Abstract
An acquired palatal defect in dogs is a pathologic condition in which there is communication between the oral cavity and nasal cavities, maxillary recesses, or orbits. Many different causes exist and must be considered. Two dogs were presented for severe palatal defects secondary to a foreign body trapped between maxillary dental arches. Numerous techniques have been previously described to repair palatal defects, and the most appropriate choice is based on the clinical features of the defect and information obtained from advanced diagnostic imaging. Acquired palatal defects are not predictable in shape, size, and location; therefore, although the literature contains many different surgical techniques, these are not always reliable. The aim of this article is to describe an innovative surgical approach to repair severe acquired caudal palatal defects in two different dogs.
Keywords
Introduction
The palate is the anatomical barrier between the digestive and respiratory systems, separating the oral cavity and oral pharynx below from the nasal cavities and nasal pharynx above. 1 It is composed of the primary palate which comprises the upper lips and the incisive bones and the secondary palate which contains the palatine processes of the maxillary bones, palatine bones, and soft palate. It can be further divided into hard and soft palates. 1 Whenever communication between the oral cavity and the overlying structures is present due to a defect in any part of the palate, a pathological condition is established.
Palatal defects could be congenital or acquired. Regarding the former, it is crucial to bear in mind that palatogenesis is a complex and unique process. It begins on the 23rd day of gestation and continues through different stages during which the incisive bones, the palatine processes of maxillary bones, and the palatine bones fuse with the vomer bone to separate the oral cavity from the nasal cavity.2,3 When this process is disturbed by genetic factors, environmental teratogens, or mechanical forces, failure to fuse bony shelves and soft tissue may occur.4–7
These defects are typically linear in shape, and they occur as cleft lips when the involved portion is the primary palate or cleft palate when the defect lies in the secondary palate distal to the palatine fissures. A cleft lip could be further categorized as incomplete when it does not extend into the nostril or complete when the defect continues into the nostril with or without the involvement of the alveolar process between the maxillary third incisor and canine teeth. Both cleft lip and palate could be additionally categorized as unilateral or bilateral defects. 8
Acquired palatal defects are properly named fistulas and, according to location, they are defined as oro-nasal fistulas, oro-antral fistulas, and oro-orbital fistulas if the pathologic communication exists between the oral cavity and nasal cavities, maxillary recess, or orbit, respectively. These defects could affect any part of hard or soft tissues and exist in differing shapes and sizes.
Etiologies of acquired palatal defects are many, including trauma, dental pathology, electric burns, radiotherapy, malocclusion, dehiscence, tumors, and foreign bodies.9–18
Communication between the oral cavity and nasal cavity, orbits, or maxillary recesses leads to secondary infections such as rhinitis, aspiration pneumonia, and ocular infections.19–23
Treatment planning and the decision-making process are highly linked to advanced diagnostic imaging techniques such as fan beam computed tomography (FBCT) or cone beam computed tomography (CBCT).24–26 It is crucial to investigate the underlying bone defect and the vascular supply to ensure the success of surgical treatment.27–29
Many surgical techniques are described in the literature and the decision-making process is related to the location, shape, and dimensions of the defect.30–40 However, these features can be extremely variable, especially in acquired defects, and prevent already existing techniques from being feasible. In this article, an innovative surgical technique is described for severe palatal caudal defects in two different dogs.
Materials and Methods
Case 1
A 7-year-old male Bull Terrier with a history of chronic rhinitis was referred to the author's clinic for a suspected acquired palatal defect caused by a foreign body. Oral examination revealed bilateral nasal mucopurulent discharge, severe halitosis, and on opening the mouth a wooden stick was found trapped on the palate between 108 and 208 (Figure 1). Findings on the patient's general physical examination were unremarkable and preanesthetic blood work results were within the normal range.
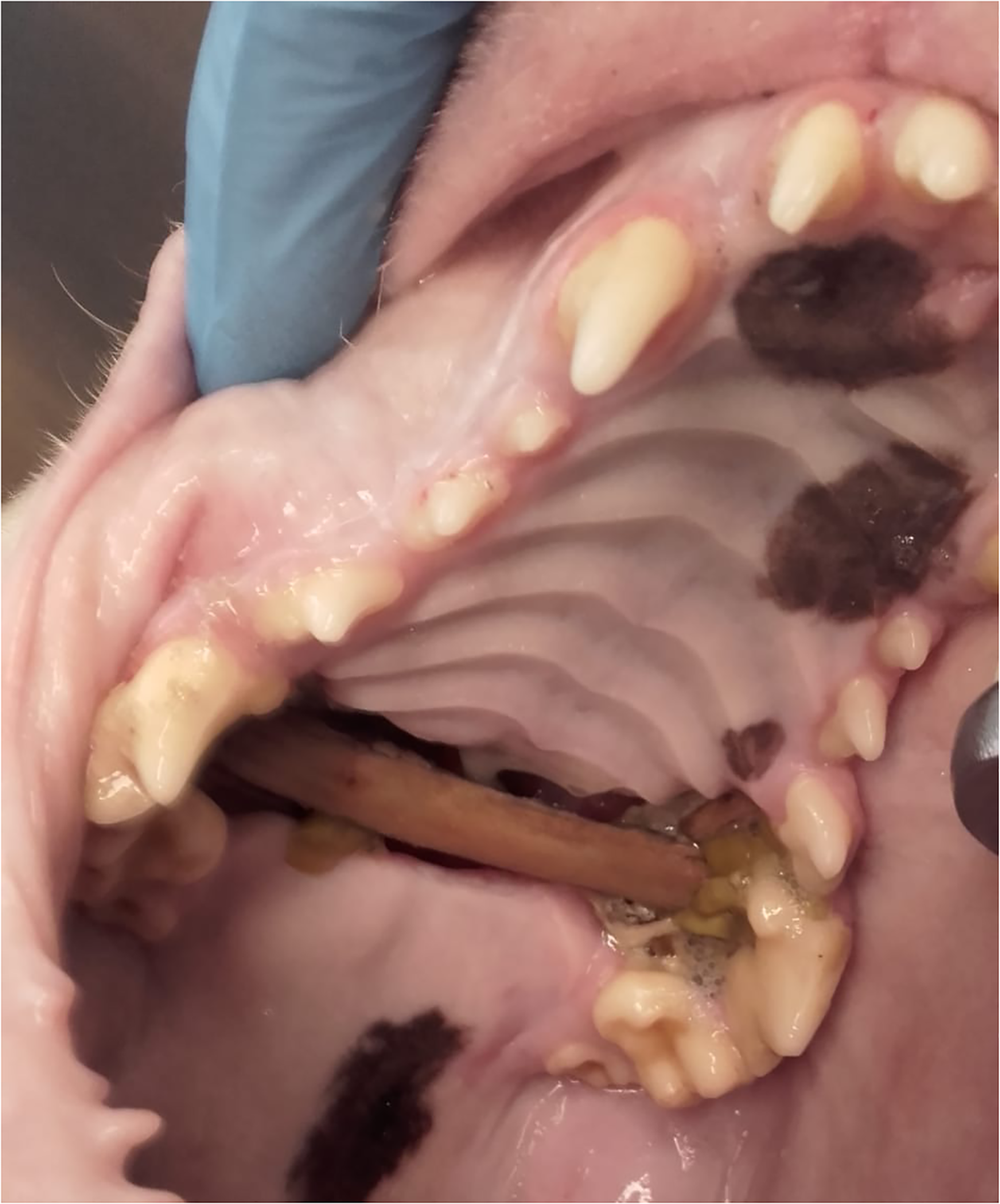
Case #1. Wooden stick trapped between the right and left maxillary fourth premolar and first molar teeth. Note the debris, mucopurulent material, and underlying palatal defect.
The patient was anesthetized for initial evaluation and stick removal. Premedication with dexmedetomidinea (2 mcg/kg) intramuscularly (IM) and butorphanolb (0.15 mg/kg) IM was given 10 min prior to anesthetic induction. General anesthesia was induced with propofolc (4 mg/kg) intravenously (IV) and maintained with oxygen and isofluraned (1%-2%).
With the patient under general anesthesia, the wooden stick was removed, and the maxillary fourth premolar teeth were surgically extracted bilaterally exposing an underlying severe palatal defect 6 cm in length and 1 cm in width. Nasal cavities and bilateral exposure of maxillary recesses were observed through the defect (Figure 2). Subsequently, the nasal cavities were copiously flushed, and the patient underwent a computed tomography (CT) scan to fully investigate the bony defect, which revealed a slightly wider bony palatal defect. The patient recovered uneventfully.
