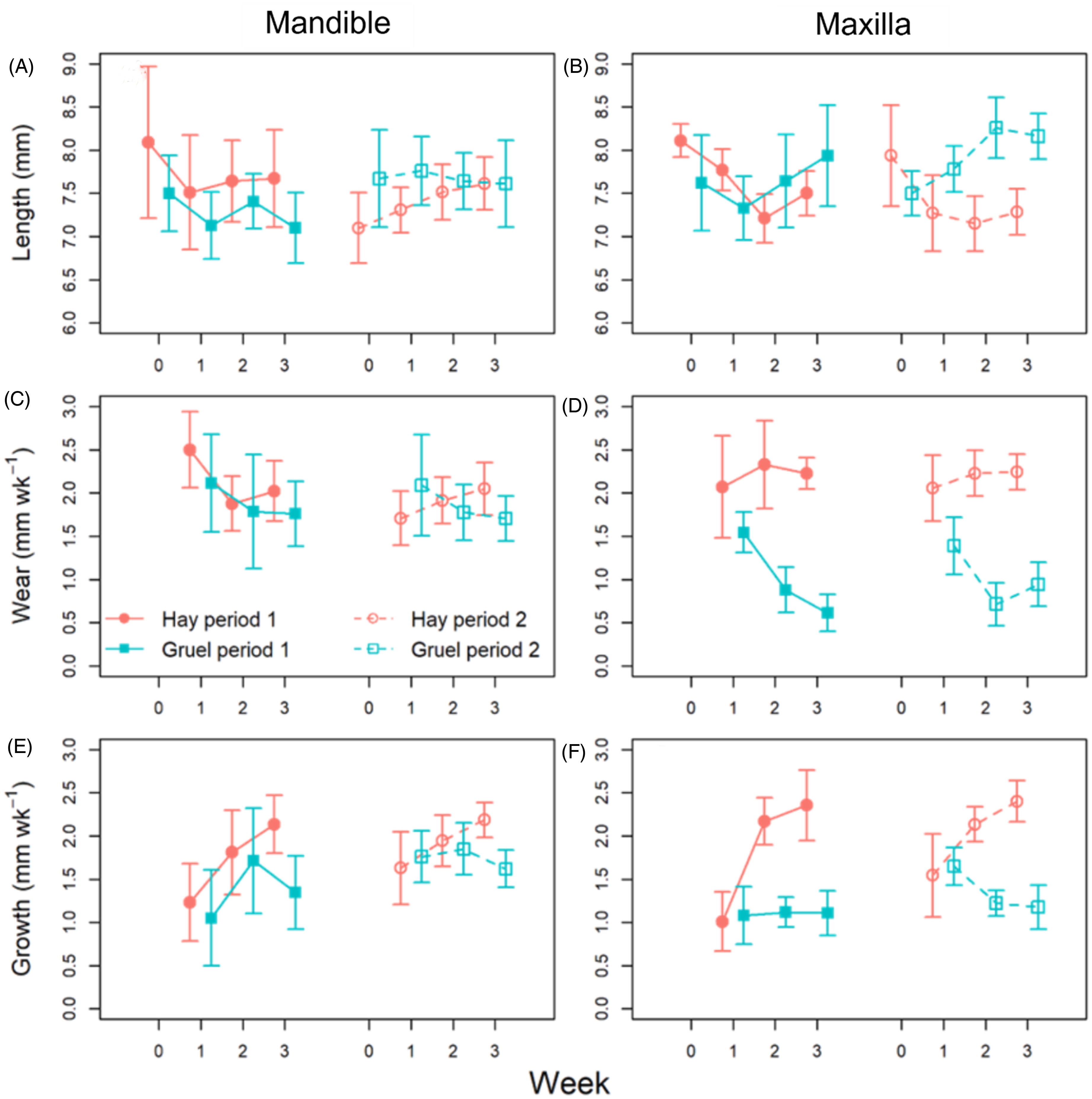
Comparison of mean (symbols) tooth length (A, B), growth (C, D), and wear (E, F) of rabbits between the 2 experimental diets (gruel and hay) over time, and across periods 1 and 2 of the crossover design. Error bars are 95% confidence intervals of the mean. Individually measured growth and wear values for each rabbit are visualized in Figure 8.
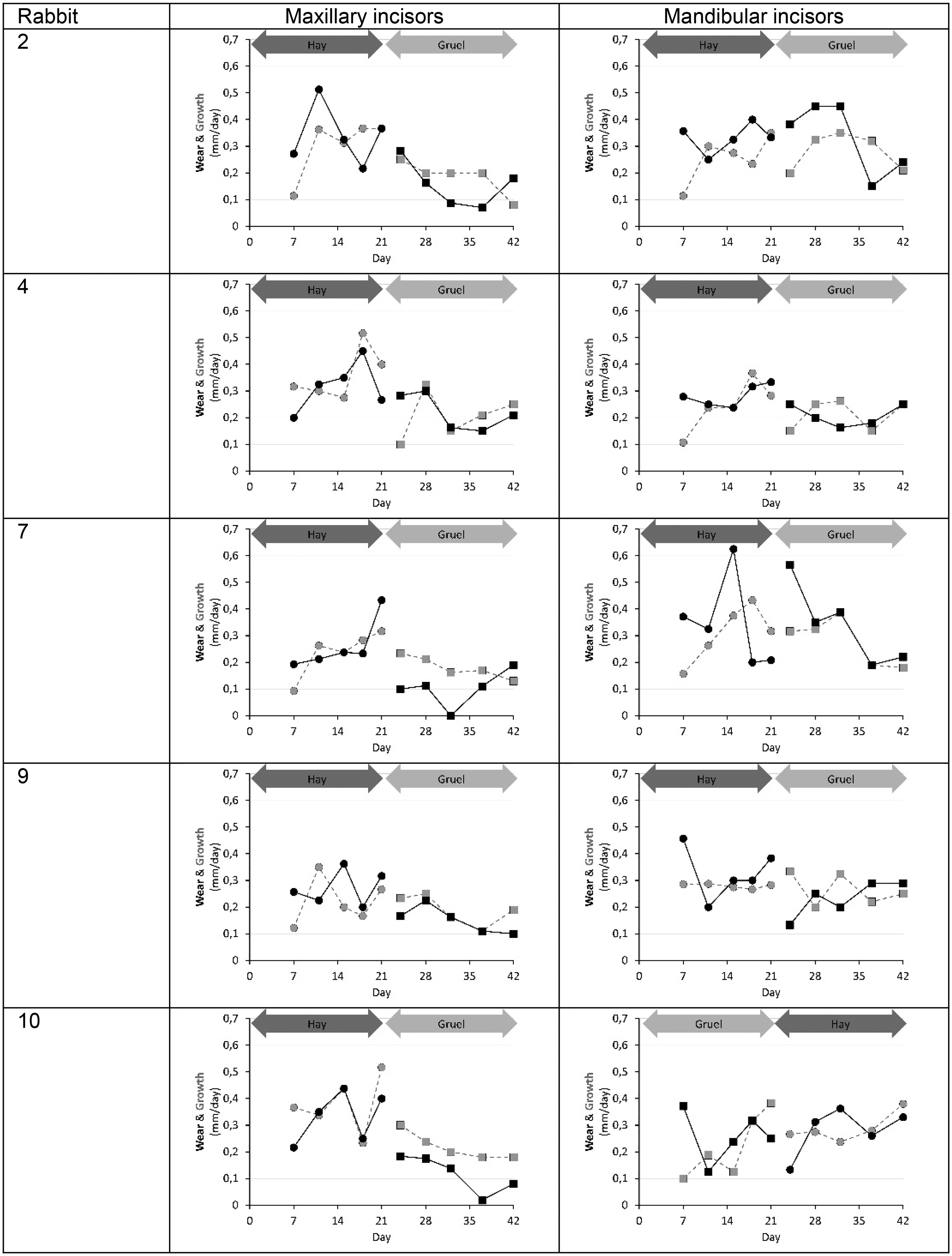
Wear and growth of the maxillary and mandibular incisors recorded individually for each rabbit during period C (hay) and period L (gruel). Day 21 marked both the end of the first period, as well as the start of the second.
Effect of Dental Trimming
Although BM variance in this study was best explained by the null model, there is evidence (wi = 0.248) that rabbits experienced a decrease in BM over time (time point effect [−0.002; −0.001]) (Table 3); however, since this result is after ln-transformation of BM, the decrease was evidently nonlinear, limited to the first few time points of the study, after which BM stabilized (Figure 9A). Variation in tooth length (only evaluated in the noncut teeth) was also best explained by the null model, but