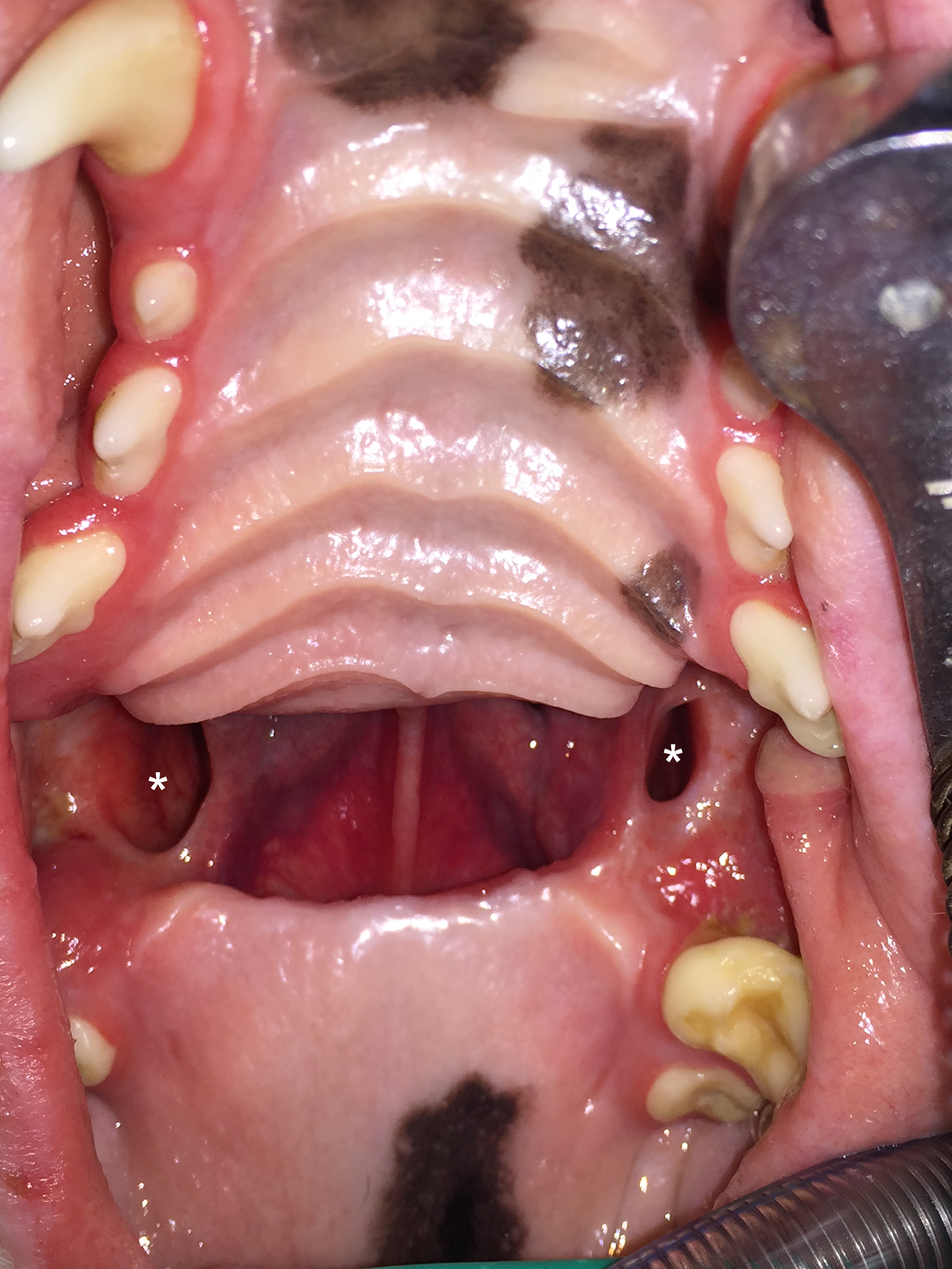
Case #1. Palatal defect after removal of the wooden stick, and extraction of right and left fourth premolar teeth, exposing the nasal cavities and right and left maxillary recesses (white asterisks).
Since the defect was exactly where the major palatine arteries emerge through the major palatine fissures, an angiographic CT study was performed to investigate the blood supply of the remaining palatal tissues and to properly plan the surgical treatment. The study showed the absence of the major palatine arteries as expected; however, it was unclear which vessel was responsible for the blood supply due to the presence of a copious network of small vessels embracing the rostral part of the palate. It was then assumed that the vascular supply of the palate rostral to the defect was derived from arteries of nasal origin. The surgical technique described in this article is based on this concept.
Specifically, a staged surgical treatment was planned, and the first step was to selectively extract teeth to allow the development of the planned flaps. Two weeks later the patient was anesthetized for repair of the defect. They were premedicated with dexmedetomidine (2 mcg/kg) IM and methadonee (0.2 mg/kg) IM 10 min prior to anesthetic induction. General anesthesia was induced with propofol (4 mg/kg) IV and maintained with oxygen and isoflurane (1%-2%).
Prior to surgical treatment, bilateral maxillary nerve blocks were performed with 2 ml ropivacainef (3 mg/kg). Subsequently, the right maxillary third premolar, first and second molars, and left maxillary first, second, and third premolars, and first and second molars were surgically extracted. To reduce the complications of oro-nasal and oro-antral communications during the gingival healing process, a custom-made vinyl polysiloxaneg temporary obturator was placed in the palatal defect (Figure 3).
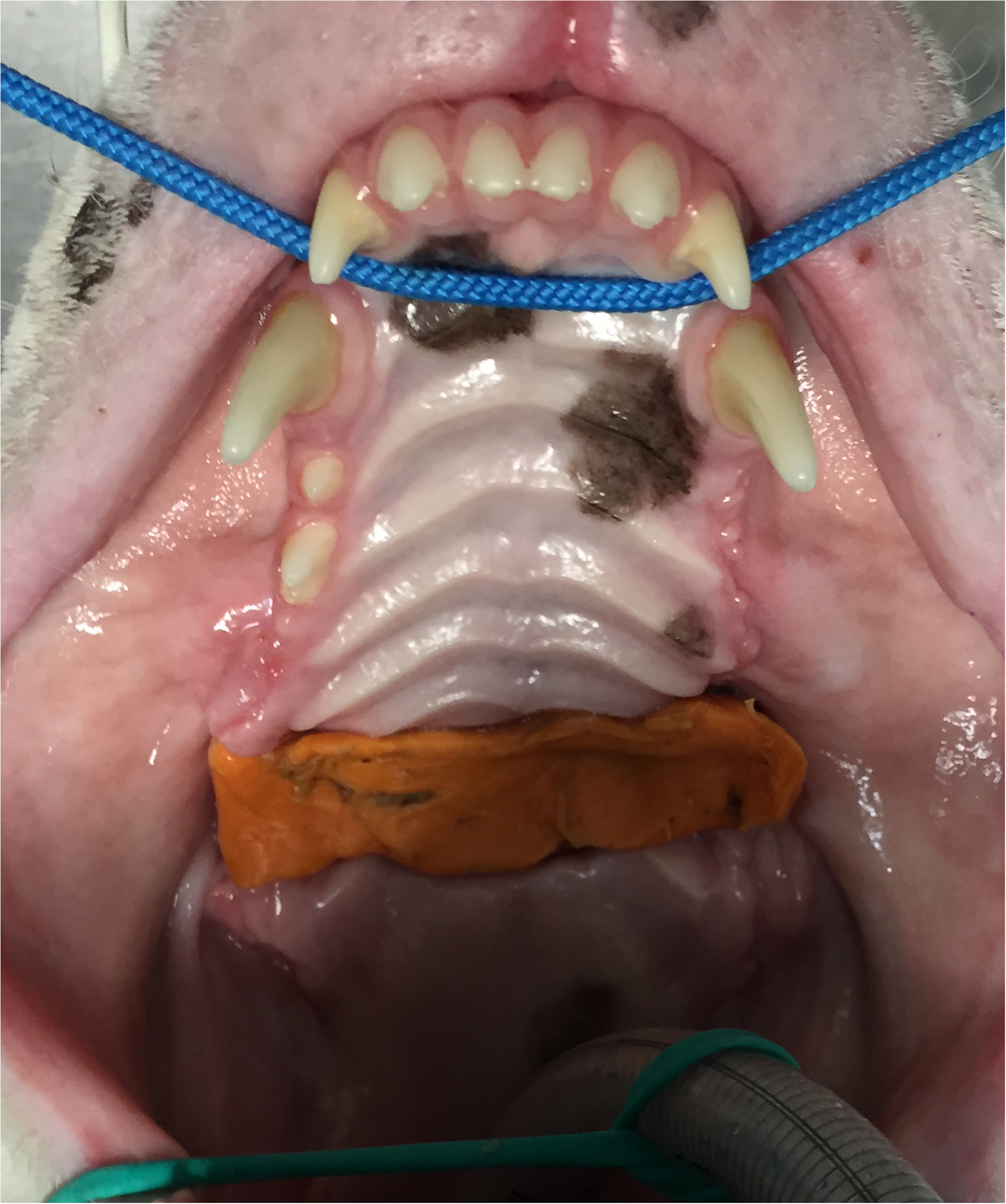
Case #1. Picture showing healed extraction sites and the temporary custom-made vinyl polysiloxane obturator covering the defect.
Four weeks postoperatively, the patient was anesthetized using the previous protocol described to close the palatal defect. The extraction sites were completely healed and the obturator was removed. Maxillary nerve blocks were performed bilaterally, and the planned flaps were marked with a mucosal tissue marker (Figure 4). The first flap was a full-thickness flap obtained from the rostral part of the defect. The mucosal incision was made immediately palatal to the maxillary gingiva beginning from the mesial aspect of the defect on one side, bordering the maxillary dental arches, and ending on the other side creating a unique rostral palatal U-flap. Care was taken to incise distally to the palatine fissures and to not expose them. The mucoperiosteal flap was undermined subperiosteally with a periosteal elevatorh being careful to not damage the vascular supply. The full-thickness flap was reversed distally, placed under the distal border of the defect previously elevated subperiosteally, and then sutured to the mucoperiosteum of the caudal part of the palate with a simple interrupted pattern using 5-0 monofilament absorbable suture materiali (Figure 5). To increase the strength of the surgical site, bilateral pedicle mucosal flaps were developed and sutured together in the midline (Figure 6). The exposed donor site bone was left to heal by the second intention.
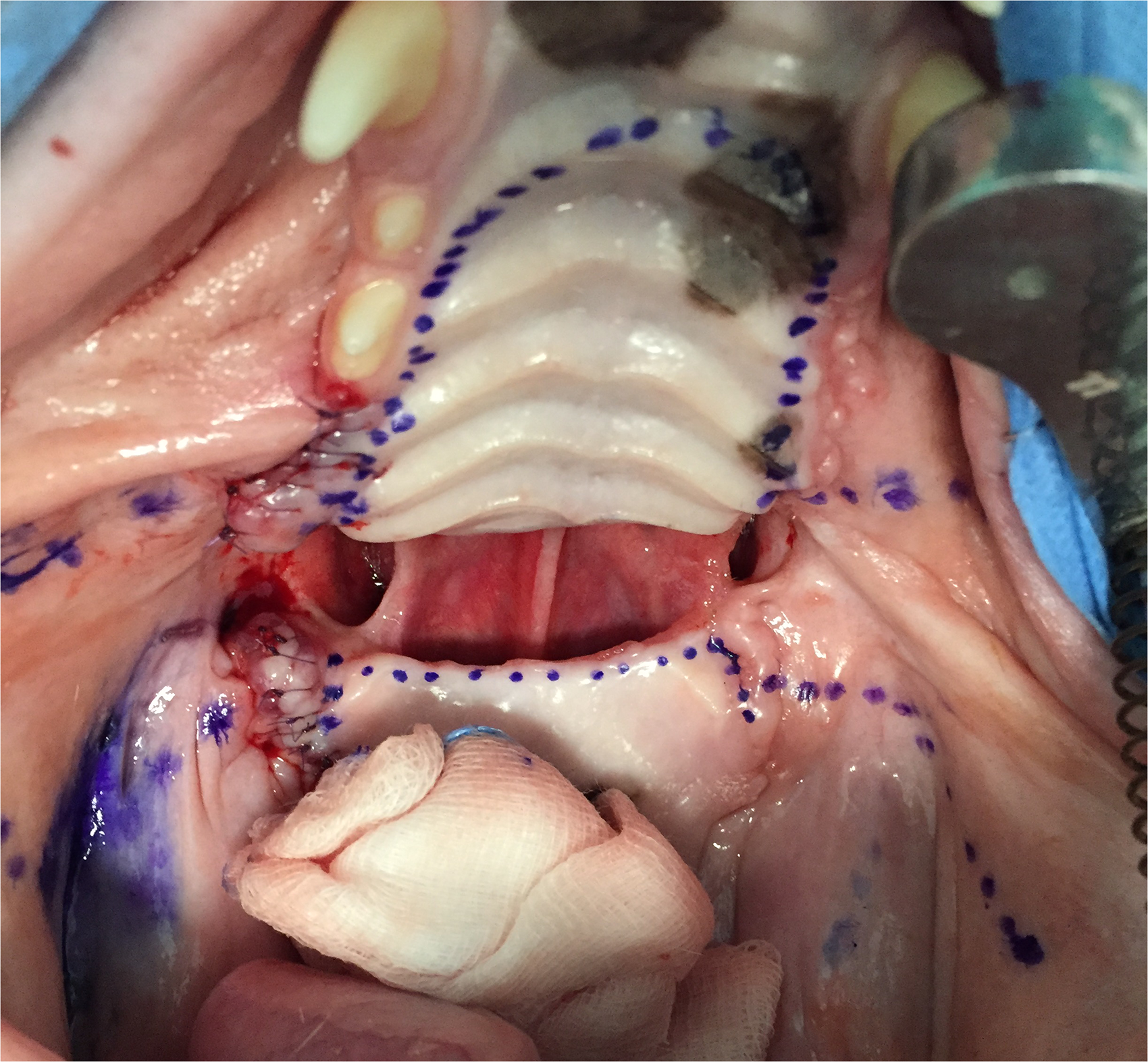
Case #1. Preoperative images showing the planned incision lines for the creation of the rostral reversed and caudal palatal flaps and for the vestibular mucosal flaps.
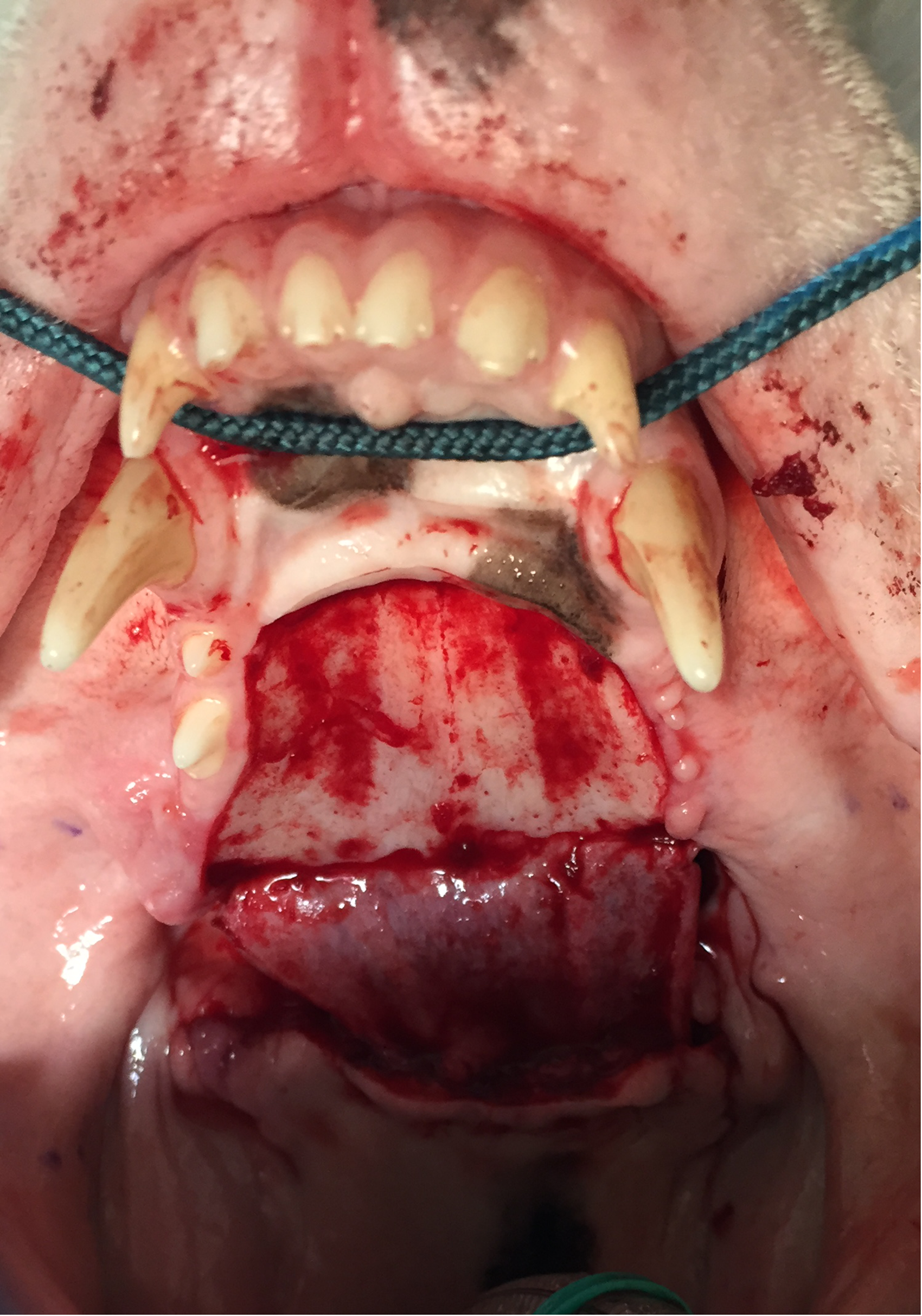
Case #1. The rostral full-thickness palatal flap was raised, caudally reversed, and hinged beneath the caudal palatal mucosa previously raised along the caudal border of the palatal defect.
Case #1. Bilateral pedicle mucosal flaps were raised and sutured as a second layer over the palatal reversed flap.
The patient was observed overnight and discharged the next day with amoxicillin and clavulanic acidj (24 mg/kg) per os (PO) twice daily and robenacoxibk (2 mg/kg) PO once daily for 7 days.
On oral examination 15 days postoperatively, the exposed bone showed granulation tissue and the reversed mesial palatal flap was healing perfectly. However, dehiscence of mucosal flaps was observed, and surgical revision was planned after 2 weeks (Figure 7). The patient was anesthetized using the aforementioned anesthetic protocol; subsequentially bilateral maxillary nerve blocks were performed and bilateral rotational vestibular mucosal flaps were created (Figures 8 and 9). The patient was discharged the same day with amoxicillin and clavulanic acidi and robenacoxibj at the previous prescription. After 2 weeks and 1 month, the oral examination revealed no signs of dehiscence. The referring veterinarian reported perfect healing after 1 year.