again there is evidence for a directional change over time (wi = 0.101), in this case an increase [0.001; 0.004], with no evidence for a different response in the maxilla versus the mandible (Figure 9B). Analysis of tooth wear and growth revealed similar trends to each other: in both cases, the best-fit models included the terms time point and the cut × jaw interaction (Table 3). Both measures showed a decrease over time ([−0.025; −0.014] and [−0.047; −0.009] for wear and growth, respectively), and both variables were also higher in cut teeth (uncut parameter estimates [−0.539; −0.420] and [−2.446; −2.152], respectively) (Figure 9C and D). Moreover, the difference between cut and uncut teeth was less pronounced in the maxilla than in the mandible ([0.053; 0.152] and [0.800; 1.173], respectively).
Effect of Dental Trimming. Comparative Goodness-of-Fit of the Best Models (Maximum 5) Fit to Data for Body Mass, Tooth Length, Wear, and Growth of Rabbits from the Dental Trimming Experiment.
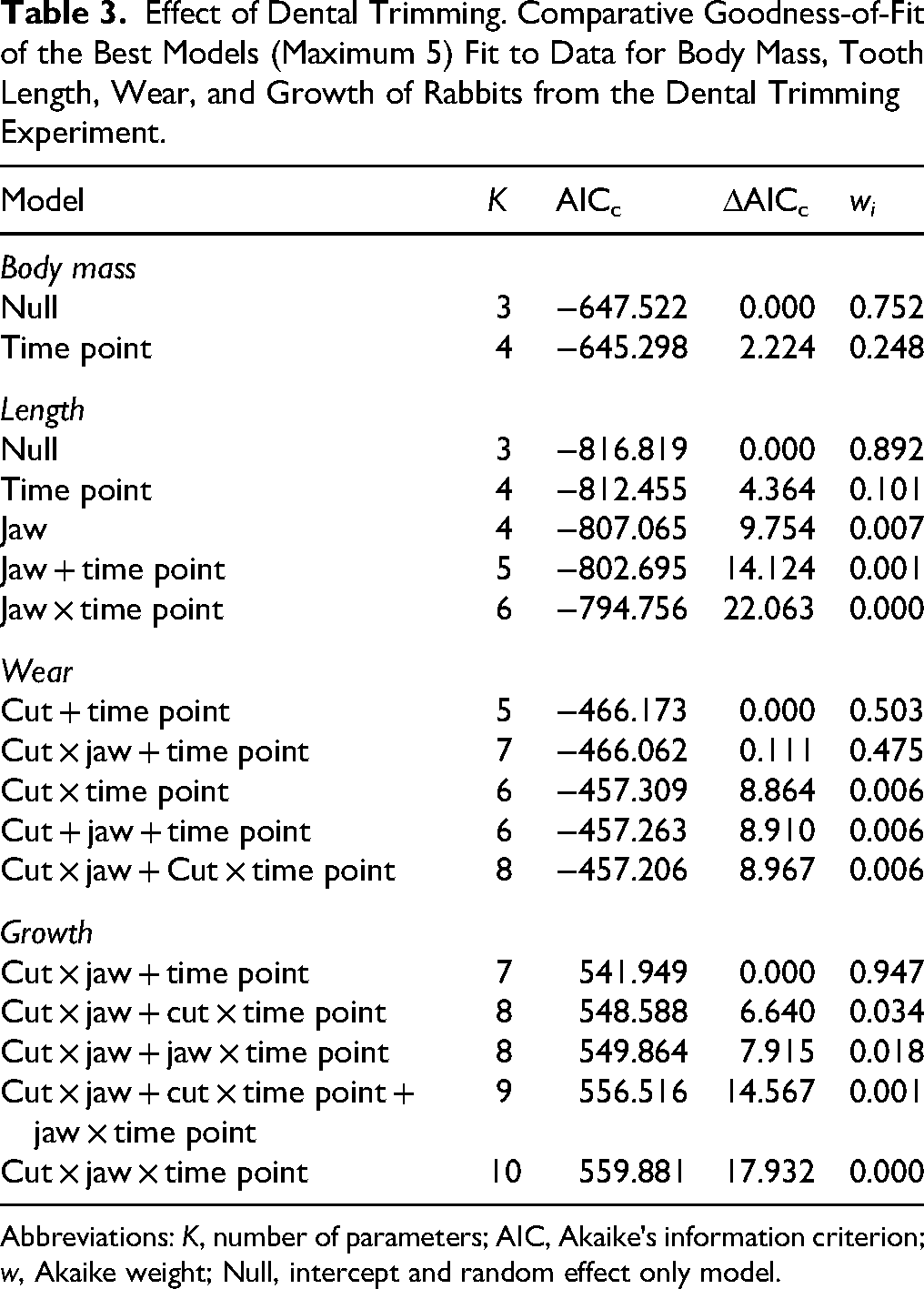
Abbreviations: K, number of parameters; AIC, Akaike's information criterion; w, Akaike weight; Null, intercept and random effect only model.