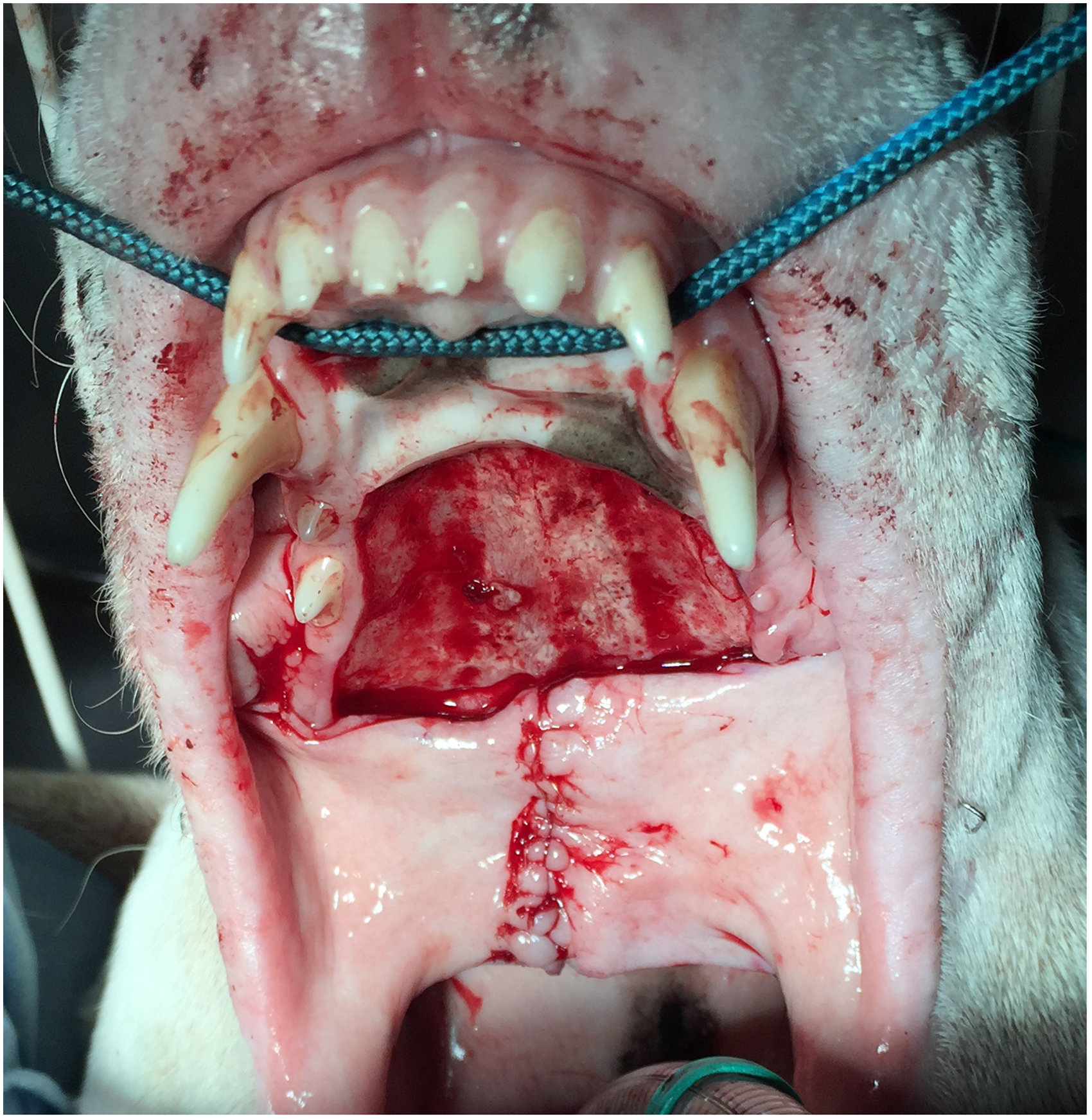
Case #1. Perfectly healed reversed palatal flap and exposed bone with dehiscence of mucosal flaps and bilateral oro-antral fistulas.
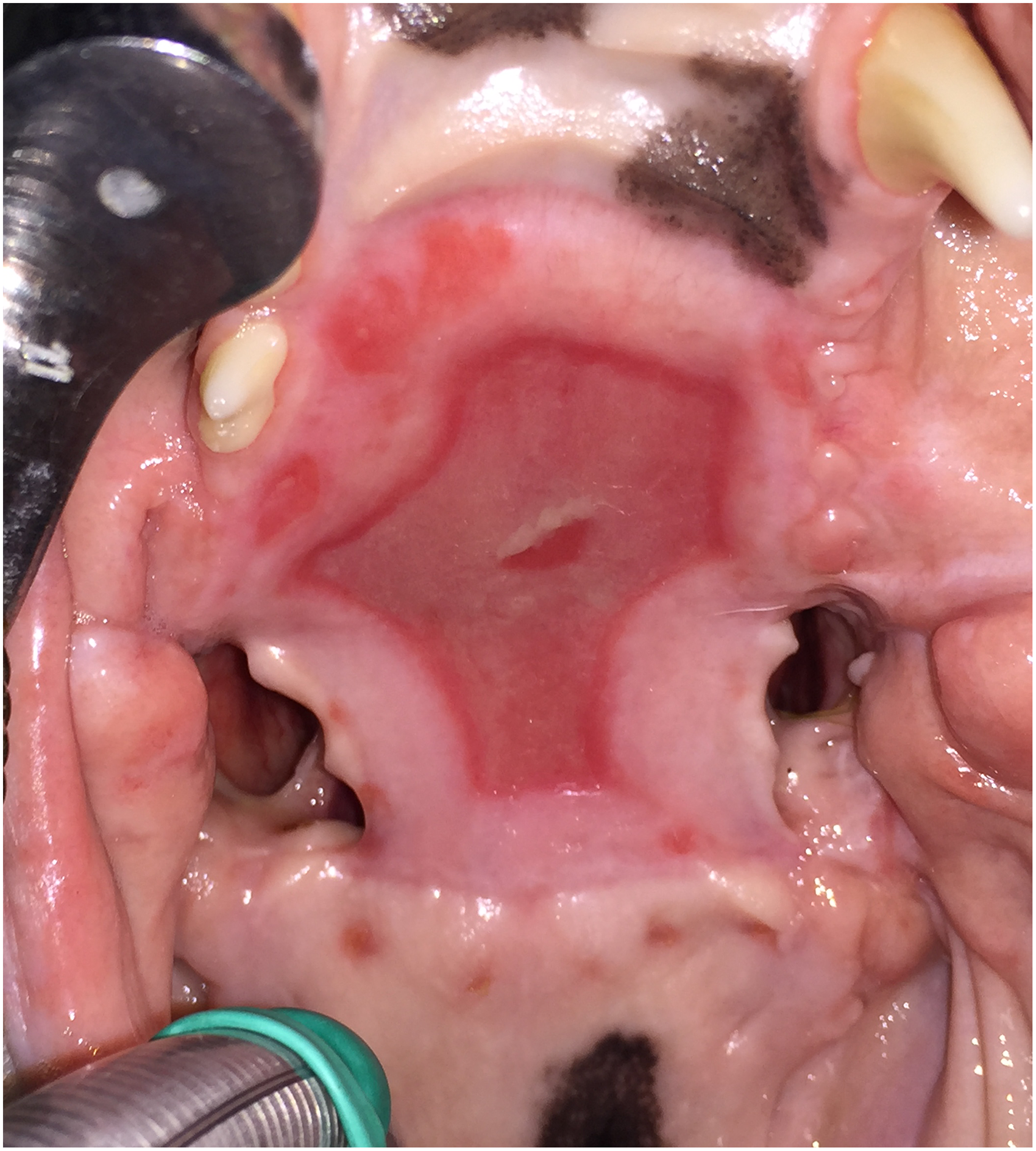
Case #1. Preoperative images showing the planned incision lines for the creation of the vestibular mucosal flap on the right side (dotted line).
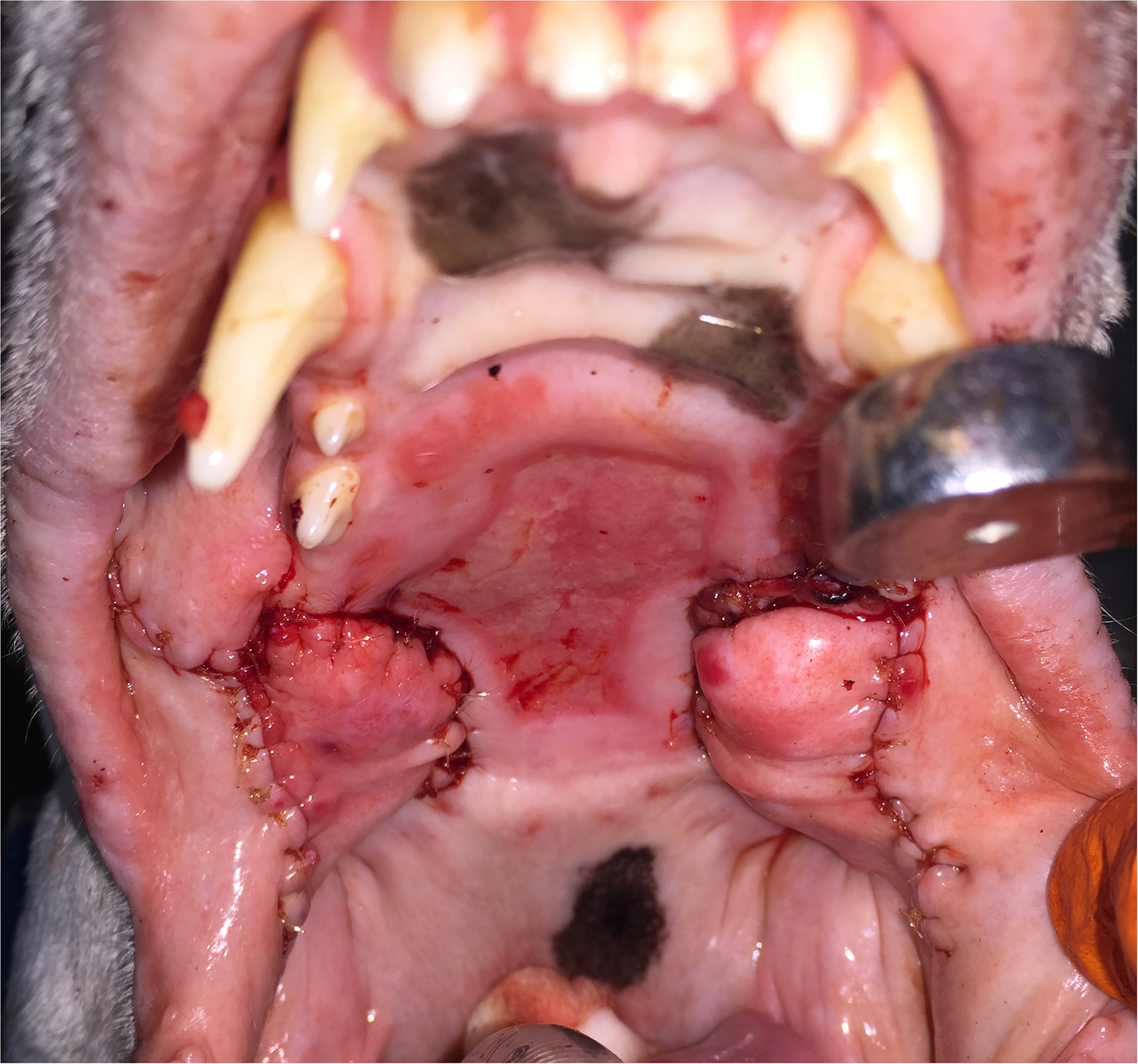
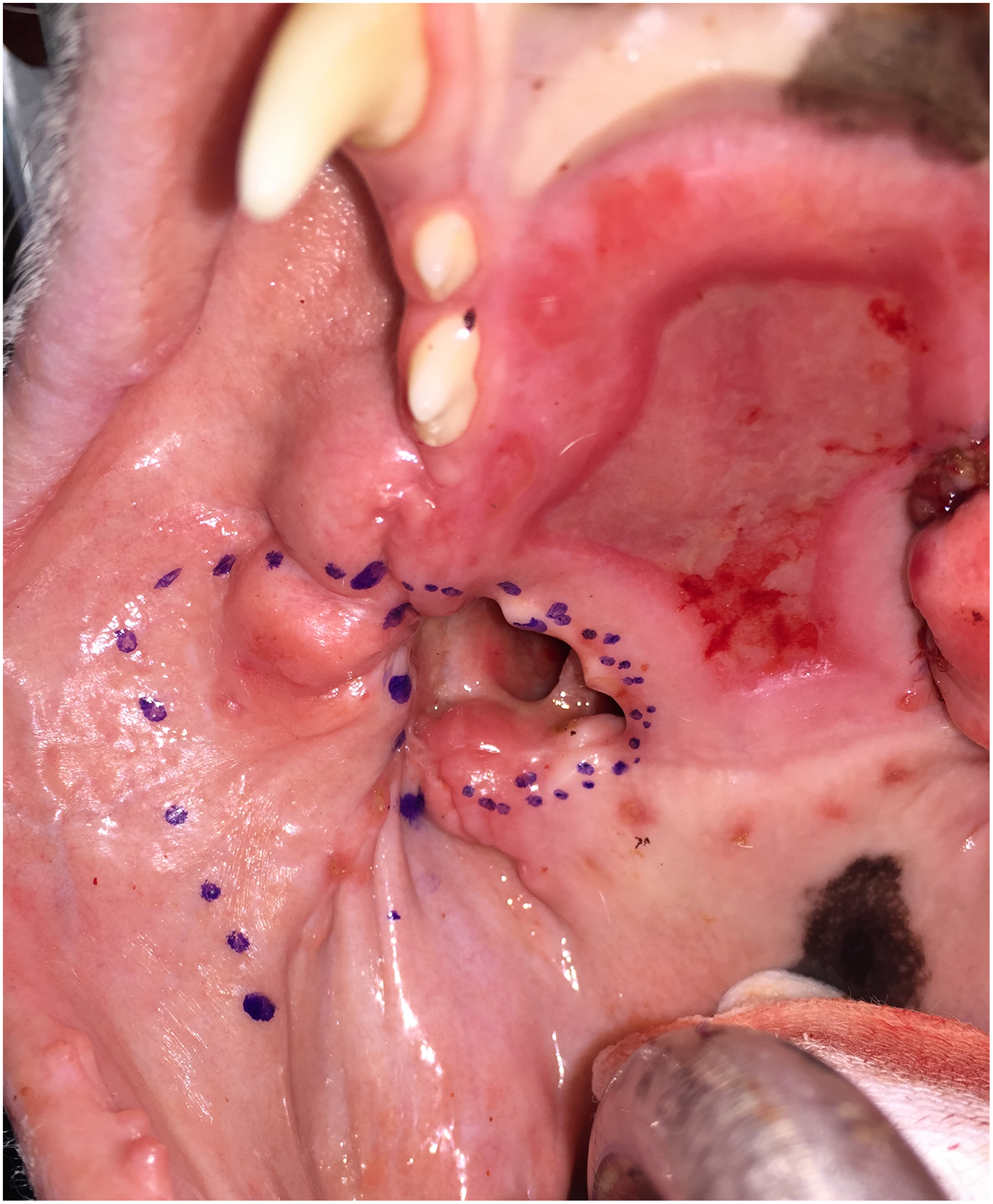
Case #1. Postoperative closed bilateral oro-antral fistulas with right and left mucosal flaps.
Case 2
A 10-year-old male Dachshund was referred for a severe acquired palatal defect secondary to a foreign body lodged between the caudal maxillary teeth which was removed by the referring veterinarian.
At the first visit, the patient showed symptoms referable to chronic rhinitis such as sneezing and bilateral nasal mucopurulent discharge and upon opening the mouth a caudal palatal defect was observed (Figure 10). The remaining physical examination was unremarkable. Preanesthetic blood tests revealed a slight increase in white blood cells attributable to upper airway infection. The patient was anesthetized with dexmedetomidine (2 mcg/kg) IM and methadone (0.2 mg/kg) IM 10 min prior to anesthetic induction. General anesthesia was induced with propofol (4 mg/kg) IV and maintained with oxygen and isoflurane (1%-2%).
Case #2. Caudal palatal defect between the maxillary fourth premolar teeth. The exposed nasal cavities are seen through the defect.
During the first anesthetic session, the nasal cavities were copiously flushed, and a thorough investigation of the defect was performed through careful oral examination and advanced imaging using CT. The oral exploration revealed a severe palatal defect extending from the right maxillary fourth premolar and first molar teeth to the left counterparts, exposing the nasal passages. The defect's dimensions were 2 cm in length × 1 cm in width.
The CT study confirmed the absence of palatal shelves beneath the palatal mucosal defect and revealed a slightly larger bony defect. The location of the defect was exactly at the level of the major palatine foramena, where the major palatine arteries emerge to run rostrally under the palatal mucosa. Because this portion of the palate was destroyed, the palatine arteries were considered to be irreversibly damaged. Therefore, without resorting to angiography CT, the palate rostral to the defect was believed to be supplied by nasal vascular vessels.
Based on the extension of the lesion, a staged treatment was planned. The patient was anesthetized using the protocol aforementioned. Subsequently, bilateral maxillary nerve blocks with 2 mL ropivacaine (3 mg/kg) and extraction of the right and left maxillary third and fourth premolars and first and second molars were performed to increase tissue availability for the planned closure (Figure 11). The patient was discharged the same day with amoxicillin and clavulanic acid and robenacoxib as prescribed in Case #1.
Case #2. Selective tooth extractions were performed including left and right maxillary third and fourth premolars and first and second molars.