Effect of Trimming the Neighboring Tooth
As predicted, tooth growth increased linearly with tooth wear. The overall effect was an average 0.43 mm increase in growth for each 1 mm increase in wear (MMI weighted 95% CI [0.262; 0.598]). However, all well-supported models for effects on growth included the effects of jaw as well as neighbor (i.e., whether the neighboring tooth had been cut) (Table 4). This was evident because, in the maxilla, the rate of increase in growth per 1 mm increase in wear was on average 0.35 mm more if the neighboring tooth had been cut compared to if it had not been cut (95% CI [0.040; 0.650]) (Figure 10), whereas in the mandible no clear effect of cutting the neighboring tooth was noted. The model-weighted intercept for the growth–wear relationship was 1.117 [0.748; 1.486] mm/week.
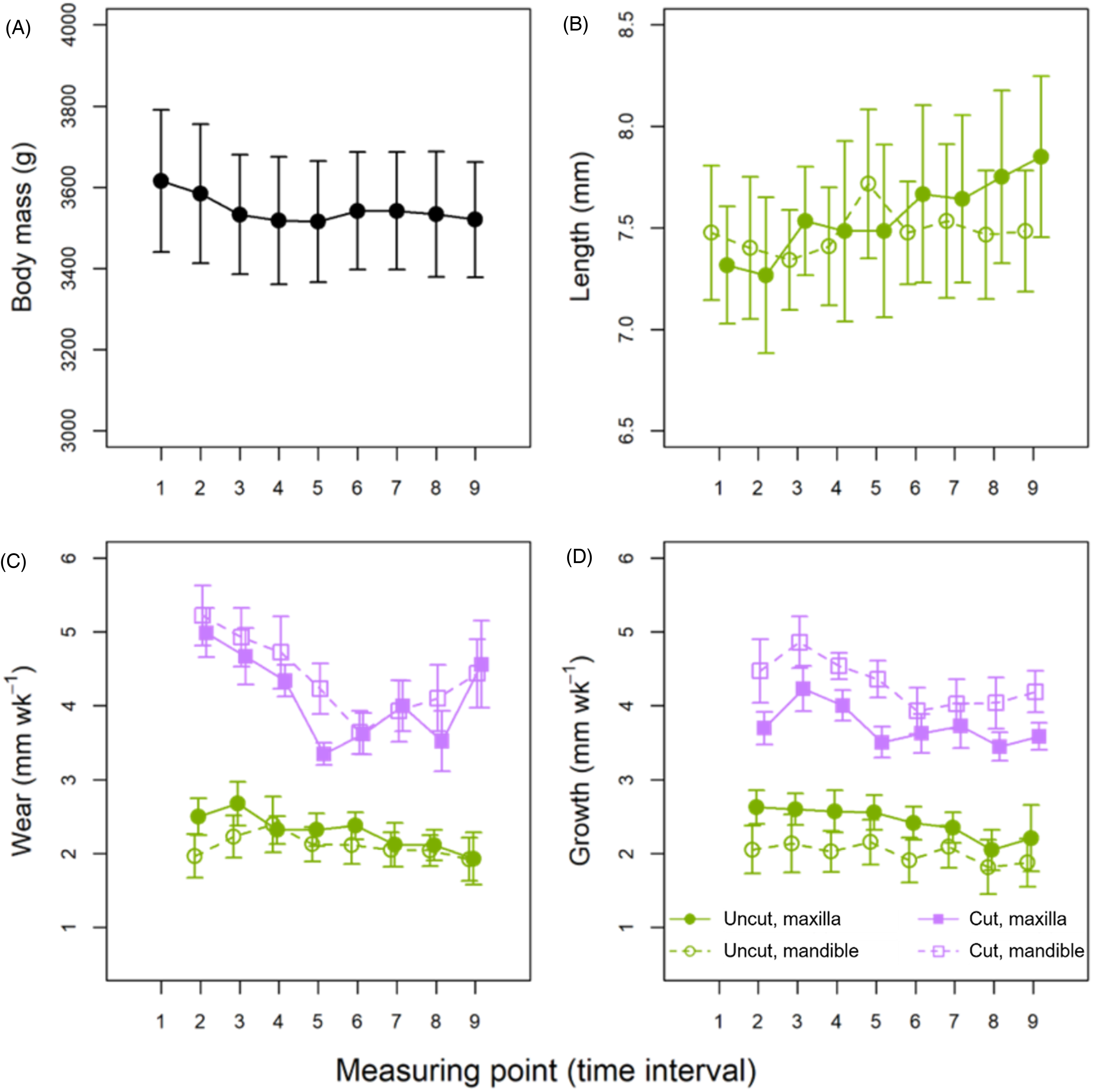
Comparison of temporal change in (A) mean body mass, (B) crown length of the maxillary and mandibular uncut incisors, (C) tooth wear and (D) growth of both uncut and cut incisors in the maxilla and the mandible. In the case of cut teeth, “wear” refers to the length of the cut part. Error bars are 95% confidence intervals of the mean.
Effect of Dental Trimming of the Neighboring Tooth. Comparative Goodness-of-Fit of the Best Models (Maximum 5) Fit to Data for Body Mass, Tooth Length, Wear, and Growth of Rabbits from the Dental Trimming Experiment.

Abbreviations: K, number of parameters; AIC, Akaike's information criterion; w, Akaike weight.