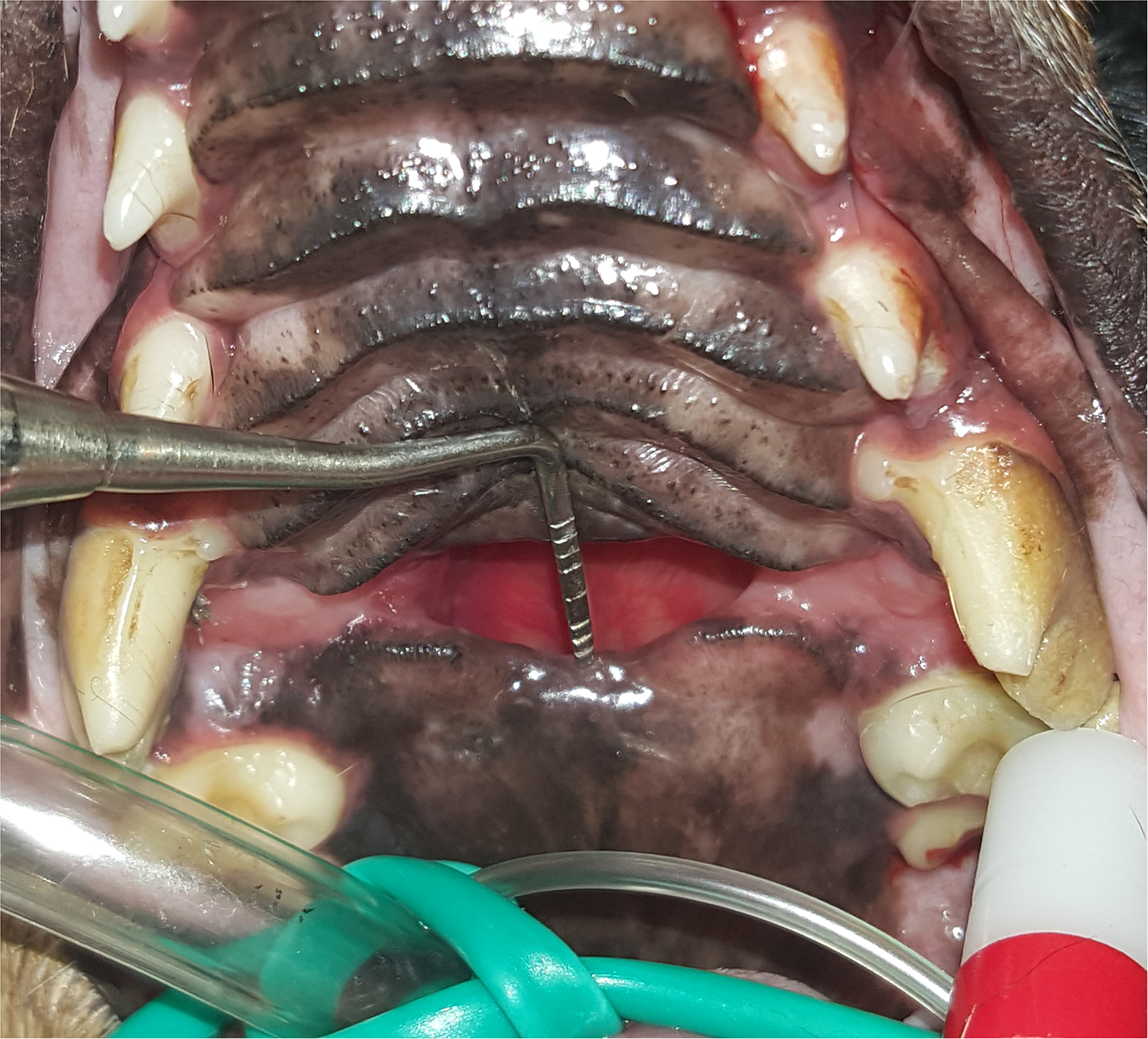
Fifteen days postoperatively, the surgical sites had healed, and the second treatment stage was planned. A single reversed pedicle U-shaped flap was designed in the palate rostral to the defect (Figure 12). A full-thickness incision of the palatal mucoperiosteum using a #15 blade was made on the right side just palatal to the gingival tissue at the mesial margin of the defect. The incision was continued rostrally bordering the maxillary dental arches, curving to the left side at the level of the second premolar distal to the palatine fissures, and it continued caudally to reach the defect on the left mesial margin. The flap obtained was a U-shaped flap that was twice the size of the defect. It was then undermined subperiosteally using a periosteal elevator, reversed over the palatal defect, and placed between the palatal shelves and the subperiosteally elevated palatal mucoperiosteum caudal to the defect (Figure 13A and B).
Case #2. Preoperative images showing the planned incision lines for the creation of palatal flaps.
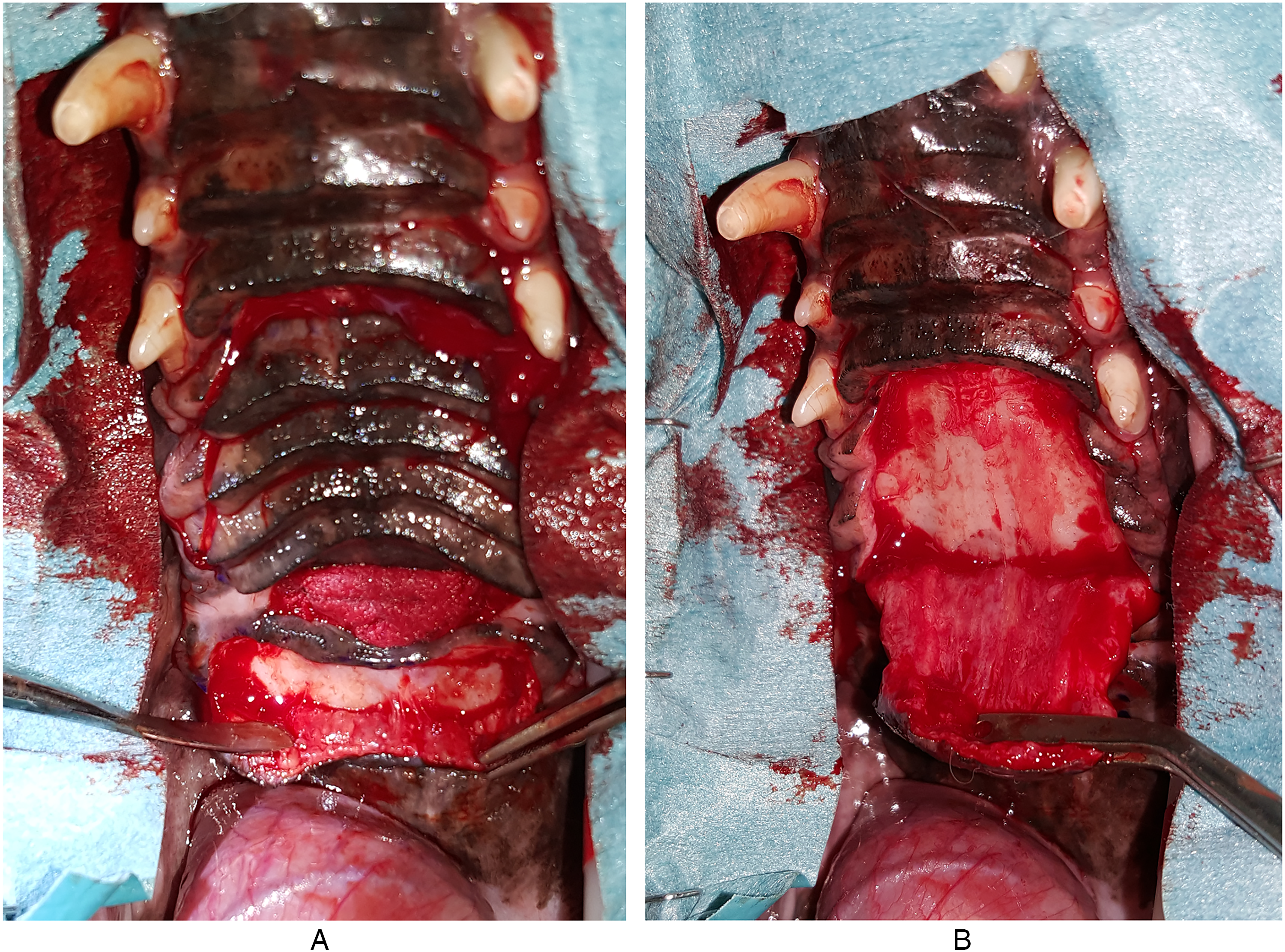
Case #2. Creation of palatal flaps: (A) caudal envelope flap and (B) raised rostral palatal flap.