Wear and Growth Correlation Including Literature Data
Combining the data of the present study with those from the literature, there was a clear correlation between incisor wear and growth, with similar parameter estimates as those reported above. The overall effect was an average of 0.71 and 0.82 mm increase in growth for each 1 mm increase in wear in the maxillary (Figure 11) and the mandibular incisors (Figure 12) respectively. The intercepts were estimated at 0.63 and 0.38 mm/week for the maxillary and the mandibular incisors, respectively. When excluding data from trimming periods, the slopes were 0.68 and 0.54 and the corresponding intercepts were 0.67 and 0.90 mm/week for the maxillary (Figure 11) and mandibular incisors (Figure 12), respectively.
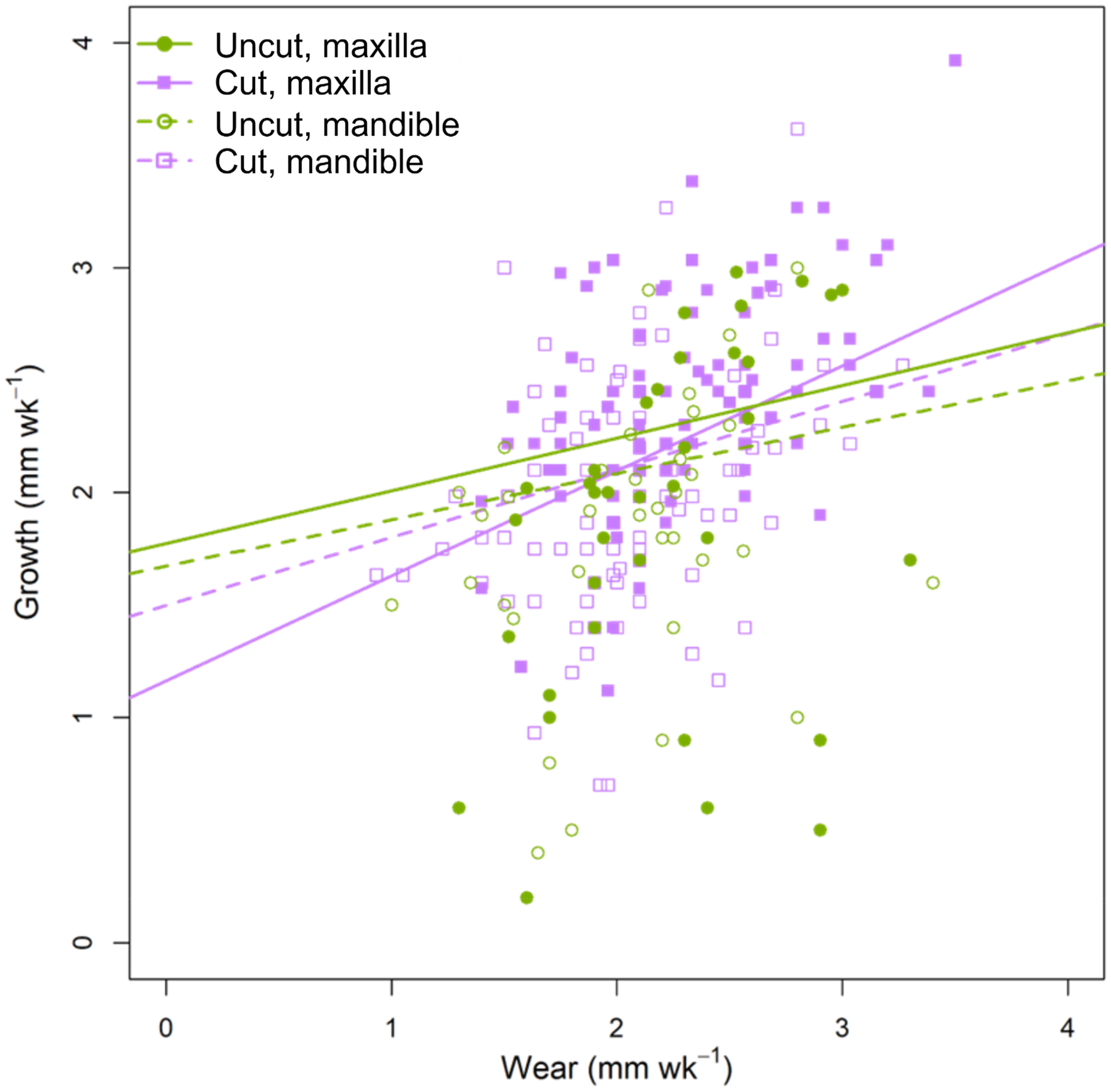
Relationship between wear and growth of the right maxillary and mandibular incisor based on whether the left incisor was intact (“uncut”) or cut.
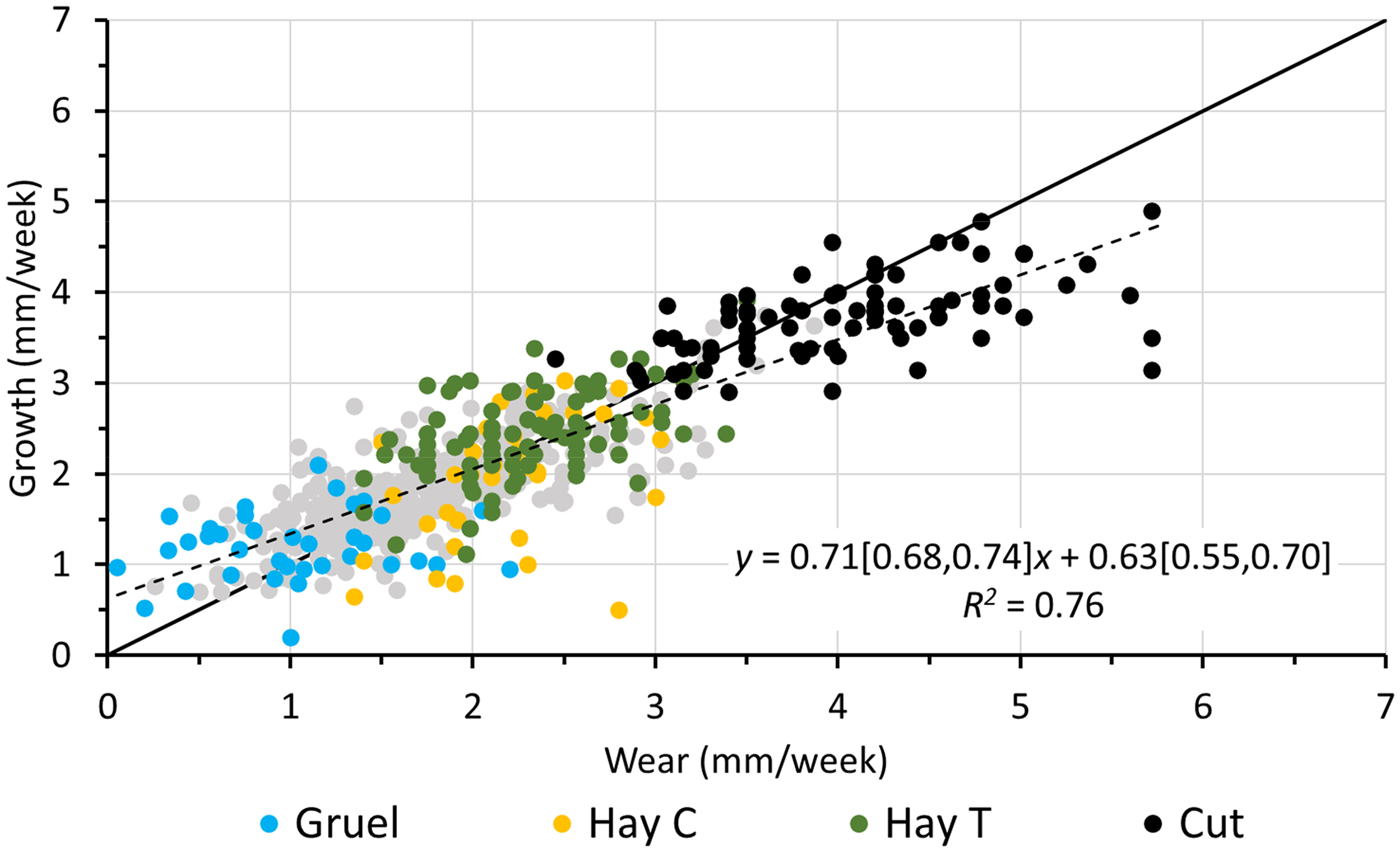
Relationship of maxillary incisor wear and growth from the present study during the dietary period on the gruel (gruel) and the hay (hay C) diets, and during the dental trimming period on the hay without shortening (hay T) and after shortening (cut), as well as from the published literature (gray dots). The solid black line denotes y = x. The dotted line represents the linear regression line; the regression equation includes the 95% confidence intervals for the intercept and the slope, respectively. The regression line for all data without any cut teeth was y = 0.68[0.62, 0.73]x + 0.67[0.57, 0.77], R2 = 0.53. The literature data is shown in Figure 2.
Discussion
The wear and growth rates for rabbit incisors obtained during this study are in agreement with previously reported values (Figures 11 and 12). This study also corroborates the hypothesis from earlier work that the speed at which dental tissue is accreted changes according to wear, either induced by dietary effects19,25,29–32 or iatrogenic manipulation,26,33 and that this effect differs between maxillary and mandibular incisors. When combining data from the literature with that of this study, a positive linear relationship between growth and wear emerges clearly, with a slope smaller than 1 and thus a significant deviation from a presumed 1:1 relationship.
Study Constraints
Before discussing the findings in more detail, it is the authors' opinion that not recording measurements in relation to time of day or feeding time represents an important constraint on the present study as well as on previously published work. One previous study found that the magnitudes of wear and growth in rabbit incisors show opposite diurnal fluctuations, with more net wear during the phase of feeding (daytime), and more net growth during the phase of resting (night-time) 47 (Figure 13A). As a result, even if tooth length considered over a longer period is constant, it fluctuates systematically according to feeding–resting patterns (Figure 13B). This might influence recorded wear–growth relationships as discussed below.
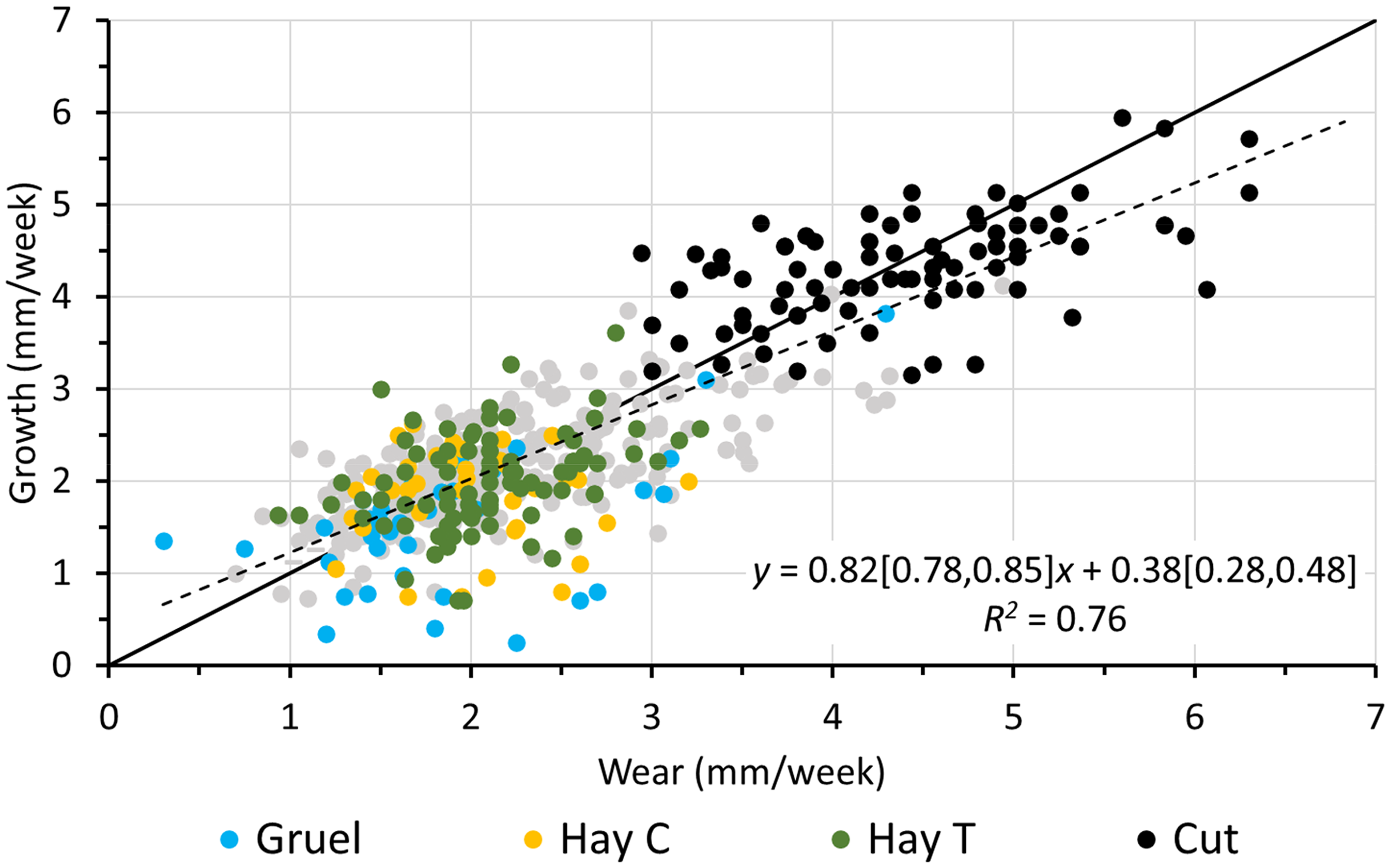
Relationship of mandibular incisor wear and growth from the present study during the dietary period on the gruel (gruel) and the hay (hay C) diets, and during the dental trimming period on the hay without shortening (hay T) and after shortening (cut), as well as from the published literature (gray dots). The solid black line denotes y = x. The dotted line represents the linear regression line; the regression equation includes the 95% confidence intervals for the intercept and the slope, respectively. The regression line for all data without any cut teeth was y = 0.54[0.47, 0.61]x + 0.90[0.75, 1.04], R2 = 0.36. The literature data is shown in Figure 3.
The values of the dental proxies may have also been slightly influenced by not taking into account the curvature of the teeth during measurements. This would, however, have required frequent anesthetics. As it was not expected that including dental curvature would greatly improve the findings, as the main focus of the present study was on differences between measurements, taken with the same “error” due to tooth curvature, and it could actually restrict comparability with data from the literature (Figures 11 and 12) where tooth curvature was also not taken into account, it was decided to not attempt to measure distances on the teeth along the curved surface.
As another theoretical constraint of the approach, it is currently unknown if identified biochemical pathways involved in dental tissue accretion7,48 are affected by nutrient intake. Theoretically, the different nutritional profiles of the gruel and hay diets could play a part in the noted effects. It should, however, be considered that the results from previous studies, that align well with those from the present study, were obtained from rabbits on vastly different diets (Figures 11 and 12). Second, the potential for tissue accretion of hypselodont incisors only appears affected by obvious dietary deficiencies.49–51 And third, when animals lost weight on the hay diet during this study (indicating reduced caloric intake), they showed higher wear and growth rates compared to when they gained weight on the gruel diet (indicating excess caloric intake). In the absence of severe deficiencies, empirical data therefore does not support a relevant limiting effect of nutrient composition per se on dental wear and growth rates.