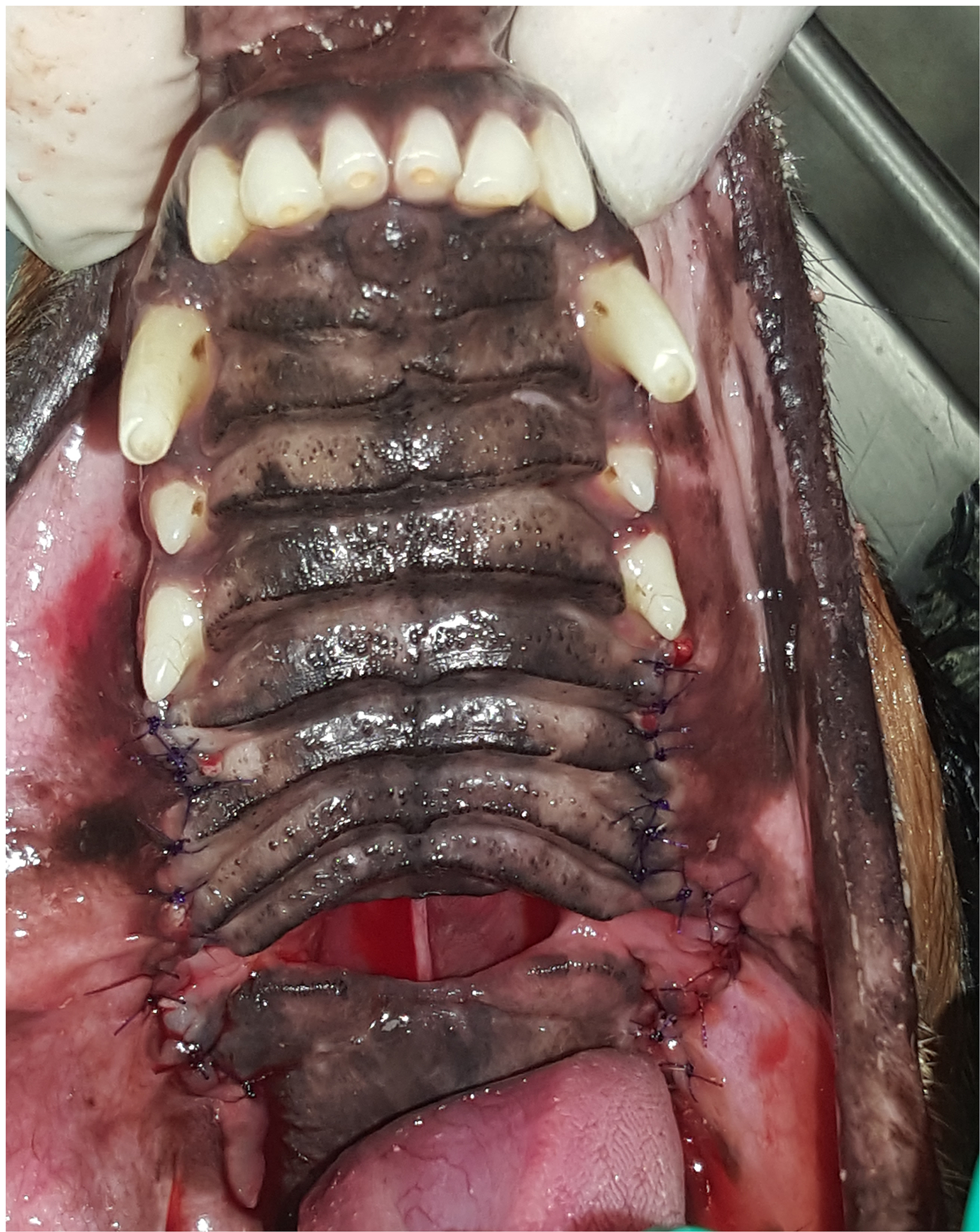
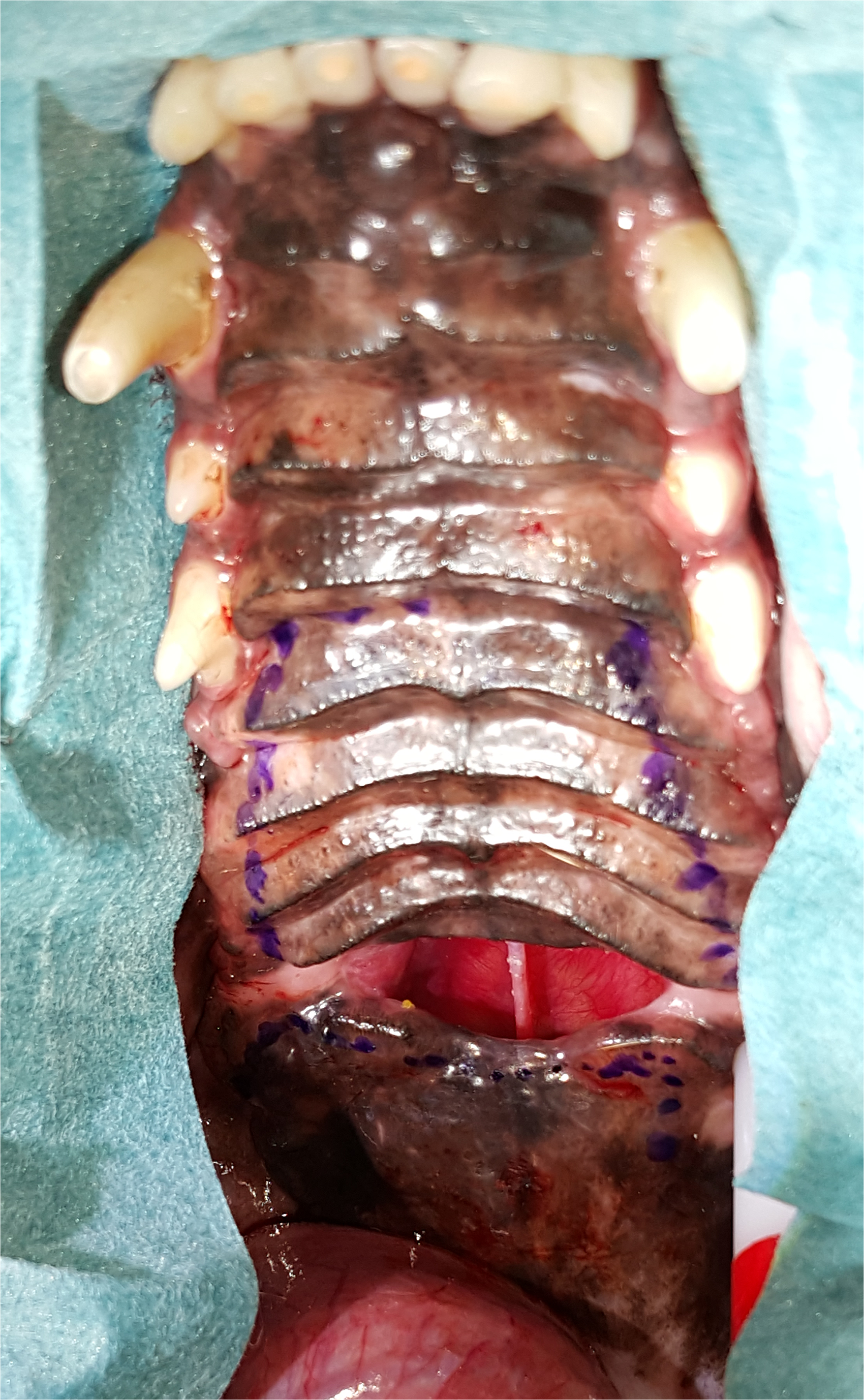
The flap was sutured to the surrounding tissues using a simple interrupted pattern with 5-0 monofilament absorbable suture material (Figure 14). The exposed bone was left to heal by the second intention. The patient recovered uneventfully and was discharged the next day with amoxicillin and clavulanic acid and robenacoxib as previously prescribed. At the 2-week, 2-month, and 6-month follow-up examinations the flap had healed well without signs of dehiscence, and the previously exposed bone was completely epithelialized (Figure 15A and B).
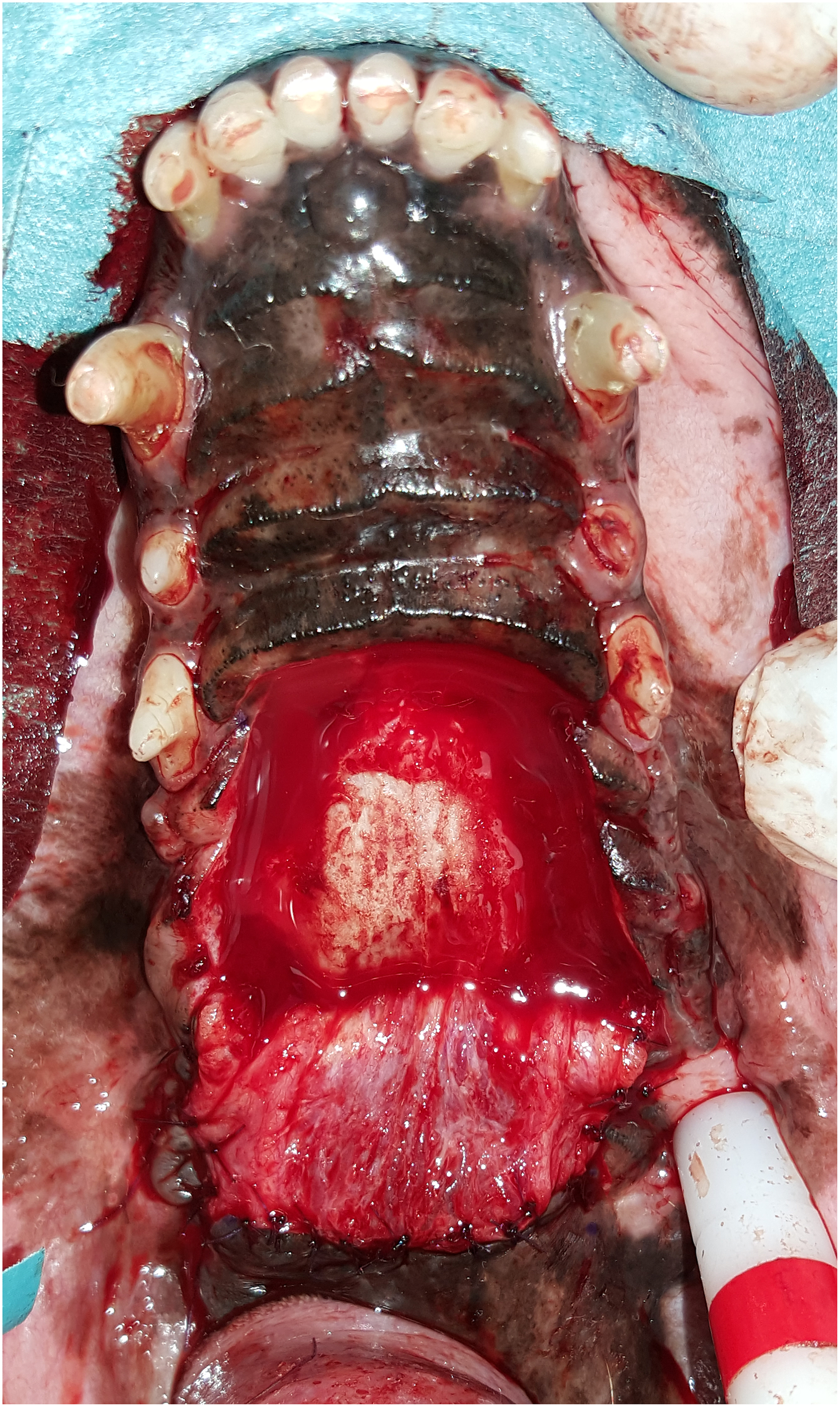
Case #2. Postoperative image with the rostral palatal flap hinged beneath the caudal envelope flap.