Finally, in order to meet standard husbandry requirements for rabbits in Switzerland, as well as to nonartificially increase attrition on the incisors, rabbits were provided ample gnawing opportunities during hay feeding. However, in the study setup, the authors did not quantify gnawing activity. Therefore, differences in gnawing behavior may have affected measurements in unknown ways.
Effect of Dietary Format
During a similar study, feeding a gruel diet also reduced dental growth. 25 However, it affected maxillary and mandibular incisors similarly, whereas tooth length remained relatively stable. In contrast, the present study indicates that dietary format alters length, wear, and subsequent growth rate in the maxillary but not the mandibular incisors. These findings corroborate other studies showing a more pronounced effect of the diet on the maxillary compared to the mandibular incisors.19,29,31 Although maxillary and mandibular incisors differ morphologically, the microstructural similarity of the enamel layer 52 renders tooth structure as an explanation for the dichotomous results less likely. The difference in their respective wear, on the other hand, provides a more promising concept. When biting or gnawing, the enamel-covered labial aspects of the mandibular and the anterior maxillary incisors first align. Thereafter, a cranial to caudal sliding motion of the mandibular incisors against the maxillary incisors occurs, wearing down the softer dentin and creating the chisel shape of the maxillary first incisor. 53 In guinea pigs, the same process has been suggested to explain why increased biting frequency, and thus decreased support of lingual dentin to labial enamel, renders maxillary incisors more labile to wear. 54 The effect of food type could be further augmented as it does not only influence the frequency of incisor use, but also the extent of the sliding motion when biting. 53 In contrast, sharpening of the mandibular incisors is considered to occur through deliberate thegosis or bruxism, which is unrelated to feeding. Although this process has only been visually confirmed in rodents,50,55,56 anatomical feasibility as well as wear patterns strongly suggest that a similar action takes place in rabbits.50,53 Theoretically, as previously indicated in rats, 55 it therefore appears that feed type affects wear more markedly in the maxillary than in the mandibular incisors. Additionally, when at rest, the labial aspect of mandibular incisors is in contact with the maxillary second incisors “peg teeth” or the lingual aspect of the maxillary first incisors. Wear patterns indicate that the labial side of the mandibular incisors experiences additional contact from lateral motions of the mandible during grinding chewing, in contrast to that of the maxillary first incisors. 53
During biting or gnawing, the incisors occlude in the mid-sagittal plane, that is, in bilateral symmetry. 53 A lack of laterality for the incisors, detected in previous studies29,30 as well as in the present study, would therefore be the default expectation, in contrast to noted laterality of cheek teeth due to unilateral use during chewing.31,53,57 The finding of some laterality in incisors in a single study 31 may therefore be considered an outlier.
Effect of Dental Trimming
The dental trimming period supports previous studies stating that tooth growth is regulated at the level of the individual tooth.26,29,33 This is shown by the significant difference in growth between uncut and cut teeth (Figure 9). In the maxillary incisors, however, a slight acceleration of growth was detected when the neighboring tooth had been cut (and hence triggered to grow particularly fast) (Table 4). Because this finding results from the comparison of data obtained during 2 different periods in the present study (C and T) and not derived during 1 period with a cross-over assay, it should be considered with caution.
The present study also corroborates earlier studies which found that taking rabbit incisors out of occlusion markedly increases their growth rate.26,33 As pressure decreases mitotic rate in hypselodont incisors,58–62 the reduction of its effect by dental trimming explains this observation. The former study 26 and the present study also note that cutting incisors out of occlusion increases growth rate more markedly in mandibular compared to maxillary incisors. Revealing the unconstrained growth potential by dental trimming thus indicates a greater possibility to increase the speed of dental accretion in the mandibular incisors, compared to the maxillary incisors. As mandibular incisors have been documented to fracture more frequently,26,50 possibly due to being more exposed in terms of a free-standing tooth body as well as in terms of angulation-related leverage, the authors speculate that their larger growth potential forms an adaptive quality to respond to more likely insults. Another hypothesis could be a difference in pressure feedback for the mandibular and maxillary incisors. The labial aspect of mandibular incisors can interact with the caudal aspect of maxillary first incisors at rest, during biting and grinding chewing.21,50,53,57,63–65 That of the maxillary first incisors, on the other hand, only interacts with the mandibular incisors during biting, but does not receive pressure feedback during grinding chewing or at rest. The labial aspect of mandibular incisors therefore experiences more tooth-on-tooth contact than that of their maxillary antagonists. Considering that the labial aspect of incisors will be the more important determinant of tooth growth7,66–68 and the duration of pressure may be more important than the absolute force,36,37 the mandibular incisors may thus receive more of this feedback under normal circumstances. Conversely, the growth of maxillary incisors should therefore be adjusted to a larger proportion of the daily time spent without pressure. If the growth of the mandibular incisors is regulated by a more time-intensive pressure feedback, then it would follow that a lack of pressure due to the artificial shortening exhibits a greater effect on them. This might also explain the effect of the neighboring tooth for the maxillary incisors. In the mandibular incisors, the more regular control of growth by the antagonistic pressure might overrule any effect that the upregulation of growth in the neighboring tooth might have. By contrast, the lesser degree of such pressure control in the maxillary incisors might make the effect of cutting the neighboring tooth more detectable (Table 4).
Interestingly, the effect of relieving occlusal pressure on growth rate, in this study and previous research on rabbits 33 and rats, 68 appears to stabilize at a slightly lower level after an initial surge. As the connection between pressure and molecular pathways 7 remains to be elucidated, it is currently unclear if this represents an exhaustion of a reserve to quickly respond to insults or an adaptation of the system to reduced feedback over time.
An unexpected result from this study was the reduction of wear and growth rate with an increase in tooth length for the uncut incisors. The authors expected the opposite, as previously documented for the mandibular incisor, 33 since only 1 incisor pair could be used for biting and thus would be exposed to relatively more wear. An explanation could be that rabbits during this study exhibited less “facultative” biting through gnawing after dental trimming, for which there was no opportunity in the previous study. 33 Reduced gnawing might lead to increased tooth length, despite a relatively increased use for food ingestion. As gnawing behavior and biting patterns were not assessed in the present study, this remains speculative.
Wear and Growth Correlation
When plotting growth as a function of wear (Figures 11 and 12), there is a clear linear relationship in both maxillary and mandibular incisors. Mathematically, the positive intercepts of the linear equations indicate a “minimal growth rate” of 0.63 and 0.38 mm/week in the absence of wear for the maxillary and mandibular incisors, respectively. This study could thus be interpreted as corroborating the occurrence of a continuous basal growth in hypselodont incisors. Previous findings have been interpreted as indicators of such a minimal basal growth, like the distortion of dental tissue and remodeling of alveolar bone at the dental apex when inhibiting eruption (by pinning or traction) or wear (by capping).33,69–71 Although the calculated minimal growth in the present study lies below the previously reported value of 0.7 mm/week, 33 it may still present an overestimation due to the time of measurement. With measurements obtained from the morning onward (i.e., after the resting period), it could be that the present as well as previous studies19,29–33,41 classically overestimate growth, and underestimate wear, due to the uneven distribution of wear and growth across the 24 h cycle (Figure 13). 47 In contrast, it could be speculated that if measurements of growth and wear had been taken systematically at the end of the feeding and before the resting period, a negative intercept might have occurred. This effect might be more pronounced with reduced feeding time and frequency (i.e., increased resting period). Less time is typically allotted to eating compound feed compared to hay,29,72–74 and the observations during the study indicated that rabbits started feeding whenever new food was provided. The putatively longer period between the last meal and point of measurement when feeding compound feeds would allow more time for regrowth and therefore would render a larger difference between growth and wear. As rabbits on such compound feeds typically experience less incisor wear,29,32 this effect likely exerts a larger influence on data closer to the y-axis (Figures 11 and 12), and thus may augment the value of the positive intercept and contribute to a slope of less than 1 in the regression analysis. Regarding the latter, on the other end of the spectrum, dental trimming is conducted in a way that growth cannot fully compensate for wear. Although this further reduces the regression's slope, the effect on the equation appears marginal (especially for the maxillary incisors) when excluding the dental trimming data from the analysis (Figures 11 and 12). Importantly, in rats, circadian patterns have also been reported for both occluding and for trimmed, nonoccluding (i.e., freely erupting) teeth, indicating potential influences beyond feeding patterns alone. 75 Research documenting wear and growth at different times during the day, as well as with a different relation to feeding time, are warranted to investigate the effect of time of measurement on the growth–wear relations rendered by this study.