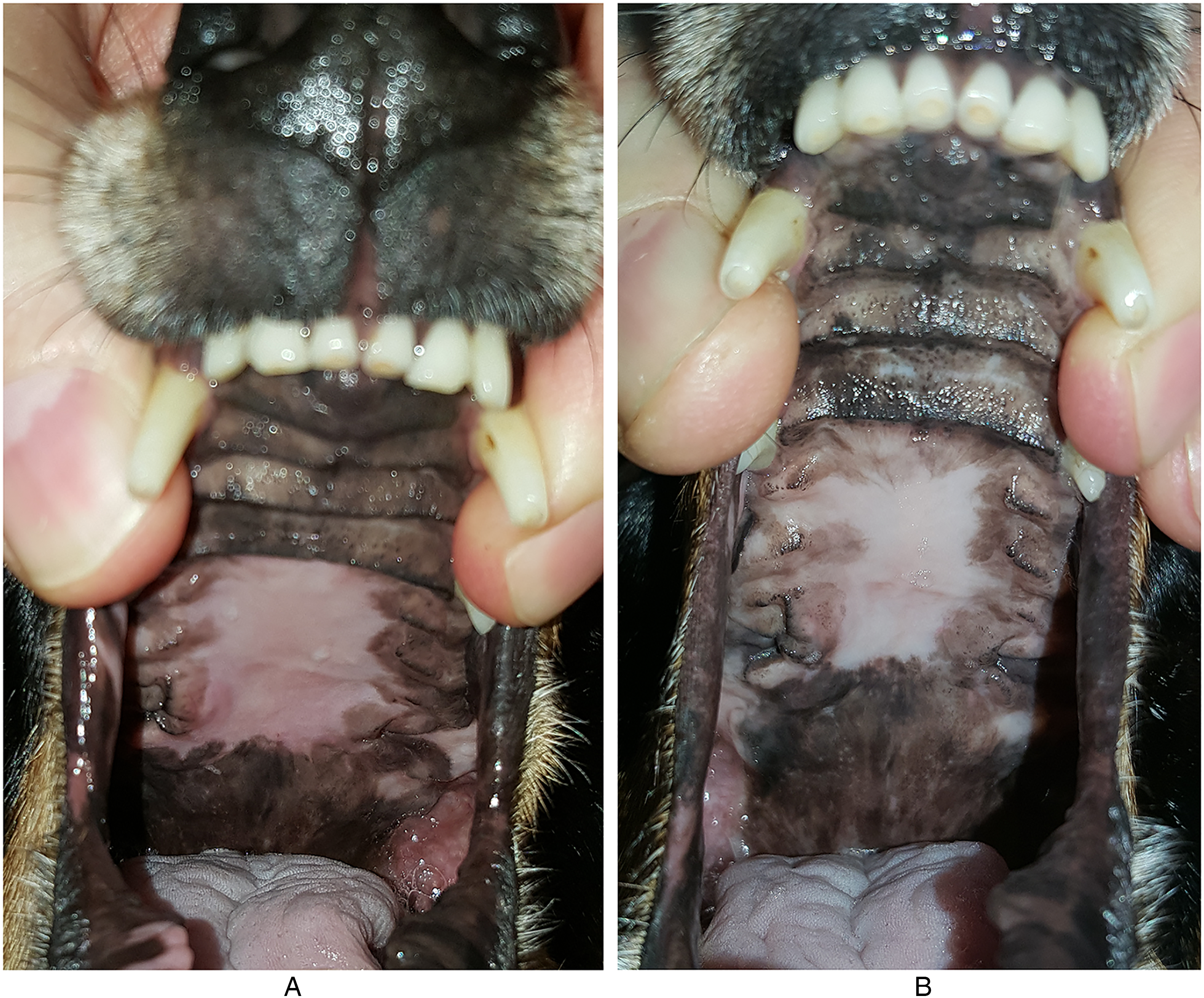
Case #2. Postoperative photographs: (A) 2-month and (B) 6-month follow-ups. The exposed bone is completely epithelialized and the flap has healed.
Discussion
In the present case series, an innovative technique is described to repair severe acquired palatal defects. This condition could be secondary to numerous etiologic factors. However, it is not always possible to trace the primary cause making a histological examination crucial to rule out pathologic conditions such as immune-mediated disease or neoplasia in which the surgical treatment should be carefully considered or would even be contraindicated in selected cases.41–43
This article describes two dogs with chronic acquired palatal defects secondary to foreign bodies trapped between the right and left maxillary teeth.
When a palatal defect is observed it is mandatory to address it as soon as possible to avoid secondary complications due to the passage of saliva, food, water, and debris into the nasal passages, maxillary recesses, or orbits.19–23,44 However, the proper timing is closely related to the etiology; for example, congenital defects should be treated between 3 and 4 months of age whereas acquired defects caused by dental pathologies should be treated at the time of diagnosis.45,46 Conversely, severe acute acquired defects due to traumatic events should wait a few days until the necrotic border of the palatal mucosa is completely outlined, while slight acute defects of the hard palate without an epithelial layer along the margins could be left to heal on their own.27,47,48 In the latter condition, if the cornified stratified epithelium of the oral cavity and the pseudostratified columnar epithelium of nasal cavities connect, the defect needs to be surgically repaired. 49
The patients, in this case report, possessed chronic acquired large palatal defects that had to be addressed as soon as possible to avoid worsening of clinical signs and complications. The treatment plan must always include advanced diagnostic imaging such as FBCT or CBCT to fully investigate the defect and understand which surgical technique could be the most appropriate.10,50,51 In this field, 3-dimensional (3D) reconstruction seems to be the most used imaging in trauma patients being of greatest spatial clarity while multiplanar reconstruction (MPR) is the appropriate modality for accurately diagnosing the medical condition. 52 Furthermore, CT study allows the creation of 3D printed models that are extremely useful for surgical planning and training. 53
Many different techniques to repair palatal defects are described including local mucosal flaps, axial pattern flaps, tongue flaps, auricular cartilage-free grafts, and artificial obturators.18,30–32,34–41,54,55 However, these defects, mostly the acquired ones, could have different shapes, locations, and dimensions making in some cases already existing techniques unfeasible. Even flaps from the vestibular mucosa could have been risky in these two dogs since the palatal defects were transversely wide and they extended minimally rostro-distally requiring long, narrow flaps. Major aspects to consider in decision-making are healthy tissue availability around the defect and blood supply of designed flaps.27,46
Another important consideration for severe palatal defects is the strength of the flap which must resist tongue movement, nasal air flow, and the contaminated oral environment, making double flap techniques preferable to one-layer closures.22,36,56 Lastly, to ensure sutures without tension and the creation of wide flaps, a staged surgical approach with selected tooth extractions could be necessary to increase tissue availability. 57
The dogs in this article underwent staged procedures beginning with the extraction of selected teeth. A custom-made vinyl polysiloxane temporary obturator was fabricated in Case #1 to close the defect as described in previous articles.36,55
The mainstream concept of the surgical technique described is the fact that the blood supply of the palate derives partially from nasal vessels such as sphenopalatine and lateral nasal branches, mostly if the major palatine arteries are compromised due to trauma or other causes. 15 However, these vessels reach the palate rostrally, not supporting the creation of the described technique. Then, to understand the vascular supply of the part of the palate rostral to the defect an angiographic CT was performed in Case #1 which showed that an extremely large number of vessels reached both the rostral and the distal part of the palate rostral to the defect without pinpointing the origin of this dense vascular network. With this information, a full-thickness flap based at the rostral border of the defect was raised, caudally reversed over the defect, and placed beneath the caudal border of the defect between the bone shelves and the mucoperiosteum.
The palatal defect of Case #1 was larger, exposing both left and right maxillary recesses and, to increase the strength of the surgical site, bilateral pedicle mucosal flaps were developed and sutured together on the midline over the reversed palatal flap. However, these two additional flaps failed with subsequent partial dehiscence and bilateral recurrence of oro-antral fistulas. The dog was therefore re-treated using bilateral rotational mucosal flaps. With hindsight, the authors consider questionable the creation of a second layer since the reversed palatal flap was already sufficiently strong. However, both dogs showed perfect healing without signs of dehiscence of the reversed palatal flap using the technique introduced in this report.
Previous studies carried out on surgical repair of congenital defects showed an 85% success rate with 50% demanding a second intervention. 58 Regarding acquired palatal defects, a recent article showed risk factors associated with hard palate mucoperiosteal flap failure, which determined that the patient's weight, presence of neoplasia at the time of surgery, flap dimensions, area to cover, and flap travel distance were factors significantly linked to failure rates. 56
Although no conclusion on failure rate can be made with only two cases, there was a 100% rate of success with this innovative technique. Therefore, the authors conclude that in severe palatal defects where the surgical treatment cannot rely on the major palatine arteries, flaps could be designed in consideration of random vessels from the nasal cavity.
Materials
(a) Sedadex, Dechra, Paesi Bassi
(b) Torphasol, Livisto, Senden-Bösensell
(c) PropoVetTM, Zoetis, Roma
(d) IsoFlo, Zoetis, Roma
(e) Semfortan®, Dechra, Paesi Bassi
(f) Ropivacaina Kabi, Fresenius Kabi, Verona
(g) Soft impression putty, iM3, Sydney
(h) Molt periosteal elevator n.9, iM3, Duleek
(i) Monolac, Chirmax, Prague
(j) Konclav, Ati, Bologna
(k) Onsior, Elanco, Cuxhaven
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