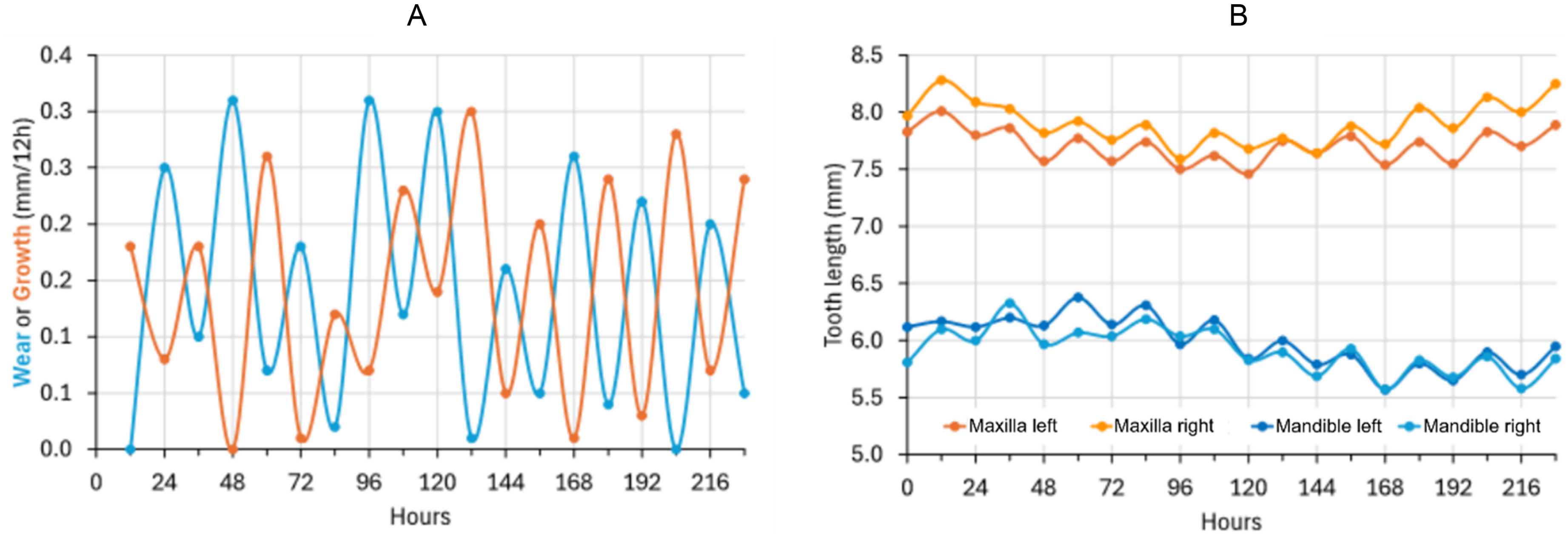
Diurnal changes (A) in wear or growth (mm/12 h) in the maxillary left incisor and (B) in the length of all incisors of a rabbit measured twice daily. 47
Based on the equations, a constant tooth length can only be expected at an experienced wear around 2.17 and 2.11 mm/week for the maxillary and mandibular incisors, respectively. It is, however, important to consider the dynamic equilibrium between wear and growth.19,47 As suggested previously, dietary aspects could simply determine a certain set-point of tooth length for opposing antagonists at which a primary balance is reached in the short term. 29 However, it remains to be studied if diets causing different wear rates would lead to differences in tooth length in the long term or if tooth length would always return to a similar (“ideal”) value.
It should be noted that the collected data never showed an absence of wear, even when rabbits are fed a liquid diet or are at rest (Figures 11 and 12). 47 This is likely due to physiological thegosis (attrition due to “empty” chewing)21,25,50,64 and might explain why liquid-fed rabbits do not necessarily develop dental issues.25,38 This finding, as well as the lack of overgrowth during this study, questions the necessity of a minimal dietary abrasiveness to account for basal dental growth. However, the relatively short time frame of most studies, as well as the paucity of large datasets on growth and wear in cheek teeth compared to incisors, do not fully exclude the “low-abrasion-leads-to-dental-disease” concept. 32 Nevertheless, based on the findings from this study, future research on the relation between diet and dental disease in rabbits should focus more on early development feeding, 76 elicited chewing patterns,21,53,57 mineral imbalances, 77 and the effect of diet on behavior,72–74 rather than on abrasiveness and hence the amount of induced wear per se. With respect to choosing diets for rabbits, these results should not be construed as an argument for why forage-based diets are not necessary, given their relevance to many other health factors.78–81 With respect to the effect of nomenclature, findings like those of the present study have led to the suggestion that rather than calling the teeth of rabbits and rodents “ever-growing”, which invokes a concept of constant growth, they should be called “occlusion-controlled growth teeth” (OCG teeth) to reflect that the growth is not constant but under the control of a feedback mechanism. 82
Conclusion
Environmental aspects such as diet and gnawing opportunities affect tooth length, wear, and growth rate differently for the maxillary incisors compared to the mandibular incisors. This is due to the different exposures to attrition during the biting and chewing cycles, resulting in different wear patterns between maxillary and mandibular incisors, as well as the putatively greater potential for growth in the latter.
Nevertheless, in both, growth rate is linearly related to experienced wear, with a corresponding slope less than 1. Summarizing the data from this study alongside previously reported measurements indicates that basal tissue accretion may occur in the mandibular and maxillary incisors. Although the calculated values of minimal growth clearly lie below previous estimates, they are likely still too high, considering a probable bias due to time of measurement usually obtained at the end of a resting phase. It should also be noted that a no-wear situation in rabbits with no morphological abnormalities appears to be purely theoretical unless artificially induced. This, as well as the lack of dental disease during the present as well as in other similar studies, questions the need for minimal dietary abrasiveness to account for basal dental growth when managing rabbits.
Materials
3525 Ranger Kaninchen Spezial, Granovit, Kaiseraugst, Switzerland
UFAG Laboratorien AG, Sursee, Switzerland
Granovit, Kaiseraugst, Switzerland
0.4 mm, KaVo® K5™ Plus, Biberach, Germany
Nanjingjinshidianzimaoyi Co., Ltd., Nanjing City, China
Technocraft® Allchemet AG, Bäretswil, Switzerland
Henry Schein AG, New York, USA
R Development Core Team, Statistics Department of the University of Auckland, Auckland, New Zealand
Footnotes
Acknowledgments
The authors would like to thank Melissa Terranova, Christoph Jenni, Sergej Amelchanka, Raphael Jendly, and the AgroVet-Strickhof team for supporting this project. In addition, the authors would like to express their gratitude to Dr Prebble and Prof. Meredith for sharing the raw data of their work with our research group.
Author Contributions
MC and J-MH designed the study; J-MH acquired the funding; JM and HO performed the animal study; HO, DC, and MC analyzed the data; RG provided additional literature review; and HO and MC wrote the manuscript with input from all coauthors.
Data Availability
The original data measured in this study are available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Vontobel Foundation and the Finnish Foundation of Veterinary Research. DC was funded by the National Research Foundation of South Africa (UID# 137968).
